Abstract
Prior research indicates that sleep restriction, sleep deprivation, and circadian misalignment diminish positive affect, whereas effects on negative affect are inconsistent. One potential factor that may influence an individual’s affective response to sleep restriction, sleep deprivation, and circadian misalignment is chronotype. Later chronotypes generally report higher negative affect and lower positive affect under typical sleep conditions; however, there is mixed evidence for an influence of chronotype on affective responses to sleep restriction and sleep deprivation. The present study examined the effect of chronotype on positive and negative affect during sleep restriction and subsequent total sleep deprivation. Sixteen healthy adults (Mage = 28.2 years, SDage = 11.6 years) were classified as earlier or later chronotypes using multiple chronotype definitions: morningness-eveningness (MEQ), mid-sleep on free days corrected (MSFsc), habitual mid-sleep timing, dim light melatonin onset (DLMO), and phase relationship between DLMO and bedtime. Participants completed a 10-day protocol with one night of sleep restriction and subsequent 28 h total sleep deprivation. Affect was assessed hourly during scheduled wakefulness with the Positive and Negative Affect Schedule (PANAS). Data were analyzed with mixed-model analyses of variance (ANOVAs). During sleep restriction and subsequent sleep deprivation, positive affect decreased and negative affect increased. Across all chronotype measures, relatively later chronotypes demonstrated vulnerability to increased negative affect during sleep loss. The influence of chronotype on positive affect during sleep loss varied by chronotype measure. These findings suggest later chronotypes are more vulnerable to affective impairments during sleep loss and circadian misalignment, even when late chronotype is not extreme.
Keywords
Sleep loss and circadian misalignment (e.g. wakefulness during the biological night) are increasingly recognized as important contributors to poor mental and physical health (Barnes and Drake, 2015; Foster, 2020; McHill and Wright, 2017). Affect is one mechanism that may link sleep loss and circadian misalignment to adverse health outcomes. Affect describes acute positive (e.g. excited, enthusiastic) and negative (e.g. distressed, hostile) mood states (Watson and Tellegen, 1985). Positive and negative affect are orthogonal states that do not represent opposite ends of a single affect spectrum, such that low positive affect states (e.g. dull, sluggish) are distinct from high negative affect states, and low negative affect states (e.g. relaxed, placid) are distinct from high positive affect states (Watson and Tellegen, 1985). Higher positive affect is associated with more favorable health outcomes (Dockray and Steptoe, 2010), including lower risk of cardiovascular disease (Sin, 2016) and mortality (Chida and Steptoe, 2008). Conversely, higher negative affect is implicated in adverse health outcomes, including mood and anxiety disorders (Hofmann et al., 2012) and increased number of chronic health conditions (Willroth et al., 2020). Furthermore, affect is impacted by sleep restriction and deprivation, such that positive affect is consistently diminished by sleep restriction (Alfano et al., 2020; Cox and Olatunji, 2021; Dagys et al., 2012; Finan et al., 2017; McMakin et al., 2016; Reddy et al., 2017; Rossa et al., 2014; Saksvik-Lehouillier et al., 2020; Talbot et al., 2010) and total sleep deprivation (Franzen et al., 2008; Grèzes et al., 2021; Stenson et al., 2021). Notably, sleep restriction and deprivation induce circadian misalignment by evoking wakefulness during the biological night (Depner et al., 2019; Eckel et al., 2015; Markwald et al., 2013), and positive affect is also decreased during circadian misalignment (Chellappa et al., 2020; Sprecher et al., 2019). Although data consistently show that positive affect is decreased during sleep restriction, sleep deprivation, and circadian misalignment, evidence for how sleep restriction impacts negative affect is mixed. Some findings suggest that negative affect increases with sleep restriction (Baum et al., 2014; Dinges et al., 1997; McMakin et al., 2016); however, others have found no significant effect of sleep restriction on negative affect (Alfano et al., 2020; Cox and Olatunji, 2021; Dunn et al., 2018; Grèzes et al., 2021; Rossa et al., 2014; Saksvik-Lehouillier et al., 2020; Stenson et al., 2021; Talbot et al., 2010), despite some observations of similar effect sizes for increased negative affect as those observed for decreased positive affect (Reddy et al., 2017; Reid et al., 2023).
These inconsistencies in the literature may be due to unmeasured individual differences that influence affective response to sleep loss and circadian misalignment. Indeed, prior work has found that some individuals are more susceptible to impairments during sleep loss than others. For example, several studies have supported trait-like vulnerability of neurocognitive deficits (e.g. attention, alertness, affect) in response to total sleep deprivation (Van Dongen et al., 2004), sleep restriction (Dennis et al., 2017), and combined sleep restriction and circadian misalignment (Sprecher et al., 2019). That is, performance impairments in response to sleep loss and circadian misalignment vary between individuals, but are consistent within an individual, across repeated trials. Interestingly, prior work attempting to identify individual difference factors that predict these trait-like impairments has yielded few reliable predictors (Galli et al., 2022). To date, the best predictor for neurocognitive performance, including mood, during sleep loss is baseline performance (Galli et al., 2022; Van Dongen et al., 2004). Importantly, previous research has largely focused on cognitive performance, rather than affect states, and there is limited understanding of factors that may predict affective response to sleep restriction and deprivation.
One factor that may influence affective responses to sleep loss and circadian misalignment is chronotype. Chronotype describes the temporal organization of an individual’s behavior (e.g. sleep timing) and represents an interaction between the biological circadian clock and environmental input (e.g. light exposure) (Duffy et al., 2001; Roenneberg et al., 2003, 2007). Later chronotypes (i.e. those who tend to go to sleep and wake up later) tend to have higher negative affect (Carciofo, 2020; Cox and Olatunji, 2019) and lower positive affect than earlier chronotypes (Biss and Hasher, 2012; Dagys et al., 2012; Miller et al., 2015). In addition, later chronotypes exhibit a lower and later positive affect amplitude compared to early chronotypes (Miller et al., 2015). Together these findings suggest that chronotype may be a potential predictor of individual differences in affective responses to sleep loss and circadian misalignment.
