Abstract
Schizophrenia is a multifactorial disorder caused by a combination of genetic variations and exposure to environmental insults. Sleep and circadian rhythm disturbances are a prominent and ubiquitous feature of many psychiatric disorders, including schizophrenia. There is growing interest in uncovering the mechanistic link between schizophrenia and circadian rhythms, which may directly affect disorder outcomes. In this review, we explore the interaction between schizophrenia and circadian rhythms from 2 complementary angles. First, we review evidence that sleep and circadian rhythm disturbances constitute a fundamental component of schizophrenia, as supported by both human studies and animal models with genetic mutations related to schizophrenia. Second, we discuss the idea that circadian rhythm disruption interacts with existing risk factors for schizophrenia to promote schizophrenia-relevant behavioral and neurobiological abnormalities. Understanding the mechanistic link between schizophrenia and circadian rhythms will have implications for mitigating risk to the disorder and informing the development of circadian-based therapies.
Schizophrenia is a severe and chronic neurodevelopmental disorder, with a lifetime prevalence of close to 1% (Jablensky, 2000; McGrath et al., 2008). It is typically diagnosed in early adulthood with characteristic positive symptoms (i.e., delusions, hallucinations), negative symptoms (i.e., affective, avolition), and impaired cognition (i.e., learning, attention; Tandon et al., 2013). Much attention has been given to the hypothesis that schizophrenia is a multifactorial disorder, triggered by a combination of risk factors including gene variants as well as environmental insults that one is exposed to in utero and during their lifetime (van Os et al., 2008; Ayhan et al., 2016; Stilo and Murray, 2019). Risk factors for schizophrenia are believed to have a small individual impact; most likely, multiple risk factors act synergistically along the life span to affect onset, symptoms, and course of the disorder. Thus, such risk factors should be studied as an integrated system (Jablensky, 2010). The complexity of the disorder is also reflected in the limited pharmacologic treatment options available. Although antipsychotic drugs, working principally on the dopamine system, are effective in controlling positive and some negative symptoms, they rarely improve cognitive and social dysfunctions that are better predictors of functional outcomes (Miyamoto et al., 2005; Kahn et al., 2015). Discovering more efficient therapeutic options may rely on exploring novel risk factors for schizophrenia and uncovering the complex mechanisms in which these risk factors interact to jointly exert their effect.
In the context of schizophrenia, a promising avenue of study is sleep and the circadian system. Indeed, disturbances in these systems are a prominent and ubiquitous feature of many psychiatric disorders, including schizophrenia (Wulff et al., 2010; Wulff et al., 2012; Jagannath et al., 2013; Monti et al., 2013; Kaskie et al., 2017; Logan and McClung, 2019). In a healthy individual, the sleep/wake cycle displays a characteristic 24-h periodicity and is the most documented output of the circadian (daily) system. Circadian clocks are cell autonomous, producing rhythms with a period of ~24 h, but they can also be entrained by external timing cues (zeitgebers), such as light. The proper alignment of circadian clocks to environmental cycles is essential for organisms to anticipate and prepare for local challenges, and it directly affects the fitness and longevity of many species (Evans and Davidson, 2013). In mammals, a “master” circadian clock is located in the hypothalamic suprachiasmatic nucleus (SCN), which receives photic information directly from the retina (Hastings et al., 2018). The SCN clock is formed by many single-cell circadian oscillators that synchronize with one another to generate coordinated circadian outputs (Hastings et al., 2018). Circadian clocks are also located in most other tissues and organs. These “peripheral” clocks can also function in an autonomous manner. However, they can be reset or synchronized by rhythmic cues that are directly or indirectly controlled by the SCN (Dibner et al., 2010; Hastings et al., 2018).
In this review, we explore the interaction between schizophrenia and circadian rhythms from 2 complementary angles. First, we assert that sleep and endogenous circadian rhythm disturbances are a fundamental component of schizophrenia. Second, we discuss the idea that environmental and genetic circadian rhythm disruption interacts with risk factors for schizophrenia to promote schizophrenia-relevant behavioral and neurobiological abnormalities.
Sleep and Circadian Disturbances Constitute a Fundamental Component of Schizophrenia
Sleep and Circadian Disturbances in Patients with Schizophrenia
Sleep disturbances in individuals with schizophrenia have been documented as early as the 19th century by Kraepelin et al. (1919), with “care for sleep” being a recommended treatment. In line with these observations, recent findings using more objective experimental tools have confirmed that various sleep and circadian disturbances appear highly prevalent in individuals with schizophrenia (Fig. 1). Indeed, up to 80% of individuals with schizophrenia exhibited difficulties with sleep (Cohrs, 2008), and sleep disturbances preceded prodromal psychotic symptoms in more than 77% of patients (Tan and Ang, 2001). Sleep disturbances in subjects with schizophrenia included reduced slow-wave sleep, reduced total sleep time and efficiency, and increased rapid eye movement sleep latency that persisted after accounting for medication, social isolation, and the absence of a social routine (Chouinard et al., 2004; Wulff and Joyce, 2011; Zanini et al., 2013). In individuals with schizophrenia, poorer sleep quality correlated with greater symptom severity (Korenic et al., 2019; Laskemoen et al., 2019), and patients with disturbed circadian rhythms performed worse in cognitive tests compared with patients with normal circadian rhythms (Bromundt et al., 2011). In a study measuring both actigraphy and melatonin rhythms, a substantial portion of individuals with schizophrenia had severe disruptions of the circadian sleep/wake timing. This included delayed and/or free-running sleep/wake cycles compared with matched healthy subjects (Wulff et al., 2012). A meta-analysis of sleep parameters accessed by actigraphy reported various sleep disturbances in patients with treated and remitted schizophrenia compared with healthy controls. These included greater total sleep time, longer time in bed, greater sleep latency, greater wake after sleep onset, and reduced motor activity (Meyer et al., 2020).
