Abstract
Circadian clocks confer 24-h periodicity to biological systems, to ultimately maximize energy efficiency and promote survival in a world with regular environmental light cycles. In mammals, circadian rhythms regulate myriad physiological functions, including the immune, endocrine, and central nervous systems. Within the central nervous system, specialized glial cells such as astrocytes and microglia survey and maintain the neuroimmune environment. The contributions of these neuroimmune cells to both homeostatic and pathogenic demands vary greatly across the day. Moreover, the function of these cells changes across the lifespan. In this review, we discuss circadian regulation of the neuroimmune environment across the lifespan, with a focus on microglia and astrocytes. Circadian rhythms emerge in early life concurrent with neuroimmune sculpting of brain circuits and wane late in life alongside increasing immunosenescence and neurodegeneration. Importantly, circadian dysregulation can alter immune function, which may contribute to susceptibility to neurodevelopmental and neurodegenerative diseases. In this review, we highlight circadian neuroimmune interactions across the lifespan and share evidence that circadian dysregulation within the neuroimmune system may be a critical component in human neurodevelopmental and neurodegenerative diseases.
The circadian system is an evolutionarily adaptive system that produces rhythmic anticipatory signals in response to the 24-h periodicity on Earth (Patke et al., 2020). These rhythmic anticipatory signals decide the “whens” in life: When do we stay alert? When do we need peak motor function? When do we have time and energy for digestion? When can we rest and clear debris from our systems? Importantly, activities that are regulated by the circadian system result in time-of-day differences in encountering pathogens and injury (Costantini et al., 2020). Thus, it follows that the circadian system also regulates the immune system. Immune processes are energetically expensive, involving the recruitment of specialized cells and production of inflammatory molecules to combat pathogens and clear up dead or damaged cells (Ganeshan and Chawla, 2014). Thus, circadian regulation of immune function can facilitate resource allocation and lead to optimized functioning (Scheiermann et al., 2013).
This review will focus on the circadian regulation of neuroimmune function. The immune system and the central nervous system (CNS) have tightly controlled bi-directional communication (Dantzer et al., 2008). Specialized neuroimmune cells, such as microglia and astrocytes, monitor and maintain the CNS environment throughout the lifespan (Ortinski et al., 2022). Neuroimmune cells not only protect the CNS from pathology but also participate in homeostatic processes which facilitate neuronal functioning and regulate behavior (Reemst et al., 2016). Neuroimmune cells are regulated by the circadian system (Fonken et al., 2015; Takayama et al., 2016), and disruption of circadian rhythmicity can lead to dysfunction in these cells (Erblich et al., 2011; Fonken et al., 2016; Inokawa et al., 2020; Takayama et al., 2017). In this review, we will discuss circadian regulation of the neuroimmune system across the lifespan, from the emergence of circadian rhythms in childhood to the dampening of circadian rhythms late in life. Neuroimmune and circadian dysfunction are implicated in multiple neurodevelopmental disorders (NDDs) and neurodegenerative diseases. Thus, exploring the intersection of circadian rhythms and the neuroimmune system could lead to new mechanistic and therapeutic insights.
The Circadian System Regulates Rhythmic Daily Activities to Maintain Homeostasis
In mammals, daily rhythms are controlled by the primary circadian oscillator located in the suprachiasmatic nucleus (SCN) of the hypothalamus. The SCN responds to external cues: light is a potent zeitgeber that entrains or “fine-tunes” the SCN to the diurnal environment. Light information is detected by a specialized population of melanopsin-containing intrinsically photosensitive retinal ganglion cells (ipRGCs). ipRGCs project to the SCN via a monosynaptic connection through the retinohypothalamic tract (Berson et al., 2002; Hattar et al., 2002). The SCN receives this direct photic input from the retina as well as indirect input from the intergeniculate leaflet (IGL) and the brainstem (Reghunandanan and Reghunandanan, 2006). In the absence of external light cues, SCN neurons maintain a synchronized, endogenous, free-running rhythm of approximately 24 h (Armstrong et al., 1986; Vriend and Reiter, 2015). Rhythmic SCN output is communicated to extra-SCN (or peripheral) molecular clocks that exist in nearly every cell via neural and humoral signals. Extra-SCN clocks have self-sustained circadian oscillations; however, their circadian phase is synchronized to the SCN by efferent information from the SCN including endocrine and autonomic signals, body temperature, exercise, and food intake (Astiz et al., 2019; Brown et al., 2002; Tahara et al., 2017). For example, the SCN drives the rhythmic secretion of hormones from the hypothalamus and pituitary. In turn, endocrine molecules such as melatonin, serotonin (5-hydroxytryptamine, 5-HT), and glucocorticoids act via cellular receptors to feedback on expression of genes regulating circadian clocks (reviewed by Vriend and Reiter, 2015) (Mistlberger et al., 2000; Vriend and Reiter, 2015; Woodruff et al., 2016). This endocrine feedback is critical as disruptions in these hormone rhythms can desynchronize circadian rhythms (Carroll et al., 2007; Daut and Fonken, 2019).
At the molecular level, 10%-40% of all protein-coding genes in rodents and more than 50% in non-human primates and human tissues show 24-h rhythmic oscillations in a highly tissue-specific manner (Baldi et al., 2021; Li et al., 2022; Mure et al., 2018; Ruben et al., 2018; Storch et al., 2002; Zhang et al., 2014). Oscillating genes are regulated by a feedback inhibition loop of “core-clock” genes and their proteins (summarized in Figure 1). The core-clock transcription factors BMAL1 and CLOCK dimerize and form the positive arm of this loop. The CLOCK-BMAL1 dimer binds to a DNA cis-element E-box to promote the transcription of various target genes, including Per and Cry. Once PER and CRY proteins accumulate in the cytosol to sufficient levels, they are transported into the nucleus to inhibit the binding of the BMAL1-CLOCK dimer to the E-box. This feedback loop is necessary for circadian oscillation (Bae et al., 2001; Ueda et al., 2005; Yoshitane et al., 2019). In addition, BMAL1-CLOCK activity is regulated by REV-ERB/ROR-binding element (RRE). ROR and REV-ERBα/β (a.k.a. Nr1d1/2) compete for the response element to activate or repress Bmal1 transcription (Guillaumond et al., 2005; Yin and Lazar, 2005) and thus form an additional stabilizing loop within the clock. REV-ERBs also adjust rhythms to exogenous factors (Jolley et al., 2014). The core-clock loop does not solely define cellular timing—this circadian transcriptional loop also controls the transcription of clock-controlled genes that regulate rhythmic biological and physiological activities.
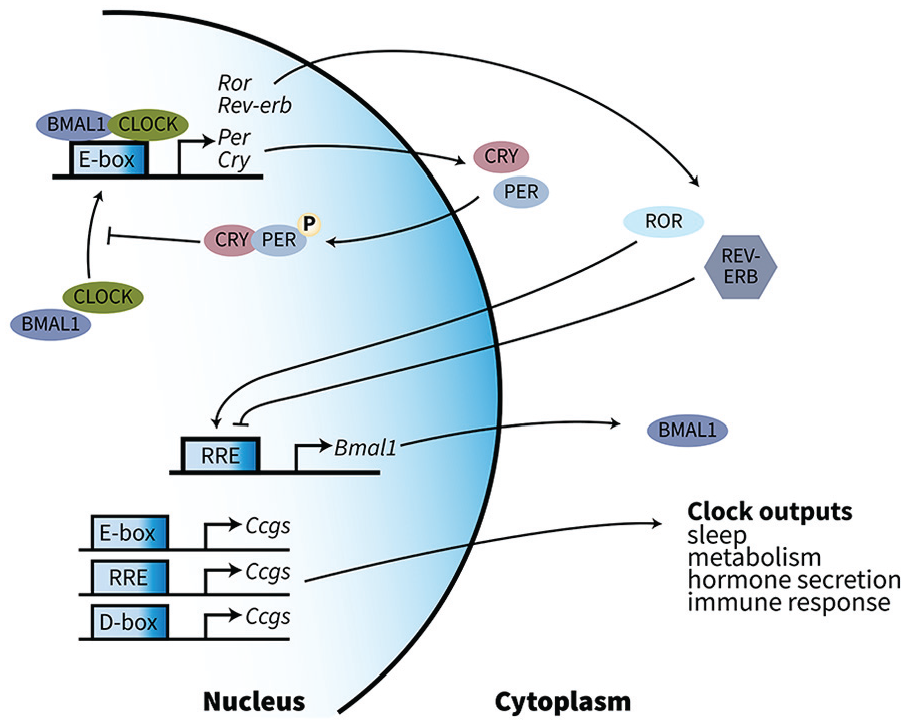
Circadian-related feedback inhibition loops control gene oscillation in mammals. The transcription factors BMAL1 and CLOCK dimerize and form the positive arm of this loop. The dimer binds to a DNA cis-element E-box to promote transcription of various target genes, including per and cry, whose protein products inhibit the binding of the BMAL1-CLOCK dimer. In addition, BMAL1-CLOCK activity is regulated by REV-ERB/ROR-binding element (RRE). REV-ERBα/β represses the transcription of Bmal1 and in turn stabilizes rhythms (Yin and Lazar, 2005), and adjusts rhythms to exogenous factors (Jolley et al., 2014). The “core-clock” loop controls the production of clock-controlled genes that are involved in rhythmic biological and physiological activities. Abbreviation: RRE = REV-ERB/ROR-binding element.