However, limited work has been conducted on this question, and findings are mixed. Though later chronotype has been found to be associated with increased negative affect following sleep restriction in adults (Cox and Olatunji, 2021), no influence of chronotype has been found on affective response to total sleep deprivation in adults (Galli et al., 2022) or to sleep restriction in adolescents (Dagys et al., 2012). To further complicate matters, one study found that later chronotypes reported decreased negative affect following total sleep deprivation and sleep restriction (Selvi et al., 2007). These discrepant findings could be due to how affect was measured. In these studies, affect was measured before and after sleep loss (Cox and Olatunji, 2021; Dagys et al., 2012; Selvi et al., 2007) or aggregated by day (Galli et al., 2022). Recent evidence has shown that negative affect increases and positive affect decreases during the biological night (Emens et al., 2020). As such, findings could be influenced by the time of day that affect was measured, and the potential influence of chronotype may not be captured if affect was only measured at one or a few time points. Furthermore, extant research has exclusively defined chronotype based on self-report measures of morningness-eveningness, which assess personality traits and preferences. The biological chronotype construct is better understood as phase of entrainment, or individual differences in circadian timing relative to the T-cycle or daylength (Duffy and Wright, 2005; Pittendrigh and Daan, 1976; Roenneberg, 2015). Thus, measures of behavior (i.e. sleep timing) or physiology (i.e. circadian melatonin phase, phase relationships between melatonin phase and bedtime) that more closely approximate/estimate the phase of entrainment are needed to examine the influence of chronotype on affective responses to sleep loss. Finally, the majority of the extant chronotype literature has focused on extreme types. Although such work is informative for understanding the health implications of extreme typology, it is unclear whether findings in the extremes generalize to those in the intermediate range. This is a notable gap, as the majority of the population are categorized as intermediate chronotypes.
To address these limitations in the extant literature, we examined whether intermediate earlier and later chronotypes, defined by self-report (morningness-eveningness, mid-sleep on free days corrected), behavior (mid-sleep timing from 2 weeks of a consistent sleep schedule), and physiology (dim light melatonin onset [DLMO] timing, phase relationship between DLMO and bedtime), differ in their positive and negative affective response, measured frequently, during sleep restriction and subsequent sleep deprivation. We examined intermediate (i.e. relatively earlier and later) chronotypes, rather than extreme early and late chronotypes, to improve our understanding of how differences in degree of chronotype within healthy individuals influences affective responses to sleep loss. In this preliminary investigation, we hypothesized that positive affect would decrease and negative affect would increase during sleep restriction and sleep deprivation, later chronotypes would report lower positive and higher negative affect than earlier chronotypes overall, and later chronotypes would report lower positive and higher negative affect than earlier chronotypes in response to sleep restriction and subsequent sleep deprivation.
Methods
Participants
Sixteen healthy adults (6 female [i.e. biological sex]; Mage = 28.2, SD = 11.6, range: 18-52; MBMI = 23.2, SD = 2.4, range = 18.80-26.3) were recruited from the community by online advertisements, flyers, and word-of-mouth. Of the 16 participants, 14 were non-Hispanic White (5 female, 9 male), 1 male was non-Hispanic Asian, and 1 female was Hispanic or Latino White. A health history interview and self-report questionnaires were conducted at the Sleep and Chronobiology Laboratory to confirm the following inclusion criteria: no medical, psychiatric, or sleep disorders, not taking medications, not pregnant, lactating, or had given birth in the past 6 months, no travel outside > 1 time zone in the past 3 weeks, no night shift work in the 3 months, habitual sleep duration 7-9 h. There were no exclusion criteria related to chronotype.
Procedure
Participants completed the Morningness-Eveningness Questionnaire (MEQ) (Horne and Ostberg, 1976) and the Munich Chronotype Questionnaire (MCTQ) (Roenneberg et al., 2003) at screening. Prior to the in-laboratory visit, participants abstained from caffeine, alcohol, and nicotine for 4 days. Participants kept a consistent, habitual 8-h sleep schedule for 1 week at home, which was verified by wrist-worn actigraphs (Actiwatch-L, Respironics), a sleep diary, and time-stamped sleep-wake times (voicemail or online form). The sleep schedule was selected by the participants to be comparable to their self-reported habitual bedtime and waketime on work/school days and free days at screening. During the 1 week of a consistent 8-h sleep schedule, participants went to bed and woke up at the same time every day, including free days. Participants who deviated by > 30 min from their sleep schedule more than once were dismissed from the study. Mid-sleep timing was calculated from the time-stamped sleep-wake times. Participants then spent 3 days in the laboratory. Urine toxicology and alcohol breath testing (Lifeloc Technologies Model FC10) were conducted at the start of the laboratory visit to confirm participants were free of recreational substances. Participant’s sleep was restricted to relative clock hour 0400-0800 h (defined as time in the protocol relative to the participants habitual sleep schedule, where 2400 and 0800 are arbitrarily defined as bedtime and waketime, respectively) on night 1 and subsequent 28 h of total sleep deprivation. Participants then had 10 h of recovery sleep in the lab (see Figure 1).

Experimental protocol. Timing was based on participants habitual sleep schedules, where habitual bedtime was arbitrarily defined as relative clock hour 2400. Black bars indicate scheduled sleep. Gray bars indicate scheduled wakefulness in dim light. Numbers in gray bars indicate Positive and Negative Affect Schedule (PANAS) administrations.
During the in-laboratory visits, participants remained in an environment free from time cues in individual temperature-controlled and sound-attenuated suites. Light levels were dim (<10 lux maximum at 72 inches in the direction of the ceiling light fixtures, ~1.9 lux, ~0.6 W/m2 in the angle of gaze) during scheduled wakefulness and dark (0 lux) during scheduled sleep. Wakefulness during sleep restriction and sleep deprivation was ensured via continuous monitoring by research staff.
