Abstract
Under conditions of prolonged durations of warmth, flies counter potential temperature stress by shifting their locomotor activity from day into night when the conditions are likely to be less harsh. Modulation of a rhythmic behavior such as this in response to the environment would require interaction between at least 2 neuronal systems: (1) a sensory system to receive input from the environment, and (2) the internal clock to correctly time rhythmic activity in response to this thermosensory input. Our previous studies found that a thermosensory mutant of the ion channel Drosophila Transient Receptor Potential-A1 (dTRPA1) failed to shift activity into the dark like control flies do and also identified the role of a specific cluster of the dTRPA1-expressing neurons, the dTRPA1sh+neurons necessary for this. In this study, we extended our previous findings and characterized the identity of these dTRPA1sh+ neurons based on their overlap with circadian neurons. Utilizing various genetic manipulations, we asked whether the overlapping neurons could be potential points of intersection between the 2 circuits that modulate behavior under warm temperature, meaning whether they function as both—sensory and clock neurons. We found that the molecular clock within the dTRPA1sh+ cluster was not necessary, but the expression of dTRPA1 in a subset of circadian neurons, the small ventrolateral neurons (sLNvs), was necessary in modulating phasing of behavior under warm temperature. Furthermore, attempting to identify the neuronal circuit, we were able to uncover the potential roles of serotonin and acetylcholine in modulating this temperature-dependent behavior. Finally, we also discuss possible parallel neuronal pathways that may exist to give rise to this modulation of behavior under warm temperature, thereby supporting and expanding the knowledge of the field about circuits that control temperature-mediated behavioral outcomes.
Keywords
In metazoans, the circadian clock composed of specific neurons in the brain controls the timing of various physiological and behavioral rhythms such as locomotion and sleep, eliciting a periodicity of ~24 h. The circadian clock comprises endogenously regulated oscillations in protein and mRNA abundance and post-transcriptional and post-translational modifications that constitute its molecular basis (reviewed in Zheng and Sehgal, 2012). In flies, the circadian clock is known to be a group of ~150 neurons bilaterally distributed in the brain and clustered into different subgroups. Based on location, there are 2 groups of
The circadian clock utilizes daily changes in environmental factors such as light, temperature, humidity, and food availability as zeitgebers (German for “time-givers”) to phase different aspects of behavior and molecular oscillations. Furthermore, in response to temperature conditions that are relatively warm, like constant warm ambient temperature such as 30 °C (LD30, where LD is 12 h each of light and dark), wild-type (WT) flies phase their activity-rest behavior such that they are visibly more nocturnal than their behavior under cooler temperature conditions of 25 °C (LD25) (Das et al., 2016; Majercak et al., 1999). For flies to be able to phase behavior appropriately with respect to temperature, there must be at least 2 components involved—(1) a temperature-sensory pathway that senses the ambient temperature, and (2) a timing mechanism that controls and regulates behavior in response to the temperature input. The first component involved in phasing of activity-rest behavior under relatively warm LD30 conditions appears to be the ion channel, Drosophila Transient Receptor Potential-A1 (dTRPA1). Lack of dTRPA1 results in flies that do not shift their behavior into the dark, thus suggesting the role of this ion channel in modulating phasing of behavior under warm LD30. On the other hand, constitutive excitation of a neuronal subset expressing the dTRPA1, referred to as the dTRPA1sh+neurons, is sufficient to shift the activity into the dark even under relatively cool conditions of LD25 (Das et al., 2016), further confirming the role of neurons expressing dTRPA1 as the thermosensory component involved in phasing of behavior under LD30. The second component that regulates the timing of behavior in response to ambient conditions points toward the circadian clock neurons that either interact with the dTRPA1sh+neurons directly or are thermosensory themselves, and hence allow the timing of behavior appropriately under different ambient temperature conditions. Interestingly, observations made by Das et al. (2016) with immunocytochemistry revealed that a subset of dTRPA1sh+ neurons co-localize with at least 3 types of circadian clock neurons in the fly brain: 2 PDF+ sLNvs, 2 PDF− 5th-sLNvs, 3-4 LNds, and 1-2 DN1s (Figure 1a) (also see Lee, 2013; Yoshii et al., 2015). This suggests a possible connection between the thermosensory circuit and the circadian clock circuit that could potentially be responsible for coordinating the behavioral modification in response to temperature. We asked what the relative contributions are of the circadian and non-circadian dTRPA1sh+ neurons in mediating phasing of behavior under LD30.
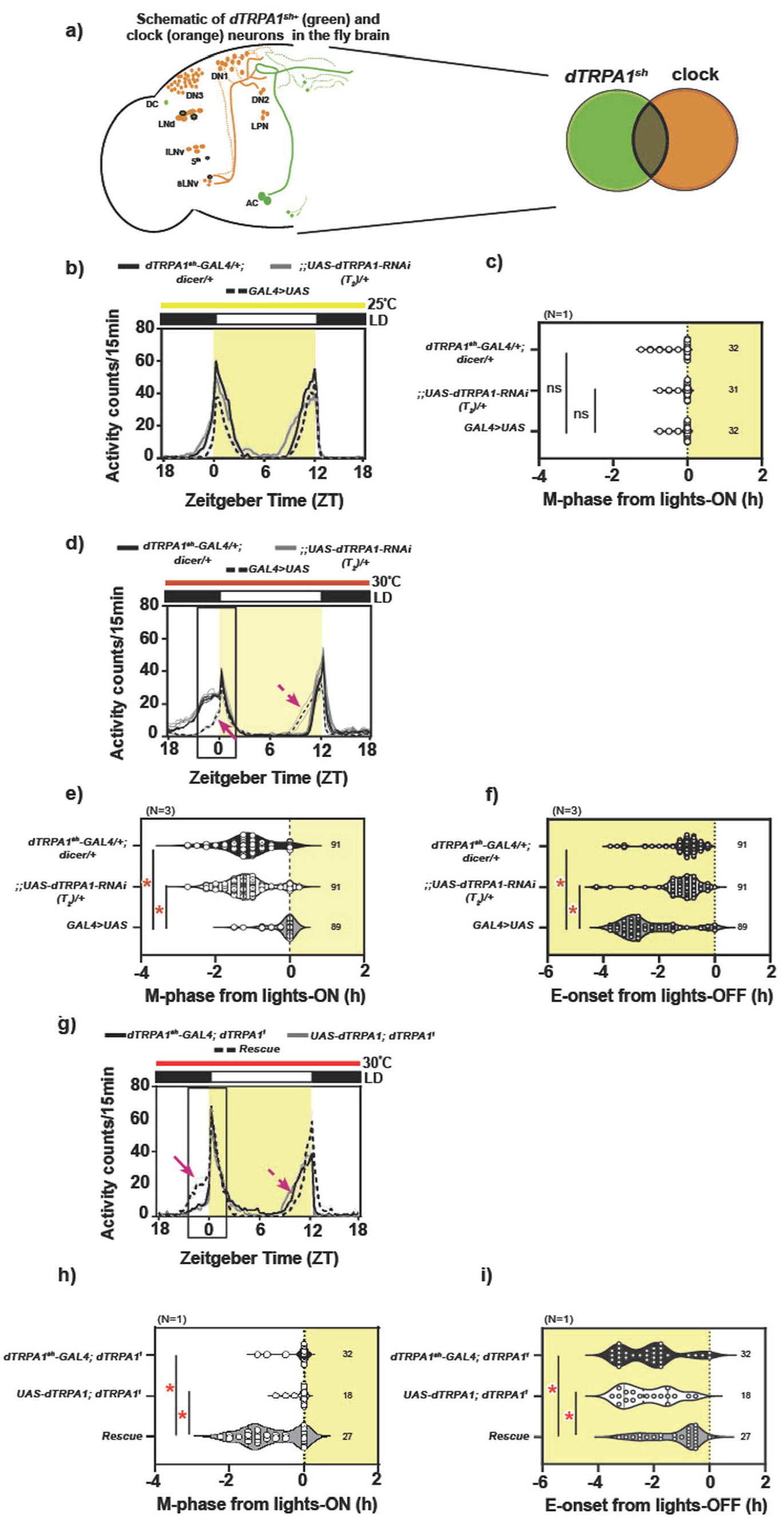
dTRPA1 expression in the dTRPA1sh+ neurons is necessary and sufficient in modulating phasing of behavior under warm ambient condition. (a, left) Cartoon representation of fly brain depicting clock (orange circles, LNvs, LNds, DNs, LPNs) and dTRPA1sh (green circles, ACs, DCs, some cells in the anterior and SOG region of the brain) circuits. Circles with bold outline indicate neurons that overlap between the 2 circuits. (Right) Schematic of the overlap between the 2 circuits. (b) Average activity counts/15 min plotted across zeitgeber time (ZT, h) for flies kept under LD25. Black and white bar on top represent night and day, respectively. Shaded region depicts daytime, and horizontal bar represents constant temperature. Bold traces depict parental controls, dTRPA1sh-GAL4/+; dicer/+ (black trace) and;;UAS-dTRPA1-RNAi(T2)/+ (gray trace) and experimental genotype (dashed trace, dTRPA1sh-GAL4/+; UAS-dicer/UAS-dTRPA1-RNAi(T2)). Faint traces depict SEM across individuals. (c) Quantification of M-phase from lights-on: Violin plots depict the spread of phases of individuals (symbols). Number indicates number of individuals for each genotype. The M-phase of all 3 genotypes is comparable under LD25 (Welch ANOVA, F2,57.8 = 1.975, p = .148, N = 1). (d) Average activity counts/15 min plotted across ZT (h), for flies kept under LD30. Error = SEM across replicate experiment means. All other details same as above. Box indicates region around lights-on where the morning peak is observed; this is expanded to visualize quantification in (e). (e) Quantification of M-phase from lights-on: Compared to both controls, the mean M-phase of experimental flies is significantly delayed (mixed-model ANOVA with genotype as fixed and replicate as random factor, F2,2 = 37.930, p = .002, N = 3). (f) Quantification of E-onset from lights-off: Compared to both controls, the mean phase of E-onset of experimental flies is significantly advanced (mixed-model ANOVA with genotype as fixed and replicate as random factor, F2,2 = 10.463, p = .026, N = 3). (g) Average activity counts/15 min plotted across ZT (h), for flies kept under LD30. Error = SEM across individuals. All other details same as above. (h) Quantification of M-phase from lights-on: Compared to both parental controls, dTRPA1sh-GAL4; dTRPA11 and UAS-dTRPA1; dTRPA11, the mean M-phase of experimental flies, dTRPA1sh-GAL4/UAS-dTRPA1; dTRPA11, is significantly advanced (Welch ANOVA F2,45.04 = 16.225, p << .001, N = 1). (i) Quantification of E-onset from lights-off: Compared to both controls, the mean phase of E-onset of experimental flies is significantly advanced (one-way ANOVA with genotype as factor, F2,74 = 8.258, p << 0.001, N = 1). Abbreviation: ANOVA = analysis of variance; dTRPA1 = Drosophila Transient Receptor Potential-A1. Color version of the figure is available online.
