Abstract
Jet lag is a circadian disruption that affects millions of people, resulting, among other things, in extreme sleepiness and memory loss. The hazardous implications of such effects are evident in situations in which focus and attention are required. Remarkably, there is a limited understanding of how jet lag recovery and associated memory loss vary year round under different photoperiods. Here we show, using different cycles representing winter, summer, and equinox in male mice, that jet lag recovery and memory vary significantly with photoperiod changes. We uncover a positive correlation of acute light effects on circadian-driven locomotion (known as negative masking) with photoentrainment speed and memory enhancement during jet lag. Specifically, we show that enhancing or reducing negative masking is correlated with better or worse memory performance, respectively. This study indicates that in addition to timed-light exposure for phase shifting, the negative masking response could also be biologically relevant when designing effective treatments of jet lag.
Each year, millions of people who board flights that cross multiple time zones are subjected to jet lag, a misalignment of the internal circadian rhythm with the external day-night cycle. For many who travel for business or work, including airline pilots, being alert is paramount to their performance; jet lag increases daytime sleepiness and diminishes memory and executive functions (Weingarten and Collop, 2013; Barner et al., 2019). Jet lag experiments in rats and hamsters demonstrated poor spatial memory and inhibition of hippocampal neurogenesis (Devan et al., 2001; Gibson et al., 2010). These studies provide crucial information on how jet lag affects memory formation; however, because the experiments were conducted in only one light cycle (12 h:12h light-dark [12:12 LD]) with 1 kind of phase shift (phase advance), more studies are necessary to further understand how memory is affected during jet lag under different photoperiods.
Jet lag treatments aim to speed up recovery by administering sleeping aids, such as Ambien, Lunesta, or melatonin, as well as exposing individuals to light pulses before departure (Arendt, 2018; Bin et al., 2018; Morgenthaler et al., 2007). These remedies are based on studies using the phase response curve, which was established in constant darkness (DD), or jet lag data usually from one type of photoperiod with either an eastward or westward shift in the LD environment (Arendt, 2018; Crowley and Eastman, 2015; Gooley, 2017; Khalsa et al., 2003; Cho et al., 2015; Kori et al., 2017; Kiessling et al., 2010; Sack et al., 2007; Yamaguchi et al., 2013). Seasonal changes in photoperiod can negatively affect physical and mental health and cause desynchrony of metabolic processes such as melatonin release (Arendt and Middleton, 2018; Arendt, 2012; Chen et al., 2016). Therefore, to develop effective jet lag therapies, multiple photoperiods must be analyzed.
In addition to the changes in the LD environment experienced with jet lag, acute light exposure at specific times can have an added impact on behavior. Light directly regulates behavior through what is known as negative masking, which is the suppressive effect of light on activity in nocturnal animals. This effect is thought to be independent of circadian control (Mrosovsky, 1999). During jet lag, travelers often experience daytime sleepiness and nighttime wakefulness while the circadian clock realigns to the destination time zone. Suppression of daytime sleepiness and nighttime wakefulness, by light or other means, would be useful for efficient adaptation following transmeridian travel. It is still unclear whether the direct effects of light can positively affect circadian photoentrainment and memory performance during jet lag.
Here, we discuss the impact of negative masking (here on referred to as “masking”) on circadian entrainment and memory in a jet lag paradigm under three photoperiods: long day 16:8 LD, the commonly used 12:12 LD, and short day 8:16 LD cycles. We find that recovery from jet lag and memory varies across different photoperiods. Specifically, memory loss and the time to adjust to jet lag are significantly worse in 8:16 LD for the 6-h phase-advance condition compared with all other tested conditions. Adjusting the light-dark environment to allow induction of a negative masking response substantially improves memory deficits.
Methods
Mice
Male C57BL/6 NCrl mice were used for our experiments. The age-matched male mice were used between 4 and 8 months old. Historically, only males are used in circadian rhythm behavioral studies because female mice have a 4- to 5-day estrous cycle, in which the variations in hormones can affect the outcome of circadian behavioral studies (Jud et al., 2005). Another study showed that while locomotor activity was similar between males and females at some stages of the estrous cycle, there were also differences at other stages of the estrous cycle (Datta et al., 2016). For these reasons, we decided to focus on behavior in male mice in the current study and reserve the exploration of behavior in female mice in the context of the estrous cycle for future experiments. Mice were housed and treated in accordance with National Institutes of Health and institutional animal care and use committee guidelines and used protocols approved by the Johns Hopkins University and Oakwood University Animal Care and Use Committees. Six to 10 mice were used per experimental group.
Wheel-Running Activity
Mice were placed in cages with a 4.5-inch running wheel, and their activity was monitored with VitalView software (MiniMitter). Analyses of wheel-running activity were calculated with ClockLab (Actimetrics, Wilmette, IL). We used 500-lux light intensity (moderately bright-office lighting) for all wheel-running experiments. Mice were bred in 12:12 LD and maintained in 12:12 LD cycle for at least 2 weeks before any experiments. Naïve mice were used for each photoperiod. For each photoperiod, the same mice were used for the pre–jet lag, 6-h phase advance, and 6-h phase delay experiments.