Affective response to combined sleep restriction and sleep deprivation was measured with the Positive and Negative Affect Schedule (PANAS) (Watson et al., 1988). The PANAS is a 20-item self-report measure of positive and negative affect. Participants were instructed to respond to each item considering how they were feeling “right now, that is, at the present moment.” Instead of Likert-type scales, participants rated items with visual analog scales, such that each item was measured on a 0-100 scale, and positive and negative affect items were summed to calculate total positive and negative affect scores, respectively. The highest possible score on each subscale was 1,000 (Sprecher et al., 2019). The PANAS was administered every hour during scheduled wakefulness at the end of a cognitive performance battery that contained self-report measures of sleepiness and mood, a reaction time task, and an addition task, from an unpublished study.
Saliva samples were collected between relative clock hour 1600 h and 1100 h during sleep deprivation to measure melatonin levels (hourly 1800-0200 and 0800-1100; otherwise every 2 h). Melatonin was assessed by radioimmunoassay (Alpco Inc.) DLMO25% was defined as the linear interpolated point in time at which melatonin levels reached 25% of the fitted peak-to-trough amplitude of the 3-harmonic of each individual’s data, and DLMO3pg was defined as the point in time at which melatonin levels exceeded 3 pg/mL (Wright et al., 2001).
All participants provided written informed consent, and the protocol was approved by the Institutional Review Board of the University of Colorado Boulder.
Statistical Analysis
Earlier and later chronotypes were identified from the highest and lowest ~30% of the sample based on MEQ, mid-sleep on free days corrected (MSFsc) from the MCTQ, average mid-sleep timing the week prior to laboratory testing, DLMO25%, DLMO3pg, phase relationship between DLMO25% and bedtime, and phase relationship between DLMO3pg and bedtime. We calculated phase relationships as the difference between DLMO clock hour and bedtime (week of consistent, habitual sleep schedule at home), such that negative values indicate a DLMO earlier than bedtime, and positive values indicate a DLMO later than bedtime (Duffy and Wright, 2005). See Supplemental Table S1 for participant classification to chronotype groups by each chronotype measure.
We conducted statistical analysis in SPSS version 29. We confirmed differences between chronotype groups with independent samples t-tests. We examined sex differences with chi-square tests. We examined associations between chronotype measures across all participants with bivariate correlations. We tested the effect of chronotype on positive and negative affective response to sleep restriction and subsequent total sleep deprivation with 14 linear mixed model analyses of variance (ANOVAs) including baseline affect (i.e. affect reported at the first testing battery), time (i.e. relative clock hour), chronotype, and a chronotype x time interaction term as fixed factors. Given unequal distribution of sex between earlier and later chronotypes defined by DLMO25% (see below), sex was included as a covariate in this model. Planned pairwise comparisons using simple main effects with modified Bonferroni correction (Keppel, 1991) were performed comparing affect at each timepoint between chronotypes. We calculated partial η2 in R version 4.3.0 to evaluate effect sizes for model main effects and simple main effects using standard thresholds (0.01 = small, 0.06 = medium, 0.14 = large). We report effect size (small, medium, large) immediately prior to the significance for all results (e.g. small, significant; medium, non-significant).
Results
Descriptive Differences Between Earlier and Later Chronotypes
Descriptive statistics and group comparisons of earlier and later chronotypes by each metric are shown in Supplemental Table S2. As expected, later chronotypes were significantly later on all chronotype measures compared to earlier chronotypes (ps < 0.05). There was a significant sex difference in earlier and later chronotypes defined by DLMO25%, χ2(1, 8) = 4.80, p < 0.05, such that females were overrepresented in the earlier chronotype group and underrepresented in the later chronotype group. There were no sex differences on any other chronotype measure (ps > 0.10). Chronotypes did not differ on self-reported sleep duration at baseline (ps > 0.10) (Suppl. Table S2). Across the full sample, self-report measures of chronotype tended to associate more strongly with each other than with physiological measures, and vice versa (Suppl. Table S3). According to typical chronotype definitions on the MEQ, 1 participant was a late chronotype, 7 were intermediate chronotypes, and 8 were early chronotypes.
Chronotype Defined by MEQ
There was a large, significant decrease in positive affect during sleep restriction and subsequent sleep deprivation (p < 0.001; Table 1). Later chronotypes reported small, significantly higher positive affect throughout the protocol (p < 0.01; Table 1). There was not a significant chronotype x time interaction (p = 0.91), and results of planned pairwise comparisons revealed no significant differences between chronotypes at any relative clock hour (ps > 0.05) (Table 1; Figure 2a).
Results of mixed model ANOVAs examining differences in positive and negative affect in response to sleep restriction and subsequent sleep deprivation between earlier and later chronotypes, controlling for baseline affect.
