Abstract
Heavy water lengthens the periods of circadian rhythms in various plant and animal species. Many studies have reported that drinking heavy water lengthens the periods of circadian activity rhythms of rodents by slowing the clock mechanism in the suprachiasmatic nucleus (SCN), the mammalian circadian center. The SCN clock is stable and robust against disturbance, due to its intercellular network. It is unclear whether this robustness provides resistance to the effects of heavy water. Here, we report that heavy water lengthened the rhythm period of clock gene expression of the SCN and peripheral tissues in vitro using a PERIOD2::LUCIFERASE bioluminescence reporter. Our results show that the period-elongation rate of the SCN is similar to those of other tissues. Therefore, the intercellular network of the SCN is not resistant to the period-elongation effect of heavy water.
Keywords
Heavy water, also known as deuterium oxide (D2O), is water in which all the hydrogen atoms are replaced by its isotope, deuterium (Urey et al., 1932). Deuterium differs from hydrogen in that each atom of deuterium contains a proton and a neutron. The additional neutron slightly increases the molecular mass and confers different physical and chemical properties compared with normal water.
Replacement of the hydrogen atoms with deuterium primarily leads to slower rates of chemical reactions (Katz and Crespi, 1970). In organisms, heavy water affects many metabolic reactions. It is known that heavy water slows various biological timings with a wide range of periods, from milliseconds to daily alterations (Enright, 1971; Richter, 1977; Yamazaki et al., 1995).
Most organisms have a daily rhythm in behavior and physiology governed by an internal biological clock named the “circadian clock” that sustains an approximately 24-h period (Harmer et al., 2001). Heavy water slows down the circadian rhythms of a wide range of species, including unicellular organisms, plants, invertebrates, and vertebrates (Bruce and Pittendrigh, 1960; Caldarola and Pittendrigh, 1974; Suter and Rawson, 1968; Thomson, 1963). Among mammals, drinking heavy water lengthens the periods of activity rhythms of mice, rats, hamsters, and deer mice (Daan and Pittendrigh, 1976; Lesauter and Silver, 1993; Richter, 1970; Suter and Rawson, 1968). The period of circadian activity increases linearly with the concentration of heavy water, and this increase of period is reversible (Daan and Pittendrigh, 1976; Dowse and Palmer, 1972). The center of the mammalian circadian clock resides in the suprachiasmatic nucleus (SCN) in the hypothalamus (Hastings et al., 2018; Moore and Eichler, 1972; Stephan and Zucker, 1972). SCN lesion and transplant experiments indicated that the extension of the circadian period by drinking heavy water was due to a slowdown of the biological clock in the SCN (Lesauter and Silver, 1993; Pickard and Zucker, 1986).
Recent studies have revealed a molecular mechanism of the circadian clock. In mammals, most cells have an intracellular molecular circadian oscillator composed of interacting positive and negative transcription-translation feedback loops (TTFLs) of the clock gene (Dunlap, 1999; Reppert and Weaver, 2002; Zhang and Kay, 2010). The circadian clock of the SCN is also driven by the molecular circadian oscillator but has different characteristics to other tissues and cells. The SCN clock exhibits high stability and robustness to disturbances by reciprocal synchronization with other oscillators through intercellular networks (Buhr et al., 2010; Welsh et al., 2010). This robustness may offer some resistance to the effects of heavy water. Pickard and Zucker (1986) indicated that the response to heavy water was more variable in rodents having SCN lesions than in intact counterparts. As another example, drinking heavy water slows the circadian clock, but takes several days to reach maximum effect (Daan and Pittendrigh, 1976; Lesauter and Silver, 1993). It was thought that this delay is due to the increase in heavy water concentration in body fluids over several days. However, the robustness of the SCN clock may have provided resistance to the effects of heavy water.
To test this hypothesis, it is necessary to strictly control the concentration of heavy water in the fluid surrounding the SCN and to compare the period-elongation rate with those of other tissues. In the present study, we investigated the effects of heavy water on the circadian oscillator of the SCN, peripheral tissues, and cell lines using real-time bioluminescence recordings of clock gene expression in vitro.
Materials and Methods
Animals
We used PERIOD2::LUCIFERASE (PER2::LUC) knock-in homozygous mice (Yoo et al., 2004). PER2::LUC knock-in mice (C57BL/6J) obtained from RIKEN (Wako, Osaka, Japan) were backcrossed to Jcl:ICR (Crea, Tokyo, Japan) for 10 generations. N10 generation mice were then intercrossed, and their offspring were used. All mice were housed in plastic cages (200 mm × 200 mm × 200 mm; Crea) under a 12 h light/12 h dark cycle (LD) at a constant temperature (23 °C). Each cage was equipped with a water bottle and rodent laboratory chow pellets (CE-2, Crea). All experiments were approved by the Committee of Animal Care and Use of Kindai University School of Medicine, and all experimental procedures were conducted in accordance with the Kindai University School of Medicine guidelines for use of experimental animals.
