Abstract
Transplant studies demonstrate unequivocally that the suprachiasmatic nucleus (SCN) produces diffusible signals that can sustain circadian locomotor rhythms. There is a vascular portal pathway between the SCN and the organum vasculosum of the lamina terminalis in mouse brain. Portal pathways enable low concentrations of neurosecretions to reach specialized local targets without dilution in the systemic circulation. To explore the SCN vasculature and the capillary vessels whereby SCN neurosecretions might reach portal vessels, we investigated the blood vessels (BVs) of the core and shell SCN. The arterial supply of the SCN differs among animals, and in some animals, there are differences between the 2 sides. The rostral SCN is supplied by branches from either the superior hypophyseal artery (SHpA) or the anterior cerebral artery or the anterior communicating artery. The caudal SCN is consistently supplied by the SHpA. The rostral SCN is drained by the preoptic vein, while the caudal is drained by the basal vein, with variations in laterality of draining vessels. In addition, several key features of the core and shell SCN regions differ: Median BV diameter is significantly smaller in the shell than the core based on confocal image measurements, and a similar trend occurs in iDISCO-cleared tissue. In the cleared tissue, whole BV length density and surface area density are significantly greater in the shell than the core. Finally, capillary length density is also greater in the shell than the core. The results suggest three hypotheses: First, the distinct arterial and venous systems of the rostral and caudal SCN may contribute to the in vivo variations of metabolic and neural activities observed in SCN networks. Second, the dense capillaries of the SCN shell are well positioned to transport blood-borne signals. Finally, variations in SCN vascular supply and drainage may contribute to inter-animal differences.
Keywords
The suprachiasmatic nucleus (SCN) is the locus of the brain’s clock, serving to synchronize physiological activities and behaviors to the local environment. Much work has identified both neural and diffusible output signals of the SCN though the former are better characterized (reviewed in Li et al., 2012; Morin, 2013; Patton and Hastings, 2018; Starnes and Jones, 2023). Functional diffusible signals of SCN transplants are effective even when the grafted tissue is encapsulated in a semipermeable polymer plastic that prevents neural fiber outgrowth, indicating that circadian activity rhythms can be sustained by diffusible signals (Silver et al., 1996). Schibler et al. (2003) suggested that the amount of signaling proteins produced by a small nucleus like the SCN is sufficient to reach only nearby loci. The transplant studies also suggest that effective diffusible signals can travel through fluids for limited distances. The host SCN can entrain rhythms in SCN grafts implanted into the third ventricle (3V) but not those placed in the lateral ventricle (Servière et al., 1994). Transplant studies also provide evidence of a possible target site. The attachment site of the graft within the 3V impacts the precision of the restored rhythm and suggests that the target of diffusible signals lies just rostral to the anatomical locus of the SCN itself (LeSauter et al., 1997). A possible target and route for these signals is the organum vasculosum of the lamina terminalis (OVLT) and connecting portal vessels (Yao et al., 2021). Vascular portal systems are specialized structures in which the capillary beds of 2 distinct regions are joined by connecting portal vessels. This arrangement permits blood-borne signals to travel without dilution in the systemic circulation. The SCN-OVLT portal pathway is a candidate route for SCN’s diffusible outputs.
Historically, the brain’s blood vessels (BVs) were thought to be passively regulated by systemic circulation with little direct impact on the brain activity (Friedland and Iadecola, 1991). However, accumulating evidence showed that regional blood flow in the brain is related to local metabolic needs and that BVs participate in shaping the activities of the nervous system and ultimately behavior. This has led to the concept of “neurovascular unit (NVU)” (reviewed in Iadecola, 2017). The SCN produces neuropeptides known to regulate vascular activities including vasopressin (AVP), vasoactive intestinal polypeptide, gastrin-releasing peptide, and angiotensin II (Ang II), among others (Abrahamson and Moore, 2001; Azushima et al., 2020; Gonkowski, 2020; Japundžić-Žigon et al., 2020; Qu et al., 2018). However, how these vasoactive peptides participate in the neurovascular coupling within the SCN is barely understood, partly due to a limited understanding of the SCN vasculature in relation to its peptidergic phenotypes.
The SCN is a heterogeneous nucleus in terms of neuronal phenotypes in its core and shell subregions (Abrahamson and Moore, 2001; Varadarajan et al., 2018; Wen et al., 2020). Thus, it is important to identify where the SCN-OVLT portal vessels emerge from within this nucleus, and how these elements are anatomically related to SCN’s arteries, veins, and capillary network. In the present study, we sought to lay the groundwork for understanding how neurosecretions might impact circadian regulation by the SCN vasculature. We explored the arterial and venous vasculature of the SCN, as well as the vascular organization of its core and shell subdivisions. To this end, we examined the arterial supply and venous drainage of the SCN using iDISCO-cleared mouse brains imaged with a light-sheet microscope, allowing preservation of the 3-dimensional structure of the vasculature. We also compared BV diameter and density in SCN subdivisions with confocal and light-sheet images.
Materials and Methods
Animals and Housing
Male C57BL/6NJ mice aged 8-10 weeks were purchased from the Jackson Laboratory (Sacramento, CA). The mice were housed for at least 2 weeks after arrival. All animals were provided with ad libitum access to food and water, and maintained in a 12:12 h light:dark (LD) cycle (lights-on at 0700 h, where zeitgeber time [ZT] 0 refers to the time of lights on). All experiments were carried out in accordance with the guidelines of Columbia University’s Institutional Animal Care and Use Committee protocol number AC-AABH1603.
Perfusion
At ZT6 animals were deeply anesthetized (11% ketamine + 2% xylazine, 10 mL/kg body weight i.p.) and perfused intracardially with 50 mL 0.9% saline followed by 100 mL 4% paraformaldehyde (PFA) in 0.1 M phosphate buffer (PB), pH 7.3. Brains were post-fixed at 4 °C overnight. The next day, brains for iDISCO were washed with phosphate-buffered saline (PBS) and stored in PBS with 0.02% sodium azide at 4 °C until future use. Brains for analysis of free-floating sections were transferred to PBS with 20% sucrose for at least 48 h.
iDISCO Clearing
The iDISCO protocol (Renier et al., 2014) was used for tissue clearing with the following modification: Prior to clearing, the brain was trimmed and 2500 µm on each side of the midline in the sagittal plane was retained in order to include the entire circle of Willis. Tissues were incubated with primary antibodies for 2 weeks and then with secondary antibodies for 1 week.
Free-Floating Sections
Brains were sectioned (50 µm) coronally or horizontally on a cryostat (Microm HM 500M, Walldorf, Germany). The following tissue was collected for each brain: 14-16 sections for the coronal plane, and 8-10 sections for the horizontal plane. Free-floating sections were washed in PBS with 0.1% Triton X-100 (0.1% PBST) and blocked in 1:100 normal donkey serum in 0.3% PBST for 1 h. Sections were then incubated with primary antibodies for 1 h at room temperature and 36 h at 4 °C. Then they were washed with 0.1% PBST and incubated with secondary antibodies for 2.5 h. Immunostained sections were washed with PBS and mounted on subbed slides and coverslipped with Fluoromount Aqueous Mounting Medium (Sigma-Aldrich, F4680, St. Louis, MO) and cover glass No. 1 (Fisher Scientific, 12-544-18, Waltham, MA).