Circadian rhythmicity helps maintain physiological homeostasis. The circadian system regulates the sleep-wake cycle, metabolic and cardiovascular processes, hormone rhythms, and immune function (Beersma and Gordijn, 2007; Edgar et al., 1993; Scheiermann et al., 2013; Vriend and Reiter, 2015; Zhang et al., 2020). Importantly, with the advent of modern electricity, humans are no longer constrained to natural light cycles. Disruption of the circadian system due to artificial light at night, shift work, and long-distance air travel increases the risk of metabolic dysfunction (Cheng et al., 2021), cardiovascular disease (Knutsson et al., 1999), poorer mental health (Torquati et al., 2019), and cancer (Straif et al., 2007). Importantly, many of these issues are likely associated with impaired immune responses in shift workers (Ruiz et al., 2020).
In summary, the SCN is the primary circadian oscillator of the circadian system. The SCN is entrained by external zeitgeber cues and synchronizes the extra-SCN molecular clocks. Together these body clocks maintain homeostasis of various daily rhythms including physiological activities such as immune responses.
Circadian Regulation of Immune Function
The circadian system potently regulates immune function. Halberg and colleagues (1960) published one of the first studies to suggest this association, revealing that an Escherichia coli lipopolysaccharide (LPS) challenge during a mouse’s inactive phase induces substantially higher (80%) mortality than injection during the active phase (20%) (Halberg et al., 1960). This notable time-of-day difference in sepsis-induced mortality has been confirmed in subsequent work (Curtis et al., 2015; Deng et al., 2018; Feigin et al., 1969; Hrushesky et al., 1994; Lang et al., 2021); this association is circadian in nature (Lang et al., 2021).
The circadian system modulates sickness behaviors and susceptibility to infection (Scheiermann et al., 2018; Wang et al., 2022). For example, there is diurnal regulation of the sickness response in both humans and rodents. In humans, peak presentation of symptoms in response to viral infection and allergic asthma tends to occur during the late evening to early morning (Smolensky et al., 1995). Rodents exhibit time-of-day differences in susceptibility to infection. Rats and hamsters administered LPS during the inactive compared with the active phase exhibit prolonged and exacerbated sickness behaviors (Fonken et al., 2015; Franklin et al., 2003; Prendergast et al., 2015). A similar time-of-day regulation exists in rodents following viral infection/mimics: infection during the inactive phase leads to higher cytokine production and mortality (Edgar et al., 2016; Ehlers et al., 2018; Gagnidze et al., 2016).
These time-of-day-dependent changes in behavior and mortality are likely due to circadian regulation of immune cell activation and mobilization. The circadian system regulates both innate and adaptive immune responses (Scheiermann et al., 2018). For example, innate immune cells such as macrophages display heightened pro-inflammatory response during the inactive phase when presentation of sickness behaviors is highest. Direct transcriptional regulation of TLR4 signaling by the circadian clock underlies this circadian inflammatory response in macrophages, regulating the release of the pro-inflammatory cytokine tumor necrosis factor alpha (TNF-α; Keller et al., 2009). Moreover, the circadian clock gene, Bmal1, regulates nuclear factor kappa B (NFκB) transcription in macrophages (Curtis et al., 2015). Time-of-day variation in immune responses also occurs with adaptive immune cells such as T cells (Bollinger et al., 2011) and B cells (Silver et al., 2012). The circadian clock regulates inflammatory responses within these cells as well as their migration to different tissues throughout the day (Druzd et al., 2017; Ince et al., 2019). Diurnal regulation of adaptive immune cells has crucial implications for human health and disease, as revealed in recent studies of vaccination timing: In subjects receiving a vaccine against influenza or severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), vaccines administered in the morning are more effective than those given in the afternoon (Hazan et al., 2023; Ince et al., 2023; Long et al., 2016; Zhang et al., 2021). Although a full discussion of circadian regulation of the innate and adaptive immune system is beyond the scope of this review, several excellent recent reviews have addressed this topic (Curtis et al., 2014; Scheiermann et al., 2018; Westwood et al., 2019).
The Neuroimmune System: Development and Function of Specialized Glial Cells Across the Lifespan
The CNS is immunologically unique. The neuroimmune system is tailored to maintain the health and homeostasis of the CNS, which is sensitive to immune stressors. Immune signals are both initiated in the brain (e.g., following a brain injury) and communicated to the brain during peripheral immune activation (e.g., following infection or injury). To protect relatively vulnerable CNS tissue from pathogens and potentially overzealous peripheral immune responses, the brain has a physical blood-brain barrier (BBB) and is considered immune privileged. However, immune privilege is relative and dynamic. Multiple innate and adaptive immune cells circulate in spaces surrounding the parenchyma (e.g., the choroid plexus, meningeal lymphatic vasculature, perivascular spaces, and the skull and bone marrow) and relay immune information to the brain (Rustenhoven and Kipnis, 2022). For example, the choroid plexus—located within the cerebral ventricles—houses a large population of antigen-presenting resident macrophages and dendritic cells and is a gateway for CD4+ T-cell trafficking into the cerebrospinal fluid (CSF) and brain parenchyma (Dani et al., 2021; Meeker et al., 2012). Moreover, recent work highlights that the skull and vertebral bone marrow are immune cell reservoirs for the CNS (Cugurra et al., 2021). Importantly, the CNS also contains specialized resident immune cells, which support neuronal functions and regulate inflammatory reactions in the brain. This review will focus primarily on 2 glial cells: microglia and astrocytes. Neuroinflammatory responses are also regulated by additional cell types (e.g., endothelial cells, pericytes, macrophages) that are beyond the scope of this review (see review by Villabona-Rueda et al., 2019).
Microglia Functions
Microglia are the resident innate immune cells of the CNS. With their unique highly motile processes, microglia actively survey their territory and recognize signals from “healthy” neurons to maintain a homeostatic state. Under homeostatic conditions, adult microglia actively support and sculpt their environment by engaging in phagocytosis and secreting factors including cytokines and neurotrophic factors. Homeostatic microglia are responsible for synaptic pruning, a process that is crucial for proper brain morphogenesis and learning. Microglia dysfunctions impair activity-dependent synaptic pruning (Hoshiko et al., 2012; Schafer et al., 2012). Other functions of homeostatic microglia include supporting myelination, neurogenesis, and regulating BBB permeability.
Microglia derive from erythro-myeloid progenitors (EMPs) that are generated in the yolk sac blood islands at E8.5 in mice (Ginhoux et al., 2010). Microglia proliferate and gradually colonize the brain perinatally, with distinct morphologies depending on spatiotemporal localization. In early brain development, microglia are crucial for vasculature development and neuronal circuit assembly (Casano and Peri, 2015; Mosser et al., 2017; Perdiguero and Geissmann, 2016; Reemst et al., 2016; Thion and Garel, 2017), participating in cell death, axonal tract formation, and morphogenesis in the brain (Butovsky et al., 2014; Hattori, 2022; Thion and Garel, 2017). Embryonic microglia depletion leads to higher neural progenitor cell (NPC) proliferation and neuronal oligodendrocyte differentiation in the hypothalamus (Marsters et al., 2020). Microglia also regulate the balance of neuronal connections within specific circuits. For example, microglia accumulate at dopaminergic axon terminals at E14.5 and limit the outgrowth of dopaminergic neurons to the striatum (Squarzoni et al., 2014). Embryonic depletion of microglia leads to an imbalance in dopaminergic neuron innervation and altered neocortical inhibitory interneuron positioning, which may influence postnatal synaptogenesis (Paolicelli et al., 2011; Squarzoni et al., 2014). Taken together, before eventually reaching adult-like patterns by the end of the second postnatal week in mice (Bennett et al., 2016), a critical function of microglia in the developing brain is to support proper wiring of brain circuits.
In the adult CNS, a major function of microglia is to maintain the integrity of the nervous system. Following immune system activation, microglia adapt altered molecular, morphological, and functional properties to initiate an immune response and repair the damaged tissue or eliminate the pathogen. Importantly, the responses of microglia to stimuli vary between developing and aged brains, with microglia taking on a more pro-inflammatory phenotype later in life (Bisht et al., 2016; Fonken and Gaudet, 2022).
Astrocyte Functions
Astrocytes are the most numerous cell type in the brain and critically regulate the brain environment. Astrocytic end-feet tightly ensheathe the vasculature and help maintain the BBB. Specifically, Aquaporin-4 (AQP4) channels on astrocyte end-feet regulate the glymphatic system to remove toxic metabolic byproducts from the brain parenchyma (Mestre et al., 2018). Astrocytes support neurotransmission by gliotransmitter release, storing and releasing glucose, regulating extracellular ion concentration (Harada et al., 2015; Walz, 2000), and recycling glutamate at the synaptic cleft (Schousboe and Waagepetersen, 2005). In response to high neural activity, astrocytes can increase oxygen supply by regulating neurovascular coupling (Iadecola, 2017; Otsu et al., 2015) and secrete glutathione to promote neuronal redox homeostasis and prevent excitotoxicity (Bolanos, 2016). Like microglia, astrocytes also mediate synapse maturation (Allen et al., 2012) and elimination (Chung et al., 2013; Vainchtein et al., 2018). Moreover, astrocytes coordinate communication between neurons and oligodendrocytes; for instance, in response to adenosine triphosphate (ATP) from neuronal activity, astrocytes release cytokines that trigger myelination by oligodendrocytes (Ishibashi et al., 2006).