Tissue and Cell Preparation
Pups (6-8 days old) were deeply anesthetized by cooling with crushed ice and euthanized by decapitation. Their brains and organs were quickly removed and immersed in ice-cold Hank’s Buffered Saline (HBS). The brain was frontal sectioned at 300 µm using a micro slicer (DSK-1000, DOSAKA EM, Kyoto, Japan), and the hypothalamic slice containing the central part of the SCN was obtained. The bilateral SCN was dissected from this slice under a stereomicroscope. The kidney and lung were isolated as square tissues about 1 mm across with a scalpel. The SCN or organ tissue was placed on a culture membrane (Millicell-CM PICM0RG50; Merck Millipore, Darmstadt, Germany) in a 35-mm petri dish. The preparation up to this step was performed in ice-cold HBS to prevent tissue damage. The explants were cultured in air at 37 °C with 1.2 mL of medium throughout this study. The air culture medium consisted of 13.4 g/L DMEM (12100-046, Gibco, Carlsbad, CA, USA), 2.383 g/L HEPES (Dojindo, Kumamoto, Japan), 0.35 g/L NaHCO3 (Nacalai Tesque, Kyoto, Japan), 20 µL/mL B-27 supplement (Gibco), 100 U/mL penicillin, and 100 µg/mL streptomycin (Nacalai Tesque).
We used 2 cell lines: mouse embryonic fibroblasts (MEFs) taken from PER2::LUC knock-in mice, and rat C6 glioma cells that carry luciferase gene driven by the mouse Per2 promotor (Kurosawa et al., 2017). The cells were cultured in 35 mm petri dishes with 1.2 mL of medium at 37 °C in a 5% CO2 atmosphere throughout this study. The medium for maintenance consisted of DMEM (D6429-500ML; Merck, Darmstadt, Germany) supplemented with 10% fetal bovine serum (FBS; ICN, Cleveland, OH, USA).
Preparation of Deuterium-containing Medium
To examine the effects of heavy water, we made a bioluminescence monitoring medium containing deuterium. To prepare the concentration series of deuterium-containing media, we made normal medium and deuterium medium using normal water and heavy water, respectively (D2O; Tokyo Chemical Industry, Tokyo, Japan). The air culture medium consisted of 13.4 g/L DMEM (12100-046; Gibco), 2.383 g/L HEPES, 0.35 g/L NaHCO3, and 20 µL/mL B-27 supplement. The medium for 5% CO2 atmosphere culture consisted of 13.4 g/L DMEM (12100-046; Gibco) and 3.7 g/L NaHCO3. These media were mixed at a ratio adjusted to the target concentration (0%, 30%, 50%, 70%, 90%, and 100% deuterium medium), and D-luciferin potassium salt (200 µM; Wako), 100 U/mL penicillin, and 100 µg/mL streptomycin were added. The supplement and antibiotics did not contain deuterium. Therefore, the final concentrations of deuterium in the media were 0%, 29.1%, 48.5%, 67.9%, 87.3%, and 97% (v/v), respectively. For 5% CO2 atmosphere, medium containing no B-27 supplement was used, but the heavy water concentration was adjusted to be the same as that for the air condition. The concentration values described in this article were rounded to the nearest whole number.
Bioluminescence Monitoring for Period Analysis
The detailed methods for real-time measurement of bioluminescence from tissues or cells in vitro have been described in our previous reports (Kurosawa et al., 2017; Sujino et al., 2018). The tissue explants were transferred to the bioluminescence monitoring medium for the air condition. For the cell culture, once C6 cells reached confluence, cells were treated with dexamethasone (DEX, 100 nM; Wako) for 1 h to synchronize the oscillation. After washing out DEX, the medium was replaced by bioluminescence monitoring medium for 5% CO2 atmosphere without serum. The amounts of luminescence emissions during 1 min were recorded every 10 min using a photomultiplier tube (PMT) (Kronos; ATTO, Tokyo, Japan). Bioluminescence recordings were processed to suppress noise using the Kronos Data Analysis software and to remove long-term trends using Microsoft Excel (Abe et al., 2002). Period lengths were estimated by the cosine or sine curve fitting method in software (Microsoft Excel or Origin8.1J; OriginLab, Northampton, MA, USA; Halberg et al., 1972). The double-plotted histogram shown in Figure 1d was drawn by ClockLab (Actimetrics, Wilmette, IL, USA).
