Abstract

Clinicians, scientists, industry, occupational health and public health professionals, and patients are excited about the promising health and safety implications of circadian medicine. The award of the 2017 Nobel Prize in Physiology or Medicine to three scientists (Hall, Rosbash, and Young) for their work in elucidating the mechanisms of circadian rhythmicity introduced many people to the field. Now we and others are frequently asked when the results of basic research will be available for diagnosis, monitoring, and/or treatment of individual patients.
Overwhelming evidence indicates that the circadian clock is essential for health and its disruption causes pathologies (Figure 1). A triad of circadian medicine approaches was recently proposed (Kramer et al., 2022): (a) detecting the clock (e.g. new diagnostic tools), (b) targeting the clock (e.g. improving or resynchronizing disrupted rhythms), and (c) exploiting the clock (e.g. using time-of-day adapted treatment regimens). Examples of each of these approaches already in use include the dim light melatonin onset (DLMO) for detecting circadian phase, light treatment for circadian and other disorders, and time-of-day recommendations for statin-type medications. These and other encouraging past and recent successes in circadian medicine suggest that greater and more comprehensive translation of basic chronobiological findings into medical practice is possible.
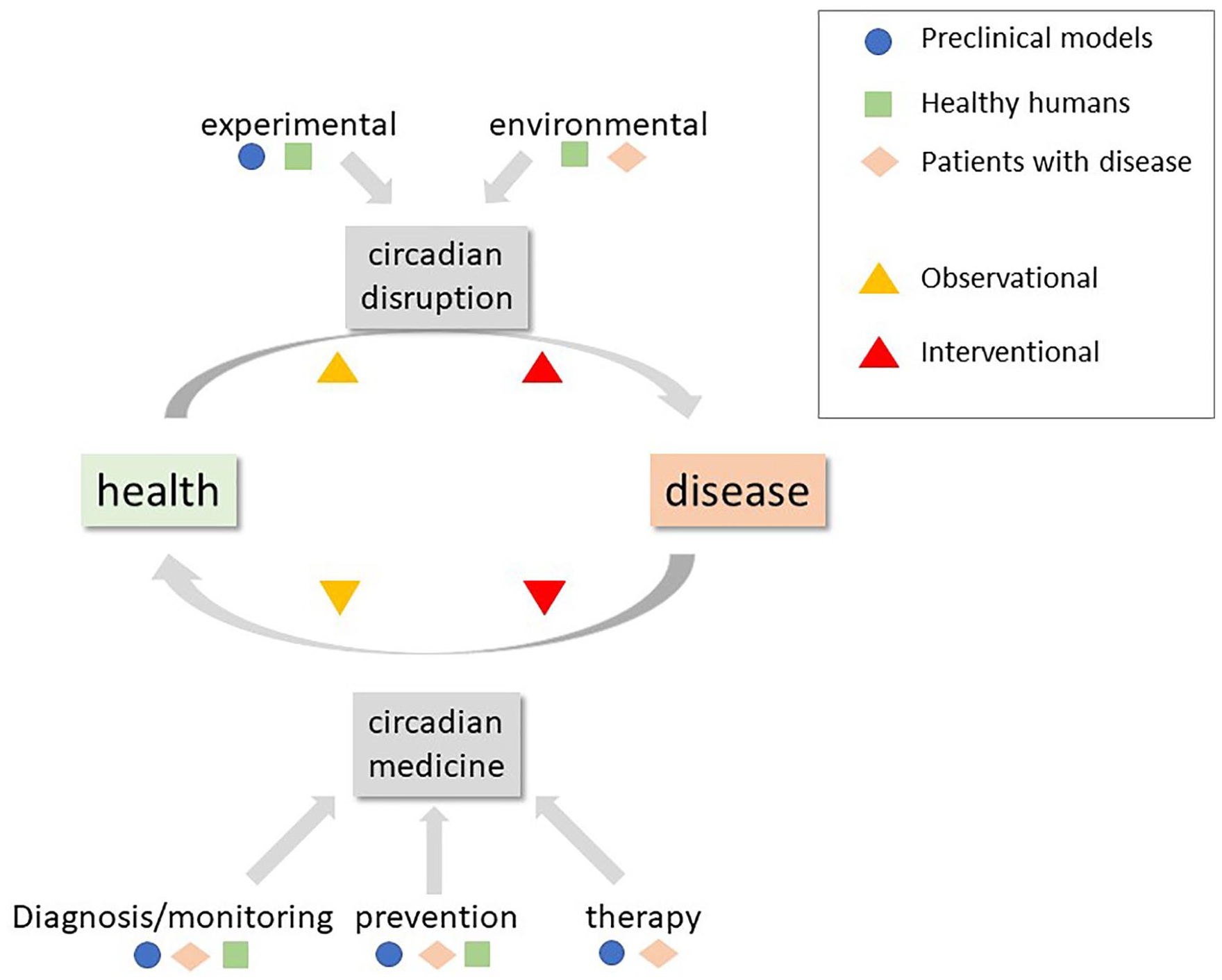
Schematic of health/disease, circadian disruption, and circadian medicine.