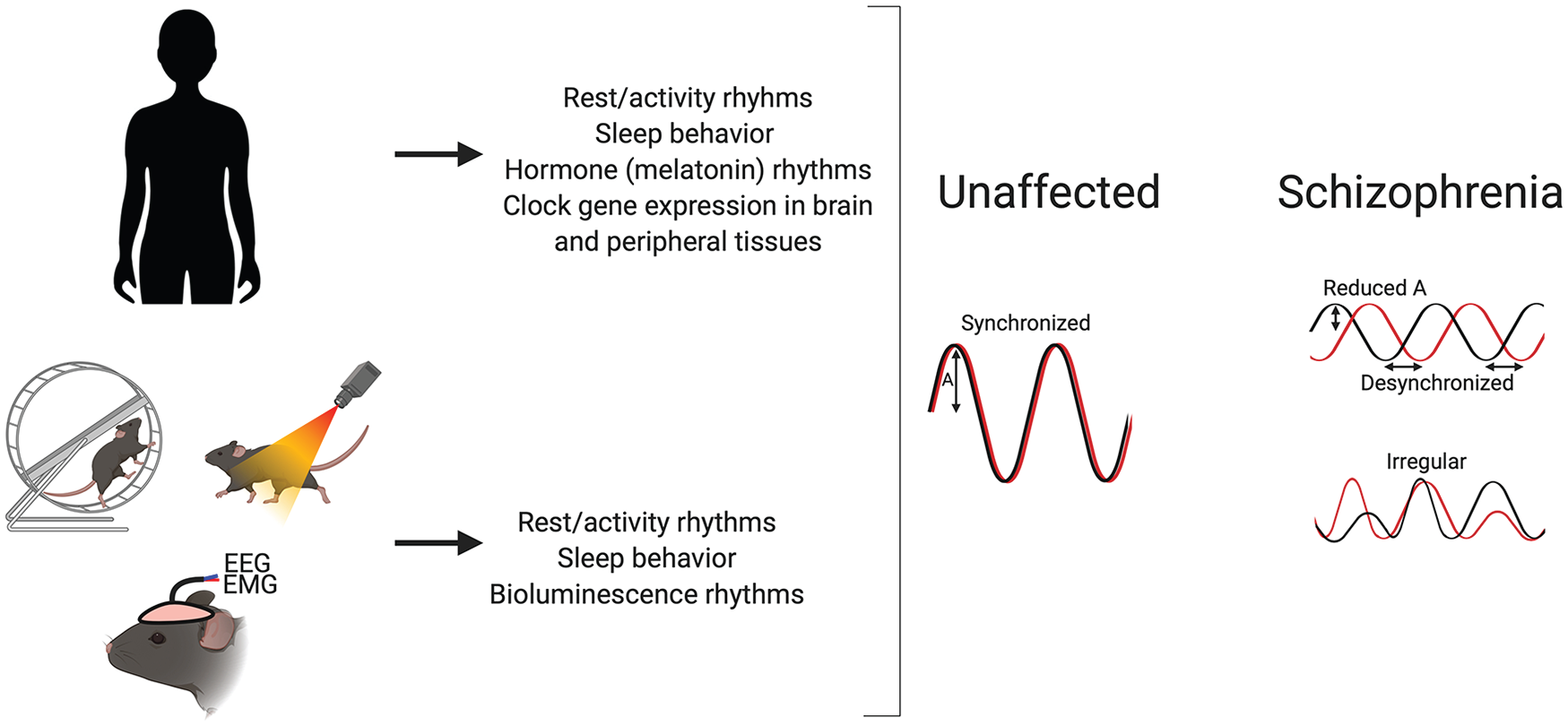
Sleep and circadian rhythm disturbances are comorbid with schizophrenia in human patients and related animal models. There is evidence to suggest that sleep and circadian rhythm disturbances constitute a fundamental component of schizophrenia, which we have reviewed in both human studies and animal models that contain genetic mutations related to schizophrenia. Overall, most studies suggest disrupted rhythms in various tissues and assays in schizophrenia patients and related animal models. This disruption is usually seen as loss of circadian rhythmicity, fragmented rhythms, a decreased amplitude (A), and/or irregularity of rhythms. Note that color version is available online.