During CNS injuries astrocytes respond quickly with a process called “astrogliosis,” with altered morphology, gene expression, and function to limit cascading neural damage (Sofroniew, 2014; Zamanian et al., 2012). For example, reactive astrocytes form glial scars that improve tissue repair (Anderson et al., 2016). In addition, although microglia are the predominant source of inflammatory cytokines in the brain, reactive astrocytes also produce inflammatory cytokines in response to immune challenges (Leone et al., 2006).
Historically, a neuro-centric view of the CNS has focused on neurons as the key components in regulating animal behavior. However, microglia and astrocytes sculpt and regulate the lifelong plasticity of the CNS, demonstrating glial cells are not merely passive supporters of neurological function but rather active influencers of neurophysiology and behavior. Improper function of these neuroimmune cells can affect behavior: for example, embryonic microglia depletion using CSF1R inhibitor PLX5622 induces hyperactivity and alters anxiety-like behaviors (Rosin et al., 2018). Microglia and astrocyte dysfunction can influence cognitive function (Bissonette et al., 2010; Politis et al., 2011; Wadhwa et al., 2017; Zhang et al., 2019), circadian rhythms (Brancaccio et al., 2017; Sominsky et al., 2021), and reward-related behaviors (Adeluyi et al., 2019; Bull et al., 2014; Northcutt et al., 2015) in adult animals (reviewed by Ortinski et al., 2022). Furthermore, neuroimmune dysregulation during vulnerable periods may have long-term effects. For example, early-life immune activation and immune activation in late aging are associated with long-lasting behavioral consequences.
Circadian Regulation of Microglia and Astrocytes
Like the broader immune system, the neuroimmune system is regulated by the circadian system (summarized in Figure 2). Microglia display diurnal differences in their morphology and function. For example, during the inactive phase, microglia are less “ramified” with thicker processes: a morphology associated with an elevated pro-inflammatory response (Barahona et al., 2022; Takayama et al., 2016). Microglia cytokine production also peaks during the inactive phase both at baseline (Taishi et al., 1997) and in response to exogenous stimuli such as LPS (Fonken et al., 2015). Moreover, microglia exhibit time-of-day variations in phagocytosis (Griffin et al., 2020). Purinergic receptor P2Y12 in the CNS is expressed exclusively by microglia and mediates microglial recognition of cellular adenosine diphosphate (ADP)/ATP, which is released during the normal activity of neurons, astrocytes, and oligodendroglia, or in response to tissue damage (Lin et al., 2020). The microglial molecular clock regulates P2Y12 receptor via lysosomal cysteine protease cathepsin S and modulates spine density and synaptic strength in a time-of-day manner, which is important for memory consolidation (Hayashi et al., 2013; Nakanishi et al., 2021). Disruption of the microglial clock impairs cathepsin S activity and, in turn, disturbs sleep and sociability (Hayashi et al., 2013; Takayama et al., 2017). Furthermore, depleting microglia using a diphtheria toxin-receptor-knock-in rat model disrupts daily oscillations in temperature, activity, and energy metabolism (Sominsky et al., 2021). These time-of-day variations in microglial activity persist under 24-h constant darkness (Hayashi et al., 2013), suggesting a circadian pattern independent of light cues.
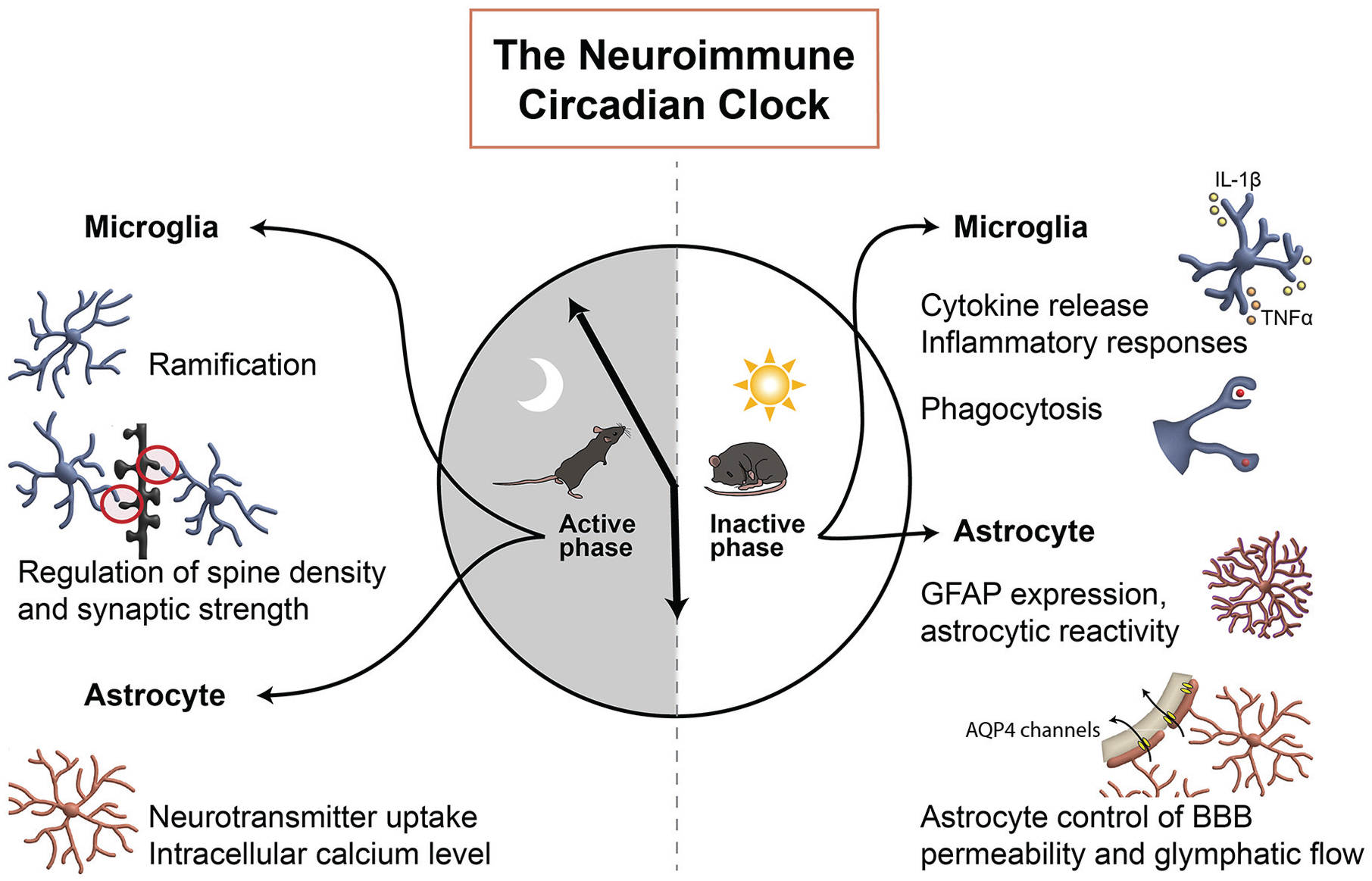
Simplified representation of some of the circadian features of neuroimmune cells. Two major neuroimmune cells of the CNS, microglia and astrocytes, display diurnal rhythmicity in morphology and functions that are critical for maintaining diurnal homeostasis of the CNS. The peak timings of microglial and astrocytic key functions are highlighted on the 24-h clock. Abbreviations: CNS = central nervous system; GFAP = glial fibrillary acidic protein; BBB = blood-brain barrier.
At the molecular level, adult microglia exhibit rhythmic expression of clock genes, including Per1, Per2, Rev-erbα, and Bmal1 (Fonken et al., 2015; Hayashi et al., 2013). The nuclear receptor REV-ERBα is one factor that acts as a hub between the circadian and immune systems (Amir et al., 2018; Chang et al., 2019; Gibbs et al., 2012; Pariollaud et al., 2018; Yu et al., 2013). REV-ERBα regulates the immune system through binding to response elements in promoter regions of pro-inflammatory genes such as nucleotide-binding oligomerization domain (NOD), leucine rich repeat (LRR), and pyrin domain-containing protein 3 (Nlrp3) and interleukin 1β (Il1β) (Pourcet et al., 2018). The binding of REV-ERBα then inactivates NF-κB signaling (interleukin [IL]-1β, IL-18, and TNF-α) and represses the expression and activation of the NLRP3 inflammasome (Duez and Pourcet, 2021; Griffin et al., 2019; Pourcet et al., 2018). Therefore, daily REV-ERBα abundance regulates the timing of NLRP3 expression and inflammatory cytokines production (Pourcet et al., 2018). Importantly, the NLRP3 inflammasome links the complement system and IL-1β production (Laudisi et al., 2013). The complement system, as a central part of innate immunity, consists of protein complexes that opsonize pathogens and promote inflammation and phagocytosis. REV-ERBα expression is positively controlled by BMAL1 and, in turn, negatively regulates Bmal1 transcription (Figure 1). This BMAL1-REV-ERBα axis serves as a regulator of complement expression and synaptic phagocytosis in the brain (Griffin et al., 2020). A synthetic REV-ERBα agonist dampens microglial circadian responses and reduces phagocytosis (Wolff et al., 2020). Conversely, Rev-erbα deletion increases microglial reactivity and induces enhanced but arrhythmic phagocytosis (Griffin et al., 2020). Modulating BMAL1 similarly targets neuroimmune cells: microglia-specific Bmal1 knockdown reduces inflammation and increases phagocytosis, both of which may elicit synapse loss (Curtis et al., 2015; Griffin et al., 2020; Wang et al., 2020).
