Abstract
“Non-image-forming” (NIF) effects of light are mediated primarily by a subset of intrinsically photosensitive retinal ganglion cells (ipRGCs) expressing the photopigment, melanopsin (OPN4). These NIF functions include circadian entrainment, pupillary reflexes, and photic effects on sleep, mood, and cognition. We recently reported that mice of multiple genotypes exhibit reduced voluntary ethanol intake under both constant darkness (DD) and constant light (LL) relative to standard light-dark (LD) conditions. In the present study, we sought to determine whether these effects are mediated by melanopsin-expressing ipRGCs and their potential relationship to photic effects on the circadian system. To this end, we examined the effects of environmental lighting regimen on both ethanol intake and circadian activity rhythms in a genetically engineered mouse model (Opn4aDTA/aDTA) in which melanopsin expression is completely blocked while ipRGCs are progressively ablated due to activation of attenuated diphtheria toxin A (aDTA) transgene under the control of the Opn4 promoter. As expected from previous studies, Opn4aDTA/aDTA mice displayed dramatic attenuation of circadian photosensitivity, but surprisingly, showed identical suppression of ethanol intake under both DD and LL as that seen in controls. These results demonstrate that the effects of lighting regimen on voluntary ethanol intake are independent of melanopsin-expressing ipRGCs and ipRGC-mediated photic effects on the circadian system. Rather, these effects are likely mediated by classical retinal photoreceptors and central pathways.
In addition to its traditional role in mediating visual perception, environmental light exerts several “non-visual” or “non-image-forming” (NIF) effects on physiology and behavior. Such NIF effects include entrainment of the circadian clock, mediation of pupillary reflexes, and modulation of sleep, arousal, activity, mood, and cognitive function. Research conducted over the last two decades has revealed that NIF effects in mammals are mediated largely by a small population of intrinsically photosensitive retinal ganglion cells (ipRGCs) that contain the photopigment, melanopsin (OPN4) (Schmidt et al., 2011; LeGates et al., 2014; Lazzerini Ospri et al., 2017; Duda et al., 2020), first identified in non-retinal photosensitive cells, such as dermal melanophores and deep-brain hypothalamic photoreceptors in amphibia (Provencio et al., 1998).
In humans, the earliest evidence that distinct photoreceptors might mediate visual perception and NIF effects was the finding that environmental light could suppress pineal melatonin secretion in some blind individuals, even in the complete absence of conscious light perception (Czeisler et al., 1995). Similarly, animal experiments revealed that circadian photosensitivity persists in rodless mice carrying the retinal degeneration (RD) mutation (Foster et al., 1991; Provencio et al., 1994) and even in genetically engineered mice completely lacking both rods and cones (Freedman et al., 1999). Furthermore, anatomical tracing studies demonstrated that melanopsin-containing ipRGCs project selectively to the master circadian pacemaker, the suprachiasmatic nucleus (SCN), and to other “non-visual” brain areas, but not to classic visual structures such as the dorsal lateral geniculate nucleus (Hattar et al., 2002). Surprisingly, however, melanopsin knockout (Opn4-/-) mice display typical entrainment to light-dark (LD) cycles (Panda et al., 2002; Ruby et al., 2002), indicating that while melanopsin signaling may be sufficient for circadian entrainment, it is clearly not necessary. On the contrary, Opn4-/- mice display attenuated circadian phase-shifting responses to acute light pulses and blunting (but not elimination) of period-lengthening normally seen under constant light (LL; Panda et al., 2002; Ruby et al., 2002), indicating that circadian photosensitivity is reduced, but not eliminated, in the absence of melanopsin-based photoreception. These results led to the hypothesis that both classical photoreceptors and ipRGCs normally contribute to circadian photoreception in intact animals, and indeed, this hypothesis was subsequently confirmed by findings that circadian photoreception is fully abrogated in both (a) “triple-knockout” mice lacking functional rod-, cone-, and melanopsin-dependent photoreception (Hattar et al., 2003; Panda et al., 2003) and (b) after genetically targeted ablation of melanopsin-expressing ipRGCs using Opn4 promoter-driven expression of either attenuated diphtheria toxin subunit A (aDTA) or full-length DTA (Güler et al., 2008; Chew et al., 2017). This latter finding is particularly significant since it demonstrates that while conventional retinal ganglion cells may also innervate the SCN (Sollars et al., 2003), classical photoreceptors contribute to circadian photoreception via retinal circuitry requiring intact ipRGCs (Güler et al., 2008).
Growing research has revealed widespread effects of environmental lighting on mood- and cognition-related behaviors (Stephenson et al., 2012; LeGates et al., 2014; Landgraf et al., 2014; Bedrosian and Nelson, 2017). These studies have employed a variety of non-traditional lighting regimens, including short- and long-photoperiod LD cycles, repeated LD phase-shifts, LL and constant darkness (DD), nocturnal light exposure, and non-24-h LD cycles. To date, however, direct evidence linking melanopsin and/or ipRGCs to the photic modulation of mood and cognition is actually quite limited. LeGates et al. (2012) showed that depression-like behavior, learning deficits, and impaired hippocampal long-term potentiation normally induced by exposure to an ultradian LD cycle (3.5 h light alternating with 3.5 h darkness) were prevented in Opn4aDTA/aDTA mice. Furthermore, LeGates et al. (2012) argued that these effects were unlikely to be related to either circadian or sleep disturbances since Opn4aDTA/aDTA mice displayed coherent free-running activity rhythms and normal sleep architecture. Interestingly, a subsequent study (Fernandez et al., 2018) showed that the affective disturbances and cognitive deficits seen under the ultradian LD cycle are mediated by separate subpopulations of ipRGCs with distinct central projections. Nevertheless, there is currently no evidence extending such findings to other atypical lighting regimens.
Our lab recently showed that multiple strains of mice all exhibit reduced voluntary ethanol intake in a two-bottle free-choice access protocol under either DD or LL, relative to standard LD 12:12 conditions (Rosenwasser and Fixaris, 2013; Rosenwasser et al., 2020). Given that essentially identical effects are seen under both DD and LL, these effects are unlikely to be mediated by circadian disruption, photoperiodism, or direct photic effects on mood or arousal. While ethanol intake is influenced by psychopharmacological, sensory, caloric, and genetic factors (Grahame and Cunningham, 1997; Tabakoff and Hoffman, 2000; Bachmanov et al., 2003; Crabbe et al., 2010), it is well-known that ethanol consumption reciprocally interacts with depression- and anxiety-like affective states (Stevenson et al., 2009; Lee et al., 2015; McCool and Chappell, 2015). Therefore, we have hypothesized that attenuated ethanol intake under DD and LL may be related to a stress-like state associated with a lack of stable circadian entrainment. This hypothesis is consistent with findings that depression-like behavior is increased under both DD and LL (Gonzalez and Aston-Jones, 2008; Işman et al., 2010; Martynhak et al., 2011; Tapia-Osorio et al., 2013) and that environmental stressors tend to reduce ethanol intake in non-dependent mice (Becker et al., 2011; Lopez et al., 2016).
The present study was designed to explore the retinal mechanisms mediating the effects of DD and LL on voluntary ethanol intake in mice, and the possible relationship of these effects to photic effects on the circadian system. To this end, we examined the effects of environmental lighting regimen (LD, DD, LL) on both circadian activity rhythms and voluntary ethanol intake in Opn4aDTA/aDTA mice and wild-type (WT) controls. As described briefly above, this mutation abrogates melanopsin expression and results in progressive age-dependent ablation of ipRGCs, thus effectively blocking rod, cone, and ipRGC input to the SCN circadian pacemaker, as well as to certain other non-visual brain areas (Güler et al., 2008). Our data demonstrate that suppression of ethanol intake under DD and LL occurs independent of either melanopsin-expressing ipRGCs or photic effects on the circadian pacemaker.