Further, some reports have outlined an association between psychosis and sleep and circadian disturbances (Reeve et al., 2015; Freeman et al., 2017; Waite et al., 2019). This association was mainly studied in healthy participants; however, preliminary research showed that sleep disturbances are especially prevalent in clinical high risk (CHR) individuals for psychosis (Poe et al., 2017). Using actigraphy and sleep diaries, youth with CHR for psychosis had more fragmented diurnal rhythms and later onset of nocturnal rest than controls, and the level of these disruptions predicted psychosis symptom severity 1 year later (Lunsford-Avery et al., 2017). Using wrist actimetry, older individuals with schizophrenia were found to spend more time in bed, had more fragmented sleep, and were awake more during the day compared with older nonpsychiatric individuals (Martin et al., 2001; Martin et al., 2005). Of note, the sparsity of research exploring aging and sleep and circadian rhythms in individuals with schizophrenia likely speaks to the challenges in conducting longitudinal studies in patients. The clinical findings summarized above suggest that sleep and circadian disturbances are likely a characteristic feature of schizophrenia, potentially involved in the development of the disorder.
With respect to the molecular markers of the circadian clock, individuals with chronic schizophrenia were found to have a loss of rhythmic mRNA expression of the clock genes CRYPTOCHROME1 (CRY1) and PERIOD2 (PER2) in skin fibroblasts cultured ex vivo and subjected to a clock-synchronizing treatment (Johansson et al., 2016). In the same study, blood mononuclear cells from patients experiencing their first episode of psychosis had decreased expression of CRY1, PER2, and CLOCK compared with controls (Johansson et al., 2016). Further, using RNA sequencing, Seney and colleagues (2019) explored gene expression in the dorsolateral prefrontal cortex of postmortem brains of subjects with schizophrenia. Using time-of-death data from each subject, researchers found that the set of genes with daily rhythms in subjects with schizophrenia were very distinct from those identified in the dorsolateral prefrontal cortex in healthy controls and had a different rhythmic pattern (Seney et al., 2019). Distinct cellular pathways were enriched among genes of each of these rhythmic groups. Therefore, these kinds of studies might point toward the molecular underpinnings of circadian disturbances in schizophrenia.
Overall, while the current evidence highlights the prevalence of sleep and circadian disturbances in individuals with schizophrenia, further studies would benefit from measuring additional biological markers of the endogenous circadian clock and peripheral clocks in individuals with schizophrenia over an extended period of time.
Targeting Sleep and Circadian Disruptions for Therapeutic Intervention
In a clinical setting, sleep and circadian disruptions are seldom assessed or targeted directly for treatment in individuals with schizophrenia, despite “sleeping better” being a desired treatment preference (Freeman et al., 2019). Luthringer and colleagues (2007) were among the first to explore the effects of the antipsychotic treatment, paliperidone ER, on sleep parameters in patients with schizophrenia. Interestingly, they found that patients showed improved sleep architecture and continuity after paliperidone ER treatment (Luthringer et al., 2007). A recent study by Mishra and colleagues (2020) explored the sleep-promoting drug ramelteon, an MT1/MT2 melatonin receptor agonist, as an add-on treatment for schizophrenia patients. Compared with patients who received antipsychotics alone (haloperidol and risperidone), add-on ramelteon appeared to significantly improve sleep as well as the severity of positive and negative symptoms. This was accompanied by increased levels of serum melatonin and aryl-alkylamine-N-acetyl-transferase (the rhythmically expressed and rate-limiting enzyme in the melatonin synthesis pathway) and urinary melatonin (Mishra et al., 2020). Cognitive behavioral therapy for insomnia (CBT-I) is emerging as an effective sleep-promoting therapy for individuals with schizophrenia who experience insomnia. CBT-I was most effective in patients with “classic severe insomnia”, denoted by short sleep duration, very poor sleep efficiency, and prolonged sleep onset latency (Chiu et al., 2018), and the benefits on sleep in patients lasted 4 weeks after treatment (Hwang et al., 2019). In addition, in male patients admitted to a psychiatric ward, CBT-I, in combination with controlling light/dark exposure and discussion on sleep monitoring, led to larger reductions in self-reported insomnia symptoms and earlier discharge compared with standard care alone (Sheaves et al., 2018). Future studies should conduct a more detailed assessment of the role of different antipsychotics and sleep or circadian-promoting therapies or drugs on symptom severity in individuals with schizophrenia and related animal models. For example, one could hypothesize that promoting a proper phase relationship between the circadian system and environmental timing cues may improve schizophrenia-related symptoms by strengthening and stabilizing circadian rhythmicity.
The studies above demonstrate a high prevalence of sleep and circadian disturbances among patients with schizophrenia, which may highlight these disturbances as a core component of schizophrenia pathology. Despite this seemingly high prevalence, there is surprisingly little research targeting these disruptions for therapeutic intervention, leaving much to be discovered.