Entrainment
The number of days to entrain to the jet lag paradigm was calculated using ClockLab (Actimetrics), to measure the onset of activity for the 6-h phase advance and offset of activity for the 6-h phase delay. The average of onset or offset was used to determine the first day of stable circadian (24-h period) entrainment following jet lag.
Transition between Light Cycles in the Jet Lag Paradigm
The approach to transitioning to jet lag was aimed at providing the least disruptive method to circadian-driven wheel-running activity. During the transition day for the 6-h phase delay and the 6-h phase advance, there was either an additional 6 h of darkness added or the light period was shortened by 6 h to compensate for the initial adjustment in day length on the first day in jet lag. In both cases, the additional darkness or shortened day did not overlap with the time of wheel-running activity in the photoperiod prior to the jet lag. The experiments were designed only to allow changes in the timing of light exposure during one time of day as it related to activity prior to jet lag (dashed black boxes in Fig. 2).
The addition or removal of light from the LD cycle during jet lag or experiments in Figure 3 caused a temporary change of 6 h in day and night ratio for each specified photoperiod during the transition day. The adjustment in the amount of light and dark on the jet lag transition day is a natural consequence of transmeridian travel. This temporary ~1-day LD ratio disruption should not be considered a photoperiod change because the effect of a photoperiod on circadian activity is a multiday effect that is measured each day by the circadian system.
6-h Phase Delay
To account for the difference in time experienced when delaying the LD cycle during the jet lag paradigm, the last night of the previous LD cycle was lengthened (from 8 h to 14 h in 16:8 LD, from 12 h to 18 h in 12:12 LD and from 16 h to 22 h in 8:16 LD). The first new light exposure during jet lag, 6 h of light, is outlined by dashed black boxes (Fig. 2).
6-h Phase Advance
To account for the difference in time experienced when advancing the LD cycle during the jet lag paradigm, the last day of the previous LD cycle was shortened (from 16 h to 10 h in 16:8 LD, from 12 h to 6 h in 12:12 LD, and from 8 h to 2 h in 8:16 LD). The first new light exposure during jet lag, 6 h of light, is outlined by dashed black boxes (Fig. 2).
Memory Tests
Mice used in the memory tests were different sets of mice than those used for the wheel-running experiments. All mice were habituated for 5 min at ZT 11 to the arena before memory tests to increase their familiarity with the area, void of objects, and the experimental room. Experiments for each photoperiod were conducted on different sets of mice. Different sets of mice were also used for each condition in both the object placement test and the novel object recognition test. Data were recorded with an aerially placed camcorder and visually analyzed, blind to the experimental condition. All experiments, before jet lag and during jet lag, were conducted in a dimly lit room of approximately 100 lux at the position of the mice in the arena. All mice were exposed to light for 10 to 15 minutes during the experiment, which likely did not confound results since immediate early genes, such as
Object Placement Test
The object placement test’s premise is to determine if mice will remember where an object was previously placed and spend a significant amount of time searching for the object, even though the object is no longer there. The test was conducted between ZT14 and ZT15 on the first full day (night 2) in the new light cycle for the jet lag paradigm and after at least 1 week of entrainment in the pre–jet lag condition. The pretest and the test period were both 5 min, with an intermediate 1-min rest period. Memory in the object placement test was analyzed using the following formula: % time in quadrant 2 (original location of object − object now absent) − % time in quadrant 4 (new location of object − object now present) (Discrimination Index − positive value = more time in the object-removed quadrant 2 = good memory). There were 2 control quadrants: 1 with no object ever and 1 with a stationary object. Mice behaved similarly in the control quadrants across photoperiods and experiments. To score a mouse as having entered into a quadrant, at least half the body length of the mouse had to be located in the quadrant. The protocol was modified from procedures assessing spatial memory in mice (Vogel-Ciernia and Wood, 2014; Denninger et al., 2018; Lesburguères et al., 2017).
Novel Object Recognition Test
The premise of the novel object recognition test is to determine if mice will remember an old/familiar object, lose interest in the old/familiar object, and spend more time with a novel object due to their innate curiosity. The test was conducted between ZT14 and ZT15 on the first full day (night 2) in the new light cycle for the jet lag paradigm and after at least 1 week of entrainment in the pre–jet lag condition. The pretest and the test period were both 5 min, with an intermediate 30-min rest period before exposure to the novel object. Memory in the novel object recognition test was analyzed using the following formula: % time with novel object − % time with old/familiar object during the second 5 min of the test (Discrimination Index − positive value = more time with the novel object = good memory). Mice exhibited a similar preference to objects in the pretest period across photoperiods and experiments. We considered interactions with the objects for the novel object recognition test if mice were less than half their body length from the object for 2 sec or longer. This protocol was adapted and modified (Bevins and Besheer, 2006; Denninger et al., 2018; Lueptow, 2017).
