Abstract
Delayed sleep phase syndrome (DSPS) is the most common sleep disturbance in adults with attention-deficit/hyperactivity disorder (ADHD). We previously showed that chronotherapy with melatonin effectively advanced the dim-light melatonin onset (DLMO), a biomarker for the internal circadian rhythm, by 1.5 h and reduced ADHD symptoms by 14%. Melatonin combined with bright light therapy (BLT) advanced the DLMO by 2 h, but did not affect ADHD symptoms. This article explores whether sleep times advanced along with DLMO, leading to longer sleep duration and better sleep in general, which might explain the working mechanism behind the reduction in ADHD symptoms after treatment with melatonin. This article presents exploratory secondary analysis on objective and self-reported sleep characteristics from a three-armed double-blind randomized placebo-controlled clinical trial (RCT), which included 49 adults (18-55 years) with ADHD and DSPS. Participants were randomized to receive sleep education and 3 weeks of (1) 0.5 mg/day placebo, (2) 0.5 mg/day melatonin, or (3) 0.5 mg/day melatonin plus 30 min of bright light therapy (BLT) between 0700 and 0800 h. Sleep was assessed at baseline, directly after treatment, and 2 weeks after the end of treatment. Objective measures were obtained by actigraphy, self-reported measures by various sleep questionnaires and a sleep diary. Melatonin with or without BLT did not advance sleep times, improve sleep in general, or strengthen wake-activity rhythms. So even though the DLMO had advanced, sleep timing did not follow. Adding extensive behavioral coaching to chronotherapy is necessary for advancing sleep times along with DLMO and to further alleviate ADHD symptoms.
Keywords
Attention-deficit/hyperactivity disorder (ADHD) in adults is often concurrent with sleep disorders (Bijlenga et al., 2019). As many as 78% of adults with ADHD have a delayed circadian rhythm, as objectively marked by a delay in their dim-light melatonin onset (DLMO) measured in saliva (Van Veen et al., 2010). A delayed circadian rhythm often causes an inability to fall asleep or wake up at a desired time and may lead to a diagnosis of circadian rhythm sleep disorder-delayed sleep phase type (CRSD-DSPT), also termed delayed sleep phase syndrome (DSPS; American Psychiatric Association, 2013).
Even in the absence of a formal sleep disorder diagnosis, sleep is often disturbed in adults with ADHD. A recent meta-analysis investigated the differences in self-reported and objective sleep measures between adults with and without ADHD (Díaz-Román et al., 2018). Adults with ADHD reported longer sleep onset latency (Arns et al., 2014; Boonstra et al., 2007) and more awakenings during the night resulting in lower sleep efficiency (Arns et al., 2014; Boonstra et al., 2007). Compared with the controls, ADHD is associated with more general sleep problems (Arns et al., 2014; Philipsen et al., 2005; Weibel et al., 2017), more daytime sleepiness (Bioulac et al., 2020; Sobanski et al., 2016), and lower sleep quality (Boonstra et al., 2007; Kooij et al., 2001; Philipsen et al., 2005; Weibel et al., 2017). ADHD symptom severity has been related to delayed sleep timing and daytime sleepiness (Gamble et al., 2013). Overall, adults with ADHD experience more disturbed and less restful sleep than adults without ADHD.
Despite the many differences in self-reported sleep between adults with ADHD and controls, objective measures generally show less dissimilarity. This is not surprising since they relate to different constructs. The meta-analysis by Díaz-Román and colleagues (2018) showed that in actigraphy studies, adults with ADHD had longer sleep onset latency and lower sleep efficiency than adults without ADHD. No differences were found in studies using other objective measures (Díaz-Román et al., 2018).
It is clear that ADHD and sleep, and particularly a delayed circadian rhythm, are strongly intertwined (Bijlenga et al., 2019). ADHD and sleep problems seem to have a reciprocal relationship and may have a shared etiology (Hvolby, 2015). Several studies in children support this notion, as ADHD symptoms decreased after treatment of specific sleep disorders (Corkum et al., 2016; Huang et al., 2007; Walters et al., 2000). Less is known about the effect of treatment of DSPS, which often consists of sleep hygiene and chronotherapy with melatonin and/or bright light therapy (BLT) with the intention of advancing the circadian and sleep rhythm (Lack and Wright, 2007). Two small studies suggested that BLT could advance the rhythm and reduce ADHD symptoms even in study populations that did not exclusively consist of people with a delayed sleep phase (Fargason et al., 2017; Rybak et al., 2006).
We recently presented the first results from the randomized clinical controlled PhASE trial (Phase shift in adult ADHD of Sleep and Appetite), investigating the effects of DSPS treatment on DLMO and ADHD symptoms in adults with ADHD and DSPS (van Andel et al., 2021). A low dose of melatonin (0.5 mg) effectively advanced the DLMO by 1.5 h and decreased ADHD symptoms by 14%; ADHD Rating Scale score went from 33 to 27, where a score of 28 or higher indicates a clinical level of ADHD symptoms (DuPaul et al., 1998). The 14% decrease was considered clinically relevant, even though remission requires a reduction of 30% (Kooij et al., 2004; Spencer et al., 1995). Surprisingly, melatonin in combination with BLT did not affect ADHD symptoms, despite a 2 h advance in DLMO (van Andel et al., 2021). We suggested this absence of additional effects of BLT may have been due to sleep times not advancing along with DLMO, resulting in shorter sleep duration as this group had to get up between 0700 and 0730 h for BLT. This was the standard BLT protocol at our clinic at the time of study implementation, already a decade ago. It was aimed at treating seasonal depression rather than DSPS and we now realize this was a major limitation in our study protocol.