Animal Models to Study Sleep and Circadian Disturbances In Relation to Schizophrenia
A number of animal models exploring genetic risk factors for schizophrenia have been used in an attempt to reproduce the sleep and circadian disruptions seen in individuals with schizophrenia (Fig. 1; see Table 1 for an overview of the animal models related to schizophrenia that have been studied for sleep and circadian phenotypes). Overall, the studies provide insights on a link between schizophrenia risk genes in animal models and sleep and circadian rhythm–related phenotypes. Some of the genes mutated in the mouse models—or related genes (including GRM3, SNAP91, TSNARE1, and EGR1)—were highlighted in a multistage schizophrenia genome-wide association study (GWAS; Schizophrenia Working Group of the Psychiatric Genomics Consortium, 2014).
Overview of mouse models related to schizophrenia and their known sleep and circadian phenotypes.
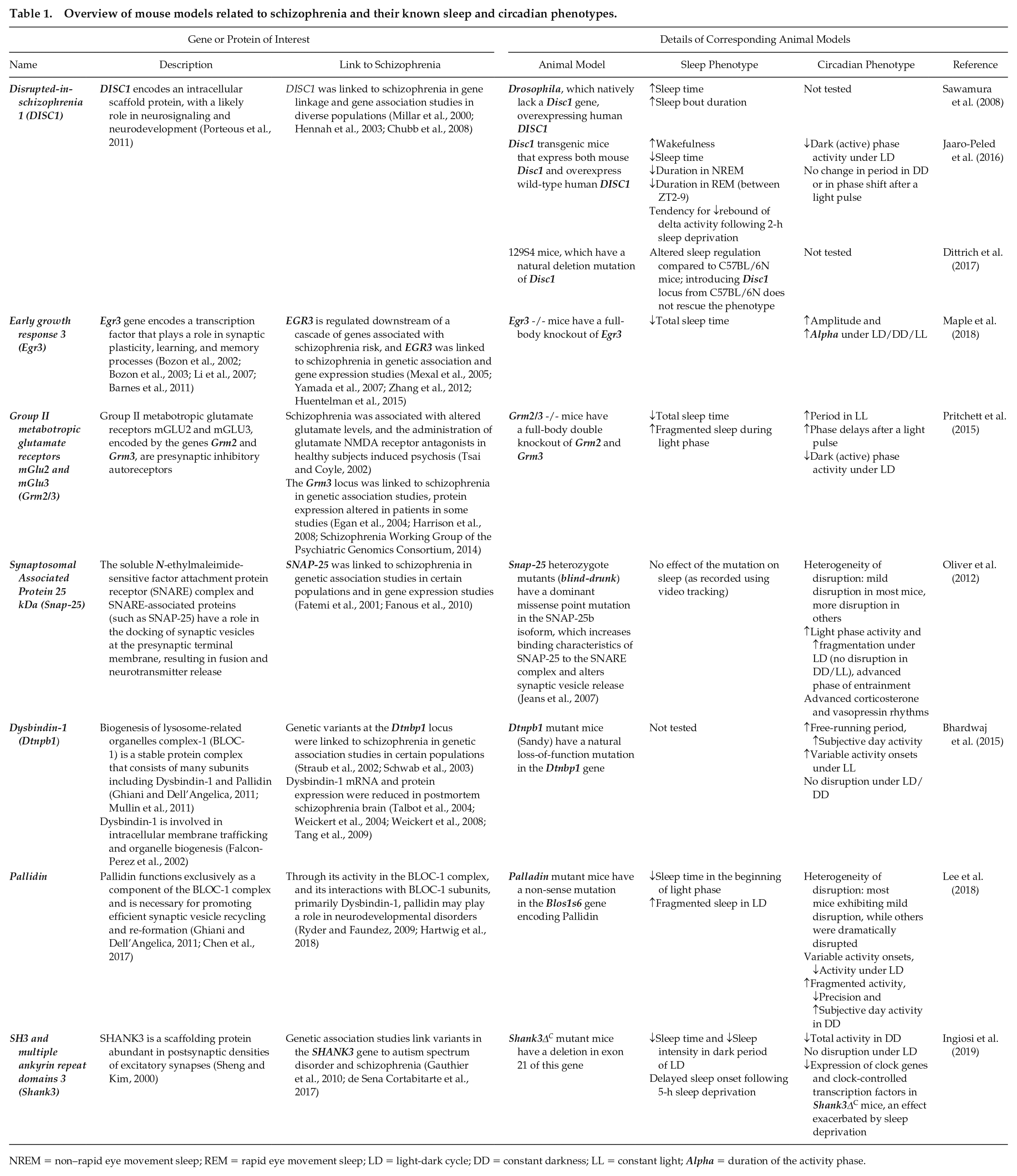
NREM = non–rapid eye movement sleep; REM = rapid eye movement sleep; LD = light-dark cycle; DD = constant darkness; LL = constant light; Alpha = duration of the activity phase.