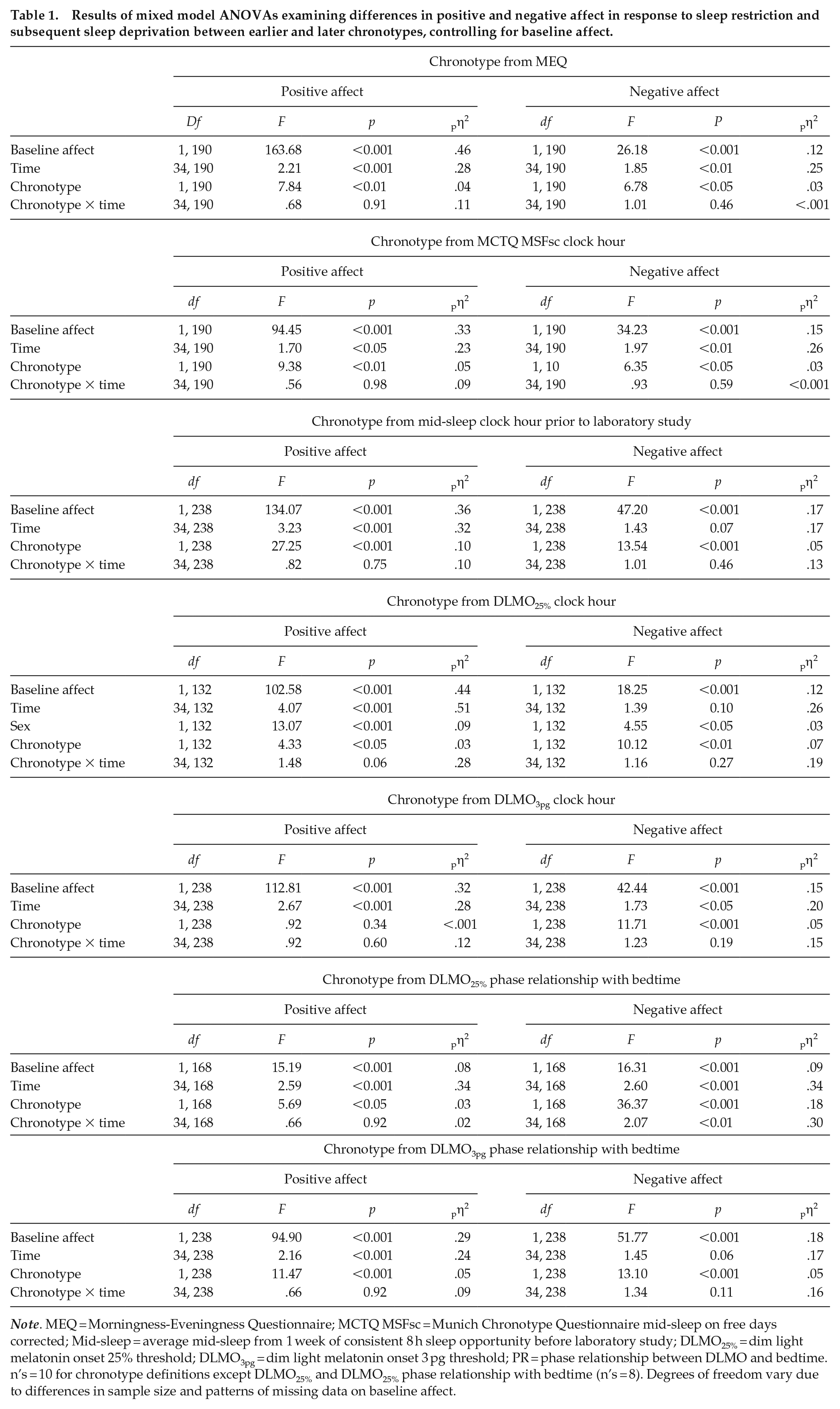
Note. MEQ = Morningness-Eveningness Questionnaire; MCTQ MSFsc = Munich Chronotype Questionnaire mid-sleep on free days corrected; Mid-sleep = average mid-sleep from 1 week of consistent 8 h sleep opportunity before laboratory study; DLMO25% = dim light melatonin onset 25% threshold; DLMO3pg = dim light melatonin onset 3 pg threshold; PR = phase relationship between DLMO and bedtime. n’s = 10 for chronotype definitions except DLMO25% and DLMO25% phase relationship with bedtime (n’s = 8). Degrees of freedom vary due to differences in sample size and patterns of missing data on baseline affect.
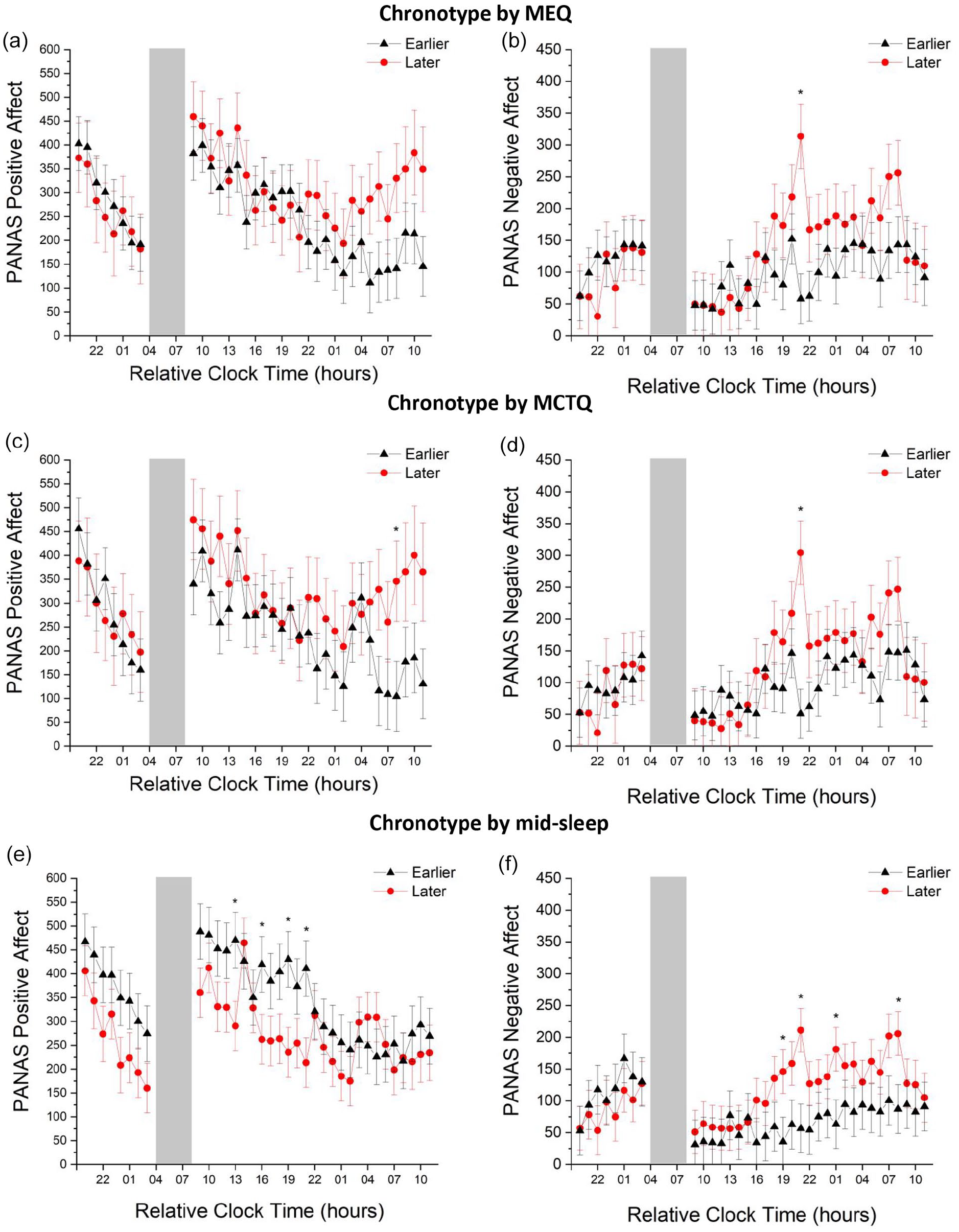
Marginal means of affect reported on the Positive and Negative Affect Schedule (PANAS) over time during sleep restriction and subsequent sleep deprivation in earlier and later chronotypes measured by self-report and behavioral metrics. (a and b) positive and negative affect, respectively, in earlier and later chronotypes defined by the Morningness-Eveningness Questionnaire (MEQ). (c and d) positive and negative affect, respectively, in earlier and later chronotypes defined by Munich Chronotype Questionnaire (MCTQ) mid-sleep on free days corrected (MSFsc). (e and f) positive and negative affect, respectively, in earlier and later chronotypes defined by average mid-sleep during 1 week of a consistent sleep schedule prior to laboratory study. Time is relative to the participants habitual sleep schedule. Black lines with triangles depict earlier chronotypes. Red lines with circles depict later chronotypes. Gray bar indicates scheduled sleep opportunity. Error bars show standard error of the mean. Asterisks (*) indicate significant differences in affect between earlier and later chronotypes. Modified Bonferroni-corrected p value = 0.048.
There was a large, significant increase in negative affect during sleep restriction and subsequent sleep deprivation (p < 0.01; Table 1). Later chronotypes reported small, significantly higher negative affect throughout the protocol (p < 0.05; Table 1). Although there was not a significant chronotype x time interaction (p = 0.46), results of planned pairwise comparisons revealed later chronotypes reported a medium, significant increase in negative affect compared to earlier chronotypes at relative clock hour 2100 (p < 0.001; pη2 = 0.08) (Table 1; Figure 2b).
Chronotype Defined by MCTQ MSFsc Clock Hour
There was a large, significant decrease in positive affect during sleep restriction and subsequent sleep deprivation (p < 00.05; Table 1). Later chronotypes reported small, significantly higher positive affect throughout the protocol (p < 0.01; Table 1). Although there was not a significant chronotype x time interaction (p = 0.98), results of planned pairwise comparisons revealed later chronotypes reported a small, significant increase in positive affect compared to earlier chronotypes at relative clock hour 0800 (p < 0.048; pη2 = 0.02) (Table 1; Figure 2c).