This article extends our previous work by presenting exploratory secondary analyses on the effects of the chronotherapeutic interventions on sleep characteristics and sleep-related measures, including actigraphy, symptoms of various sleep disorders and sleep-related problems, and self-reported experience of sleep. We expect sleep and wake times to advance along with DLMO after treatment with melatonin with and without BLT, both objective and self-reported measures of sleep to improve, and the circadian rhythm to become stronger.
Materials and Methods
Study Design
This article presents secondary analysis of data from the PhASE study (Phase Shift in ADHD of Sleep and Appetite). PhASE is a three-armed (1:1:1) placebo-controlled randomized clinical trial (RCT) studying the effects of chronotherapy in adults with ADHD and DSPS on DLMO, sleep, appetite hormones, and biomarkers for several chronic diseases, as ADHD is associated with poorer health (Instanes et al., 2018). The three conditions were sleep education plus a 3-week intervention period of (1) 0.5 mg/d melatonin (MEL), (2) 0.5 mg/d placebo (PLAC), or (3) 0.5 mg/d melatonin plus BLT (MEL + BLT). Two arms were double-blind (PLAC and MEL); researchers and participants were not blind to the melatonin plus BLT condition. PhASE was approved by the Medical Ethical Committee of Leiden, protocol #NL39579.058.12, and registered in the Netherlands Trial Register, #NTR3831. The study protocol complies with the Helsinki Declaration of 1975, as revised in 2008, and international ethical standards for biological rhythms research (Portaluppi et al., 2010). More information on the study design and interventions can be found in our previous report, where sample size calculation and the randomization and blinding process are described as well (van Andel et al., 2021).
Participants
Participants were recruited from the specialized PsyQ out-patient adult ADHD clinic in The Hague, The Netherlands from June 2013 to August 2016 and from April 2018 to June 2019. Patients aged 18 to 55 years with a clinical diagnosis of both ADHD and DSPS, based on the
Interventions
Following standard protocol used at PsyQ Program Adult ADHD in The Hague, The Netherlands, participants received one face-to-face psycho-education session on sleep and rhythm from the researcher the day before the start of treatment. Recommendations with regard to sleep hygiene were extensively discussed and individual advice was given and the participant then received the allocated 3-week intervention. After the first and second week, the researcher telephoned the participant to ask about their experiences and their adherence to sleep hygiene recommendations, and to motivate them to further improve their sleep hygiene and advance their bed times.
Melatonin or Placebo
Study medication consisted of 0.5 mg of either melatonin or placebo, produced by an independent pharmacist. The medication was taken daily for 3 weeks, following an individualized intake schedule based on baseline DLMO. Medication intake in the first week took place 3 h before individual DLMO and advanced weekly by one hour, up to 5 h before individual baseline DLMO in the last week (van Andel et al., 2021) and participants were instructed to go to bed as soon as they began to feel tired. To measure compliance, participants wrote down daily whether they had taken their study medication and at what time. The number of medication intakes was checked by counting any remaining tables after the 3-week treatment period.
BLT
To study additional effects of BLT, participants in the MEL + BLT group took home a Philips Energy Lightbox® Type HF3308/01, which emits light with an intensity of 10,000 lux. BLT was used daily at 20 cm from the eyes without glasses or contact lenses, for 30 min between 0700 and 0800 h. This protocol was aimed at treatment of seasonal depression rather than DSPS, for which no specific protocol existed at our clinic at the time of study implementation. To measure compliance, participants wrote down daily whether they had followed BLT and at what time. After the 3-week intervention period, participants rated the ease of adhering to the BLT schedule on a 10-point Likert-type scale ranging from 1 (very difficult) to 10 (very easy).
Study Assessments
Objective and self-reported sleep characteristics were assessed at baseline (T0), the day after the 3-week treatment period (T1), and at 3 weeks follow-up after end of treatment (T2).
General Characteristics
Age, sex, ADHD subtype (combined, inattentive, or hyperactive/impulsive), smoking (yes/no), and body mass index (BMI; kg/m2) were assessed at T0.
Objective Sleep Characteristics
To objectively assess sleep at T0, T1, and T2, participants wore an actometer (Actiwatch Score, Mini Mitter, Respironics; Bend, Oregon) on their non-dominant wrist for 3 consecutive nights before free days. The actigraphic data were analyzed using Respironics Actiware 5 (Philips Respironics, Bend, OR) and data from times when the actometer had not been worn were manually excluded from further analysis. A standard algorithm was used to obtain sleep characteristics, from which the following variables were calculated: sleep onset time, wake time, sleep onset latency (time between going to bed and falling asleep), wake after sleep onset (WASO) duration, average number and duration of wake bouts, and net sleep duration (total time spent asleep during time in bed). The DLMO-midsleep phase angle difference (PAD), that is, the time between DLMO and midsleep, was calculated as a marker for internal circadian alignment: PAD = DLMO – midsleep, where midsleep = sleep onset + (sleep offset – sleep onset)/2. For all variables, the average of the 3 nights was calculated and used for subsequent analyses.
In addition, nonparametric circadian rhythm analyses (NPCRA) were performed to study the strength of the circadian rest-activity rhythm (Van Someren et al., 1998; Witting et al., 1990). A full hour was excluded when the actimeter had been taken off for more than 15 min during that hour. The interdaily stability (IS) represents the strength of the coupling of the sleep-wake rhythm to supposedly stable zeitgebers, with higher values indicating better synchronization to external zeitgebers and thus a strong circadian rhythm. The intradaily variability (IV) reflects fragmentation of rest-activity rhythms, with high IV indicating a high number of transitions between rest and activity, such as naps or nighttime arousals. The relative amplitude (RA) is based on the difference between the mean activity levels during the 5 least active (L5) and the 10 most active hours (M10) in a 24-h period: RA = (M10-L5)/(M10 + L5) (Van Someren et al., 1998; Witting et al., 1990). A higher RA indicates a strong circadian rhythm with strong and regular differences between periods of sleep and activity.