Our experiments reveal that although about 10-12 circadian neurons overlap with the dTRPA1sh+ circuit, the molecular clock within these neurons does not play a role in phasing behavior under LD30. Yet, the expression of dTRPA1 in the circadian neurons and specifically the PDF+ LNvs is necessary for appropriate phasing of behavior under LD30. Finally, we attempt to delineate a circuit for this temperature-dependent phasing of behavior under LD30 and present a role for serotonin receptors on the LNvs and acetylcholine (ACh) within the dTRPA1sh+ neurons.
Materials and Methods
Fly Strains
All genotypes were reared on standard cornmeal medium under LD (12 h light:12 h dark) and 25 °C. Lines Pdf-GAL4 and Pdf-GAL80 were obtained from Todd Holmes; R6-GAL4 from Charlotte Helfrich-Förster; dTRPA1ins mutant and dTRPA1sh-GAL4 from Paul Garrity; Clock-856(8.2/2)-GAL4 from Orie Shafer; the following lines from the Bloomington Drosophila Stock Centre (BDSC): dTRPA11 (#26504), Clk4.1M-GAL4 (#36316), UAS-dicer (#24650 and #24651), UAS-dTRPA1 (#26263), UAS-per-RNAi (#25802), UAS-tim-RNAi (#40864), UAS-5HT1B-RNAi (#33418, #51842), and UAS-dTRPA1-RNAi (Janelia Farms [JF]) (#36780); and the following lines from Vienna Drosophila Research Centre (VRDC): UAS-5HT1B-RNAi (VDRC no. 617, GD construct) and UAS-dTRPA1T2-RNAi (VDRC no. 37250).
Behavioral Assays
Locomotor assay in brief: Flies were reared and collected at 25 °C. Virgin males of age 2-3 days were housed individually, inside glass tubes (6.5 cm × 0.7 cm), with standard corn food (filled till approximately one-fifth of length) at one end that was sealed with wax and plugged with cotton at the other end. These tubes were loaded into Drosophila activity monitors (DAM; Trikinetics, Waltham, USA) (DAM, RRID:SCR_021798) (Pfeiffenberger et al., 2010) in incubators manufactured by Sanyo (MIR-134, Japan), and constant temperature and light intensity (approximately 200-250 lux) were maintained.
Analysis
Raw activity counts per fly were binned into 15-min bins, averaged across 5 days for individuals. These were used to plot activity profiles. Phase of morning peak from lights-on (M-peak): The phase of highest activity before lights-on was identified for each fly from activity profiles and the phase- relationship with lights-on was calculated. Phase of onset of evening activity (E-onset): The time point at which post-siesta activity levels reached 20% of maximum daily activity (for individual flies) was identified for each fly from activity profiles and the phase relationship with lights-off was calculated. Morning anticipation index: For each fly, mean raw activity counts (averaged across 5 days) 3 h before lights-on were divided by the mean raw activity counts 6 h before lights-on.
Statistical Analysis
Where N > 1, analyses were performed on means of replicate experiments. A mixed-model analysis of variance (ANOVA) with genotype as fixed and technical replicate as random factors was implemented, and Tukey’s HSD (honest significance test) was utilized as post hoc test, keeping α = 0.05. Analyses were performed on STATISTICATM software (RRID: SCR_014213) by StatSoft Inc (USA), version 7, and Tukey’s post hoc differences were calculated in MS Excel. Where N = 1, 1-way ANOVA was utilized with genotype as the main factor followed by Tukey’s HSD. If Levene’s test of homogeneity was not satisfied for any data set, a Welch ANOVA was utilized, which was followed by the Games-Powell post hoc test. All figures were plotted using GraphPad Prism (RRID:SCR_002798), version 9.0.0 for Windows (www.graphpad.com) and arranged using Adobe InDesign CS (RRID: SCR_021799), version 3.0. Details are provided in Supplementary Tables T1.0-T1.33.
Results
dTRPA1 Expression in a Subset of dTRPA1+ Neurons, the dTRPA1sh+ Neurons, Is Necessary and Sufficient for Phasing of Behavior Under LD30
To first validate the use of dTRPA1sh-GAL4 and UAS-dTRPA1-RNAi (T2) constructs, we downregulated the levels of dTRPA1 using RNA interference along with the dTRPA1sh-GAL4 driver and observed the activity-rest behavior of flies under LD25 and LD30. dTRPA1sh-GAL4 targets a small subset of dTRPA1+ neurons, referred to as the dTRPA1sh+ neurons, that include the anterior cells (ACs), ventral cells (VCs), and dorsal cells (DCs), along with neurons in the supra-oesophagal ganglion (SOG) (Hamada et al., 2008) and also a small subset of circadian neurons (Das et al., 2016; Lee, 2013, cartoon in Figure 1a). Under relatively cooler LD25 conditions, when dTRPA1 levels were reduced using the dTRPA1sh-GAL4 driver, experimental flies, dTRPA1sh-GAL4/+;UAS-dicer/UAS-dTRPA1T2-RNAi, displayed phasing of morning peak comparable to both parental controls, dTRPA1sh-GAL4/+;UAS-dicer/+ (black trace) and UAS-dTRPA1T2-RNAi/+ (gray trace) (Figure 1b). Quantification revealed no effect of genotype on the mean phase of the morning peak (Figure 1c). In contrast, under warmer conditions of LD30, experimental flies displayed a defect in advancing the phase of the M-peak and E-onset compared to the parental controls (Figure 1d-1f), thus mimicking the behavioral response of dTRPA1ins loss-of-function mutants under LD25 and LD30 (Das et al., 2016). Quantification revealed a main effect of genotype on the mean phase of the morning peak as well as the onset of evening activity. This was also supported by observations made using a second construct for the downregulation of dTRPA1 levels (Suppl. Fig. S1a-S1c). Although not very evident in the activity profiles (individual profiles shown in Suppl. Fig. S1), quantification revealed a significant effect of genotype on the M-phase and E-onset, with both parental controls, dTRPA1sh-GAL4/+;UAS-dicer/+ (black trace) and;;UAS-dTRPA1 -RNAi/+ (gray trace), displaying a significantly advanced M-phase from lights-on and delayed phase of E-onset as is expected under warm temperature, while the experimental genotype, dTRPA1sh-GAL4/+;UAS-dicer/UAS-dTRPA1-RNAi (dotted trace), fails to advance their M-peak or delay their E-onset. These results together indicated that the expression of dTRPA1 in the dTRPA1sh+ neurons is necessary for phasing of behavior under LD30.
Under a relatively cool temperature of 25 or 27 °C (LD25 or LD27, respectively), targeted activation of the dTRPA1sh+ neurons using UAS-NaChBac (Nitabach et al., 2002) resulted in experimental flies with advanced phase of M-peak compared to parental controls, mimicking the behavioral response of WT flies under relatively warm LD30 (Das et al., 2016). Thus, the activation of dTRPA1sh+ neurons was sufficient to elicit a behavioral phase-advance of the M-phase. With this background, we aimed to test the sufficiency of dTRPA1 expression in the dTRPA1sh+ neurons in modulating phasing of behavior under LD30 by expressing dTRPA1 in the dTRPA1sh+ neurons in the background of loss-of-function mutants, dTRPA11 (Kwon et al., 2008; Lee and Montell, 2013) or dTRPA1ins (Hamada et al., 2008). If the expression of dTRPA1 within the subset of neurons is sufficient, experimental flies would display a rescue of behavior with a significant advance in M-phase and a delay in E-onset, compared to both parental controls. Upon exposing flies to relatively warm conditions of LD30, we observed a significant effect of genotype such that the experimental flies with rescued dTRPA1 expression using dTRPA1sh-GAL4 (dTRPA1sh-GAL4/UAS-dTRPA1; dTRPA11) showed an advance in M-phase compared to both parental controls (dTRPA1sh -GAL4; dTRPA11 and UAS-dTRPA1; dTRPA11) and a delay in E-onset (Figure 1g-1i). Taken together, we conclude that the expression of dTRPA1 in the dTRPA1sh+ neurons is necessary as well as sufficient (Figure 1) to modulate phasing of behavior under LD30.
We also utilized a second mutant, dTRPA1ins, and implemented a similar strategy to test the sufficiency of the dTRPA1sh+ neurons. In 2 replicate experiments conducted, we observed a significant effect of genotype such that the experimental flies with rescued dTRPA1 expression using the dTRPA1sh-GAL4 showed an advance in M-phase compared to only 1 parental control (Suppl. Fig. S1d and S1e), with no defect in the phasing of the E-onset (data not shown). Despite a visual trend in the expected direction for the M-phase (Suppl. Fig. S1d), there was lack of rescue in behavior in a dTRPA1ins mutant background, and we speculate it could be due to the contribution of neurons that lie outside the target of the driver (dTRPA1sh-GAL4) utilized here. If these neurons contribute significantly to phasing behavior under LD30, then despite a restoration of dTRPA1 expression in the dTRPA1sh+ neurons, the behavior of the experimental flies would be defective (displaying lack of M-peak advance). This could suggest that appropriate phasing of behavior under LD30 is brought about by the combined effects of at least 2 populations of neurons, one dTRPA1sh+ and one non- dTRPA1sh+.