Interestingly, most of the genes or proteins of interest in these mouse models have related cellular functions, namely, in neuronal communication and/or protein and vesicle trafficking. Indeed, disrupted synaptic connectivity and function and abnormal neurotransmitter release have been suggested as key factors in the pathogenesis of schizophrenia (Frankle et al., 2003; Harrison and Weinberger, 2005). Notably, SNAP-25 is a SNARE (soluble N-ethylmaleimide-sensitive factor attachment protein receptor) associated protein, with a role in the docking of synaptic vesicles at the presynaptic terminal membrane, resulting in fusion and neurotransmitter release (Mohrmann et al., 2010). Dysbindin-1 is a SNARE-modulating presynaptic protein, essential for synaptic homeostasis (Dickman and Davis, 2009). Dysbindin-1 has been reported to modulate Snap-25 expression through a possible interaction with Snapin (Feng et al., 2008), and it might regulate dopamine release in the central nervous system through its actions on the SNARE complex (Kumamoto et al., 2006). Dysbindin-1 and Pallidin also have related mechanisms; they are both subunits in the BLOC-1 (the biogenesis of lysosome-related organelles complex-1) stable protein complex. It was hypothesized that they exert their effects on phenotypes via affecting BLOC-1 stability (Lee et al., 2018).
Dysfunction of vesicle trafficking and synaptic communication may contribute to the pathogenesis of schizophrenia but also to the mechanism underlying the interaction between schizophrenia and sleep and circadian rhythms. The studies presented in Table 1 are mainly descriptive reports, and future studies should aim to uncover mechanistic links between schizophrenia and the sleep and circadian systems.
The Challenge of Modeling Schizophrenia in Animal Models
A limitation of studying risk factors for schizophrenia in animal models is the difficulty in faithfully recapitulating the etiological factors and behavioral phenotypes that are often heterogeneous and uniquely human. It is widely accepted that the pathogenesis of schizophrenia is explained by a complex interaction between multiple interacting genetic and environmental risk factors. Such risk factors are thought to act synergistically, affecting brain development early in life. Thus, the outcome is first seen in young adulthood. A limitation in the studies discussed in Table 1 is that most used only animal models with monogenic mutations, which likely have only a small impact on the pathology of schizophrenia. Studying interacting risk factors for schizophrenia as an integrated system would be more appropriate. However, this poses various challenges, including complicated experimental designs and data analyses.
Although no animal model can recapitulate the full intricacies of schizophrenia, they are nonetheless an essential hypothesis-testing preclinical tool. Animal models can be genetically or pharmacologically engineered. Moreover, they offer a high level of experimental control that is necessary when exploring mechanistic links and testing therapeutic targets. Therefore, such studies remain necessary but should be increasingly refined in an attempt to account for the complex nature of schizophrenia.
Distinguishing Sleep versus Circadian Rhythm Phenotypes
Sleep/wake cycles and circadian rhythms involve interconnected yet distinct biological mechanisms. However, it is difficult to disentangle these 2 phenomena, especially when attempting to measure sleep and circadian phenotypes in animal models. The sleep/wake cycle is one of the most documented outputs of the circadian system. Sleep regulation involves a circadian component, whereby the SCN circadian clock controls the activity of brain sleep- and wake-promoting centers (Saper et al., 2005). Sleep regulation also involves a homeostatic component, whereby the sleep pressure (need for sleep) experienced by an individual increases with extended wakefulness and dissipates with sleep (Borbely, 2009).
In a stable and predictable environment, the sleep/wake cycle is aligned with other circadian-regulated rhythms in the organism. However, a distinction between the two becomes apparent when either cycle is disrupted. The studies presented in Table 1 highlight the dichotomy between the sleep/wake cycle and circadian system, as several mouse models, including Egr3, DISC1, and Grm2/3, showed disruptions in one system and not in the other, perhaps shedding light on distinct biological mechanisms that lead to these disruptions.
Comparing Animal Models: Different Assays, Different Methods
Comparing the sleep and circadian phenotypes of various mouse models across studies is difficult because of the different assays used and nonstandardized experimental protocols for data collection and analysis. First, measuring sleep stages in rodent models can be done using electroencephalography and electromyography, or general sleep can be measured less invasively through motion detection, whereby sleep is defined by a bout of immobility. Measuring circadian rhythms in rodent models can also be done in multiple ways, such as through wheel-running assessment, bioluminescence recordings, and measuring other markers of the circadian clock such as body temperature and clock gene expression. Thus, the observed effects depend on the assays chosen, which need to be considered when making comparisons between studies (Pritchett et al., 2012).
Second, it is important to standardize experimental conditions, such as wavelength composition and intensity of light and the type and order of light challenges, and data analysis measures between studies. For example, light challenges can be tested in the following order: first, a standard lighting condition of 12 h of light and 12 h of dark (LD), then proceed to phase shifts (delay and/or advance) and constant conditions (constant darkness [DD], constant light [LL]). LL exposure is known to induce desynchrony between the neurons of the SCN (Ohta et al., 2005). Thus, it is beneficial for this to be the last lighting conditions to which the animals are exposed.
