Abstract
Millions of people worldwide are working at times that overlap with the normal time for sleep. Sleep problems related to the work schedule may mediate the well-established relationship between shift work and increased risk for disease, occupational errors and accidents. Yet, our understanding of causality and the underlying mechanisms that explain this relationship is limited. We aimed to assess the consequences of night-shift work for sleep and to examine whether night-shift work-induced sleep disturbances may yield electrophysiological markers of impaired maintenance of the waking brain state. An experimental model developed in rats simulated a 4-day protocol of night-work in humans. Two groups of rats underwent 8-h sessions of enforced ambulation, either at the circadian time when the animal was physiologically primed for wakefulness (active-workers, mimicking day-shift) or for sleep (rest-workers, mimicking night-shift). The 4-day rest-work schedule induced a pronounced redistribution of sleep to the endogenous active phase. Rest-work also led to higher electroencephalogram (EEG) slow-wave (1-4 Hz) energy in quiet wakefulness during work-sessions, suggesting a degraded waking state. After the daily work-sessions, being in their endogenous active phase, rest-workers slept less and had higher gamma (80-90 Hz) activity during wake than active-workers. Finally, rest-work induced an enduring shift in the main sleep period and attenuated the accumulation of slow-wave energy during NREM sleep. A comparison of recovery data from 12:12 LD and constant dark conditions suggests that reduced time in NREM sleep throughout the recorded 7-day recovery phase induced by rest-work may be modulated by circadian factors. Our data in rats show that enforced night-work-like activity during the normal resting phase has pronounced acute and persistent effects on sleep and waking behavior. The study also underscores the potential importance of animal models for future studies on the health consequences of night-shift work and the mechanisms underlying increased risk for diseases.
Millions of people worldwide work at times that overlap with their circadian phase when sleep propensity is high, and are thus obligated to sleep when the propensity is low. As a consequence, shift workers report more sleep problems than the general public (Ursin et al., 2009), and night workers report more insomnia and non-restorative sleep as compared with day and evening workers (Torsvall et al., 1989b; Pilcher et al., 2000). These sleep disturbances have consequences for functioning, as night-shift workers often complain about sleepiness during night-shifts as well as during daytime (Akerstedt, 2003). A study on Norwegian night-working nurses reported a prevalence of insomnia and excessive daytime sleepiness as high as 44% (Flo et al., 2012). In general, shift work is associated with occupational errors and accidents (Van Dongen et al., 2016) and, in the long run, night-shift workers also have increased risk of developing a wide range of adverse health outcomes, such as cancer, cardiovascular disease, diabetes, and gastrointestinal disorders (Knutsson, 2003). In many cases, the relationship between shift work and health issues may be mediated by shift work-related sleep problems (Knutsson, 2003).
Numerous studies in both humans and laboratory animals have shown that prolonged wakefulness induces deficits in brain function, which affect alertness, performance, mood, and memory (Banks and Dinges, 2007; Havekes et al., 2015). Our understanding of the causal mechanisms underlying night-shift-related sleep disturbances is limited. Rodent models established to study the effects on night-shift work have already provided important contributions to the understanding of its metabolic consequences (Opperhuizen et al., 2015). However, the sleep-wake-specific impact of night-shift work has not yet been addressed. We therefore set out to develop an animal model for research on the consequences of night-shift work for the regulation of sleep and wakefulness. Despite the fact that most night work in Europe and Norway seldom last for more than 3-4 consecutive nights, the great majority of studies conducted on mimicking night-shift work has examined night work periods of at least 5-6 days (Opperhuizen et al., 2015). We aimed to simulate one typical cycle of night-shift work in humans by subjecting rats to forced ambulation over a 4-day work schedule in LD conditions. Animals were studied in 2 conditions: active-workers (mimicking human day-shift workers) and rest-workers (mimicking night-shift workers). Both groups were subjected to four 8-h sessions of enforced ambulation in an activity wheel: active-workers at the circadian time when the animal is physiologically timed for wakefulness, and rest-workers at the time of day when the animal is primed for sleep and thus was forced to recover being in an endogenous active phase. The first specific aim of the present study was to determine whether rest-workers and active-workers demonstrate distinct changes in sleep-wake patterns and EEG markers of sleep drive; that is, 1) acute changes during the 4-day work protocol, and 2) persistent changes across a 7-day recovery phase after the end of the work protocol. We also assessed the effect of circadian factors on the sleep recovery process by measuring recovery in both LD and DD conditions. We further aimed to determine how the different work protocols affect waking behavior and, particularly, whether repeated rest-work leads to waking impairments and sleep-like EEG features during the waking state. Recently, we have shown that waking-state EEGs show distinct spectral properties during the waking sub-states of active wakefulness (associated with high muscle tone) and quiet wakefulness (devoid of locomotion, associated with low muscle tone). The accumulation of sleep drive during protracted wakefulness can be detected by quantitative EEG during quiet wakefulness (Gronli et al., 2016).
Materials and Methods
Ethical Approval
The experimental protocol was approved and registered by the Norwegian Animal Research Authority (Permit Number: 2012463), and performed according to Norwegian laws and regulations controlling experiments in live animals and the European Convention for the protection of Vertebrate Animals used for Experimental and other Scientific Purposes, March 18 1986.
Animals and Housing
Male rats (Wistar strain, NTac:WH, Taconic, Denmark) weighing approximately 300 g at arrival, were acclimatized to the laboratory conditions before being group-housed in individually ventilated type IV cages (480 × 375 × 210 mm, IVC system, Tecniplast, Italy). After surgery, animals were housed individually in type III cages (425 × 266 ×185 mm). The ventilation in the IVC-cages was 75 air changes/h, with temperature maintained at 23 ± 1 °C, and humidity at 40% ± 1% throughout the experiment. Food (rat and mouse no.1, Special Diets Services, Witham, Essex, England) and water were provided ad libitum. The animals were kept under 12:12 LD conditions with gradual transitions between the light and dark phases, from 0600 to 0700 for lights-on and from 1800 to 1900 for lights-off, respectively. Accordingly, 0600 was labelled as lights on (zeitgeber time, ZT0) and 1800 was labelled as lights off (ZT12).
Experimental Protocol
To simulate shift work in humans, rats were exposed to forced ambulation, 8 h per day for 4 days, in motorized running wheels (Rat Running Wheel, TSE, Germany). The wheels, with a diameter of 24 cm, were set to 3 RPM (a total of 1440 revolutions per session; yielding 1.086 km of linear distance) and were equipped with feeders to provide food and water ad libitum. The 8-h sessions were centered either during the rats’ normal rest phase (ZT2 - ZT10) or during their active phase (ZT14 – ZT22). In the 16-h intervals between forced ambulation sessions, all animals remained undisturbed in their home cages. All wheels, feeders and water bottles were cleaned with a 5% ethanol solution after each workday.
For assessment of sleep-wake patterns, EEG and electromyogram (EMG) were monitored continuously throughout a 24-h, undisturbed, baseline period followed by the 4-day work protocol (workdays W1 to W4) and 7-day of undisturbed recovery (R1 to R7). See Figure 1 for an overview of the protocol. Animals were randomly assigned to either rest-work (n = 15) or active-work (n = 12). To examine if changes in the sleep-wake distribution were under the influence of an endogenous circadian process and not a passive response to light and dark (Tobler et al., 1994; Deboer, 2009), each group was subdivided into a 12:12 LD conditions (n = 6 and n = 7, respectively), or constant darkness DD (rest-workers DD: n = 9 and active-workers DD: n = 5, respectively) in the recovery phase.
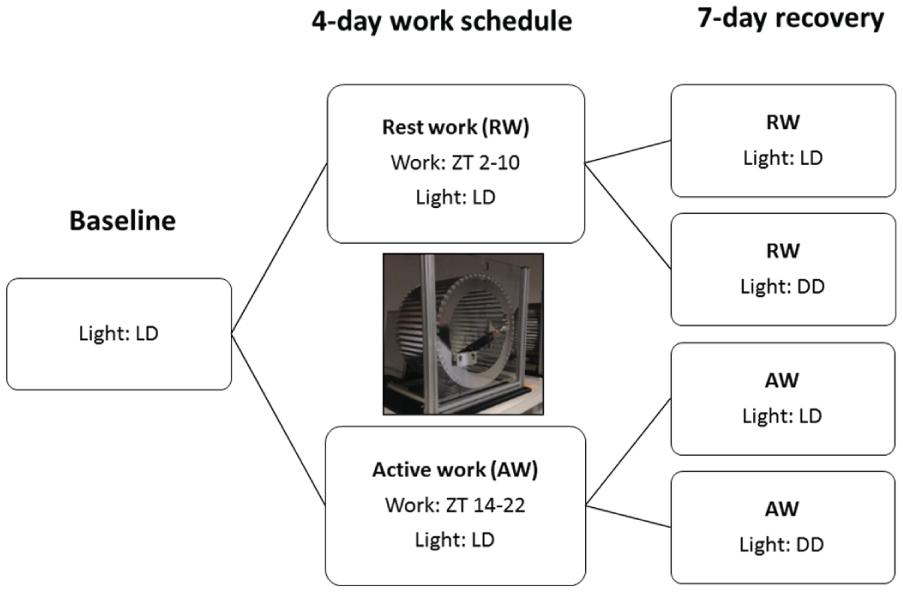
An overview of the design for the different experimental conditions.
