Abstract
The molecular mechanisms that maintain circadian rhythms in mammalian as well as non-mammalian systems are well documented in neuronal populations but comparatively understudied in glia. Glia are highly dynamic in form and function, and the circadian clock provides broad dynamic ranges for the maintenance of this homeostasis, thus glia are key to understanding the role of circadian biology in brain function. Here, we highlight the implications of the molecular circadian clock on the homeodynamic nature of glia, underscoring the current gap in understanding the role of the circadian system in oligodendroglia lineage cells and subsequent myelination. Through this perspective, we will focus on the intersection of circadian and glial biology and how it interfaces with global circadian rhythm maintenance associated with normative and aberrant brain function.
Circadian rhythms are intrinsic and self-sustaining periodical patterns in biological processes that allow organisms to anticipate dynamic environmental changes and maintain homeostatic control in response to daily physiological variations. Identified in living organisms ranging from bacteria to humans, this endogenous oscillating clock system regulates a broad range of physiological functions, including body temperature, metabolism, and rest-activity cycles, throughout each day (Honma, 2018). While circadian rhythms are sustained through transcriptional-translational feedback loops at the cellular level, higher vertebrates also have tissue-specific synchronicity (peripheral clocks) which are regulated through a combination of neural and hormonal signaling. The suprachiasmatic nucleus (SCN), known as the “primary clock,” is the coordinating control center of the brain that functions to synchronize other oscillating tissues (Hastings et al., 2018). Comprised of ~20,000 heterogenous neurons, the SCN’s powerful autonomous rhythmicity is maintained even when isolated (Green and Gillette, 1982; Yamaguchi et al., 2003). The instrumental significance of SCN control has been best demonstrated through lesion and ablation studies, which have resulted in altered phases of the heart, liver, kidney as well as other peripheral tissues (Husse et al., 2014; Kolbe et al., 2019).
Intrinsic Circadian Rhythmicity
Molecular Circadian Clock Machinery
Although global synchrony is essential for organismal homeostasis, intrinsic cellular rhythmicity maintained by the molecular clock is necessary for controlling the dynamic cellular function. The mammalian molecular circadian clock has a period of ~24 h and functions through a transcriptional-translational feedback loop where the basic-helix-loop-helix (bHLH) transcription factors BMAL1, CLOCK, and NPAS2 act as the core effectors (Takahashi, 2017). When BMAL1 heterodimerizes with CLOCK or NPAS2, it binds to E-box motifs in clock genes upregulating the expression of the negative factors Per1-3, Cry1, 2, and Rev-Erbα/β (Takahashi, 2017). PER1-2 and CRY1-2 accumulate during the daytime and then act as repressors during the second half of the circadian cycle (zeitgeber time [ZT] 12-24) by binding to a majority of the E-boxes that CLOCK: BMAL1 bind in a counterphase fashion, inhibiting their own transcription (Koike et al., 2012).
Bmal1 expression also oscillates in response to the auxiliary factors REV-ERBα/β and retinoid-related orphan receptors (RORα/β/γ). REV-ERBα, and its paralog REV-ERBβ, are ligand-regulated transcription factors of the nuclear receptor superfamily (Cho et al., 2012) that directly repress Bmal1 expression by binding to a common response element (RORE; Liu et al., 2008). Initially hypothesized to play a non-essential role in intracellular rhythmicity, later work recognized that REV-ERBα/β control a second loop of the core clock and are thereby critical for the generation and maintenance of circadian rhythms (Cho et al., 2012). The ubiquitous double knockdown of REV-ERBα/β in adult mice shows reduced and severely fragmented activity, with various features also found among Bmal1−/− mice (Bunger et al., 2000). Like REV-ERBα/β, RORα/β/γ are part of the auxiliary loop, enhancing Bmal1 transcription (Sato et al., 2004). Also important in the maintenance of circadian rhythms, RORα-deficient mice have aberrant locomotor activity and unstable rhythmicity due to reduced Bmal1 expression (Akashi and Takumi, 2005). While Bmal1 expression oscillates following a circadian pattern as a consequence of the auxiliary loop, Clock expression is relatively constitutive (Shearman et al., 2000; Preitner et al., 2002), making BMAL1 the main pacemaker effector of the molecular clock.
Clock-controlled Mechanisms of the Molecular Clock
During the last decade, advances in high-throughput sequencing techniques helped characterize BMAL1 regulation of several mouse tissues and cells (Rey et al., 2011; Koike et al., 2012; Menet et al., 2014; Trott and Menet, 2018; Beytebiere et al., 2019; Hatanaka et al., 2010; Oishi et al., 2017; Kitchen et al., 2020; Perelis et al., 2015; Mavroudis et al., 2018). These studies have shown that CLOCK: BMAL control is not only restricted to clock genes, but also binds to regulatory regions of thousands of clock-controlled genes and up to 43% of protein-coding genes cycle in at least one tissue in mice (Zhang R, et al., 2014). Moreover, BMAL1 also acts as a translational regulator independent of its role as a transcription factor (Lipton et al., 2015), a function that is much less studied. BMAL1 is necessary for proliferation and differentiation of neuronal progenitor cells (Malik et al., 2015), as it regulates its rhythmic cell-cycle entrance and exit by controlling the transcription of numerous cell-cycle factors (Bouchard-Cannon et al., 2013; Miller et al., 2007). The crosstalk between both cycles is evidenced in CLOCK: BMAL1 regulation of the expression of the G0/G1 checkpoint factor c-Myc (Fu et al., 2002), the G1 and G1/S checkpoint factor p21 (Gréchez-Cassiau et al., 2008), modulators of the G2/M checkpoint like Wee1 (Matsuo et al., 2003) and CyclinB1 (Farshadi et al., 2019), in addition to interactions with p53 (Mullenders et al., 2009; Jiang et al., 2016). The circadian clock and the cell cycle are, thus, tightly phase coupled and share the same oscillatory frequency (Bieler et al., 2014; Feillet et al., 2014). A similar degree of interplay between BMAL1 and metabolism has been described in tissues with central roles in glucose and lipid metabolic processes (as reviewed in Panda, 2016), emphasizing the broad control of BMAL1 in different biological functions outside of its direct role in circadian biology.
Clock Genes in the Intrinsic Control of Glia
The evolutionary importance of the molecular circadian clock to neuronal physiology and homeostatic function is well documented in numerous species from Drosophila to humans. The molecular clock is not restricted to neurons, and recent evidence has highlighted that it plays a fundamental and largely underappreciated role in glial biology. Microglia, astrocytes, and oligodendrocyte lineage cells are collectively referred to as glia. Traditionally considered to be the non-functional “glue” of the central nervous system (CNS), decades of research have shown the vital function of these cells in health and disease.