The goal when comparing animal models is to confidently attribute the different phenotypes observed to the different genetic mutations. This can be promoted using a wide range of assays, standardizing variables related to the experimental conditions, using various lighting conditions, and analyzing multiple measures in the data.
Genes or Proteins of Interest for Schizophrenia that are also Linked to Sleep and Circadian Rhythms
Although all of the animal models in Table 1 are associated with schizophrenia, a subset of the genes involved is also linked to sleep or the circadian system. Below, we discuss how these models are related to sleep or circadian rhythms and the implications of this.
Briefly, Snap-25 has a rhythmic 24-h pattern of expression in the rodent SCN (Panda et al., 2002). Second, Egr3 appears to regulate sleep homeostasis through the serotonin 2A receptor (5-HT2aR; Maple et al., 2015; Gronli et al., 2016). Indeed, dysfunction of the 5-HT2aR may be involved in the pathogenesis (Hurlemann et al., 2005) and/or treatment of schizophrenia (Garay et al., 2016). Egr3 is also implicated in the circadian system, as it is upregulated in the SCN following a nighttime light pulse (Morris et al., 1998). Lastly, glutamate is also an important neurotransmitter for conveying photic entrainment cues to the SCN (Golombek and Rosenstein, 2010) for communication between SCN hemispheres (Michel et al., 2013) and between the SCN and other brain regions (Zhang et al., 2006). In addition, glutamate shows rhythmic fluctuations in the prefrontal cortex during the sleep/wake cycle (Lopez-Rodriguez et al., 2007). Group II metabotropic glutamate receptors mGLU2 and mGLU3 are encoded by the genes Grm2 and Grm3, which are expressed in the SCN (Ebling, 1996) and have various effects on sleep structure (Feinberg et al., 2002).
In addition, in a study by Dittrich et al. (2017), mice of the 129S4 strain, which have a natural deletion mutation of Disc1, showed altered sleep regulation compared with C57BL/6N mice. Interestingly, introducing normal Disc1 locus from C57BL/6N mice did not rescue the phenotype. This may highlight the involvement of additional mutations in 129S4 mice in the sleep-related pathway that may or may not involve Disc1.
This emphasizes that caution should be exercised when attributing causality between single mutations in mouse models and resulting phenotypes. Overall, it is possible that a gene or protein of interest involved in both schizophrenia and sleep or circadian rhythms is involved in the interaction of these factors, but in some cases, the effect could be in parallel, with no causal relationship.
Circadian Disturbances May Affect Disorder Onset and Severity of Schizophrenia Symptoms
Negative Health and Behavioral Consequences of Circadian Rhythm Disruption
With the entry into a highly digital era and the high demands of the current 24-h society, many people prioritize aligning sleep/wake times with social and work times, instead of natural circadian timing cues, such as the rising and setting of the sun (Roenneberg et al., 2019). Compelling evidence suggests that there are adverse mental and physical consequences of perturbations of our circadian biology, especially if exposure to these perturbations is chronic (Evans and Davidson, 2013). Circadian rhythm disruption has been linked to cardiovascular disease, different types of cancers, diabetes, and obesity risk (Vetter et al., 2015; Strohmaier et al., 2018; Wang et al., 2018). Chronic circadian disruption was shown to increase mortality rates in Drosophila (Pittendrigh and Minis, 1972) and aged mice (Davidson et al., 2006), to alter innate immune responses (Castanon-Cervantes et al., 2010), and to directly regulate mood-related behaviors and cognitive functions in mice (LeGates et al., 2012; Fisk et al., 2018). In a study by Karatsoreos and colleagues (2011), the researchers reported that housing mice in 20-h LD cycles resulted in a number of effects such as weight gain, a reduction of dendritic length in neurons in the brain, and decreased cognitive flexibility.
These observations suggest that sleep and circadian disruption lead to negative health and behavioral consequences. It is largely unknown whether sleep and circadian disruptions affect schizophrenia and if so, how? As mentioned above, reports have shown that sleep disturbances can precede prodromal psychotic symptoms in patients with schizophrenia (Tan and Ang, 2001; Chouinard et al., 2004; Wulff and Joyce, 2011; Zanini et al., 2013). However, even if a causal relationship between such disturbances and the etiology of schizophrenia is suggested, the presented results are correlations. More work would be needed to demonstrate a causal relationship between circadian disruptions and schizophrenia. Below, we describe the few studies that have attempted to address such a causal link.
Testing Whether Circadian Rhythm Disruption is a Risk Factor for Schizophrenia
A commonly accredited hypothesis is that schizophrenia is a multifactorial disorder in which numerous risk factors interact within a genetically vulnerable individual, resulting in schizophrenia illness (Davis et al., 2016). Perhaps circadian rhythm disruption increases the risk for schizophrenia and interacts with other risk factors to exacerbate schizophrenia-relevant behavioral and neurobiological abnormalities (Figure 2).