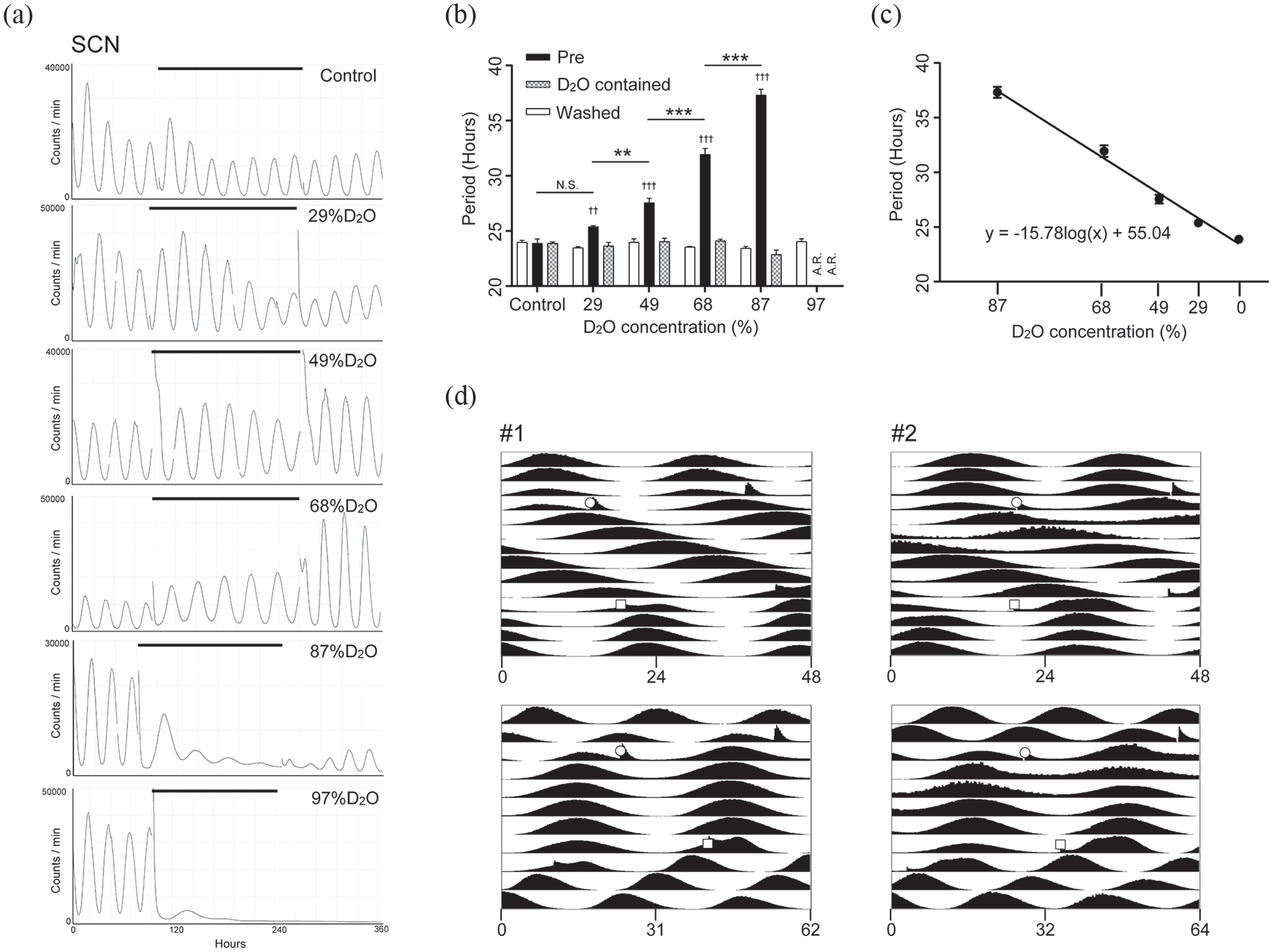
Heavy water extended the period of PER2::LUC bioluminescence rhythm in SCN explants. (a) Representative traces of PMT bioluminescence recordings of the SCN explants cultured in medium containing heavy water. The vertical axis shows the bioluminescence intensity and the horizontal axis shows the number of hours of PMT recording. The horizontal line indicates the period of heavy water treatment. (b) Period lengths of the bioluminescence rhythms, which are defined as the fitted cosine curves. Fitted periods were 2 cycles immediately before transferring to heavy water medium, 2nd to 3rd cycles during heavy water treatment, and 2nd to 3rd cycles after washout. Mean ± SEM, Dunnett (vs. pre) and Tukey (concentration) test. A.R. Arrhythmic. (c) Heavy water concentration-dependent period extension represented by semi-logarithmic graph. The vertical axis shows the period of PER2::LUC rhythms. The horizontal line (log2 scale) indicates the concentration of heavy water in the medium. A regression line is shown. (d) Double-plotted histograms representing PMT bioluminescence recordings of the SCN explants cultured in 68% heavy water medium. #1 uses the same data as (a). The horizontal axis of the upper graph is for 24 h and that of the lower graph is for the extended period. Open circles indicate the time of transferring to the heavy water medium, and open squares indicate the time of washout. Abbreviations: SCN = suprachiasmatic nucleus; PMT = photomultiplier tube; SEM = standard error of the mean; D2O = deuterium oxide.