Materials and Methods
Animals
Breeding pairs (n = 4/sex) of homozygous knock-in Opn4aDTA/aDTA mice were shipped to the University of Maine from colonies maintained at Johns Hopkins University (Baltimore, MD). To generate Opn4aDTA/aDTA mice, the Opn4 coding region was replaced with a transgene encoding aDTA. This both prevents Opn4 expression and results in aDTA-induced age-dependent degeneration of ipRGCs under the control of the Opn4 promoter. Breeder mice arrived in the laboratory at approximately 6 to 8 weeks of age and were immediately housed individually in standard mouse cages (30 × 18 × 12 cm) under a LD 12:12 lighting regimen. Subsequently, after a week of acclimation, male and female mice were bred in-house (1M:1F), and progeny were weaned at 21 days. Weaned mice were housed (under LD 12:12) with up to five same-sex animals until experimentation began at approximately 8 weeks of age. The total number of Opn4aDTA/aDTA mice used for experimentation was 46 (M: n = 24, F: n = 22).
Since Opn4aDTA/aDTA mice are maintained as homozygotes, we were unable to utilize littermate controls. Therefore, WT mice (n = 22/sex) of the same mixed B6x129 genetic background as the mutant line (B6129SF2/J; The Jackson Laboratory, Bar Harbor, ME) served as approximate controls and arrived at the University of Maine at approximately 6 weeks of age.
Procedures
At approximately 6 weeks of age, all animals were individually housed in running-wheel cages (32 × 20 × 14 cm) under a LD 12:12 lighting regimen for a 2-week acclimation period with ad libitum access to food and water (but not ethanol). Running-wheel cages were placed within light-shielded and sound-attenuating metal cabinets equipped with 15-watt fluorescent bulbs on each shelf (approximately 100 lux depending on cage position) and ventilation fans that also provided masking noise. Each cabinet held a total of 15 running-wheel cages and both males and females were housed within each cabinet.
Following the acclimation period, mice were maintained under one of two distinct sequences of lighting conditions for 18 days per condition, as follows: Sequence 1: LD 12:12 (LD1), DD, LD 12:12 (LD2); Sequence 2: LD 12:12 (LD1), LL, LD 12:12 (LD2). At the onset of LD1, 10% v/v ethanol solutions were introduced via a second drinking bottle with continued access throughout the remainder of the experiment (as described below). Food (Prolab RMH 3000; LabDiet, St. Louis, MO) and tap water were available ad libitum throughout the experiment. All experimental procedures were approved by the University of Maine Institutional Animal Care and Use Committee (IACUC).
Wheel-running
Animals were given continuous access to an in-cage running-wheel (wheel diameter: 11.5 cm; model ACT-551; Coulbourn Instruments, Whitehall, PA) for the duration of the experiment. Wheel-turns were logged in 1-min bins using the ClockLab interface system (Actimetrics Co., Wilmette, IL), with compiled wheel-turns per day providing a marker for daily activity. In addition, for each individual animal, in each lighting condition, circadian period and periodogram amplitude were calculated using the Lomb-Scargle periodogram (Ruf, 1999; Refinetti et al., 2007), as implemented in ClockLab.
Two-bottle Free-choice Ethanol Consumption
Animals were given continuous free-choice access to bottles containing either a 10% v/v ethanol solution or plain water for a total of 54 days. The physical location (right or left) of the water and ethanol solutions was switched daily to reduce potential side preference effects. Pre- and post-measurements of bottle weight, along with animals’ respective body weights following each 18-day period of ethanol access (data not shown), were used to obtain body weight–adjusted ethanol intake per day (g/kg/day) over the course of the experiment. Progressive increases in body weight were observed over the course of the experiment in all groups (DD/F, LL/F, DD/M, LL/M), regardless of lighting condition. While there were no consistent effects of genotype, both initial (start of LD1) and final (conclusion of LD2) body weight was somewhat lower in KO relative to WT mice. Ethanol preference was determined by dividing ethanol solution intake by total fluid intake and multiplying this ratio by 100 to represent as a percentage. Water intake was measured in grams per day (g/day), which is not body weight–adjusted since water is not a drug and does not possess any caloric value. Finally, separate empty cages (n = 3) were set up and handled identically to account for bottle leakage caused by evaporation, cage handling, and/or bottle weighing. Obtained leakage values (g) were subtracted from individual mouse raw intake data (g) to calculate corrected ethanol intake (g/kg/day), preference ratios, and water intake (g/day).
Design and Statistics
In all, this experiment involved eight independent groups of mice, based on genotype (Opn4aDTA/aDTA vs. WT), sex (F vs. M), and sequence (Sequence 1: LD1/DD/LD2 vs. Sequence 2: LD1/LL/LD2). Thus, each animal in all groups was exposed to a series of three lighting conditions, creating a repeated-measures factor referred to here as lighting condition. Because (a) the primary objective of this study was to identify possible interactions between genotype and lighting condition, (b) DD and LL are known to exert dissimilar effects on circadian rhythm parameters, and (c) female mice are known to consistently display higher levels of voluntary ethanol intake than males, data were analyzed by conducting separate two-factor (Genotype × Lighting Condition) mixed analysis of variance (ANOVA) for each Sex × Sequence combination (DD/F, LL/F, DD/M, LL/M). All statistical tests used p < 0.05 as the criterion for significance, although comparisons with p < 0.10 are occasionally mentioned in the text. Data analyses were performed using SPSS 25.0 (IBM Inc., Armonk, NY) while graphs and actograms were generated using SigmaPlot 10.0 (Systat Software Inc., San Jose, CA) and ClockLab (Actimetrics Co., Wilmette, IL), respectively.
Results
Qualitative Features of Circadian Activity Rhythms
Inspection of circadian actograms (Figures 1 and 2) shows that circadian behavior in WT mice conformed closely to well-established expectations for nocturnal mice. All WT mice displayed well-entrained activity rhythms with activity onsets occurring at or near dark-onset under LD1; coherent free-running activity rhythms with periods somewhat shorter than 24 h under DD; free-running periods substantially longer than 24 h and frequently associated with moderate to severe loss of rhythmic coherence under LL; and rapid (1-3 days) re-entrainment under LD2. In contrast, activity rhythms in Opn4aDTA/aDTA mice displayed several features that were clearly distinct from those of WT mice. As seen previously (Güler et al., 2008), the period-lengthening effects of LL were completely abrogated in Opn4aDTA/aDTA mice; indeed, Opn4aDTA/aDTA mice displayed essentially identical free-running periods in both LL and DD. In addition, activity rhythms in LL were more coherent and consolidated in Opn4aDTA/aDTA relative to WT mice. Despite these marked differences in LL, there were no apparent differences between genotypes in either circadian period or rhythmic coherence in DD.