Self-reported Sleep Characteristics
The Sleep Diagnosis List (SDL), a validated Dutch variant of the Sleep Diagnosis Questionnaire (SDQ), assessed indications of 13 common sleep disorders and sleep-related problems: insomnia (Cronbach’s α = 0.93), anxiety/depression (Cronbach’s α = 0.92), restless legs syndrome (Cronbach’s α = 0.90), sleep apnea (Cronbach’s α = 0.88), hypersomnia (Cronbach’s α = 0.81), daytime dysfunctioning (Cronbach’s α = 0.80), hypnagogic hallucinations/dreaming (Cronbach’s α = 0.76), sexual and social dissatisfaction (Cronbach’s α = 0.73), sleep paralysis (Cronbach’s α = 0.70), cataplexy (Cronbach’s α = 0.64), restless sleep (Cronbach’s α = 0.74), negative conditioning surrounding sleep and the bedroom (Cronbach’s α = 0.49), and automatic behavior (Cronbach’s α = 0.69) (Sweere et al., 1998a, 1998b). Each factor had a continuous value (0-5) used in statistical analyses, with higher values representing stronger indications, and a binary value, with a score of >3 representing a positive screen.
The Sleep Hygiene Questionnaire (SHQ) evaluated sleep hygiene. It was based on the Adolescent Sleep Hygiene Scale (LeBourgeois et al., 2004) and included 22 items on a 5-point Likert-type scale ranging from 1 (never) to 5 (always). Better sleep hygiene was reflected by lower scores on the total score ranging from 22 to 100.
In addition, participants kept a daily sleep diary during the 3 weeks of study intervention in which they estimated their sleep and wake times, sleep onset latency, and the number of awakenings. They also rated the difficulty of falling asleep, sleep quality, and at the end of the day how well-rested they had felt that day on 5-point Likert-type scales ranging from 1 (not at all/very bad) to 5 (a lot/very well). Average values for each week of treatment were calculated for all values.
DLMO and ADHD-RS
We used total score on the ADHD Rating Scale (ADHD-RS) and, extending our previous work (van Andel et al., 2021), calculated inattention and hyperactivity-impulsivity subscores (DuPaul et al., 1998; Kooij et al., 2005). DLMO was measured by radio-immunoassay at the Gelderse Vallei Hospital (Ede, The Netherlands) from saliva taken hourly between 2000 and 0300 h. DLMO measurements always took place at least a day after the last melatonin (or placebo) intake. Time of DLMO was defined as the time at which the melatonin concentration crossed the threshold of 3.0 pg/mL (Benloucif et al., 2008) and calculated by linear interpolations of melatonin concentration between time points. A full description of DLMO and ADHD-RS assessment was reported previously (van Andel et al., 2021).
Analyses
To check for any differences in general characteristics or study outcome variables between randomization groups at baseline, we used analyses of variance (ANOVAs) for continuous variables, Fisher’s exact test for categorical variables including cell counts below 5, and chi-square tests for categorical variables with all cell counts ≥5. The effects of the three interventions (MEL, PLAC, and MEL + BLT) on various objective and self-reported sleep variables were explored using linear mixed model analyses corrected for baseline values of outcomes and with T0 as reference, or week 1 for sleep diary variables. Multiple testing adjustments were considered, but any corrections would greatly reduce statistical power. Instead, interpretation was focused on finding patterns rather than any particular associations. Analyses with non-normally distributed variables were repeated with ln-transformed data. Since this did not impact the results, regression coefficients (β) and Cohen’s
We then investigated which objective or self-reported sleep characteristics were related to ADHD symptoms: the effects of the various sleep variables (and DLMO) on ADHD-RS score over the entire study period for all randomization groups were investigated using linear mixed models clustered at the individual level.