Surgery
Animals underwent surgery for the implantation of transmitters to record EEG and electromyogram (EMG). Two types of transmitters were used, 4ET and F40-EET (Physiotel, Data Sciences International; St. Paul, MN). Animals were given an antibiotic in drinking water up to 3 days before surgery (trimethoprim, 0.16 mg/ml; sulfamethoxazole, 0.8 mg/ml; Bactrim, Roche; Basel, Switzerland). Animals were then anaesthetized by subcutaneous (s.c.) injection of a mixture of fentanyl 0.227 mg/kg, fluanizone 8.8 mg/kg and midazolam 2.5 mg/kg (Hypnorm, Janssen Pharmaceuticals, Beerse, Belgium; Dormicum, Roche; Midazolam, Actavis, Teva Pharmaceuticals, Peta Tikva, Irsael) for implantation of frontal-parietal EEG electrodes (bregma coordinates: AP = 2.0 mm, ML = −2.0 mm; lambda coordinates: AP = 2.0 mm, ML = 2.0 mm). An additional EEG derivation for the 4ET transmitter was placed frontal-frontal (bregma coordinates: AP = 2.0 mm, ML = 2.0 mm). EMG electrodes were implanted bilaterally in the neck muscle. The sterile transmitters were implanted in an s.c. pocket in the dorsomedial lumbar region (4ET transmitters) or in the neck region (F40-EET transmitters).
Immediately after surgery, Ringer’s acetate solution was given to compensate for fluid loss during surgery (5 ml i.p., Baxter). For 2 days after surgery, the rats received antibiotics in their drinking water (trimethoprim, 0.16 mg/ml; sulfamethoxazole, 0.8 mg/ml; Bactrim, Roche). For 3 days following surgery, an analgesic was given twice daily (buprenorphine; 0.30 mg/ml, s.c., Temgesic, Reckit & Benckiser; Slough, UK), and anti-inflammatory treatment was given once a day (meloxicam; 5 mg/ml, s.c., Metacam, Boehringer Ingelheim, Germany). Daily care was provided throughout the post-operative period and at least 2 weeks were allowed for recovery (Moscardo and Rostello, 2010).
Collection, Processing and Analysis of EEG and EMG Data
The wireless recording device (input voltage: –1.25 to +1.25 mV) acquired EEG and EMG signals at a sampling rate of 250 Hz (Dataquest A.R.T, version 4.1, Data Sciences International). Telemetry signals were collected through receivers (RPC-2/RPC-3, Data Sciences International) placed directly beneath the animals’ home cage or next to the rotating wheel during forced ambulation. The different amplification of the signals (4ET: 240-fold; F40-EET: 200-fold) was corrected for by the use of calibration in the software Dataquest ART. As a result, the built-in bandwidth of 4ET is 1-100 Hz and F40-EET is 1-50 Hz. Because a 2-pole filter in F40-EET on the low-pass band displayed a significant impact on the low gamma analyses (30-50 Hz), only data from the 4ET devices (rest-workers: n = 11 and active-workers: n = 7) were analyzed for low and high gamma activity.
Sleep stages were scored offline with Neuroscore software (version 2.0.1, Data Sciences International). Wakefulness, NREM sleep and REM sleep were manually scored in 10-s epochs. For the purpose of manual scoring based on criteria from Neckelmann and Ursin (1993), the EEG signals were filtered with high-pass at 0.5 Hz and low-pass at 35 Hz. EMG signals were filtered with high-pass at 5 Hz. The latency to stable sleep was defined as 1 min of consecutive sleep. The latency to REM sleep was defined as 30 sec of consecutive REM sleep. Epochs of wakefulness were subdivided into active, intermediate and quiet wakefulness using EMG peak-to-peak amplitude values of all wakefulness epochs across the interval analyzed. Active wakefulness was defined as those epochs of wakefulness in which the EMG peak-to-peak amplitude was
In addition to the time spent in different vigilance states, we also analyzed the spectral characteristics of the EEG during these states (Neuroscore, version 2.0.1). Offline Fast Fourier Transform (FFT) analysis was performed on unfiltered EEG signals averaged across consecutive 10-sec epochs (average of five 2-sec intervals) with a Hamming window and 50% overlap. Artefacts were removed in 2 steps: 1) visual inspection of EEG signals, where all epochs containing electrical artefacts were excluded; and 2) an automated algorithm that detected epochs with EEG power spectral values in any 1-Hz band from 1-20 Hz that exceeded the mean value for that band across the entire recording (across all vigilant states) by at least 8 standard deviations (Gronli et al., 2016). The artefacts eliminated from the analysis ranged from 0.07 to 2.9%, with a mean of 0.76% across all files. To account for individual differences in EEG signal strength, EEG spectral power tracked across states was normalized to the average power values of these sleep states during each animal’s 24 h baseline recording or the corresponding circadian time period during baseline. We calculated EEG slow-wave activity (SWA, the average spectral power in the 1-4 Hz range) in NREM sleep as a measure of sleep drive. In addition, the accumulation of SWA over time (slow-wave energy, SWE) was calculated by multiplying the number of epochs spent in a given state per h and the average amount of EEG spectral power in the 1-4 Hz range across all epochs of that state in that h (Gerashchenko et al., 2008). Cortical oscillations in the higher frequencies, low gamma (30-50 Hz) and high gamma (80-90 Hz), were analyzed as indicators of changes in the waking brain fast network oscillations (Sohal, 2012; Kann, 2016).
Statistical Analyses
ANOVA for repeated measures was used to gauge experimental effects across time in the 2 groups of rats. Sleep parameters were tested for group effects separately within each of the different phases of the experiment (baseline, work and recovery). For the baseline phase, the analysis included the between-subjects factor ‘work-time’ (active-work vs. rest-work) to verify the absence of differences between the groups before they experienced different work schedules. For the work phase of the experiment, the analysis included the between-subjects factor ‘work-time’ (active-work vs. rest-work) and within-subjects factors ‘workday’ (day W1 to W4) and ‘time interval’ (successive 2-h intervals within the days). For the recovery phase of the experiment, the analysis included the between-subjects factors ‘work-time’ (active-work vs. rest-work) and ‘light condition’ (LD vs. DD) and the within-subjects factors ‘recovery day’ (day R1 to R7) and ‘time interval’ (successive 2-h intervals within the days). All significant main and interaction effects were followed up with Fisher LSD post hoc tests. Significance level was set at α ≤ 0.05. Different degrees of freedom are due to exclusion of some animals because of missing values (if a state did not occur in a particular interval) or poor EEG signals. The software package MATLAB (The MathWorks, Natick, MA) was used for analysis and modelling, and Statistica (version 12.0, StatSoft, Inc., Dell. Round Rock, TX) was used for statistics.
Results
Waking Behavior during Work Hours
During baseline, all rats displayed a typical daily rhythmic, polyphasic, organization of sleep and wakefulness, with wakefulness predominating during the dark phase (66% during D and 25% during L), and sleep during the light phase (29% NREM sleep and 5% REM sleep during D, and 58% NREM sleep and 17% REM sleep during L). At baseline, the groups did not differ in time spent in wakefulness, NREM sleep or REM sleep (F1,24 < 0.3, p > 0.60); sleep consolidation measures (number and duration of wakefulness and sleep episodes: F1,24 < 0.3, p > 0.59); or EEG power spectra (F1,15 < 0.1, p > 0.84).
The forced wheel rotation effectively kept the rats awake for almost the full 8-h work-sessions, except for occasional micro-sleep episodes. During work, rest-workers spent more time in sleep than active-workers (F1,24 = 10.7, p = 0.004). The average min ± SEM in sleep during work across W1 to W4 in rest-workers were 0.8 ± 0.2, 0.7 ± 0.4, 0.3 ± 0.2 and 1.0 ± 0.3 (average, 0.7 ± 0.1) and, in active-workers, 0.0 ± 0.3, 0.0 ± 0.5, 0.1 ± 0.2 and 0.0 ± 0.4 (average, 0.0 ± 0.2), respectively. Total sleep time during work varied from 0.0 to 4.5 min.
Rest-work was also characterized by a slowing of the waking EEG, manifested by an accelerated accumulation of SWE in epochs of quiet wakefulness (work-time, F1,24 > 5.1, p < 0.038), but not in active or intermediate wakefulness (work-time, F1,24 < 1.3, p > 0.27; data not shown). In rest-workers, SWE in quiet wakefulness progressively increased across the work-session (ZT2-10), to a greater extent than in active-workers (ZT14-22). The change was global, as it was seen in both frontal (Fig. 2) and parietal (data not shown) EEGs. The cumulative SWE in quiet wakefulness relative to baseline after the 4-day work cycle was almost twice as high in rest-workers (820%) as in active-workers (455%). This difference was not a function of time spent in quiet wakefulness, since quiet wakefulness during work represented either equal time (days W1, W2) or less time (days W3, W4) in rest-workers relative to active-workers (Fig. 3).