Activity before and after Masking
This is the potential for masking responses based on circadian control and activity output. During the 6-h period of the night that mice would be exposed to light following the 6-h phase delay and 6-h phase advance during the jet lag paradigm, we averaged the activity over 6 days and divided it by the total activity during the same 6 days, to determine the percentage of nightly activity that would be subject to masking (white boxes in Fig. 1). To assess masking during jet lag, we compared the amount of activity during the 6-h light period on the first day of jet lag (dashed black boxes in Fig. 1) to the activity prior to jet lag during the same 6-h window (white boxes in Fig. 1).
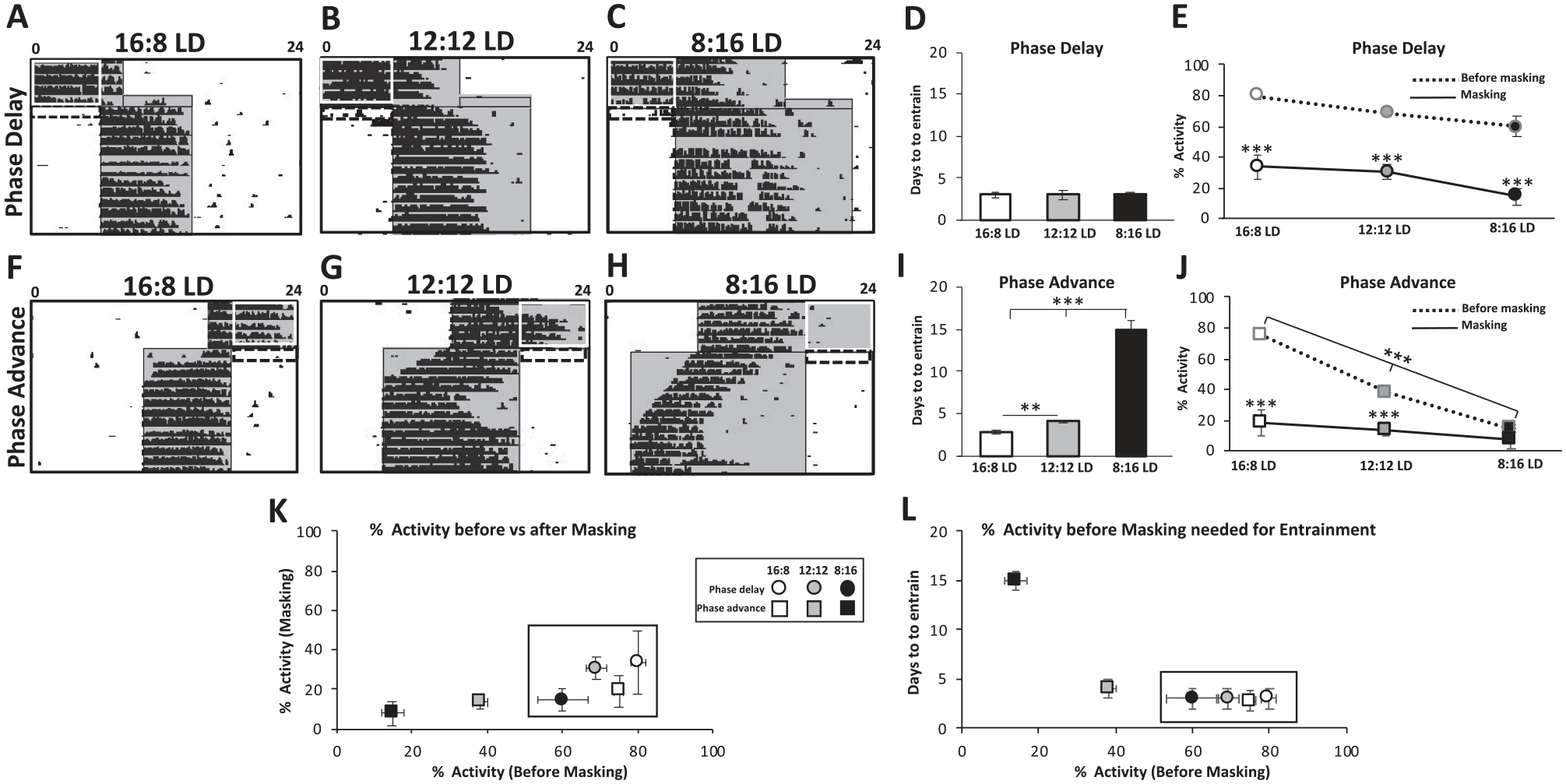
Jet lag recovery depends on photoperiod and masking. (A-C) 6-h phase delay Jet lag during 16:8 LD, 12:12 LD, and 8:16 LD photoperiods, respectively. Actograms depict wheel running activity (A-C, F-H). Shaded gray boxes outlined in black is the dark period of the day. Actograms depict a range of 17-22 days plotted on the Y-axis. Dashed black boxes- first-day jet lag masking period. White boxes- jet lag activity to suppress period. (D) Days to entrain in the 6-h phase delay jet lag during 16:8 LD, 12:12 LD, and 8:16 LD photoperiods. F(2,13) = 2.97, p = 0.09. (E) Percent activity before and during jet lag masking period during 16:8 LD, 12:12 LD, and 8:16 LD photoperiods. (F-H) 6-h phase advance jet lag during 16:8 LD, 12:12 LD, and 8:16 LD photoperiods, respectively. (I) Days to entrain in the 6-h phase advance jet lag during 16:8 LD, 12:12 LD, and 8:16 LD photoperiods. F(2,20) = 90.74, p = 1.22 × 10-9. (J) Percent activity before and during jet lag masking period during 16:8 LD, 12:12 LD, and 8:16 LD photoperiods. (K) Percent activity before versus after masking in jet lag. (L) Percent activity before versus days to entrain in jet lag. Black rectangle (K & L): inside values are similar and statistically different from outside values. Data are represented as mean ± SEM. **P-value <0.01 and ***P-value <0.001. Color version of the figure is available online.
