Abstract
Mammals have evolved circadian rhythms in internal biological processes and behaviors, such as locomotor activity (LA), to synchronize to the environmental conditions they experience. Photic entrainment of LA has been well established; however, non-photic entrainment, such as ambient temperature (Ta), has received much less attention. To address this dearth of knowledge, we exposed two subterranean endothermic-homeothermic African mole-rat species, the solitary Cape mole-rat (Georychus capensis [GC]) and social Mahali mole-rat (Cryptomys hottentotus mahali [CHM]), to varying Ta cycles in the absence of light. We showed that the LA rhythms of these two species entrain to Ta cycles and that the majority of LA occurred during the coolest 12-h period. LA confined to the coolest Ta periods may be the direct consequence of the poor heat dissipation abilities of African mole-rats brought about by physiological and ecological constraints. Recently, it has been hypothesized that Ta is only a strong zeitgeber for circadian rhythms in species whose thermoregulatory abilities are sensitive to changes in Ta (i.e., heterotherms and ectotherms), which previously has excluded endothermic-homeothermic mammals. However, this study demonstrates that Ta is a strong zeitgeber or entrainer for circadian rhythms of LA in subterranean endothermic-homeothermic mammals as a consequence of their sensitivity to changes in Ta brought about by their poor heat dissipation abilities. This study reinforces the intimate link between circadian rhythms and thermoregulation and conclusively, for the first time, provides evidence that Ta is a strong zeitgeber for endothermic-homeothermic mammals.
Mammals have evolved morphological, physiological, and behavioral traits in response to predictable environmental fluctuations (Piersma and Drent, 2003). One crucial trait is the ability to synchronize their internal biological processes and behaviors, such as locomotor activity (LA) patterns, to cyclic patterns of abiotic variables, for example, daily changes in light or ambient temperature (Ta) (Cloudsley-Thompson, 1961; Aschoff, 1966; Daan and Aschoff, 1975; Patel et al., 2014). The adaptive significance of innate circadian rhythmicity is thought to be related to the changes that arise in the environment of an organism, such as predator avoidance or food availability (so-called extrinsic advantages) (Boulos and Terman, 1980; O’Reilly et al., 1986; Boulos et al., 1989; Lima and Dill, 1990; Vaze and Sharma, 2013; Alagaili et al., 2020). It may also be through benefits derived from orchestrating internal biological processes such as maintaining metabolic activity and core body temperatures (Tb) (i.e., intrinsic advantage) (Boulos and Terman, 1980; O’Reilly et al., 1986; Boulos et al., 1989; Lima and Dill, 1990; Vaze and Sharma, 2013; Alagaili et al., 2020).
Light is arguably the most influential zeitgeber for the entrainment of circadian rhythms, and in particular, LA (DeCoursey and DeCoursey, 1964; Stephan and Zucker, 1972; Daan and Aschoff, 1975; Erkert et al., 1986; Goldman, 1999; Mahoney et al., 2001; Okamura et al., 2002; Verwey et al., 2013). It follows that the neuroanatomical pathways related to photic entrainment of the master circadian oscillator are well established (Reppert and Weaver, 2000; Shearman et al., 2000; Reppert and Weaver, 2001; Okamura et al., 2002; Mohawk et al., 2012). In contrast, non-photic entrainment of the circadian clock and the importance of circadian rhythms in conditions void of photic stimuli are far less understood.
In environments where reliable photic cues are absent, circadian rhythmicity has been demonstrated in several taxa, ranging from deep-sea or cave-dwelling invertebrates to subterranean rodents (reviewed by Abhilash et al. (2017)). Given the relatively stable environments of these dark habitats, the adaptive advantage of these circadian rhythms is primarily related to orchestrating internal physiological processes (Vaze and Sharma, 2013). However, data on the exact zeitgeber used for entrainment in species inhabiting environments where reliable photic cues are absent are limited, as most studies only report the presence or absence of a rhythm (Beale et al., 2016; Abhilash et al. 2017). Notwithstanding, changes in other non-photic cues have been proposed as potential zeitgebers in these stable dark environments, and one of importance to this study is Ta (Abhilash et al., 2017).