Immunostaining
The following primary antibodies were used in the iDISCO protocol: for labeling the SCN, AVP antibody (1:2500, rabbit; ImmunoStar, Hudson, WI); for labeling arteries, smooth muscle actin (SMA) antibody (1:67, mouse; Dako, M0851, Santa Clara, CA); for labeling the entire vasculature, Type IV Collagen-UNLB antibody (1:50, goat; SouthernBiotech, Birmingham, AL). For the sections, the rabbit AVP antibody was used at 1:5000. The following secondary antibodies were used in the iDISCO protocol; for AVP, donkey anti-rabbit Cy2 (1:200; Jackson ImmunoResearch); for SMA, donkey anti-mouse Cy3 (1:200; Jackson ImmunoResearch, West Grove, PA); for collagen, donkey anti-goat Cy5 (1:200; Jackson ImmunoResearch), and for sections, for AVP, donkey anti-rabbit Cy3 (1:200; Jackson ImmunoResearch); for the entire vasculature, tomato lectin-fluorescein (1:100; Vector Labs, Newark, CA).
Light-Sheet Microscopy
Cleared and immunostained tissues were imaged with UltraMicroscope II (LaVision BioTec, Bielefeld, Germany) equipped with an Olympus MVX10 zoom body (Olympus, Bartlett, TN), a LaVision BioTec Laser Module, and an Andor Neo sCMOS Camera with a pixel size of 6.5 µm. The following lasers and filters were used: Cy2-AVP was excited at 488 nm and emission was acquired at 525 ± 50 nm; Cy3-SMA was excited at 561 nm and acquired by 605 ± 50 nm; Cy5-collagen was excited at 639 nm and emission was collected at 705 ± 72 nm. The vasculature of the SCN region was scanned horizontally with a voxel size 1.21 µm × 1.21 µm × 1.5 µm (LE-RI × R-C × D-V). Stitching was performed using 3 × 3 1940 µm × 2750 µm × 960 µm stacks with 20% overlap.
Confocal Microscopy
Images were captured on a Nikon Eclipse Ti2E confocal microscope (Nikon Inc., Melville, NY) with a 20× objective and acquired with NIS Advanced Research software.
Image Preprocessing and BV Tracing
For light-sheet microscopy, multi-position stacks were stitched in Imaris Stitcher software (Bitplane AG, Zurich, Switzerland). The stitched images were imported into Imaris software (Bitplane AG) for image rendering. AVP staining was used to delineate the core and shell regions using Imaris. The AVP-sparse core and AVP-rich shell were extracted using the Surface tool of Imaris. Next, stacks of BV in these SCN subregions were imported to Vesselucida 360 (MicroBrightField, Colchester, VT) for tracing using the user-guided mode with the directional kernel. For confocal images, the Polygon Selection tool in ImageJ was used to delineate the SCN shell and core based on AVP staining. This delineation permitted identification of core vs shell BV in the lectin channel. The BVs of confocal images were traced in Vesselucida 360 using the same method as for the light-sheet images.
Image Analysis and Statistics
Branch points and diameter of BVs were measured with automated analysis tools in Vesselucida Explorer (MicroBrightField) for both confocal (N = 11 brains, 22 SCNs) and light-sheet (4 brains, 8 SCNs) images in core and shell regions (Suppl. Fig. S1). All statistical analyses were based on within-brain pairwise comparison between the SCN core and shell using the paired t test. The following parameters were compared: The average diameter of a BV segment (a segment is the part of a BV lying between a 2 branch points, or BV between a branching point and a starting point, or BV between a branching point and an ending point, or an isolated vessel without branching points); length density of BV, measured as length of vasculature/unit volume; surface area density of BV, measured as surface area of vasculature/unit volume; the capillary diameter, where capillary is defined as BV segment of the average diameter <10 μm; capillary length density, measured as length of capillary/unit volume.
Preliminary analysis indicated that the mean diameter per BV segment was not normally distributed for either SCN core or shell for either confocal or light-sheet data (Shapiro-Wilk test in Suppl. Tables S1 and S2). The mean diameter per capillary segment of light-sheet data is normally distributed in the core but not in shell in all 4 samples (Suppl. Table S3). In contrast, the difference between the paired core and shell median BV diameter for both the confocal and light-sheet data and the differences between the paired core and shell median capillary diameter were normally distributed (Suppl. Fig. S2a-c, Shapiro-Wilk test in Suppl. Table S4). Thus, hypothesis testing was based on median diameter of segments.
In light-sheet data, core and shell SCN, BVs were compared for 2 additional parameters: length density and surface area density. Both parameters are normally distributed (Suppl. Fig. S2d, e; Suppl. Table S4). The light-sheet data were also used to analyze differences in capillary length density between shell and core region. This parameter is also normally distributed (Suppl. Fig. S2, Suppl. Table S3). The BV segment diameter and length data sets are in Research data 1 for confocal images and Research data 2 for cleared SCN. Vascular network information for cleared SCN, including the total length, surface area, volume, is in Supplementary Table S5. The volumes of SCN in the cleared brains are in Suppl. Table S6.
All statistical analyses were done with Python 3.0 and its packages NumPy (Harris et al., 2020), Pandas (McKinney, 2010), and Scipy (Virtanen et al., 2020). Graphs were created with Python packages Matplotlib (Hunter, 2007) and Seaborn (Waskom, 2021).
Results
Unless otherwise noted, the terminology used for BV nomenclature is that of McKinley et al. (2003). A description of the arterial and venous organization of the SCN is presented in Figures 1 -4. Quantitative measurements of the core and shell vasculature are shown in Figure 5.