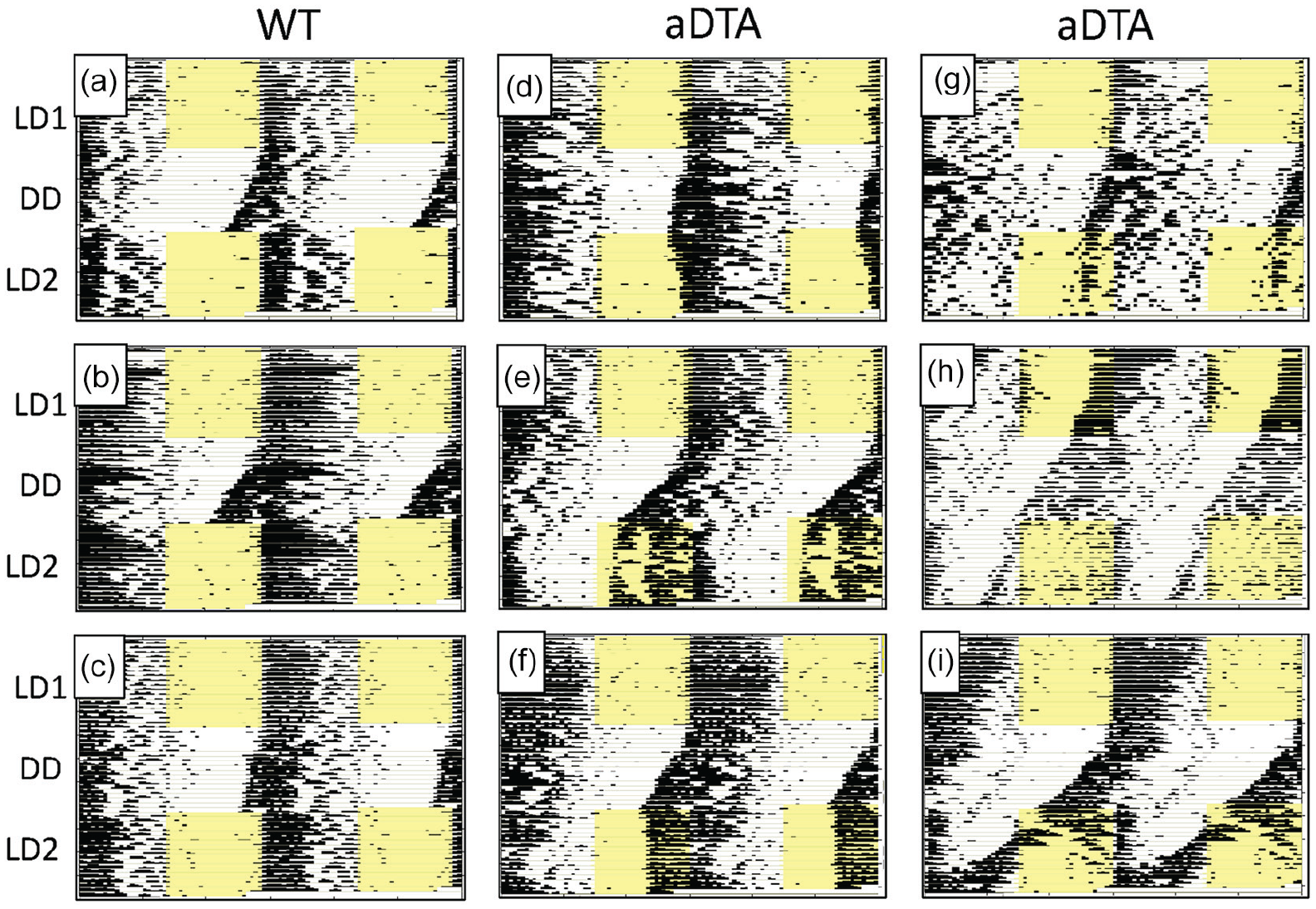
Raster-style circadian actograms illustrating temporal patterning of wheel-running activity of representative WT (a-c) and Opn4aDTA/aDTA (d-i) mice from the DD Group. Most Opn4aDTA/aDTA mice displayed circadian entrainment abnormalities under LD2, including extended transients (d), stable entrainment at an abnormal entrainment phase (e-g) and failed re-entrainment (h, i). Abbreviations: WT = wild-type; aDTA = attenuated diphtheria toxin A; DD = constant darkness; LD = light-dark.
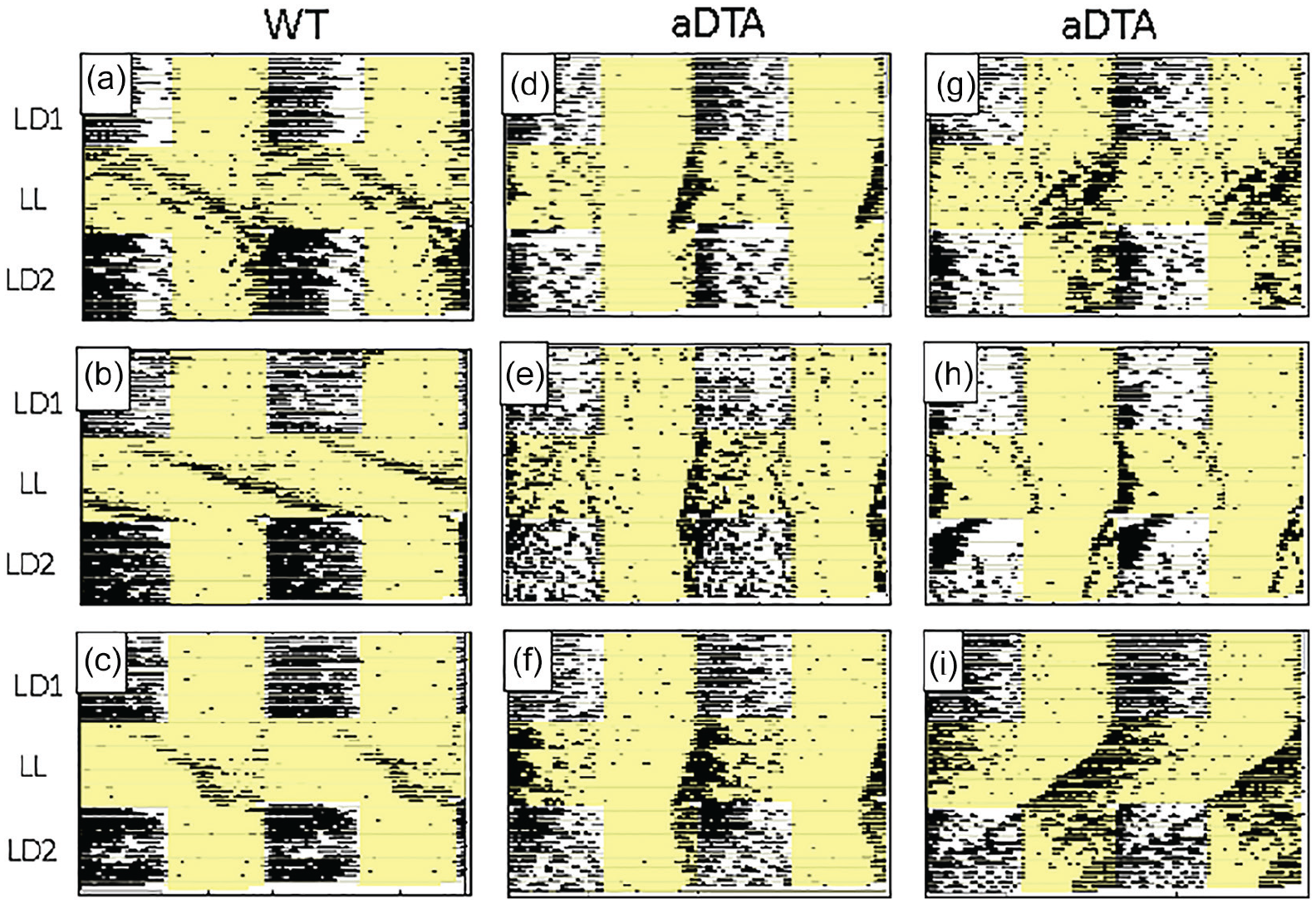
Raster-style circadian actograms illustrating temporal patterning of wheel-running activity of representative WT (a-c) and Opn4aDTA/aDTA (d-i) mice from the LL Group. All plotting conventions as in Figure 1. Opn4aDTA/aDTA mice consistently showed shorter free-running periods than WT mice under LL. In addition, most Opn4aDTA/aDTA mice displayed circadian entrainment abnormalities under LD2, including extended transients (e, f), atypical entrainment phase (g), and failed re-entrainment (h, i). Abbreviations: WT = wild-type; aDTA = attenuated diphtheria toxin A; LL = constant light; LD = light-dark.