There was a large, significant increase in negative affect during sleep restriction and subsequent sleep deprivation (p < 0.01; Table 1). Later chronotypes reported small, significantly higher negative affect throughout the protocol (p < 0.05; Table 1). Although there was not a significant chronotype x time interaction (p = 0.59), results of planned pairwise comparisons revealed later chronotypes reported a medium increase in negative affect compared to earlier chronotypes at relative clock hour 2100 (p < 0.001; pη2 = 0.08) (Table 1; Figure 2d and e).
Chronotype Defined by Mid-Sleep Timing Clock Hour
There was a large, significant decrease in positive affect during sleep restriction and subsequent sleep deprivation (p < 0.001; Table 1). Later chronotypes reported medium, significantly lower positive affect throughout the protocol (p < 0.001; Table 1). Although there was not a significant chronotype x time interaction (p = 0.75), results of planned pairwise comparisons revealed later chronotypes reported small decreases in positive affect compared to earlier chronotypes at relative clock hours 1300, 1600, 1900, and 2100 (ps < 0.048; pη2’s = 0.02-0.03) (Table 1; Figure 2e).
There was a large, non-significant trend increase in negative affect during sleep restriction and subsequent sleep deprivation (p = 0.07; Table 1). Later chronotypes reported small, significantly higher negative affect throughout the protocol (p < 0.001; Table 1). Although there was not a significant chronotype x time interaction (p = 0.46), results of planned pairwise comparisons revealed later chronotypes reported small increases in negative affect compared to earlier chronotypes at relative clock hours 1900, 2100, 0100, and 0800 (ps < 0.048; pη2’s = 0.02-0.04) (Table 1; Figure 2f).
Chronotype Defined by DLMO25% Clock Hour
There was a large, significant decrease in positive affect during sleep restriction and subsequent sleep deprivation (p < 0.001; Table 1). Female participants and later chronotypes reported medium and small, significantly lower positive affect throughout the protocol, respectively (p < 0.05; Table 1). There was a non-significant trend chronotype x time interaction (p = 0.06), such that later chronotypes reported small to medium decreases in positive affect compared to earlier chronotypes at relative clock hours 2300, 1300, 1600, 1900, and 2100 during total sleep deprivation following sleep restriction (ps < 0.048; pη2’s = 0.02-0.07) (see Table 1, Figure 3a).