As undoubtedly important as neurons are to all facets of brain function, glia, which constitute the majority of the cells of the CNS, are increasingly recognized to not only be necessary for neural circuit formation, physiology, and plasticity, but also facilitate these processes through a broad range of dynamical functions. This feature of glial cell homeodynamic ability lends itself to being modulated in a circadian manner. Through this review, we aim to expand on the precise role of specific molecular clock components on glial function as well as the implications of glial circadian disruption in brain disorder.
Circadian Regulation of Astrocytes
Astrocytes (sometimes referred to as GFAP-expressing glia in older literature) provide a key regulatory role in the brain, including structural support between neurons and vasculature, and maintenance of the blood-brain barrier (Volterra and Meldolesi, 2005). Astrocytes are critical modulators of metabolites in the brain, releasing lactate to be used as an aerobic metabolite by neurons and supporting high neuronal activity by breaking down stored glycogen under hypoglycemic conditions (Deitmer et al., 2019). In addition, astrocytes are the major site of lipoprotein-like particles and cholesterol synthesis in the brain, aiding in oligodendroglial myelination (Camargo et al., 2017). Astrocytes contribute to both intercellular communication through neurotransmitter clearance in response to neuronal activity as well as maintenance of pH homeostasis in the brain by expressing transporters and pH modulators (Deitmer et al., 2019).
Astrocyte Reactivity
The first evidence of a molecular clock in the mammalian brain outside neurons and neural stem cells comes from in vitro cultures of mouse cortical GFAP-expressing astrocytes, showing rhythmic bioluminescence under the control of clock genes that can be entrained independently of neurons (Prolo et al., 2005). This rhythmicity can be sustained by co-culturing astrocytes with SCN explants (Prolo et al., 2005) and inhibited by disrupting Bmal1, Clock, Npas2, or Per1-2 (Marpegan et al., 2011). Later work subsequently demonstrated that Bmal1 is also expressed in the cerebral cortex of mice in vivo (Musiek et al., 2013). Through the use of a mouse model that expresses Cre recombinase under the control of the Nestin promoter, they assessed the effects of Bmal1 knockdown on glial populations relative to global Bmal1 deletion in mice, focusing on its effect on astrocyte reactivity. Reduced BMAL1 in the brain was associated with age-dependent astrogliosis, increased oxidative stress, and chronic neuroinflammation. While this study involved a neural-specific Bmal1 knockout, it highlighted the circadian nature of astrocytic behavior, especially through the expression of key genes of the oxidative stress defense pathway. Relatedly, Nqo1 and Aldh2 redox genes, which have been implicated in neurodegeneration and are highly expressed in astrocytes, carry E-boxes which are transcriptionally regulated by BMAL1, confirming a mechanism of BMAL1 control of redox homeostasis in this glial population (Musiek et al., 2013). Follow-up work determined that while a small component of the astrogliosis appears to be non-cell-autonomously related to BMAL1 control of neurons, as evidenced by deleting Bmal1 selectively from neurons versus Aldh1l1+ astrocytes, astrocyte activation and inflammatory gene expression depends on a cell-autonomous pathway regulated by BMAL1 (Lananna et al., 2018). The transcriptional profile of Bmal1 knockout astrocytes shows the upregulation of transcripts that encode key factors involved in astrocyte activation (Gfap, Il6, and Il33), astrocyte-mediated synaptic elimination (C4b and Megf10), inflammatory mediators (Cxcl5 and Ccr7), and metalloproteinases (Mmp14 and Mmp2), confirming an anti-inflammatory effect of the molecular clock in astrocytes. Subsequently, as RORα expression and modulation were demonstrated in astrocytes, it was found to directly regulate astrocytic inflammation via the inhibition of the NF-κB pathway and control IL-6 expression (Journiac et al., 2009). In vitro astrocyte experiments further show that RORα is naturally upregulated by hypoxia and has a neuroprotective function through the downregulation of hypoxic inducible factor 1α (Jolly et al., 2011). This mechanism was implicated in the cerebellar degeneration reported among RORα-deficient mice (Jolly et al., 2012). The lack of Rev-Erbα in the brain also induces the astrocytic inflammatory response in vivo but, as opposed to the effect produced by Bmal1 deletion, it does so in a non-cell-autonomous manner (Griffin et al., 2019b). Counter to this, the global deletion of Per1 and Per2 does not induce astrogliosis in the cortex (Musiek et al., 2013), even though PER1 controls the production of pro-inflammatory cytokines in isolated spinal astrocytes (Sugimoto et al., 2014), challenging our understanding of circadian versus pleiotropic effects of BMAL1 on astrogliosis.
Astrocyte Morphology
Much of astrocytes’ ability to control CNS homeostasis depends on their dynamical morphological plasticity (Schiweck et al., 2018). A recent study shows that BMAL1 regulates the morphology of astrocytes, including the number, thickness and length of distal processes, and the number of branching bifurcations (Ali et al., 2020). Even prior to the onset of astrocytosis, the deficiency of Bmal1 in astrocytes of young mice leads to subtle morphological changes evidenced in soma diameter and surface area. These changes are enough to affect astrocytic processes involved in hippocampal mossy fiber synapses that are necessary for learning and memory formation. The disorganization of actin filaments is associated with a decrease in the actin-binding protein CTTN and the cytoskeleton regulator Rho-GTP, that is subsequently controlled by CTTN. CTTN is known to be transcriptionally regulated by BMAL1 (Ali et al., 2020). This highlights the importance of the molecular clock in modulating the changes in astrocyte morphology necessary for their homeodynamic plasticity.