Statistical Analysis
We performed statistical analyses independently for each photoperiod when comparing pre–jet lag to jet lag. We statistically compared mice during the pre–jet lag conditions for memory at ZT14 because they were photoentrained in each photoperiod. One-way analysis of variance was used for statistical analysis. When comparing all 3 photoperiods with 1 independent variable or 3 conditions in 1 photoperiod, we used the Tukey–Kramer posttest to determine statistical significance between groups. Six to 10 mice were used per experimental group. Significance was determined by a
Results
Acute Light on Activity Is Necessary for Photoentrainment after Jet Lag
We placed mice in 16:8 LD, 12:12 LD, and 8:16 LD cycles and subjected them to a jet lag paradigm, a 6-h phase delay, and 6-h phase advance to determine the effects of photoperiod on photoentrainment (Fig. 1 A-C, F-H). Entrainment was measured using wheel-running activity. There was no difference in the amount of time to entrain to the 6-h phase delay jet lag paradigm among all photoperiods (Fig. 1D; ~4 days to entrain). However, we observed a remarkable slowness in the ability of 8:16 LD maintained mice to entrain to a 6-h phase advance compared with both 12:12 LD and 16:8 LD cycles (Fig. 1I; ~15 days). This shows for the first time that photoperiod has a major effect on the speed of photoentrainment.
Negative masking was studied predominantly during the phase delay aspect of a 6-h shift (Fig. 1A-C, dashed black boxes; Fig. 1E). However, upon careful inspection of the actograms, it became apparent that masking can also be observed in the 6-h phase advance treatment (Fig. 1F-H, dashed black boxes; Fig. 1J), where it is most apparent in the 16:8 LD cycle at the end of the dark on the first day after jet lag (Fig. 1F, dashed black box; Fig. 1J). Interestingly, the only cycle that lacked this masking component, as well as sufficient activity before jet lag for masking (Fig. 1H, white box), was the 8:16 LD 6-h phase advance jet lag (Fig. 1J). Thus, we generated the hypothesis that masking of available activity on the first day of jet lag speeds up circadian photoentrainment. We found that if 60% to 80% of activity during the night is available for masking during jet lag (white boxes in Fig. 1), then masking as well as rapid jet lag recovery, as measured by wheel-running activity, occur (Fig. 1K, L, black boxes). This finding suggests a novel role for negative masking in circadian photoentrainment. Another possibility is that without negative masking, rapid phase shifting necessary for jet lag recovery is absent in the 8:16 LD 6-h phase advance cycle.
Memory Is Impaired When Masking Does Not Occur during Jet Lag
Changes in spatial and recognition memory are associated with the speed of photoentrainment. We employed an object placement test to assess spatial learning and novel object recognition test to assess recognition memory (Suppl. Fig. S1) as a metric to determine the jet lag effects under different photoperiods. We chose to perform these tests at zeitgeber time 14 (ZT 14), a time of high activity and alertness in mice (Pendergast and Yamazaki, 2011). Prior to jet lag, spatial and recognition memory are not different across photoperiods (Fig. 2A-C). Experiments were conducted on the second day of jet lag after either a 6-h phase delay or advance in the different photoperiods (Fig. 2D, G). Spatial and recognition memory were not altered during the 6-h phase delay across all photoperiods (Fig. 2E, F). However, both memories were reduced during the 6-h phase advance, particularly in 8:16 LD (Fig. 2H, I). This indicates that the speed of recovery from jet lag, as measured by wheel-running activity and memory performance, is correlated with photoperiod and the availability of sufficient activity for masking.