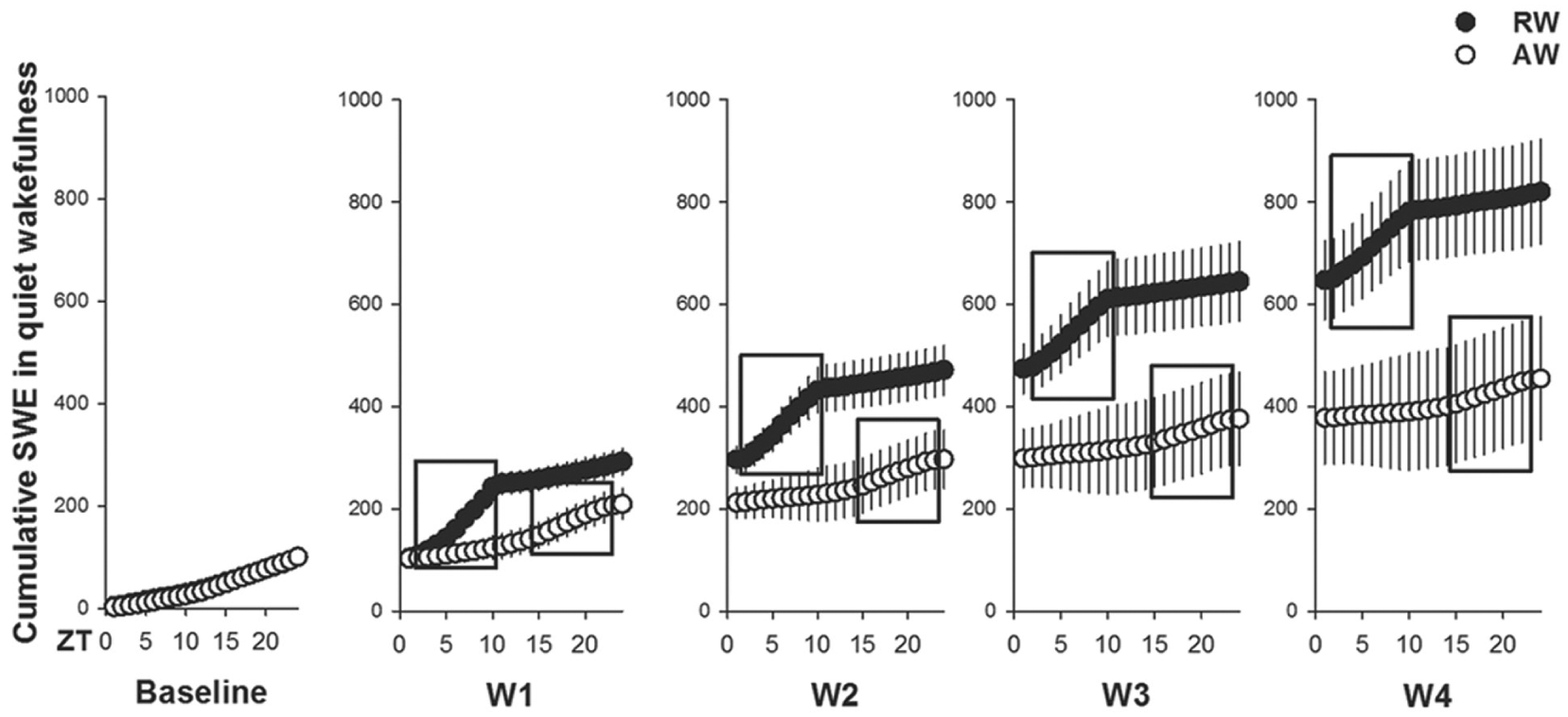
The cumulative burden of rest-work on wake function. Cumulative slow-wave energy (SWE; total integrated power in the SWA (1-4 Hz) over all 10-sec epochs of quiet wakefulness) expressed per 1 h during baseline and 4-day work schedule; workday 1 (W1) to W4 in rest-workers (RW, n = 11, work-hours: ZT2-10) and active-workers (AW, n = 7, work-hours: ZT14-22). Work sessions are shown in black boxes. Data are shown as mean values ± SEM.
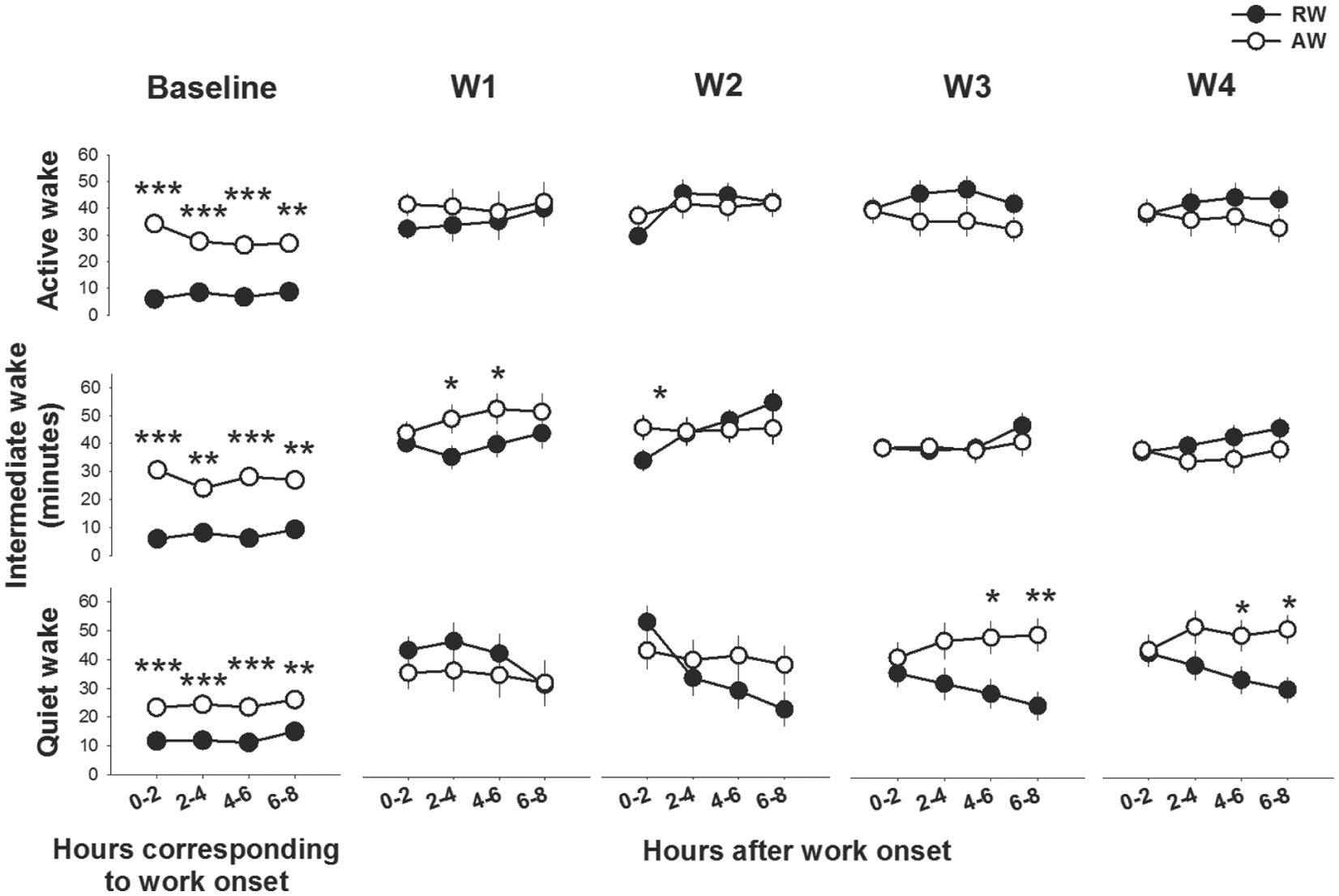
Waking behavior during 8 h of work and during the corresponding circadian time in baseline. Both rest-workers (RW, n = 15) and active-workers (AW, n = 12) increased time spent in active wakefulness (RW: F = 13.0, p < 0.001; AW: F = 2.9, p = 0.003), intermediate wakefulness (RW: F = 30.1, p > 0.001; AW: F = 15.9, p < 0.001) and quiet wakefulness (RW, F = 13.5, p < 0.001; AW: F = 8.1, p < 0.001) across the 4 consecutive workdays in the shift work schedule; workday 1 (W1) to W4 as compared with corresponding h in baseline condition. Asterisks indicate significant differences between the groups; *p < 0.05, **p < 0.01. Data are shown as mean values ± SEM.
The rats’ waking behavior during work-sessions differed across the workdays (workday X work-time, F3,75 = 5.5, p = 0.0018). Both rest-workers and active-workers spent more time in all 3 sub-states of wakefulness during work-sessions as compared with their waking behavior in the corresponding circadian hours (see Fig. 3). At W1, rest-workers spent less time in intermediate wakefulness than did active-workers. At W3 and W4, rest-workers spent less time in quiet wakefulness than did active-workers, specifically in the second half of the 8-h work-session. Time spent in active wakefulness during work was not different.
Rest-workers Spent More Time Awake and Displayed Higher Gamma Activity Immediately following Work Sessions than Active-workers
Upon returning to the home cage after work sessions, the waking behavior (all 3 sub-states of wakefulness) differed markedly between rest-workers and active-workers (work-time X time interval, F6,150 > 2.8, p < 0.035, data from W1 and W4 are shown in Fig. 4). Consistently, across the 16 h between work h, rest-workers spent less time in all 3 sub-states of wakefulness as compared with their corresponding circadian waking behavior. The time course of waking behavior showed that active-workers were more awake (all sub-states) than rest-workers the first 2 h after work (p < 0.05, all days). This difference may reflect the endogenous circadian phase of the 2 groups, since active-workers returned to their home cage in their active phase (ZT22) and rest-workers in their rest phase (ZT10). Across the following 12 h (D in rest-workers and L in active-workers), rest-workers spent more time awake (all sub-states) as compared with active-workers. During the 2 h before the onset of the next work session, rest-workers were less awake than active-workers. This pattern was rather consistent throughout the 4-day work schedule. The total amount of quiet wakefulness after W1 was higher in rest-workers relative to active-workers, but lower at W4 (see insets in Fig. 4). Profiles of the normalized high (80-90 Hz) and low (30-50 Hz) gamma activity to the circadian baseline h across the interval between work-sessions showed an elevated higher gamma activity in all sub-states of wakefulness in rest-workers during the 2-8 h following work, whereas the active-workers displayed a lower higher gamma activity than baseline condition and rest-workers (work-time X time interval, F6,135 > 3.0, p < 0.075: Fig. 5). Low gamma activity differed significantly between the groups in intermediate wakefulness (work-time X time interval, F6,135 = 2.2, p = 0.039).