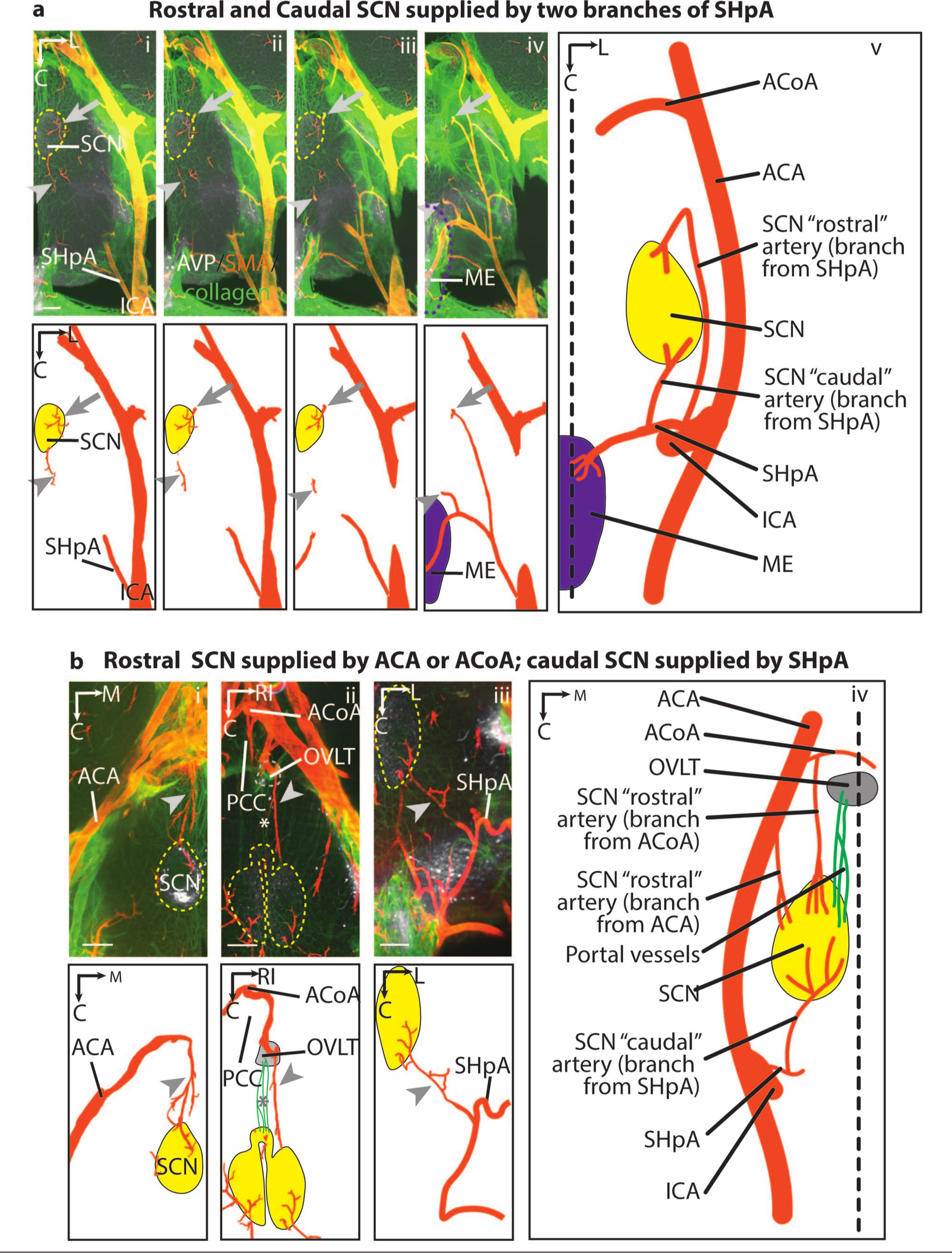
Arterial supply of the rostral and caudal SCN (horizontal view). In both panels a and b, the top row shows immunostaining in optical slices of cleared images and an explanatory drawing is shown immediately below. The larger panel to the right shows an overview of all the images in each panel. Panels a and b show the three variations observed in arterial supply. Panel a(i-v) depicts the SHpA as it branches from the ICA to reach the SCN and ME. One branch of SHpA enters the SCN at the rostral aspect (arrow) and another enters at the caudal aspect (arrowhead). In some mice (panel b(i)), the artery supplying the rostral SCN (arrowhead) is from the ACA, while in others, (panel b(ii)) it is from ACoA (arrowhead). The portal vessels (panel b(ii), asterisk) connect the rostral SCN and the OVLT. Panel b(iii), the artery supplying the caudal SCN (arrowhead) is a branch of the SHpA. Same brain as panel b(ii). Legend: AVP = white, SMA = orange, collagen = green in immunostaining images; arteries = orange, veins = blue, portal vessels = green in illustrations; SCN = yellow dashed line in immunostained images and yellow solid shape in illustrations; ME = purple dashed line in immunostained images and purple solid shape in illustrations; OVLT = gray dashed line in immunostained images and gray solid shape in illustrations; midline of the brain = black dashed line. For this and all subsequent figures, please consult the online version of the manuscript to visualize colors. Abbreviations: ACA = anterior cerebral artery; ACoA = anterior communicating artery; AVP = vasopressin; C = caudal; ICA = internal carotid artery; L = lateral; M = medial; ME = median eminence; OVLT = organum vasculosum of the lamina terminalis; PCC = prechiasmatic cistern; RI = right; SCN = suprachiasmatic nucleus; SHpA = superior hypophyseal artery; SMA = smooth muscle actin. Slice orientation: Panel a: serial dorsal-to-ventral optical slices, 200 μm thick with 50 μm overlap; panel b: optical slices, 200 μm thick. Scale bar = 200 μm.
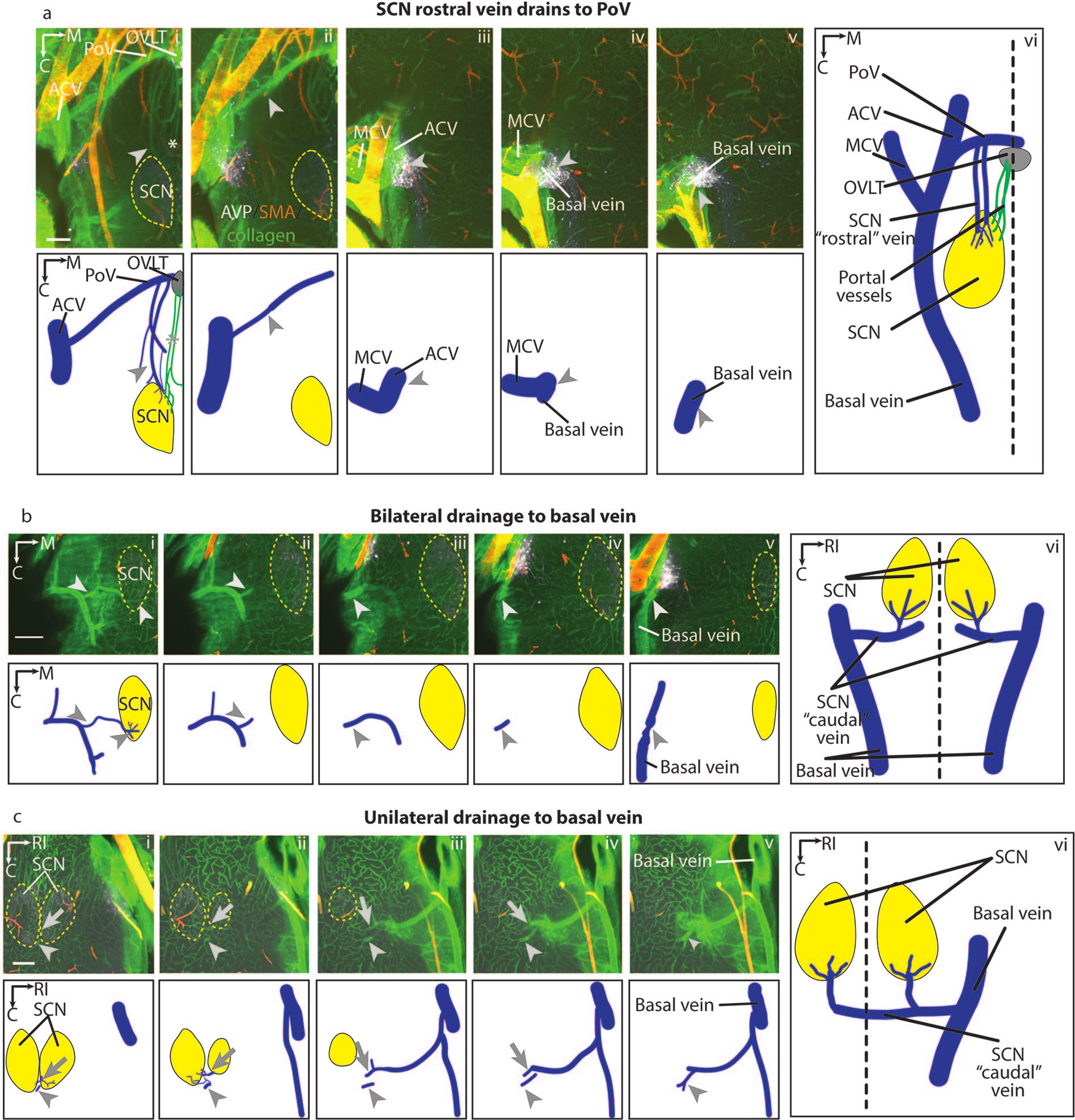
Venous drainage of the rostral and caudal SCN (horizontal view). In all panels a-c, the top row shows immunostaining in optical slices of cleared images and an explanatory drawing is shown immediately below. The larger panel to the right shows an overview of all the images in each panel. Panel a(i-vi) shows several veins emerging from the rostral SCN (arrowhead) draining to the PoV and finally merging with the ACV and then the basal vein. Panels b and c demonstrate that at the ventral aspect of the SCN, multiple capillaries converge upon the “caudal” vein. In some animals, the “caudal” vein on each side drains into the ipsilateral basal vein (panel b (i-vi), arrowhead), while in others (panel c (i-vi)), this vein drains into the basal vein on one side of the brain (arrowhead, vein from the left SCN; arrow, vein from the right SCN). Abbreviations: ACV = anterior cerebral vein; AVP = vasopressin; C = caudal; M = medial; MCV = medial cerebral vein; OVLT = organum vasculosum of the lamina terminalis; PoV = preoptic vein; RI = right; SCN = suprachiasmatic nucleus; SMA = smooth muscle actin. Slice orientation: Panels a, b, and c: serial ventral-to-dorsal optical slices, 150 μm thick with 50 μm overlap. Scale bar = 200 μm.