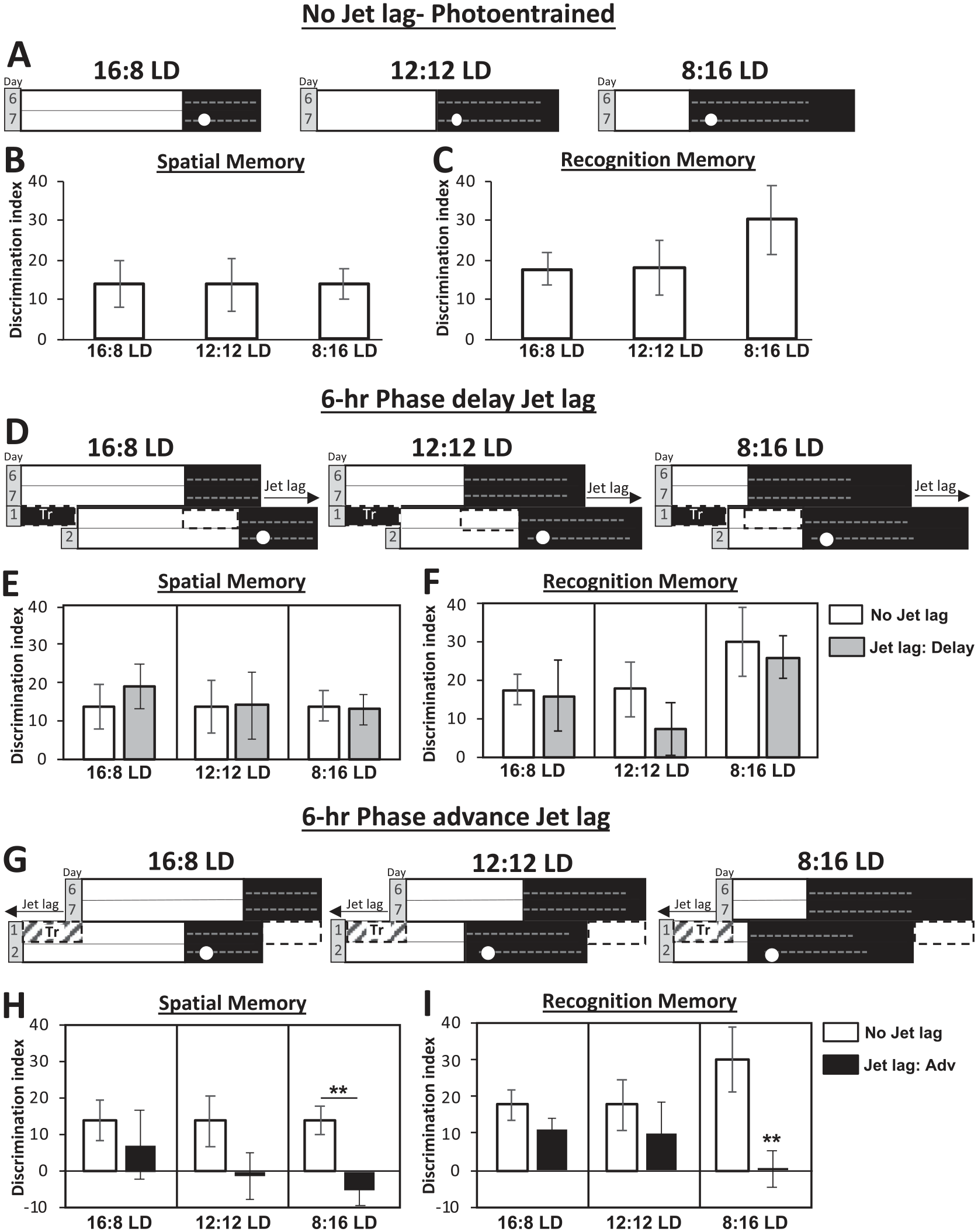
Spatial and recognition memory are impaired in 8:16 LD 6-h phase advance jet lag where masking is absent. (A) Schematic of 16:8 LD, 12:12 LD, and 8:16 LD photoperiods. Experiments conducted on day 7 in each photoperiod (All were previously in 12:12 LD, shifted by change in the time of lights on) at ZT 14 (white circle). Dashed lines represent wheel running activity and activity distribution across the night. (B) Object Placement test. Positive value for the discrimination index indicates good memory of the location of the missing object compared to its new location. F(2,20) = 0.06, p = 0.94. (C) Novel Object Recognition test. Positive value for the discrimination index indicates good memory by more time spent with the new object compared to the old object. F(2,19) = 0.94, p = 0.41. (D) Schematic of 6-h phase delay jet lag in 16:8 LD, 12:12 LD, and 8:16 LD. Experiments conducted on day 2 of jet lag in each photoperiod at ZT 14 (white circle). Dashed black boxes- indicate masking period during the first day of jet lag. Tr-extended 6-h of dark on the transition day. (E) Spatial memory during 6-h phase delay jet lag as measured by the object placement test. Negative values in the discrimination index indicate preference for new location of the object. F-values: 16:8 LD, F(1,10) = 0.2, p = 0.66; 12:12 LD, F(1,11) = 0.002, p = 0.97; 8:16 LD, F(1,10) = 0.06, p = 0.81. (F) Recognition memory during 6-h phase delay jet lag as measured by the novel object recognition test. F-values: 16:8 LD, F(1,10) = 0.03, p = 0.87; 12:12 LD, F(1,9) = 1.2, p = 0.29; 8:16 LD, F(1,12) = 0.14, p = 0.71. (G) Schematic of 6-h phase advance jet lag in different photoperiods- 16:8 LD, 12:12 LD, 8:16 LD. Tr-truncated 6-h of light, which allowed earlier onset of dark on the transition day. (H) Spatial memory during 6-h phase advance jet lag as measured by the object placement test. F-values: 16:8 LD, F(1,9) = 1.9, p = 0.2; 12:12 LD, F(1,7) = 1.5, p = 0.26; 8:16 LD, F(1,15) = 6.7, p = 0.01. (I) Recognition memory during 6-h phase advance jet lag as measured by the novel object recognition test. F-values: 16:8 LD, F(1,13) = 1.53, p = 0.23; 12:12 LD, F(1,16) = 0.52, p = 0.48; 8:16 LD, F(1,9) = 10.14, p = 0.01. Data are represented as mean ± SEM. **P-value <0.01. Color version of the figure is available online.