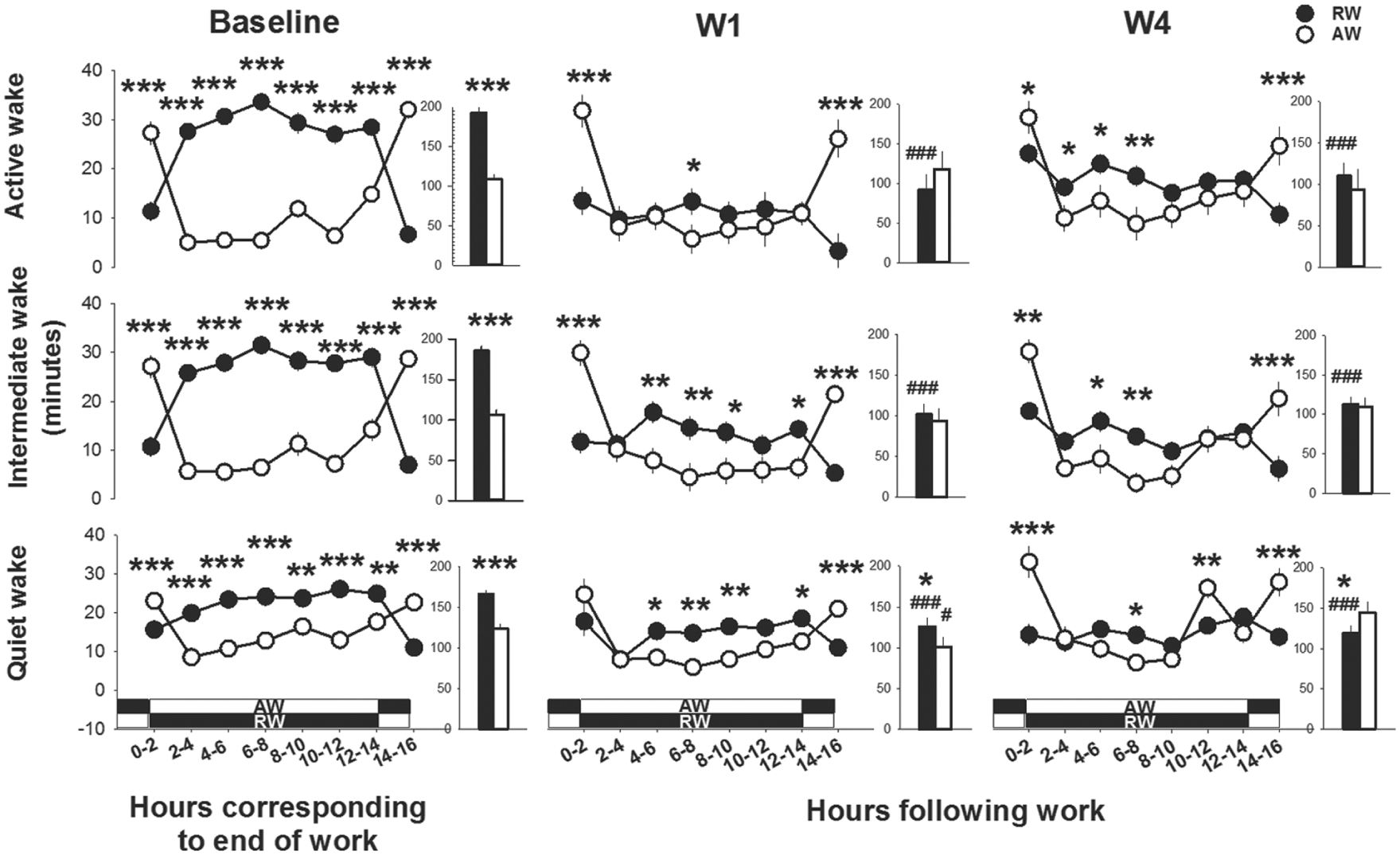
Time course of waking behavior directly after work-hours; workday 1 (W1) and W4 and during the corresponding circadian time in baseline. Asterisks indicate significant differences between rest-workers (RW, n = 15) and active-workers (AW, n = 12); *p < 0.05, **p < 0.01, ***p < 0.001. The white-black bands represent the light conditions in RW and AW following work. Dark represents the dark phase and white indicates the light phase. Data are shown as mean values of 2 h ± SEM. The insets show the time spent in the different wake states across the total 16 h. Hashtags denote significant differences as compared with the baseline: #p < 0.05 and ###p < 0.001.
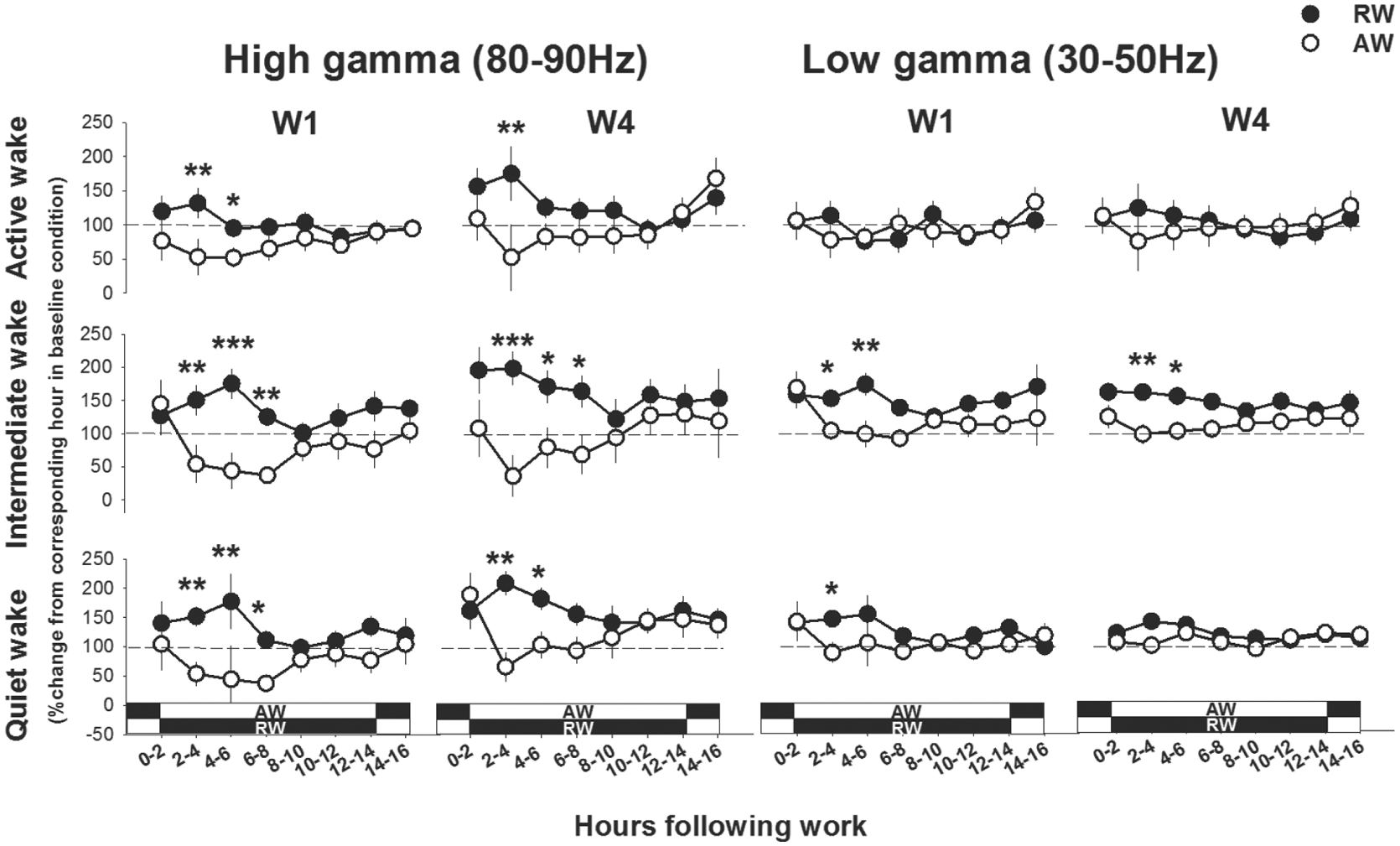
Time course of high and low gamma activity during wakefulness directly after work-hours; workday 1 (W1) and W4. Asterisks indicate significant differences between rest-workers (RW, n = 11) and active-workers (AW, n = 7), *p < 0.05, **p < 0.01, ***p < 0.001. Data are shown as mean values of 2 h ± SEM normalized to the corresponding 2-h period during baseline. The dotted line indicates baseline activity (100%). The white-black bands represent the light conditions in RW and AW following work. Dark represents the dark phase and white indicates the light phase.
Short-term Changes in Sleep during the Work Phase
The raw data and statistics from 24-h LD, 12-h L and 12-h D on time spent in wakefulness and sleep during baseline and the 4-day work schedule are shown in Supplementary Table S1. Forced ambulation during the normally rest (L) phase in rest-workers caused a redistribution of sleep to the normally active (D) phase (see Suppl. Fig. S1 and Suppl. Table S1). In D, the time in sleep (NREM sleep and REM sleep) increased and time in wakefulness (all sub-states) decreased in rest-workers relative to both their own baseline and to that of the active-workers. In L, time awake was elevated in rest-workers relative to both their own baseline and to the active-workers (p < 0.001, all stages; all 4 days). Active-workers showed an increased time in wakefulness with a concomitant decrease in NREM sleep and REM sleep during the active phase as compared with baseline data (p < 0.001, all stages; all 4 days). As a consequence, these animals spent more time in NREM sleep during their rest (L) phase at W2 and W3 as compared with the baseline condition (p = 0.03 and p = 0.01, respectively). Time spent in wakefulness and REM sleep during L was not significantly different from baseline in active-workers.
The latency to stable sleep, i.e., 1 min of uninterrupted sleep immediately after work-sessions, was shorter in rest-workers as compared with active-workers (work-time, F1,63 = 10.7, p = 0.004; work-time X workday: F3,63 = 10.4, p = 0.00001, Fig. 6A). After W1, rest-workers showed stable sleep after 28.9 ± 5.0 min as compared with 54.7 ± 6.3 min in active-workers. The sleep latency in rest-workers was rather con-stant throughout the work schedule, and significantly shorter than that in active-workers after W3 and W4. Active-workers displayed shorter sleep latency at W2 and W3 compared with W1, but returned to the value of W1 at W4. Latency to REM sleep was not different between the groups of workers until W4 when rest-workers showed shorter latency to REM sleep as compared with the active-workers (Fig. 6A; work-time X workday, F3,63 = 2.7, p = 0.049).