To date, only a limited number of studies have experimentally shown entrainment of LA patterns by daily Ta in mammals (Aschoff and Tokura, 1986; Rajaratnam and Redman, 1998; Pálková et al., 1999; Refinetti, 2010; Farsi et al., 2020). This existing body of research suggests that, in comparison with photic cues, Ta exhibits a varied effect on the circadian system depending on the species in question (Rajaratnam and Redman, 1998; Rensing and Ruoff, 2002; Refinetti, 2010). Recently, Farsi et al. (2020) hypothesized that the strength of Ta as a zeitgeber depends on the specific sensitivity of animals to changes in Ta. Indeed, for those species whose thermoregulatory states depend on Ta, such as heterothermic large desert mammals (Allali et al., 2013; Farsi et al., 2020) and ectotherms (Rensing and Ruoff, 2002), their circadian rhythms, like their Tb, are sensitive to changes in Ta and readily entrain to it. In contrast, for endothermic homeotherms, like many rodent species, a weak effect of Ta on the circadian system has been found (Rajaratnam and Redman, 1998; Rensing and Ruoff, 2002; Refinetti, 2010). The weak effect of Ta on the circadian rhythms of endothermic homeotherms has been hypothesized as the direct consequence of their constant Tb irrespective of the daily Ta cycle, resulting in these species being less sensitive to Ta changes (Rajaratnam and Redman, 1998; Rensing and Ruoff, 2002; Refinetti, 2010). However, these studies have been confined to mammals that are readily exposed to photic cues, and as a consequence, the evolutionary pressures behind this form of entrainment and its prevalence among mammals not exposed to photic cues are still unclear.
African mole-rats (Bathyergidae) are a subterranean family of rodents endemic to sub-Saharan Africa who rarely, if ever, venture to the surface (Bennett and Faulkes, 2000). Sociality within this family ranges from solitary to eusocial species (Bennett and Faulkes, 2000; Hart et al., 2021), and their thermoregulatory abilities range from endothermic homeothermy (Šumbera, 2019; Wallace et al., 2021) to heterothermy (Boyles et al., 2012; Oosthuizen et al., 2021) and even poikilothermy (Buffenstein and Yahav, 1991). The subterranean niche provides a relatively thermostable environment (Bennett et al., 1988; Šumbera et al., 2004; Holtze et al., 2018; Šumbera, 2019) as well as a shelter from above-ground predators and temperature extremes (Bennett and Faulkes, 2000; Finn et al., 2020), but the burrow systems have hypoxic and hypercapnic atmospheres with poor ventilation, high humidity, and limited access to light (Kennerly, 1964; Darden, 1972; Roper et al., 2001; Ivy et al., 2020; Logan et al., 2020). Although the burrow systems of African mole-rats are considered to possess a muted Ta range, a minor Ta range does exist (~10 °C) (Roper et al., 2001; Haupt et al., 2017). Furthermore, the LA of mole-rats under natural conditions has been suggested to be constrained by the risk of overheating during bouts of activity (Šklíba et al., 2014; Oosthuizen et al., 2021; Vejmělka et al., 2021), but there is no direct evidence to support this. The combination of subterranean existence, varied thermoregulatory abilities, and apparent sensitivity to overheating during activity make the members of the family Bathyergidae an ideal taxon to test the idea that entrainment to Ta is dependent on the sensitivity to temperature (i.e., thermoregulation strategy).
The LA patterns of African mole-rats have been extensively investigated and shown to entrain to photic stimuli, which suggests the possession of endogenous rhythms and a functional circadian system (Oosthuizen et al., 2003; Vasicek et al., 2005; Schöttner et al., 2006; De Vries et al., 2008; Streicher et al., 2011; Haupt et al., 2017; van Jaarsveld et al., 2019). Nevertheless, the degree of rhythmicity and ability to entrain varies within and between species (reviewed by Bennett (2009)). A recent study on the social endothermic-homeothermic Mahali mole-rat (Cryptomys hottentotus mahali [CHM]) suggested that both LA and Tb may be capable of synchronizing to Ta cycles; however, the experimental setup was unable to definitively prove this as the authors could not differentiate between masking and entrainment (van Jaarsveld et al., 2019).