With this, we validate the use of dTRPA1sh-GAL4 and UAS-dTRPA1-RNAi (T2) constructs and demonstrate the importance of dTRPA1 in the dTRPA1sh+ neurons in enabling appropriate phasing of M-peak and E-onset under warm temperature conditions of LD30.
The Expression of dTRPA1 in the Circadian Neurons Is Necessary in Modulating Phasing of Behavior Under LD30
As described previously, the dTRPA1s + neurons include approximately 2 PDF+ LNvs, 2 PDF− sLNvs, 3-4 LNds, and 1-2 DN1s (Das et al., 2016; Yoshii et al., 2015). This overlap elicits the question as to whether they act as points of communication between 2 circuits, thermosensory and circadian, that could together bring about phasing of behavior under LD30. We decided to test the role of these neurons using 2 neurogenetic strategies: first, by testing the role of the expression of dTRPA1 ion channel within the subset of clock neurons that overlap with the dTRPA1sh+ circuit, and second, by testing the role of the molecular clock within the circadian neurons of the dTRPA1sh+ circuit.
To test the role of dTRPA1 expression in the circadian neurons, we utilized 2 GAL4 drivers: Pdf-GAL4 (Renn et al., 1999) and Clk-856-GAL4 (Gummadova et al., 2009; Yao and Shafer, 2014). Pdf-GAL4 targets only the PDF+ LNvs (Figure 2a), while Clk-856-GAL4 has been described to target all known circadian pacemakers in the brain. If the expression of dTRPA1 is necessary in the circadian neurons, then downregulating the levels must result in a defect in phasing of behavior in the experimental flies compared to respective parental controls.
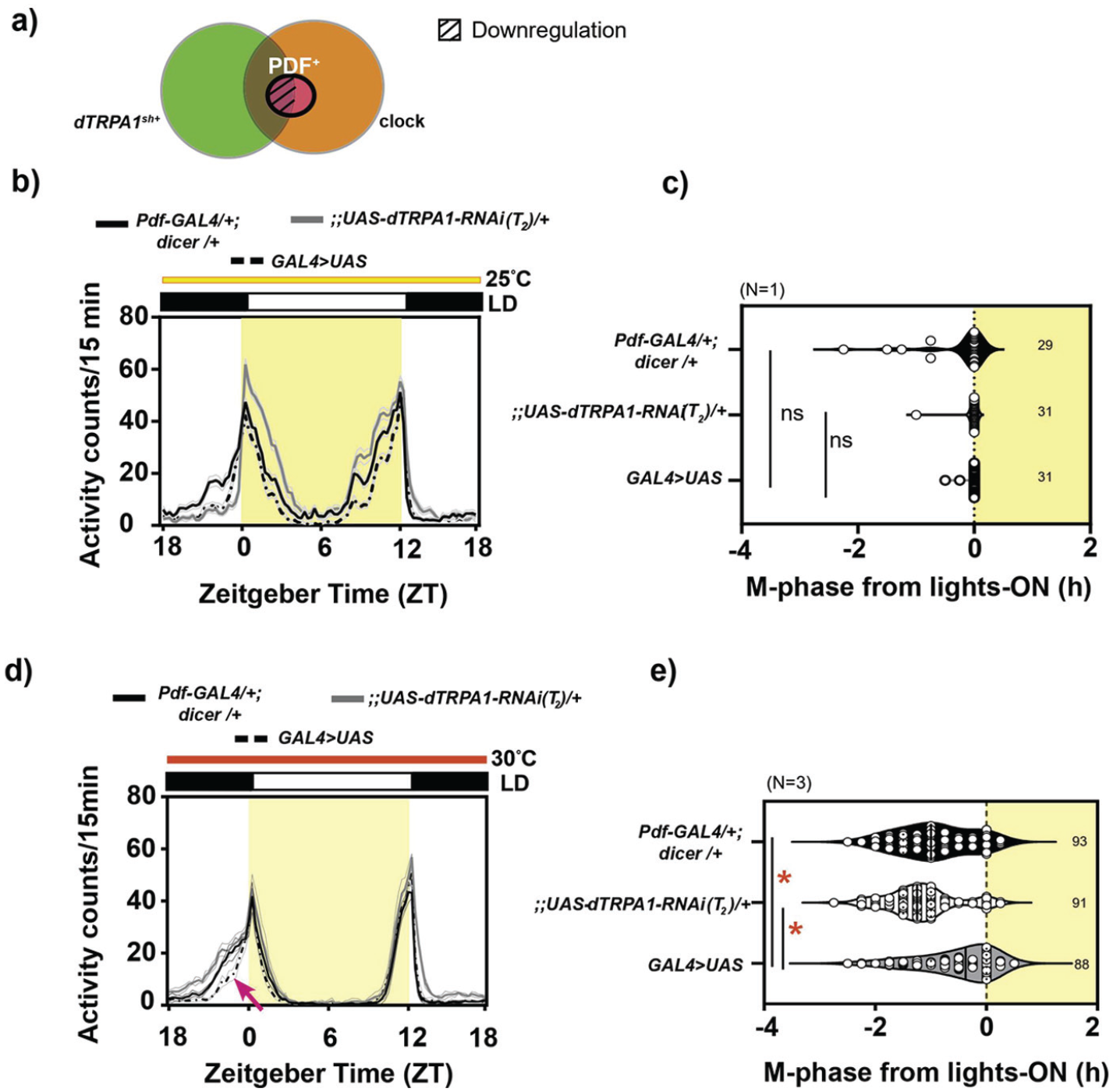
dTRPA1 expression in the PDF+ neurons is necessary to modulate phasing of behavior under warm ambient condition: (a) Diagrammatic representation of the target of Pdf-GAL4 driver (bold outline) and downregulation of the dTRPA1 expression in a subset of neurons (hatched region) (b) Average activity counts/15 min plotted across zeitgeber time (ZT) for flies kept under LD25. Other details same as Figure 1. Bold traces depict parental controls, Pdf-GAL4/+; dicer/+ (black trace) and;;UAS-dTRPA1-RNAi(T2)/+ (gray trace), and experimental genotype (dashed trace, Pdf-GAL4/+;UAS-dicer/UAS-dTRPA1-RNAi(T2)). (c) Quantification of M-phase from lights-on: The M-phase of all 3 genotypes is comparable under LD25 (Welch ANOVA, F2,47.5 = 1.82, p = .173, N = 1). (d) Average activity counts/15 min plotted across ZT ( h) for flies kept under LD30. Error = SEM across replicate experiment means. All other details same as Figure 1. (e) Quantification of M-phase: Compared to both controls, the mean M-phase of experimental flies is significantly delayed (mixed-model ANOVA with genotype as fixed and replicate as random factor, F2,2 = 14.294, p = 0.015, N = 3; asterisk = significant difference). Abbreviations: ANOVA = analysis of variance; dTRPA1 = Drosophila Transient Receptor Potential-A1; PDF = pigment dispersing factor.
Under LD25, RNA interference (RNAi)-mediated downregulation of dTRPA1 under the Pdf-GAL4 driver did not give rise to any defects in activity profiles across genotypes (Figure 2b and 2c). But under LD30, while both the parental controls, Pdf-GAL4/+; UAS-dicer/+ and;UAS-dTRPA1-RNAi/+, displayed an advanced M-phase around ZT 21.5 compared to phase of lights-on at ZT 0, experimental flies, Pdf-GAL4/+;dicer/UAS-dTRPA1-RNAi, failed to show this advance (Figure 2d and 2e, Suppl. Fig. S2a), mimicking the behavior of dTRPA1ins mutants described before (Das et al., 2016). This was also supported by observations made using a second construct for the downregulation of dTRPA1 levels (Suppl. Fig. S2b-S2d). Here, the mean M-phases of both parental controls, Pdf-GAL4/+;UAS-dicer/+ (black trace) and UAS-dTRPA1-RNAi/+ (gray trace), were significantly advanced, while the experimental genotype, Pdf-GAL4/+;UAS-dicer/UAS-dTRPA1-RNAi, failed to do so (dotted trace, Suppl. Fig. S2b and S2c). We also observed that the phase of E-onset was significantly different between experimental flies and parental controls (Suppl. Fig. S2d). These results indicated that the expression of dTRPA1 in PDF+ neurons is necessary for phasing of behavior under LD30.
Curiously, when we utilized the broad clock-neuron driver, we observed different results. Downregulation of dTRPA1 expression using Clk-856-GAL4 that targets all LNvs, LNds, and DNs, as well as the DN1ps and LPNs (Gummadova et al., 2009) revealed a significant defect in phasing compared to parental controls under LD30, but surprisingly, the defect observed here was in the opposite direction than described before. Experimental flies of Clk-856-GAL4/UAS-dicer;UAS-dTRPA1-RNAi/+ showed no difference in M-phase from controls under baseline conditions of LD25 (Suppl. Fig. S3b and S3c), but under relatively warmer LD30, experimental flies advanced their M-phase further more than their parental controls, Clk-856-GAL4/+ and UAS-dicer/+; UAS-dTRPA1-RNAi/+ (Suppl. Fig. S3d and S3e), overriding the potential lack of advance displayed due to reduced dTRPA1 expression in the PDF+ LNvs (Figure 2). This suggested a possible role for the non-LNv, circadian dTRPA1sh+ neurons in mediating appropriate phasing of behavior under LD30. Since we had identified a role for PDF+ LNvs in phasing of behavior under LD30 (Figure 2), we also tested whether retaining the expression of dTRPA1 in the PDF+ LNv in the above background could improve the extreme phase-advanced defect displayed by experimental flies of Clk-856-GAL4/UAS-dicer;UAS-dTRPA1-RNAi/+ genotype. For this, we utilized an intersectional strategy and combined Pdf-GAL80 with Clk-856-GAL4, which restricted the UAS-RNAi expression to only the PDF− circadian neurons (Suppl. Fig. S4a). Thus, experimental flies are expected to possess the expression of dTRPA1 in all non-circadian dTRPA1+ neurons and PDF+ neurons. Activity profiles of experimental flies, Clk-856-GAL4/UAS-dicer;Pdf-GAL80/UAS-dTRPA1-RNAi, showed no differences under LD25 (Suppl. Fig. S4b and S4c), verifying no baseline differences across genotypes. Under LD30, an advance in M-phasing was observed, but this was not significantly different from both parental controls (Suppl. Fig. S4d and S4e) or Clk-856-GAL4/UAS-dicer;UAS-dTRPA1-RNAi/+ (also see Suppl. Table T1.15), suggesting that retention of dTRPA1 in the PDF+ LNvs resulted in an improvement of the M-phase defect in Clk-856-GAL4/UAS-dicer;Pdf-GAL80/UAS-dTRPA1-RNAi.