Cohen’s
Results
Baseline data were available for
General characteristics and study outcomes at baseline (T0) for total study population (
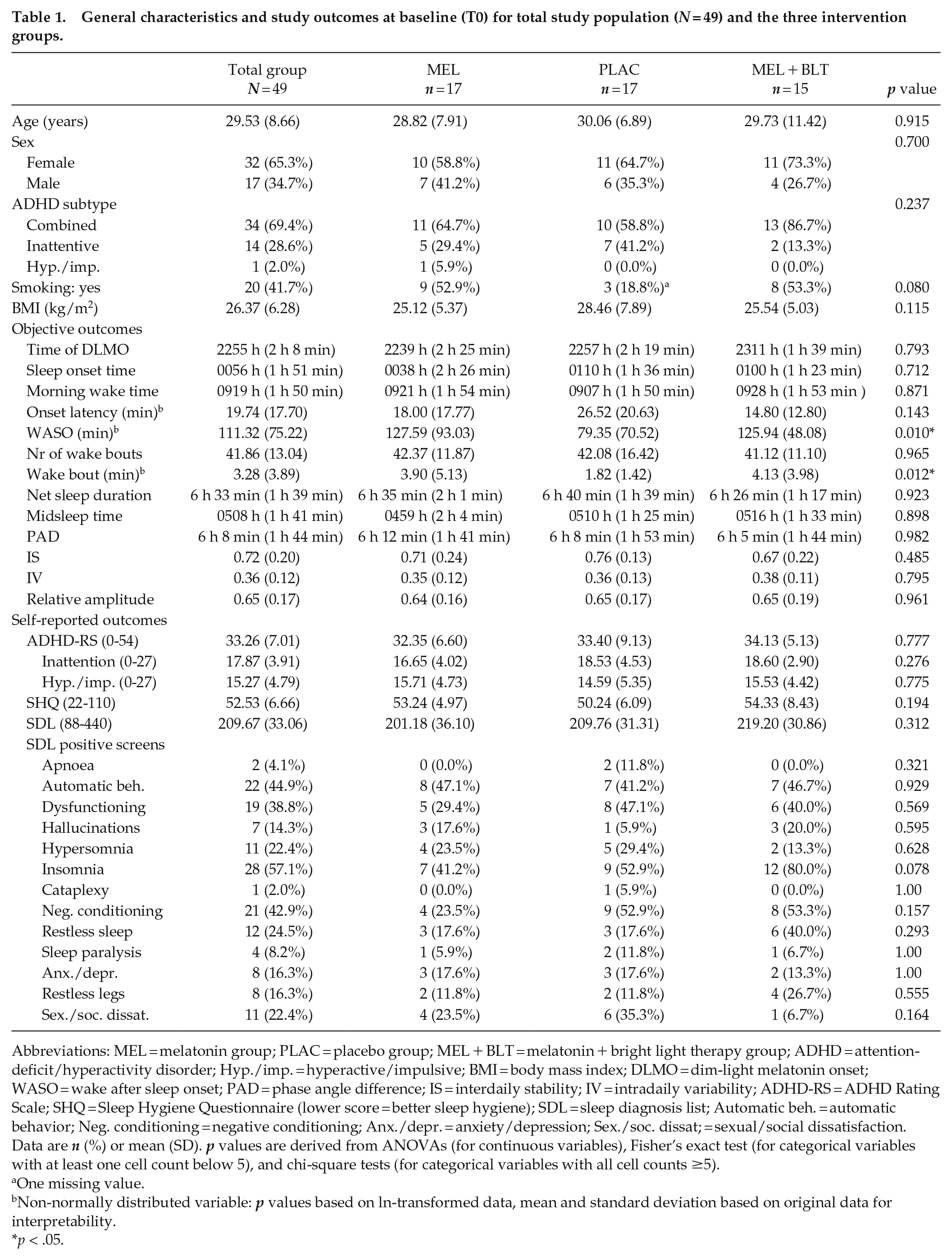
Abbreviations: MEL = melatonin group; PLAC = placebo group; MEL + BLT = melatonin + bright light therapy group; ADHD = attention-deficit/hyperactivity disorder; Hyp./imp. = hyperactive/impulsive; BMI = body mass index; DLMO = dim-light melatonin onset; WASO = wake after sleep onset; PAD = phase angle difference; IS = interdaily stability; IV = intradaily variability; ADHD-RS = ADHD Rating Scale; SHQ = Sleep Hygiene Questionnaire (lower score = better sleep hygiene); SDL = sleep diagnosis list; Automatic beh. = automatic behavior; Neg. conditioning = negative conditioning; Anx./depr. = anxiety/depression; Sex./soc. dissat; = sexual/social dissatisfaction. Data are
One missing value.
Non-normally distributed variable:
Baseline Characteristics
Table 1 shows general characteristics and outcome values at baseline (T0) for the total group (
Effects of Interventions on Sleep
The raw data of study outcomes for the three randomization groups across time are reported in Table 2. The results of the linear mixed models investigating the effects of interventions on various sleep outcomes are displayed in Table 3 (objective measures) and Table 4 (self-reported measures).
Values of study outcomes for the three randomization groups at T0, T1, and T2 or weeks 1 and 3.

Abbreviations: MEL = melatonin group; PLAC = placebo group; MEL + BLT = melatonin + bright light therapy group; DLMO = dim-light melatonin onset; WASO = wake after sleep onset; SHQ = Sleep Hygiene Questionnaire (lower score = better sleep hygiene); SDL = sleep diagnosis list; ADHD-RS = attention-deficit/hyperactivity disorder Rating Scale.