We attempt to prove that Ta is a strong zeitgeber of circadian rhythms of LA in endothermic-homeothermic African mole-rats, given the lack of a reliable photic cue in the subterranean environment and the suggested sensitivity of LA to Ta, as a consequence of avoiding overheating while being active. To accomplish this, we used two species of African mole-rats, namely, the solitary Cape mole-rat (Georychus capensis [GC]) (Lovegrove and Papenfus, 1995; Oosthuizen et al., 2003) and social Mahali mole-rat (van Jaarsveld et al., 2019; Hart et al., 2020; Fagir et al., 2021). Both genera of Cryptomys (Boyles et al., 2012) and Georychus (Okrouhlík et al., 2021) have not been observed to exhibit heterothermy or poikilothermy, but are considered strict homeothermic endotherms.
Materials and Methods
Study Animals
Twelve CHM (82.6 ± 24.4 g, n = 12; male = 6, female = 6) and 12 GC (161.2 ± 43.7 g, n = 12; male = 5, female = 7) were used for this study. All animals were maintained and acclimated in captivity for longer than 1 year prior to the initiation of the experiments. All mole-rats were captured using modified Hickman traps (Hickman, 1979). GCs were caught during the austral summer in 2018 (January) in the Western Cape, South Africa (33°23′55.2″S, 18°25′06.4″E). CHM individuals were captured from the north-western most region of Gauteng, South Africa (25°39′48.0″S, 28°02′22.5″E), during the austral spring (September) 2018. Captured animals were transported back to the University of Pretoria Small Animal Physiological Research Facility (25°44′49.9″S, 28°15′31.7″E), where they were kept in a temperature constant room (26 °C) on a 12L:12D lighting regime. All animals were housed individually in plastic containers (83 × 44 × 37 cm) and maintained on an ad libitum diet of sweet potatoes, which were replaced on a daily basis to avoid any increases in activity levels associated with foraging (Vasicek et al., 2005). To avoid entrainment to a fixed feeding time, the animals were fed at varying times of the day and night (Golombek and Rosenstein, 2010). All animals were provided with wood shavings and paper towels for nesting material. A study conducted by Bennett and Jarvis (1995) showed that sweet potatoes provide mole-rats with more than 90% of their water and nutritional needs.
Between experimental cycles, plastic containers were cleaned by replacing the wood shavings and the paper toweling. Rooms were fitted with double doors to ensure the integrity of the light and temperature cycles inside the room. A dim red light was used during feeding and cleaning times if this occurred during the dark phase as well as during the constant dark (DD) conditions (light intensity <1 lux).
Experimental Protocol
The experimental protocol was conducted in the climate-controlled rooms of the Small Animal Physiological Research Facility at the University of Pretoria. We are able to regulate the temperature and lighting in the climate rooms in this facility to duplicate sinusoidal patterns in Ta and light cycles. Animals were subjected to a habituation period of 14 days (Cycle 1), followed by three experimental cycles ranging from 21 to 42 days (see Table 1). During Cycle 1, animals were subjected to 10L:14D and constant Ta of 25 °C, and lights went on at 0700 h and off at 1700 h (Figure 1a). To investigate whether the circadian rhythms of LA of CHM and GC are capable of entraining to Ta cycles in the absence of light, we subjected animals to complete darkness (DD) and a simulated Ta cycle, Cycle 2, mimicking the natural range of temperatures experienced by these animals in their burrow systems (18-28 °C) (Roper et al., 2001; Haupt et al., 2017) (Figure 1b). Cycle 2 was followed by constant conditions (DD, Ta = 25 °C), Cycle 3, to differentiate whether any observed rhythms were due to masking or entrainment (Figure 1c). Thereafter, we moved the Ta cycle forward 6 h—Cycle 4—to test whether rhythms would re-entrain to the temperature stimulus (Figure 1d). All procedures involving live animals and sample collection described in this article were conducted in accordance with the appropriate national and provincial guidelines, permits, and regulations. The study was approved by the animal ethics committee of the University of Pretoria, and the ethics certificate approval number is EC038-18.
Number of days spent in each experimental cycle for 12 Mahali mole-rats (Cryptomys hottentotus mahali [CHM]) and 12 Cape mole-rats (Georychus capensis [GC]).

