Abstract
Understanding behavioral rhythms in a pest species can contribute to improving the efficacy of control methods targeting that pest. However, in some species, the behavioral patterns recorded in artificial conditions contrast greatly with observed wild-type behavioral rhythms. In this study, we identify the determinants of daily activity rhythms of the soft and stone fruit pest Drosophila suzukii. The impact of gender, space, social housing, temperature, light, fly morph, and the circadian clock on D. suzukii locomotor rhythms was investigated. Assays were performed under artificial laboratory conditions or more natural semifield conditions to identify how these factors affected daily locomotor behavior. Daily locomotor activity patterns collected under semifield conditions varied very little between the various sex and social condition combinations. However, in lab-based assays, individual and group-housed males often exhibited divergent activity patterns, with more prominent hyperactivity at light/dark transitions. In contrast, hyperactivity responses were suppressed under lab protocols mimicking summer conditions for groups of females and mixed-sex groups. Moreover, when environmental cues were removed, flies held in groups displayed stronger rhythmicity than individual flies. Thus, social interactions can reinforce circadian behavior and resist hyperactivity responses in D. suzukii. Fly morph appeared to have little impact on behavioral pattern, with winter and summer morph flies displaying similar activity profiles under April semifield and laboratory mimic environmental conditions. In conclusion, separate and combined effects of light, temperature, circadian clock function, and social interactions were apparent in the daily activity profiles of D. suzukii. When groups of female or mixed-sex flies were used, implementation of matching photoperiods and realistic daily temperature gradients in the lab was sufficient to re-create behavioral patterns observed in summer semifield settings. The ability to leverage lab assays to predict D. suzukii field behavior promises to be a valuable asset in improving control measures for this pest.
Behavioral and physiological processes are regulated in a wide range of organisms by an internal pacemaker: the circadian clock (Allada and Chung, 2010). Light and temperature, along with other biotic and abiotic factors, entrain the clock, which is vital in synchronizing organisms to their environment (Dubruille and Emery, 2008). Observations of model organisms, within a laboratory setting, are often made under simplified environmental conditions. While this approach has expanded knowledge and provided an invaluable understanding of the circadian clock (Young, 2018), there is a need for complementary studies involving more realistic environmental conditions. Behavioral and physiological rhythms in a natural setting remain relatively unexplored for many species, but their analysis could provide valuable insights into the most relevant phenotypic outcomes of daily timekeeping and highlight opportunities to exploit such rhythms in pest species (Miyata, 2011).
Drosophila suzukii (Matsumura) is a global pest of soft, stone, and ornamental fruit and has dispersed from its native range in Japan, aided by the importing and exporting of contaminated fruit (Cini et al., 2014). Female D. suzukii are equipped with serrated oviscapts, which enable them to cut into the epicarp of ripening, otherwise healthy soft fruits to deposit eggs (Atallah et al., 2014). After hatching, the larvae consume the fruit from within, which causes collapse, resulting in yield and economic losses (Walsh et al., 2011; Lee et al., 2011). A wide range of integrated pest management options are emerging to tackle this pest (Landolt et al., 2012; Cha et al., 2014; Cha et al., 2017; Haye et al., 2016; Gabarra et al., 2015; Wiman et al., 2016). A greater understanding of behavioral patterns in D. suzukii would enable specific targeting of rhythms, which could result in an increased efficacy of these methods and higher level of control (Ferguson et al., 2015).
It is not always possible to perform behavioral assays under natural conditions, because limiting factors, such as providing reliable power sources, often restrict the ability to monitor specimens in a remote, semifield location. This has driven groups to investigate behavioral patterns within laboratory settings, instead often using simplified environmental cycles (i.e., 12:12 L:D, 23 °C) or constant environmental conditions for convenience and ease of interpretation (Lin et al., 2014). However, successful field and semifield observations of Drosophila have highlighted the large variation in behavioral responses between laboratory and more natural settings (Green et al., 2015; Vanin et al., 2012; De et al., 2013; Menegazzi et al., 2012; Prabhakaran et al., 2013). Mammals also exhibit discrepancies between the 2 settings, with some species switching from diurnal to nocturnal when housed within a laboratory (Daan et al., 2011; Gattermann et al., 2008). More realistic environmental cycles can be introduced in the laboratory to reduce the divergence between natural and artificial conditions and has been explored in Drosophila diapause (Nagy et al., 2018) and, to a limited extent, locomotion (Hamby et al., 2013). For observations of oviposition (Shaw et al., 2018b) and emergence (Kannan et al., 2012), the success of laboratory-based simulation of seminatural behavioral profiles was addressed directly. Such an investigation of the parameters needed to reproduce seminatural behavior in the laboratory has, to date, not yet been conducted for the locomotor behavior of D. suzukii. Thus, the aim of this research was to identify the determinants of daily rhythms of D. suzukii locomotor activity that may help predict field behavior. In the experiments presented here, the impact on D. suzukii locomotor rhythms was assessed for gender, space, social housing, temperature, light, and the circadian clock. Fly morph was also a parameter. Within wild populations, winter morph D. suzukii are found through the colder months once temperatures drop below 10 °C. Winter morph flies are better able to withstand cooler temperatures and have a longer life span than summer morph flies when exposed to low temperatures (Dalton et al., 2011; Jakobs et al., 2015; Kacar et al., 2016; Plantamp et al., 2016; Ryan et al., 2016). To test whether there were quantitative or qualitative differences in daily activity patterns between winter or summer morph flies, a limited number of comparative analyses including winter morph flies was run. All of these factors were assessed in a controlled manner in the laboratory, while the impact of gender and social housing setting was also addressed at semifield conditions sampled at 4 different times of year. Understanding how environmental factors affect key behaviors in D. suzukii could enable us to target specific behaviors and improve pest control measures.
Materials and Methods
Drosophila Culturing
D. suzukii cultures were established at the University of Southampton from a wild strain collected in 2013 in Trento, Italy. Populations of D. suzukii were housed in glass 25- × 95-mm Opticlear vials (Kimble Chase, Fisher Scientific, Loughborough, UK) at 23 °C in a 12:12 L:D cycle at a constant 65% relative humidity. Cultures were maintained on standard yeast cornmeal diet (6 g agar, 48 g table sugar, 73 g yellow maize meal, 17 g yeast, 10 g soya flour, 46 g malt extract, 0.2% propionic acid, 0.07% methyl-4-hydroxybenzoate, distilled H2O to 1 L) and were transferred into new vials every week to promote egg laying, as D. suzukii prefer less densely colonized oviposition sites (Mitsui et al., 2006). After the transferal of the flies, the vials were returned to the prior conditions to await the subsequent generation emergence. Once emergence began, the vials were kept for a week before the offspring were transferred to new vials. At this point, the existing vials were discarded. This ensured that overlapping aged populations were established. Cultures were kept genetically diverse by randomly mixing offspring between multiple populations to reduce inbreeding at each transferal. Summer morph flies were held within the conditions stated above. Winter morph flies were produced by transferring stage 3 larvae to the same diet within a 10 °C incubator under constant darkness until a minimum of 200 adults of each sex emerged. Under these conditions, D. suzukii emerged as a larger and darker fly able to withstand cooler temperatures than the summer morph (Dalton et al., 2011; Shearer et al., 2016; Stephens et al., 2015; Toxopeus et al., 2016; Wallingford and Loeb, 2016). Prior to commencing the assays involving winter morph flies, a sample of 10 females was removed from the culture and dissected to determine their reproductive state. All females was categorized as stage 1 using the scale proposed by Gerdeman and Tanigoshi (2012), corresponding to “no distinguishable ovaries.” We would not expect a switch to the summer reproductive state during the experiments, as temperatures used in the behavioral assays with the winter morph flies (8-15 °C) were within the range required to produce winter morph D. suzukii (Dalton et al., 2011; Hamby et al., 2016; Stockton et al., 2018; Wallingford et al., 2016) and winter morph females have been found to delay reproduction under comparable conditions (Zhai et al., 2016; Wallingford et al., 2016).