Enhancing or Reducing Masking Correlates with Memory Performance
To directly test whether masking affects memory formation, we either increased the masking response by giving a light pulse (Fig. 3A) or decreased the masking response by prolonging darkness (Fig. 3F). We found a positive correlation between masking and memory. Our observation that entrainment and memory are normal when there is 60% to 80% of activity available for masking during jet lag led us to the hypothesis that exposing mice in 8:16 LD 6-h phase advance to a 10-h light pulse in the latter portion of the night will lead to an enhanced masking response in 8:16 LD as well as normal memory (Fig. 3A). The 10-h light pulse, aimed at suppressing up to 80% of activity, induced a significant masking response during 8:16 LD 6-h phase advance jet lag (Fig. 3B, C). Furthermore, both spatial and recognition memory were improved to the pre–jet lag levels in 8:16 LD 6-h phase advance (Fig. 3D and E; Fig. 3I and J, solid horizontal line [pre-jet lag] and black triangle data point). Based on our observations that masking and entrainment during the jet lag paradigm were normal in 16:8 LD and 12:12 LD 6-h phase advance, we tested the hypothesis that replacing light with darkness during jet lag would reduce masking and result in poorer memory (Fig. 3F). We found that both spatial and recognition memory were negatively affected by the lack of light to drive a masking response (Fig. 3G and H; Fig. 3I and J, solid horizontal line [pre-jet lag] and white and gray triangle data points). These findings indicate that memory functions during jet lag are correlated with negative masking.
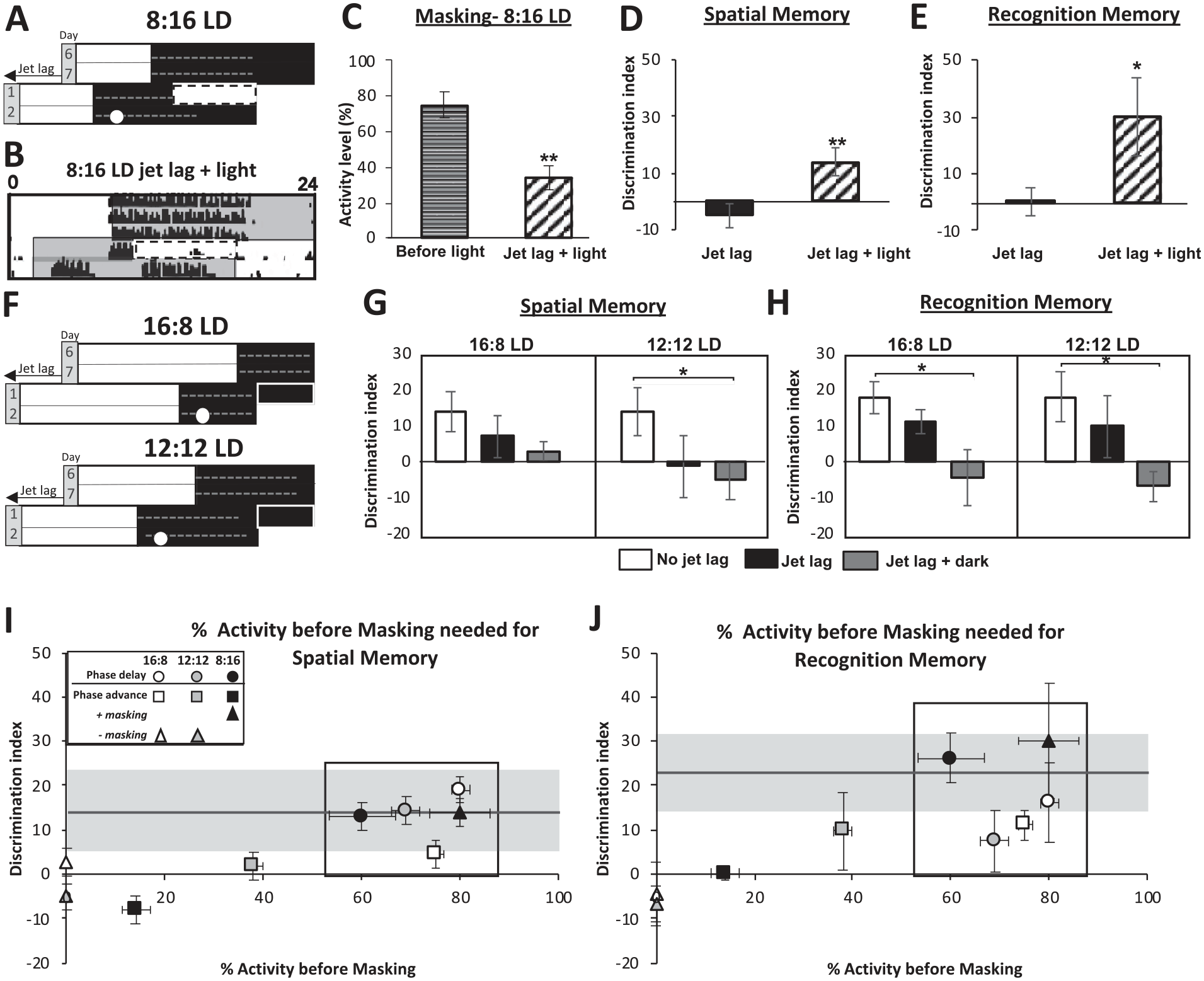
Permitting or blocking masking on the first night of jet lag improves or worsens spatial and recognition memory, respectively. (A) Schematic of 6-h phase advance jet lag in 8:16 LD with added light period during the latter 10 hrs of the 16-h night (black triangle). Experiments conducted on day 2 of jet lag at ZT 14 (white circle). (B) Actogram of 6-h phase advance jet lag with 10-h light exposure (black triangle). (C) Quantification of masking (% activity) with the 10-h light exposure in 8:16 LD jet lag compared to the day before during the same time as the 10-h light exposure. F-value (1,23) = 16.8, p = 0.01. (D) Spatial memory as measured by the object placement test. F(1,16) = 7.21, p = 0.01. (E) Recognition memory as measured by the novel object recognition test. F(1,10) = 5.22, p = 0.04. (F) Schematic of 6-h phase advance jet lag in 16:8 LD and 12:12 LD with added darkness during the masking period (white and gray triangles). Experiments conducted on day 2 of jet lag in each photoperiod at ZT 14 (white circle). (G) Spatial memory as measured by the object placement test. F-values: 16:8 LD, F(2,17) = 1.5, p = 0.37; 12:12 LD, F(2,18) = 3.8, p = 0.05. (H) Recognition memory as measured by the novel object recognition test. F-values: 16:8 LD, F(2,20) = 4.12, p = 0.03; 12:12 LD, F(2,19) = 4.1, p = 0.03. (I) Spatial memory and (J) Recognition memory versus percent activity to mask during jet lag (phase advance and phase delay) and jet lag (phase advance) with or without masking. Horizontal solid gray line is the average memory performance before jet lag. Horizontal shaded gray area is the range of SEM for memory before jet lag. % Activity for masking (Black rectangle): inside values are similar and statistically different from outside values. Data are represented as mean ± SEM. *P-value <0.05 and **P-value <0.01. Color version of the figure is available online.