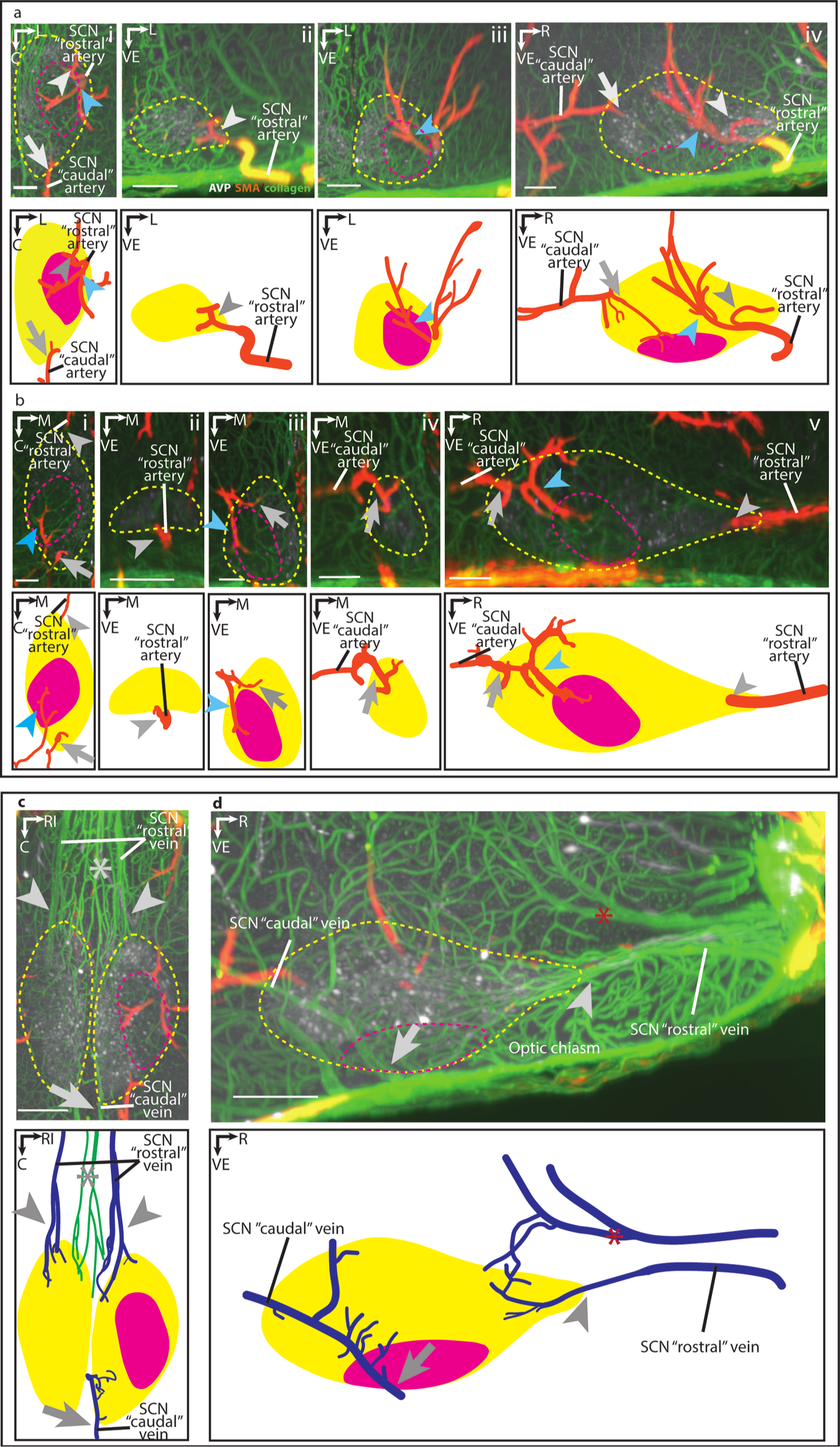
Arterial supply and venous drainage of the SCN core and shell (in horizontal, coronal and sagittal views). In panels a-d, immunostaining in optical slices of cleared images is shown in the top row and the accompanying explanatory drawing is shown immediately below. Panels a and b present the arterial supply in of SCN subregions. Panels c and d present the venous drainage of the SCN subregions. Panel a(i-iv), presents the horizontal (i), coronal (ii, iii), and sagittal (iv) orientations. In the case where the SCN is supplied only by the SHpA (not shown), a branch of the “rostral” artery supplies the rostral SCN shell (gray arrowhead), and it ramifies to supply the dorsal shell and core (blue arrowhead). The “caudal” artery from the SHpA supplies the caudal shell of the SCN (gray arrow). Same brain as in Figure 1a. Panel b depicts the horizontal (i), coronal (ii-iv), and sagittal (v) orientations. In the case where rostral SCN is supplied by the ACoA and caudal SCN is supplied by the SHpA (not shown), the “rostral” artery (gray arrowhead) enters the SCN at the rostral tip. The “caudal” artery enters the SCN at the aspect and ramifies into 2 branches, with one supplying the core and the dorsal shell (blue arrowhead), and the other supplying the caudal shell (gray arrow). Same brain as in Figure 1b(iii). Panel c presents the horizontal view, showing that at the rostral SCN are several veins (arrowhead) lying lateral to the small portal vessels (asterisk). Caudally, a draining vein forms at the nasal part of the shell (arrow). Panel d presents the sagittal view showing that a vein running rostral-dorsally to the SCN forms anastomosis with rostral shell capillaries (red asterisk). Legend: Arteries = orange; veins = blue; SCN core = yellow dashed line in immunostained images and yellow solid shape in illustration; SCN shell = magenta dashed line in immunostained images and magenta solid shape in illustration. Abbreviations: AVP = vasopressin; C = caudal; L = lateral; M = medial; R=rostral; RI = right; SCN = suprachiasmatic nucleus; SMA = smooth muscle actin; VE = ventral. Optical slice = 200 μm. Scale bar = 100 μm.