Drosophila Activity Monitoring
The TriKinetics Drosophila Activity Monitoring (DAM; TriKinetics Inc, Waltham, MA) system was used to record activity by analyzing the number of infrared beam breaks in 5-min bins (Pfeiffenberger et al., 2010). D. suzukii were taken from mixed-sex cultures and were 3 to 7 days old at the start of the assay and presumed mated. Flies were immobilized and held on a CO2 pad before being transferred by the wing with soft forceps to either cuvettes (for individuals, 5 mm × 65 mm glass cuvettes; volume ≈1.28 cm3) or vials (for groups, 25-mm × 95-mm glass vials; volume ≈44.8 cm3). Tubes and vials were monitored within Trikinetics DAM2 and LAM25 monitors for narrow cuvettes and population vials, respectively. Both contained a basic sugar agar food (100% dH2O, 1% agar, 5% table sugar, 0.07% Tegosept). Cuvettes and vials were filled with the sugar agar food up to a height of ~10 and ~30 mm, respectively, and left to set for 24 h. Cuvettes were loaded with individual males or females. Population vials were loaded with either individual males or females, single-sex groups of 10 males or 10 females, or mixed-sex groups of 10 males and 10 females. Both vessels were sealed by a breathable cotton bung. Males were identified by the presence of sex combs and a single spot on each forewing and females by the oviscapt morphology. Both vessel types were held horizontal while the flies became active. Both vials and cuvettes were monitored for a minimum of 6 days after a 24-h “settling” period.
Semifield Locomotion
Individual and populations of flies were exposed to natural environmental conditions in a sheltered, semifield setting in the southeast of England at 4 different times in the year: April, June, August, and October (Table 1). In this location, artificial light was absent. The DAM systems were held within a well-ventilated waterproof housing unit to protect the equipment from rain and direct sunlight. Environmental monitors (DEM Model DEnM, TriKinetics Inc) were deployed alongside the DAM to record temperature and humidity conditions. The average sunrise and sunset times through the assay period were recorded (Table 1). Summer morph flies were used in June, August, and October collections. Winter morph and summer morph fly locomotor activity was collected in parallel during April conditions to mimic the variation in morph in the field at this time of the year in the United Kingdom (Fountain et al., 2017). For winter morph assays, only individual males, individual females, and mixed-sex groups were assayed because of the difficulty in culturing large populations of winter morph flies in the laboratory.
Environmental conditions for semifield locomotor assays.
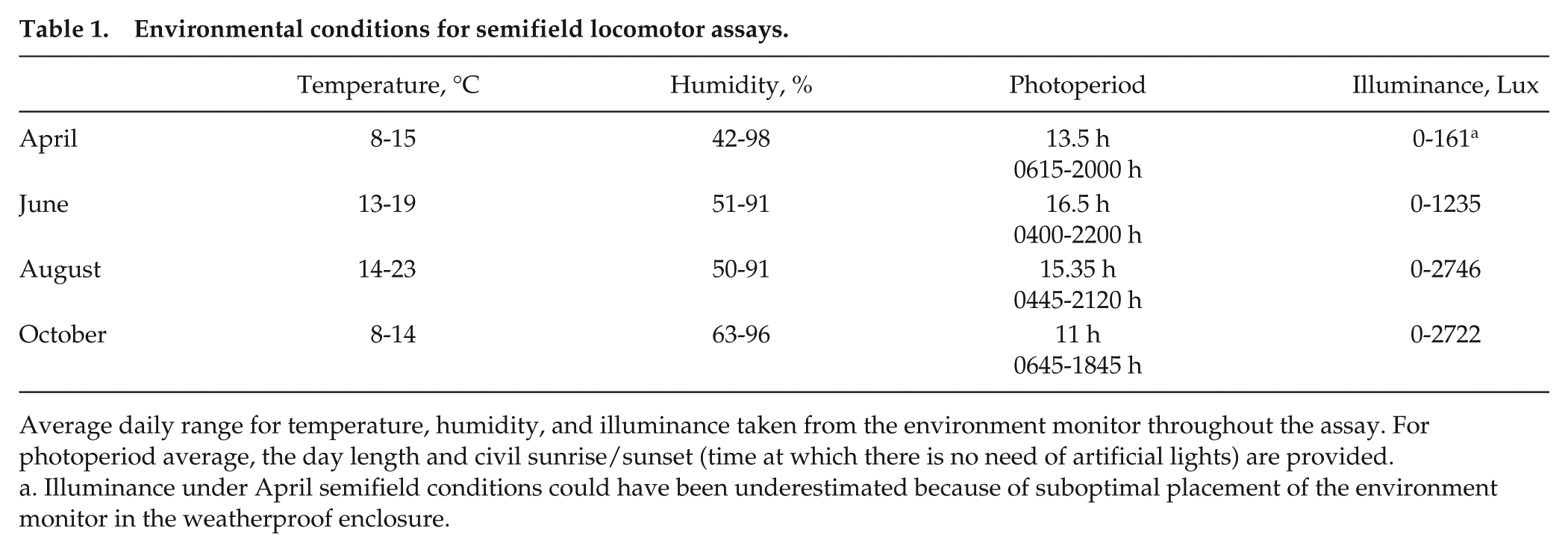
Average daily range for temperature, humidity, and illuminance taken from the environment monitor throughout the assay. For photoperiod average, the day length and civil sunrise/sunset (time at which there is no need of artificial lights) are provided.
Illuminance under April semifield conditions could have been underestimated because of suboptimal placement of the environment monitor in the weatherproof enclosure.