Degradation Assay of PER2 Protein
For real-time monitoring of PER2 protein levels, we used MEFs taken from PER2::LUC mice. Once MEFs reached confluence in a petri dish, the medium was replaced by bioluminescence monitoring medium containing 68% or 0% (control) heavy water. The amounts of bioluminescence during 1 min produced by PER2::LUC protein were recorded every 10 min using PMT. Before performing the degradation assay, we measured the periods of circadian luminescence rhythm of MEFs under 68% or 0% heavy water. The periods were estimated from the luminescence traces for 60 h from the 24th hour after stimulation of 100 nM DEX for 60 min. In the degradation assay, no initial entraining stimulus was applied to average the effects of phase differences. Because the change of medium evoked a weak rhythm in heavy water group, cycloheximide (CHX, a protein synthesis inhibitor, 20 µg/mL; Wako) was added after the amplitude of bioluminescence rhythms had become highly attenuated. Petri dishes of the heavy water group and the control group were examined alternately to avoid any influence of the time difference. After measuring protein degradation over 12 h, the medium was replaced by new medium containing no heavy water with 100 nM DEX to confirm recovery of the bioluminescence rhythm. On the vertical axis of the degradation curve, 100% and 0% were defined as the luminescence intensity at time 0 after administration of CHX and the average luminescence intensity between the 780th and 900th minutes, respectively.
Statistical Analysis
Data were analyzed by using Prism 7 (GraphPad, San Diego, CA, USA). Results are expressed as mean ± standard error of the mean (SEM). In two-variable experiments, two-way repeated-measures analysis of variance (ANOVA) was used to evaluate the significance of differences between group means. If an interaction effect was detected, post hoc analysis was performed using the Dunnett, Bonferroni, or Tukey multiple comparisons test. p < 0.05 was considered significant.
Results
Heavy Water Lengthened the Circadian Oscillator Period in Cultured Mouse SCN
To investigate the effect of heavy water on the molecular circadian oscillator of the SCN, we measured the bioluminescence of SCN explants from newborn PER2::LUC mice with heavy-water-containing medium. In normal medium, the SCN explants showed robust and stable circadian bioluminescence rhythms (Figure 1a). A medium change did not affect the circadian period of these explants (control in Figure 1b). We found that the circadian period of cultured SCNs drastically lengthened in the heavy water medium (Figure 1a and 1b), and this period elongation was dose-dependent (Figure 1c). The circadian periods of each condition are shown in Table 1. The circadian period at a heavy water concentration of 87% reached 37.33 ± 0.51 h. However, a large decrease in amplitude was observed. In the case of 97% heavy water, the bioluminescence decreased and remained minimal; it did not recover even after the medium was replaced with 0% heavy water medium. These results suggest that high concentrations of heavy water (87% or greater) caused irreversible damage to the SCN explants. For heavy water conditions other than 97%, the period returned to its original value after washing out the medium (Figure 1b).
Effect of heavy water on the period of PER2::LUC rhythms in SCN explants.
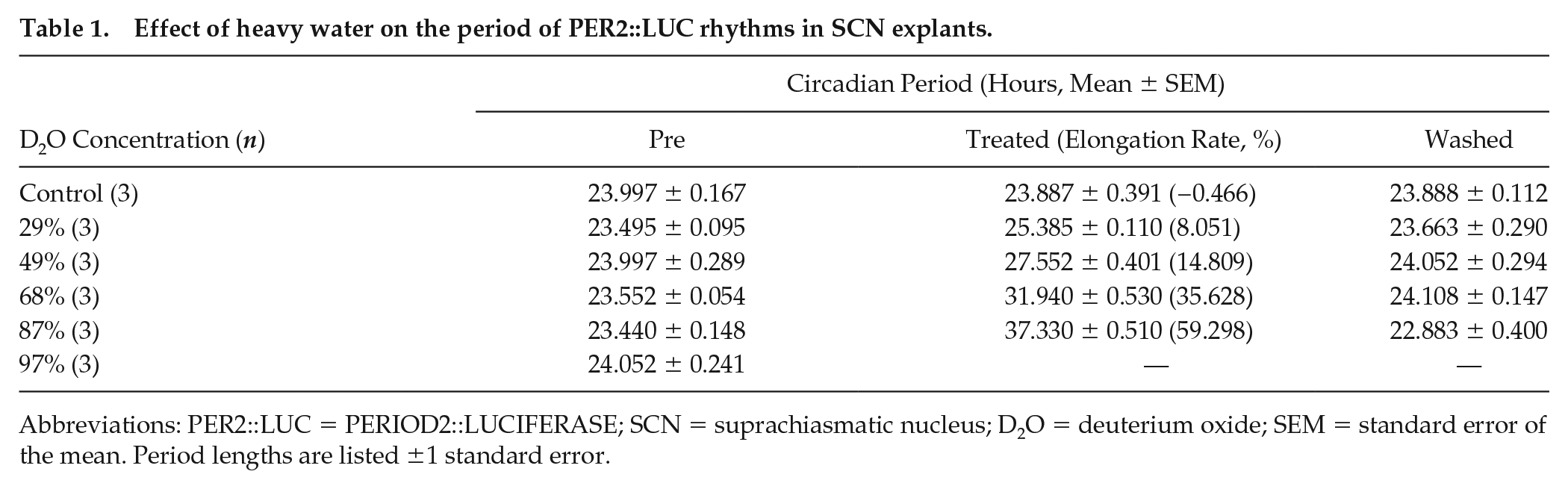
Abbreviations: PER2::LUC = PERIOD2::LUCIFERASE; SCN = suprachiasmatic nucleus; D2O = deuterium oxide; SEM = standard error of the mean. Period lengths are listed ±1 standard error.