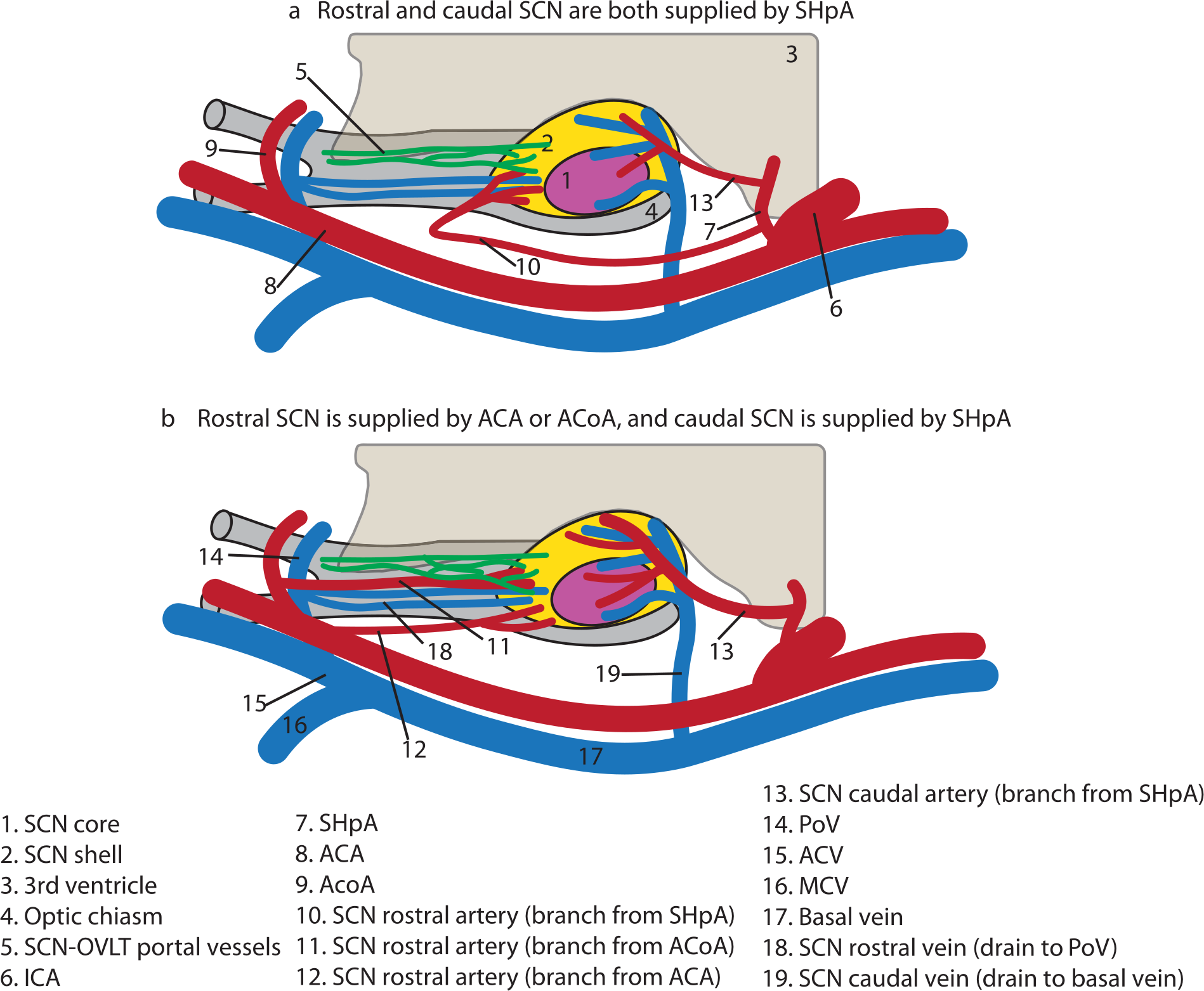
Diagrammatic summary of major SCN arteries and veins: sagittal view. Rostral SCN is supplied by SHpA (a), ACA(b), or ACoA (b). The caudal SCN is supplied by SHpA (a, b). The rostral SCN is drained by the PoV and caudal SCN is drained by the basal vein (a, b); the lateralization variations in venous drainage are not shown. The portal vessels form at the rostral-most SCN (a, b). Abbreviations: ACA = anterior cerebral artery; ACV = anterior cerebral vein; ACoA = anterior communicating artery; ICA = internal carotid artery; MCV = medial cerebral vein; OVLT = organum vasculosum of the lamina terminalis; PoV = preoptic vein; SCN = suprachiasmatic nucleus; SHpA = superior hypophyseal artery.
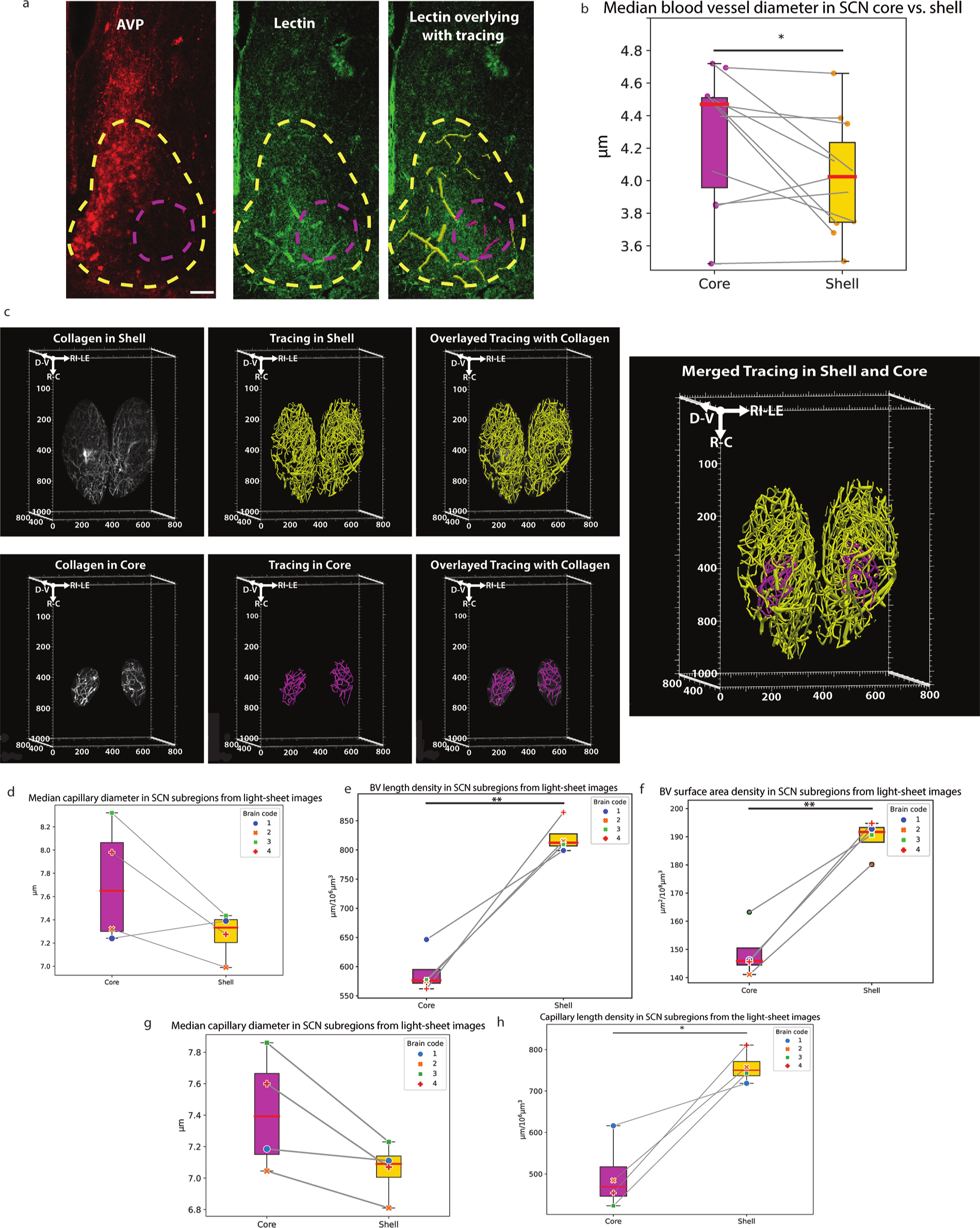
Characterization of SCN core versus shell vascular network. (a) SCN and its BV, and BV traces in the core and shell of confocal images. (b) The median BV diameter in the shell is significantly smaller than the core, paired t test, α = .05, same as for the following tests (4.02 ± 0.33 vs 4.27 ± 0.38 μm, t(10) = 2.41, p = .037). (c) BV traces of cleared SCN core and shell. Left panel, immunostaining of SCN BV labeled by collagen, traces, and BV traces overlaid with immunostaining in the shell (top row, from left to right) and the core (bottom row). Right panel, merged shell and core tracing. (d) The shell median BV diameter is smaller in three out of four brains, but not statistically significant (7.27 ± 0.17 vs 7.72 ± 0.45 μm, t(3) = 1.93, p = .149). (e) The BV length density is significantly higher in the shell than in the core (821.90 ± 25.12 vs 590.26 ± 32.96 μm/106 μm3, t(3) = 7.54, p = .004). (f) The BV surface area density is significantly higher in the shell than in the core (189.58 ± 5.64 vs 149.04 ± 8.43 μm2/108 μm3, t(3) = 8.34, p = .004). (g) Capillary (BV segment diameter < 10 μm) tends to be thinner in the SCN shell compared with the core but it is not statistically significant (7.06 ± 0.15 vs 7.42 ± 0.32 μm, t(3) = 2.86, p = .06). (h) The SCN shell has higher capillary length density (757.42 ± 33.73 vs 494.41 ± 73.60 μm/106 μm3, t(3) = 4.68, p = .02). The data are presented as boxplots showing the interquartile range (IQR, between 25th percentile and 75th percentile). Red line is the median. Upper whisker represents 75th percentile + 1.5 IQR. Lower whisker represents 25th percentile − 1.5 IQR. SCN shell = magenta dashed line; SCN core = yellow dashed line; shell BV trace = magenta; core BV trace = yellow. Abbreviations: AVP = vasopressin; BV=blood vessel; C=caudal; D=dorsal; IQR=interquartile range; LE=left; R=rostral; RI=right; SCN = suprachiasmatic nucleus; V=ventral.
Overview of Arterial Supply and Venous Drainage of the SCN
To visualize the arterial supply and venous drainage of the intact SCN, we scanned iDISCO-cleared tissues in horizontal view (Figures 1 and 2). This allowed visualization of the relationship between the circle of Willis originating from the internal carotid artery (ICA) and each of the arterial branches and veins that serve the nucleus.
Arteries
In the rostral SCN, there are variations among animals in arterial supply, and in some cases, the right and left sides differ within an animal (Figure 1). In contrast, the origins of caudal arteries are consistent. The rostral arterial supply, termed the “rostral” artery, was examined in 6 animals (12 SCNs). Surprisingly, the “rostral” artery is either from the superior hypophyseal artery (SHpA, Figure 1a: N = 7 SCNs), or from the anterior cerebral artery (ACA, Figure 1b(i), N = 3 SCNs), or from the anterior communicating artery (ACoA, Figure 1b(ii), N = 2 SCNs). Two animals have symmetrical arterial supply on bilateral SCNs. These 2 animals are supplied only by the SHpA. The rest 4 animals have different sources of arterial supply of the bilateral SCNs at the rostral aspect. In remaining animals in which the rostral SCN is supplied by the SHpA, the branch starts near the level of median eminence (ME) and runs rostrally and then turns sharply back before entering the SCN (Figure 1a). In animals in which the rostral SCN is supplied by the ACA (Figure 1b(i)), the ramification of the ACA first runs toward the preoptic