Discussion
Masking during Jet Lag Depends on Sufficient Available Activity during Each Photoperiod
Our study provides an advancement in the understanding of jet lag recovery in 3 different photoperiods: 16:8 LD, 12:12 LD, and 8:16 LD. We found that photoentrainment was most difficult for our mice during the 8:16 LD 6-h phase advance experiment but not during the 6-h phase delay or other photoperiods. A previous study showed that in long day (16:8 LD) photoperiods, a 4-h light pulse induces a larger phase advance than in the short day (8:16 LD) photoperiod (Ramkisoensing et al., 2014). However, in the short-day photoperiod experiment, there was very little activity available for masking. Based on our findings, we propose a longer light exposure of 6 to 10 h would induce a much larger phase shift in their short-day paradigm and result in a larger and similar phase shift to the long-day paradigm.
Although our study is not the first report on differences between jet lag recovery for phase advance or delay (Diekman and Bose, 2018; Boulos et al., 1995),we observed differences in negative masking and photoentrainment as it correlates with available light cues in different photoperiods, which has the most relevance for phase advances where activity tapers as the night progresses. In the 6-h phase advance experiment, the masking light exposure falls at different effective circadian times in each photoperiod. This is a natural result of the relationship between the photoperiod and the circadian-driven wheel-running activity distribution throughout the night. When we temporarily adjusted the LD cycle to allow light exposure to occur during the active phase, negative masking was induced and memory improved (Fig. 3A-E). Furthermore, strategically removing light from falling during the active phase during the 6-h phase advance resulted in no improvement as well as deficits in memory (Fig. 3G and H).
Inducing a Masking Response Correlates with Improvement in Memory Performance
We selected ZT14-15 as the time for memory experiments. Although the circadian time (CT) is likely different among the 3 photoperiods tested, during day 2 of jet lag, mice exhibit little wheel-running activity at ZT14 because they were not fully entrained. This allowed us to view ZT14 on jet lag day 2 as the earliest and best point during jet lag to compare memory across different photoperiods. To restrict our analyses to the ZT14-15 time point during jet lag, we analyzed short-term memory with the object placement and novel object recognition tests. In a study that was not as ZT restricted as our study, testing memory at 2 h or 24 h after the initial pretest demonstrated that a 20:4 LD cycle positively affected memory performance when compared with 12:12 LD (Dellapolla et al., 2017). Although we did not observe a difference in memory in our pre–jet lag analyses, this study supports our findings during jet lag that the least severe disruption to memory was observed in 16:8 LD compared with 8:16 LD.