Astrocyte Neuromodulatory Function
A series of studies have further shown that circadian rhythms play a regulatory role on astrocyte neuromodulatory function. Adenosine triphosphate (ATP) levels, necessary for astrocyte-astrocyte and astrocyte-neuron communication, oscillate over a 24-h cycle (Womac et al., 2009; Marpegan et al., 2011). The molecular clock upregulates extracellular ATP release through CLOCK, BMAL1, and PER, indicating the circadian control of neuromodulation within glia. In addition, the molecular clock contributes to glutamate homeostasis in the brain as it modulates Glutamate Aspartate Transporter 1 (Slc1a3), a transcript that is restricted to astrocytes (Spanagel et al., 2005), in an oscillatory manner. Per2 Brdm1 mutant mice that lack a functional protein-protein interaction PAS domain in all cells express less Slc1a3 and have deficient glutamate uptake. As this transporter is responsible for glutamate clearance in the synaptic gap, this deficiency leads to a hyper-glutamatergic brain (Spanagel et al., 2005). In accordance, Slc1a3 expression and glutamate uptake are also reduced in cortical astrocytes that lack functional Clock, Npas2, or Per2 (Beaulé et al., 2009), reinforcing the broad ethos of circadian control of astrocyte neuromodulation. Similarly, the molecular clock of astrocytes regulates cortical gamma aminobutyric acid (GABA) signaling by enhancing the rhythmic expression of the GABA transporters Gat1 and Gat3 (Barca-Mayo et al., 2017). Glutamate and GABA levels in the arcuate nucleus of the hypothalamus are decreased following astrocytic deletion of BMAL1, resulting in a severe disruption of energy homeostasis which can be rescued by GABA receptor inactivation (Barca-Mayo et al., 2020). Complementary studies show that PER2 regulates GABA, glutamate, and dopamine signaling in the nucleus accumbens and hypothalamus by controlling cyclic gene expression of the GABA transporters Gat1 and Gat2, dopamine receptor Drd3, and glutamate levels (Martini et al., 2021). The deletion of Per2 from GFAP+ cells using either Per2fl/fl and GFAP:: Cre mice or a GFAP-driven adenovirus strategy leads to reduced anxiety and despair (Martini et al., 2021). However, this effect is independent of the nucleus accumbens astrocytic BMAL1, as its deletion does not alter Gat2 expression, reinforcing the importance of studying multiple clock genes to determine circadian and non-circadian roles of clock genes. A more comprehensive analysis of these molecular regulators and their varied circadian and non-circadian determinants is needed to fully elucidate the biological importance of the molecular clock in astrocyte neuromodulation.
Astrocytic Contributions to Global Circadian Rhythmicity
Of particular interest has been the expression of the molecular clock among astrocytes in the SCN and their role in maintaining global biological rhythms in mice, as astrocytes are abundant in this pacemaker region (Lavialle and Servière, 1993; Santos et al., 2005). Approximately 60% to 80% of the mouse SCN astrocytes express Bmal1 (Barca-Mayo et al., 2017; Tso et al., 2017). As virtually every neuron in the SCN is GABAergic, neuronal activity and maintenance of circadian rhythms in the SCN rely on tight control of GABA extracellular clearance (Albers et al., 2017). The loss of BMAL1 in astrocytes during adulthood dysregulates GABA homeostasis and affects VIP neuropeptide expression in the SCN (Barca-Mayo et al., 2017). These deficits prevent neuronal entrainment and alter clock-genes rhythmicity, leading to changes in locomotor activity and memory. Using 2 alternative approaches to target BMAL1 expression only in SCN astrocytes, Herzog and colleagues showed a similar and complementary result that BMAL1 is necessary among SCN astrocytes to maintain the length of circadian rhythms and proper locomotor periodicity (Tso et al., 2017). During the dark phase, SCN astrocytes suppress the activity of the mainly GABAergic SCN neurons by releasing glutamate into the extracellular space, which depends on the astrocyte-specific expression of glutamine synthase (Brancaccio et al., 2017). Moreover, in the absence of Cry1-2 expression in neurons, astrocyte clocks alone can autonomously initiate and modulate self-sustained circadian oscillations in the SCN (Brancaccio et al., 2019). However, astrocytes alone cannot influence SCN phase, as this is only regulated by neurons (Patton et al., 2022). Notably, as opposed to eliminating Bmal1, the lack of Per2 only in GFAP+ cells does not alter circadian period or locomotor activity (Martini et al., 2021), further emphasizing the importance of extra-circadian signaling roles of these clock genes. Whether BMAL1 regulatory effects on SCN astrocytes is fully circadian-dependent is still an open question. Taken together, these findings (Table 1) suggest a bidirectional interaction between astrocytes and the circadian system, with astrocytes directly and indirectly modulating circadian rhythmicity and the circadian molecular machinery regulating astrocytic form and function (Figure 1).
Summary of clock gene control in glial cells.
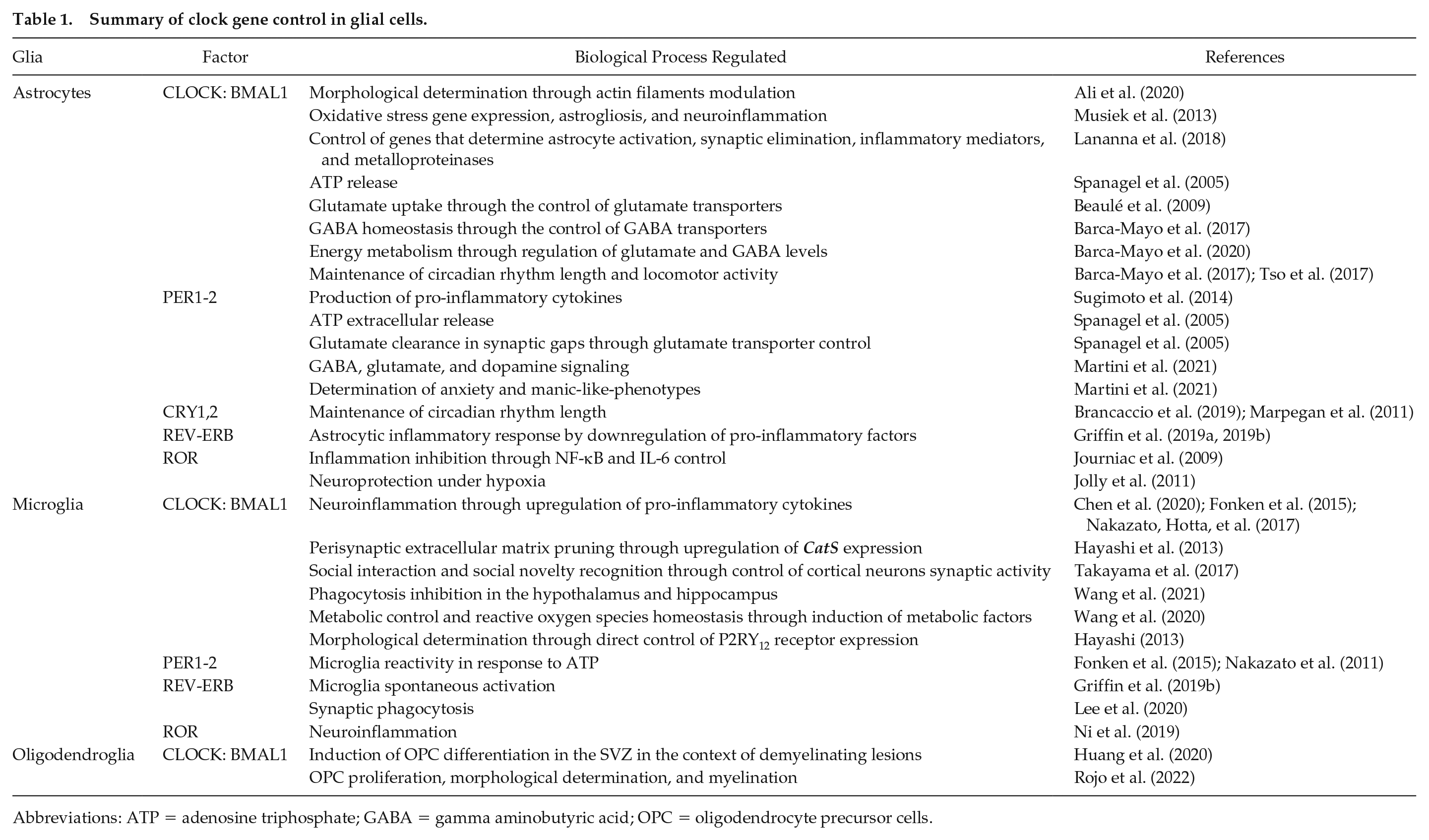
Abbreviations: ATP = adenosine triphosphate; GABA = gamma aminobutyric acid; OPC = oligodendrocyte precursor cells.