Laboratory Locomotion
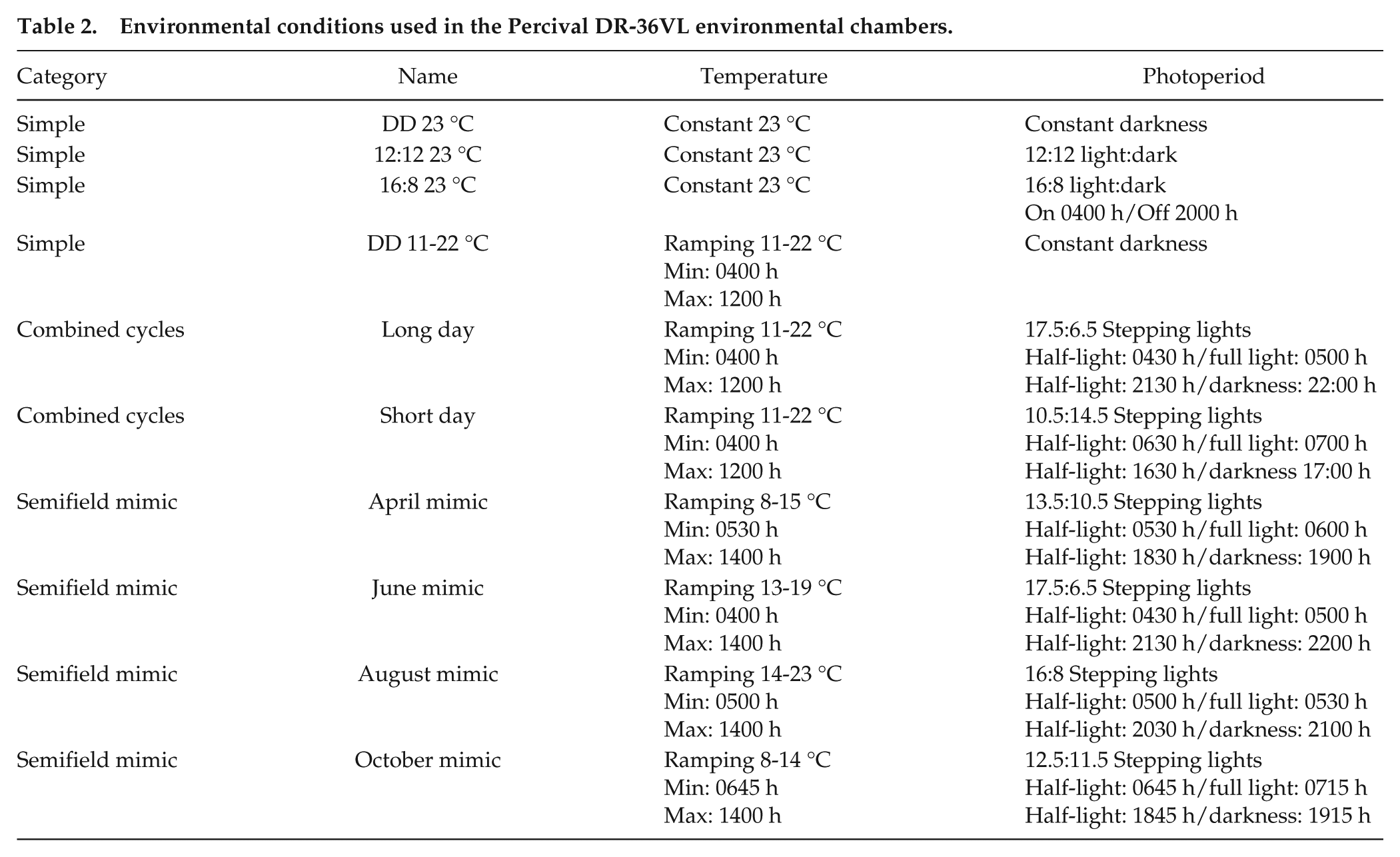
A range of environmental conditions was established in Percival DR-36VL environmental chambers within the insectary facility at the University of Southampton, United Kingdom. Conditions were divided into 3 categories: simple cycles, combined temperature/light cycles, and semifield mimic (Table 2). Humidity was kept at a constant 65% relative humidity in all laboratory conditions. The semifield mimic environmental conditions were extrapolated from data loggers deployed alongside the DAM system in the semifield assays. Average daily temperature and light cycles were established from the data. To replicate dawn, stepping light was used, in which half the lighting banks came on initially, followed half an hour later by the remaining half (full illuminance: 2670 lux, half illuminance: 1386 lux). This was reversed for dusk. We also tested whether an increase in space for individual flies affected locomotor behavior by comparing the behavior of individual males and individual females in narrow cuvettes versus population vials for the simple and combined environmental cycles in the laboratory.
Environmental conditions used in the Percival DR-36VL environmental chambers.
Analysis
Only flies that survived the whole assessment period were included in all analyses, resulting in varying replication (Table 3). Groups in which any mortality occurred were also discarded. The CLOCKLAB software package (Actimetrics, Inc.) was used to analyze locomotor activity counts for the generation of actograms, chi-square periodograms, and daily activity profiles. The number of beam breaks in 5-min intervals was collected for 6 days, and these data were used to generate the activity profiles. The beam break counts were then binned into 30-min intervals for further analysis.
Number of each fly sample recorded within each environmental condition and used for analysis.
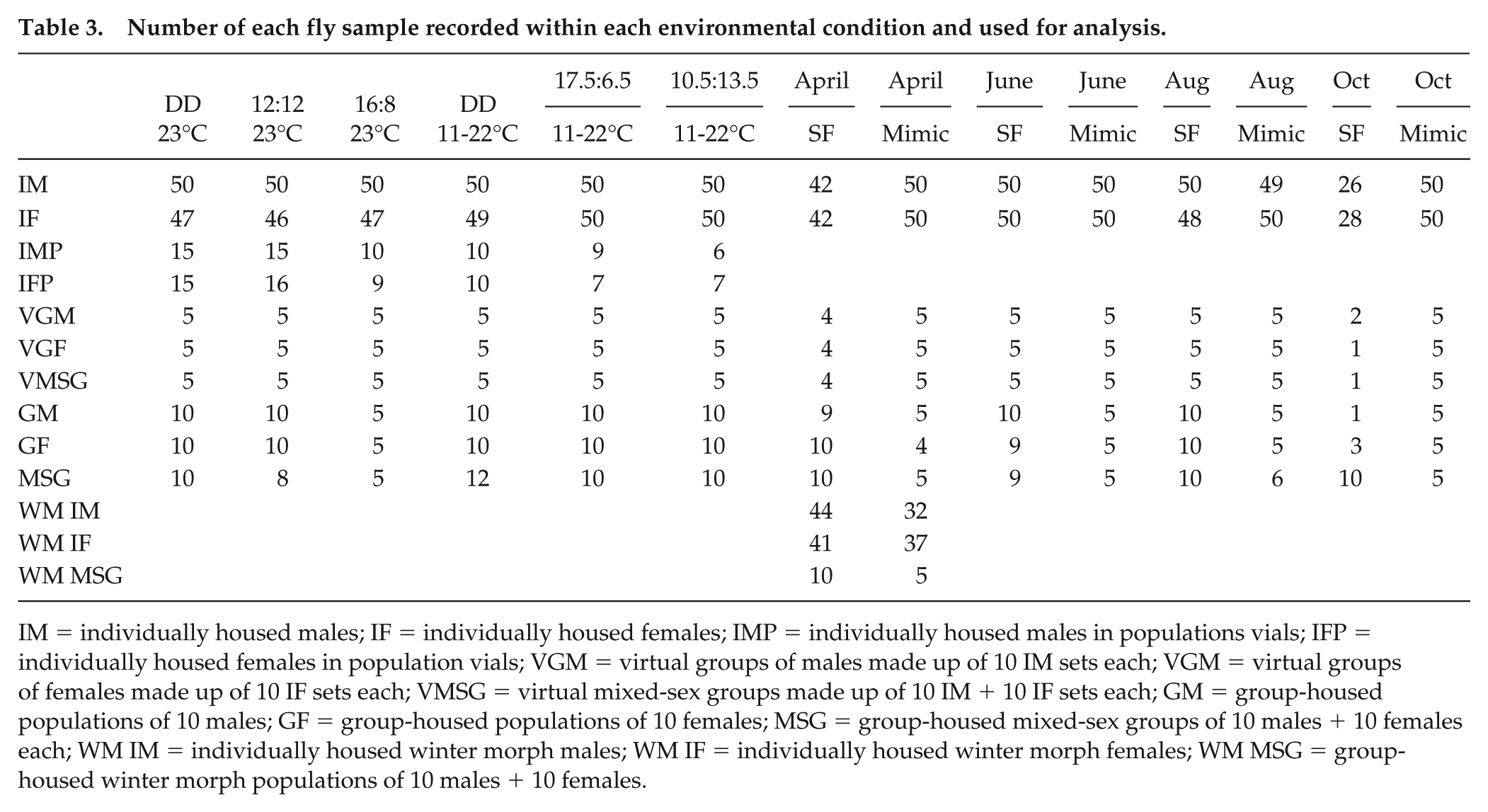
IM = individually housed males; IF = individually housed females; IMP = individually housed males in populations vials; IFP = individually housed females in population vials; VGM = virtual groups of males made up of 10 IM sets each; VGM = virtual groups of females made up of 10 IF sets each; VMSG = virtual mixed-sex groups made up of 10 IM + 10 IF sets each; GM = group-housed populations of 10 males; GF = group-housed populations of 10 females; MSG = group-housed mixed-sex groups of 10 males + 10 females each; WM IM = individually housed winter morph males; WM IF = individually housed winter morph females; WM MSG = group-housed winter morph populations of 10 males + 10 females.
To calculate anticipation, the method suggested by Zhang and Emery (2013) was performed on data collected from the simple and combined laboratory cycles (12:12 23 °C, 16:8 23 °C, long-day, and short-day cycles). In brief, the average activity counts of each social grouping were measured in 30-min bins over a 2.5-h period during the middle of the dark phase for each day. This was subtracted from the average activity counts measured in 30-min bins over the 2.5 h prior to lights-on. This was also performed for the evening anticipation by subtracting the average activity counts during 2.5 h during the middle of the light phase from the average of 2.5 h prior to lights-off. The data were tested for normality with the Shapiro-Wilk test based on the results (normality was confirmed in only 2 of 8 data sets); the Kruskal-Wallis nonparametric alternative to the 1-way analysis of variance was conducted with Dunn’s post hoc tests for the analysis with different days as replicates (7 days).