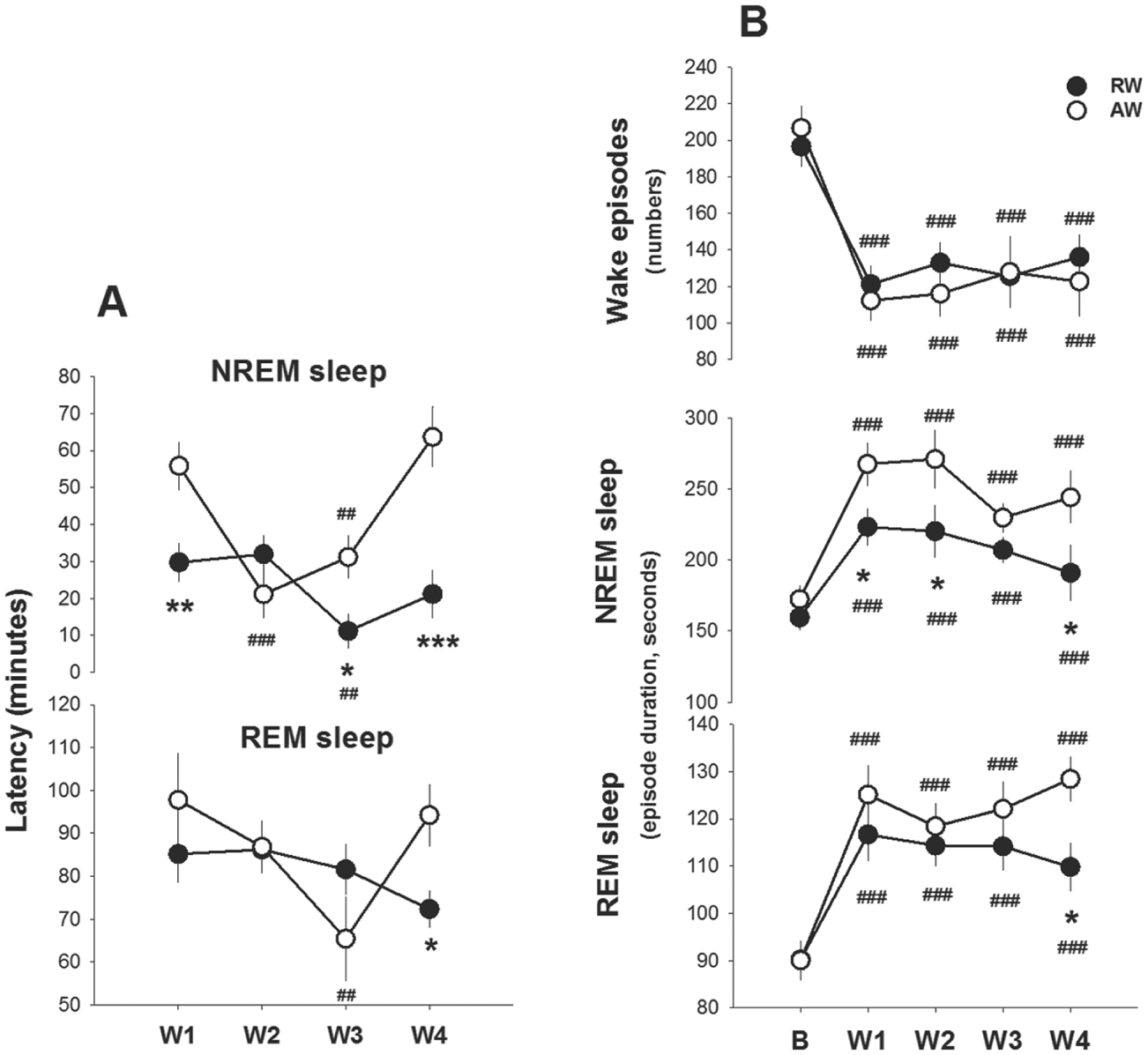
Work-induced changes in latency to sleep and sleep consolidation measures. A) Latency to 1 min of consecutive sleep and 30 sec of consecutive REM sleep following work-hours. B) Number of wake episodes and duration of NREM- and REM-sleep episodes between work-hours. Data are shown as mean values ± SEM after the 4 workdays (W1 to W4). Asterisks indicate significant differences between rest-workers (RW, n = 15) and active-workers (AW, n = 12); *p < 0.05, ** p < 0.01, *** p < 0.001, and hashtags denote significant differences as compared with W1: #p<0.05, ##p < 0.01 and ###p < 0.001.
Work increased the average episode durations of NREM sleep and REM sleep and reduced the number of wakefulness episodes in both groups as compared with the baseline (workday, F4,76 > 13.9, p < 0.000, Fig. 6B). Yet, the increase in NREM sleep episodes was less pronounced in rest-workers (work-time X workday, F4,76 > 2.6, p < 0.043). Compared with active-workers, the average duration of NREM sleep was shorter in rest-workers throughout the work schedule, and the duration of REM-sleep episodes was shorter after W4. The number of wakefulness episodes did not significantly differ between the groups.
Sleep timing and spectral data from the groups were aligned relative to the end of work-hours to determine whether sleep after equivalent work-sessions would vary as a function of circadian timing. Time spent asleep differed between rest-workers and active-workers after work (work-time X time interval, F6,133 > 20.8, p < 0.0001, Fig. 7). Consistently, rest-workers spent more time in sleep across the 16-h between work hours as compared with their sleep in the corresponding circadian h during baseline. The time course of sleep showed that rest-workers spent more time in NREM sleep (p < 0.001, all days) and REM sleep (p < 0.05, all days) than active-workers in the first 2 h after work. Thereafter (ZT12 in rest-workers and ZT0 in active-workers), rest-workers spent less time in NREM sleep as compared with active-workers. Rest-workers slept more than active-workers during the 2 h immediately before the start of another 8-h work-session. This pattern was fairly consistent throughout the 4-day work schedule. Time spent in REM sleep after workdays was less affected than NREM sleep. Across the 16-h opportunity to sleep after work, the total amount of NREM sleep was significantly less in rest-workers relative to active-workers after W4 (see insets in Fig. 7). High, though not equivalent, values of SWA in NREM sleep were reached for both groups of rats immediately after a work-session. Rest-workers showed a significantly lower SWA during the first 2-h after work on all workdays (work-time X time interval, F3,135 > 2.9; p < 0.008, Fig. 8). SWA rapidly decreased with time in NREM sleep in both groups. The cumulative SWA or SWE during NREM sleep across the 4-day work protocol did not differ between the groups or between frontal (Suppl. Fig. S2) and parietal EEG (data not shown) (work-time, F1,24 < 0.1; p > 0.71, both EEG derivations). The SWE in NREM sleep progressively increased after work-sessions. The total SWE in NREM sleep after baseline and the 4-day work schedule was not different between rest-workers (487%) and active-workers (491%).
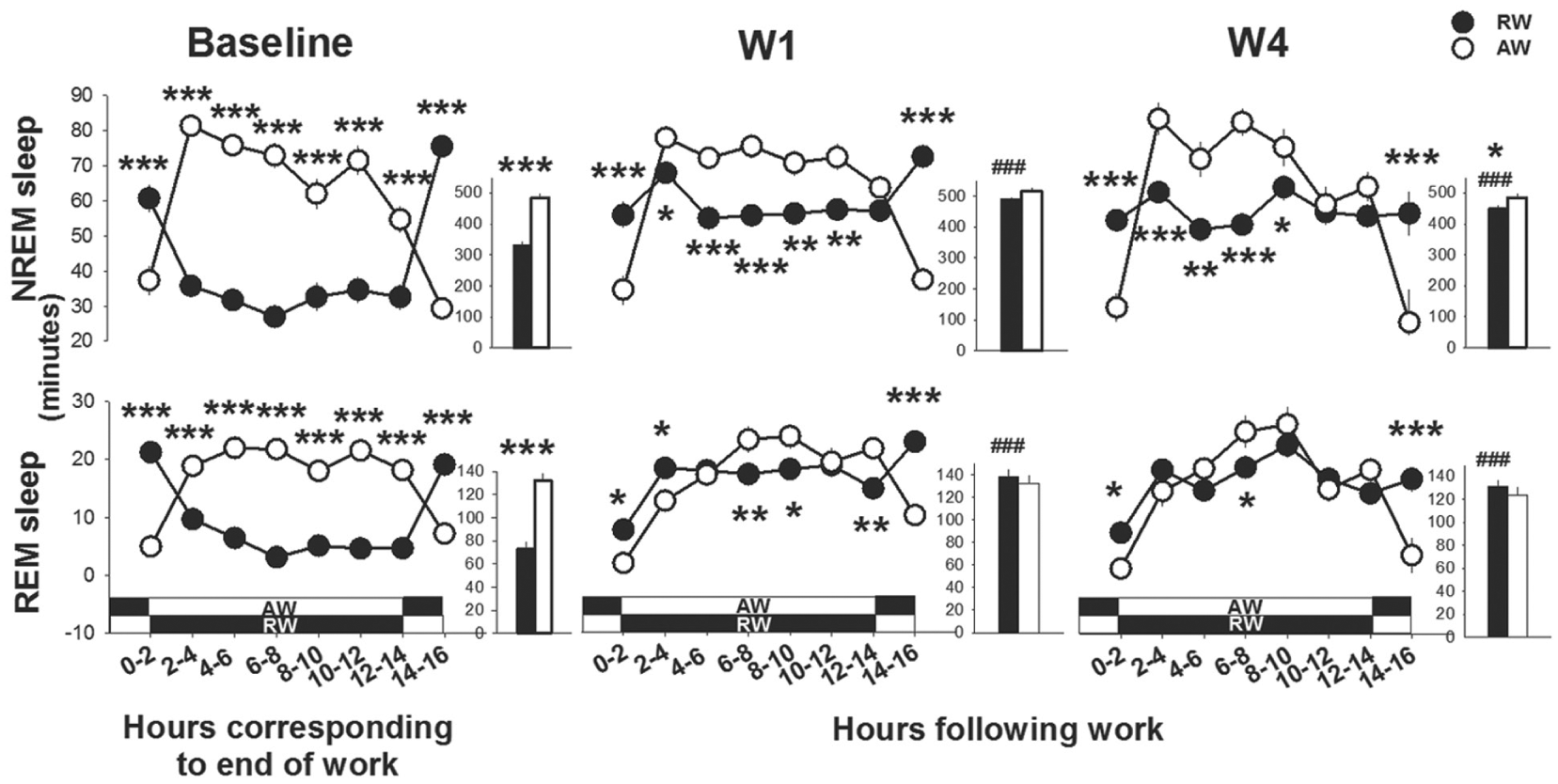
Time course of sleep directly following work-hours; workday 1 (W1) and W4 and during the corresponding circadian time in baseline. Asterisks indicate significant differences between rest-workers (RW, n = 15) and active-workers (AW, n = 12). *p < 0.05, **p < 0.01, ***p < 0.001. Data are shown as mean values of 2 h ± SEM. The insets show the time spent in the different sleep states across the total 16 h. Hashtags denote significant differences compared with the baseline: ###p < 0.001. The white-black bands represent the light conditions in RW and AW following work. Dark represents the dark phase and white indicates the light phase.