We therefore recognize a need to provide for the circadian rhythms research community an outline of the steps, processes and time frame required to translate basic science findings to evidence based clinical care and/or public health and safety recommendations (Figure 2): how does a discovery become an approved approach, and then a guideline, a clinical care recommendation, and finally a policy? Each step along the way is performed with specific questions, protocols, variables, and analyses within a specific population, and each step should consider whether the target is etiology, health/disease balance, and/or recovery (Roenneberg et al., 2022). Each step applies to diagnosis/monitoring tests, interventions (e.g. drug, device), and/or analysis methods [biochemical or algorithmic (e.g. to calculate circadian phase or the optimal timing and/or dose of an intervention)]. Most of the following steps apply to interventions, but the underlying principles are the same. Before efficacy and effectiveness conclusions/clinical guidelines can be drawn, data from multiple experiments and studies will have to be compiled in a stepwise approach.
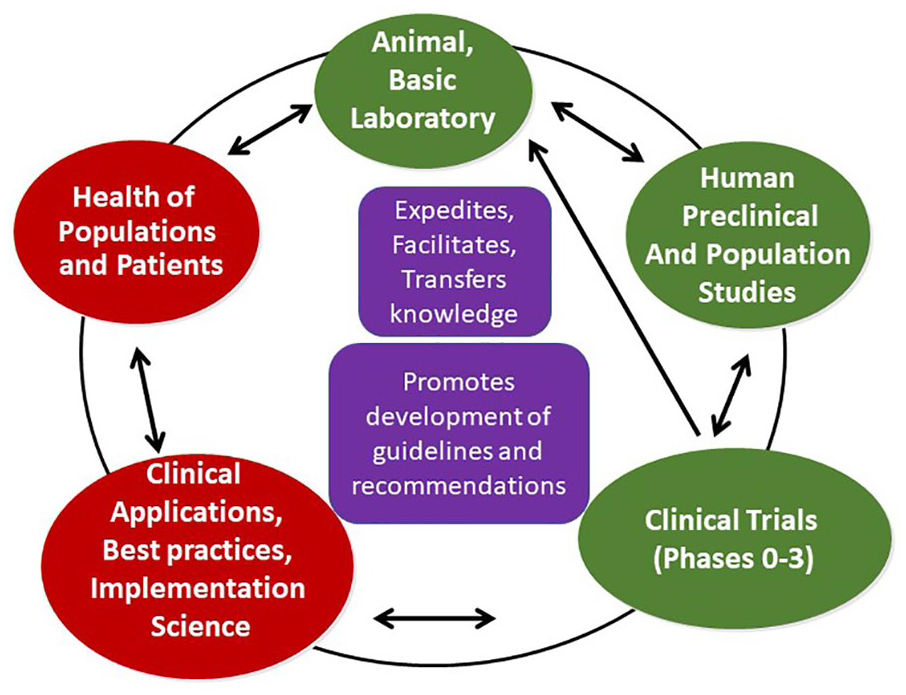
Schematic of multiple interacting steps from bench to bedside and back again.
Overview of Steps, Processes, and Approaches
We have identified five stages of investigation and implementation before a diagnostic tool or therapeutic approach is clinically approved or recommended: (a) preclinical (basic science using animal models of disease) or epidemiological to identify targets of investigation, (b) clinical mechanistic (i.e. in highly controlled protocols) that can be conducted with or without an intervention, (c) efficacy and safety of interventions or diagnostic tools in populations or communities, (d) implementation research, and (d) development of clinical practice guidelines. These five stages need not necessarily proceed sequentially, and the transition from “bedside” observations to “bench” studies (i.e. reverse translation) is often critical for understanding pathophysiology and mechanism. Ultimately establishing policies through advocacy will facilitate (or be necessary for) widespread adoption of the clinical practice. We will use two examples in the following sections to illustrate these points: (a) biomarkers of circadian phase, two of which we are developing (Braun et al., 2018; Wittenbrink et al., 2018), and drug X that can affect circadian rhythms in flies. For the latter, drug X’s specific effect is not relevant here: examples include affecting the stability of rhythms (e.g. for neurodegenerative diseases), shifting rhythms (e.g. for circadian rhythm sleep wake disorders), and/or affecting people with otherwise disrupted rhythms.