region, where it penetrates the brain parenchyma. It then courses about 600 µm along the floor of the third ventricle and enters the SCN near its narrow rostral tip. In a third variation, the rostral SCN is supplied by ACoA (Figure 1b(ii)). Here, a branch from ACoA lying in the subarachnoid space penetrates the prechiasmatic cistern and enters the brain parenchyma by traveling under the OVLT and third ventricle to reach the rostral tip of the SCN. The caudal SCN, in all animals, is supplied by a branch from the SHpA, termed the “caudal” artery (Figure 1a, b(iii-iv)). Variation among animals occurs, however, in the origin of the SHpA artery that feeds the caudal SCN. Where the rostral SCN is supplied by the SHpA, the origin lies at the lateral of the ME. In cases where the rostral SCN is supplied by the ACA or ACoA, then the origin is more lateral to the midline than in the foregoing case.
Veins
There are inter-animal differences in the lateralization of venous drainage (Figure 2). There are 2 SCN draining pathways, here termed the “rostral” and the “caudal” veins. Emerging from the rostral area of both sides of the SCN, there are several veins running directly forward along the floor of the third ventricle above the optic chiasm and lying laterally to the portal vessels (Figure 2a). Once the “rostral” veins reach the preoptic vein (PoV), they course laterally in the prechiasmatic cistern and join the anterior cerebral vein (ACV). From this point, they run caudally and join the basal vein. The “caudal” vein forms near the SCN’s caudal-nasal edge and runs laterally and ventrally. As it leaves the parenchyma, it joins a branch of the basal vein (Figure 2b). In some animals (N = 3), the caudal veins primarily drain to the ipsilateral basal vein (Figure 1b(vi)), while in others (N = 3), both sides of the caudal veins drain to only one side of the basal vein (Figure 2c).
Portal vessels
The portal vessels emerge from the rostral SCN (Figures 1b(ii), 2a(i)), course along the floor of the ventricle and join the capillaries of the OVLT at its base. A detailed description of the SCN-OVLT portal vessels has been published (Yao et al., 2021).
Arterial Supply of SCN Core and Shell
The foregoing analyses focused on the entry point of arteries into the SCN, and we next examined the arterial sources of SCN core and shell subregions in optical slices of iDISCO-cleared brains in horizontal coronal and sagittal orientations (Figure 3a and 3b). In the case where the SCN is supplied by only the SHpA (Figure 3a(i), horizontal view), the “rostral” artery reaches the SCN rostral shell from the lateral side of the nucleus and it arborizes into capillaries near the lateral edge of the shell (Figure 3a(ii), coronal view). Another ramification from the “rostral” artery runs into the medial SCN, where it further branches near the ventral border of the SCN. Seen in the coronal view, this branch primarily supplies the core and the dorsal shell of the SCN (Figure 3a(iii)). The area supplied by this branch in the core and dorsal shell is also shown in the sagittal plane (Figure 3a(iv)). Here, the artery supplying the caudal shell can be seen entering the SCN from its caudal-dorsal edge. In the case where the rostral SCN is supplied by ACoA or ACA and the caudal SCN is supplied by SHpA (Figure 3b(i), horizontal view, ACoA), the “rostral” artery reaches the SCN from its nasal-ventral area of the shell (Figure 3b(ii), coronal view). This “rostral” artery originating from ACoA lies more medially compared with the variation in which the rostral SCN is supplied by SHpA (shown in Figure 3a).
The “caudal” artery reaches the SCN from the dorsal edge of the shell (Figure 3b(iii), coronal view). It forms 2 ramifications: One penetrates the core and supplies it (Figure 3b(iii)). The other arborizes into capillaries supplying the dorsal shell (Figure 3b(iii) also). These 2 ramifications are also connected by anastomoses. Another ramification of the “caudal” artery enters the SCN caudal shell from the dorsal side (Figure 3b(iv), coronal view). The path of the artery supplying the rostral shell and the artery supplying the core and the dorsal-caudal shell is also seen in the sagittal view (Figure 3b(v)).