To measure normalized hyperactivity at daily light/dark transitions, the 30-min interval that commenced with the lighting event was identified for both dawn and dusk, and its number of counts was then normalized by dividing it by the daily average activity (per 30 min). The dawn and dusk periods were each defined as a 2-h period that encompassed 30 min before a lighting event and 1.5 h after the event. In those conditions that experienced stepping light events (i.e., the semifield mimic cycles within the laboratory) the 2-h period commenced 30 min before the first event. For profiles collected under constant darkness, subjective dawn/dusk was defined by daily phases corresponding to the most recent dawn and dusk periods. To ensure the best signal-to-noise estimates, normalized hyperactivity was determined subsequent to averaging over both the available replicate days and flies. Prior to analyses addressing the impact of environmental or social housing conditions on dawn- or dusk-associated hyperactivity, data sets were tested for the normality of their distribution using the Shapiro-Wilk test. Based on these results (instances of nonconfirmed normality in all data sets), nonparametric statistical tests (Kruskal-Wallis) were employed with an additional Dunn’s multiple comparison post hoc tests in GraphPad Prism 7.03 on these results. Only summer morph flies were used in this analysis.
To determine the impact of social housing on timekeeping ability in constant conditions (DD 23 °C), the period length and the strength of the rhythm (relative rhythmic power [RRP]) of locomotor behavior was identified by the CLOCKLAB software. The RRP was calculated from chi-square periodogram analyses in CLOCKLAB by dividing the “power” by the “significance” threshold values (using a threshold of p = 0.01) manually in Excel (Goda et al., 2011). To explore the impact of social interactions on activity, data from group-housed flies were compared with that of individually housed flies both directly and after the creation of “virtual fly groups” by summing the activity data from matching numbers of individually housed flies (10 individual males, 10 individual females, or 10 individual males and 10 individual females, respectively). Circadian activity patterns were categorized as arrhythmic (in which period length could not be identified), weakly rhythmic (RRP 1-1.5), and rhythmic (RRP >1.5), and the distribution of observed behavioral patterns across these categories was compared for the different social groupings. Significant differences in the distribution of flies across the 3 rhythmicity categories as a function of social setting was explored using pairwise Fisher’s exact tests, and the results were annotated relative to a multiple-testing corrected (Bonferroni) p value threshold.
For social groupings in which a period length could not be identified, data were not included within the overall analysis or figures in which statistical comparisons of RRP and period length were made. Subsequently, based on the outcome of Shapiro-Wilk tests for normality, a Kruskal-Wallis test with Dunn’s post hoc tests was performed in GraphPad Prism 7.03 on both of these parameters. Only summer morph flies were used for this experiment.
Average activity counts per hour per fly were calculated by dividing hourly activity by the number of flies in the sample group to enable comparison between all social groupings (including virtual populations) and environments. This was done to ensure that activity counts based on equal numbers of individually versus group-housed flies were compared. Nonparametric Kruskal-Wallis tests with post hoc pairwise comparison of average activity counts per hour per fly (using Mann-Whitney tests with multiple testing correction) were performed in SPSS. In October, semifield collections survival was low, and so, in some cases, fewer replicates were included and are indicated in figure legends or tables. In addition to the summer morph flies used across all conditions, winter morph flies were included for a subset of these analyses as annotated.