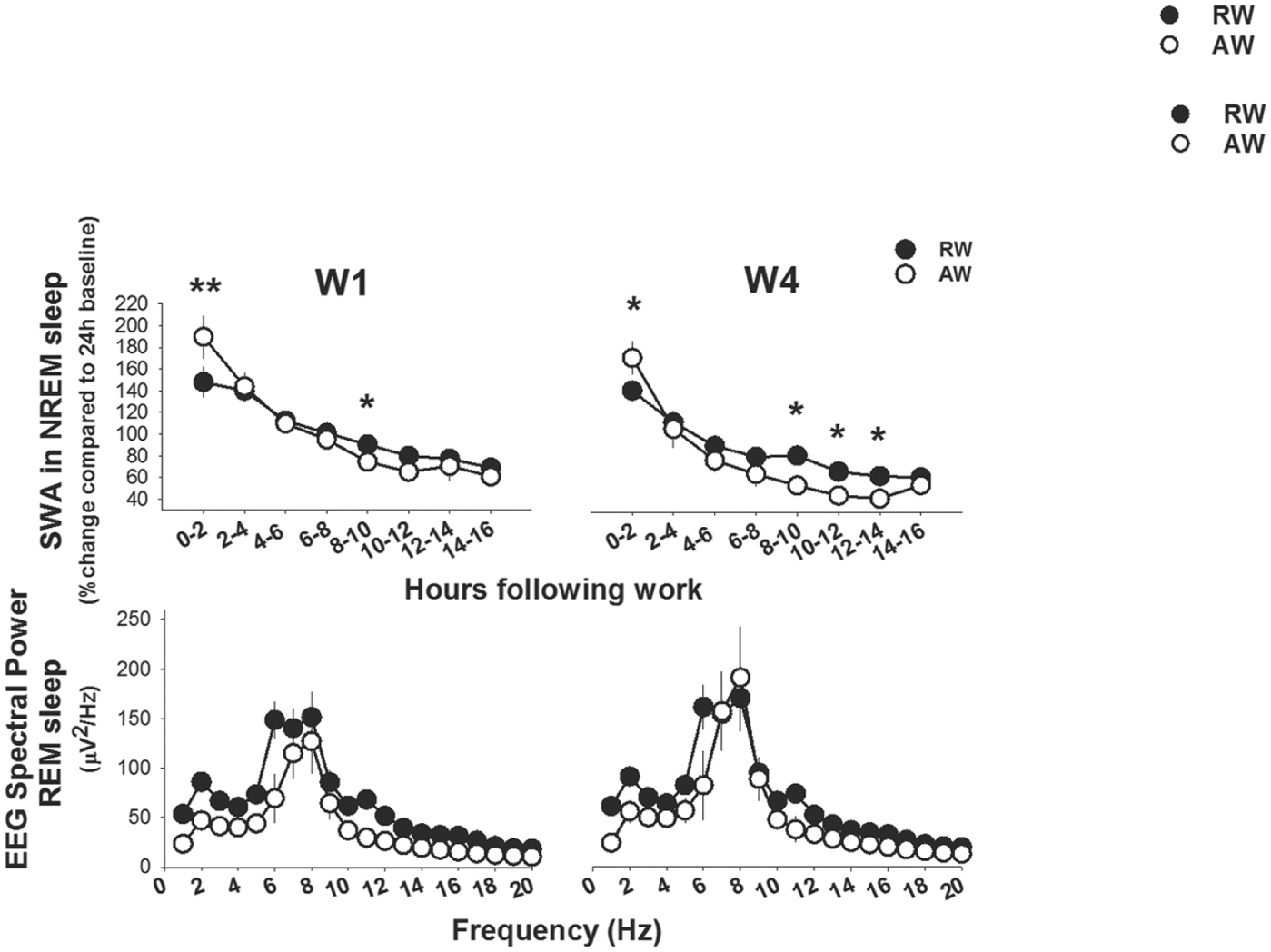
Frontal slow-wave activity (SWA; 1-4 Hz) in NREM sleep and parietal EEG power profile in the 1-20 Hz range in REM sleep directly after work-hours. Asterisks indicate significant differences between rest-workers (RW: frontal EEG, n = 11 and parietal EEG, n = 15) and active-workers (AW: frontal EEG n = 7 and parietal EEG, n = 12). *p < 0.05, **p < 0.01, ***p < 0.001. Data are shown as mean values of 2 h ± SEM.
Despite a redistribution of wakefulness and sleep in rest-workers during the 4-day work schedule, both groups showed the effects of work on the overall time spent sleeping per day (24 h LD) (workday, F4,100 > 23.4, p < 0.001, in all cases). Only quiet wakefulness displayed an interaction effect with ‘work-time’ (work-time X workday, F4,100 = 3.4, p = 0.012). The 4-day work schedule increased the time spent awake (12% in active-workers vs. 11% in rest-workers), with concurrent reductions in the time spent in NREM sleep (–10% vs. –9%) and REM sleep (–2% vs. –2%), compared with their baseline readings.
Rest-work Leads to Long-term Redistribution of Wakefulness and Sleep
Raw data and statistics from 24-h LD, 12-h D and 12-h L for time spent in wakefulness and sleep during baseline and recovery days in LD and DD conditions are shown in Supplementary Table S2.
The redistributed pattern of wakefulness and sleep in the rest-worker group persisted over several days during the unrestricted 7-day recovery period in LD following the work schedule. In the normal rest phase (L), the increased time spent in active and intermediate wakefulness persisted to R3 and the increased time spent in quiet wakefulness to R4, relative to baseline (Fig. 9). Parallel effects were found in DD, except for quiet wakefulness, which was not different from baseline during the first recovery days. Active-workers did not exhibit any significant changes in wakefulness in their rest (L) phase; however, during DD, the time in active wakefulness increased until R4, as compared with the baseline.
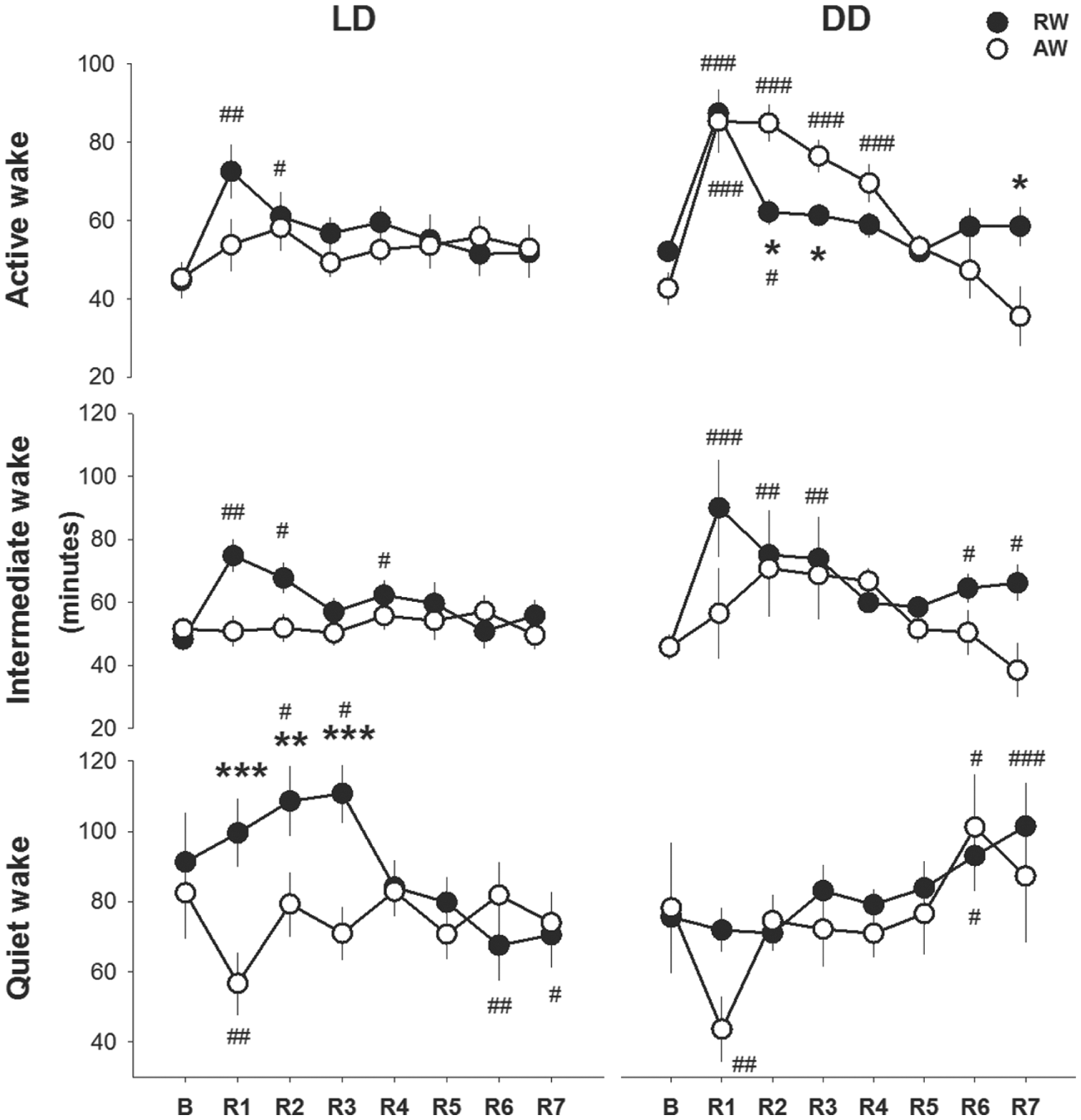
Wake profiles during the light (rest) phase in the unrestricted recovery phase after the 4-day work schedule; recovery days 1 (R1) to R7 in LD and DD conditions. Asterisks indicate significant differences between rest-workers (RW) and active-workers (AW), 12:12 LD conditions (RW n = 6 and AW n = 7) and constant darkness DD (RW n = 9 and AW n = 5). *p < 0.05, **p < 0.01, ***p < 0.001. Hashtags denote significant differences compared with the baseline. #p < 0.05, ##p < 0.01 and ###p < 0.001. Data are shown as mean values of 12 h ± SEM.