Interestingly, microglia cell state of reactivity also influences circadian rhythms in these cells: polarizing microglia toward a pro-inflammatory phenotype (by co-treatment with LPS and interferon [IFN]-γ) results in a blunted and shortened microglial PER2::LUC rhythm (Honzlova et al., 2023). Conversely, anti-inflammatory polarization via IL-4 treatment enhances microglial rhythmicity and, more importantly, increases the amplitude of PER2::LUC rhythm in the SCN. A similar phenotype has been previously reported in bone marrow–derived macrophages: PER2::LUC oscillation in macrophages can be suppressed by pro-inflammatory polarization and enhanced by anti-inflammatory polarization (Chen et al., 2020). This novel finding supports that microglia polarization may regulate rhythmicity at both cell and tissue levels.
Although a growing body of literature suggests that circadian machinery in microglia may contribute to their physiological functions and some related behaviors, whether microglia prune all synapses in a similar Bmal1- and Rev-erbα-dependent manner, and whether microglia synaptic pruning is dependent on sleep, remains unclear.
Similar to microglia, astrocytes display circadian patterns. Astrocytes first appear in the SCN at E20 in rats (Munekawa et al., 2000). Astrocytes isolated from neonatal rat brains at P1-P2 show rhythmic expression of circadian clock genes Bmal1 and Rev-erbα (Carver et al., 2014), which continues in adulthood (Wen et al., 2020). Astrocytes also display diurnal differences in functions and activities. Glial fibrillary acidic protein (GFAP) is a classic marker of astrocyte reactivity and is involved in many important astrocytic functions in the CNS (Tykhomyrov et al., 2016); during the inactive phase, astrocytes have intensified GFAP expression indicating higher inactive phase astrocytic reactivity (Leone et al., 2006; Santos et al., 2005). During the active phase in the SCN, astrocytes exhibit a higher level of intracellular calcium, which is antiphase to that in SCN neurons (Hastings et al., 2019; Noguchi et al., 2017).
Astrocyte rhythms may contribute to neuronal and behavioral rhythms. The circadian oscillation in astrocytic intracellular calcium is phase-locked to extracellular glutamate levels, through which astrocytes suppress the activity of SCN glutamatergic neurons (Brancaccio et al., 2017). Similarly, ex vivo cortical astrocytes entrain their daily rhythms to co-cultured cortical neurons in a Bmal1-dependent manner (Barca-Mayo et al., 2017; also see review by Hastings et al., 2023). SCN astrocytes are sufficient to establish circadian rhythms in locomotion, although their rhythms are not required. Loss of SCN astrocytic clock by conditional Bmal1 knockout lengthens, but does not abolish, circadian rhythms of locomotor activity. On the other hand, the shortened circadian period of locomotor activity in CK1ε-tau mutant mice is restored by increasing the period of SCN astrocytes via astrocyte-specific removal of the tau mutation (Tso et al., 2017). Furthermore, rescuing Cry1 in SCN astrocytes restores circadian rhythms in locomotion in Cry1/2 knockout mice (Brancaccio et al., 2019), suggesting that circadian rhythms in SCN astrocytes contribute to SCN function during the active phase. In addition to its effect on circadian rhythms of locomotor activity, studies using conditional Bmal1 deletion suggest that astrocytic rhythms regulate many physiological functions, such as glucose metabolism (Barca-Mayo et al., 2020) and cognition (Barca-Mayo et al., 2017).
Given these striking recent results highlighting the role of astrocytes in the circadian system, several knowledge gaps remain to be filled. For example, future studies should explore the extent that astrocytes “control” SCN neurons and whether depleting astrocytes would diminish (or reduce) the ability of SCN neurons to generate daily rhythms. In addition, the activity of glutamatergic neurons is known to be regulated by astrocytes, but a more thorough investigation on the type(s) of SCN neurons whose activities are regulated by astrocytes, as well as the pathways of the astrocytic regulation of SCN neuronal activities, will further our understanding of how neuroimmune cells like astrocytes contribute to daily rhythms in physiology and behavior.
The circadian regulation of microglia and astrocytes during early development remains understudied; however, the evidence presented below (see section “Circadian rhythms influence neuroimmune wiring of neuronal circuitry”) suggests that circadian dysregulation may interrupt critical functions of glia in the developing brain. Future studies should explore the emergence of diurnal changes in microglia and astrocytes during early development. This information could help illuminate mechanisms underlying how glial clocks influence the developing brain, and whether circadian-neuroimmune dysfunction contributes to NDDs.
Circadian Rhythms Emerge Early in Life, Influence Neuroimmune Modeling of Brain Circuits, and Affect Neurodevelopmental Outcomes
During prenatal and early postnatal periods, a massive number of cells are generated and form the brain and the spinal cord. Morphogenesis and accurate brain circuit wiring during these critical periods are essential for proper brain function throughout the lifespan. Glial cells support brain development and support developing brain circuits and structures. However, most of the evidence demonstrating circadian regulation in neuroimmune components comes from studies using adult animals, and few studies have explored how circadian mechanisms influence microglial and astrocytic roles in early development. Indeed, the strengthening of rhythmicity during the first postnatal weeks in rodents coincides with the maturation of brain circuits. This section of the review will discuss the emergence of circadian rhythms early in life, and how these rhythms may govern the neuroimmune sculpting of the CNS to influence animal behaviors into adulthood. We further discuss evidence implicating circadian neuroimmune dysregulation in the pathophysiology of NDDs, including autism spectrum disorder (ASD), bipolar disorder (BD), and schizophrenia (Figure 3).
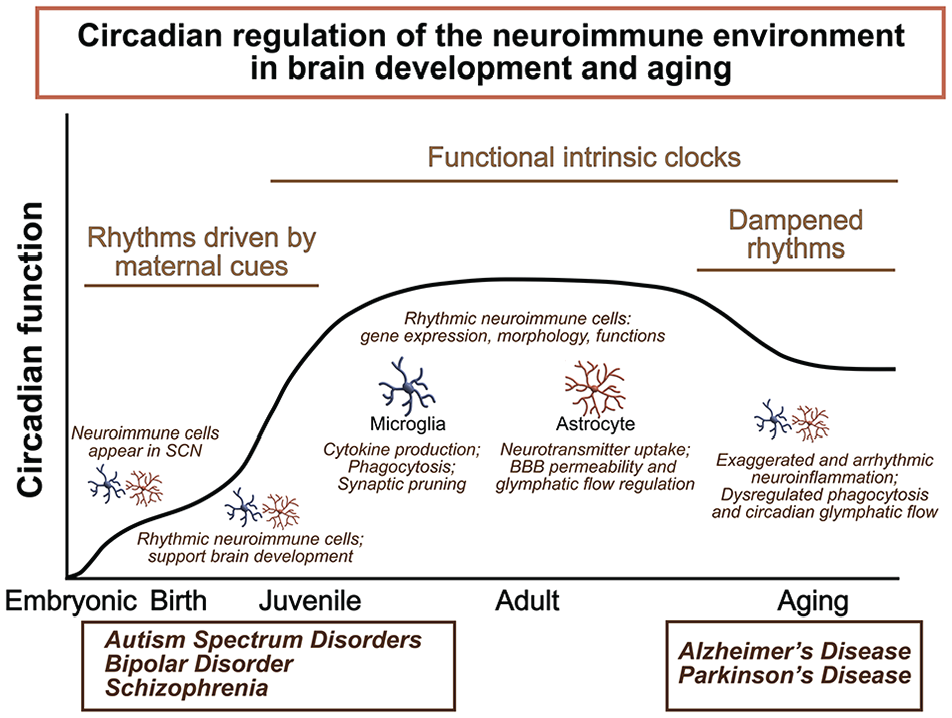
Summary of circadian regulation of the neuroimmune environment in brain development and aging. Within the central nervous system, specialized glial cells such as microglia and astrocytes surveil and maintain the neuroimmune environment, and their activities vary in a time-of-day manner. Circadian dysregulation can alter immune function, thereby contributing to susceptibility to neurodevelopmental and neurodegenerative diseases. Abbreviations: SCN = suprachiasmatic nucleus; BBB = blood-brain barrier.