In several countries, drug/device intervention studies are classified according to test phases.
Stages from Bench to Bedside
Preclinical and Epidemiological Studies to Identify Potential Targets
For preclinical studies, we consider both studies in non-human models and epidemiological studies. We will not discuss biochemical or in vitro studies here. For studies using non-human models, it is important to consider how different the model is from humans. Many studies are conducted in nocturnal rodents, in which (by definition) the relationship between the timing of activity/rest and associated behaviors and the activity in the hypothalamic circadian pacemaker is different from that of diurnal humans (Schwartz et al., 1983). This difference alone reduces the probability of successful translation (Esposito et al., 2020). The physiologically relevant (for translation) differences from non-mammals (e.g. drosophila, birds) to humans are likely to be much larger, since regulatory pathways (including specific cells, molecules, hormones, receptors), and therefore targets for intervention, may differ.
In animal research, there are two approaches: alter the circadian clock (e.g. create a mutant with missing or altered genes) and then look for disease, or use a disease model (e.g. obesity) and investigate circadian dysregulation at multiple levels of organization. These studies are critical for identifying and understanding potential physiological mechanisms. For the example of circadian phase determination from blood sample(s), these animal studies identified the hypothalamic circadian clock and defined its influence on multiple aspects of daily physiology that could then be incorporated in the design of a test of circadian phase using blood samples. For the example of drug X affecting the circadian pacemaker, animal studies have tested the ability of drugs to shift the circadian pacemaker, which applies to circadian rhythm sleep-wake disorders of jet lag or shiftwork.
Epidemiological studies may also begin with altered clocks (e.g. shiftwork) and look for disease or begin with a disease and examine circadian causes (e.g. chronotype) based on pre-clinical or clinical observations.
Clinical Mechanistic
We limit this section to studies in humans. These studies can be observational or interventional. The goal is to define physiology in humans.
Non-intervention studies (e.g. a diagnostic or monitory marker) involve healthy humans and/or humans with a specific disease. Timed samples (e.g. of blood, heart rate, alertness) are collected and analyzed for daily rhythms. Studies that are initially conducted in healthy individuals need also to be repeated on individuals with specific conditions or diseases or under different conditions (e.g. shiftwork). For the example of determining circadian phase from blood sample(s), these studies identified substances in blood that can be used to predict circadian phase in healthy individuals and should now be conducted in other populations and under different conditions.
Studies involving an intervention are conducted for several reasons: proof of concept, feasibility, estimation of the effect size of an outcome, information for planning of a randomized clinical trial (RCT) (e.g. what measures should be collected, for sample size calculations), and to determine potential mechanisms. RCTs are considered the gold-standard for intervention trials. These intervention studies may produce results that are then investigated using pre-clinical models (i.e. bedside to bench). They may be conducted with healthy humans and/or people with a specific disease and either in the laboratory under highly controlled conditions or in the participants’ home environment. For the example of determining circadian phase using blood sample(s), these studies might involve using the method to time an intervention (e.g. with drug X), and then document whether there are changes in the outcome.
Since some interventions do not require safety testing (e.g. light or time-restricted eating), the initial experiments can be conducted in humans. The same is true for chrono-pharmacology, where already approved medications are tested for efficacy and side effects at different times of day. In both cases, if results are promising, animal models can be used for mechanistic insight.
Epidemiological or Community-Based Study
This is the step of documenting the relationships and relevance of the monitoring or intervention in people with potentially other medical conditions besides the target condition and who are living at home in their daily lives. It studies the relationships between biological, behavioral, and environmental factors and risk for disease, and is often useful in identifying targets for intervention to prevent disease. It can be conducted using epidemiological or RCT methods. For the example of defining circadian phase from blood sample(s), these studies may test whether their use in using circadian timing when planning an intervention improves clinical care. For the example of drug X, these will test the drug’s efficacy and safety in the general population.
Implementation Research
Implementation research tests the best methods for implementing knowledge into practice. It utilizes many methods and approaches and focuses on specific steps that health care professionals or patients can take to influence the benefits of an approach; for the example of drug X, this may include the education of health care professionals (HCPs)(e.g. physician, nurse, pharmacist) and patients about using the drug.
Implementation research is important for the final step that occurs at the level of the HCP–patient relationship. The HCP and patient must evaluate available evidence and guidelines (e.g. Oxman, 1993; Murad et al., 2014) and how they apply to that patient’s medical condition(s) and situation (e.g. Jaeschke, 1994; Guyatt, 1994), and balance them against implementation difficulties (e.g. intervention at 8 pm, multiple saliva samples in dim light) or side effects.