To better understand the characteristics of the effect of heavy water, 2 representative bioluminescence rhythms under 68% heavy water concentration were combined as a double-plotted histogram (Figure 1d). Before heavy water addition, the periods of bioluminescence rhythms were stable at almost 24 h. When the explants were transferred to heavy water medium, the first acrophase appeared more than 24 h after the previous acrophase. Thereafter, the period of bioluminescence rhythms was stable in the heavy water condition. After replacing with normal medium, the extended period immediately returned to the original 24 h.
Elongation Rates of Period Length Were Similar Among the SCN, Peripheral Tissues, and Cell Lines
We observed the effect of heavy water of the circadian clock in the peripheral tissues and cultured cells. Figure 2a and 2b shows the circadian bioluminescence rhythms of the kidney and lung tissue explants taken from newborn PER2::LUC mice. All measured tissues exhibited circadian rhythm period elongation in a heavy water concentration-dependent manner (Figure 2d and 2e). The circadian period of cultured C6 glioma cells was also lengthened by the heavy water (Figure 2c and 2f). The periods for each condition are shown in Table 2. Figure 2g-2i indicates the heavy water concentration dependence of the period length.
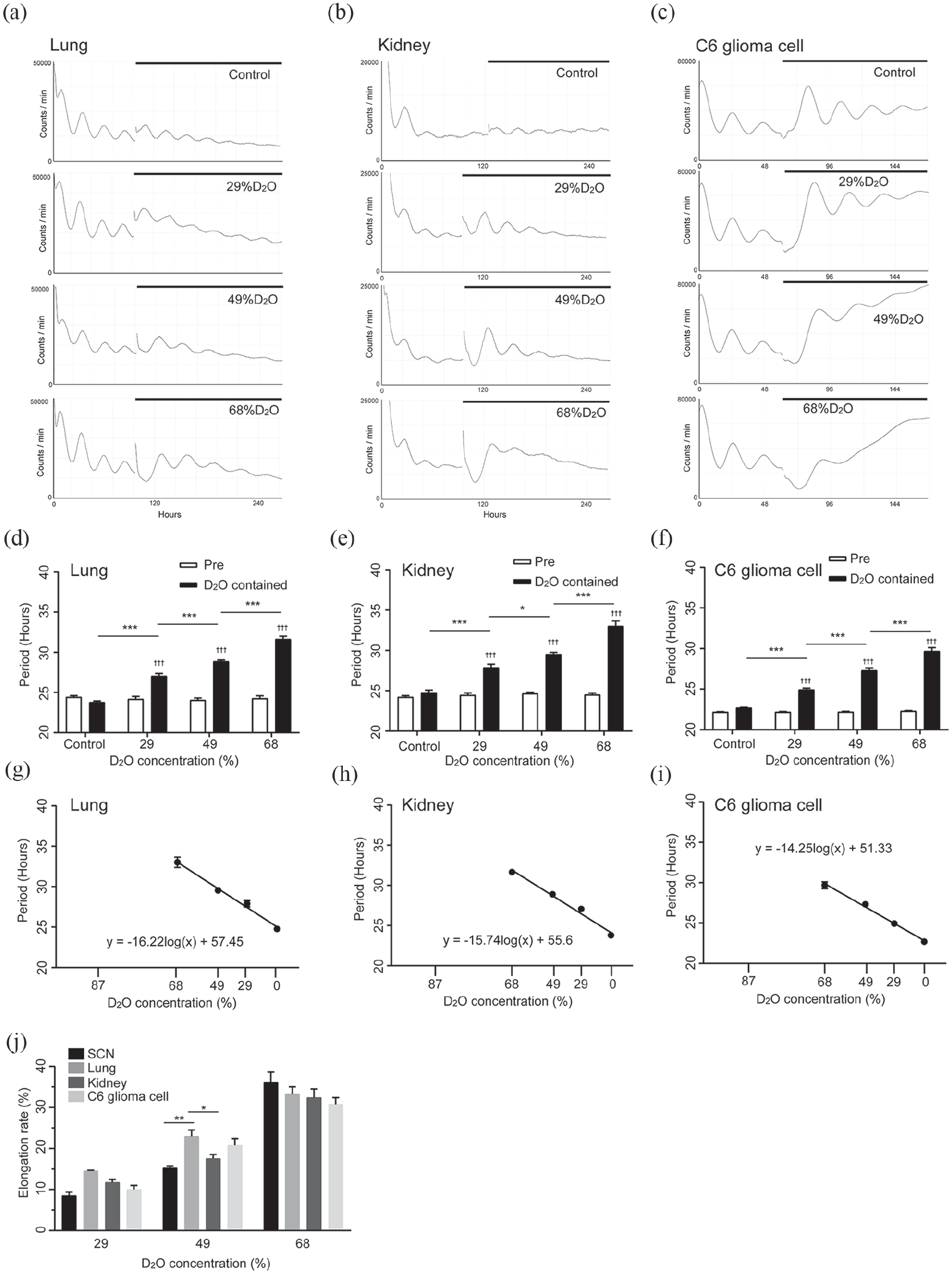