The Developmental Emergence of Circadian Rhythms
In rodents, neurogenesis in the SCN region of the embryonic hypothalamus happens between E10 and E18 (reviewed by Landgraf et al., 2014; Carmona-Alcocer et al., 2020). Development of SCN neurons displays a spatiotemporal order: a mid-SCN core forms earlier than the surrounding SCN-shell with a ventrolateral to dorsomedial gradient (Altman and Bayer, 1978, 1986; Davis et al., 1990; Kabrita and Davis, 2008). Following SCN neuronal development, by E17, mRNA expression of clock components are detectable in the mouse SCN region (Shearman et al., 1997; Shimomura et al., 2001); however, daily oscillation of these clock genes emerges only after birth (Ansari et al., 2009; Greiner et al., 2022; Li and Davis, 2005; Sladek et al., 2004). Likewise, rhythmic clock gene expression in developing extra-SCN tissue such as the heart (Sakamoto et al., 2002) and the liver (Sladek et al., 2007) is detected gradually after birth (except for transcriptional repressor gene Rev-erbα that shows rhythmic activity in embryonic liver at E20; Sladek et al., 2007). In contrast to transcriptomic analyses, rhythmic expression is observed in ex vivo embryonic SCN cells from clock gene-driven bioluminescent promoter PER::LUC as early as E12 (Saxena et al., 2007). A recent study reports self-sustained, synchronized PER2::LUC rhythm in SCN cells from E15.5 mice (Carmona-Alcocer et al., 2018). Together these and other studies suggest that the fetal SCN expresses rhythmic oscillations of metabolic and electrical activities (reviewed in Carmona-Alcocer et al., 2020). In ex vivo embryonic liver, heart, and kidney cells, rhythms have been observed by E18 (Dolatshad et al., 2010; Umemura et al., 2017). This may suggest the existence of rhythms at the cellular level in embryonic tissue and a lack of synchronized rhythms at the whole tissue level.
In adult animals, the SCN is entrained to the surrounding light-dark environment by direct photic input via melanopsin-containing ipRGCs. ipRGC neurogenesis occurs between E11 and E14 in mice (McNeill et al., 2011), and melanopsin expression is detectable in the retinal ganglion cell layer by E15 (Fahrenkrug et al., 2004; McNeill et al., 2011). By E17, ipRGC axons reach the optic chiasma near the ventral SCN and gradually innervate the SCN during the first postnatal week. Although ipRGCs are present prenatally, they are not functional until postnatal day 7 (P7) (McNeill et al., 2011). Thus, during early development, circadian rhythms in rodent pups likely depend primarily on maternal cues instead of direct photic input (Bates and Herzog, 2020; Takahashi and Deguchi, 1983). For example, in the 1980s, Reppert and Schwartz first showed that the embryonic SCN has a time-of-day difference in glucose utilization. This daily variation is influenced by the circadian phase in maternal SCN (Davis and Gorski, 1985; Reppert et al., 1984; Reppert and Schwartz, 1983, 1986). Maternal signals such as dopamine and melatonin directly trigger embryonic SCN metabolic rhythms (Davis and Mannion, 1988; Weaver and Reppert, 1995). Interestingly, recent work indicates that these maternal signals are sufficient, but may not be necessary for entrainment (see review by Bates and Herzog, 2020); this suggests that the maternal clock may provide a parallel pathway for the fetus to perceive or reinforce phase of daily timing. Likewise, recent studies indicate that maternal metabolic signals directly elicit growth factor signaling, whereas maternal activity/feeding rhythms entrain neuronal activities in the embryonic SCN, suggested by enrichment analysis of rhythmically expressed genes (Greiner et al., 2022). The influence of maternal activity/feeding rhythms on pup SCN activity persists during postnatal development until weaning age (Olejnikova et al., 2018). Furthermore, maternal-fetal synchronization is abolished when the maternal SCN is ablated (Davis and Gorski, 1988; Greiner et al., 2022; Reppert and Schwartz, 1986).
Circadian Rhythms Influence Neuroimmune Wiring of Neuronal Circuitry
Disruption of the circadian system can have detrimental effects on various body systems. Modern-day causes of circadian disruption—including jetlag and shift working schedules—are modeled in animals by manipulating the light-dark environment. Mimicking chronic jet lag in adult mice by repeatedly advancing the light phase for 4 h over a year desynchronizes SCN neurons and activates immune pathways (Inokawa et al., 2020).
Early in life, microglia and astrocytes play essential roles in many neurodevelopmental processes (Erblich et al., 2011), so circadian disruptions during critical periods of development may alter neurodevelopmental trajectories (Estes and McAllister, 2016; Knuesel et al., 2014). Animal models of maternal immune activation (MIA) (Bauman et al., 2014; Missig et al., 2020; Rose et al., 2017), which recapitulate symptoms of ASDs (Fernandezde et al., 2017) or schizophrenia (Meyer and Feldon, 2012), have revealed that prenatal immune activation can induce long-term upregulation in inflammatory gene expression (Rose et al., 2017) and decrease complement-dependent homeostatic synaptic pruning, potentially interfering with neurodevelopmental synaptic refinement (Fernandez de Cossio et al., 2017). The developmental timing, type of immune challenge, and specific immune receptors involved collectively determine the effect on development, with unique MIA protocols recapitulating symptoms from distinct NDDs. For example, Poly I:C (viral mimetic) given to pregnant rats activates TLR3 and TLR4 receptors, resulting in lasting autism-related social and anxiety-like behaviors, whereas a similarly timed imiquimod (IMQ) immune challenge activates TLR7 and results in opposite behaviors (Missig et al., 2020). In some cases, MIA effects are sex-dependent, underscoring the complex interactions between immune and neurodevelopmental programs early in life (Haida et al., 2019). As several NDDs display a sex bias (Aleman et al., 2003; Christensen et al., 2016; Ferri et al., 2018; Loomes et al., 2017), and symptoms may differ between males and females (Diflorio and Jones, 2010; Erol et al., 2015; Sommer et al., 2020; Werling and Geschwind, 2013), a better understanding of sex-specific effects of MIA is likely to enhance prevention and treatment options for both sexes.
Accordingly, recent evidence has revealed that disrupting environmental light/dark cycles in utero or postnatally alters neuroimmune responses with long-lasting behavioral repercussions, similar to the effects of MIA during gestation. Early-life exposure to constant light (LL), dim light at night (dLAN), and chronic jet lag (CJL) affects developmental trajectories.
Constant Light
From conception, the maternal immune environment influences embryonic development, so even in utero, developing offspring can be affected by their mother’s response to altered environmental light. Pregnant mice exposed to constant light (LL) exhibit decreased placental expression of Serpinf1 and upregulation of macrophage markers, Iba1 and CD11b (Clarkson-Townsend et al., 2021). If changes in placental macrophages (Edlow et al., 2019) are mirrored in embryonic microglia, these immune shifts may alter the neuroimmune environment even during the earliest periods of brain development. Early postnatal LL also shifts the neuroimmune environment by increasing astrocyte number and decreasing astrocytic arborization (Canal et al., 2009). Behaviorally, mice raised from birth to weaning under LL have weakened circadian rhythms in wheel running (Ohta et al., 2006), which persist into adulthood, long after the animals are switched to LD. Therefore, developmental LL disrupts the immune system and induces lasting changes in animal behaviors.
Light at Night
It is increasingly recognized in humans that artificial lighting at night, especially blue light from electronic devices, negatively affects sleep/wake cycles and mood (Bedrosian and Nelson, 2013). Using a mouse model of dLAN, we showed that temporary exposure to dLAN during juvenile or adolescent stages alters microglial reactivity and mood behaviors long into adulthood (Chen et al., 2021). Female mice were most susceptible to these early-life circadian disruptions, which primed the immune system and increased microglial reactivity to an LPS challenge in adulthood. These dLAN-induced neuroimmune shifts were accompanied by increased anxiety-like behaviors in female adults and increased depressive-like behaviors in both male and female adults (Chen et al., 2021). Another study found similar anxiety-like behaviors in adult mice after in utero or postnatal dLAN exposure (Borniger et al., 2014). These data suggest that early life is a sensitive period during which circadian disruption via aberrant timing of dim light can induce negative long-term developmental outcomes.
Chronic Jet Lag
In an elegant cross-fostering study, Smarr and colleagues (2017) recently explored the behavioral effects of pre- and postnatal chronodisruption using a murine CJL model. In this CJL study, pregnant dams or newborn offspring were exposed to 6 h phase advances every 4 days. In utero or postnatal CJL decreased social behaviors and increased stereotyped behaviors, similar to the phenotype observed in other animal models for ASD. Interestingly, CJL had the greatest impact during gestation. CJL effects were also magnified in the offspring who experienced both in utero and postnatal CJL, implying cumulative impacts over developmental time. Collectively, the body of experimental data in rodent models shows that environmental light cycles affect developmental neuroimmune and behavioral outcomes.
Circadian Neuroimmune Dysregulation in the Etiology of Neurodevelopmental Disorders
Epidemiological studies have repeatedly shown a connection between early-life immune challenges and NDDs, such as ASDs, schizophrenia, and BD (Atladottir et al., 2010; Brown and Derkits, 2010; Jiang et al., 2016). Viral or bacterial infection during pregnancy is associated with neurodevelopmental disruptions to the offspring. Gestational immune activation, measured by maternal plasma cytokine levels, correlates with autism risk (Goines et al., 2011; Jones et al., 2017). Inflammation resulting from birth complications or acute perinatal hypoxia similarly increases the risks of long-term neurological complications (Hagberg et al., 2015). Early-life immune challenges can result in a sensitized immune system throughout life and prolonged disruptions to neurodevelopmental processes with lasting cognitive and psychiatric consequences (Knuesel et al., 2014).
The circadian system may be critical for balancing inflammatory and homeostatic neuroimmune functions throughout early brain development, as dysregulation of circadian neuroimmune function early in life is implicated in the etiology of several NDDs. Individuals with NDDs commonly exhibit increased neuroinflammation and sleep disruptions, but it is unclear to what extent these are correlating symptoms or if they interact to exacerbate disorder pathology. Many human disorders, including NDDs, do not stem from a single cause but involve interlocking molecular pathways, called the interactome, in which cellular processes influence one another to jointly contribute toward disorder progression (Barabasi et al., 2011; Paci et al., 2021). The emerging field of network medicine treats disease states as a network perturbation and attempts to characterize diseases by their distinct alterations to the interactome. By exploring the impact of gene and protein pathways on one another, this approach can help identify novel targets for treatment and intervention.