Beyond these effects on free-running rhythms in LL, many, but not all, Opn4aDTA/aDTA mice also displayed distinct anomalies in circadian entrainment. While these observations were more common and more pronounced in LD2, a few Opn4aDTA/aDTA mice displayed entrainment deficiencies even during LD1. Thus, of the 46 Opn4aDTA/aDTA mice, 2 failed to entrain in LD1 (Figure 1h) and 3 entrained at a clearly aberrant phase, with activity onsets occurring in the mid-light phase (not shown). In LD2, 8 Opn4aDTA/aDTA mice failed to re-entrain (including the 2 that had failed to entrain in LD1) (Figures 1h, 1i, 2h, and 2i); 14 re-entrained but at an aberrant phase (Figures 1e, 1f, 1g, and 2g); and 2 eventually re-entrained at a normal phase, but only after an extended number of transient cycles (Figures 1d and 2f). Opn4aDTA/aDTA mice were somewhat more likely to display entrainment abnormalities in LD2 following LL than following DD: Of the 24 mice that showed atypical circadian entrainment in LD2, 10 (42%) were from the DD group and 14 (58%) were from the LL group. Finally, male Opn4aDTA/aDTA mice were somewhat more likely to display entrainment abnormalities than females: While 15 of 24 (62.5%) males showed abnormal entrainment under either LD1 or LD2 (or both), similar abnormalities were seen in 9 of 22 (40.9%) females.
Circadian Activity Parameters
Circadian Period
ANOVA (Genotype × Lighting Condition) revealed significant main effects of lighting condition on circadian period in all four groups (DD/F: F2,38 = 9.902, p < 0.001,
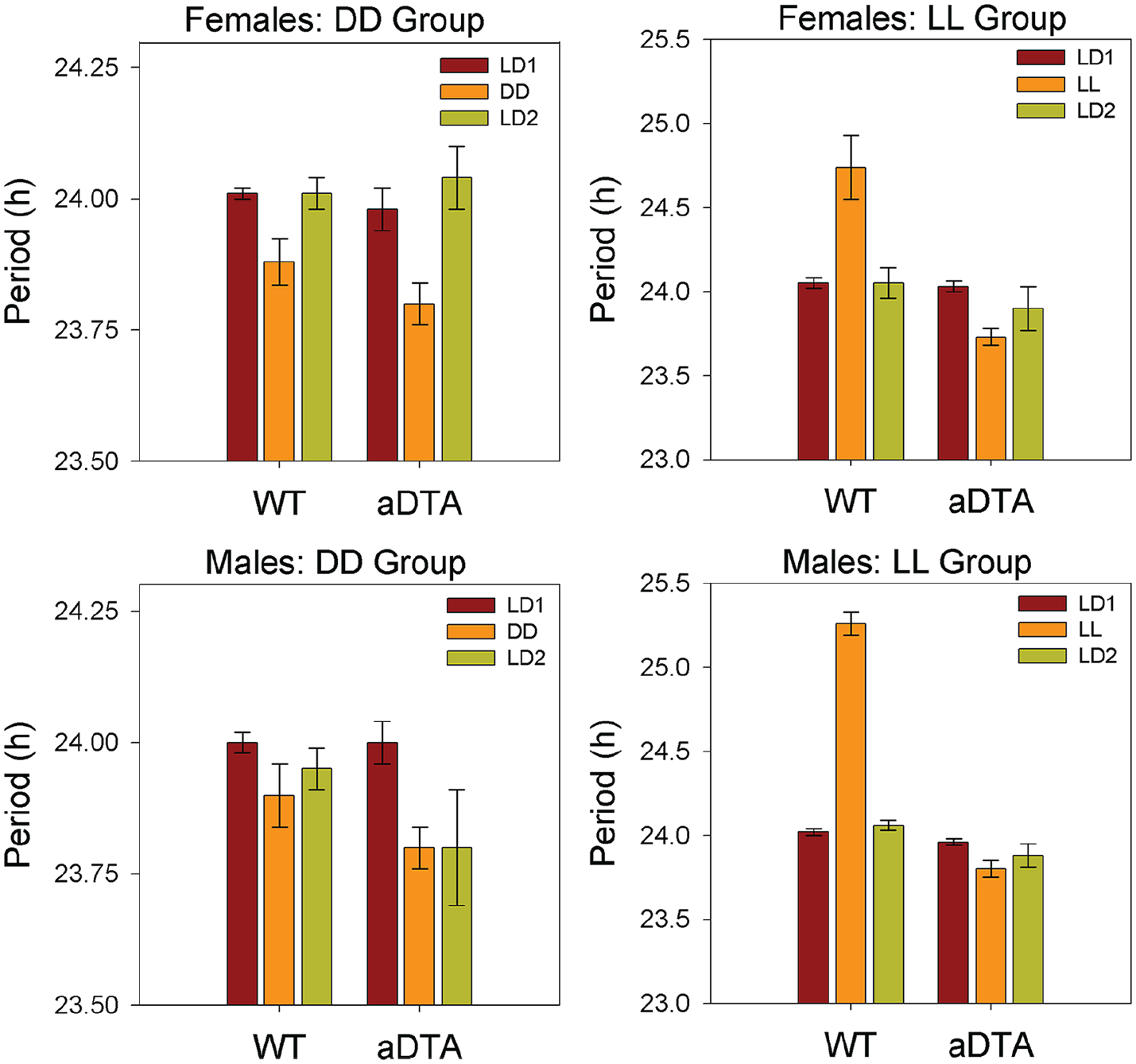
Means ± standard error of the mean for circadian period in WT and Opn4aDTA/aDTA mice, plotted by sex (rows) and lighting group (columns). Data are collapsed across days within each lighting condition. Lighting conditions are identified as in Figures 1 and 2. Abbreviations: WT = wild-type; aDTA = attenuated diphtheria toxin A; DD = constant darkness; LD = light-dark; LL = constant light.