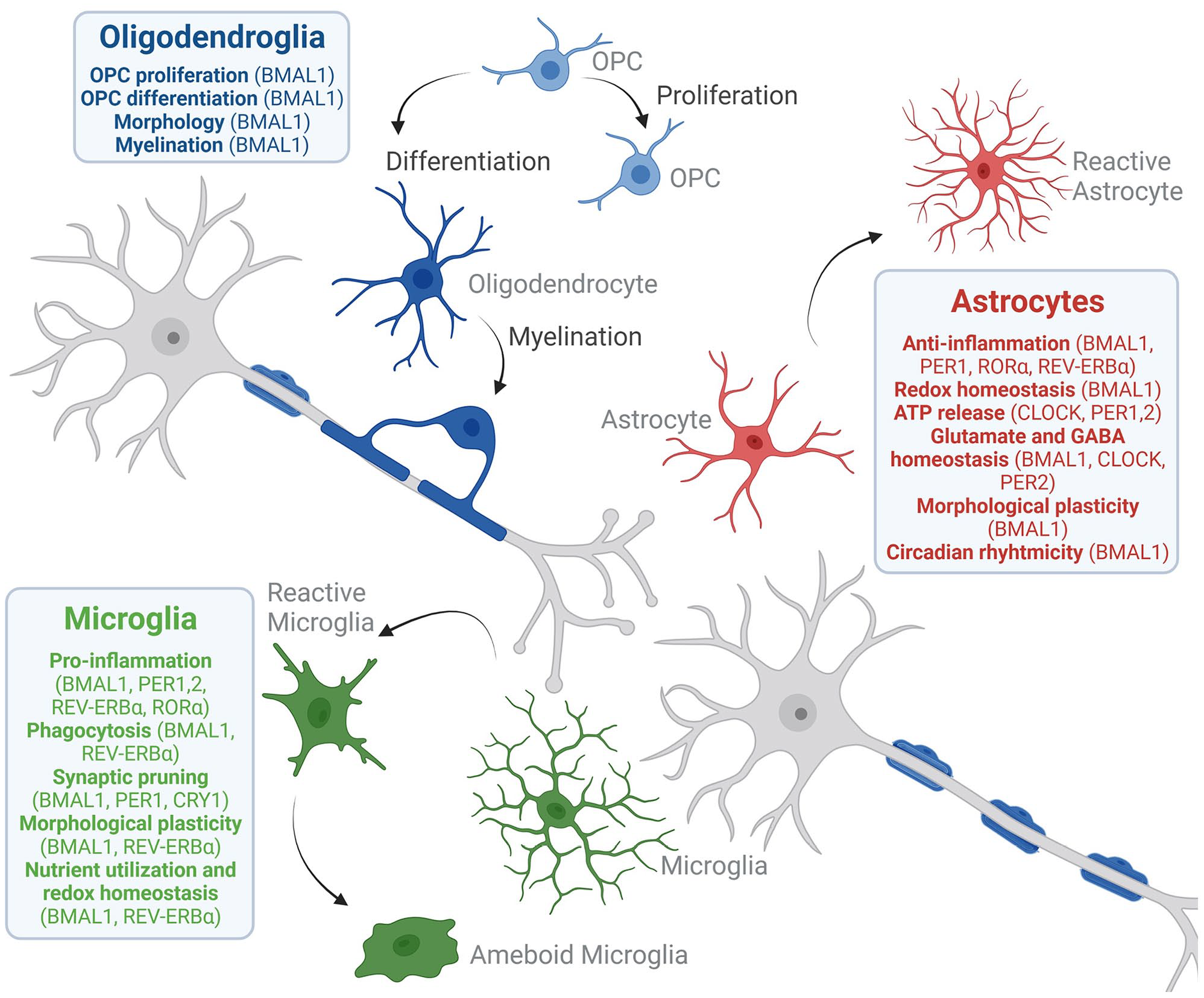
The circadian clock participates in maintaining the homeostasis of dynamic functions in glia populations. Circadian rhythms, acting through BMAL1, PER1-2, CRY1,2, RORα,β, and REV-ERBα,β factors, control not only the circadian period length but also the expression of genes that participate in diverse glial functions. In astrocytes, microglia, and OPCs, the molecular clock modulates morphological factors necessary for their highly dynamic processes. It also regulates glucose metabolism and redox homeostasis in astrocytes and microglia through the enhancement of expression of glucose transporters, metabolic enzymes, and anti-oxidant factors. Circadian rhythms also promote synaptic plasticity by shaping glutamate and GABA transporters in astrocytes and several factors necessary for synaptic pruning and phagocytosis in microglia. Moreover, most of the circadian factors are associated with a modulatory role in inflammatory processes by controlling leukins, metalloproteinases and inflammatory mediators, and modulating astrocyte and microglia activation. Even though the role of the circadian clock in oligodendroglial lineage cells, including OPCs and oligodendrocytes, remains to be fully elucidated, evidence points toward a circadian regulation of the homeostasis between proliferation and differentiation, as well as myelination. Figure created in BioRender. Abbreviations: ATP = adenosine triphosphate; GABA = gamma aminobutyric acid; OPC = oligodendrocyte precursor cell.