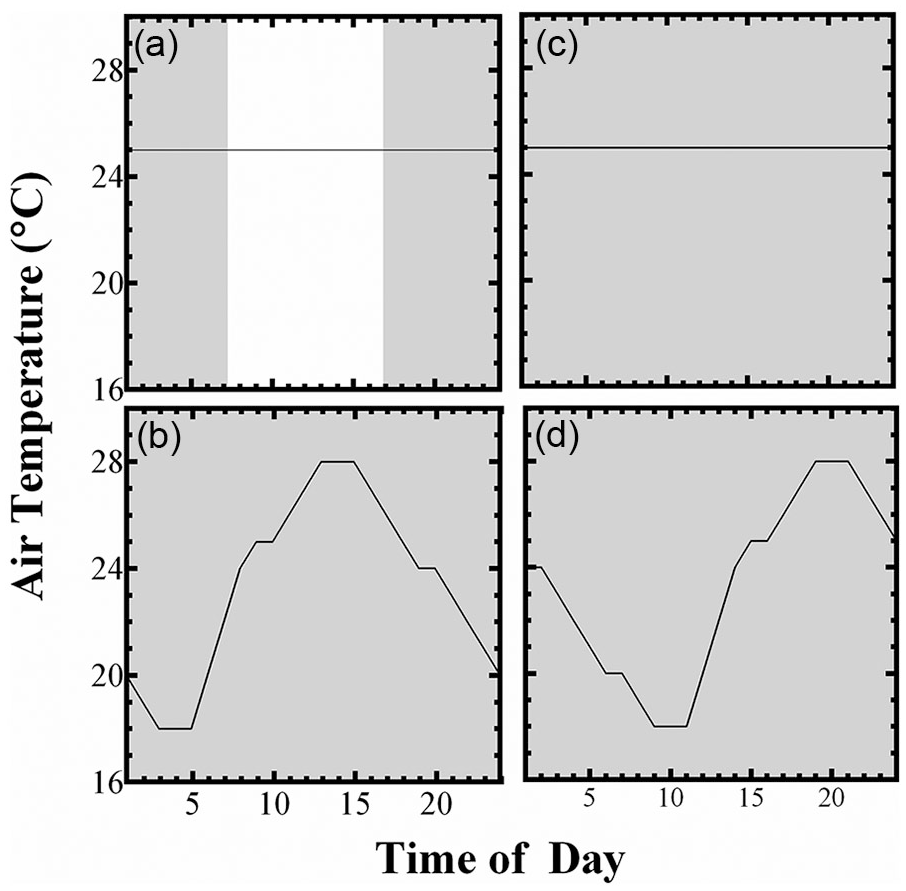
Experimental conditions during the (a) habituation period/Cycle 1, (b) Cycle 2, (c) Cycle 3, and (d) Cycle 4. Solid lines indicate the Ta profile, whereas gray and white bars indicate the absence or presence of light, respectively.
LA Recording
Infrared motion detector (Quest PIR internal passive infrared detector; Elite Security Products [ESP], Electronic Lines, UK) was used to record LA for each mole-rat. The motion detectors were positioned over the containers such that all LA on the floor space of the container was detected. Consequently, the recorded LA readings were summed per minute and relayed to a computer using the program Vitalview (Vital ViewTM, Minimitter Co., Inc., Sunriver, OR, USA; www.minimitter.com).
Statistical Analysis
Actograms and tau (τ) of LA were calculated using RythmicAlly in R (R Development Core Team 2018; Abhilash and Sheeba, 2019). To assess whether body mass and sex influenced proportion, we conducted generalized linear mixed-effects models on hourly proportions data for Cycles 2 and 4, for each species, separately, using the lme4 package (Bates et al., 2015). Each model contained cycle, the body mass and sex of the mole-rat, and called on the binomial distribution (Bates et al., 2015). Each model included individual ID as a random factor to account for pseudoreplication. We refrained from commenting on total counts of activity as biotic and abiotic factors readily influence this. Reported values are presented as the percentage of activity within a cycle for each hour of the day (Equation 1):
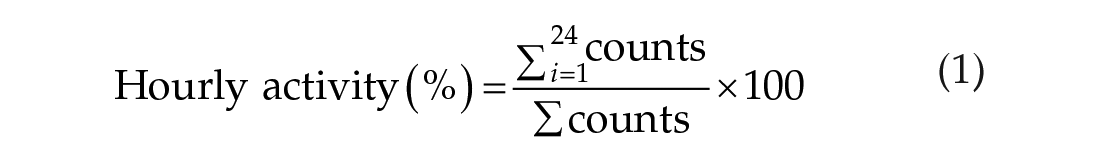
Results
During Cycle 1, both species confined the majority of their activity to the dark phase of the LD cycle (Figure 2). CHM and GC displayed 86% and 79% of activity during the dark phase (1700 h to 0700 h).