Periodogram Amplitude
ANOVA (Genotype × Lighting Condition) revealed significant main effects of lighting condition on periodogram amplitude in all four groups (DD/F: F2,38 = 31.863, p < 0.001,
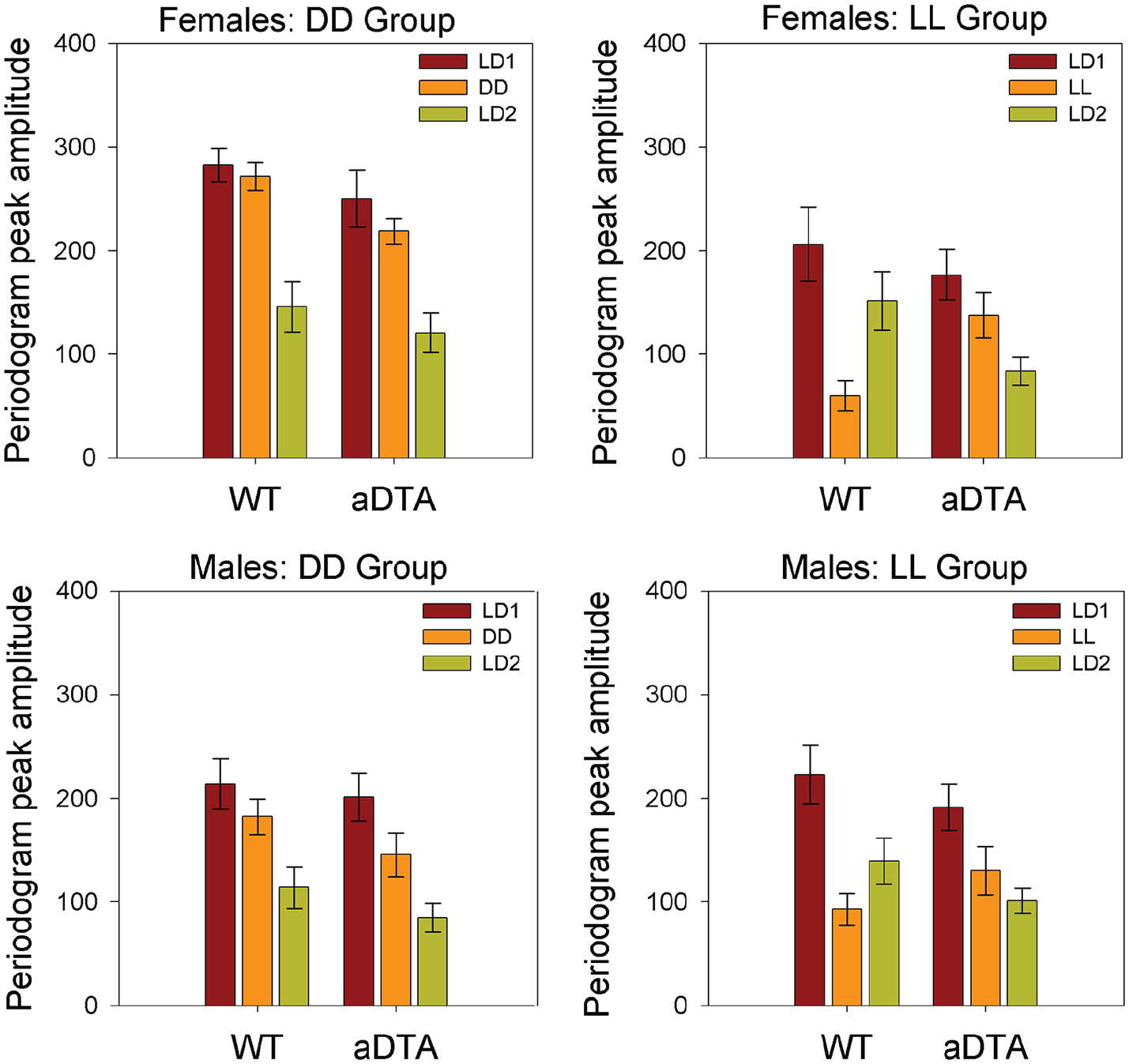
Means ± standard error of the mean for periodogram amplitude in WT and Opn4aDTA/aDTA mice, plotted by sex (rows) and lighting group (columns). Data are collapsed across days within each lighting condition. Lighting conditions are identified as in Figures 1 and 2. Abbreviations: WT = wild-type; aDTA = attenuated diphtheria toxin A; DD = constant darkness; LD = light-dark; LL = constant light.
Activity Level
ANOVA (Genotype × Lighting Condition) revealed significant main effects of lighting condition on daily activity level in all four groups (DD/F: F2,38 = 5.115, p = 0.011,
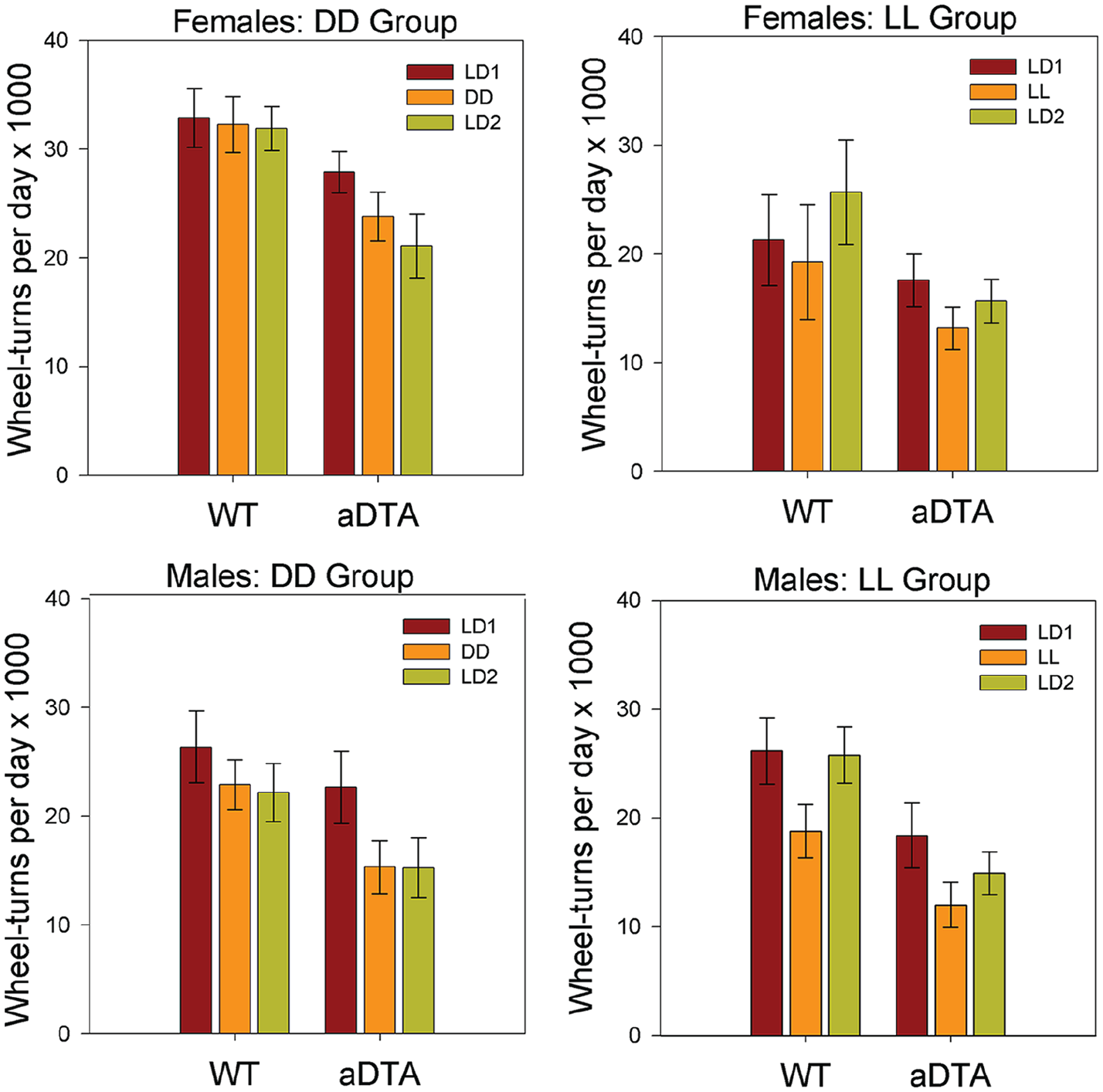
Means ± standard error of the mean for daily wheel-turns in WT and Opn4aDTA/aDTA mice, plotted by sex (rows) and lighting group (columns). Data are collapsed across days within each lighting condition. Lighting conditions are identified as in Figures 1 and 2. Abbreviations: WT = wild-type; aDTA = attenuated diphtheria toxin A; DD = constant darkness; LD = light-dark; LL = constant light.