Our results suggest a role for dTRPA1 expression in the circadian neurons to be necessary for phasing of behavior under LD30, with a role for dTRPA1 expression in the circadian dTRPA1sh+ neurons (that include LNds, LNv, and DNs) in delaying the phase of M-peak (since its absence resulted in extreme phase-advance; Suppl. Fig. S3). Furthermore, we find that dTRPA1 expression in PDF+ LNvs neurons is needed for M-peak advance under LD30 (Figure 2). Moreover, if among the circadian neurons dTRPA1 is retained in the PDF+ LNv, then they (in concert with non-circadian dTRPA1+ neurons) effectively delay the occurrence of the M-peak to its appropriate phase (Suppl. Fig. S4) (also see Discussion). Thus, we infer that the overall effect on M-peak phase is determined by the cohort of neurons, circadian and non-circadian, that express dTRPA1.
The Restoration of dTRPA1 in the PDF-Expressing Ventrolateral Neurons (LNvs) Is Not Sufficient in Modulating Phasing of Behavior Under LD30
To further investigate the role of dTRPA1 ion channel in the PDF+ dTRPA1sh+ neurons, we tested the sufficiency of dTRPA1 expression in these neurons in modulating phasing of behavior under LD30. For this, we used the intersectional strategy of the GAL4 suppressor, GAL80, expressed only in the PDF+ neurons (Pdf-GAL80) along with dTRPA1sh-GAL4 (Das et al., 2016) and downregulated the levels of dTRPA1 (Figure 3a) as before. Activity profiles of experimental flies, dTRPA1sh-GAL4/UAS-dicer;UAS-Pdf-GAL80/UAS-dTRPA1-RNAi, showed no differences under LD25 (Figure 3b and 3c), verifying no baseline differences across genotypes. Under LD30, experimental flies, dTRPA1sh-GAL4/UAS-dicer;UAS-Pdf-GAL80/UAS-dTRPA1-RNAi, which experienced downregulation of dTRPA1 in the entire dTRPA1sh+ neurons excluding the 2 PDF+ dTRPA1sh+ neurons, failed to advance their M-peak compared to parental controls (Figure 3d and 3e). The lack of advance in dTRPA1sh-GAL4/UAS-dicer;UAS-Pdf-GAL80/UAS-dTRPA1-RNAi flies was comparable to that of dTRPA1sh-GAL4/+;UAS-dicer/UAS-dTRPA1-RNAi in 1 experiment under LD30 (see Suppl. Table T1.18), further suggesting that the retention of dTRPA1 expression in the 2 PDF+ dTRPA1sh+ neurons was not sufficient in function to the rest of the dTRPA1sh+ for appropriate phasing of behavior under warm LD30. Taken together, we conclude that the retention of dTRPA1 in the PDF+ neurons is necessary (Figure 2) but not sufficient (Figure 3) within the dTRPA1sh+ circuit for modulating phasing of behavior under LD30.
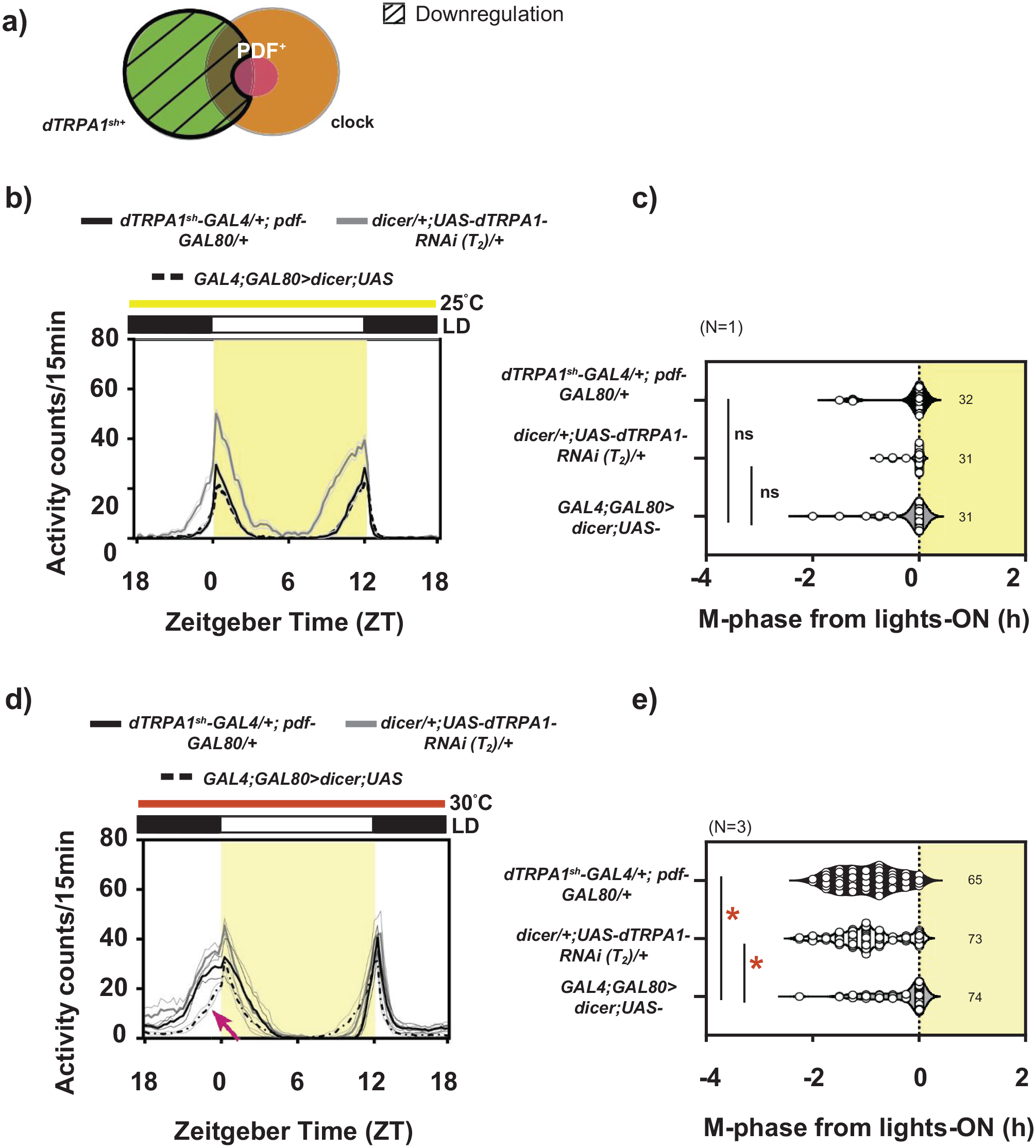
dTRPA1 restoration in the PDF+ neurons is not sufficient to modulate phasing of behavior under warm ambient condition: (a) Diagrammatic representation of the target of dTRPA1sh-GAL4;Pdf-GAL80 driver (bold outline) and downregulation of the dTRPA1 expression in a subset of neurons (hatched region), leaving dTRPA1 expression only in the PDF+ve dTRPA1sh+ neurons. (b) Average activity counts/15 min plotted across zeitgeber time (ZT) for flies kept under LD25. Error = SEM across individuals. Other details same as Figure 1. Bold traces depict parental controls, dTRPA1sh-GAL4/+;Pdf-GAL80/+ (black trace) and dicer/+; UAS-dTRPA1-RNAi(T2)/+ (gray trace) and experimental genotype (dashed trace, dTRPA1sh-GAL4/UAS-dicer;Pdf-GAL80/UAS-dTRPA1-RNAi(T2)). (c) Quantification of M-phase from lights-on: The M-phase is comparable across all genotypes (Welch ANOVA, F2,50.46 = 48.977, p = .204, N = 1). (d) Average activity counts/15 min plotted across ZT for flies kept under LD30. Error = SEM across means of replicate experiments. Other details same as Figure 1. Bold traces depict parental controls, dTRPA1sh-GAL4/+;Pdf-GAL80/+ (black trace) and dicer/+; UAS-dTRPA1-RNAi(T2)/+ (gray trace) and experimental genotype (dashed trace, dTRPA1sh-GAL4/UAS-dicer;pdf-GAL80/UAS-dTRPA1-RNAi(T2)). (e) Quantification of M-phase from lights-on: Compared to both controls, the mean M-phase of experimental flies is significantly delayed (mixed-model ANOVA with genotype as fixed and replicate as random factor, F2,2 = 48.977, p = .001, N = 3). Abbreviations: ANOVA = analysis of variance; dTRPA1 = Drosophila Transient Receptor Potential-A1; PDF = pigment dispersing factor.
The Molecular Clock in the dTRPA1sh Circuit Is Not Necessary for the Phasing of Behavior Under LD30
To address the second aspect of the role of the overlapping set of neurons between the thermosensory dTRPA1sh+ and circadian circuits, we probed the role of the molecular clock in these neurons. We hypothesized that if the molecular clock in these neurons was crucial for conveying timing information for behavior of flies under relatively warm LD30, then disrupting the molecular clock within these neurons would result in defective phasing of the M-peak. To test this, we downregulated the levels of the canonical clock genes, timeless and period, using RNA interference in the dTRPA1sh+ circuit (Figure 4a) and observed behavior under LD30. Given the large number of tim- and per-expressing neurons, this approach allowed us to obtain a nuanced understanding of the contribution of neuronal clusters, namely, the clock-positive, dTRPA1sh-positive and the clock-positive, dTRPA1sh-negative clusters, which have not been previously reported in this context. In addition, from the perspective of understanding the clock circuit itself, the above manipulation would result in disruption of the molecular clock only in a subset of 7-8 core clock neurons.