The centrality of the circadian system to a wide range of physiological functions suggests that whether or not molecular clock dysfunction is a primary cause of NDD, it may play a role in mediating the progression of a disorder, through its interactions with other cellular processes (Mullegama et al., 2015). Here, we discuss evidence that circadian neuroimmune disruptions may play a role in the pathophysiology of NDDs, including ASDs, BD, and schizophrenia. We highlight cellular pathways which have been implicated in these disorders and discuss how the molecular clock is impacted by and may synergize with these pathways to exacerbate neuroinflammation and influence disorder progression.
Autism Spectrum Disorders
ASDs are a heterogenous group of early-onset NDDs characterized by social and communication impairments and restricted, repetitive behaviors (American Psychiatric Association [APA], 2013). Neuroinflammation may play a role in the heterogenous developmental disruptions that occur in ASD. Positron emission tomography (PET) scans in young adults with ASD reveal increased microglial reactivity across many brain regions, most markedly in the cerebellum (Suzuki et al., 2013). Postmortem samples from individuals with autism reveal increased levels of brain cytokines (Li et al., 2009; Wei et al., 2011) and altered microglial morphology compared with typically developing individuals (Lee et al., 2017).
Circadian disruption is common in individuals with ASD and may contribute to neuroinflammation. Most patients with ASD experience chronic sleep disruption (Veatch et al., 2015) and exhibit altered rhythms in body temperature, activity, and melatonin and serotonin secretion (Glickman, 2010; Melke et al., 2008; Rossignol and Frye, 2011). Genome-wide association studies have uncovered gene polymorphisms and allele variants that confer significant risk for ASDs (Satterstrom et al., 2020; Stessman et al., 2017). Many of these disrupt the circadian system either directly through the molecular clock (Yang et al., 2016) or by altering the central pace-making function of the SCN (Ingiosi et al., 2019; Ma et al., 2022). Mutations in Npas, Per2, and Rev-erb (Goto et al., 2017; Hu et al., 2009) and hypomethylation of the Rora promoter (Nguyen et al., 2010) are implicated as ASD risk factors.
Outside of core clock components, other ASD-related molecular targets, such as FMRP and mTOR, interrupt both neuroimmune and circadian systems. Transcriptional silencing of Fmr1, which encodes the fragile X mental retardation protein (FMRP), causes Fragile X Syndrome, which is the most common single-gene cause of ASD (Richter and Zhao, 2021). FMRP interacts with several circadian transcripts, and Fmr1 knockout mice show reduced hippocampal clock gene expression and time-of-day-specific memory impairments (Sawicka et al., 2019). FMRP-deficient microglia show exaggerated inflammatory responses to LPS (Parrott et al., 2021), and loss of FMRP results in an increased number of dendritic spines (Bagni and Greenough, 2005), possibly from defects in microglial-mediated synaptic pruning during development (Jawaid et al., 2018). The mTOR-eIF4E pathway provides another intriguing link between central clock dysfunction and inflammation/homeostasis imbalance in ASDs. The mTOR pathway is responsible for photic entrainment in the SCN (Cao et al., 2010), as well as regulating the molecular clock (Ramanathan et al., 2018; Zheng and Sehgal, 2010). Overactive mTOR-dependent protein synthesis increases autism-like behaviors in mice, and microglia-specific eIF4E overexpression results in increased microglial reactivity, increased phagocytosis, decreased spine pruning, and autism-like behaviors (Xu et al., 2020). Disruptions to either FMRP or mTOR signaling pathways impair circadian rhythmicity and alter neuroimmune function early in life, contributing to aberrant developmental processes.
Bipolar Disorder
BD is an affective disorder with typical onset in adolescence or early adulthood. BD is characterized by cyclic mood disturbances, alternating between mania and depression, with euthymic periods of normal mood function interspersed (Vieta et al., 2018). Maternal influenza infection increases the risk of offspring developing BD (Canetta et al., 2014; Parboosing et al., 2013). Depressive and manic episodes are associated with increased plasma concentrations of inflammatory cytokines (O’Brien et al., 2006), and postmortem analysis in prefrontal cortex from humans with BD revealed higher cytokine levels and increased astrogliosis and microgliosis than in healthy controls (Rao et al., 2010). RNA sequencing from human tissue has also implicated neuroimmune pathways in BD (Pacifico and Davis, 2017).
In the general population, disrupted circadian rhythms are associated with increased rates of mood disorders and poorer subjective well-being (Lyall et al., 2018). In BD, sleep disruptions occur in the early prodromal phase (i.e., mild symptoms experienced before the major signs of a disease state), sometimes occurring up to 10 years before the onset of the first manic episode (Conus et al., 2008). Furthermore, actigraphy studies demonstrate that circadian activity in bipolar individuals is distinct from that of control individuals, even in euthymic cycles (the stable mental state) (Salvatore et al., 2008). Also, genetic polymorphisms in core clock components occur at higher rates in individuals with affective disorders (Etain et al., 2011; Kripke et al., 2009; Severino et al., 2009), and a Per3 variable-number tandem repeat influences the onset age of BD (Benedetti et al., 2008). Collectively, these data suggest that circadian disruptions represent a persistent pathological feature of the disorder and not simply a symptom of mood state.
Disturbance to the molecular clock may cause the development of BD, as suggested by studies using gene disruption in mouse mutants. Mice with Clock gene knockdown develop mania-like behaviors, including hyperactivity and reduced anxiety (Roybal et al., 2007). When restricted to the ventral tegmental area (VTA) alone, Clock knockdown recapitulates both manic and depressive-like behaviors, analogous to those seen in humans with BD (Mukherjee et al., 2010). Early in life, the molecular clock influences the wiring of the dopaminergic system, which plays a role in motivation, attention, learning, and mood regulation. Through adulthood there are circadian rhythms in the transcription of monoamine oxidase A, which degrades dopamine in the synaptic cleft (Hampp et al., 2008). Clock knockdown interferes with this regulation, increasing dopaminergic signaling, which may underlie bipolar mood fluctuations. Behavioral phenotypes in Clock mutant mice are ameliorated by lithium treatment, which is a potent inhibitor of glycogen synthase kinase 3 (GSK-3), suggesting a central role for GSK-3 in modulating circadian neuroimmunity. In Drosophila, GSK-3 shortens the circadian period (Martinek et al., 2001). In rodents, GSK-3 is expressed in a circadian fashion and modulates the expression of multiple clock genes in SCN, promoting the phosphorylation and degradation of CRY2 (Kurabayashi et al., 2010), and dampening the amplitudes of BMAL1 and PER rhythms (Besing et al., 2015). GSK-3 is also known to increase microglial pro-inflammatory reactivity (Martinez et al., 2002). Lithium’s inhibition of GSK-3 activity may therefore act as a circadian neuroimmune therapy, working by increasing amplitude of Per2 rhythmic expression (Li et al., 2012) and decreasing inflammation.
Schizophrenia
Schizophrenia is a disabling psychiatric disorder involving disordered thinking, hallucinations, or delusions, and often first presents in adolescence or young adulthood (Tandon et al., 2008). Schizophrenia is associated with a decrease in gray matter, due to excessive developmental synaptic pruning (Prasad et al., 2016). The complement system regulates synaptic pruning by tagging synapses for removal with molecular signals that stimulate microglial engulfment (Schafer et al., 2012; Stevens et al., 2007). The complement component C4 is implicated in the altered pruning in schizophrenia, as greater schizophrenia risk is associated with the C4A allele compared with the C4B allele (Sekar et al., 2016). Serum complement analyte concentrations can be used as a biomarker of first-episode psychosis (Kopczynska et al., 2019) and induced microglia-like cells from patients with schizophrenia exhibit greater synaptic engulfment, which correlates with C4 risk variant (Sellgren et al., 2019).
Like ASD and BD, several lines of evidence suggest that circadian dysfunction may be a central contributor to pathology in schizophrenia. Individuals with schizophrenia often experience lifelong sleep difficulties and disrupted cycles of melatonin production (Johansson et al., 2016; Wulff et al., 2012). For adolescents with a familial risk of psychosis, circadian disturbances correlated with worse outcomes 1 year later (Lunsford-Avery et al., 2017). Patients with schizophrenia exhibit loss of rhythmicity in several core clock genes, including Clock, Per2, and Cry (Johansson et al., 2016), and postmortem cortical tissue from individuals with schizophrenia reveals reduced rhythmicity in typical circadian transcripts (Seney et al., 2019). Genetic risk factors may underlie this circadian arrhythmicity, as the Clock gene T3111C polymorphism is more highly prevalent in individuals with schizophrenia than in controls (Takao et al., 2007). One additional gene implicated in the etiology of schizophrenia (Hennah et al., 2003), Disrupted-in-Schizophrenia1 (Disc1), is also linked to the molecular clock. Disc1 was discovered through a gene-linkage study of a Scottish family with high penetrance of schizophrenia and adolescent affective and conduct disorders (St Clair et al., 1990). The DISC1 protein is a direct target of the BMAL1/CLOCK heterodimer, and it stabilizes BMAL1 activity through interactions with GSK-3β (Lee et al., 2021). Faulty DISC1 activity may alter circadian rhythms, disrupt neuroimmune homeostasis, and could shift microglia into a state of excessive pruning, contributing to schizophrenia pathology.