Heavy water extended the period of the bioluminescence rhythm driven by Per2 promotor in peripheral tissues and cells. (a-c) Representative traces of PMT bioluminescence recordings of the lung, kidney, and C6 glioma cells, cultured in heavy water medium. The vertical axis shows the bioluminescence intensity, and the horizontal axis shows the number of hours of PMT recording. The horizontal line indicates the period of heavy water treatment. (d-f) The period lengths of the bioluminescence rhythms, which are defined as the fitted cosine or sine curves. Fitted periods were 2 cycles immediately before transferring to heavy water medium and the 2nd to 3rd cycles during heavy water treatment. Mean ± SEM, Bonferroni test. (g-i) Heavy water concentration-dependent period extension is represented as a semi-logarithmic graph. The vertical axis shows the period of bioluminescence rhythms. The horizontal line (log2 scale) indicates the concentration of heavy water in the medium. A regression line is shown. (j) Difference in the period-elongation rate among the SCN, lung, kidney, and C6 glioma cells. The elongation rate was calculated by subtracting the amount of change in the control group. Mean ± SEM, Tukey test. Abbreviations: PMT = photomultiplier tube; SEM = standard error of the mean; SCN = suprachiasmatic nucleus; D2O = deuterium oxide.
Effect of heavy water on the period of bioluminescence rhythms driven by Per2 promotor in cultured tissues and cells.
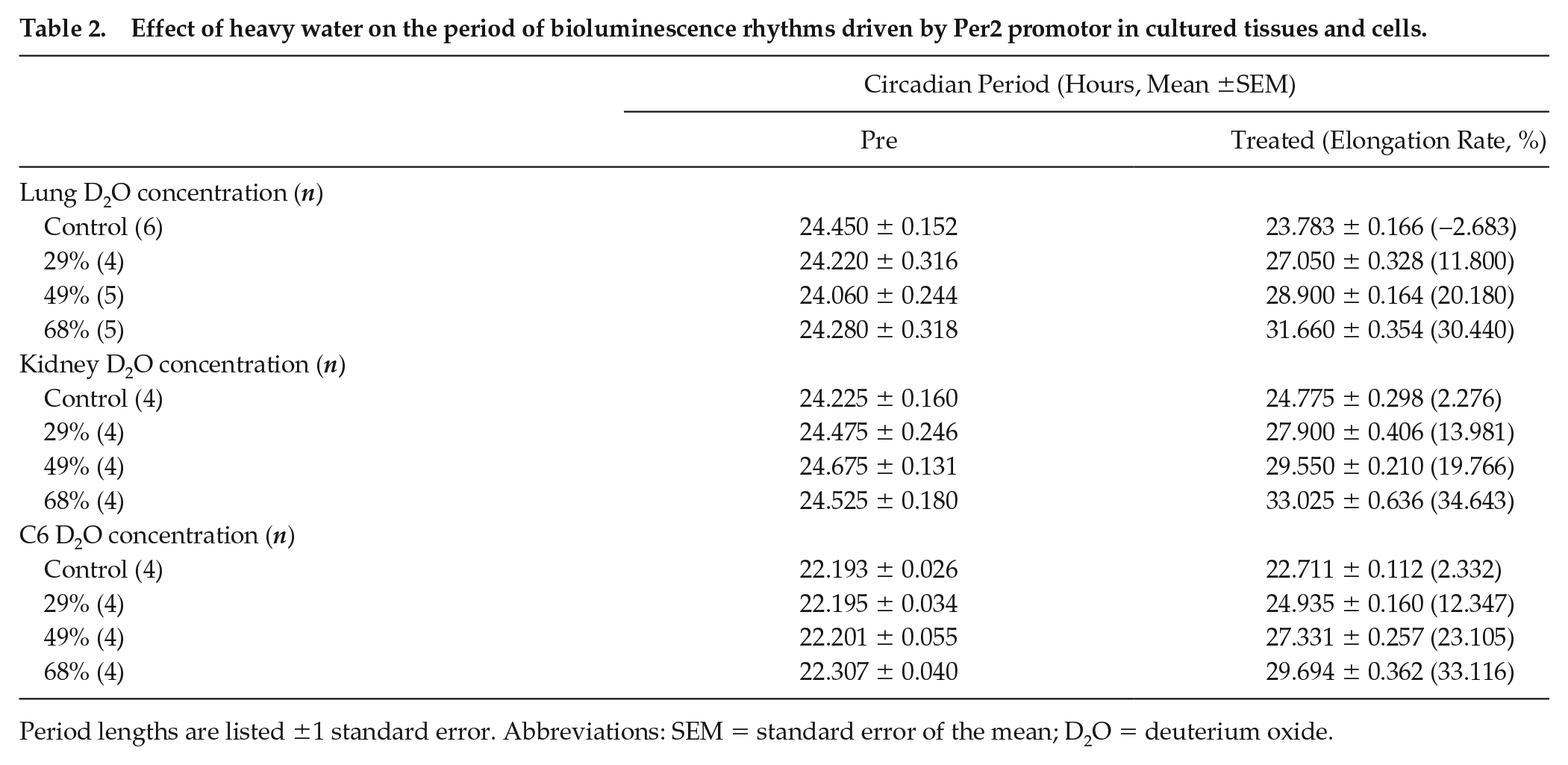
Period lengths are listed ±1 standard error. Abbreviations: SEM = standard error of the mean; D2O = deuterium oxide.