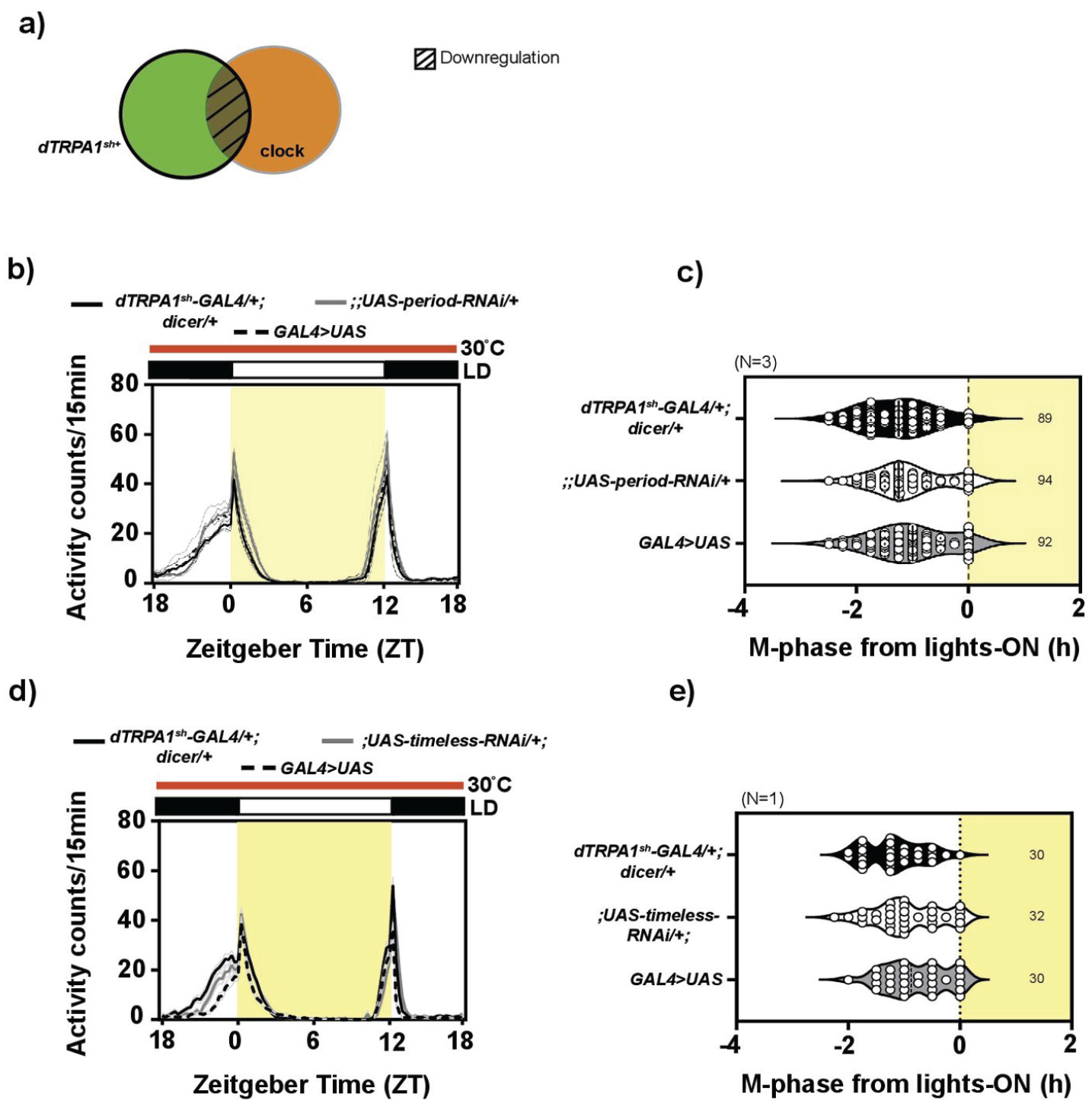
The clock in the dTRPA1sh+ neurons is not necessary for modulating phasing of behavior under warm ambient condition: (a) Cartoon depicting the target of the genetic manipulation: The dTRPA1sh-GAL4 driver is expressed in a broad set of cells (bold outline) and period or timeless gene downregulation is expected to occur in the overlapping subset of neurons (hatched region) between the dTRPA1sh+ neurons and circadian neurons. (b) Average activity counts/15 min plotted across zeitgeber time (h) for flies kept under LD30. Bold traces indicate parental controls, dTRPA1sh-GAL4/+; dicer/+ (black trace) and;;UAS-period-RNAi/+ (gray trace), and experimental flies (dashed trace, dTRPA1sh-GAL4/+;UAS-dicer/UAS-period-RNAi). Error = SEM across means of replicate experiments. All other details same as Figure 1. Phase of the morning peak is advanced with respect to lights-on in all the 3 genotypes. (c) Quantification of M-phase from lights-on: The mean M-phases for the 3 genotypes are comparable, with no significant differences (mixed-model ANOVA with genotype as fixed and replicate as random factor, F2,2 = 1.146, p = .404, N = 3). (d) Average activity counts/15 min plotted across zeitgeber time (h) for flies kept under LD30. Bold traces indicate parental controls, dTRPA1sh-GAL4/+; dicer/+ (black trace) and;UAS-timeless-RNAi /+ (gray trace), and experimental flies are displayed in dashed trace (dTRPA1sh-GAL4/UAS-timeless-RNAi; UAS-dicer/+). Error = SEM across individuals. All other details same as Figure 1. Phase of the morning peak is advanced with respect to lights-on in all the 3 genotypes. (e) Quantification of M-phase from lights-on: The mean M-phases for the 3 genotypes are comparable, with no significant differences (1-way ANOVA with genotype as factor, F2,89 = 3.057, p = .052, N = 1). Abbreviations: ANOVA = analysis of variance; dTRPA1 = Drosophila Transient Receptor Potential-A1.
No defect was visible in phasing of M-peak under LD30 for experimental flies, dTRPA1sh-GAL4/+;UAS-dicer/UAS-per-RNAi and dTRPA1sh-GAL4/+; UAS-dicer/ UAS-tim-RNAi (Figure 4b and 4d), when compared to controls, dTRPA1sh-GAL4/+; UAS-dicer/+ and UAS-per-RNAi/+ or UAS-tim-RNAi/+. Quantification confirmed this and revealed no main effect of genotype—all the 3 genotypes advanced M-phase by around 1.5 h with respect to lights-on (Figure 4c and 4e). Thus, the molecular clock within the dTRPA1sh+ circuit is not necessary for modulating phasing of behavior under LD30. While this is not entirely surprising, it is worth noting that the disruption of the molecular clock in a subset of 8-12 circadian neurons does not hamper the behavior of the flies under LD30, suggesting that the rest of the clock circuit is able to convey timing information for behavior of flies under LD30.
Expression of Serotonin Receptors, 1B, on the PDF+ Neurons May Be Necessary for the Phasing of Behavior Under LD30
How might the dTRPA1sh+ and circadian neurons communicate to bring about modulation of behavior with respect to warm ambient temperature? One candidate neurotransmitter is serotonin (5-
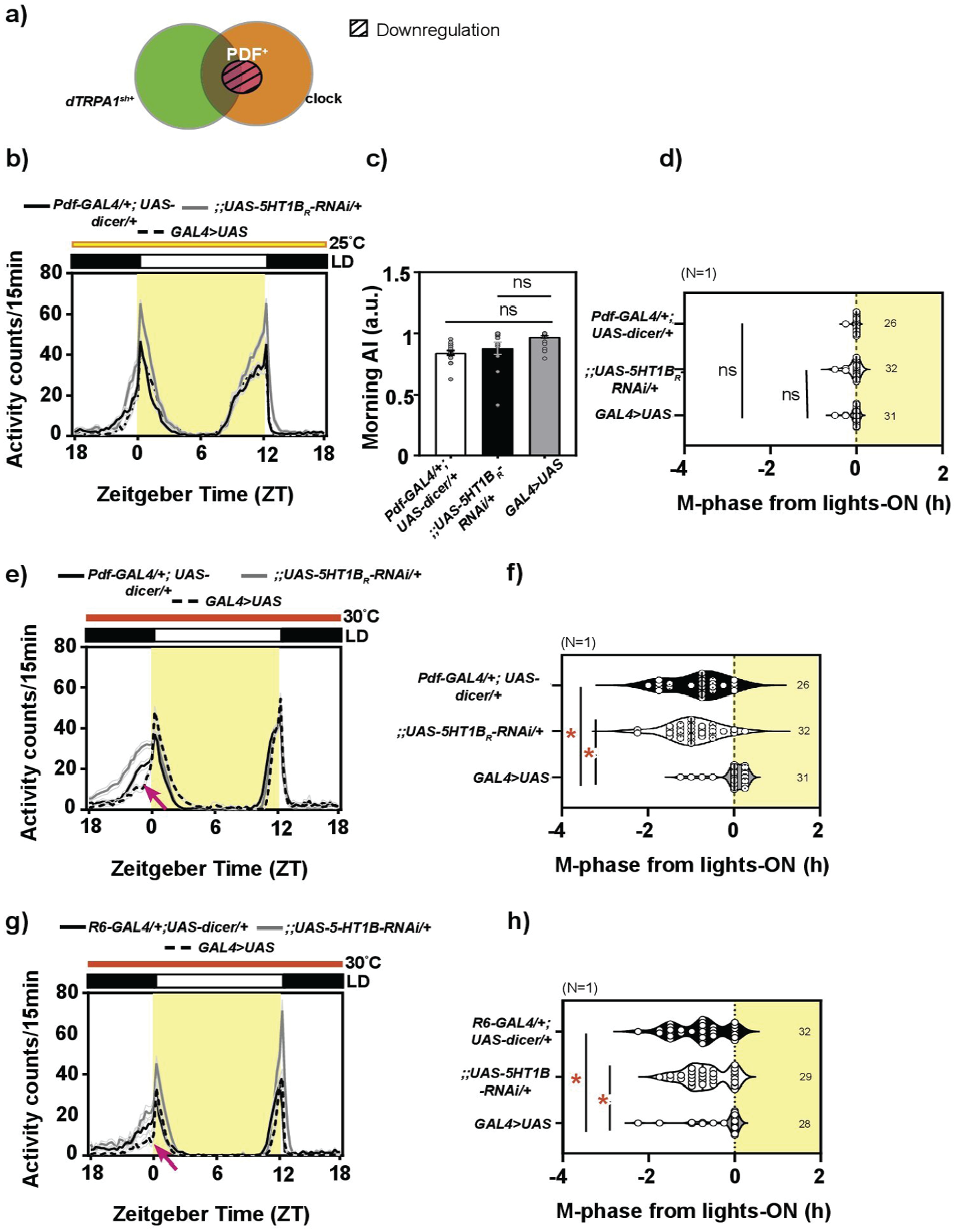
Expression of serotonin receptors, 5HT1B, on the PDF+ neurons is necessary in modulating phasing of behavior under warm ambient condition: (a) Diagrammatic representation of the target of Pdf-GAL4 driver (bold outline) and expected downregulation of the 5HT1B expression in the neurons (hatched region). (b) Average activity counts/15 min plotted across zeitgeber time (ZT) for flies kept under LD25. Error = SEM across individuals. Other details same as Figure 1. Bold traces depict parental controls, Pdf-GAL4/+; dicer/+ (black trace) and;;UAS-5HT1B-RNAi/+ (gray trace) and experimental genotype (dashed trace, Pdf-GAL4/+;UAS-dicer/UAS-5HT1B-RNAi). (c) Quantification of morning anticipation index: There is no significant difference across genotypes (Welch ANOVA, F2,24.2 = 18.11, p > .05, N = 1). (d) Quantification of M-phase: There is no significant differences across genotypes (Welch ANOVA, F1,52.3 = 2.442, p = .09, N = 1). (e) Average activity counts/15 min plotted across ZT for flies kept under LD30. Error = SEM across individuals. Other details same as Figure 1. (f) Quantification of M-phase from lights-on: Compared to both controls, the mean M-phase is significantly delayed for experimental flies, Pdf-GAL4/+;UAS-dicer/UAS-5HT1B-RNAi (1-way ANOVA, F2,85 = 