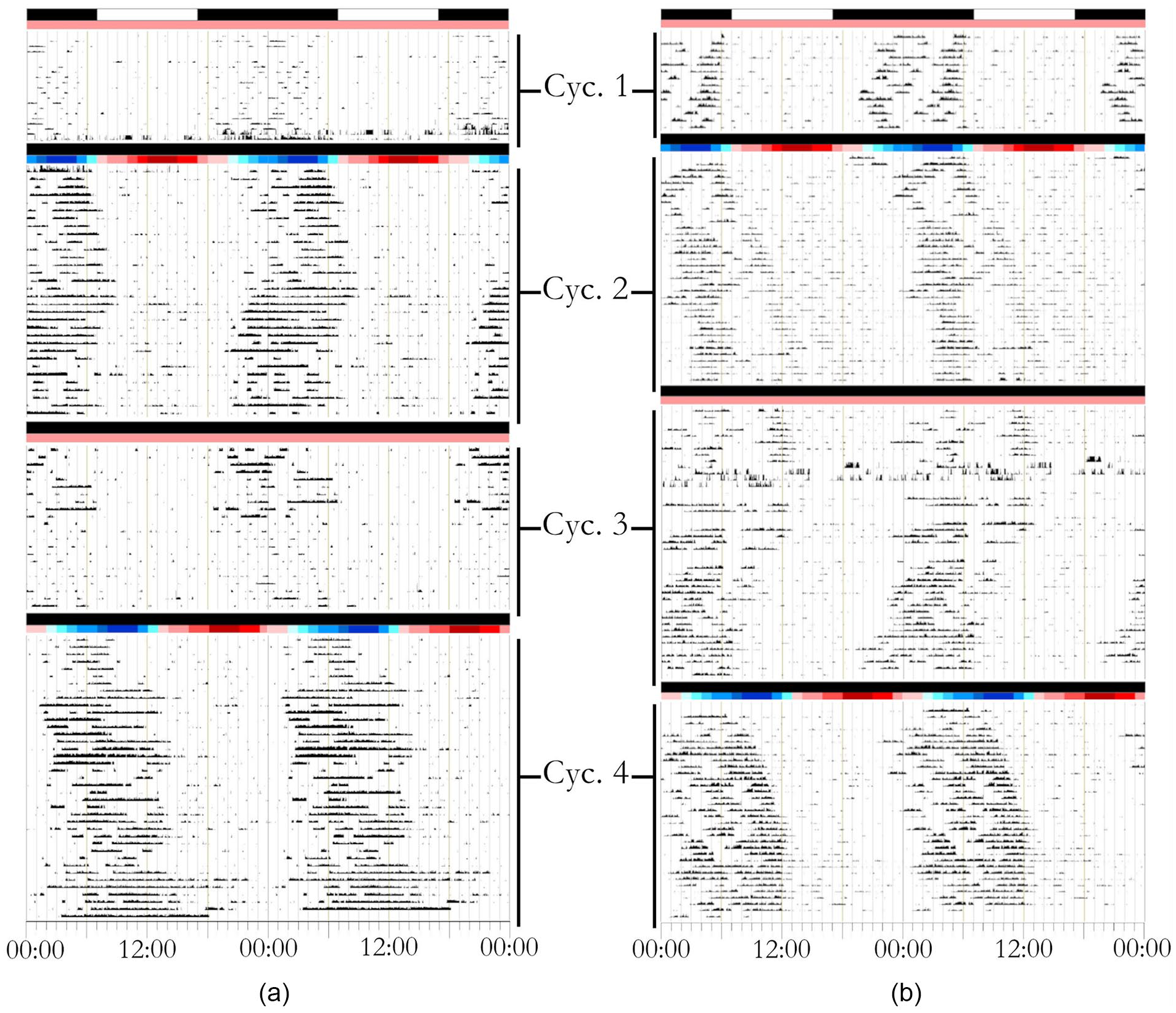
Representative actograms of locomotor activity for (a) Mahali mole-rat (Cryptomys hottentotus mahali) and (b) Cape mole-rat (Georychus capensis) individuals. Actograms are double-plotted with time on the x-axis. During the habituation period/Cycle 1, air temperature (Ta) was 25 °C, and the lights went on at 0700 h and off at 1700 h. Cycle 2 consisted of constant darkness (DD) and a natural temperature ramp (18-28 °C), with a nadir at 0300 h. During Cycle 3, animals were subject to DD and 25 °C. Cycle 4 consisted of DD and a 6-h shift from Cycle 2 in the natural temperature ramp, with a nadir at 0900 h. Color bar indicates temperature gradient, with dark red representing the hottest period (28 °C) and dark blue representing the coolest period (18 °C).