Results
Impact of Environment and Social Conditions on Daily Locomotor Activity
Daily locomotor activity profiles exhibited clear differences across combinations of environment and social setting (Fig. 1a, b). Profiles collected in semifield conditions (exposed to natural light and temperature cycles; Fig. 1b, columns 1, 3, 5, and 7; Fig. 1c, columns 1 and 3) and those collected in the laboratory under constant darkness (Fig. 1a, columns 1, 2), showed less variation in locomotor pattern between social groupings than those collected in the laboratory with a light/dark cycle. In the presence of light/dark cycles, increased activity was observed during the light phase relative to the dark phase. In the laboratory, individual males (Fig. 1a, b, c, row 1), individual females (Fig. 1a, b, c row 2), and group-housed males (Fig. 1a, row 5 and 1b, row 3) displayed behavioral profiles featuring prominent bouts of hyperactivity, immediately following lights-on and lights-off transitions. This response was, in most cases, more subdued for group-housed females (Fig. 1a, row 6 and 1b, row 4) or summer morph mixed-sex groups (Fig. 1a, row 7 and 1b, row 5) under the same environmental conditions. Flies monitored under semifield conditions (Fig. 1b, columns 1, 3, 5, 7 and Fig. 1c, column 1, 2) did not show these bursts of activity at dawn and dusk to the same extent, suggesting this behavioral response may be an artifact caused by abrupt changes in light levels, associated with the use of fluorescent lights.
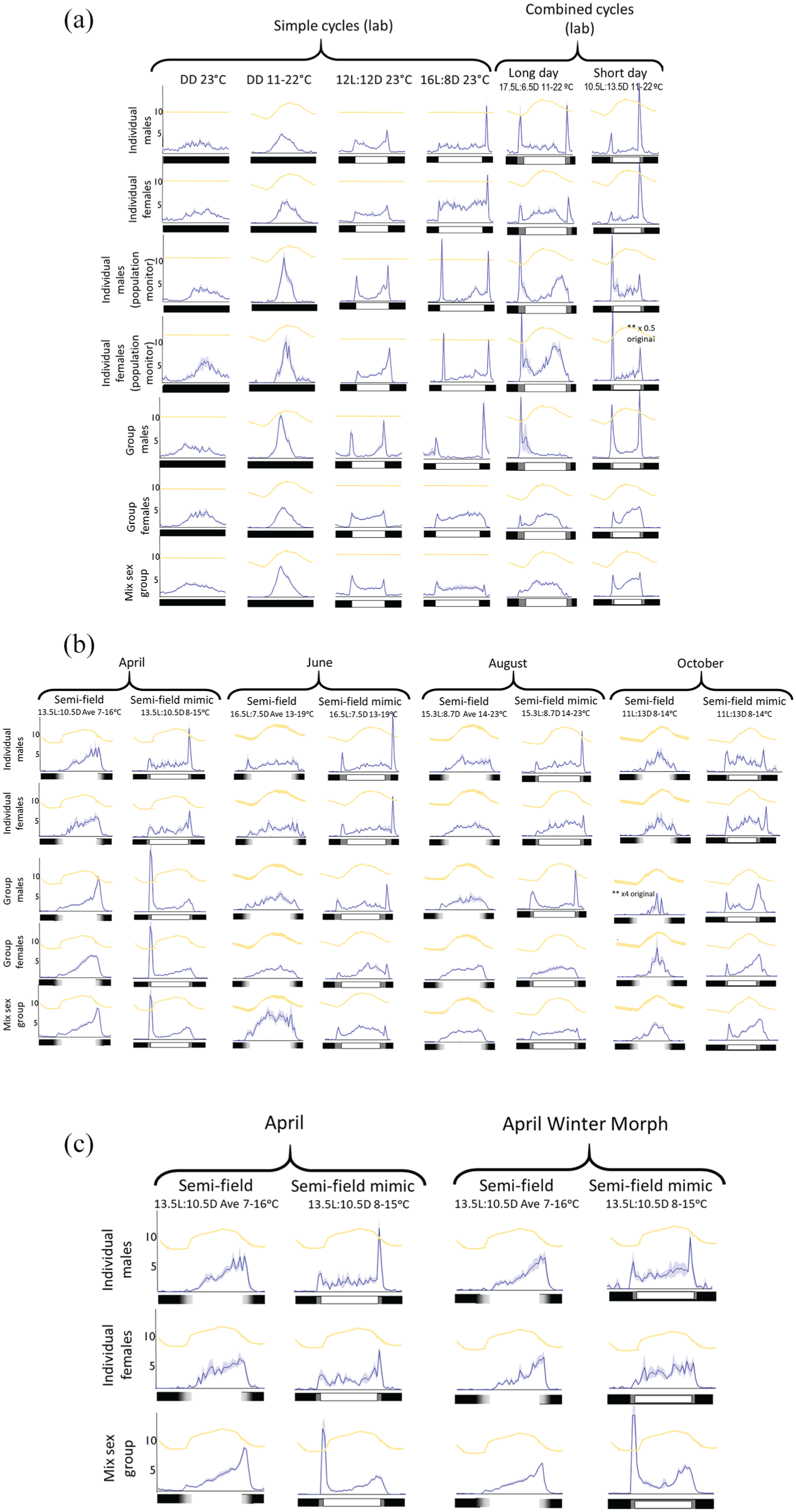
Normalized activity profiles of summer morph D. suzukii adults under (a) simple and combined laboratory conditions, (b) semifield and laboratory-based mimic conditions, and (c) winter and summer morph flies under April semifield and laboratory-based mimic conditions. For ease of comparison, data for summer morph flies in (c) is duplicated from (b). Profiles were collected under different laboratory and semifield conditions (columns) and in various social groupings (rows) collected over a 6-consecutive-day period. The blue line and shaded area for each profile represent the average activity counts per half hour ±SEM across these 6 days. The temperature regime is displayed by the orange line at the top of each activity profile and is not relative to axis counts. The temperature cycle for semifield conditions represents the average daily cycle temperature over the collection period. Temperature maxima and minima are indicated in the column captions, with additional information provided in Tables 1 and 2. The y-axes solely refer to average activity counts. The laboratory light regime is indicated by the black (no light banks), gray (2 light banks), and white bar (4 light banks) at the bottom of each activity profile. Light in semifield shows night as black, gray as dusk or dawn, and white as day. All flies were kept in a 12:12 LD 23 °C laboratory environment prior to experimental conditions, and this is particularly relevant as the entraining condition for the laboratory DD 23 °C conditions (as in column 3). The number of replicates contributing to each behavioral profile are annotated in Table 3. In all cases, group-housed sets contained either 10 males or 10 females or 10 males + 10 females. Because of low survival, October semifield activity collections were based on 1 group of male flies and 3 groups of females. **Indicates activity profile on different axis scale to all others.