Standardization for Jet Lag Light Therapy Is Needed
Improvement in memory when negative masking was induced during our 8:16 LD 6-h phase advance experiment is an encouraging indicator that a parallel process for light therapy may exist for improving jet lag symptoms in humans. While translating our results into a manner that is applicable for improving human health outcomes is beyond the scope of this study, current reports on the effectiveness of light therapy for jet lag recovery in humans are inconsistent. We believe this is due to the wide variety in the LD environments used for testing. For example, in an eastward shift study in which men were administered evening light therapy, entrainment to the new time zone did not occur, and early-morning sleepiness persisted (Boivin and James, 2002). Another study comparing 3 morning bright-light exposure patterns revealed that bright-light–advanced schedules did not result in alignment with a 3-h shifted sleep schedule but rather produced small phase advances that might have intensified jet lag symptoms if allowed to continue (Crowley and Eastman, 2015). Nevertheless, other studies indicate successful acclimatization to several conditions and different photoperiods due to light therapy, such as the study indicating that light therapy improves the sleep-wake cycle for seasonal and nonseasonal disorders (Wirz-Justice, 2018). Yet some studies conflict in the timing of light therapy, such as light therapy in the morning and evening (Kim et al., 2018), only the evening before and the day of departure (Choy and Salbu, 2011), or several cycles over 2 to 3 days (Czeisler et al., 1990). The varying protocols for light therapy that result in different outcomes, some of which were positive, still need to be standardized to yield effective outcomes for those suffering from circadian disruptions (Al-Karawi and Jubair, 2016; Chen et al., 2016). Our studies show the need for a reevaluation of when light therapy treatments are being administered as it relates to the exogenous photoperiod.
Environmental Considerations for the Use of Pharmaceuticals for Jet Lag
Although it may be beneficial to combine therapeutic approaches such as light treatment and pharmaceuticals, the photoperiod must at least be considered in predicting the effectiveness of such treatments. Research aimed at ameliorating the effects of jet lag–associated memory loss indicated that melatonin improved spatial memory loss and stimulated hippocampal neurogenesis experienced during jet lag during 12:12 LD. While this study suggests that melatonin is a good therapy option, the mice were exposed to extreme jet lag conditions of a 6-h phase advance every 3 days for 3 weeks (Iggena et al., 2017). In contrast, our study explores the impact of different photoperiods on memory while simulating jet lag with either one 6-h advance or delay shift in the light-dark cycle. Our findings indicate that an optimal range of 60% to 80% activity is needed for masking with light therapy during jet lag recovery. This is currently an unconsidered facet in the development of light therapy treatments. Future studies in humans could determine whether considering photoperiod, activity distribution, and light responsiveness would increase the effectiveness of pharmaceuticals used to aid in jet lag recovery.
Conclusion
This body of work demonstrates that the masking response is an indicator of normal jet lag recovery and memory response. The jet lag masking response varies with available light during different photoperiods and relies on the distribution of activity throughout the night. Evaluating the role of both masking and phase shifting in jet lag recovery will likely strengthen current jet lag therapies (Arendt, 2018; Crowley and Eastman, 2015; Gooley, 2017). While further studies would be necessary to determine how these findings would translate to humans, we are hopeful that this body of work leads to more fine-tuned remedies for jet lag, based on the specific responses of the circadian clock to light in different LD environments. Our light therapy approach in mice was administered on one day to be as nonintrusive to scheduling as possible. We hope that future studies will determine the impact of light exposure during the first 24 h of jet lag in humans, which will hopefully lead to enhanced recovery and improved memory performance for travelers.
Supplemental Material
Richardson_Supplemental_Figure_June_2020_Revised – Supplemental material for Jet Lag Recovery and Memory Functions Are Correlated with Direct Light Effects on Locomotion
Supplemental material, Richardson_Supplemental_Figure_June_2020_Revised for Jet Lag Recovery and Memory Functions Are Correlated with Direct Light Effects on Locomotion by Melissa E. S. Richardson, Samuel Parkins, Isabelle Kaneza and Amy-Claire Dauphin in Journal of Biological Rhythms
Footnotes
Acknowledgements
Dr. Samer Hattar, PhD, was instrumental in allowing the authors to use his lab equipment and resources to conduct some of the experiments for this study at Johns Hopkins University and revise the article. Gregory Darville and Ashleigh Moten are former students of Oakwood University who contributed to this study by assisting in establishing the object placement test’s behavioral apparatus. The memory analyses in this study were conducted at Oakwood University and monetarily supported by the IMARI grant and departmental funds from the Department of Biological Sciences. Funding source: NIH-GM076430 (Samer Hattar).
Author Contributions
M.E.S.R. conceived the study, performed experiments, and wrote the manuscript; S.P. performed experiments and wrote the manuscript; I.K. and A.C.D. established experiment protocols and wrote the manuscript.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