Here, we discussed links between circadian and neuroimmune function in human NDDs and possible mechanisms. Each of these disorders involves complex disruptions to the typically developing nervous system, with heterogenous symptomatology and multifaceted and interacting mechanisms. These disorders express comorbidly in the general population, and studies suggest that ASDs, BD, and schizophrenia share overlapping genetic and environmental risk mechanisms (Carroll and Owen, 2009; Gandal et al., 2018; Rees et al., 2021; Stefansson et al., 2009). Many of the molecular players already discussed have also been implicated in neurodegenerative disorders (Bie et al., 2019; Bleuze et al., 2021), which will be discussed in the next section. Circadian disruptions and altered neuroimmune pathways represent a common theme in a wide range of neurological disorders throughout the lifespan. Future studies should explore whether strengthening the circadian system can reduce neuroinflammation and alleviate symptoms in these disorders.
Age-Associated Circadian Changes and Neurodegenerative Diseases
The neuroimmune system plays multiple roles in promoting healthy brain development and optimal nervous system functioning throughout the lifespan. During natural aging, neuroimmune function gradually shifts toward higher levels of basal inflammation (Erraji-Benchekroun et al., 2005; Soreq et al., 2017). Both neuronal and glial transcription change over time, with a general decrease in neuronal transcription and an increase in immune-related transcripts with age (Boisvert et al., 2018; Grabert et al., 2016). In healthy aging, microglia increase the basal release of pro- inflammatory cytokines, such as IL-1α, TNF, and C1qA (Clarke et al., 2018), and astrocytes exhibit increasingly reactive transcriptional profiles with upregulation of complement and major histocompatibility complex (MHC) (Boisvert et al., 2018). These changes result in an increase in synaptic pruning and a gradual loss of dendritic spines and gray matter with age.
The aging neuroimmune system may also be compromised by progressively weakening circadian rhythms. Circadian amplitudes of activity and body temperature decrease with aging (Czeisler et al., 1992; Musiek et al., 2018), and aged adults wake earlier and have melatonin peaking earlier in the circadian cycle (Duffy et al., 2002). Elderly individuals experience more fragmented sleep (Carskadon et al., 1982), with over half of individuals aged 65 or older experiencing some form of chronic sleep disruption that is linked to negative health outcomes (Foley et al., 1995).
There are several mechanisms implicated in this circadian decline. First, clock gene oscillations throughout the brain dampen with aging (Chen et al., 2016). Second, there is a decline in SCN synchronization with aging, although there are mixed findings regarding whether this is due to the death of vasoactive intestinal polypeptide (VIP) neurons (Roberts et al., 2012; Wang et al., 2015) or the desynchronization of physiological properties of SCN neurons (Curran et al., 2019). Transplantation of fetal or young SCN tissue into aged hamsters can restore disrupted circadian rhythms and increase longevity, but the mechanisms for this effect are unclear. Electrophysiological recordings in rodent SCN demonstrate that neuronal membrane properties lose rhythmicity with age, due to decreasing GABAergic input and decreased A-type potassium currents (Farajnia et al., 2012).
Another mechanism for dampened rhythms with aging may involve a reduced expression of the NAD+-dependent deacetylase, sirtuin 1 (SIRT1) (Meng et al., 2020). Sirtuins were initially noted for their ability to increase longevity in yeast and have since been implicated in many aging-related processes across animal species, with a role in glucose metabolism, mitochondrial oxidation, telomere regulation, transcriptional repression, DNA repair, and maintaining genome integrity (Toiber et al., 2011). SIRT1 directly modulates the cellular clock by both binding the BMAL1-CLOCK heterodimer and deacetylating PER2 to enhance the amplitude of the feedback loop. SIRT1 protein levels naturally decline with age, contributing to weakening circadian rhythms, but transgenic mice overexpressing SIRT1 retain robust rhythms in gene expression, even in old age (Chang and Guarente, 2013).
Circadian Dysregulation of Neuroimmune Cells With Aging
Animal models show that the neuroimmune system loses circadian rhythmicity with aging, possibly contributing to the increased inflammation seen in immunosenescence. Hippocampal microglia from young rats (3 months) show robust circadian rhythms in clock genes and inflammatory markers, but aged rats (24 months) have reduced rhythms in Per genes and tonically increased inflammation, without circadian variation (Fonken et al., 2016). In addition, SIRT1 is also involved in regulating the immune system (Gamez-Garcia and Vazquez, 2021). Nuclear sirtuins inhibit microglial inflammation, but in microglia in the aged brain, an increase in nuclear cathepsin B degrades sirtuins, thereby increasing neuroinflammation (Meng et al., 2020). SIRT1 hereby contributes to both circadian weakening and neuroinflammation, significantly affecting longevity (Satoh et al., 2013).
Circadian regulation in the glymphatic system also decreases in the aged brain. The glymphatic system is a perivascular network that regulates CSF influx into the brain parenchyma and interstitial solute clearance by draining into the lymphatic system. In young animals, glymphatic clearance is highest during sleep, as slow-wave brain activity drives an increase in interstitial volume, allowing for the clearance of accumulated metabolites (Xie et al., 2013). However, the knockout of AQP4 channels on astrocytic end-feet abolishes glymphatic rhythmicity (Hablitz et al., 2020), highlighting the importance of AQP4 channels to proper timing of metabolite clearance. In human postmortem tissue, a decrease in perivascular AQP4 is associated with increasing age (Zeppenfeld et al., 2017), suggesting that human glymphatic function may similarly lose circadian rhythmicity in aging.
Circadian Components in Age-Related Neurodegenerative Disorders
Age-related changes in circadian neuroimmunity may predispose certain individuals to develop neurodegenerative disorders, which involve a toxic accumulation of protein aggregates, exaggerated and detrimental neuroinflammation, excessive neuronal death, and precipitous loss of cognitive or motor functions. Although protein misfolding and aggregation are pathological hallmarks of neurodegenerative diseases, there is no single driver of pathology; rather, degeneration involves interactions across multiple systems (Gan et al., 2018). Because the neuroimmune system plays a central role in phagocytosis and clearance of these protein aggregates, neuroimmune dysfunction, which may be exacerbated by underlying circadian dysrhythmia, may contribute to the progression of these diseases. Neurodegeneration is distinct from the “healthy” aging process. Here, we review the links between circadian and neuroimmune disruption in neurodegeneration, most of which are being studied in the context of two of the most common forms of neurodegeneration: Alzheimer’s disease (AD) and Parkinson’s disease (PD). Again, it is important to delineate correlation from causation. Neurodegenerative disorders often display concurrent shifts in both immune and circadian systems; accumulating evidence suggests that a pathological interaction of these dysfunctioning systems may play an additional role in disease progression.
Alzheimer’s Disease
AD is characterized by progressive memory impairment, along with psychiatric conditions like depression, aggression, and sleep disturbances. Pathological characteristics of AD include a buildup of protein aggregates, including intracellular neurofibrillary tangles and amyloid-beta (Aβ) plaques, diffusely affecting the entire brain, including the hippocampus, cortex, as well as hypothalamus (Ogomori et al., 1989). Inflammation is a key component of AD susceptibility. Inflammatory genes are highly implicated in AD, including complement receptor 1 and Trem2, which regulate phagocytosis and are important for the clearance of aβ plaques (Karch and Goate, 2015).
In humans, preclinical AD pathology is associated with fragmentation of sleep patterns, with 70% of AD patients exhibiting sleep disruptions, when measured by in-home actigraphy (Musiek et al., 2018), suggesting that sleep could be used as a biomarker for early AD diagnosis and intervention. For individuals with AD, sleep disturbances exacerbate behavioral symptoms, including aggression (Moran et al., 2005). Human postmortem samples suggest that aberrant rhythms in DNA methylation and Bmal1 expression could underlie circadian disruption in AD patients (Cronin et al., 2017).
While the SCN is less susceptible to plaque-induced cell death than the hippocampus or cortex (Stopa et al., 1999), this brain region displays a course of pathology in AD that is more severe than what occurs with healthy aging and is likely to play a role in the progression of AD (Van Erum et al., 2018). Through mild and moderate AD, some patients maintain typical circadian rhythmicity in hormone patterns including plasma cortisol (Hatfield et al., 2004), but AD patients have fewer SCN VIP neurons than age-matched controls (Stopa et al., 1999; Zhou et al., 1995). Severely affected individuals show significant death of VIP neurons and astrogliosis in the SCN (Stopa et al., 1999). Loss of vasopressin or neurotensin neurons in the SCN is associated with specific fragmentation in activity rhythms (Harper et al., 2008), suggesting that pathology underlies the progressive sleep disturbances associated with AD.