Effects of the three interventions on objective sleep: linear mixed model analyses (
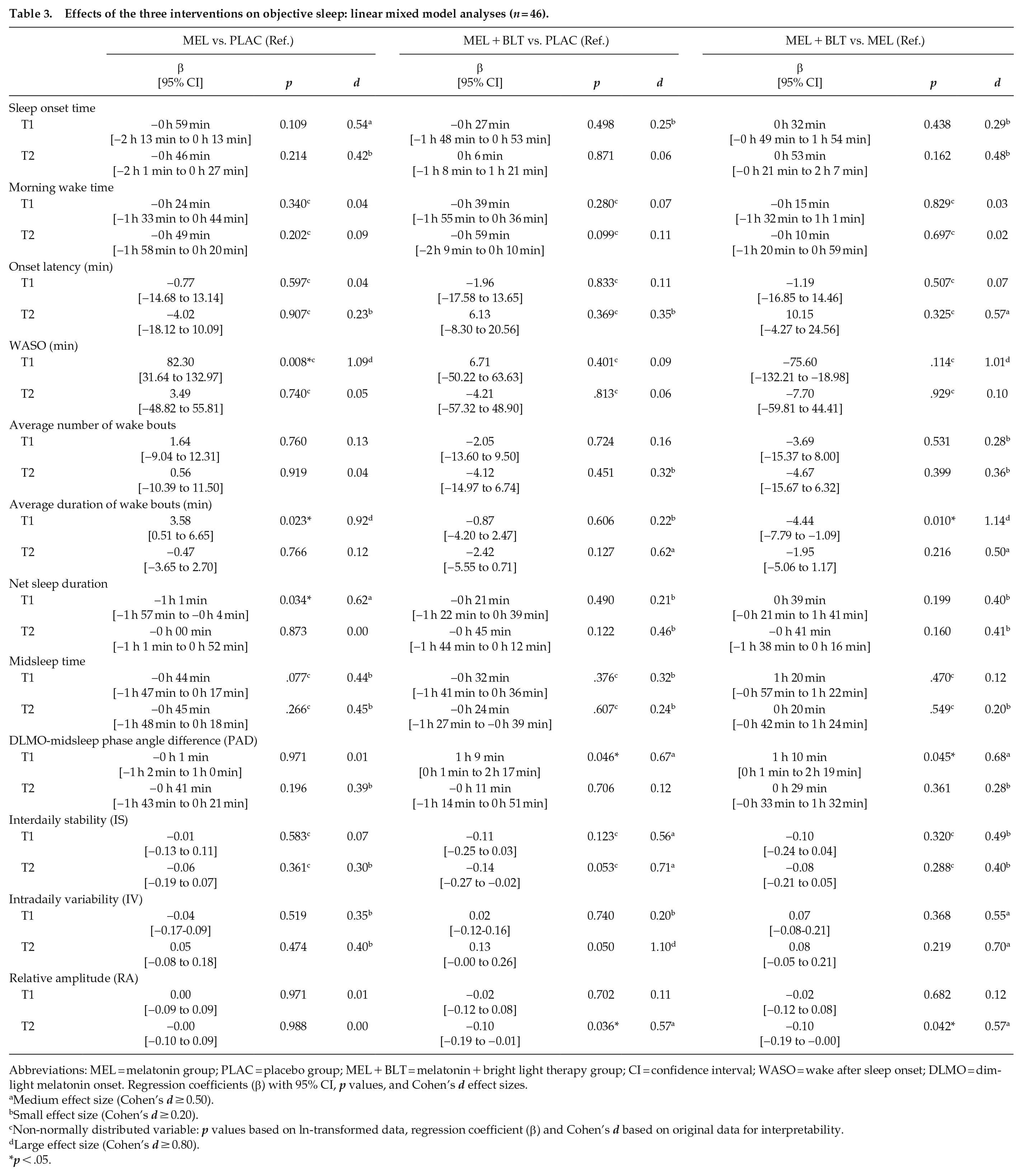
Abbreviations: MEL = melatonin group; PLAC = placebo group; MEL + BLT = melatonin + bright light therapy group; CI = confidence interval; WASO = wake after sleep onset; DLMO = dim-light melatonin onset. Regression coefficients (β) with 95% CI,
Medium effect size (Cohen’s
Small effect size (Cohen’s
Non-normally distributed variable:
Large effect size (Cohen’s
Effects of the three interventions on self-reported sleep and ADHD-RS scores: Linear mixed model analyses (
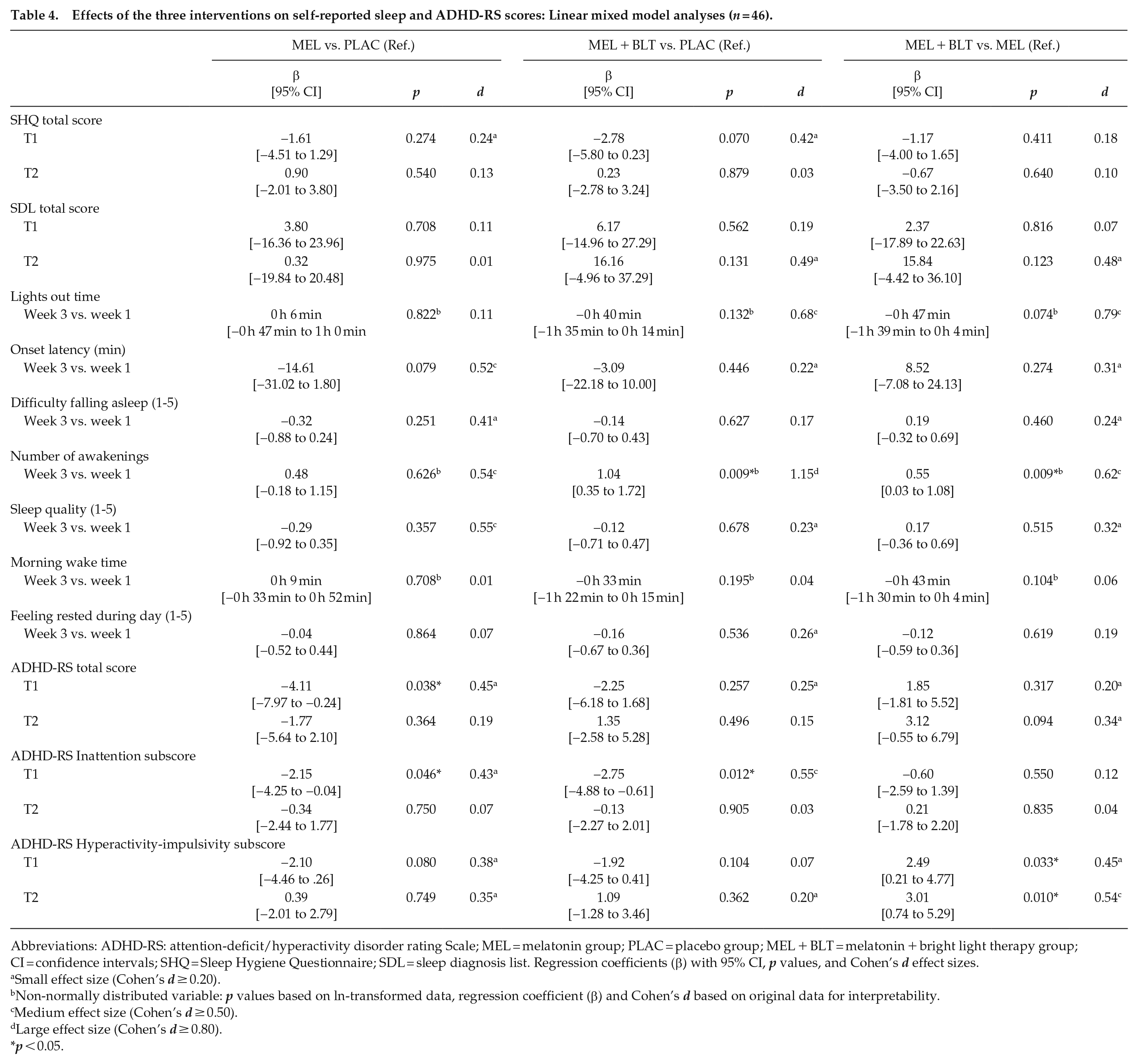
Abbreviations: ADHD-RS: attention-deficit/hyperactivity disorder rating Scale; MEL = melatonin group; PLAC = placebo group; MEL + BLT = melatonin + bright light therapy group; CI = confidence intervals; SHQ = Sleep Hygiene Questionnaire; SDL = sleep diagnosis list. Regression coefficients (β) with 95% CI,
Small effect size (Cohen’s
Non-normally distributed variable:
Medium effect size (Cohen’s
Large effect size (Cohen’s
Objective Sleep Characteristics
Due to the inflexible planning of the study schedule, it was not always practically feasible to collect data from 3 consecutive nights before free days only. The number of work days during the actigraphy measurements was not quantified, but estimated at 10% across the whole study, based on comparisons between wake times in the actigraphy data and self-reported wake times during the treatment period. Because of the small estimated percentage of work days, comparisons between free and work days could not be made, and analyses were performed on all data.
Sleep onset and wake times had not significantly advanced in any group at either T1 or T2. Yet, sleep onset in the MEL group at T1 was 59 min earlier compared with PLAC, which was not statistically significant but had a medium effect size,
The NPCRA analyses included on average 65.48 (SD 13.92) valid hours with a median of 68 h. The number of valid hours was not related to any of the outcomes. Only the fragmentation of rest-activity rhythms (IV) was increased in the MEL + BLT group compared with PLAC at T2, β = 0.13,
Self-reported Sleep Characteristics
Sleep hygiene (SHQ score) had improved over time in all randomization groups (not shown in Table 4), but did not differ between groups at T1 or T2. SDL total score did not change significantly over time, although indications on two SDL subscale scores were significantly stronger with small to medium effect sizes (not shown in Table 4): SDL hallucinations was higher in MEL + BLT than PLAC at T2, β = 0.35,