In the presence of a temperature cycle, the locomotor behavior of flies, under semifield and some laboratory conditions, tended to rise and fall along with the temperature; this was especially clear for flies maintained in the presence of a daily temperature gradient in the dark within the laboratory. Dramatic changes in the daily activity profile were observed when a long- or short-day exposure to fluorescent lights was integrated with a daily temperature gradient for the combined cycles (Fig. 1a, column 2 versus 5, 6). Again, hyperactivity was prominently observed in conjunction with the turning on or off of fluorescent lights. The alignment between environmental temperature cycle and daily locomotor activity pattern appeared to be well preserved for all social conditions tested under June, August, and October semifield conditions. However, peak activity was delayed relative to peak temperature for the April semifield conditions that featured long days combined with somewhat cooler temperature cycles (Fig. 1b, column 1, 2). The peak in locomotor activity in the April semifield conditions occurred between 1900 and 1930 h, whereas the peak in temperature occurred on average between 1500 and 1600 h. Light transition–associated hyperactivity for June, August, and October lab mimic conditions was relatively subdued in groups of females or mixed-gender flies compared with individual male or female flies or group-housed males (Fig. 1b, columns 4, 6). Divergent results were, however, obtained for the April lab mimic conditions, which featured more prominent lights-on hyperactivity for group-housed flies (Fig. 1b, column 2 rows 3, 4, 5) and more pronounced lights-off hyperactivity in individual flies (Fig. 1b, column 2, rows 1, 2). Finally, the October lab mimic profiles differed from the corresponding semifield originals by lights-on–associated hyperactivity as well as either acute lights-off–associated hyperactivity in the case of individual flies (Fig. 1b, column row 1,2) or a delay in the main activity peak in the case of group-housed flies (Fig. 1b, column row 3-5).
Average daily activity profiles for individual males and females in population vials showed higher circadian locomotor activity peaks under constant conditions than individuals in narrow cuvettes (Fig. 1a, column 1). The behavior of individual flies in population vials in other simple and combined cycles in the laboratory was also suggestive of improved circadian rhythmicity in the wider vial environment, as behavioral anticipation of the lights-off transition appeared more prominent (Fig. 1, column 3-6).
A limited analysis of possible behavioral differences between winter and summer morph flies was included for April semifield and corresponding laboratory mimic conditions based on the rationale that a mixture of summer and winter morph flies may be present at this time of year within the United Kingdom. Moreover, as differences in the Drosophila female reproductive state have been reported to affect locomotor activity (Ferguson et al., 2015; Isaac, 2019), we hypothesized that analogous differences might be observed between reproductively active summer morph flies and winter morph flies that have entered reproductive diapause. As described above (in the Materials and Methods section), reproductive quiescence was, indeed, confirmed for the winter morph flies prior to their use in our behavioral analyses. Because of the difficulty in culturing the large numbers needed to assay all social groupings, only individual males, individual females, and mixed-sex groups were monitored in winter morph form. The resulting activity profiles collected for both morphs under the April conditions displayed very similar daily locomotion patterns, with the winter morph flies under semifield conditions being representative of semifield summer morph activity (Fig. 1c, column 1-4). However, regardless of morph, laboratory mimic profiles diverged from their semifield counterparts, as described above.
Anticipation
The anticipation of lighting events varied with social parameters. In the simple and combined laboratory cycles involving a light cycle (12:12 23 °C, 16:8 23 °C, long-day and short-day combined cycles), both morning and evening anticipation was evaluated. Kruskal-Wallis tests indicated a significant association between social grouping, and anticipation was found for all conditions except morning anticipation during the long-day combined cycles (see Fig. 2). Dunn’s post hoc tests revealed further pairwise significant differences, as indicated in Fig. 2. In general terms, more significant differences were observed in evening versus morning anticipation, and the impact of social setting on anticipatory behavior diverged for the single short-day condition that was included. Under 12-h or longer photoperiods, single males in population monitors showed a significant increase in evening anticipation, while mixed-sex groups showed a significant decrease in evening anticipation. Furthermore, the above-noted trend for individual flies to exhibit higher levels of evening anticipation when housed in population vials rather than narrow vials did not result in statistically significant differences in these analyses.
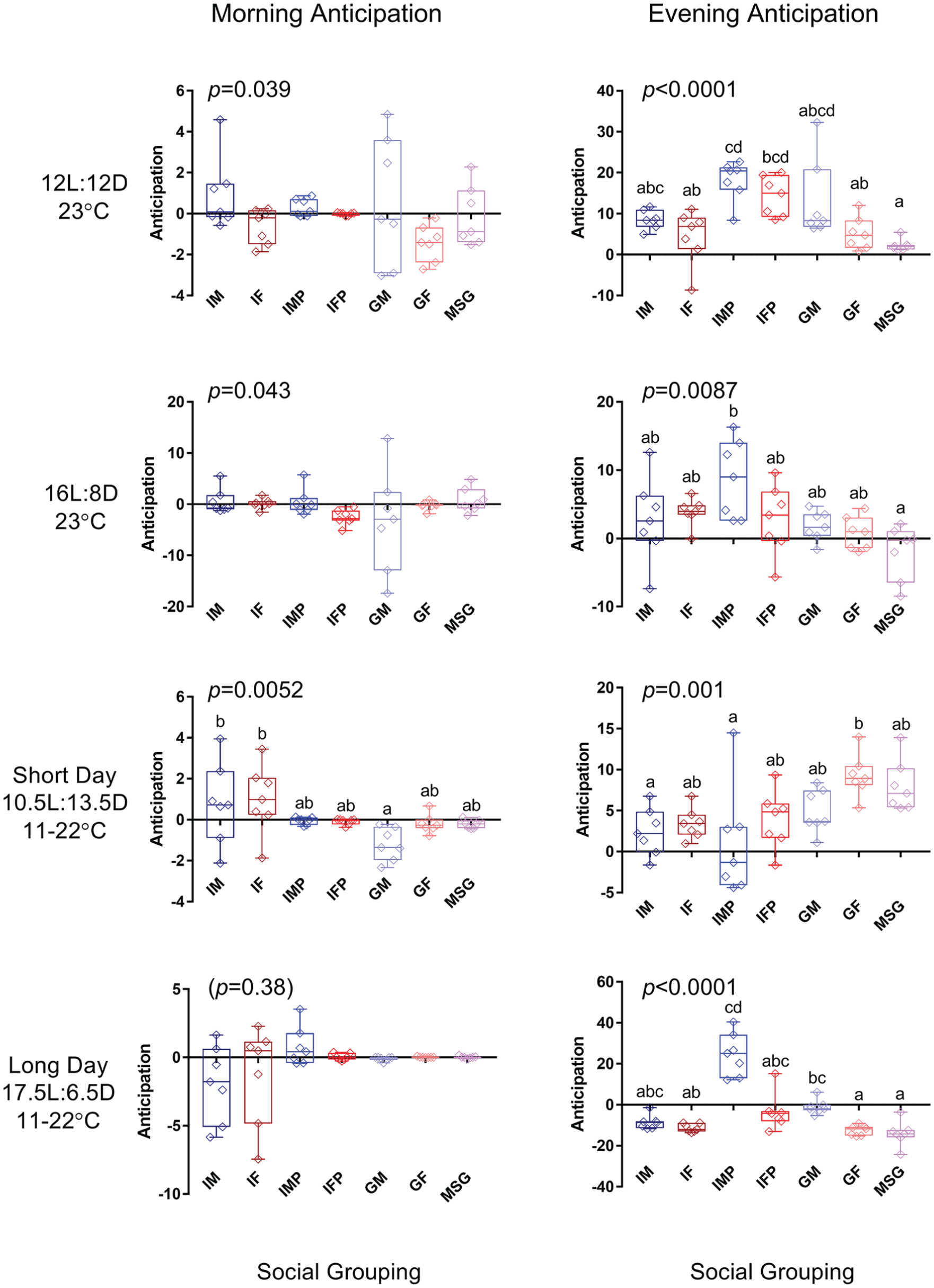
Morning (left) and evening (right) anticipation of various D. suzukii social groupings under 12:12 and 16:8 light cycles at 23 °C as well as long-day and short-day combined light/temperature cycle conditions. IM = individual males; IF = individual females; IMP = individual males in population monitors; IFP = individual females in population monitors; GM = group males; GF = groups of females; MSG = mixed-sex groups. Group sizes and replicate numbers are as described above. Anticipation analysis from 2.5 h prior to lighting events, not including the event itself. Kruskal-Willis p values are indicated above each panel, and pairwise Dunn’s post hoc test differences are annotated on the graphs.