29.470, p << .001, N = 1). (g) Average activity counts/15 min plotted across ZT for flies kept under LD30. Bold traces depict parental controls, R6-GAL4/+; dicer/+ (black trace) and;;UAS-5HT1B-RNAi/+ (gray trace) and experimental genotype (dashed trace, R6-GAL4/+;UAS-dicer/UAS-5HT1B-RNAi). Error = SEM across individuals. Other details same as Figure 1. (h) Quantification of M-phase from lights-on: Compared to both controls, the mean M-phase is significantly delayed for experimental flies, R6-GAL4/+;UAS-dicer/UAS-5HT1B-RNAi (1-way ANOVA, F2,86 = 6.918, p = .001, N = 1). Abbreviations: ANOVA = analysis of variance; dTRPA1 = Drosophila Transient Receptor Potential-A1; PDF = Pigment Dispersing Factor.
To further validate the role of sLNvs in the given context, we performed a similar experiment using a second driver, R6-GAL4, known to target the sLNvs and to test the role of the sLNvs in activity-rest behavior (Nagy et al., 2019). Supporting our above findings, we found that while the phasing of behavior was comparable across genotypes under baseline, standard laboratory conditions of LD25 (Suppl. Fig. S5c and S5d), experimental flies, R6-GAL4/+;UAS-5HT1B-RNAi/UAS-dicer, differed significantly in their phasing of the M-peak under LD30 from both its parental controls, R6-GAL4/+;UAS-dicer/+ and;;UAS-5HT1B-RNAi/+, in the expected direction (Figure 5g and 5h). Thus, taken together, we conclude that signaling via serotonin receptor on the PDF+ sLNvs is necessary for phasing of behavior, specifically under warm ambient condition.
Acetylcholine, But Not Serotonin Expression, in the dTRPA1sh+ Circuit Is Necessary for the Phasing of Behavior Under Warm Ambient Temperature
Based on the proposed model, 5HT1B+ PDF+ neurons would receive serotonin signal from serotonergic dTRPA1sh+ neurons. To test this, we probed the role of serotonin within the dTRPA1sh+ circuit (Figure 6a) in modulating phasing of behavior under LD30 by downregulating the levels of serotonin in the dTRPA1sh+ circuit via RNAi against the biosynthetic enzyme for serotonin,
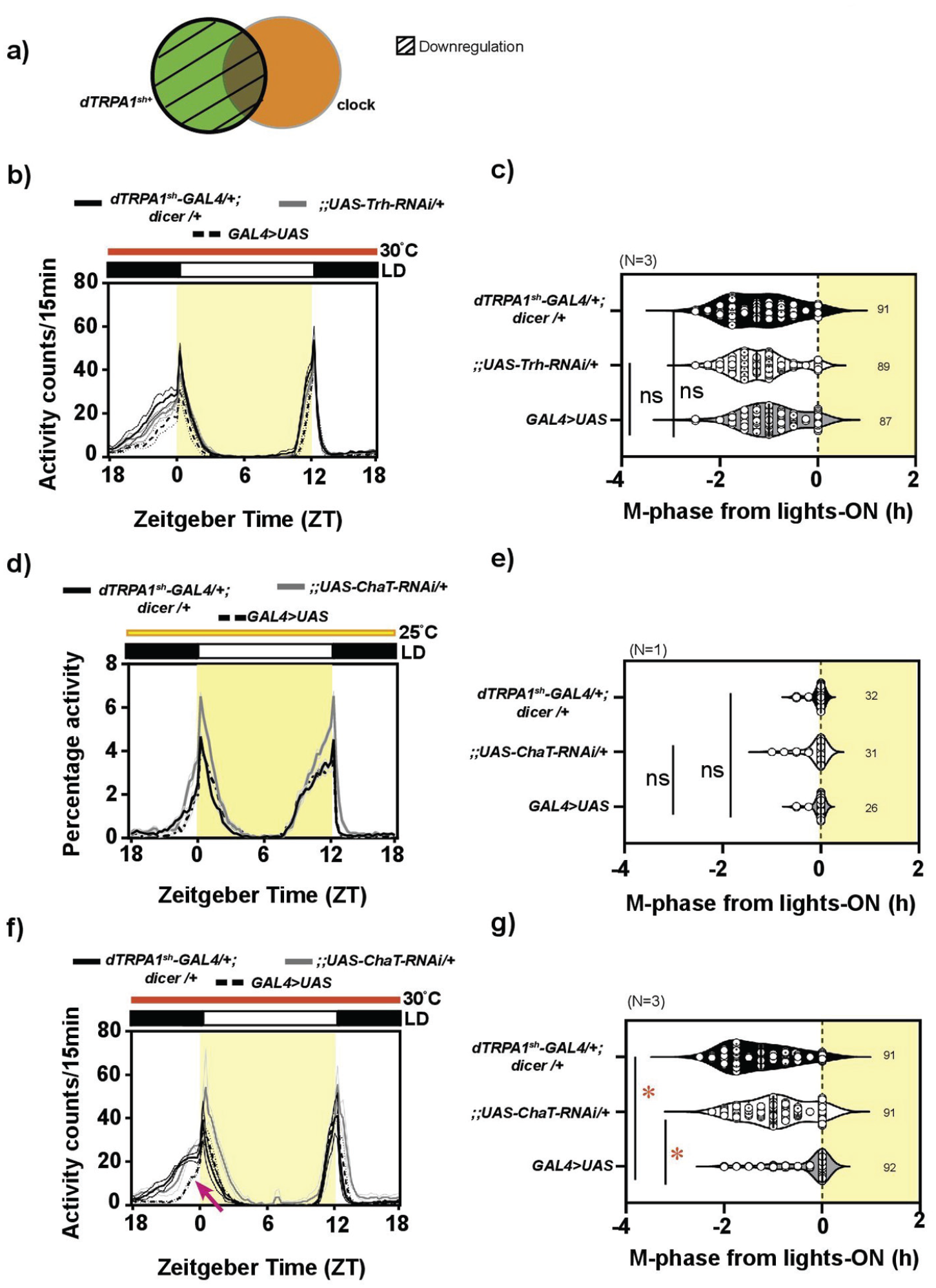
Expression of acetylcholine but not serotonin in the dTRPA1sh circuit is necessary in modulating phasing of behavior under warm ambient condition: (a) Schematic depicting target of dTRPA1sh-GAL4 driver (bold outline) and the downregulation (hatched region) of expression of tryptophan hydroxylase (Trh) and acetylcholine transferase (ChAT), required for the synthesis of serotonin and acetylcholine, respectively. (b) Average activity counts/15 min plotted across zeitgeber time (ZT) for flies kept under LD30. Bold traces indicate parental controls, dTRPA1sh-GAL4/+; dicer/+ (black trace) and;;UAS-Trh-RNAi/+ (gray trace), and experimental flies (dashed trace, dTRPA1sh-GAL4/+;UAS-dicer/UAS-Trh-RNAi). Error = SEM across means of replicate experiments. All other details same as Figure 1. (c) Quantification of M-phase from lights-on. The mean M-phases for the 3 genotypes are comparable, with no significant differences (mixed-model ANOVA with genotype as fixed and replicate as random factor, F2,2 = 1.456, p = .334, N = 3). (d) Average activity counts/15 min plotted across ZT for flies kept under LD25. Bold traces indicate parental controls, dTRPA1sh-GAL4/+; dicer/+ (black trace) and;;UAS-ChAT-RNAi/+ (gray trace), and experimental flies (dashed trace, dTRPA1sh-GAL4/+; UAS-dicer/UAS-ChAT-RNAi). Error = SEM across individuals. All other details same as Figure 1. (e) Quantification of M-phase from lights-on. The mean M-phase is comparable across genotypes, with no significant difference (1-way ANOVA, F2,86 = 0.709, p = .495, N = 3). (f) Average activity counts/15 min plotted across ZT for flies kept under LD30. Bold traces indicate parental controls, dTRPA1sh-GAL4/+; dicer/+(black trace) and;;UAS-ChAT-RNAi/+ (gray trace), and experimental flies (dashed trace, dTRPA1sh-GAL4/+;UAS-dicer/UAS-ChAT-RNAi). Error = SEM across means of replicate experiments. All other details same as Figure 1. Compared to controls, the phase of the morning peak is not advanced in experimental flies (arrow) (g) Quantification of M-phase from lights-on. Compared to both controls, the experimental flies have a significantly delayed M-phase (mixed-model ANOVA with genotype as fixed and replicate as random factor, F2,2 = 52.308, p = .001, N = 3). Abbreviations: ANOVA = analysis of variance; dTRPA1 = Drosophila Transient Receptor Potential-A1.