Fluid Intake Parameters
Ethanol Intake
ANOVA (Genotype × Lighting Condition) revealed significant main effects of lighting condition on ethanol intake for all four groups (DD/F: F2,38 = 9.466, p < 0.001,
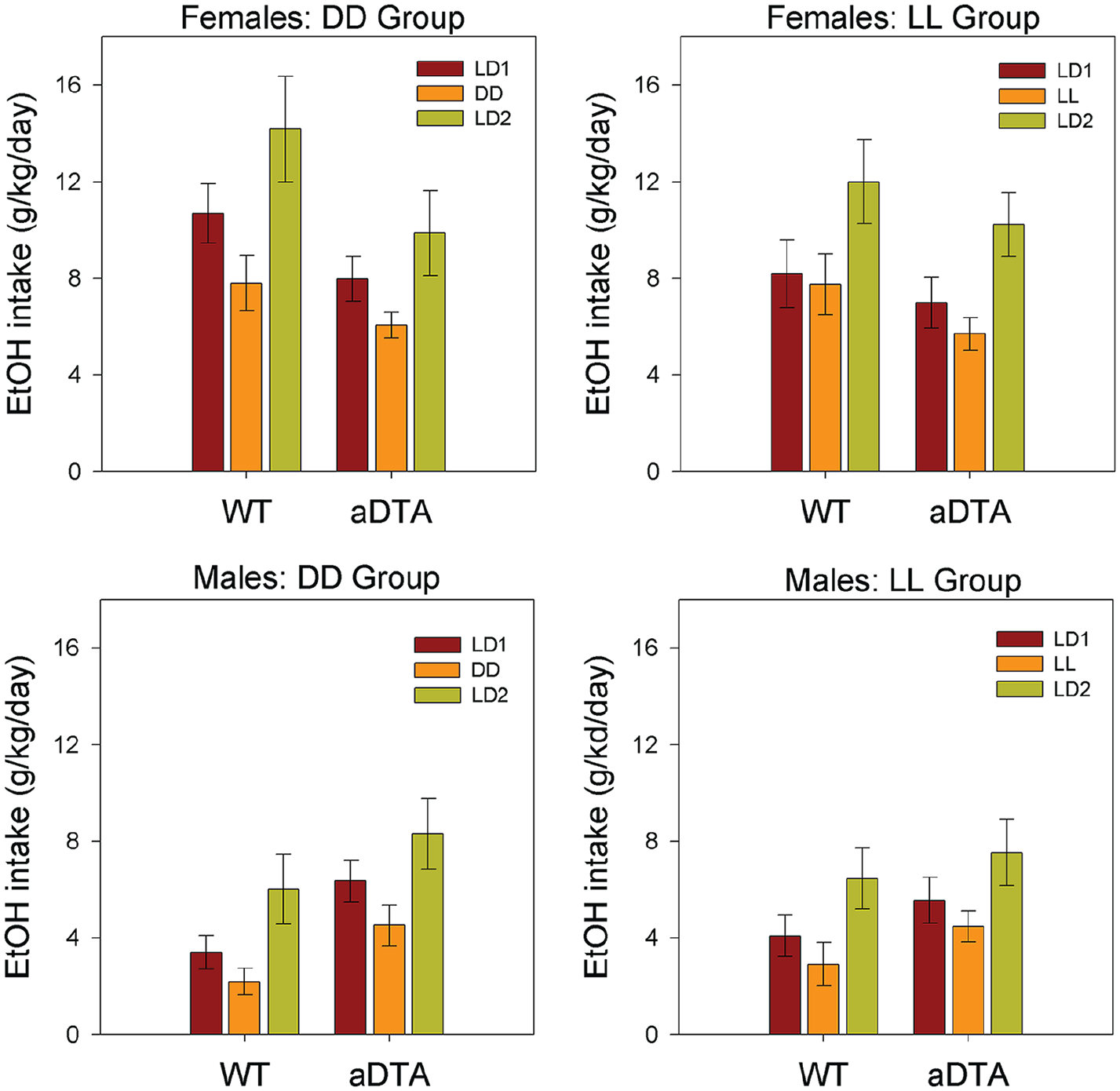
Means ± standard error of the mean for ethanol intake in WT and Opn4aDTA/aDTA mice, plotted by sex (rows) and lighting group (columns). Data are collapsed across days within each lighting condition. Lighting conditions are identified as in Figures 1 and 2. Abbreviations: WT = wild-type; aDTA = attenuated diphtheria toxin A; DD = constant darkness; LD = light-dark; LL = constant light.
Ethanol Preference
ANOVA (Genotype × Lighting Condition) revealed significant main effects of lighting condition on ethanol preference for both male groups (DD/M: F2,44 = 4.476, p = 0.018,
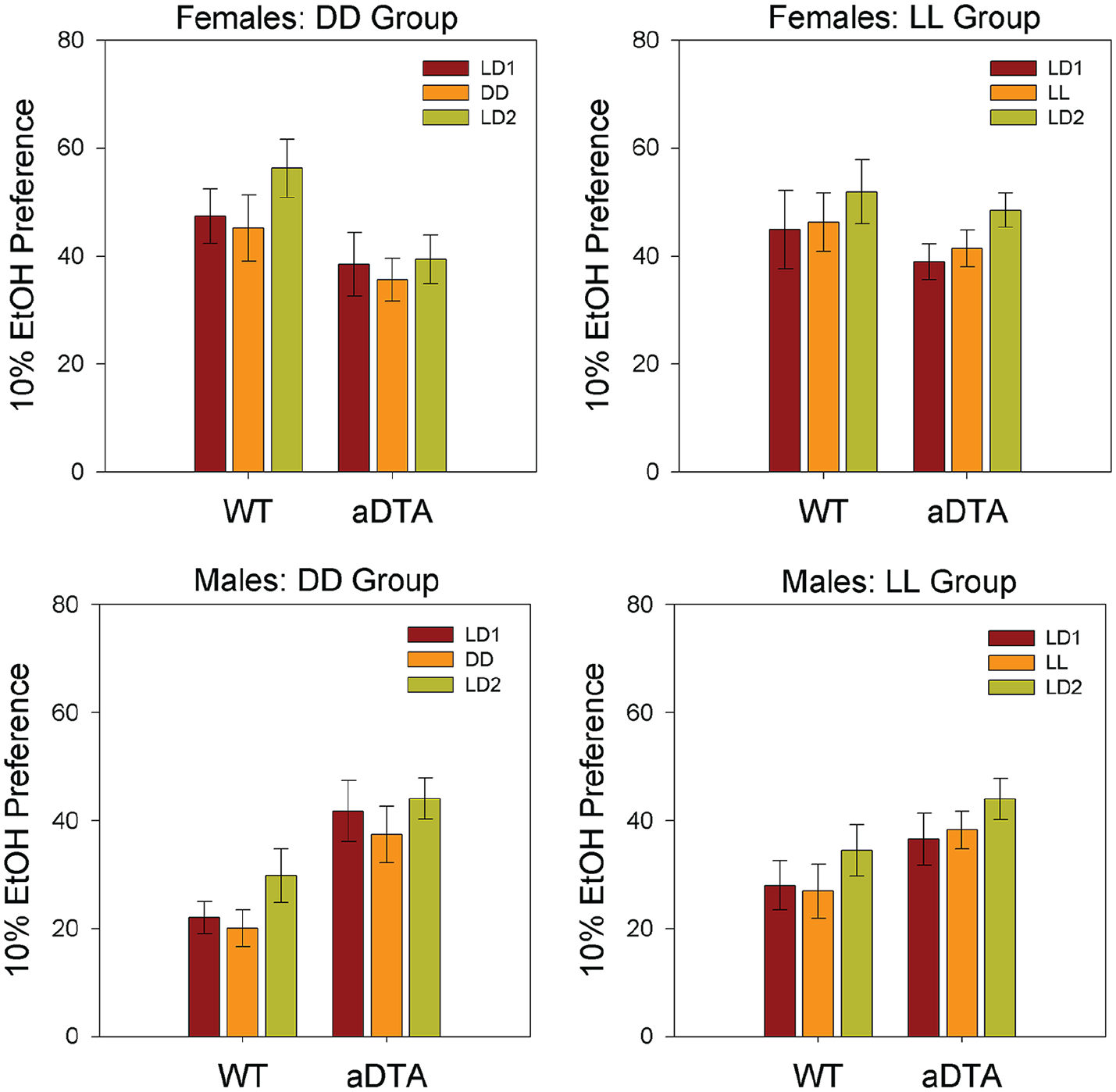
Means ± standard error of the mean for 10% ethanol preference in WT and Opn4aDTA/aDTA mice, plotted by sex (rows) and lighting group (columns). Data are collapsed across days within each lighting condition. Lighting conditions are identified as in Figures 1 and 2. Abbreviations: WT = wild-type; aDTA = attenuated diphtheria toxin A; DD = constant darkness; LD = light-dark; LL = constant light.