Circadian disruptions exaggerate neuroinflammation and increase neurotoxicity in AD rodent models with genetic mutations that alter amyloid or tau processing. Overexpressing amyloid precursor protein (APP) promotes an early AD phenotype in young animals. Two-month-old APP-knock-in mice have altered clock gene expression and increased time-of-day-dependent expression of inflammatory markers, including IL-6 and TNF-α (Ni et al., 2019). In another AD mouse model with the human tau variant, tauP301S, inhibiting microglial Sirt1 increases the expression of IL-1β in young mice and exacerbates memory deficits in aged mice (Cho et al., 2015). As previously discussed, Sirt1 deletion also disrupts circadian rhythms. As Sirt1 naturally declines with aging, dampened circadian rhythms and increased IL-1β expression may combine with other genetic risk factors to promote inflammatory pathology in individuals with AD.
Animal studies provide the strongest evidence that the circadian system directly modulates AD progression: in mice, a robust molecular clock facilitates the clearance of Aβ plaques in AD models, whereas circadian disruptions inhibit plaque clearance (Lee et al., 2020; Zhu et al., 2018). Bmal1 expression drives microglial phagocytosis of fAβ1-42 in immortalized BV-2 mouse microglial cells. In 5xFAD mice, pharmacological or siRNA inhibition of REV-ERBα/β increases Bmal1 expression and microglial fAβ1-42 clearance (Lee et al., 2020). Crossing the Rev-erba knockout mouse with the 5XFAD mouse decreases amyloid plaque burden and disease-associated microglial gene signatures (Lee et al., 2020). In the P301S mouse model of tauopathy, early-life sleep disruption increases glia cell reactivity, increases the accumulation of soluble tau, and hastens neurodegeneration in the hippocampus and amygdala, resulting in motor and memory impairments (Zhu et al., 2018).
Beyond local protein degradation mechanisms, circadian dysregulation inhibits the clearance of plaques through the glymphatic system in AD. Acute sleep deprivation increases the amount of Aβ accumulation in humans (Shokri-Kojori et al., 2018). AQP4 is implicated in the pathophysiology of AD due to its role in the clearance of Aβ plaques (Iliff et al., 2012). Furthermore, Aβ pathology and AD severity correlate with decreasing astrocytic AQP4, even when controlling for age (Zeppenfeld et al., 2017).
Parkinson’s Disease
PD is a progressive neurodegenerative disorder involving the death of dopaminergic neurons projecting from the substantia nigra to the basal ganglia resulting in disrupted motor planning, initiation, and execution. Motor symptoms include bradykinesia, tremor, rigidity, and postural instability, while non-motor features include cognitive impairment, emotional dysregulation, and sleep disruptions (Jankovic, 2008). Sleep irregularities including nighttime insomnia or daytime sleepiness are reported in a majority of individuals with PD (Stefani and Hogl, 2020), and cognitive, motor, and mood symptoms tend to increase at dusk. Termed “sundowning,” this time-of-day-dependent exacerbation of symptoms implies the involvement of circadian mechanisms in PD behaviors.
Several studies suggest that maintaining the integrity of the molecular clock in aging may reduce or delay neuronal death in PD. Rodent models of PD include intraperitoneal injection of 6-hydroxydopamine (6-OHDA) or 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) which causes acute death of dopaminergic neurons in the substantia nigra, motor impairments, and time-of-day dependent depression and anxiety-like behaviors. Bmal1 knockout mice exhibit increased microglial number and inflammatory cytokines like IL-1β and TNF-α after MPTP injection, exacerbating dopaminergic cell death, compared to wild-type mice (Liu et al., 2020). The opposing limb of the molecular clock, REV-ERBα, is also protective in PD models. Rev-erbα knockout mice exhibited worsened motor impairments and neuronal death in the substantia nigra (SNpc) and VTA after 6-OHDA injection compared to wild-type controls (Kim et al., 2018). Rev-erbα knockouts also display a prolonged increase in the number of microglia, but not astrocytes, after 6-OHDA injection. Local injection of the REV-ERBα antagonist, SR8278, into the VTA ameliorated these mood-related behaviors (Kim et al., 2022). Both Bmal1 and Rev-erbα may be neuroprotective for dopaminergic neurons in the striatum, although the mechanisms through which these opposing limbs may contribute to neuroprotection are unknown.
Future Perspectives
Additional research using adult animal models will unveil the mechanisms by which clock genes direct neuroimmune activities. Because the circadian system involves both cellular and system-level controls, it is difficult to disentangle the mechanisms linking circadian disruption and neuroinflammation. For example, global Rev-erbα knockout increases pro-inflammatory cytokines in the brain, but this inflammation may indirectly result from increased neuronal oxidation levels which then secondarily drive pro-inflammation (Griffin et al., 2019). Rather than global gene knockouts, cell-type-specific clock gene knockouts allow researchers to induce circadian disruptions in a single cell type and analyze its effects on network functioning and its broader impacts on animal behavior. For example, microglia-specific Bmal1 knockdown has revealed a cell-autonomous role for Bmal1 in mediating neuroinflammation and synaptic phagocytosis (Griffin et al., 2020). Continued work is needed using cell-type-specific clock gene knockouts to dissect cell-autonomous and nonautonomous mechanisms.
Microglia and astrocytes exhibit unique morphological and functional characteristics depending on the brain region (Grabert et al., 2016). Targeting cell-specific manipulations of clock genes to specific brain regions is also a research priority because regions might differ in sensitivity to circadian disruptions. For example, is the dopaminergic system more sensitive to circadian perturbations than other neuromodulatory systems? Dopaminergic dysfunction may be central to many psychiatric and neurodegenerative disorders because it requires tighter circadian regulation, hence may be more sensitive to circadian disruption than other systems.
Circadian disruptions alter neuroimmune homeostasis and interfere with healthy brain development or aging. Their involvement in a broad range of NDDs and neurodegenerative diseases highlights the need for a deeper exploration of circadian neuroimmune functions at all life stages. Despite the importance of early-life brain development, few studies have explored the role of circadian rhythms in early-life neuroimmune activities. How do maternal circadian rhythms influence fetal neuroimmune functions, and how might perinatal circadian disruption influence the immune system to contribute toward NDDs? At the other end of life, in aged individuals, a loss of circadian rhythmicity and increased pro-inflammatory responses characterize immunosenescence. Additional research on the extreme ages of life will help discern the importance of circadian rhythms in developmental processes and in neuroprotection.
The circadian system is central to human health and disease, and many pharmaceuticals target clock-controlled molecular mechanisms (Zhang et al., 2014). There is increasing awareness that timed dosing is an important consideration for treatments, such as cancer treatments (Dallmann et al., 2016). In addition, several pharmaceuticals have been developed that directly target the circadian system, including drugs that modulate the molecular clock (via RORs or REV-ERBs) as well as drugs that influence SCN phase, via melatonin or orexin receptors (Ruan et al., 2021). As our understanding of circadian neuroimmune mechanisms increases, circadian dosing or circadian modulators may be useful for the treatment of various neurological diseases.
Behavioral interventions that strengthen circadian rhythms are some of the most effective ways to protect cognitive and psychiatric health across all ages. Early morning light exposure (Tao et al., 2020), daytime exercise (Hughes et al., 2021), time-restricted diets (Balasubramanian et al., 2020; Hatori et al., 2012; Hu et al., 2019; Upadhyay et al., 2019), and proper sleep hygiene (Prince and Abel, 2013) are beneficial for combatting symptoms of depression, protecting against memory loss, and increasing longevity. The precise mechanisms connecting these behavioral interventions with the molecular clock and cognitive health are not fully known, and further investigation is needed into the role of the neuroimmune system in mediating these therapeutic strategies. In addition, it is unclear whether similar chronotherapies may support brain development early in life (Novakova et al., 2010; Prates et al., 2022) and ameliorate behavioral deficits in NDDs. Bright light therapy helps alleviate the symptoms of attention-deficit hyperactivity disorder (ADHD), and exercise is similarly beneficial for sleep and motor behaviors in children with autism (Brand et al., 2015; Turner and Johnson, 2013). Bolstering circadian rhythms early in life may help with immediate symptoms and could also ameliorate disorder trajectory. Early-life circadian reinforcement might lead to lasting effects on health, aging, and neurodegeneration.
Conclusions
The CNS directs a diverse repertoire of complex behaviors, including cognition, memory, personality, motor control, emotional regulation, and social interactions. As such, the rich social-emotional depth of the human lived experience relies on preserving the integrity of the CNS. Critically, the neuroimmune system maintains CNS homeostasis to optimize neural development and function throughout the lifespan. Neuroimmune cells such as microglia and astrocytes actively sculpt brain circuitry early in life, participating in axonal guidance, synaptogenesis, and spine pruning; in adulthood, the neuroimmune system facilitates learning and memory; and in aging, neuroimmune cells protect against the accumulation of misfolded proteins and neuronal death.
As this review highlights, neuroimmune responses to injury or illness are dynamic. Circadian regulation of neuroimmune activities is essential for protecting neural networks throughout the lifespan. On the other hand, circadian dysregulation may alter immune function at any age, disrupting CNS neuroimmune homeostasis, and thereby contributing to susceptibility to cognitive or psychiatric disorders. A growing body of evidence increasingly implicates circadian neuroimmune dysfunction in many neurodevelopmental and degenerative disorders—beginning before birth and extending throughout the lifespan. A deeper exploration of circadian neuroimmune functions across all ages will likely uncover novel avenues for treating a broad spectrum of human neurological conditions.
Footnotes
Acknowledgements
This work was supported by NIA grants R01AG078758 and R01AG062716 to L.K.F. and R01NS131806 to A.D.G. R.C. was supported by T32AA007471 and B.N.R. was supported by T32DA018926.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