When subjected to DD conditions in the presence of a natural Ta ramp (Cycle 2), LA became confined to the coolest hours of the cycle (18-23 °C) in both species (Figures 2 and 3). CHM displayed 80% of activity in the coolest 12 h of the cycle, and GC displayed 68% of their activity during the same period (Figure 3).
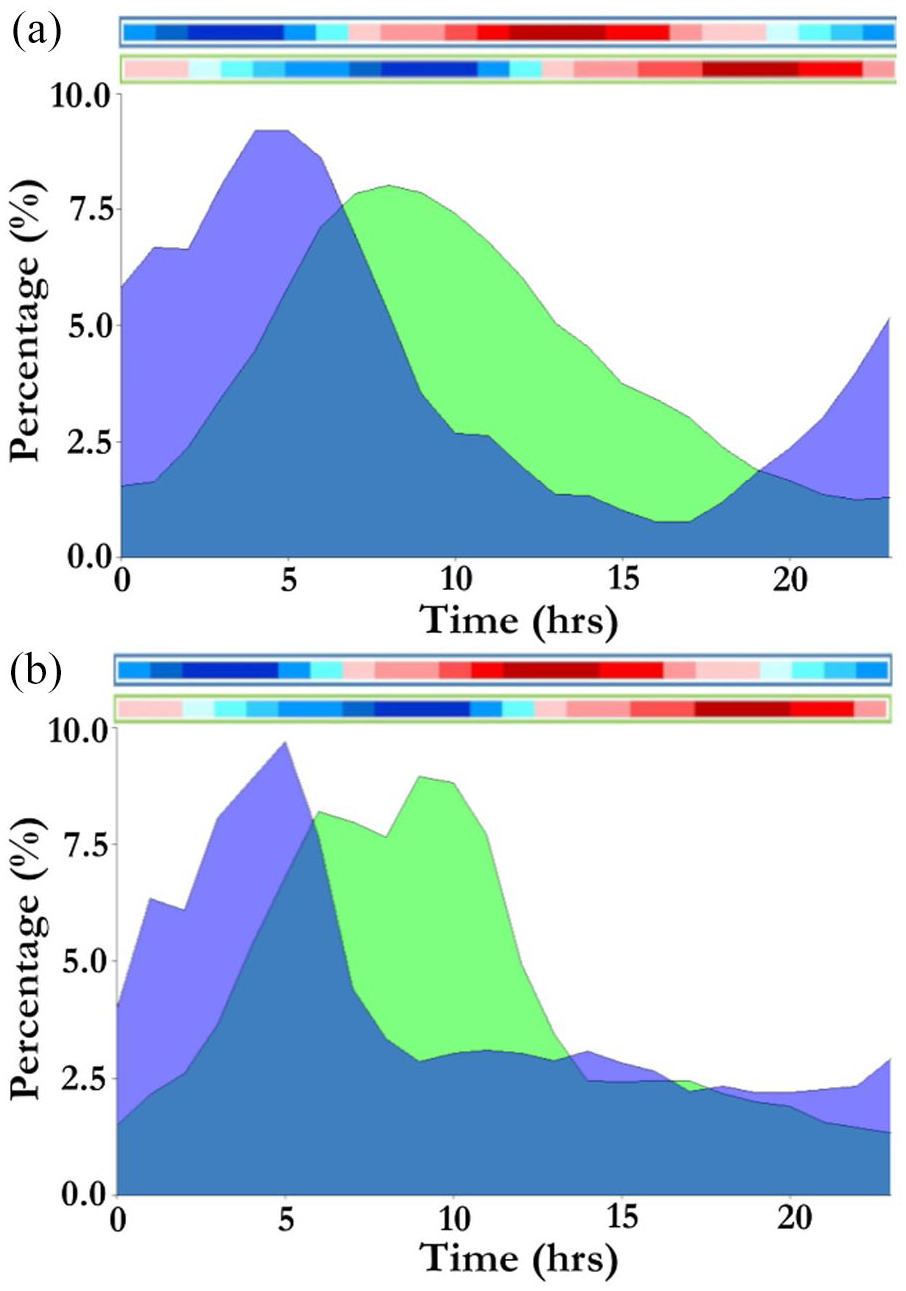
Percentage (%) of daily locomotor activity expressed per hour for (a) 12 Mahali mole-rats (Cryptomys hottentotus mahali) and (b) 12 Cape mole-rats (Georychus capensis). Cycle 2 (purple) consisted of constant darkness (DD) and a natural temperature ramp, with a nadir at 0300 h. Cycle 4 (green) consisted of DD and a 6-h shift in the natural temperature ramp, with a nadir at 0900 h. Color bar indicates temperature gradient, with dark red representing the hottest period (28 °C) and dark blue representing the coolest period (18 °C).
Eight of the 12 CHM displayed a free-running period under constant DD conditions, with an average τ of 23.9 ± 0.37 h (Cycle 3; Figure 2). Ten of the 12 GC individuals displayed a free-running period under constant conditions, with an average τ of 24.12 ± 0.39 h (Cycle 3; Figure 2).
When released from constant conditions and subjected to a Ta cycle advanced by 6 h, both CHM and GC displayed a phase advance and re-entrainment of LA to the coolest hours of the cycle (Cycle 4; Figures 2 and 3). The proportion of activity expressed during the 12 coolest hours (18-23 °C) was 75% in CHM, whereas GC displayed 75% of their activity in the same period (Figure 3).
Experimental cycle had a significant effect on hourly proportions of LA for both species (CHM: z = 4.231, p < .001; GC: z = −6.889, p < .001). For CHM, hourly proportions were higher in Cycle 4 compared to Cycle 2, while the reverse was observed for GC. No effect of sex (CHM: z = 0.614, p = .539; GC: z = 1.795, P = .073) or body mass (CHM: z = −0.713, p = .476; GC: z = −0.004, p = .997) was reported for either species.