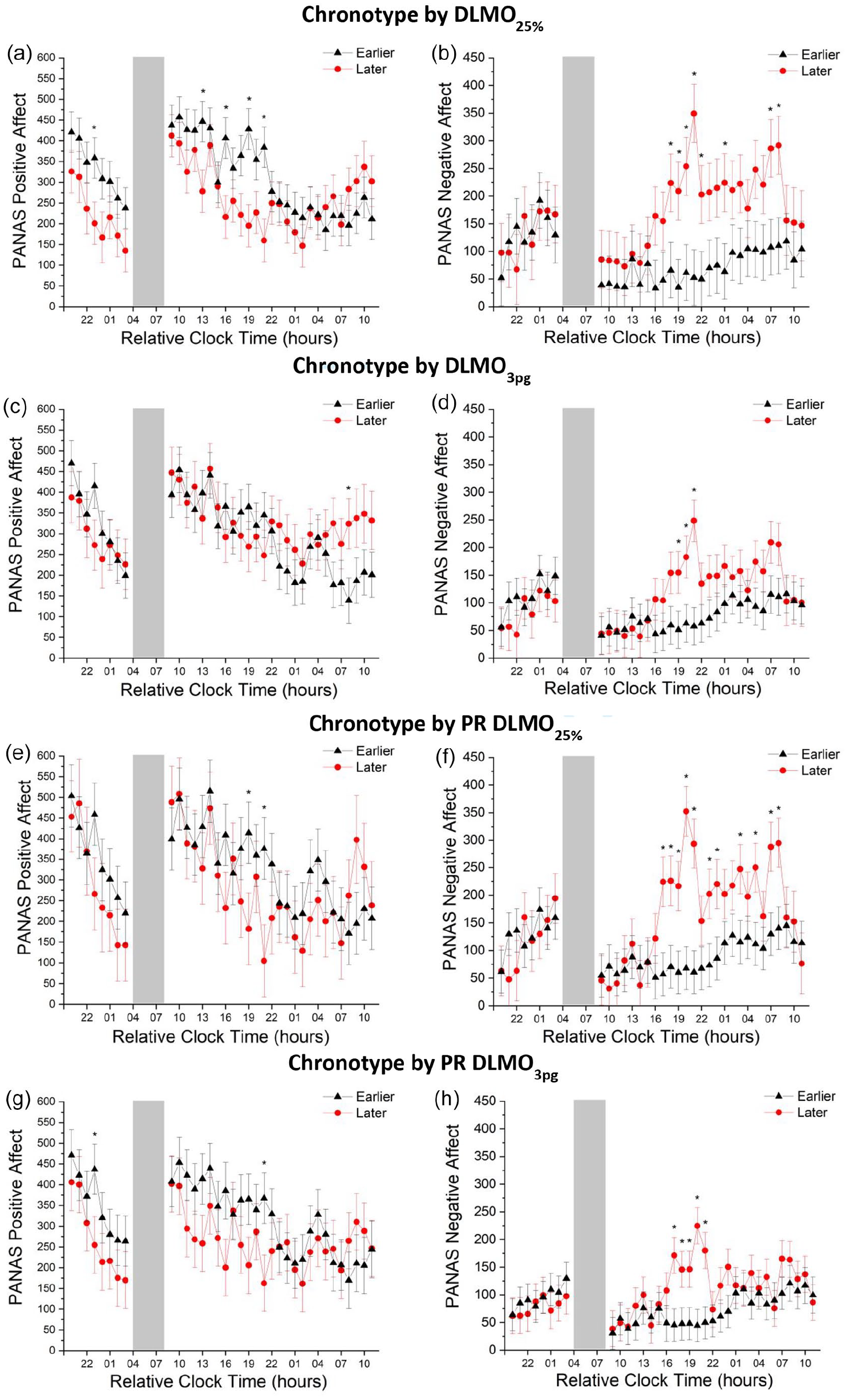
Marginal means of affect reported on the Positive and Negative Affect Schedule (PANAS) over time during sleep restriction and subsequent sleep deprivation in earlier and later chronotypes defined by DLMO clock hour with sex included as a covariate. (a and b) positive and negative affect, respectively, in earlier and later chronotypes defined by DLMO using the 25% threshold method. (c and d) positive and negative affect, respectively, in earlier and later chronotypes defined by DLMO using the 3 pg threshold. (e and f) positive and negative affect, respectively, in earlier and later chronotypes defined the phase relationship between DLMO (25% threshold method) and bedtime. (g and h) positive and negative affect, respectively, in earlier and later chronotypes defined the phase relationship between DLMO (3 pg threshold method) and bedtime. Time is relative to the participants habitual sleep schedule. Black lines with triangles depict earlier chronotypes. Red lines with circles depict later chronotypes. Gray bar indicates scheduled sleep opportunity. Error bars show standard error of the mean. Asterisks (*) indicate significant differences in affect between earlier and later chronotypes. Modified Bonferroni-corrected p value = 0.048.
There was a large, trend-level increase in negative affect during sleep restriction and subsequent sleep deprivation (p = 0.10; Table 1). Female participants and later chronotypes reported small and medium, significantly higher negative affect throughout the protocol, respectively (p < 0.05; Table 1). Although there was not a significant chronotype x time interaction (p = 0.27), results of planned pairwise comparisons revealed later chronotypes reported small to medium increases in negative affect compared to earlier chronotypes at relative clock hours 1800-2200, 0100, 0700, and 0800 during total sleep deprivation following sleep restriction (ps < 0.048; pη2’s = 0.03-0.11) (see Table 1, Figure 3b).
Chronotype Defined by DLMO3pg Clock Hour
There was a large, significant decrease in positive affect during sleep restriction and subsequent sleep deprivation (p < 0.001; Table 1). Chronotypes did not significantly differ in positive affect throughout the protocol (p = 0.34). Although there was not a significant chronotype x time interaction (p = 0.60), results of planned pairwise comparisons revealed later chronotypes reported a small increase in positive affect compared to earlier chronotypes at relative clock hour 0800 (p < 0.048; pη2 = 0.02) (see Table 1, Figure 3c).
There was a large, significant increase in negative affect during sleep restriction and subsequent sleep deprivation (p < 0.05; Table 1). Later chronotypes reported small, significantly higher negative affect throughout the protocol (p < 0.001; Table 1). Although there was not a significant chronotype x time interaction (p = 0.19), results of planned pairwise comparisons revealed later chronotypes reported small to medium increases in negative affect compared to earlier chronotypes at relative clock hours 1900-2100 (ps < 0.048; pη2’s = 0.02-0.06) (see Table 1, Figure 3d).