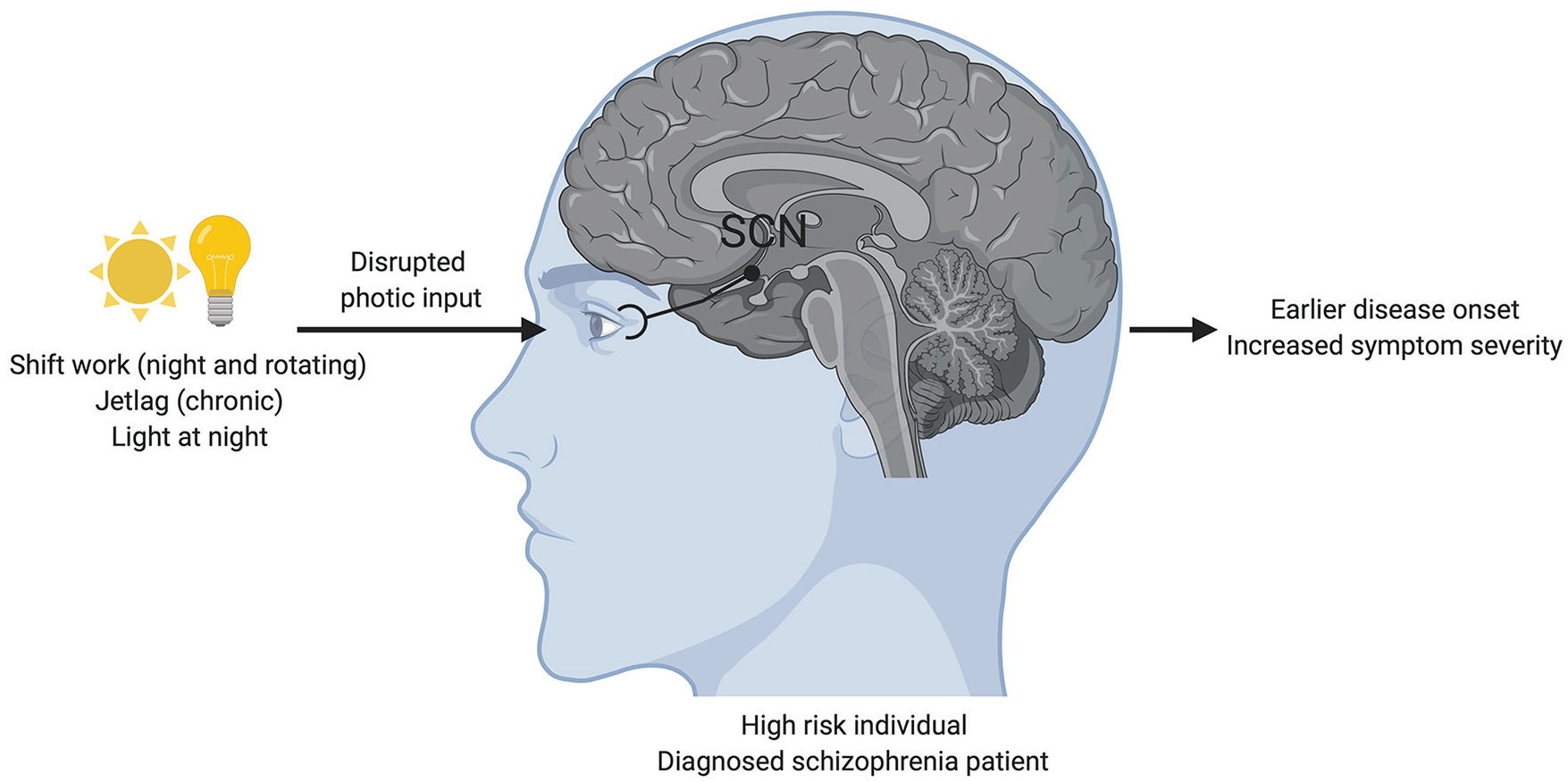
Circadian disruption is proposed to interact with other schizophrenia risk factors to promote schizophrenia-relevant behavioral and neurobiological abnormalities. Disrupted photic input hits light-sensing retinal ganglion cells, which relay the signals to the SCN. We hypothesize that because of the link between circadian rhythms and schizophrenia, disturbances to the circadian system can affect disorder outcomes and symptom severity in patients. This suggests that “vulnerable” individuals would benefit from limiting exposure to environments that can disturb circadian rhythms and that restoring circadian rhythmicity could be used as an adjunctive therapy for schizophrenia. Note that color version is available online.