Circadian Regulation of Microglia
Despite the evidence of circadian regulation of gene expression in the immune system (Hergenhan et al., 2020), it was not until recently that the interplay between the molecular clock and microglia, the primary immune-modulating and phagocytic cells in the CNS, has begun to be elucidated. The expression of clock genes in microglia was first described in the murine BV-2 cell line as well as primary microglia cultures from neonatal mice (Nakazato et al., 2011) and adult mouse and rat brains (Hayashi et al., 2013; Fonken et al., 2015). Cortical microglia exhibit rhythmic expression of clock genes that depend on CLOCK: BMAL1, where rhythmicity is abolished in mice that lack Clock (Hayashi et al., 2013). Furthermore, microglia isolated from adult rat hippocampi rhythmically express the clock genes Bmal1, Per1, Per2, and Rev-Erb, whereas Clock is constitutively expressed throughout the circadian day (Fonken et al., 2015).
Microglial Reactivity
The initial studies on the circadian regulation of microglia showed that when microglia are exposed to exogenous stress signals like glucocorticoids or ATP, the expression of microglial Per1 and Per2 increases in vitro (Nakazato et al., 2011; Fonken et al., 2015), potentially acting as modulators of microglial reactivity. Knock down of Per1 inhibits the recovery of microglia to a resting-state morphology, and they remain in an activated ameboid-like shape (Nakazato et al., 2011). In agreement, microglia isolated during the light phase respond to the pro-inflammatory lipopolysaccharide (LPS) stimulation producing more cytokines compared with microglia isolated during the dark phase, when Per1-2 expression peaks (Fonken et al., 2015). Expression of the pro-inflammatory cytokines IL-6, IL1β, and TNFα oscillate across the circadian day, reaching their peaks during the light phase, when Bmal1 is at its highest levels (Fonken et al., 2015). This depends on an intrinsic direct control of BMAL1 on IL-6 expression through E-box binding (Nakazato et al., 2017a). Bmal1 overexpression doubles IL-6 release after LPS exposure in vitro, while its knock down limits the LPS-induced IL-6 expression and release. BMAL1 also directly activates the expression of the pro-inflammatory chemokine RANTES/CCL5 after LPS induction by binding to a regulatory element in its promoter (Chen et al., 2020). As the dysregulation of the microglial molecular clock impairs the upregulation of IL-6 and CCL5 pro-inflammatory factors in vivo, it attenuates neuronal damage after a severe brain inflammatory event (Nakazato et al., 2017a) and prevents the depressive behavior induced by the immune-inflammatory reaction caused by CCL5 (Chen et al., 2020).
Microglial Phagocytic and Pruning Functions
In the context of REV-ERB regulation of microglia, the Musiek lab has been a leader in the field, demonstrating that REV-ERBα directly binds to promoters of NF-kB-related genes repressing their transcription and regulating basal microglia activation throughout the day. The global deletion of REV-ERBα causes spontaneous microglial activation and increased phagocytic function, whereas its pharmacological activation using SR9011 reduces LPS-induced neuroinflammation (Griffin et al., 2019b). Microglial phagocytosis is antiphase to REV-ERBα expression, and REV-ERBα deletion causes persistently elevated phagocytotic behavior (Griffin et al., 2020). In accordance, REV-ERBα activation not only attenuates microglia pro-inflammatory response by downregulating the increase in pro-inflammatory cytokines expression in response to TNFα, but also decreases their phagocytic function and upregulates the anti-inflammatory cytokine Il10 expression (Wang et al., 2020). In contrast, limited work has assessed RORα in microglia, as some literature has even suggested an absence of RORα expression among this cell population (Chi-Castañeda and Ortega, 2017). However, a more recent study exploring microglia from an Alzheimer’s disease (AD) mouse model found an inflammatory microglial phenotype associated with a reduction in RORα, which in turn reduces IκBα and enhanced NF-κB activation (Ni et al., 2019). RORα likely has a direct immunoregulatory role in microglia that remains to be fully elucidated.
Given that microglia are now recognized to prune synapses to refine neural circuits and drive neural plasticity during postnatal development (Paolicelli et al., 2011; Schafer et al., 2012), there has been growing interest in the temporal rhythmicity of this behavior. Synaptic pruning depends on a tight regulation of different molecular signals. Both during normal neurodevelopment and in response to brain injury, cortical microglia play a critical role in synapse pruning through pre- and post-synaptic engulfment and remodeling perineural nets (Paolicelli et al., 2011; Schafer et al., 2012; Filipello et al., 2018; Andoh et al., 2019; Pantazopoulos et al., 2020; Mehl et al., 2022). One factor responsible for homeostatic maintenance of the perineural environment is the microglia-specific protease Cathepsin S (CatS) that cycles along the circadian day peaking early in the dark phase (Hayashi et al., 2013). The mechanism through which CLOCK: BMAL1 modulates CatS depends on the transactivation of at least 2 E-boxes on its promoter region that can be repressed by PER1 and CRY1. As evidenced by mice that lack functional CatS or Clock genes, the circadian modulation of the perisynaptic extracellular matrix pruning by CatS is necessary for refining diurnal variations in synaptic activity, subsequently leading to a proper time-of-day-dependent locomotor activity (Hayashi et al., 2013). The molecular clock in microglia, acting through CatS, also controls social interaction and social novelty recognition by affecting synaptic activity of cortical neurons (Takayama et al., 2017). Moreover, BMAL1 in microglia participates in modulating a normative hippocampal and hypothalamic neural microenvironment, necessary for critical cognitive functions like memory and regulation of energy homeostasis, respectively. Through the generation of a mouse model with a microglia-specific Bmal1 deletion using an inducible Cre recombinase under the control of the Cx3cr1 promoter, Wang and colleagues demonstrate that BMAL1 inhibits microglia phagocytosis in 2 brain regions associated with adaptive responses (Wang et al., 2021). In the hypothalamus, the lack of expression of Bmal1 in microglia was reflected by an increase in microglia phagosomes and proopiomelanocortin (POMC) neuron density within the arcuate nucleus. Microglial phagocytosis promotes POMC neuronal survival under hypercaloric stress (Gao et al., 2017). As POMC neurons regulate energy homeostasis and inhibit food intake, mice that do not express Bmal1 in microglia and have more POMC neurons are resistant to high-fat diet-induced weight gain by decreasing their food intake (Wang et al., 2021). Through a similar phagocytic mechanism, memory formation and consolidation in the hippocampus and cognitive flexibility are improved by the lack of microglial Bmal1 (Wang et al., 2021). These results suggest that BMAL1 controls microglia phagocytic capacity and, consequently, synaptic pruning in response to brain challenges. Moreover, it reinforces previous evidence of a cell-autonomous circadian control of microglia. However, neuronal activity has also been shown to induce microglial synaptic pruning (Faust et al., 2021). Since neuronal activity oscillates throughout the circadian day, it may potentially influence microglial circadian behavior in a non-cell-autonomous fashion. Future work will be necessary to parse out the complex cell-cell interactions that could be driving 24-h oscillation in microglial behavior.