The above result prompted us to ask which other neurons and neurotransmitters other than serotonin from the dTRPA1sh+ neurons may be involved in modulating this phasing of behavior. Previous reports have indicated that dTRPA1sh+ AC neurons are targeted by a driver specific for ACh (Hamada et al., 2008), suggesting the expression of ACh in these neurons. Hence, we tested the role of ACh from the dTRPA1sh+ neurons in modulating phasing of behavior. We investigated the role of ACh in the dTRPA1sh circuit using RNAi for the biosynthesis enzyme,
Serotonin Expression in Dorsal Neuron-1 (DN1) Circuit Is Not Necessary for Phasing of Behavior Under Warm Ambient Temperature
Jin, Tian, and colleagues demonstrate a role for the circadian cluster of
Based on this, we proposed a model where the DN1s are intermediate neurons for the AC→DN1→sLNv circuit involved in mediating warm-temperature-dependent phasing of M-peak under LD30 and asked whether serotonin expression within the DN1s could signal to serotonin receptor–expressing 5HT1B+ sLNvs described above. To this end, we targeted DN1ps using Clk4.1M-GAL4 (Zhang et al., 2010) and reduced the levels of serotonin using RNAi against the biosynthesis enzyme for serotonin, as utilized before. Under relatively cool, LD25 conditions, experimental flies of UAS-dicer/+;Clk4.1M-GAL4/UAS-Trh-RNAi showed a slight advance in M-phase, but this is not significantly advanced from controls, UAS-dicer/+;Clk4.1M-GAL4/+ and;;UAS-Trh-RNAi/+ (Suppl. Fig. S7a and S7b). Similarly, under warmer LD30 conditions as well, there was no difference between experimental flies and controls with all the 3 genotypes advancing their phase with respect to lights-on (Suppl. Fig. S7c and S7d). This suggests that serotonin from these DN1s may not be involved in modulating warm temperature–specific phasing of M-peak. However, we exercise caution in making this conclusion, since the driver used in the experiment does not target all the DN1s but only a subset of 8-10 DN1ps, and hence it is possible that serotonin present in other DN1ps may mediate phasing of behavior under warm ambient conditions. This could be investigated in further studies.
Discussion
When exposed to warm environmental temperatures for prolonged durations, flies can modulate the phasing of their activity-rest behavior, such that much of the activity occurs during the night (Das et al., 2016; Majercak et al., 1999). A subset of neurons, some overlapping with the circadian pacemaker circuit, expresses the gene for the thermosensitive ion channel, dTRPA1, and have been previously implicated in being crucial for this modulation of phasing of behavior under warm temperature and standard light conditions (LD30) (Das et al., 2016). Here, we report the relative contributions of the circadian and non-circadian, dTRPA1sh+ neurons in this context and conclude that the expression of dTRPA1 in the circadian neurons, especially in 2 PDF+ dTRPA1sh+ neurons, is necessary in modulating behavior under LD30. Furthermore, we posit a role for serotonin receptors on the PDF+ small LNvs of the circadian circuit and the role for acetylcholine from the thermosensory circuit of dTRPA1sh+ neurons in modulating this behavior. With this, our results contribute to the growing understanding of this temperature-related neuronal circuit that can modulate behavior.
Temperature Sensitivity of Circadian Neurons
This study focuses on the contribution of PDF+ sLNvs that are part of the dTRPA1sh circuit but does not inform whether these neurons are indeed temperature-sensitive as a consequence of expressing dTRPA1. A recent study aimed to identify which circadian neurons respond to temperature by exposing adult flies to a peltier plate maintaining temperatures of 29 or 23 °C, and revealed the temperature sensitivity of the DNs of the circadian circuit via fluorescence changes tagged to endogenous calcium levels (Yadlapalli et al., 2018). Some of our own work has also been able to allude to the role of the LNvs in regulating the allocation of activity levels during the night via PDF signaling under warm LD conditions (Iyengar et al., 2022). We report there that lack of proper PDF signaling and ablated LNvs results in increased activity in the middle of the night under LD30, and we speculate it could be mediated via the DN1s. One other explanation for the defect in phasing of behavior under LD30 in flies that lack dTRPA1 in the LNvs (Figures 2 and 3) could be a consequence of improper neuromodulation of the dTRPA1sh+ PDF+ neurons over post-synaptic neurons, like the DN1s. Our observations with the PDF+ neurons lacking dTRPA1 expression leading to defects in phasing of behavior also suggest that the rest of the dTRPA1sh+ circuit relies on the neuromodulation offered by the PDF+ dTRPA1sh+ neurons under LD30, since the lack of dTRPA1 expression in the PDF+ neurons alone results in a defect in phasing of behavior. Thus, taken together with results presented here, we propose the role of the sLNvs in mediating a temperature-dependent modulation of phasing of behavior.
On the other hand, the PDF+ dTRPA1sh+ neurons, independent of the rest of the dTRPA1sh+ neurons, are not sufficient to independently modulate proper phasing of behavior under LD30 (Figure 3). Collectively, these results suggest 2 subgroups of neurons within the dTRPA1sh+ cluster that are involved in phasing of behavior under LD30. It is interesting to note that such a point of clarity about the relative contributions of groups of neurons has been made possible only due to the availability of genetic manipulations in flies at the level of single neuronal subgroups.
Other Rhythmic Behaviors That Depend on Temperature Sensation Could Have Common Neuronal Circuits
Another temperature-related behavior is the temperature-preference behavior (TPR) of flies under LD25, where flies prefer cool temperature during the night and warmer temperature during the day (Kaneko et al., 2012). TPR is observed even under constant darkness and is disrupted under constant light, indicating that it is clock-controlled. The circadian neuronal subgroups, DN1s, DN2s, and the sLNvs, and the dTRPA1-expressing ACs underlie the neuronal basis of this behavior (Goda et al., 2016; Kaneko et al., 2012; Tang et al., 2017). Although one cannot compare the TPR with activity-rest behavior, the observation that LNvs play a role in modulating behavioral preference for temperature that is time-of-day-dependent suggests that this circuit could potentially play a role in timing of a clock-dependent activity-rest rhythm as well. Similarly, the dTRPA1+ neurons have been implicated in modulating activity and sleep levels under cyclic temperature cues of 20-28/29 °C and constant darkness (Das et al., 2016; Roessingh et al., 2015). This indicates that these neurons could be part of circuits that modulate circadian behavior with respect to temperature. Taken together, it appears that temperature-related behaviors like the TPR and phasing of activity-rest behavior, utilizing common players like LNvs and ACs, could be modulated via similar neuronal pathways. There is also emerging evidence that these common players utilize similar neuromodulation, for instance, serotonin and ACh (Jin et al., 2021; Tang et al., 2017), to bring about behavioral responses under different contexts.
In this study, we demonstrate behavioral defects in flies that have reduced the expression of serotonin receptors 5HT1B on the sLNvs (Figure 5) and it is also worth noting that some of the sLNvs that express 5HT1B receptors could potentially also overlap with the dTRPA1sh+ sLNvs, suggesting an auto-regulation within the circuit. Furthermore, our results (Figures 4, 5b and 5c) suggest LNvs receive input via serotonin-expressing neurons that are most likely outside the dTRPA1sh+ circuit and are involved in modulating phasing of behavior under LD30, and future studies could pursue this. Our data also suggest a role of ACh in the dTRPA1sh+ circuit in modulating phasing of behavior under LD30. There are ACh receptors detected on the large LNvs (McCarthy et al., 2011) but not the sLNvs, and hence the question of other downstream partners of the dTRPA1sh+ circuit partaking in completing the circuit involved in phasing of behavior under LD30 has remained unclear. Work from the group of Junhai Han has provided insight in this direction where they discovered a role of DN1ps as downstream targets of ACs to promote wakefulness under prolonged warm conditions (Jin et al., 2021). However, in our investigation of the role of serotonin in DN1s (that could potentially signal to the 5HT1B receptors on LNvs), we do not observe any defects in behavior that expressed RNAi for the biosynthesis enzyme for serotonin, Trh (Suppl. Fig. S7). Thus, although the AC←→LNv-DN1p circuit model appears to be converging on various aspects of rhythmic behavior of flies in the context of temperature, roles for DN1s in the context of phasing of behavior under LD30 need further investigation. Future studies could be aimed to investigate which level of brain processing gives rise to the divergence in behavioral output based on the internal and external contexts. Some of this has been discussed in a detailed review by Roessingh and Stanewsky (2017).