Chronotype Defined by Phase Relationship Between DLMO25% and Bedtime
There was a large, significant decrease in positive affect during sleep restriction and subsequent sleep deprivation (p < 0.001; Table 1). Later chronotypes reported small, significantly lower positive affect throughout the protocol (p < 0.05; Table 1). Although there was not a significant chronotype x time interaction (p = 0.92), results of planned pairwise comparisons revealed later chronotypes reported small decreases in positive affect compared to earlier chronotypes at relative clock hours 1900 and 2100 (ps < 0.048; pη2’s = 0.02-0.03) (see Table 1, Figure 3e).
There was a large, significant increase in negative affect during sleep restriction and subsequent sleep deprivation (p < 0.001; Table 1). Later chronotypes reported large, significantly higher negative affect throughout the protocol (p < 0.001; Table 1). There was a significant chronotype x time interaction (p < 0.01), and results of planned pairwise comparisons revealed later chronotypes reported small to medium increases in negative affect compared to earlier chronotypes at relative clock hours 1700-2100, 2300, 2400, 0300, 0500, 0700, and 0800 (ps < 0.048; pη2’s = 0.03-0.12) (see Table 1, Figure 3f).
Chronotype Defined by Phase Relationship Between DLMO3pg and Bedtime
There was a large, significant decrease in positive affect during sleep restriction and subsequent sleep deprivation (p < 0.001; Table 1). Later chronotypes reported small, significantly lower positive affect throughout the protocol (p < 0.001; Table 1). Although there was not a significant chronotype x time interaction (p = 0.92), results of planned pairwise comparisons revealed later chronotypes reported small decreases in positive affect compared to earlier chronotypes at relative clock hours 2300 and 2100 (ps < 0.048; pη2’s = 0.02) (see Table 1, Figure 3g).
There was a large, non-significant trend increase in negative affect during sleep restriction and subsequent sleep deprivation (p = 0.06; Table 1). Later chronotypes reported small, significantly higher negative affect throughout the protocol (p < 0.001; Table 1). Although there was not a significant chronotype x time interaction (p = 0.11), results of planned pairwise comparisons revealed later chronotypes reported small to medium increases in negative affect compared to earlier chronotypes at relative clock hours 1700-2100 (ps < 0.048; pη2’s = 0.02-0.07) (see Table 1, Figure 3h).
Discussion
Consistent with our hypothesis, we found a large decrease in positive affect during sleep restriction and subsequent sleep deprivation. This finding adds to the robust literature indicating that sleep loss and circadian misalignment detrimentally impact positive affect. We also found a large increase in negative affect during sleep restriction and subsequent sleep deprivation. Though consistent with findings from a small number of studies (Baum et al., 2014; Dinges et al., 1997; McMakin et al., 2016), this result is in contrast with findings from the majority of prior studies suggesting no effect of sleep loss on negative affect. We may have been able to detect an effect of sleep loss on negative affect due to the high frequency with which we measured affect, whereas most of the extant literature measured affect before and after sleep loss, and since the later types appeared to show larger increases in negative affect. Indeed, findings from one recent study suggested a medium (though non-significant) effect size for increased negative affect following one night of experimental sleep fragmentation when affect was measured hourly (Reid et al., 2023). Still, significant increases in negative affect following sleep loss have been found in some prior studies which did not utilize frequent affect sampling (Baum et al., 2014; Dinges et al., 1997; McMakin et al., 2016). Together these findings suggest the detrimental effects of sleep loss and circadian misalignment on negative affect are less robust than those on positive affect.
We also found small to large effects for higher negative affect in later chronotypes compared to earlier chronotypes during sleep restriction and subsequent sleep deprivation across all chronotype metrics. Higher negative affect in later chronotypes was most commonly reported during relative evening hours during total sleep deprivation. The extant literature on the influence of chronotype on negative affect during sleep loss is mixed, with studies showing effects in opposite directions (Cox and Olatunji, 2021; Selvi et al., 2007) or no effect of chronotype (Dagys et al., 2012). Notably, these studies all used morningness-eveningness measures of chronotype, which we found to more weakly influence negative affect relative to behavioral (mid-sleep) or physiological (DLMO, phase relationship) measures of chronotype. Frequent affect sampling may also account for our ability to detect the effect of chronotype on negative affect. Indeed, given that negative affect differences between chronotypes emerged in the middle of the sleep deprivation procedure, an effect of chronotype would not be observed if affect were only sampled pre- and post-sleep loss.
In contrast, the influence of chronotype on positive affect during sleep restriction and subsequent sleep deprivation varied by chronotype measure. When measured by behavior (mid-sleep) and physiology (DLMO, phase relationship), we found small to medium effects for lower positive affect in later chronotypes compared to earlier chronotypes, with the exception of chronotype measured by DLMO3pg. It is unclear why DLMO3pg showed a different pattern of results from the DLMO25% and both DLMO phase relationship variables. When measured by self-report (MEQ, MCTQ), we found small effects for higher positive affect in later chronotypes compared to earlier chronotypes. These findings are inconsistent with prior work showing no effect of chronotype on positive affective response to sleep loss (Cox and Olatunji, 2021; Dagys et al., 2012), as well as work demonstrating lower positive affect in late chronotypes under conditions of typical sleep (Miller et al., 2015). These opposing findings may reflect the limitations of self-report measures of chronotype. Relatedly, given that our chronotype groups are relative to the sample, self-report measures of chronotype may demonstrate better agreement with behavioral or physiological measures when predicting affective responses to sleep loss in more extreme chronotypes. Notably, physiological measures of chronotype have not been previously used to examine whether chronotype impacts affective response to sleep loss. Indeed, though preliminary, our findings suggest that circadian physiology measures of chronotype may be more robust than self-report measures of chronotype in this context.