Chronotype is an individual’s preferred timing of sleep and activity, which is a reflection of one’s endogenous circadian clock, the social environment, and age-related factors (Takahashi et al., 2018; Roenneberg et al., 2019). In general, the more extreme the chronotype (i.e., extreme morning or extreme evening preference), the greater the misalignment with conventional social schedules (for example, work and social schedules), resulting in social jetlag (Wittmann et al., 2006). Social jetlag imposes a chronic disruption on the circadian system, and late chronotypes in particular show substantial sleep debt (Roenneberg et al., 2003) and greater social jetlag than early chronotypes (Roenneberg et al., 2019). In a GWAS, Jones et al. (2019) explored the genetics of chronotype in relation to mental health disorders including schizophrenia. In a subset of nearly 700,000 subjects, the researchers used Mendelian randomization to infer causality between morningness (being an early chronotype) and reduced risk of schizophrenia (Jones et al., 2019). Perhaps the relationship between schizophrenia and chronotype is based on social jetlag; thus, conducting large studies on populations including individuals with schizophrenia, which would include information on chronotype and social jetlag, would be informative.
Circadian rhythm disruption as a risk factor for schizophrenia could be better studied in animal models, but such studies are scarce. To our knowledge, the only study so far that has directly tested this hypothesis is the one by Bhardwaj and colleagues (2015) using Sandy (Dysbindin-1 mutant) mice. Mice with a mutation in the Dtnbp1 gene show behavioral abnormalities, including cognitive deficits, reminiscent of schizophrenia patients (Bhardwaj et al., 2009; Cox et al., 2009; Talbot, 2009). Sandy mice also have altered dopamine/D2 receptor signaling (Papaleo et al., 2012) and altered neuronal excitability. Schizophrenia-relevant behaviors of Sandy mice were tested once after exposure to an LD cycle and then once more after LL exposure, a condition that alters and dampens circadian rhythmicity. LL exposure caused significant deficits in prepulse inhibition of acoustic startle response, hyperlocomotion, and less anxiety-like behavior in Sandy mice compared to controls (Bhardwaj et al., 2015). It was hypothesized that circadian disturbances, due to LL exposure, may have exacerbated preexisting connectivity impairments in the Sandy mice and therefore unmasked behavioral deficits. Notably, 3 weeks of LD were sufficient to partially rescue the observed behavioral deficits (Bhardwaj et al., 2015).
Clearly, further research is needed to evaluate whether circadian disruption is a risk factor for schizophrenia. Studies should test whether exposure to circadian disruption that desynchronizes rhythms in the SCN or in other tissues, or genetic mutations that disrupt clock function, would exacerbate schizophrenia-relevant behavioral and neurobiological abnormalities.
Conclusion
If there is a prominent link between circadian rhythms and schizophrenia, then disturbances to the circadian system could affect disorder outcomes in patients. This suggests that at-risk and affected individuals would benefit from limited exposure to environments that promote circadian rhythm disruption. In addition, restoring circadian rhythmicity could be used as an adjunctive therapy for schizophrenia. There is a clear urgency for novel and adjunctive therapies, as the current limited treatment options are insufficient to alleviate the full range of symptoms experienced by individuals with schizophrenia. The mouse model studies suggest that the biological mechanisms that contribute to sleep disruption in the context of schizophrenia may be distinct from those responsible for altered circadian entrainment and rhythmicity, such that different treatment strategies might have to be considered to address each aspect.
Here, we argue that circadian disruption is a core pathophysiological component of schizophrenia. Circadian disruption is a consistent core feature observed in most patients. Moreover, promising preliminary evidence suggests that circadian disruption may also act as an interacting risk factor for schizophrenia, affecting aspects of the disorder.
We acknowledge that sleep and circadian disruptions are not specific to schizophrenia. Indeed, a number of studies have observed sleep and circadian disruptions in various neurodevelopmental, neurodegenerative, and intellectual disorders. Perhaps this speaks to sleep and circadian disturbances being a key psychopathological mechanism across a wide range of disorders.
The topic of sleep and circadian rhythms in schizophrenia is a burgeoning field due to recent technical and methodological advances that allow this phenomenon to be studied more precisely. Most studies exploring the link between circadian rhythms and schizophrenia so far have mainly been correlative and descriptive in nature. There is currently a significant gap in the literature with regard to the precise mechanistic link between schizophrenia and circadian rhythms, which future research should address. Another important aspect that remains to be explored is the interaction of circadian disruption with nongenetic risk factors of schizophrenia.
Footnotes
Acknowledgements
The authors thank the members of the Cermakian and Srivastava laboratories for helpful discussions. Figures were created with ![]() . The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Canadian Institute of Health Research (PJT-153299), the Natural Sciences and Engineering Research Council of Canada (RGPIN-2017-04675), and Velux Stiftung (Project 927). T.C.D. was supported by graduate scholarships from the Schizophrenia Society of Canada Foundation, the Canadian College of Neuropsychopharmacology, and the Fonds de Recherche du Québec–Santé.
. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Canadian Institute of Health Research (PJT-153299), the Natural Sciences and Engineering Research Council of Canada (RGPIN-2017-04675), and Velux Stiftung (Project 927). T.C.D. was supported by graduate scholarships from the Schizophrenia Society of Canada Foundation, the Canadian College of Neuropsychopharmacology, and the Fonds de Recherche du Québec–Santé.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