Venous Drainage of SCN Core and Shell
The venous drainage of the SCN core and shell are shown in horizontal and sagittal orientations in iDISCO cleared tissue (Figure 3c and 3d). In the horizontal view (Figure 3c), the rostral shell region of the SCN is drained by the veins running forward (arrowhead) into the PoV (not shown). These veins lie lateral to the midline portal vessels (asterisk). Capillaries from the caudal aspect of the SCN shell drain into the caudal vein (arrow). In the sagittal view (Figure 3d), the “rostral” veins emerge at the rostral-most tip of the SCN. Anastomoses can be seen with a vein running just dorsal to the SCN (red asterisk). The major trunk of this vein lies outside the SCN but may participate in draining the rostral shell area of the SCN via anastomoses. The “caudal” vein penetrates the SCN and then the optic chiasm seen in the dorsal-ventral axis (arrow). Its ramifications extend widely, collecting the capillaries from these several SCN subregions.
Overview of Inter-animal Differences in SCN Arteries and Veins
A summary of SCN arteries and veins in relation to its subregions is shown in Figure 4. The most rostral and most caudal SCN have separate arterial and venous vessels, with variations among individuals. The rostral SCN shell is supplied by a branch from either the SHpA (Figure 4a), or ACA, or ACoA (Figure 4b). The caudal SCN area is always supplied by the SHpA (in both Figure 4a and 4b). The medial part of the SCN, including the core, is supplied by ACA, ACoA, or SHpA.
As for the draining vessels, the rostral SCN has multiple parallel small veins, running forward to the PoV, merging into the ACV and then joining the basal vein. The vein draining the caudal SCN shows various lateralization patterns among animals. In some animals, the “caudal” veins of bilateral SCN drain to one side of the basal vein, while in other animals, the “caudal” vein of each SCN drains to the ipsilateral basal vein (not shown). The portal vessels emerge from the most rostral shell area of the SCN (as shown in Figure 4a and 4b).
Characterization of SCN Core Versus Shell Vascular Network
In confocal images of slices containing the SCN (example of BV staining and tracing is shown in Figure 5a), 8 of 11 brains have median BV diameters that are significantly smaller in the SCN shell compared with the core of the same brain (Figure 5b). In the cleared SCN (example of traces shown in Figure 5c), a similar trend for BV median diameter is observed with 3 of 4 brains showing smaller median BV diameter in the shell (Figure 5d). Also, in this material, the shell BV length density is significantly greater than that of the core (Figure 5e). Finally, the surface area density of the shell SCN is significantly greater than the core (Figure 5f). The capillary (BV diameter < 10 µm) was also analyzed in the cleared SCN. The capillaries occupy 88.81% ± 0.02% and 83.82% ± 0.09% of total BVs in SCN shell and core separately. There is a trend that the median capillary diameter in the SCN shell is smaller than that in the core, but it is not statistically significant (Figure 5g). Bootstrapped capillary diameter distribution in the SCN shell versus core of each cleared brain also shows the tendency wherein the median capillary diameter is smaller in the shell (Suppl. Fig. S3). Distribution analysis of the normality of capillary segment diameters indicates that in all samples the core capillary diameter are normally distributed. The shell capillary diameter distribution, however, is skewed toward smaller diameter measurements (Suppl. Table S3), supporting the finding of core-shell differences. The length density of capillaries in the shell is significantly higher than in the core (Figure 5h).
Discussion
There are very few prior studies of the SCN vasculature in rodents (Akmayev, 1971; Ambach and Palkovits, 1975; Kirst et al., 2020; Scremin, 1970) and human (Polzović et al., 1990). In some studies on hypothalamic or whole brain vasculature, the SCN was not identified (Van Den Pol, 1982) or was not included in the analysis (Di Giovanna et al., 2018; Todorov et al., 2020).
We performed a detailed analysis of the mouse SCN arterial sources, venous targets, and intra-SCN capillary network as a first step to understanding how it might be organized to contribute to the newly identified SCN-OVLT portal pathway (Yao et al., 2021). The following key features of SCN vasculature emerged. First, the rostral SCN’s artery supply and venous drainage are separate from the caudal SCN. This difference on the rostral-caudal axis in the anatomical separation of the SCN between these 2 areas may be related to functional subdivisions of the SCN. Studies of sagittal slices of the SCN indicate that AVP cells have morphological differences in the rostral and caudal regions (Taub et al., 2021). Furthermore, in vivo uptake of deoxyglucose differs along the rostro-caudal axis of the SCN (Schwartz et al., 1987). This locally differentiated metabolic rate, is perhaps related to differences in the blood supply along the rostro-caudal axis of the SCN. In addition, the release of serotonin in the rostral and caudal SCN has a significantly different pattern, which may be attributable to the anatomical difference between these subregions (Ramirez et al., 1987).
Second, the SCN rostral arterial supply is variable among mice, a finding not previously reported, although individual differences in the vasculature of the circle of Willis have been described (Moore et al., 2006; Rhoton and Perlmutter, 1980). There are notably numerous individual differences in humans in circadian behaviors, such as chronotype and photosensitivity (Chellappa, 2020; Zhang and Volkow, 2023). Experiments done with mice also show broad variations in timing of locomotor activity and rate of adaptation to jetlag, which is closely related to the between-subject variations in PER2:LUC rhythmicity (Evans et al., 2015). As the brain’s regional vasculature and the blood flow are closely related to the local metabolism and functions (Takahashi, 2022), the between-subject variation in vasculature may contribute to individual differences in behaviors.
There is some disagreement on arterial supply in the few available studies in rats, though none describe inter-animal variability. Ambach and Palkovits (1975) describe the SCN arterial supply in the rats as an isolated branch from the ICA named arteria rechochiasmatica. Two branches of this artery supply the rostral and caudal SCN. This artery supply is similar to the current study where 2 branches of SHpA supply the rostral and caudal SCN. Scremin (1970), however, reported that the rat SCN is supplied only by the ACA. For veins, as shown in Ambach and Palkovits (1975), the rat SCN is drained rostrally by parallel veins to the PoV and dorsal-caudally by a vein running laterally to the basal vein but they did not report on the drainage of the ventral SCN. They also describe the numerous venous anastomoses rostral to the optic chiasm.
Third, in our within animal analyses of sliced sections, the SCN shell tends to have narrower and denser BV and capillaries than the core region. In the iDISCO-cleared tissue, a similar trend is seen in all 4 brains studied. The differences between the BV of SCN subregions suggest different hemodynamics in core and shell and the possibility that the 2 regions may play different roles in blood-borne signaling.
The diameter of SCN capillaries in relation to red blood cells (RBCs) is of interest, but difficult to unravel based on the available literature. In mouse somatosensory cortex, capillaries have a diameter of 4.4 ± 0.1 μm (Hall et al., 2014). Similarly, in another report on mouse cortex, the diameter varies between 2.0 and 5.3 μm (Hartmann et al., 2021, Fig 7 source data).