Discussion
In this study, under DD conditions in the presence of a 24-h Ta cycle, both species displayed a preference for increased LA during the cooler period (18-23 °C) of the 24 h cycle—rather than the warmer period (23-28 °C). Therefore, it is evident that LA is able to synchronize, through entrainment, to Ta cycles, a result that is in agreement with previous studies on mammals (Rajaratnam and Redman, 1998; Rensing and Ruoff, 2002; Refinetti, 2010, van Jaarsveld et al., 2019). However, it is essential to distinguish between entrainment and masking effects of Ta on LA (Golombek and Rosenstein, 2010; Bittman, 2021). The data presented here present two lines of evidence that oppose the notion that LA rhythm is synchronized to Ta through masking. First, when animals were released into constant conditions (i.e. Cycle 3), the free-running rhythms begin from the same place as the prior entrained rhythm (i.e. Cycle 2) and display drift (Daan and Aschoff, 2001; Refinetti, 2015). In addition, a 6-h phase advance in the daily Ta cycle, from free-running conditions, resulted in a progressive shift in LA patterns (~6 h), with peak activity again present in the cooler periods of the Ta cycle (Daan and Aschoff, 2001; Refinetti, 2015). Together, LA rhythm persistence under free-running conditions and progressive phase-matching of free-running rhythms to 6-h advance in Ta cycle favor the notion of entrainment of LA by Ta in the two species of African mole-rat. To the best of our knowledge, this is the first evidence of temperature entrainment in subterranean rodents.