Microglial Morphology
The activity level of microglia has been associated to its morphological complexity as well, which changes across the circadian cycle (Hayashi, 2013). At night, during the murine active phase, cortical microglia have longer processes and higher number of branches that are associated with an increase in wrapping of dendritic spines. The expression of the microglia-specific receptor P2RY12, that is involved in morphological determination, is transcriptionally activated by the binding of CLOCK: BMAL1 to a canonical E-box on its promoter and peaks at night (Hayashi, 2013). Time-of-day microglial volume changes are prevented by Rev-Erbα deletion (Griffin et al., 2019b). As opposed to cortical microglia, the morphological complexity of hippocampal microglia decreases at night (Griffin et al., 2019b; Wang et al., 2021), a difference that could potentially be attributed to spatial heterogeneity. Whether these changes reflect alterations to microglial function in relation to synaptic remodeling and phagocytic capacity throughout the circadian day remains to be fully determined.
Microglial Metabolism
Similar to astrocytes, the molecular clock is also a key modulator of microglial metabolism and anti-oxidative stress in mice. The glucose transporter Glut5 and Lipoprotein lipase genes exhibit elevated transcription at night, when mice and microglia are more active. This rhythmicity depends on BMAL1 (Wang et al., 2020). Moreover, the expression of anti-oxidant factors glutathione reductase and heme oxygenase 1, which are necessary for eliminating the reactive oxygen species generated after microglia activation, is also induced during the dark phase (Wang et al., 2020). REV-ERBα activation inhibits mitochondrial respiration by repressing the expression of metabolic genes (Wolff et al., 2020). Altogether, the molecular clock regulates microglial nutrient utilization and redox homeostasis by inducing gene expression during its most active phase but whether it is fully circadian-dependent requires further investigation. These findings (Table 1) support a modulatory role for molecular clock genes in microglia that fosters their homeodynamic ability (Figure 1) to respond to changes in their microenvironment and influence cognition, physiology, and behavior.
The Interplay of Circadian and Oligodendroglial Biology
Despite the established role for molecular circadian control of neuronal and glial populations in the mammalian brain, oligodendroglial lineage cells remain understudied relative to other glial populations. This lineage encompasses myelinating oligodendrocytes as well as their associated heterogenous precursor population, oligodendrocyte precursor cells (OPCs). Myelin, the multilaminar structure that promotes efficient impulse conduction and metabolic support of axons, is essential to the coordination of neural network signal timing, and ultimately, to circuit function. Nearly half of the human brain is composed of white matter tracts that consist of these myelinated axons in which new myelin-forming oligodendrocyte production from OPCs is critical to neurodevelopment, maintenance, and plasticity (Kuhn et al., 2019; Gibson et al., 2018). As OPCs are uniformly distributed throughout the gray and white matter of the adult CNS, other additional roles have been suggested for this population, including a putative neuroinflammatory role (Fernández-Castañeda et al., 2020). OPCs have also been found to make synaptic junctions with neurons (Bergles et al., 2000) and may be involved in neural signaling (Lin and Bergles, 2004).
OPCs are the most consistently mitotic cells in the brain (Dawson et al., 2003; Matsumoto et al., 2011) and are remarkable at maintaining their population-level homeostasis (Hughes et al., 2013; Rosenberg et al., 2008). The balance between self-renewal and sustained differentiation into oligodendrocytes is indicative of both precision in oligodendroglia dynamics and distribution homeostasis despite an ever-changing microenvironment, a function that could be potentially modulated by circadian rhythms given the evidence in other cell populations. Notwithstanding the known importance of myelination in system-wide neural circuit function, the intrinsic circadian clock control of OPCs, myelin-forming cells, and myelination is quite understudied compared with the circadian regulation of astrocytes and microglia. To date, numerous components of the transcriptional molecular clock have been identified throughout the oligodendroglial lineage in both mice and humans (Zhang Y, et al., 2014, 2016; Rojo et al., 2022), which has been confirmed at the proteomic level with PER2 staining in oligodendrocytes (Colwell and Ghiani, 2020) and BMAL1 staining in OPCs (Rojo et al., 2022). In addition, isolated OPCs show rhythmic expression of Bmal1, Per2, and Rev-Erbα that is disrupted by the lack of Bmal1 (Rojo et al., 2022).
A series of studies have suggested that OPCs and mature oligodendrocytes have functional clocks that regulate oligodendroglia function. The OPC and myelin genes Pdgfrα, Mbp, and Mog cycle throughout the day in mice (Colwell and Ghiani, 2020). Furthermore, several oligodendroglial-specific transcripts oscillate over the sleep-wake cycle in mice and are differentially expressed during the wake phase (Bellesi et al., 2013). The level of expression of 2% of all genes expressed in oligodendrocyte lineage cells vary between ZT7 and ZT20 because of circadian factors, independent of sleep-wake states. During the rest phase, oligodendroglia display upregulated expression of genes involved in phospholipid metabolism, proliferation, and myelination. For example, Pllp and Opalin, which are involved in myelin biogenesis and stabilization, are upregulated during the rest phase. On the contrary, expression of genes linked to differentiation, cytoskeleton, stress response, and apoptosis are increased during the wake phase. Sox17, a gene that is involved in regulating the transition from OPC to immature oligodendrocytes by controlling Notch signaling (Chew et al., 2019), is upregulated during the wake phase (Bellesi et al., 2013). Consequently, OPC proliferation oscillates following a circadian pattern, as it doubles during the light phase (Matsumoto et al., 2011). It was not until recently that the functional effect of the molecular clock in oligodendroglia was corroborated by eliminating Bmal1 from OPCs, showing that the circadian rhythms participate in oligodendroglia population homeostasis by regulating OPC proliferation and allowing proper myelination (Rojo et al., 2022). In line with previous evidence showing time-of-day regulation of OPC proliferation (Matsumoto et al., 2011), BMAL1 controls the expression of genes associated with OPC proliferation, cell-cycle regulation, and morphological complexity (Rojo et al., 2022) (Figure 2).