Under high heavy water concentration conditions (87% or more), the cultured C6 cells did not show significant circadian bioluminescence rhythms (R2 < 0, coefficient of determination) (Suppl. Fig. S1). Except for the 97% heavy water condition, the circadian bioluminescence rhythm recovered its original period after washing out the medium. In the case of 97% heavy water, no spontaneous bioluminescence rhythm appeared even after washing out. However, the luminescence rhythm with the original period appeared following stimulation with DEX (Suppl. Table S1).
Next, the elongation rate in the SCN explants was compared with those in peripheral tissues and cell lines. The elongation rate in the lung explants in 49% heavy water medium was significantly higher than those in the SCN and kidney (p = 0.0056 and p = 0.0456, respectively; Figure 2j). However, there were no significant differences among the groups under 29% and 68% heavy water conditions. Therefore, no evidence was found that the elongation rate in the SCN was smaller than those in peripheral tissues and cells.
Heavy Water Altered the Shape of the PER2::LUC Degradation Curve
The overall picture of the molecular mechanisms that determine the periods of circadian rhythms is not yet clear, but one important factor is the degradation rate of clock gene products (Brenna and Albrecht, 2020; Mehra et al., 2009). The rate of PER2 degradation also greatly affects the periods of circadian rhythms (Eide et al., 2005; Vanselow et al., 2006). It has been reported that delaying PER2 degradation results in longer periods of circadian rhythms (Masuda et al., 2020; Zhou et al., 2015). If PER2 degradation is delayed by heavy water, the period would be extended. To test this possibility, we used PER2::LUC MEFs to measure the rate of PER2 degradation.
PER2::LUC MEFs exhibited elongated circadian periods in 68% heavy water (28.54 ± 0.12 h in 68% heavy water, 25.11 ± 0.06 h in control, n = 4). Supplemental Figure S2A shows representative traces of bioluminescence in 68% heavy water and control medium. Inhibition of protein synthesis caused a rapid decrease in PER2::LUC under both conditions, but the decay curves were significantly different (p < 0.001, two-way ANOVA with interaction, Suppl. Fig. S2B). In the heavy water group, a slight increase in luminescence intensity was observed after the administration of CHX (arrow in Suppl. Fig. S2A). The time to reach 75% of the initial value was significantly slower in the heavy water group than in the control group (p = 0.021, 40 ± 0 min, 25 ± 2.89 min, respectively; Suppl. Fig. S2C). However, there was no difference in the time taken for the luminescence intensity to halve between the heavy water group and the control group (p = 0.0622, 85 ± 2.89 min, 72.5 ± 2.5 min, respectively). The time to reach 25% of the initial value was significantly shorter in the heavy water group than in the control group (p < 0.001, 170 ± 5.787 min, 200 ± 4.08 min, respectively). The rhythm was restored by changing the medium under both conditions. However, the bioluminescence intensity in the heavy water group was lower than that in the control group. After the experimental period, floating cells were not observed.
Discussion
Heavy water showed a period-elongation effect in all of the SCN, kidney, lung, and cultured cells examined. The elongation rate depended on the concentration of heavy water and was almost the same for all tissue types. The elongation rate in the lung at 49% heavy water concentration was higher than those in the SCN and kidney, but the cause of this discrepancy was unknown (Figure 2j). The magnitude of the effect of heavy water may vary slightly depending on the type of tissue. No consistent differences were found in elongation rates among the SCN, peripheral tissues, and cells. Therefore, the robustness of the SCN clock did not provide resistance to the period-elongation effect of heavy water.
The mammalian circadian oscillator consists of the TTFL of the clock genes (Dunlap, 1999; Reppert and Weaver, 2002). In the present study, the characteristics of circadian rhythms were calculated by measuring the expression of Per2, a core clock gene, in real time. The elongation of Per2 expression rhythm indicated that heavy water affected the TTFL of the clock genes. The molecular clock is present in most mammalian cells and produces circadian rhythms by the same molecular mechanism (Zhang and Kay, 2010). Because the elongation rate was almost the same for all tissue types, the period-elongation effect of heavy water was considered to act at the cellular level via the same mechanism.