Both species were most active at Ta well below their thermoneutral zones, subsequently increasing the energetic costs of locomotion. The coolest 12 h of the Ta cycles were between 18 and 23 °C, while the thermoneutral zone for CHM and GC lies between 27 and 32 °C (Wallace et al., 2021) and 26 and 34 °C (Lovegrove, 1987), respectively. CHM and GC may tolerate the energetic demands of increased energetics under their respective thermoneutral zone for two possible reasons. First, by using Ta as a zeitgeber and confining LA to the cooler periods, GC and CHM may be avoiding exercise-induced hyperthermia associated with the metabolic heat gains from burrowing (McNab, 1974; Speakman and Król, 2010). This condition may be further exacerbated by the poor evaporative cooling capabilities present in these two species of burrowing mammals (Luna et al., 2020; Wallace et al., 2021). Consequently, these mole-rats are unable to physiologically dissipate heat efficiently, further increasing the risk of exercise-induced hyperthermia. This is congruent with field studies that found mole-rats confine activity to the cooler periods (Streicher et al., 2011; Šklíba et al., 2014; Vejmělka et al., 2021). Second, foraging underground is energetically more expensive than above-ground living (Vleck, 1979). Geophytes, which provide the only source of food and water, are unevenly dispersed in their habitat, resulting in even more significant energetic expenditure (digging) to locate their food sources (Bennett and Faulkes, 2000). Traditional resource-based ecological and evolutionary theories assume that energetic constraints are directly affected as a consequence of resource availability (Luna et al., 2020). However, according to the Heat Dissipation Limit theory, the energetics of an endotherm could possibly increase through enhanced heat dissipation capacity (Speakman and Król, 2010). Thus, increased activity during the cooler periods may potentially allow for increased energetic output during LA.
In the past, homeothermic mammals have been recorded to show weak entrainment to Ta (Rajaratnam and Redman, 1998; Rensing and Ruoff, 2002; Refinetti, 2010); it is permissible to predict that GC and CHM would also show weak responses to Ta. However, when considering the limited heat dissipation ability brought about by physiological and ecological constraints, it is clear that these species are indeed sensitive to Ta changes, resulting in their strong LA entrainment by Ta. Moreover, the lack of a reliable photic cue makes Ta a more reliable “time-keeping” variable in the subterranean environment to which GC and CHM entrain.
In conclusion, these results favor the hypothesis put forward by Farsi et al. (2020), namely that the strength of Ta as a zeitgeber depends on the sensitivity (often linked to the thermoregulatory abilities of the species) of a species to daily Ta changes. In addition, we show that Ta is a reliable cue that social and solitary African mole-rats are capable of perceiving and subsequently use to entrain LA rhythms. Finally, the nocturnal LA patterns in many African mole-rat species could possibly be a consequence of the requirement to be active during the cooler periods of the day. The African mole-rats may be the ideal model family, with a diverse range of thermoregulatory abilities, to unravel the relationship between thermoregulation, circadian rhythmicity, and the subsequent molecular machinery behind this relationship, which are currently unknown.
Footnotes
Acknowledgements
We are grateful to the Gauteng Department of Nature Conservation and the Western Cape Department of Nature Conservation for permits to collect CHMs (CPF6-0127) and GCs (AAA041-00169-0056), respectively. We thank the respective landowners Mr. John Duckett (Western Cape) and the Kemp family (Gauteng) for allowing us to trap mole-rats on their farms. We thank the two anonymous reviewers for their insightful comments that improved the quality of this manuscript. This study was funded by SARCHI Chair of Mammal Behavioural Ecology and Physiology to N.C.B. (grant no. 64756).
Author Contributions
D.W.H., B.v.J., M.K.O., and N.C.B. conceptualized the project. D.W.H. and K.L.G. collected data and performed animal maintenance. B.v.J. and K.G.L. analyzed the data. All authors contributed toward the writing of the manuscript and agreed to be held accountable for the content therein.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