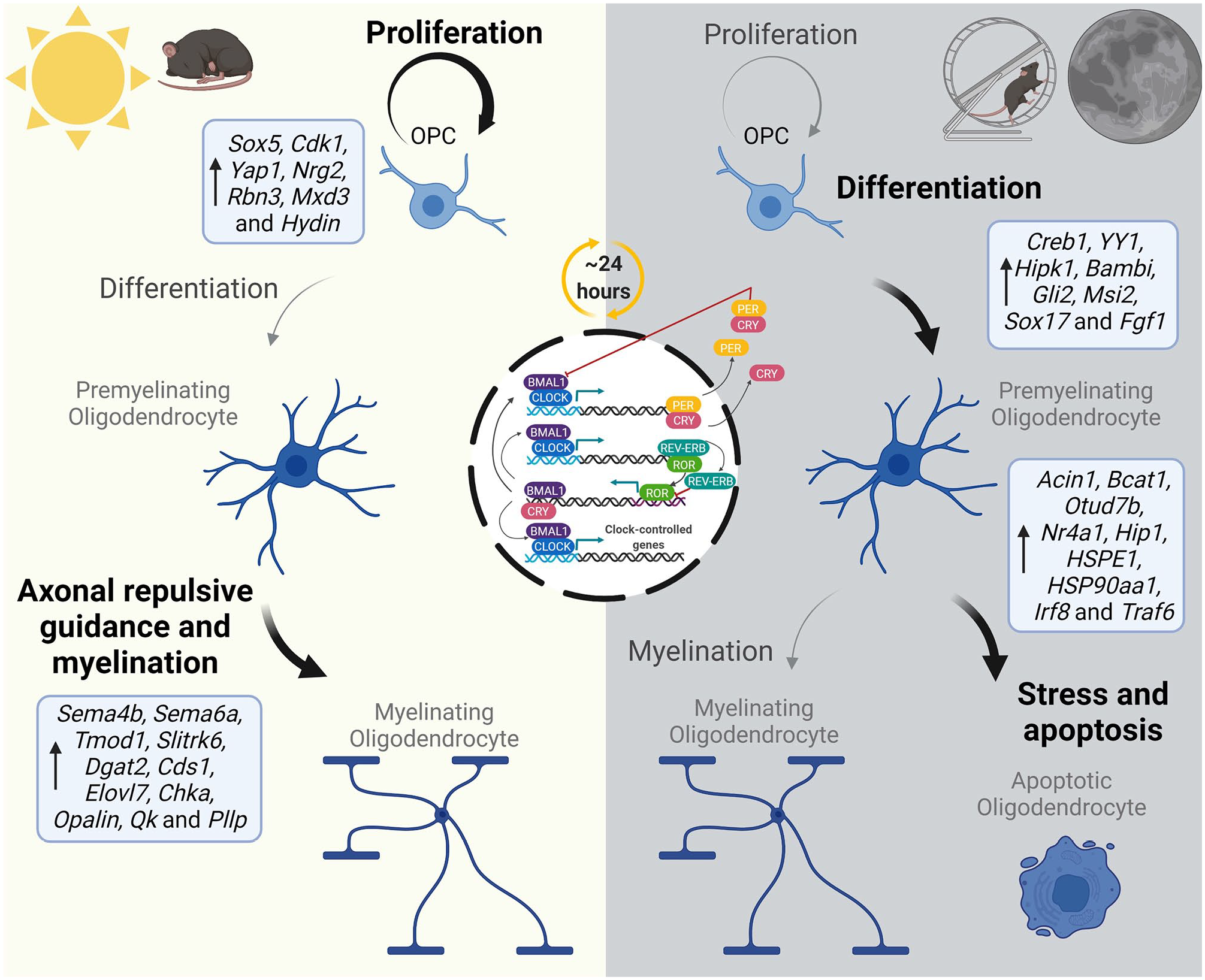
Oligodendroglia homeostatic density and myelination is affected by daily circadian variations. Circadian rhythms are generated ubiquitously by a molecular transcriptional/translational negative feedback loop that has a period of ~24 h. The products of the core clock genes Clock (blue) and Bmal1 (purple) heterodimerize and bind to DNA at E-boxes of clock genes (light blue). CLOCK: BMAL1 drives the transcription of the clock genes Period (Per; yellow) and Cryptochrome (Cry; pink). Accumulated levels of PER and CRY within the cytoplasm dimerize and feed back into the nucleus, suppressing CLOCK and BMAL1 function, and consequently their own transcription. CLOCK: BMAL1 also modulates the expression of REV-ERB (cyan) and ROR (green), that bind to RORE elements (purple) and inhibit or induce BMAL1, respectively. A complete intrinsic molecular clock is present in oligodendroglia with rhythmic levels of expression across the circadian day (Zhang Y, et al., 2014; Rojo et al., 2022). In mice, the level of expression of genes that control OPC proliferation and differentiation, myelination, and oligodendroglia apoptosis vary between the rest and wake phases (Bellesi et al., 2013). During the rest phase, genes related to OPC proliferation and myelination are upregulated. This is reflected in a net increase of OPC proliferation during the day in vivo (Matsumoto et al., 2011). Counter to this, genes that control OPC differentiation and cellular stress, and apoptosis are upregulated during the wake-dark phase (Bellesi et al., 2013). One of the main OPC proliferation regulators, Sox5, is controlled by BMAL1 and its disruption leads to decreased Sox5 expression during the light phase (Rojo et al., 2022). Consequently, the disruption of the clock leads to a decrease in OPC proliferation and density and myelination (Rojo et al., 2022). Collectively, this daily variation points toward evidence of circadian-mediated oligodendroglial density maintenance. Figure created in BioRender. Abbreviation: OPC = oligodendrocyte precursor cell. Color version of the figure is available online.