The number of wake episodes and the average duration of NREM-sleep and REM-sleep episodes during the rest (L) phase was altered during the recovery phase in both groups (recovery day, F4,76 > 13.9, p < 0.001, Fig. 10A). In rest-workers, the number of wake episodes was elevated above baseline values through R3, and the average duration of NREM-sleep and REM-sleep episodes was reduced relative to baseline to R5. In active-workers, the number of wake episodes was not different from the baseline on any of the recovery days. Active-workers showed a longer duration of NREM sleep and REM sleep at R1, and shorter durations at R2, as compared with the baseline. The duration of REM-sleep episodes was elevated at R4 and R5, both compared with the baseline and with that of rest-workers. A similar, albeit non-significant, trend was evident under constant darkness.
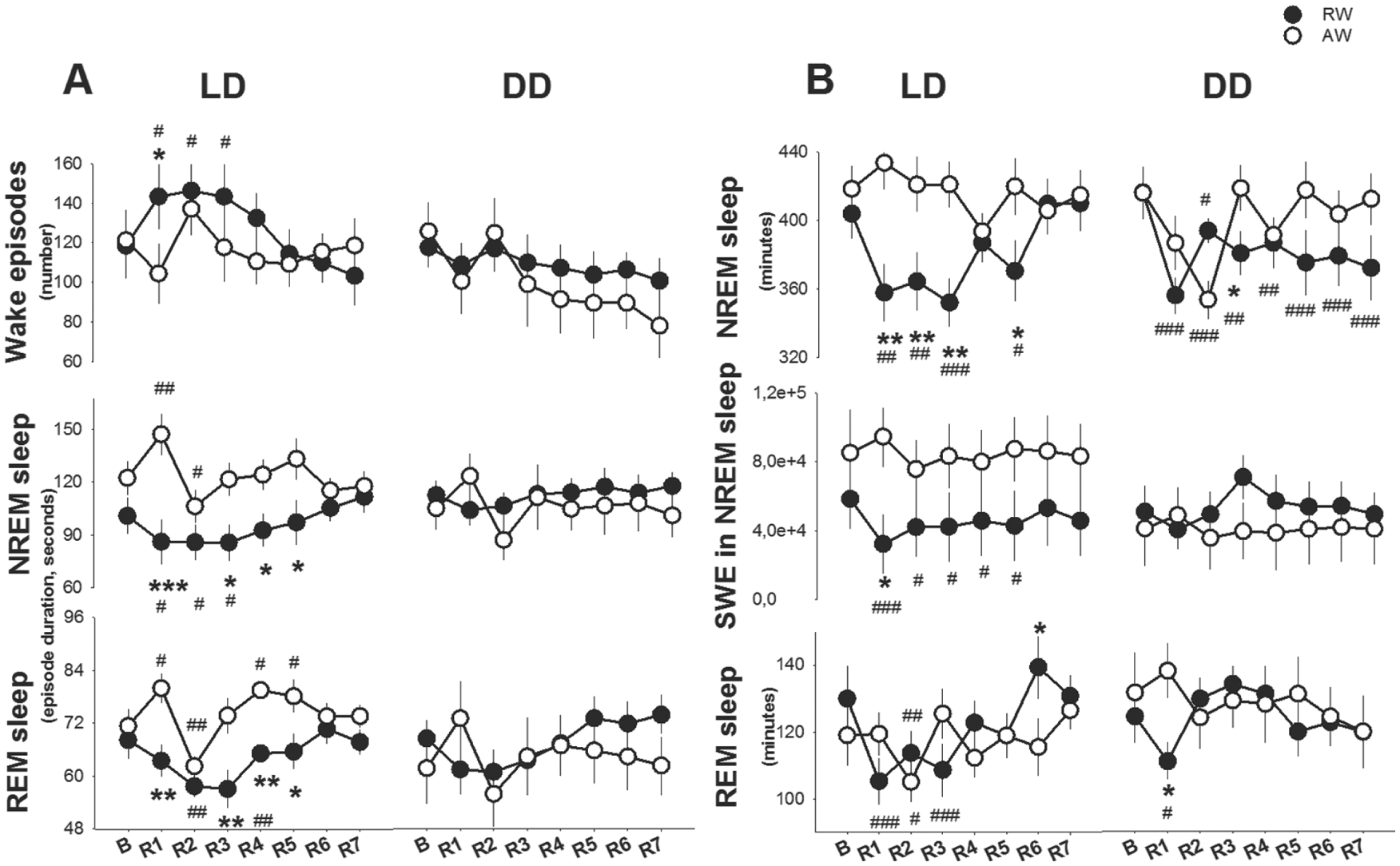
Sleep profiles during the light (rest) phase in the unrestricted recovery phase after the 4-day work schedule; recovery day 1 (R1) to R7 in LD and DD conditions. A) Number of wake episodes and duration of NREM- and REM-sleep episodes, and B) Time spent in NREM and REM sleep. Asterisks indicate significant differences between rest-workers (RW) and active-workers (AW), 12:12 LD conditions (RW n = 6 and AW n = 7) and constant darkness DD (RW n = 9 and AW n = 5). *p < 0.05, **p < 0.01, ***p < 0.001. Hashtags denote significant differences compared with baseline. #p < 0.05, ##p < 0.01 and ###p < 0.001. Data are shown as mean values of 12 hours ± SEM.
The time spent in NREM sleep and REM sleep during the rest (L) phase changed after the work schedule (recovery day X work-time, F(4,76) > 2.6, p < 0.043; Fig. 10B). In rest-workers, the time spent in NREM sleep was reduced at R1, R2, R3 and R5, both compared with baseline and active-workers. Under constant darkness, rest-workers displayed a reduced time in NREM sleep, relative to baseline—though not relative to active-workers—throughout the 7-day unrestricted recovery phase. In rest-workers recovering in LD, time in REM sleep was also reduced as compared with the baseline, but normalized at R4. Under the DD condition, REM sleep was normalized at R2. Active-workers housed in LD did not differ in time spent in NREM sleep or REM sleep in the rest phase as compared with the baseline. Active-workers housed in DD showed a transient decrease in both states at R2 only. Rest-workers housed in LD conditions showed a suppressed SWE in NREM sleep (work-time × recovery day, F11,66 > 2.5, p < 0.029). SWE was lower than the baseline at R1 through R5, and lower than that in active-workers at R1. There were no significant changes under constant darkness (see Fig. 10B).
The time spent in all states per recovery day (24-h LD) changed after the 4-day work schedule (recovery day, F4,100 >23.4, p < 0.001, in all cases). Intermediate and quiet wakefulness and NREM sleep showed an interaction effect with work-time (F7,77 > 2.3, p < 0.031). In active-workers, the work schedule increased the time spent in NREM sleep (13%) and REM sleep (10%), and reduced the time in quiet wakefulness (–28%) at R1 compared with baseline. Rest-workers did not show a sleep rebound in any of the recovery days; time spent in NREM sleep was shorter than baseline (−4 to −12%) and REM sleep was not significantly changed as compared with baseline on any of the recovery days. Compared with active-workers, rest-workers spent less time in NREM sleep R1 to R3 (p < 0.022), spent more time in intermediate and quiet wakefulness at R1, more time in quiet wakefulness at R2, and more time in active wakefulness at R3 as compared with active-workers (see also Table S4).
Discussion
This study assessed whether a 4-day schedule of rest-work leads to sleep disturbances during the work days and in recovery from the work schedule. Our data show that rest-work induced more pronounced acute changes, both in wakefulness and sleep, than active-work. The study additionally provides evidence for enduring sleep disturbances in the laboratory rat after rest-work, requiring 3-5 days to normalize in terms of different sub-states of wakefulness, and 6 days to recover both the amount of NREM sleep and slow-wave-energy in NREM sleep to obtain normal duration of NREM sleep and REM-sleep episodes. A comparison of the recovery data from the LD and DD conditions suggests that the increased time in active and intermediate wakefulness and the reduced time in NREM sleep induced by rest-work may be modulated by circadian factors.
Overall, the rodent model of shift work increased the daily time awake and reduced the daily time in sleep in both active-workers and rest-workers as compared with the undisturbed baseline condition. Under baseline conditions, all rats showed a typical polyphasic circadian sleep-wake pattern: asleep 70% to 80% of the time during the light phase (the main rest phase) and 20% to 30% of the time during the dark phase (the main active phase). The elimination of sleep during the active phase in the active-workers group resulted in a net loss of sleep time; although, the time in sleep in the active-workers’ rest phase was not impaired. Indeed, sleep consolidation measures indicated more stable sleep following the work-sessions. The active-workers showed rebound sleep during the first 24 h of recovery; albeit, this was only in animals housed in 12:12 LD conditions (Table S4). The rebound was mostly manifested as increased NREM sleep in the active (D) phase.