Under 97% heavy water conditions, both the SCN and C6 cells lost the circadian rhythm of luminescence (Figure 1 and Suppl. Fig. S1). The molecular clock mechanism may have stopped or it is possible that the physiological ability of the cell to emit luminescence was diminished. No rhythm was observed in the C6 cells even under 87% heavy water conditions. However, this may be the result of the phases of each cell becoming asynchronous due to the variation in their periods. It is considered that the SCN maintained its rhythm under 87% heavy water conditions because of intercell communication. This issue will be clarified in a future study by performing single-cell level analysis using a CCD image sensor.
Regardless of the tissue type and heavy water concentration, the period was either restored to its original value by washing out the medium or it was not restored at all. Interestingly, the C6 cells lost their rhythm with 98% heavy water medium, but washing out the medium increased their luminescence and stimulation restarted the rhythm with the original period (Suppl. Table S1). These results indicate that the period-elongation effect of heavy water on the molecular mechanism of circadian rhythms is reversible. In contrast, the luminescence of the SCN explant disappeared in 98% heavy water and did not increase even when the medium was washed out (Figure 1a). Although cell death was not verified in these experiments, the disappearance of luminescence suggests that the SCN tissue was irreversibly damaged by the higher concentrations of heavy water. The intensity of damage caused by heavy water may vary depending on the cell type.
The effects of heavy water on animals and SCN explants appear to be somewhat different. The first difference is the period length. Previous studies reported that drinking heavy water resulted in a dose-dependent elongation of the free-running periods of mice, rats, hamsters, and deer mice (Daan and Pittendrigh, 1976; Lesauter and Silver, 1993; Richter, 1970; Suter and Rawson, 1968). In our study, the circadian periods seen in the mouse SCN explants tended to be longer than those of animals (Daan and Pittendrigh, 1976). This difference may be due to the metabolized water in the body. Metabolized water is produced by metabolizing food. The bait provided to the animals does not contain deuterium, and the metabolized water does not become heavy water. The concentration of heavy water in extracellular fluid is therefore lower than the concentration of heavy water in the drinking water (Daan and Pittendrigh, 1976). The second difference is the lag between the timing of heavy water supply and the appearance of the effects. In the case of experiments using animals, it took several cycles until the elongated period stabilized during intake of heavy water (Daan and Pittendrigh, 1976; Lesauter and Silver, 1993). In contrast, in the present study, the period of the clock gene expression rhythm in the SCN changed and stabilized within 1 cycle when the concentration of heavy water in the medium was increased or decreased (Figure 1d). This shows that if the extracellular fluid in direct contact with the cells is replaced with heavy water, the effect will appear rapidly. It took several days for the concentration of heavy water in the body fluid to reach equilibrium in previous in vivo studies (Daan and Pittendrigh, 1976).
The result of the degradation test of PER2::LUC was surprising. We expected a slower degradation rate, but under heavy water conditions, the time taken for the PER2::LUC amount to reach half of the initial value was not different from that of the control group (Suppl. Figs. S2B and S2C). Moreover, the level in the heavy water group decreased to a quarter faster than that in the control group. The time to reach the 75% amount under heavy water conditions was delayed because the PER2::LUC amount increased in the initial stage (arrow in Suppl. Fig. S2A). The rate of PER2 degradation is controlled by phosphorylation by casein kinase 1 (CK1; Brenna and Albrecht, 2020; Lowrey et al., 2000). The phosphorylation of Familial Advanced Sleep Phase (FASP) sites promoted stabilization of PER2, and phosphorylation of β-TrCP sites promoted degradation of PER2 (Eide et al., 2005; Vanselow et al., 2006). If the phosphorylation in β-TrCP sites is predominant owing to mutation in the CK1ε gene or low temperature, PER2 degradation is accelerated (Lowrey et al., 2000; Ralph and Menaker, 1988; Zhou et al., 2015). In addition, an initial sharp increase in PER2 abundance was observed when a CK1δ/ε inhibitor was administered to the MEFs together with a protein synthesis inhibitor (Zhou et al., 2015). In our study, the heavy water may have affected the functions of CK1 and PER2 phosphorylation. However, cell health was clearly impaired in the heavy water group, because the luminescence intensity after washout was low. We will verify cell health and quantify PER2 phosphorylation in a future study. However, it is unlikely that this change in the degradation curve of PER2 resulted in the period elongation. It is considered that heavy water caused delays in other molecular clock processes such as transcription and translation. Further studies are needed to elucidate the cause of the period change by heavy water.
Supplemental Material
sj-pdf-1-jbr-10.1177_07487304211012905 – Supplemental material for Heavy Water Lengthens the Molecular Circadian Clock Period in the Suprachiasmatic Nucleus of Mice In Vitro
Supplemental material, sj-pdf-1-jbr-10.1177_07487304211012905 for Heavy Water Lengthens the Molecular Circadian Clock Period in the Suprachiasmatic Nucleus of Mice In Vitro by Mitsugu Sujino, Satoshi Koinuma, Yoichi Minami and Yasufumi Shigeyoshi in Journal of Biological Rhythms
Footnotes
Acknowledgements
This work was supported by Japan Society for the Promotion of Science KAKENHI Grant Numbers 17K08580 (Y.S.) and 15K08221 (M.S.).
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
NOTE
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