Development of Guidelines/Recommendations
HCP and researchers must evaluate evidence from all phases of studies using meta-analyses and other methods. These evaluations frequently uncover gaps in knowledge. To be accepted by policy makers, health insurers, or other organizations, more than one trial may be required after an initial promising study so that meta-analyses can be conducted. If the evidence is strong enough, guidelines can be developed for the use of the method (e.g. diagnostic tool, intervention) in specific populations or the general public. Recommendations are usually a stronger statement than guidelines.
Advocacy is required for acceptance and promotion of new clinical guidelines or recommendations. Medical and other societies and government agencies (e.g. the National Institutes of Health in the United States) issue guidelines and best practice statements. For example, the American Heart Association (AHA) added “Get Healthy Sleep” to its “Life’s Essentials” list in 2022, 12 years after first formulating the list with 7 other factors. This step required considerable time and effort by AHA members and other experts. Having health insurance plans pay the costs of a diagnostic test or intervention (e.g. drug, device) also requires advocacy.
Two Examples of the Timeline Before FDA Approval
Two recent examples of the multi-decade timeline from basic science to FDA approval (but not implementation research, advocacy, and/or insurance approval) are chronotherapy for rheumatoid arthritis (RA) and a melatonin agonist for treatment of non-24-sleep-wake disorder in blind people. The publications cited here are only a small selection of all available on the topics and each publication required year(s) of planning and experiments.
Observations of diurnal rhythms in RA symptoms were published in the 1950s (Wright, 1959), variation in steroid levels in 1960s (Saba et al., 1963), effect of time of steroids on RA symptoms (including awakening patients at 2 am!) in 1997 (Arvidson et al., 1997), circadian rhythm effects on underlying RA pathophysiology in the 2000s (Cutolo and Masi, 2005), a randomized trial of a delayed release steroid in 2008 (Buttgereit et al., 2008), with approval by the FDA of the relevant drug to treat RA in 2012.
Reports were published of melatonin’s ability to entrain free-running rhythms in rodents in 1984 (Redman et al., 1983), a phase response curve in humans to melatonin in 1998 (Lewy et al., 1998), multiple studies of melatonin entraining free-running rhythms in blind people in 1991-2004 [reviewed in (Arendt and Skene, 2005)], the ability of a melatonin agonist to phase shift melatonin rhythms of rodents in 2005 (Hirai et al., 2005) and humans in 2009 (Rajaratnam et al., 2009), and to entrain free-running rhythms in blind people in 2015 (Lockley et al., 2015), with approval by the FDA of the relevant drug to treat non-24-sleep-wake disorder in blind people in 2014.
Conclusion
There is an urgent need for more clinical trials of circadian medicine approaches (diagnostics and therapeutics), in each of the stages mentioned. Only a vigorous and systematic collection of evidence in all stages will give us the opportunity to ensure that the promising approaches of circadian medicine really benefit all patients. The challenge is that each stage usually takes years to complete study design, patient recruitment and follow-up, analyses, and publication. It is important to recognize that failure is possible and, from experience, likely at each phase. However, for good reasons, successfully completion of all phases is required by most official regulatory agencies in each country/region (e.g. USA FDA and European Union European Medicines Agency [EMA]) and clinical organizations. To accelerate these processes and make them efficient, we must also advocate for increased funding for all steps of this process.
Despite this necessary staying power, we are convinced that if circadian medicine, as an emerging field, pools its energy now and tackles the stages outlined above with courage, perseverance, and rigorous scientific and clinical standards, it will be extremely beneficial to our patients and general health and safety. It is time that we take time seriously in medicine.
Footnotes
Acknowledgements
Klerman: NIH R01-NS099055, U01-NS114001, U54-AG062322, R21-DA052861, R01-NS114526-02S1, R01-HD107064; DoD W81XWH201076; Leducq Foundation for Cardiovascular Research. Kramer: German Research Foundation (TRR186—project number 278001972 and KR1989/12-1). Zee: NIH R01-HL140580; P01-AG011412; R01-AG059291; R01-HD097786; R01-HL14188; R01-HL1418811; R01-AG070867; R01-MH126911
Conflict of Interest Statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Klerman: Consulting for American Academy of Sleep Medicine Foundation, Circadian Therapeutics, National Sleep Foundation, Sleep Research Society Foundation, Yale University Press; travel support from European Biological Rhythms Society; partner owns Chronsulting. Kramer: Consulting for Stanford University and Circadian Therapeutics; shareholder of BodyClock GmbH. Zee: Consulting for Eisai, Jazz, Idorsia, Sanofi-Aventis, CVS Caremark, WebMD; Stock ownership: Teva