Accumulating evidence likewise links later chronotype to poor outcomes, including lower social support (Walsh et al., 2022), mood and anxiety disorders (Antypa et al., 2016; Wood et al., 2009), metabolic disorders (Yu et al., 2015), and cardiovascular disease (Merikanto et al., 2013). Notably, our sample consisted of physically and mentally healthy individuals, indicating the link between chronotype and affect is not better accounted for by health conditions. Further, given that all participants kept a consistent sleep opportunity prior to the in-laboratory procedures, our findings suggest that the influence of chronotype on affective responses to sleep loss may not be explained by greater variability in sleep timing in later chronotypes. Likewise, earlier and later chronotypes did not differ in baseline self-reported sleep duration, and the prescribed 8 h sleep opportunity for 1 week prior to in-laboratory data collection would also minimize sleep debt prior to experimental sleep loss. Thus, our findings are likely not attributable to pre-existing sleep debt in the later chronotypes relative to earlier chronotypes, which theoretically could contribute to between-chronotype differences in naturalistic comparisons of chronotypes. The robust effect of later chronotype on negative affect is particularly notable given that we did not study extreme early and late chronotypes. These findings support conceptualizing chronotype across a continuous spectrum from early to late and suggest that even a moderate degree of lateness may be a vulnerability for adverse outcomes during sleep loss. The observation of this effect in relative, versus extreme, late chronotypes highlights a need for additional research examining non-extreme chronotype presentations, which may be more generalizable to the average adult.
Despite the small effect sizes observed, it is notable that chronotype influenced affective responses to sleep loss over and above the influence of baseline affect, given that the best known predictor of impairments following sleep loss is baseline performance (Galli et al., 2022; Van Dongen et al., 2004). Consistent with prior work on the predictive value of baseline performance, we likewise found that baseline affect significantly predicted change in affect during sleep restriction and subsequent sleep deprivation. However, with the exception of chronotype defined by DLMO3pg, we found no differences in positive or negative affect at baseline between the chronotype groups, suggesting that the observed effects are not accounted for by group differences in affect prior to experimental sleep loss. Later chronotypes exhibit more sleep disturbances than early chronotypes (Vitale et al., 2015; Vollmer et al., 2017); thus, it is possible that late chronotypes may be more vulnerable to the affective consequences of sleep loss. Notably, our sample consisted of healthy sleepers, suggesting this vulnerability may be present even in later chronotypes without significant sleep disturbance. Still, given the small sample size, it is important to replicate this finding in a larger sample with moderate and more extreme chronotypes.
In this preliminary investigation, we found support for our hypotheses that positive affect would decrease and negative affect would increase in response to sleep restriction and subsequent sleep deprivation, and later chronotypes would report higher negative affect in response to sleep restriction and subsequent sleep deprivation. We found partial support for the hypothesis that later chronotypes would report lower positive affect in response to sleep restriction and subsequent sleep deprivation. Though this study has several strengths, including rigorous control of prior sleep at participants habitual times, sleep manipulation controlling for prior time awake, frequent measurement of affect, and the inclusion of multiple indicators of chronotype spanning multiple levels of analysis, it is important to consider our findings within the context of the study limitations. First, we identified earlier and later chronotypes post hoc, which may have limited our ability to detect differences in affective responses to sleep loss. Replication in moderate and more extreme chronotypes is needed. Second, the sample size was relatively small, and future research using larger samples sizes is needed. Third, the sample was largely non-Hispanic white participants, which limits the ability to generalize these findings to other racial groups. Likewise, the strict inclusion criteria limits the ability to generalize these findings beyond healthy individuals, again necessitating replication in more representative samples. Fourth, we did not include an objective measure of affect. Given evidence for heighted physiological emotional reactivity during sleep deprivation (Franzen et al., 2008), future research would benefit from incorporating a multimethod assessment of affect. Fifth, we did not sample a full baseline day of positive and negative affect. Thus, though we included affect reported in the baseline battery as a covariate in the models, we do not have data to compare changes in affect observed during sleep loss to diurnal changes in affect that could be observed across an entire baseline day. Sixth, we used a modified version of the PANAS where Likert-type scales were replaced by visual analog scales, which limits comparability of our results with prior work. Despite these limitations, these findings offer evidence that chronotype may represent a vulnerability to the affective consequences of sleep loss and circadian misalignment, such that later chronotypes may be more susceptible to experiencing negative changes in affect during sleep loss than earlier chronotypes.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304231188204 – Supplemental material for Chronotype and Affective Response to Sleep Restriction and Subsequent Sleep Deprivation
Supplemental material, sj-docx-1-jbr-10.1177_07487304231188204 for Chronotype and Affective Response to Sleep Restriction and Subsequent Sleep Deprivation by Rebecca C. Cox, Hannah K. Ritchie, Oliver A. Knauer, Molly K. Guerin, Ellen R. Stothard and Kenneth P. Wright, Jr. in Journal of Biological Rhythms
Footnotes
Acknowledgements
The authors thank the research participants for their contribution, the staff and students of the Sleep and Chronobiology Laboratory for their assistance, and JM Ronda for his assistance with affect assessment outcomes. This work was supported by the CurAegis Technologies Inc. (formerly Torvec, Inc.), the National Heart, Lung, and Blood Institute (T32 HL149646; R01 HL109706), NIH/NCATS Colorado CTSA Grant UL1TR002535, and the University of Colorado Boulder Undergraduate Research Opportunities Grant. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Conflict Of Interest Statement
K.P.W. received consulting fees from or served as a paid member of scientific advisory boards for the Sleep Disorders Research Advisory Board-National Heart, Lung and Blood Institute, CurAegis Technologies, Circadian Therapeutics, LTD, Circadian Biotherapies, Inc., and the U.S. Army Medical Research and Materiel Command–Walter Reed Army Institute of Research during the conduct of this research and research support/donated materials from DuPont Nutrition & Biosciences, Grain Processing Corporation, and Friesland Campina Innovation Center, unrelated to this research.
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