Our rat model of human night-shift work suggests a degraded waking or reduced arousal relative to day-shift work, as there were occasional micro-sleep episodes and higher SWE in quiet wakefulness during work, particularly in the rest-workers. In rats, task performance is shown to be incompatible with SWA during quiet wakefulness. Rats performing a conditioned licking task for liquid reward, which depends on sensorimotor integration in the cortex, failed at the task when the stimulus was presented simultaneous to a spontaneous cortical slow wave in quiet wakefulness (Rector et al., 2005; Krueger et al., 2013). Neurophysiologically, SWA reflects hyperpolarization-dependent synchrony in thalamocortical circuits (Steriade, 2004) caused by decreased activity in subcortical depolarizing neuromodulators as a function of increasing sleep need. In our study, the increase in SWE in the state of quiet wakefulness of the rest-workers during work therefore marks vulnerability to performance decrements. In the normal rested brain, dynamic changes in neuronal synaptic strength are thought to be essential for cognitive functioning (Bramham and Panja, 2014). The synaptic strength is suggested to be ‘recharged’ during sleep for optimal functioning during wakefulness [for review see (Gronli et al., 2014)]. It is tempting to speculate that this may be impaired after sessions with rest-work.
Four-day rest-work resulted in a pronounced redistribution of sleep to the rats’ endogenous main active (D) phase. Similar to the present changes in sleep and wake states, a disrupted daily activity-inactivity pattern in rodents has been reported in those that were forced to be active during the active phase, or exposed to restricted food access or running wheel access (Mistlberger, 1994; Salgado-Delgado et al., 2008; Barclay et al., 2012; Leenaars et al., 2012). In our study, active-workers returned to their home cage in their active phase (ZT22) and rest-workers in their rest phase (ZT10); consequently, the rest-workers initiated sleep faster than active-workers. Total sleep during the following 12-h period was reduced, as the animals are physiologically primed for wakefulness (ZT12-24). These results reflect the interaction of sleep drive and the circadian process in sleep propensity (Borbely and Achermann, 2004). One environmental factor that may contribute to the reduced NREM sleep after rest-work in rats is the photoperiod that was kept constant, also during the sleep opportunities between the work sessions, as light has been shown to impair NREM sleep in the rat (Tobler et al., 1994).
The restorative properties of sleep depend on its duration, quality, and continuity. The present data indicate impaired restorative properties of sleep after rest-work in the laboratory rat, as NREM sleep (duration, quality and continuity) was not conserved. NREM sleep disruption has consequences for daytime functioning, as it increases daytime sleep propensity (Dijk and von Schantz, 2005), and there are reports of subsequent performance deficits and disruption to memory consolidation (Walsh et al., 1994).
Distinct dynamics in the recovery from a 4-day rest-work schedule was demonstrated in our study: 3 recovery days for time spent in active wakefulness, 4 recovery days for time spent in quiet wakefulness and number of wake episodes, and 5 recovery days for time spent in intermediate wakefulness. Finally, after 6 recovery days, all parameters—time spent in NREM sleep, SWE in NREM sleep, duration of NREM-sleep and REM-sleep episodes—were normalized to baseline rest phase. In rats, SWA is correlated with arousal thresholds (Neckelmann and Ursin, 1993). Data from rest-workers in DD indicate that the extent of increase in active and intermediate wakefulness is modulated by circadian factors at 2 and 3 days into the recovery phase, respectively, and a reduced time in NREM sleep throughout the 7-day recovery phase. Measures of sleep consolidation, time in quiet wakefulness, and SWE in NREM sleep did not show persistent changes under DD. An increase in SWA on the first day in DD would be expected, as light cues modulate SWA (Tobler et al., 1994); however, this was not evident in rest-workers or in active-workers. Taken together, in rats, rest-work changed the circadian modulation of time spent in NREM sleep.
EEG dynamics of SWA after equal work-hours were suppressed in rest-workers as compared with active-workers, suggesting a lower NREM sleep propensity in rest-workers. There are several potential explanations for this, as accumulation of sleep debt is shown to be both time- and use dependent (Vyazovskiy et al., 2000; Huber et al., 2004). One explanation is the prior sleep-wake history. Rest-workers slept more than active-workers during the 2 h before work. According to the homeostatic model of sleep, more sleep (or a daytime nap) should lead to a reduction in sleep drive at the onset of the subsequent sleep period. This is found in humans instructed to take an afternoon nap, where the sum of SWA measured during the sleep (nap) and post-sleep period is found to only be slightly reduced as compared with baseline sleep (Werth et al., 1996). Our rest-working rats showed a progressive increase in cumulative SWE in NREM sleep after work-hours. However, the total SWE in NREM sleep during the 4-day work schedule was not different from that of active-workers. Second, a lower SWA after work-hours in the rest-workers group may have been the result of different distributions of sleep and wakefulness. Both animal and human studies demonstrate an association between SWA and sleep continuity, indicating that normal individuals with a greater amount of SWA have fewer awakenings than those with less SWA (Dijk and von Schantz, 2005). The number of wake episodes did not differ between rest-workers and active-workers during the work-schedules, but rest-workers displayed shorter durations of NREM sleep episodes in off-work hours throughout the work protocol. Third, different waking behavior during work-hours and between work-hours could result in reduced build-up of sleep debt in rest-workers; however, the data did not support this notion, as rest-workers spent less time in quiet wakefulness than active-workers at W3 and W4 during work, and, between work-hours, rest-workers displayed more time in wakefulness, and wakefulness showed higher gamma activity. The cellular mechanisms of gamma oscillations (~30 to 100 Hz) include neuroenergetics, high oxygen consumption rates, and increased sensitivity to metabolic stress. One hypothesis is that increased activity of fast-spiking interneurons may result in energy failure and neuronal network dysfunction (Kann, 2016). Albeit speculative, the higher gamma activity in between rest-work sessions may be indicative of metabolic and oxidative stress in fast-spiking interneurons after night-shift work. Normally, more active waking behavior potentiates an increase in sleep drive (Vyazovskiy et al., 2000; Huber et al., 2004). Finally, the build-up of sleep debt may have been lower in the rest-workers than in active-workers because of the slowing of EEG during quiet wakefulness. Recently, we have shown that quiet wakefulness in mice is permissive to metabolic changes that occur in NREM sleep (Gronli et al., 2016). The latter, together with time spent in sleep before work, may explain the lower build-up of sleep drive in rest-workers after work-hours.
Micro-sleeps or sleep attacks intruding into wakefulness are common in night-shift workers and sleep-deprived subjects (Torsvall and Akerstedt, 1987; Akerstedt, 2003); these dynamics result in nighttime performance deficits characterized by lapses of attention, slower responses and wake-state instability (Doran et al., 2001). Hypotheses about the underlying mechanisms of behavioral decrements in night-shift workers range from global instability in arousal; local sleep within discrete circuits, as a result of prolonged neuronal use; brain region-specific impairments (Jackson et al., 2013); or incomplete dissipation of homeostatic sleep drive, causing excessive daytime sleepiness in night-shift workers in the real world (Torsvall et al., 1989a; Akerstedt, 2003; Goel, 2011) and in simulated night-shift schedules (Chapdelaine et al., 2012; Jackson et al., 2014). All of the explanations are likely to involve intrusion of sleep-related, slow oscillatory events into the cerebral cortical networks as the need for sleep accumulates. Our findings of a higher SWE in quiet wakefulness were not explained by more time spent in quiet wakefulness or less total time spent in NREM sleep. Our finding of incomplete dissipation of homeostatic sleep drive between work hours are in line with studies on human night-shift work and night-time sleep restriction paradigms that often imply a conserved total time in the deep NREM sleep (stage N3) (Brunner et al., 1993; Belenky et al., 2003; Van Dongen et al., 2011; Chapdelaine et al., 2012). At the same time, both animal and human simulation of night-shift work indicate an impaired continuity of sleep (Santhi et al., 2008; Chapdelaine et al., 2012) might be one explanation as to why night-shift workers report insomnia and non-restorative sleep more often as compared with day and evening workers (Torsvall et al., 1989b; Pilcher et al., 2000). Presently, it is not possible to conclude whether night-shift work actually leads to gradually accumulating electrophysiological impairment and concomitant alterations of the sleep-wake cycle (Webb, 1983; Brunner et al., 1993; Dumont et al., 1997; Wyatt et al., 1999; Van Dongen et al., 2003; Paech et al., 2012); however, this should be investigated in future studies.
Rodent models established to study the effects of shift work have so far mainly focused on metabolic changes (Opperhuizen et al., 2015). While these studies provide important contributions to the understanding of the metabolic consequences of shift work, there are no direct comparisons to our study. Using animal models to investigate the complexity of shift work is challenging. We used altered timing of activity to model human night work, which resulted in sleep disturbances. Nutrition (food and water intake) was left ad libitum and LD condition was held constant during the work-schedule. Each of these aspects alters the circadian system and most likely wakefulness and sleep. In view of the fact that the work schedule in our rat model was conducted under LD conditions, we cannot dismiss the possibility that sleep in rest-workers between the work sessions was impaired by the absence of light.
In summary, with this rat model of a 4-day night work schedule, we demonstrate that work in the rest phase induced a pronounced redistribution of sleep to the endogenous active phase, and a degraded waking state. Also, the study provides evidence for enduring sleep-impairing effects of rest-work in the laboratory rat and that these effects may be modulated by circadian factors. These data demonstrate the potential importance of this model of night-shift work for future studies on the relationship between night-shift work and health consequences, and the mechanism(s) underlying these consequences.
Footnotes
Acknowledgements
This research was supported by grants from Faculty of Psychology, University of Bergen (Småforsk), Helse Vest Regional Health Authority (911781) and Norwegian Competence Center for Sleep Disorders. The authors express their thanks to Anne Marie Kinn Rød and Nina Harkestad for their assistance with collecting the data.
Conflict of Interest Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