Sleep diary variables had no T0 assessment, so averages of the third week of intervention were compared with averages of the first week. Difficulty falling asleep and sleep quality did not change during the 3-week treatment period and none of the groups reported becoming more rested. The MEL + BLT group turned off the lights 47 min earlier than MEL,
ADHD-RS scores (total and subscores for inattention and hyperactivity-impulsivity) are also included in Table 4. As reported previously, total ADHD-RS score had decreased in the MEL group at T1,
Effects of DLMO and Sleep Variables on ADHD
To globally explore which sleep characteristics were associated with a change in ADHD-RS score, the associations between DLMO, sleep variables, and ADHD-RS score over the entire study period for all randomization groups were investigated. The results of the linear mixed model analyses are shown in Table 5. Of the objective measures, only DLMO was significantly related to ADHD-RS score albeit with a very small effect size, β = 0.868,
Associations between sleep variables and ADHD-RS score over the entire study period (T0, T1, T2) for the total group (
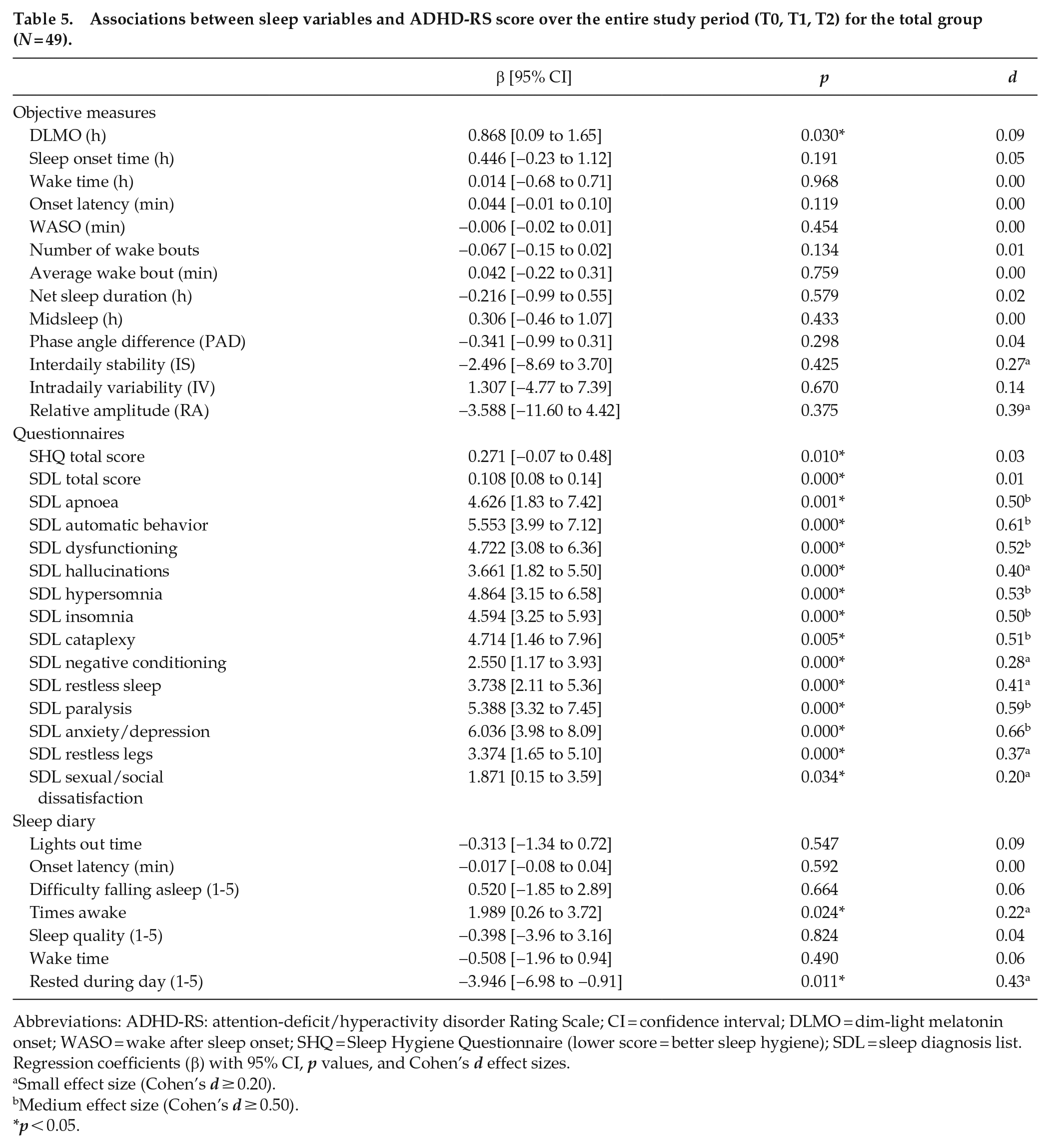
Abbreviations: ADHD-RS: attention-deficit/hyperactivity disorder Rating Scale; CI = confidence interval; DLMO = dim-light melatonin onset; WASO = wake after sleep onset; SHQ = Sleep Hygiene Questionnaire (lower score = better sleep hygiene); SDL = sleep diagnosis list. Regression coefficients (β) with 95% CI,
Small effect size (Cohen’s
Medium effect size (Cohen’s
Discussion
This exploratory secondary analysis of an RCT studied the effects of chronotherapy on sleep characteristics in adults with ADHD and DSPS. We previously confirmed our hypothesis that the DLMO, a marker for internal circadian rhythm, can advance after treatment with melatonin with and without BLT (van Andel et al., 2021). However, this does not automatically imply that sleep in general will improve, so we currently investigated whether sleep times had advanced along with DLMO and if objective and self-reported sleep had improved.
In contrast to our expectations, sleep and wake times had not advanced along with DLMO after treatment with melatonin or melatonin plus BLT. Moreover, we found no evidence that the interventions had improved sleep in general or strengthened the circadian rhythm, even though sleep hygiene had improved. So even though chronotherapeutic treatment with melatonin with or without BLT advanced the DLMO and made it biologically possible for participants to fall asleep earlier, their sleep times did not change. Only targeting the biology thus seems insufficient for changing long-standing sleep behaviors. Our current DSPS treatment is based on cognitive behavioral therapy for insomnia (CBTi; Baglioni et al., 2020) and includes more extensive behavioral coaching, where concrete, individual goals are set and compliance is frequently monitored, which shows promising results.