Social and Environmental Determinants of Hyperactivity at Light/Dark Transitions
The daily activity profiles, discussed above, showed that acute behavioral responses at light/dark transitions were much more prominent under laboratory than semifield conditions (Fig. 1; Suppl. Fig. S1). The impact of environmental conditions and social groupings on light transition–associated hyperactivity was examined quantitatively. First, normalized hyperactivity at (subjective) dawn and dusk was determined from the average daily activity profile per environmental/social grouping combination (Fig. 3a, b). Next, the resulting normalized hyperactivity values were compared across environmental conditions (Fig. 3c, d) and social groupings (Fig. 3e, f). As flies in constant dark conditions and in semifield conditions showed little to no hyperactivity at (subjective) dusk or dawn across different social or environmental settings, these conditions were used as controls for the detection of significant changes in associated laboratory cycles with light/dark transitions. Separate Kruskal-Wallis tests for dawn- and dusk-associated hyperactivity each showed highly significant impacts (p < 0.0001) of both environment and social grouping. The impact of environmental conditions and social settings on dawn- and dusk-associated hyperactivity was then assessed at the level of pairwise post hoc tests. Notably, the addition of a photoperiod to a DD condition resulted in an increase in normalized hyperactivity at dawn and dusk with statistically significant differences detected in the context of an 11 to 22 °C cycle in DD, in comparison with long- and short-day photoperiods with essentially the same temperature cycle (Fig. 3c, d). Furthermore, laboratory-based protocols designed to mimic semifield conditions inadvertently produced increases in average normalized hyperactivities at light/dark transitions, and a significant result was found for the April and October semifield mimic at dawn (Fig. 3c).
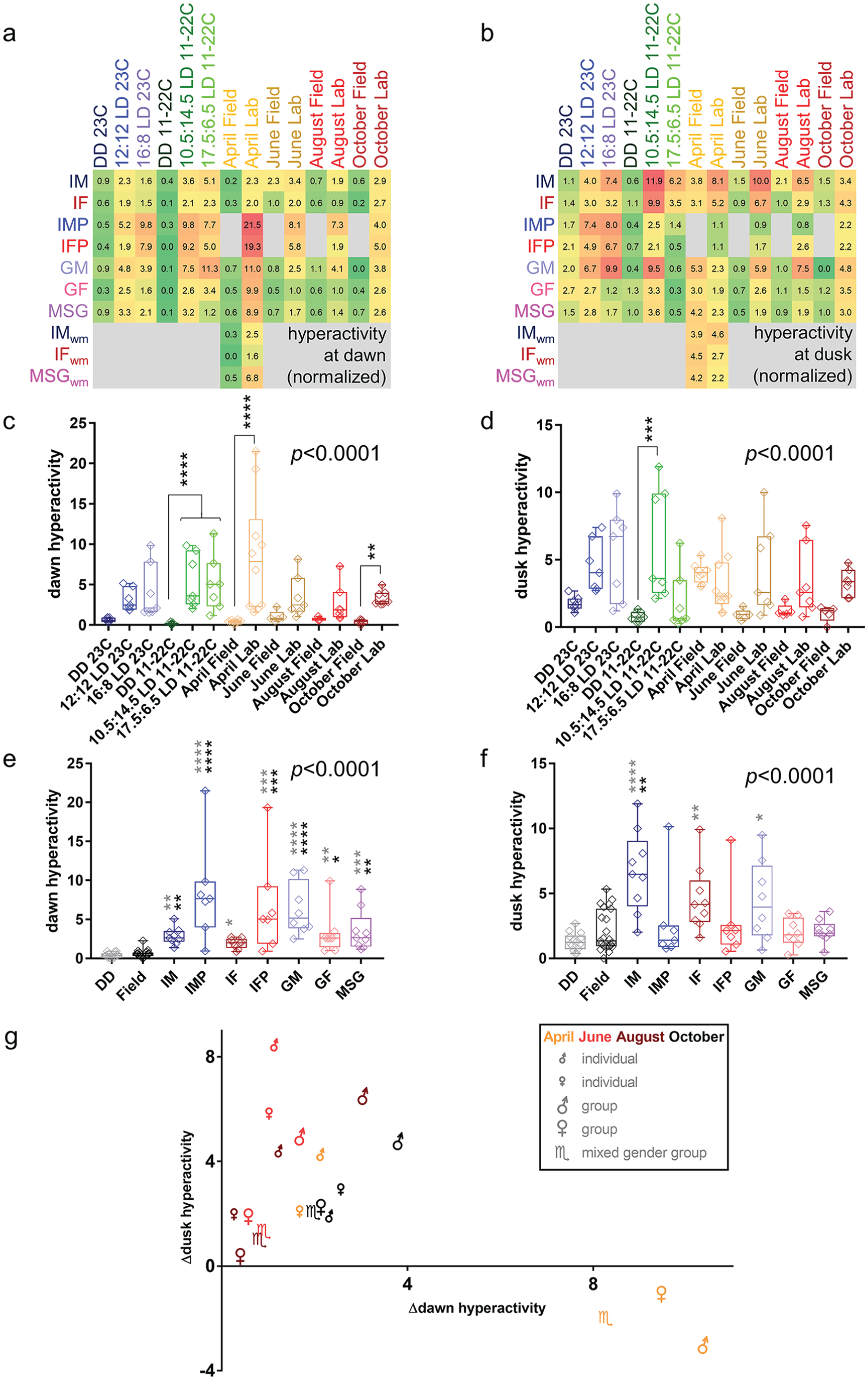
Impact of environmental and social conditions on normalized D. suzukii dawn and dusk hyperactivity. (a, b) Heat maps representing normalized hyperactivity at (subjective) dawn (a) or dusk (b) across different environmental and social settings. Missing conditions are indicated in gray. (c-f). Normalized hyperactivity at (subjective) dawn (c, e) or dusk (d, f) plotted as a function of environmental protocol (c, d) or social setting (e, l, f). Graphs are labeled with Kruskal-Wallis p values indicating the statistically significant effect of environment or social setting. Pairwise comparisons using Dunn’s post hoc tests in (c) and (d) are annotated for matching LD versus DD protocols as well as for associated semifield versus laboratory mimic conditions, while pairwise comparisons in (e) and (f) use either the lab DD or collective semifield data sets as control in comparison with data for non-DD lab conditions with the specified social settings. IM = individual males; IMP = individual males in population monitors; IF = individual females; IFP = individual females in population monitors; GM = group males; GF = groups of females; MSG = mixed-sex groups. *p < 0.05. **p < 0.01. ***p < 0.001. ****p < 0.0001. Black and gray typeface for the * annotation in (e) and (f) indicate a significant difference relative to DD and semifield conditions, respectively. (g) Distribution of different social setting/lab mimic protocol combinations based on their divergence from matching semifield data in normalized hyperactivity at light/dark transitions. Colors indicate environmental conditions and symbols indicate social setting, with groups indicated by larger symbols as annotated. Only summer morph flies were used in this analysis.