Water Intake
Water intake was rather stable across conditions in most groups, and ANOVA (Genotype × Lighting Condition) revealed a significant main effect of lighting condition on water intake only for the LL/M group (F2,40 = 7.732, p = 0.002,
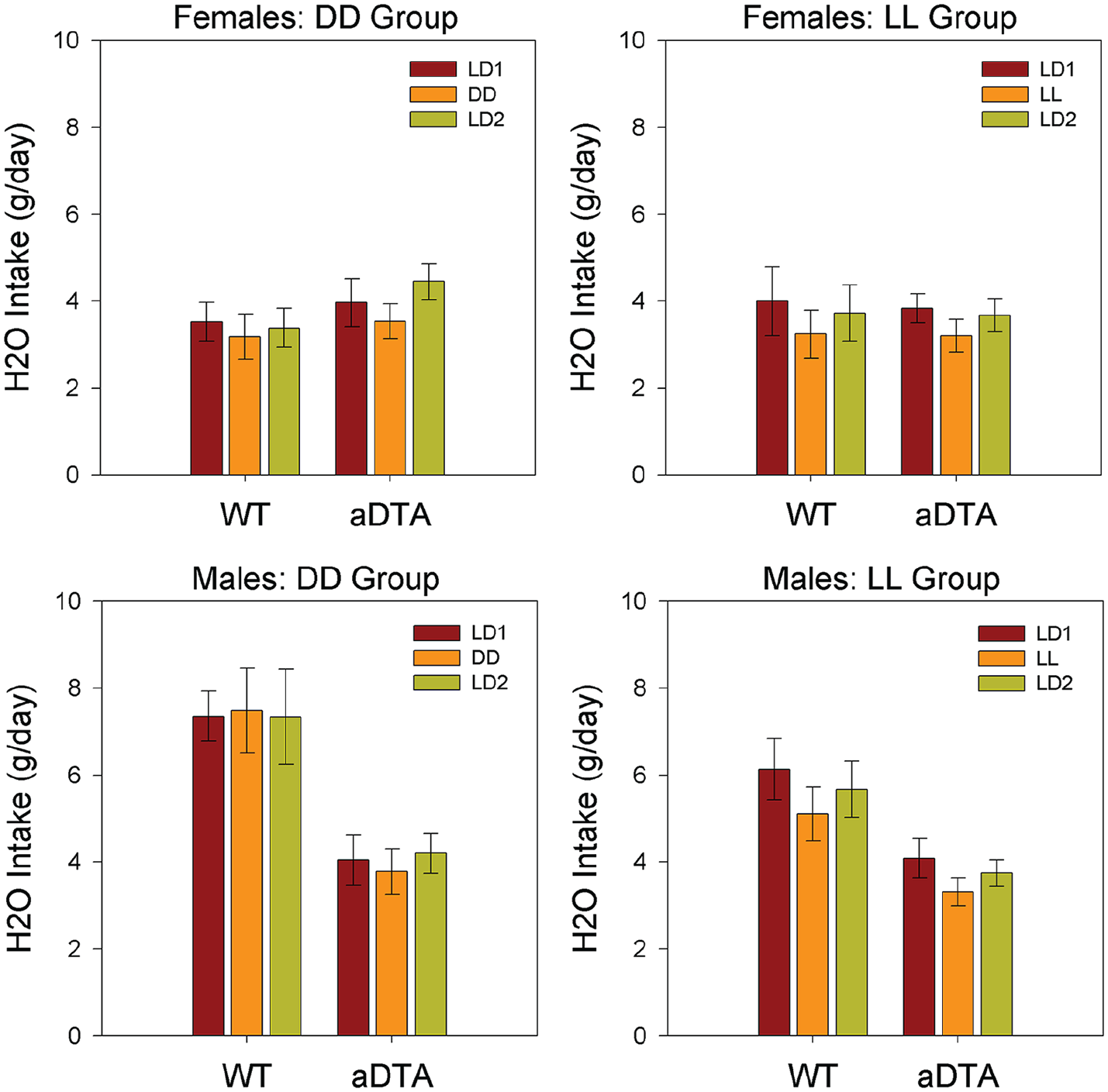
Means ± standard error of the mean for water intake in WT and Opn4aDTA/aDTA mice, plotted by sex (rows) and lighting group (columns). Data are collapsed across days within each lighting condition. Lighting conditions are identified as in Figures 1 and 2. Abbreviations: WT = wild-type; aDTA = attenuated diphtheria toxin A; DD = constant darkness; LD = light-dark; LL = constant light.
Discussion
Circadian Rhythms
Opn4aDTA/aDTA and WT mice displayed entrained and free-running circadian activity rhythms that closely conformed to prior studies utilizing these genotypes. Thus, WT mice exhibited stably entrained activity rhythms with daily activity onsets at or near dark-onset under LD1, free-running circadian rhythms with periods somewhat shorter than 24 h under DD and substantially longer than 24 h under LL, and rapid re-entrainment within 1 to 3 days under LD2. In addition, WT mice generally showed modest to extreme loss of rhythmic coherence in LL.
In contrast, circadian photosensitivity, assessed by both LD entrainment and period-lengthening under LL, was dramatically impaired Opn4aDTA/aDTA relative to WT mice, and in some animals, appeared to be completely eliminated. Overall, circadian phenotypes displayed by Opn4aDTA/aDTA mice were quite similar to, although less extreme, than those reported by Güler et al. (2008). This is likely due to the fact that Güler et al. (2008) began testing at an older age than in the present study (6 months vs. 6-8 weeks), at a time when ipRGC degeneration is more advanced (although still not complete; Güler et al., 2008, Suppl. Fig. S.2A). Therefore, while Güler et al. (2008) reported considerable variability in circadian phenotype, a higher proportion of their animals failed to entrain to LD cycles than in the present study. Nevertheless, in the present study, a much higher proportion of entrainment anomalies were seen in LD2 than in LD1, possibly due to aging of the animals, as well as to aftereffects of the preceding free-running conditions. Furthermore, even at the younger ages tested here, Opn4aDTA/aDTA mice completely failed to exhibit prototypical lengthening of free-running period under LL, and showed essentially identical periods in both LL and DD. Remarkably, this was true even in animals who successfully entrained under both LD1 and LD2. These observations suggest that rod- and cone-dependent signaling to the SCN circadian pacemaker, likely mediated via synaptic interactions with surviving ipRGCs, is sufficient to mediate entrainment to LD cycles, but not to result in period-lengthening in LL. Thus, we suggest that the contribution of classical photoreceptors to circadian photosensitivity may be greater for the so-called “phasic” rather than the “tonic,” effects of light (Daan, 1977, 2000).