Overall, self-reported measures appeared to be most strongly related to ADHD-RS score. This could, however, not explain the reduction in ADHD symptoms seen in the MEL group after treatment, as the self-reported measures had not changed in this group. Moreover, DLMO had a small but statistically significant effect on ADHD-RS score in the whole sample and the observed advances could play a role in the relationships between interventions, sleep, and ADHD-RS score, although how exactly remains unclear. Future studies are needed to elucidate the working mechanisms behind the reduction in ADHD symptoms after treatment with melatonin (van Andel et al., 2021).
Unexpectedly, melatonin-only treatment increased the average wake bout duration, causing longer WASO and shorter net sleep duration. We reckoned the administered melatonin could wear off after a couple of hours and endogenous production may lag behind. However, the raw actigraphy data showed no evidence for this and the same should then be seen in the MEL + BLT group. We could thus not explain this result, so further research is necessary to establish the cause of this effect or whether it was a chance finding.
The MEL + BLT group showed no reduction in ADHD symptoms despite a large advance in DLMO. We previously suggested this may be because this group was strictly instructed to get up early for BLT, which half of the group reported to find very difficult and which may have prevented any reduction in ADHD symptoms (van Andel et al., 2021). Still, adherence to the schedule was good and this group turned off their lights an hour earlier during the third week of treatment compared with the first. Directly after treatment, however, objectively measured sleep onset time was not different from baseline. Remarkably, 2 weeks after the end of treatment this group scored worse on a few SDL subscales and also showed a weaker circadian rhythm: IS and RA were lower and IV was higher, indicating weaker coupling of the rhythm to zeitgebers, smaller differences between periods of rest and activity, and more fragmentation, respectively. These findings suggest that the MEL + BLT group may have overcompensated for built-up sleep debt resulting from the instructed early rise times during the 3 weeks of treatment.