Circadian clock machinery in other glial cells may also indirectly influence myelin dynamics, highlighting the highly interactive nature of glial subpopulations. BMAL1 in astrocytes drives subventricular zone (SVZ) neural precursor cells toward the oligodendrocyte lineage to stimulate remyelination of demyelinating lesions (Huang et al., 2020). This work suggests that OPCs can be indirectly regulated by extrinsic circadian clocks from other glial populations. However, it contradicts previous evidence found in humans (Lavtar et al., 2018) as it suggests that Bmal1 depletion could be beneficial in a demyelinating lesion. Given the recent findings that the molecular clock in oligodendroglia influences homeostasis and remyelination (Rojo et al., 2022), it is intriguing to speculate that the high prevalence of demyelinating disorders with increased latitude (Simpson et al., 2019) and in night shift workers (Hedström et al., 2011) could be related to changes in the interactions between circadian and myelin biology. One of the many changes that accompany geographic latitudes approaching the poles is variations in the light-dark cycle with substantial portions of the year experiencing light cycles ranging from continual light to continual darkness, 2 environmental states known to cause dysregulation in the circadian system. Whether this influences the prevalence of diseases associated with aberrant myelination remains to be determined. Taken together, these findings (Table 1) lay the foundation for circadian regulation of oligodendroglial lineage cells and open new opportunities to uncover novel molecular mechanisms modulating oligodendroglial biology in normative and aberrant brain function.
Neurodegenerative Diseases
Circadian clock machinery plays an important role in normative brain homeostasis (Figure 1) and may contribute to brain disease. The earliest evidence of this relationship stems from correlative reports of both sleep and circadian disturbances among individuals with neurodegenerative disease. Altered rhythmicity has been reported among individuals with AD (Hatfield et al., 2004; Witting et al., 1990), Parkinson’s disease (PD; Niwa et al., 2011), Huntington disease (Aziz et al., 2010; Morton et al., 2005), and multiple sclerosis (MS; Melamud et al., 2012). Moreover, circadian dysregulation is known to increase with aging (Nakamura et al., 2011; Wyse and Coogan, 2010). The risk of neurodegenerative diseases increases with aging as well (Tranah et al., 2011) in part due to accumulated damage and insufficient repair of neural cell populations, especially progenitor populations. In particular, deficits in OPC density and differentiation are underlying causes for the limited remyelination in MS (Kuhlmann et al., 2008). While traditionally not viewed as a myelin disorder, myelin deficits are also a common feature in AD (Bouhrara et al., 2018; Nasrabady et al., 2018). Compared with traditional demyelinating disorders like MS, little is known about the role of OPC and myelin homeostasis in the onset or progression of AD but both clinical populations (Nasrabady et al., 2018) and experimental animal models (Desai et al., 2010; Vanzulli et al., 2020) suggest conserved mechanisms of dysmyelination in AD. How the circadian system may interact with myelin biology in disease settings such as MS and AD remain an open area of investigation. Although the implications are robust, direct interplays between circadian and glial biology in disease settings are only now gaining traction. Some of the primary mechanisms reported to date that directly link circadian disruption among glia with disease include oxidative stress, impaired metabolism, inflammation and altered protein aggregation (Musiek and Holtzman, 2016), with the caveat that these pathways are largely interrelated and limited by a focus restricted to astrocytes and microglia. Nonetheless, an altered molecular clock among microglia has been predominantly linked to neuroinflammation (Wolff et al., 2020; Nakazato et al., 2017a) and metabolic stress (Wang et al., 2020), reminiscent of AD (Lee et al., 2020) as well as PD (Liu et al., 2020) pathophysiology. Similarly, molecular clock disruptions in astrocytes have been reported to induce impaired aerobic glycolysis (Lananna et al., 2018; Yoo et al., 2020), astrogliosis (Musiek et al., 2013), and reduced blood-brain barrier integrity (Nakazato et al., 2017b), all of which have been implicated in various neurodegenerative conditions (McKee et al., 2020). While more work is necessary to understand if interactions between circadian and glial biology and their relationship to disease is causal or merely correlative, this field may encourage the development of circadian-centric therapeutic strategies that could capitalize on this biology. For example, pharmacologically targeting REV-ERBα/β activity could be potentially exploited to mitigate neuroinflammation and amyloid-β plaque clearance (Griffin et al., 2019a; Lee et al., 2020). However, a more comprehensive understanding of the pleiotropic effects of administering circadian modulators is needed prior to administering these molecules as potential therapeutic strategies to moderate neurodegenerative disorders.
Conclusion
A commonality among all glial populations is the ability to interact with other cells to foster dynamic responses to microenvironmental changes. This dynamic ability is imperative to the maintenance of short-term brain homeostasis and to create long-term neural stability. Understanding the homeodynamic nature of glial cells is essential to the study of brain circuit development, aging, and disease. Even though glia represent around half of the cells in the mammalian brain, they have been historically understudied compared with neurons across neuroscience subfields, including circadian biology. Despite the increasing amount of evidence supporting intrinsic circadian control of glia, extrinsic rhythmic signals, such as diurnal rhythms in neuronal activity, may also initiate oscillatory rhythms in a non-cell-autonomous manner. Neurons and neuronal activity reciprocally regulate microglia (Faust et al., 2021; Schafer et al., 2012), astrocytes (Tan et al., 2021), and oligodendroglial lineage cells (Gibson et al., 2014; Monje and Káradóttir, 2021). Even if a specific glial function is not regulated by the cell-autonomous molecular clock, it does not rule out the possibility of it being influenced by exogenous 24-h rhythms. This highlights the importance of studying circadian regulation of glia as the sum of the contributions of different cell populations. A shared challenge to the circadian field is to distinguish rhythmic versus non-rhythmic functions of the molecular clock, a constraint that is similarly important in glial biology. Many of the studies on glial circadian regulation focus on the effects produced by the ablation of Bmal1 that could potentially lead to pleiotropic non-circadian effects. Moreover, as most of the studies take advantage of mouse models that lack the BMAL1 DNA-binding domain, the effects produced can be attributed to transcriptional regulation but do not typically account for circadian post-translational effects. Future studies will need to address these questions in glial subpopulations. Elucidating the role of the molecular clock across glial cell subtypes will impart unique insights into normative and aberrant brain activity. Consequently, understanding the intersection of circadian and glial biology may produce novel therapeutic strategies for the numerous neurodegenerative brain disorders that are increasingly recognized to exhibit both glial and circadian abnormalities.
Footnotes
Acknowledgements
This work was supported by the Department of Defense (W81XWH-21-1-0846); the National Multiple Sclerosis Society (PP-1907-34759); National Institutes of Health (R21CA267135); The Weintz Family COVID-19 Research Fund; Stanford University School of Medicine Department of Psychiatry & Behavioral Sciences 2021 Innovator Grant Program.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