To our knowledge, this is the first study of circadian behavior in Opn4aDTA/aDTA mice to include a measure of circadian activity rhythm “robustness,” in this case, the peak amplitude of the Lomb-Scargle periodogram (Ruf, 1999; Refinetti et al., 2007). Periodogram amplitudes generally decreased over the course of the experiment in the DD groups for both sexes and genotypes, but this appeared to be independent of lighting condition. Instead, we interpret these changes to be due to the simple passage of time or perhaps to aging of the animals. In contrast, however, clear Genotype × Lighting interactions were seen in the LL groups. Specifically, while periodogram amplitude was suppressed under LL in WT mice, such suppression was not seen in Opn4aDTA/aDTA mice. Rather, Opn4aDTA/aDTA mice in the LL groups displayed similar progressive reductions in periodogram amplitude across conditions as seen in the DD groups. These observations indicate that the mechanisms normally mediating reductions in circadian amplitude under LL (like those mediating period-lengthening) are deficient in Opn4aDTA/aDTA mice, suggesting that classical photoreceptor signaling to the SCN, relayed via ipRGCs, may be critical for such effects.
Previous studies of Opn4aDTA/aDTA mice have not explored the effects of this mutation nor its possible interaction with lighting conditions on daily activity levels. In the DD groups, activity levels of WT mice showed little to no change across lighting conditions, while activity levels of Opn4aDTA/aDTA mice tended to progressively decrease. In contrast, activity levels were lower in LL than in either LD1 or LD2 for both LL/M and LL/F groups, although this was significant only in the males. In addition, Opn4aDTA/aDTA mice showed overall lower levels of activity across groups and lighting conditions than did WT mice, and a significant Lighting Condition × Genotype interaction was detected for the DD/F group. While these findings might indicate a role for ipRGCs in the regulation of activity levels, this effect should be interpreted cautiously and will require replication, given that it was impossible to employ littermate controls in the present experiments.
Since mice had access to 10% v/v ethanol under all employed lighting conditions (Sequence 1: LD1/DD/LD2 or Sequence 2: LD1/LL/LD2), it is reasonable to assume that ethanol intake might affect activity and circadian parameters. Interestingly, however, previous work from our laboratory has observed little to no effect of free-choice ethanol access on activity and circadian parameters measured during these particular lighting condition sequences. Specifically, Rosenwasser and Fixaris (2013) utilized a parallel set of water-only controls and found that although free-choice access to 10% v/v ethanol altered daily activity waveforms (only under steady-state LD entrainment), there were no effects on any other parameters measured (total daily wheel-turns, free-running period, and periodogram amplitude). Therefore, due to these previous findings, and the fact that implementing such controls would have doubled the required number of animals in the study, we did not explicitly control for possible effects of ethanol access on activity and circadian parameters.
Fluid Intake
The present results confirm our previous findings that ethanol intake is reduced similarly in either DD or LL conditions, relative to typical LD cycles (Rosenwasser and Fixaris, 2013; Rosenwasser et al., 2020). Furthermore, we show here that neither melanopsin-dependent photoreception nor ipRGC cellular integrity contribute to this effect. Despite the dramatic loss of circadian photosensitivity seen in Opn4aDTA/aDTA mice, neither main effects of genotype nor Genotype × Lighting Condition effects on ethanol intake were detected, indicating that the effects of lighting conditions on ethanol intake were quantitatively indistinguishable between WT and Opn4aDTA/aDTA mice. While the degeneration of melanopsin-expressing ipRGCs in Opn4aDTA/aDTA mice is thought to be incomplete at the ages tested here (Güler et al., 2008), cellular degeneration was clearly sufficient to completely block the circadian period-lengthening effects of LL and, in most animals, dramatically disrupt circadian entrainment. Although Opn4aDTA/aDTA mice are unable to synthesize melanopsin at any age, lack of melanopsin alone cannot fully account for the loss of circadian photosensitivity seen here, since previous studies have shown much less dramatic disruptions of entrainment and free-running periodicity in melanopsin knockout (Opn4-/-) mice (Panda et al., 2002; Ruby et al., 2002). Thus, we can conclude that photic effects on voluntary ethanol intake are mediated via photic pathways that are entirely distinct from those regulating the SCN circadian pacemaker. Presumably, these effects are mediated by components of the classical image-forming visual system, although further research would be needed to identify the critical photoreceptors and pathways.
Studies of voluntary, free-choice ethanol drinking generally monitor both ethanol and water intake to compute preference ratios, and assess whether experimental effects are selective for ethanol per se. Nevertheless, interpretation of ethanol preference is complicated by the fact that several factors can selectively affect water intake, thus produce misleading values for ethanol preference (Crabbe, 2014). In our previous studies (Rosenwasser and Fixaris, 2013; Rosenwasser et al., 2020), ethanol preference showed similar, but less robust, changes in response to altered lighting regimen than were seen for ethanol intake, while water intake was largely unchanged. Similarly, in the present experiments, consistent lighting effects on ethanol intake were seen in all groups, while water intake and ethanol preference remained rather stable across conditions. Thus, we conclude that ethanol consumption was selectively reduced under DD and LL conditions in both WT and Opn4aDTA/aDTA mice.
While this study was not specifically focused on sex differences, certain sex-related effects were apparent. Thus, as has been almost universally observed in previous mouse studies (Belknap et al., 1993; Yoneyama et al., 2008), females consumed more ethanol and less water, and showed higher ethanol preference than males, across genotype and lighting conditions. Nevertheless, careful examination of the fluid intake data suggests that these sex differences were more robust in WT than in Opn4aDTA/aDTA mice. To further examine this, we performed exploratory Sex × Genotype ANOVAs on all three fluid intake parameters, which showed significant interaction effects on water intake in both DD and LL groups. Further studies would be necessary to confirm the existence of sex-dependent behavioral effects of ipRGC ablation.
Several apparent sex differences were also noted in the circadian activity data. For example, females of both genotypes tended to display higher activity levels and periodogram amplitudes than males, especially in DD groups, while WT females showed somewhat shorter periods in LL than Opn4aDTA/aDTA females. Furthermore, Opn4aDTA/aDTA males were somewhat more likely than females to display abnormal LD entrainment. While sex differences in circadian activity parameters have been previously noted in the literature (Krizo and Mintz, 2014; Bailey and Silver, 2014), such effects tend to be small and inconsistent, and will not be discussed further here.
Conclusion
Taken together, these results indicate that reductions in voluntary ethanol intake seen under both DD and LL relative to LD entrainment appear to be independent of either melanopsin-dependent photoreception or the cellular integrity of ipRGCs. Indeed, the present results also support our earlier suggestion that such effects are unrelated to the period and/or integrity of circadian pacemaker function. A large number of recent studies have shown alterations in affective and/or addiction-related behaviors in animals maintained under atypical lighting regimens, including DD and LL, long and short photoperiods, non-24-h LD cycles, and a variety of so-called “light-at-night” protocols. While it is commonly assumed that such effects are mediated by circadian disruption and/or the melanopsin/ipRGC signaling pathways, direct evidence for this conclusion is actually rather sparse. To further complicate the issue, a recent study of animals maintained on an ultradian (3.5:3.5 h) LD cycle has revealed that mood and cognitive deficits are mediated by distinct subpopulations of ipRGCs with different central projections (Fernandez et al., 2018). Thus, the cognitive, but not the mood deficits seen in this model appear to be mediated by SCN-projecting ipRGCs. The present results serve as a reminder that much remains to be learned about the retinal and central pathways underlying the wide range of photo-behavioral phenomena.