At the time of study implementation, BLT at our clinic was given between 0700 and 0800 h to treat seasonal depressive symptoms. This protocol was incorporated into the PhASE study to treat DSPS, for which no protocol existed in our clinic at the time. It is now clear this is a major limitation, as administering BLT too early in one’s circadian rhythm can actually cause a delay. Melatonin plus BLT, nevertheless, effectively advanced the DLMO by 2 h. Additional effects of BLT could thus not be established, but individualizing BLT timing will likely prove more effective in advancing the DLMO and perhaps further reducing ADHD symptoms (Bjorvatn and Pallesen, 2009; Fargason et al., 2017; Lack and Wright, 2007; Terman and Terman, 2005a, 2005b).
Some other limitations need also be mentioned. First, the sample size was relatively small for linear mixed model analyses and the exploratory nature of the analyses and the large number of outcomes analyzed cause a high risk of type I errors and means results should be interpreted with caution. Moreover, adding covariates would compromise the reliability of the models. Second, the actigraphy and NPCRA variables were calculated on the basis of a maximum of 3 nights. Given the day-to-day variability in activity and sleep behavior, actigraphic estimates of sleep and circadian variables have a relatively large measurement error if calculated over only few days. The measurement error decreases with an increasing number of days. However, no systematic errors occur with assessments of short duration. Several studies found sufficient sensitivity to group differences or intervention effects even with a limited number of recording days. Given the limited number of recording days, we should be extra careful not to conclude absences of differences or effects if they were not found in the present study. Moreover, future studies should include logs kept during actigraphy measurements, so self-reported sleep and wake times can be matched to actigraphy data to generate more reliable results, and a distinction can be made between work and free days. Especially the accuracy of sleep onset latency would benefit from longer measurement and sleep logs, as it is difficult to distinguish between quiet wakefulness and sleep. Still, the currently reported onset latency of 19.74 min is comparable with ADHD populations in previous studies, which did include 7 days of monitoring and sleep logs (Boonstra et al., 2007; Kooij et al., 2001; Tonetti et al., 2017; Van Veen et al., 2010; for systematic review and meta-analysis see Díaz-Román et al., 2018). Third, sleep diary variables were not assessed before treatment, so only their development during 3-week treatment could be studied. Fourth, the study population had little comorbidity and is therefore not representative of the entire adult ADHD population. Finally, this was the first RCT on chronotherapy and ADHD symptoms and the focus was therefore mostly on the chronotherapeutic interventions. The current instructions on and monitoring of sleep hygiene behaviors were too lenient to generate behavioral changes in sleep times: participants received one face-to-face session with sleep education and personalized sleep hygiene recommendations and weekly telephone calls to monitor and further motivate their adherence to sleep hygiene recommendations. Stricter instructions and more frequent monitoring are necessary to change long-standing sleep behaviors. However, the fact that interventions took place in the participants’ naturalistic home environment, and a broad range of both objective and self-reported measures of sleep was included, is a major strength of this study. Another strength is the broad age range of participants. Rhythm and sleep change with age and the circadian rhythm reaches a peak in lateness in late adolescence/early adulthood (Roenneberg et al., 2004). In the general adult population, DSPS is indeed age-specific to (late) adolescents and the prevalence declines with age, but DSPS in ADHD is not age-related (Bijlenga et al., 2013), which was further evidenced by the lack of relationships between age and any of the current outcomes.
Despite these limitations, the current results are clinically relevant. They show that sleep times did not advance and sleep in general did not improve in adults with ADHD and DSPS after treatment with melatonin, with or without BLT, despite advances in DLMO and improvements in sleep hygiene. In other words, participants did not go to bed earlier despite being biologically prepared for sleep and instructed to go to bed as soon as they started feeling tired. We therefore recommend complementing chronotherapeutic interventions for DSPS with extensive behavioral coaching on sleep hygiene and sleep times. This way, sleep onset time, and consequently wake time, may advance along with DLMO and in turn further alleviate ADHD symptoms. Extensive behavioral coaching with concrete, individual goals and frequent monitoring, based on CBTi (Baglioni et al., 2020), is part of our current standard protocol for DSPS treatment, with promising results that are presently being systematically investigated. With respect to BLT, we recommend individualizing the schedule and gradually advancing BLT timing (Bjorvatn and Pallesen, 2009; Lack and Wright, 2007; Terman and Terman, 2005a, 2005b). Studies in larger samples implementing the aforementioned changes are expected to find advances in sleep timing along with advances in DLMO, better sleep, and a stronger rhythm in general, and consequently a stronger reduction of ADHD symptoms.
The current study shows that sleep times did not advance and sleep in general did not improve by advancing DLMO through melatonin treatment. Future research should include larger samples and extensive behavioral coaching to further investigate the working mechanisms behind the reduction in ADHD symptoms seen after DSPS treatment. This way, sleep characteristics that have the strongest effect on decreasing ADHD symptoms can be identified and targeted more specifically in treatment of DSPS in adults with ADHD.
Footnotes
Acknowledgements
The authors thank all participants for their time and effort, the team at PsyQ Program Adult ADHD for their help with inclusion, and Eus van Someren for calculating the NPCRA variables. PhASE was funded by an unrestricted grant from research fund Fonds NutsOhra (FNO #1102-026).
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