Role of the Neurons That Overlap Between the Thermosensory and Circadian Circuits
Using the Pdf-GAL4 driver to target the downregulation of dTRPA1 expression only in the LNvs and utilizing the Pdf-GAL80 intersectional strategy to exclude the downregulation of dTRPA1 in the LNvs, we were able to tease apart the role of the LNvs when in combination with other circadian and non-circadian dTRPA1sh+ neurons. First, we found that the expression of dTRPA1 in the PDF+ LNvs is necessary (Figure 2) but not sufficient (Figure 3) to give rise to appropriate phase-advancing of the M-peak under LD30. Interestingly, when the downregulation of dTRPA1 is targeted to the entire circadian circuit using Clk856-GAL4, a phase-advanced M-peak is observed in experimental flies compared to parental controls (Suppl. Fig. S3), which suggests that the dTRPA1 expression in the circadian neurons is necessary to give rise to appropriate phase-delay of the M-peak under LD30. In this manipulation, the expression of dTRPA1 in the non-circadian dTRPA1sh+ neurons (ACs, VCs, DCs, and some SOG neurons) is expected to be intact, and so the above phase-advancing defect is surprising because it appears to override the phase-delaying effect seen with depriving dTRPA1 in the entire dTRPA1sh+ circuit (Figure 1) as well as the phase-delaying effect seen with depriving dTRPA1 only in the PDF+ LNvs (Figure 2). Even more interestingly, in this experimental background, retaining the expression of dTRPA1 in the PDF+ LNvs (Clk856-GAL4+PdfG80) enabled flies to appropriately phase M activity to occur just before lights-on (Suppl. Fig. S4). Thus, taking the above 3 results together, we conclude that the non-circadian dTRPA1sh+ neurons, the circadian PDF+ dTRPA1sh+ neurons, and the circadian non-PDF+ dTRPA1sh+ neurons have additive roles in modulating phasing of behavior under LD30, depending on the combination of neuronal subsets that retain dTRPA1 expression: The non-circadian dTRPA1sh+ neurons may be involved in phase-advancing (from Suppl. Fig. S3), the circadian non-PDF+ dTRPA1sh+ neurons may be involved in phase-delaying (Suppl. Fig. S4), and the circadian PDF+ dTRPA1sh+ neurons play dual roles, a phase-delaying role when the rest of the circadian cluster has reduced dTRPA1 expression (Suppl. Fig. S4) and a phase-advancing role when the entire dTRPA1sh + circuit has reduced dTRPA1 expression (Figure 2) (see Table 1 and Suppl. Fig. S8 for summary). But how can the phasing of behavior be brought about if the molecular clock within the dTRPA1sh+ circuit is not necessary (Figure 4)? We speculate this may be brought about by the thermosensory role of the circadian dTRPA1sh+ neurons which may indirectly modify how the rest of the circadian circuit modulates phasing of behavior under LD30.
Summary of M-peak phenotypes obtained with various genotypes with restricted dTRPA1 expression.
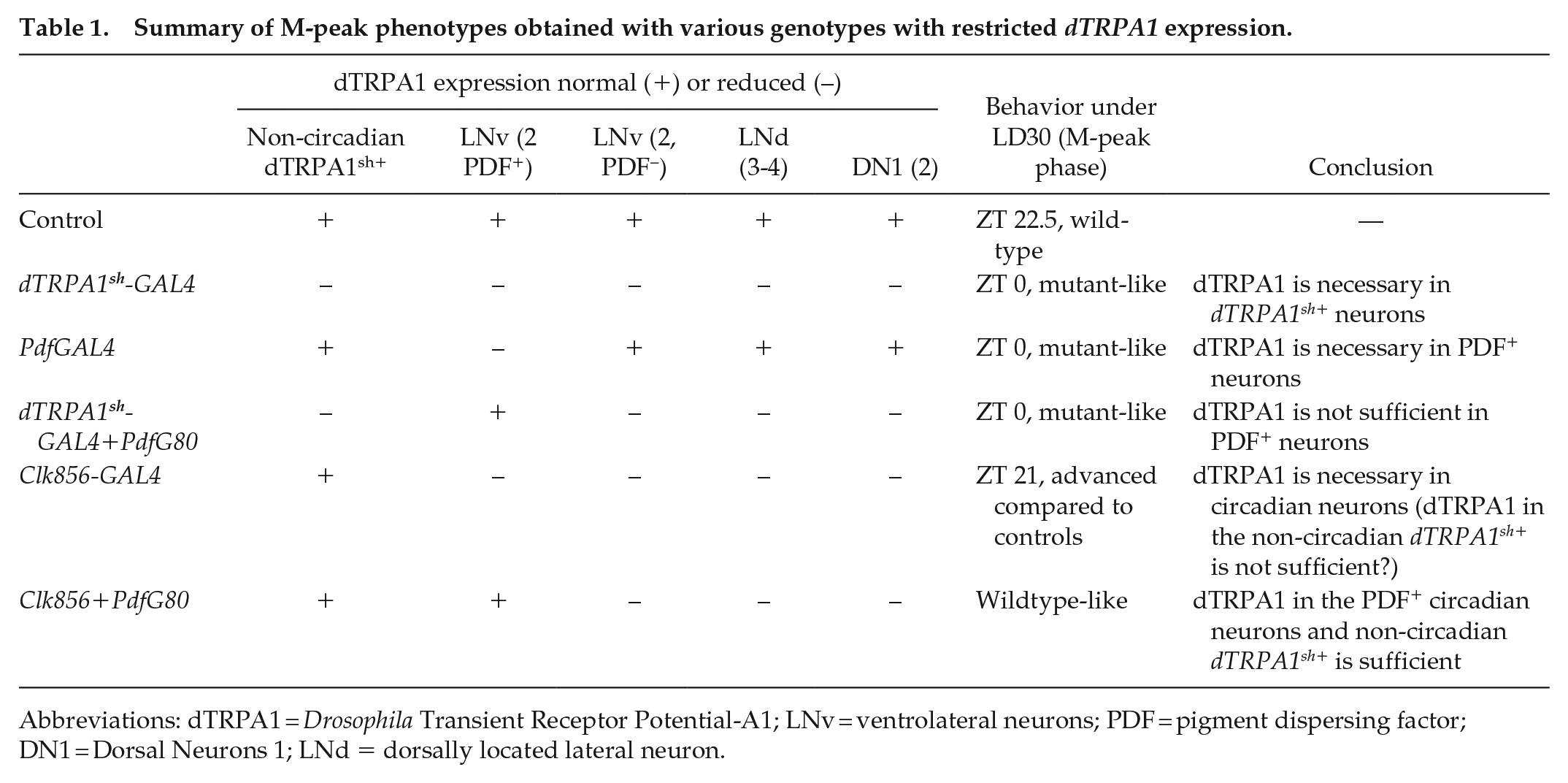
Abbreviations: dTRPA1 = Drosophila Transient Receptor Potential-A1; LNv = ventrolateral neurons; PDF = pigment dispersing factor; DN1 = Dorsal Neurons 1; LNd = dorsally located lateral neuron.
The timing of circadian behavior is understood to be brought about by a complex series of protein oscillations within the circadian neurons. Disrupting the period gene has been shown to lead to arrhythmic or aperiodic behavior (Konopka and Benzer, 1971) and has been phenocopied effectively using the GAL4-UAS strategy, restricting the disruption of the molecular clock in subsets of neurons. In our study, we reduced the levels of the period and timeless genes in only a subset of neurons in the brain, targeted by the dTRPA1sh+ driver expecting a resultant disruption of molecular clock in only a few circadian neurons alone, namely, 2 PDF+ sLNvs, 2 PDF− sLNvs, 3-4 LNds, and 1-2 DN1s. It is not surprising that the behavior we observe under LD30 is normal, since the rest of the entire circadian circuit is intact and is perhaps able to give rise to normal behavior under LD30. This also suggests that a disrupted molecular clock in the small subset of PER+ dTRPA1sh+ neurons does not hamper the phasing of behavior under LD30. In the context of their overlapping position between the circadian and thermosensory circuits, this implies that the PER+/TIM+ dTRPA1sh+ neurons perhaps only play a role in thermosensory circuit in modulating the phasing of behavior under LD30, while their circadian function may be redundant in this context. One strategy to test the necessity of molecular clock in the dTRPA1sh+ neurons for phasing of behavior under LD30 would be to disrupt the molecular clock in the entire circadian circuit, sparing only the dTRPA1sh+ neurons (dTRPA1sh-GAL80). However, the neurogenetic resources for such a manipulation are limited, and hence, this will be addressed in the future.
Conclusion
Ambient temperatures can rise to 30 °C or higher in the wild, and animals, especially insects, must be able to modulate their behavior to avoid harsh consequences. One strategy that flies can adopt is to shift their activity into the dark. In this study, we model a scenario where the mean ambient temperature remains relatively warm at 30 °C, and we attempt to elucidate a neuronal pathway that is responsible for modulation of activity-rest behavior under such conditions. We tease-apart the relative contributions of neurons that overlap between the dTRPA1sh and clock circuits, specifically the PDF+ dTRPA1sh+ neurons. Adding to the well-established role of the sLNs in modulating the morning activity under LD25, we posit the role of expression of the 5HT1B receptors and the dTRPA1 ion channels on the sLNvs in giving rise to warm temperature–specific phasing of behavior, possibly directly or via communication with the ACs and DN1s. Finally, together with recent reports, we identify a role for ACh in the dTRPA1sh circuit in modulation of this behavioral phasing under LD30.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304231159713 – Supplemental material for A Subset of Circadian Neurons Expressing dTRPA1 Enables Appropriate Phasing of Activity Rhythms in Drosophila melanogaster Under Warm Temperatures
Supplemental material, sj-docx-1-jbr-10.1177_07487304231159713 for A Subset of Circadian Neurons Expressing dTRPA1 Enables Appropriate Phasing of Activity Rhythms in Drosophila melanogaster Under Warm Temperatures by Aishwariya Srikala Iyengar, Sushma Rao and Vasu Sheeba in Journal of Biological Rhythms
Footnotes
Acknowledgements
The authors would like to thank Jawaharlal Nehru Centre for Advanced Scientific Research (JNCASR) and Department of Biotechnology (DBT) (BT/INF/22/SP27679/2018), Mr. N Rajanna and Mr. M Muniraju for technical assistance, Dr. Antara Das and other members of the lab for useful discussions and support at roadblocks, Ms. Sheetal Sahu and Ms. Anagha Muralidharan for help with preliminary experiments, Ms. Viveka Singh and 2 anonymous reviewers for comments on a previous version of the manuscript, and the fly community for reagents.
Authors’ Note
Any additional information in the form of raw data will be provided upon reasonable request to the corresponding author.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