In the current study, we found that the median BV diameter in brain slices is smaller than the iDISCO-cleared tissue. In SCN shell and core, respectively, the median BV diameter was 4.02 and 4.27 µm in brain slices, while the overall median SCN BV diameter was 7.27 and 7.72 µm and the median capillary diameter was 7.06 and 7.42 µm in the cleared tissue. The latter values are consistent with the whole brain vasculature analysis reported in the iDISCO-cleared brains (Kirst et al., 2020). In that work, the authors reported that the most capillaries are measured at a diameter of 5 pixels (1.58 μm/pixel). Some of these differences may be due to differential effects of processing on tissue shrinkage. In the brain slices, PFA fixation and sucrose infiltration can cause 30% overall tissue shrinkage (Newton et al., 2002). In the cleared tissue, the iDISCO method results in 11% shrinkage in brain volume (Renier et al., 2016).
It will be important to consider the functional significance of these differences in capillary size. It has been reported that a 1%-3% change of capillary diameter is sufficient to impact the velocity of blood flow (Kisler et al., 2017). Our subregional analysis of SCN shell and core median BV diameter indicates a difference of 6.22% (4.02 vs 4.27 μm). While the mean size of mouse RBC (5.8 μm) may be larger than capillary diameters in some tissue (Namdee et al., 2015), RBCs deform to squeeze through the capillaries of a smaller diameter (Skalak and Brånemark, 1969).
Previously, the SCN vasculature density in the whole nucleus was compared with other brain regions, but the intra-SCN comparison of its subregion vasculature had never been done. Akmayev (1971) reported that in rats SCN BV length density is about 809 mm/mm3, which is lower than the paraventricular nucleus and supraoptic nucleus but higher than other hypothalamic regions assessed. Kirst et al. (2020) analyzed BV branching density in many brain regions, including the mouse SCN using light-sheet images of cleared mouse brain. The results for the branching density of SCN were confirmed by our own study and we also showed the SCN shell has higher branching density comparing with the core (Yao et al., 2021). A higher density of capillaries lends more capacity for substance exchange in the SCN shell across the BV walls. Most of the brain vasculature is thought to have a blood-brain barrier (BBB) preventing free exchange of large non-lipid molecules between the brain and blood. However, carrier-mediated transporter system or receptor-mediated transporter system can facilitate the transportation of large molecules across the BBB (Kadry et al., 2020).
Finally, the portal vessels emerge at the very rostral SCN shell capillaries, and the neurosecretions in nearby regions are possible candidates for transport by the portal vessels. The shell is abundant in AVP which is considered as an output signal from the SCN (Buijs et al., 2021). There is a saturable carrier-mediated transport system of AVP from the brain parenchyma to the blood (Banks et al., 1987), though it remains to be determined whether AVP of SCN origin courses in portal vessels to the OVLT.
In summary, a detailed examination of the SCN vasculature presents a possible pathway for SCN’s diffusible outputs signaling downstream areas. It also highlights the anatomical basis of SCN neurovascular coupling, which emphasizes the role of the vasculature in actively regulating the brain functions. For example, the glymphatic system uses perivascular space to facilitate volume signaling and waste clearance via the transportation of cerebrospinal fluid (Hablitz and Nedergaard, 2021). This activity is under the circadian control and activated during the high-amplitude, low-frequency phase of the non-rapid eye movement sleep. Neurovascular coupling also impacts the capacity of neurotransmitter synthesis locally by influencing the oxygen-limited enzymes (Drew, 2022). The structure and function of all components of NVU, including the endothelium, pericytes, astrocytes, and tight junctions, are under circadian regulation (Schurhoff and Toborek, 2023), which, in turn, impacts the permeability of the BBB (Skapetze et al., 2023). Although it is well known that many components of NVU and BBB are under the circadian regulation (reviewed in Schurhoff and Toborek, 2023; Skapetze et al., 2023), SCN neurovascular coupling within the SCN is not well understood. Using animal models deficient in vasoactive peptides of the SCN and/or their receptors may contribute to understanding neurovascular regulation in this nucleus (Takeda et al., 2009). The current study provides the anatomical foundation for future studies in exploring the neurovascular coupling underlying SCN functions, especially in its pathways for volume signaling.
Supplemental Material
sj-eps-1-jbr-10.1177_07487304231189537 – Supplemental material for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals
Supplemental material, sj-eps-1-jbr-10.1177_07487304231189537 for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals by Yifan Yao, Isabella K. Green, Alana B. Taub, Ruya Tazebay, Joseph LeSauter and Rae Silver in Journal of Biological Rhythms
Supplemental Material
sj-eps-2-jbr-10.1177_07487304231189537 – Supplemental material for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals
Supplemental material, sj-eps-2-jbr-10.1177_07487304231189537 for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals by Yifan Yao, Isabella K. Green, Alana B. Taub, Ruya Tazebay, Joseph LeSauter and Rae Silver in Journal of Biological Rhythms
Supplemental Material
sj-eps-3-jbr-10.1177_07487304231189537 – Supplemental material for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals
Supplemental material, sj-eps-3-jbr-10.1177_07487304231189537 for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals by Yifan Yao, Isabella K. Green, Alana B. Taub, Ruya Tazebay, Joseph LeSauter and Rae Silver in Journal of Biological Rhythms
Supplemental Material
sj-xlsx-4-jbr-10.1177_07487304231189537 – Supplemental material for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals
Supplemental material, sj-xlsx-4-jbr-10.1177_07487304231189537 for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals by Yifan Yao, Isabella K. Green, Alana B. Taub, Ruya Tazebay, Joseph LeSauter and Rae Silver in Journal of Biological Rhythms
Supplemental Material
sj-xlsx-5-jbr-10.1177_07487304231189537 – Supplemental material for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals
Supplemental material, sj-xlsx-5-jbr-10.1177_07487304231189537 for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals by Yifan Yao, Isabella K. Green, Alana B. Taub, Ruya Tazebay, Joseph LeSauter and Rae Silver in Journal of Biological Rhythms
Supplemental Material
sj-xlsx-6-jbr-10.1177_07487304231189537 – Supplemental material for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals
Supplemental material, sj-xlsx-6-jbr-10.1177_07487304231189537 for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals by Yifan Yao, Isabella K. Green, Alana B. Taub, Ruya Tazebay, Joseph LeSauter and Rae Silver in Journal of Biological Rhythms
Supplemental Material
sj-xlsx-7-jbr-10.1177_07487304231189537 – Supplemental material for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals
Supplemental material, sj-xlsx-7-jbr-10.1177_07487304231189537 for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals by Yifan Yao, Isabella K. Green, Alana B. Taub, Ruya Tazebay, Joseph LeSauter and Rae Silver in Journal of Biological Rhythms
Supplemental Material
sj-xlsx-8-jbr-10.1177_07487304231189537 – Supplemental material for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals
Supplemental material, sj-xlsx-8-jbr-10.1177_07487304231189537 for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals by Yifan Yao, Isabella K. Green, Alana B. Taub, Ruya Tazebay, Joseph LeSauter and Rae Silver in Journal of Biological Rhythms
Supplemental Material
sj-xlsx-9-jbr-10.1177_07487304231189537 – Supplemental material for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals
Supplemental material, sj-xlsx-9-jbr-10.1177_07487304231189537 for Vasculature of the Suprachiasmatic Nucleus: Pathways for Diffusible Output Signals by Yifan Yao, Isabella K. Green, Alana B. Taub, Ruya Tazebay, Joseph LeSauter and Rae Silver in Journal of Biological Rhythms
Footnotes
Acknowledgements
Work from our labs described herein was supported by National Science Foundation (NSF) grant 1749500 (to RS). National Institutes of Health (NIH) grant R21NS134228 (to RS). Light-sheet microscopy was performed with support from the Zuckerman Institute’s Cellular Imaging platform and NIH (1S10OD023587-01).
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Note
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