The effect of social housing on hyperactivity at light/dark transitions was illustrated when data were collated by social, rather than environmental, conditions (Fig. 3e, f), resulting in considerably more significant pairwise differences (Table 4). Typically, more differences occurred in comparison with DD and semifield hyperactivity at dawn than at dusk. Single male and single female flies in narrow cuvettes and group males showed significantly increased hyperactivity at both dawn and dusk in laboratory-based photocycles compared with either DD or semifield conditions. Differences were less consistent for other social settings.
Significant differences in dawn- or dusk-associated hyperactivity for lab assays using various social settings.
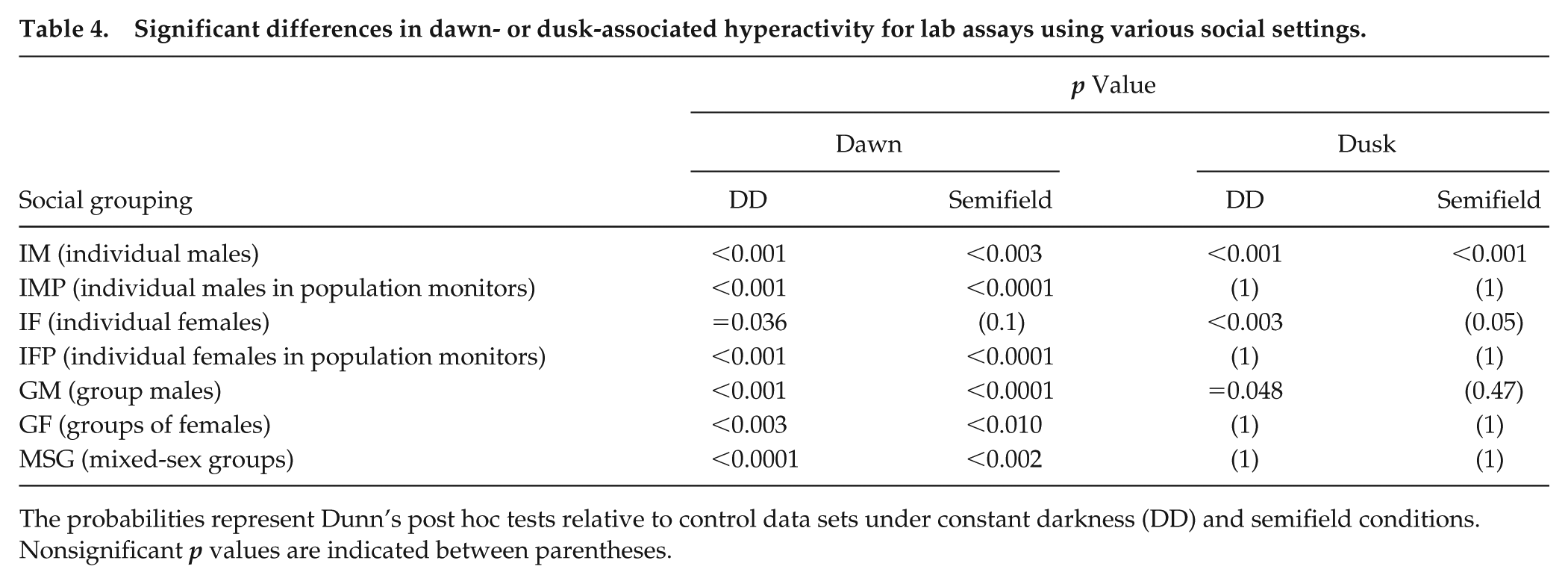
The probabilities represent Dunn’s post hoc tests relative to control data sets under constant darkness (DD) and semifield conditions. Nonsignificant p values are indicated between parentheses.
A more detailed representation of laboratory lighting-induced hyperactive episodes across different social settings and semifield paradigms is provided by the scatter plot in Fig. 3g. Social setting/environmental cycle combinations were plotted according to observed differences in normalized hyperactivity at dawn (x axis) and dusk (y axis) between semifield and matching laboratory mimic conditions, and so more successful re-creations of semifield conditions would be found closest to the origin of the graph. For June, August, and October, laboratory mimic conditions of group female and mixed-sex group conditions performed best in this regard, while the profile for individual females was closest to the semifield profile observed in April. Typically, individual males and groups of males were found on the periphery, indicating more divergence between laboratory and semifield behavior (Fig. 3g).
Impact of Social Housing on Timekeeping Ability in Constant Conditions
Monitoring D. suzukii in the absence of light and temperature cycles revealed a significant impact of social condition on daily locomotor activity patterns, rhythmic strength, and period length. Actograms displaying daily activity patterns vary greatly between social groupings, with the weakest rhythmicity in individual males and the strongest in group-housed females and mixed-sex groups (Fig. 4). Individual males and individual females assayed in narrow tubes had high percentages of arrhythmic flies and weakly rhythmic flies (Fig. 4b). In fact, 0 of 50 individual males in this condition were classified as “rhythmic.” Since the number of flies contributing to a single behavioral profile for group-housed individuals was 10-fold or 20-fold larger than that of individual flies, we considered that pooling the data from matching numbers of individually housed flies might improve the subsequently detected circadian rhythmicity. However, when virtual groups of males and females were created from individually housed fly data to address this point, no significant improvement in rhythmicity was observed relative to the original data sets (Fig. 4b). Significantly increased levels of rhythmicity were found for group-housed flies relative to individually housed flies (Fig. 4b, d; Table 5). Circadian period length was also affected and showed a much wider variation in measurements from individual rather than groups of flies (Fig. 4c; Table 5). Moreover, the weakly rhythmic individual males showed a circadian period that was longer than that of group-housed males or females (Fig. 4c; Table 5). The bulk of individual fly assays was conducted in narrow vials, and it was therefore of interest to determine whether the difference in vial size might have contributed to the observed differences in rhythmicity between individually and group-housed flies. This was addressed by conducting experiments using individual males and females in population-sized vials. Of note, individual males in larger, population-sized vials exhibited a significant shift in their distribution across categories of rhythmicity (Fig. 4b), indicating that confinement to narrow vials negatively affected circadian behavior in individual males. However, this was not the case for female flies, and group-housed females showed increased rhythmicity versus their individually housed counterparts, regardless of the vial size used for the latter (Fig. 4b, d).
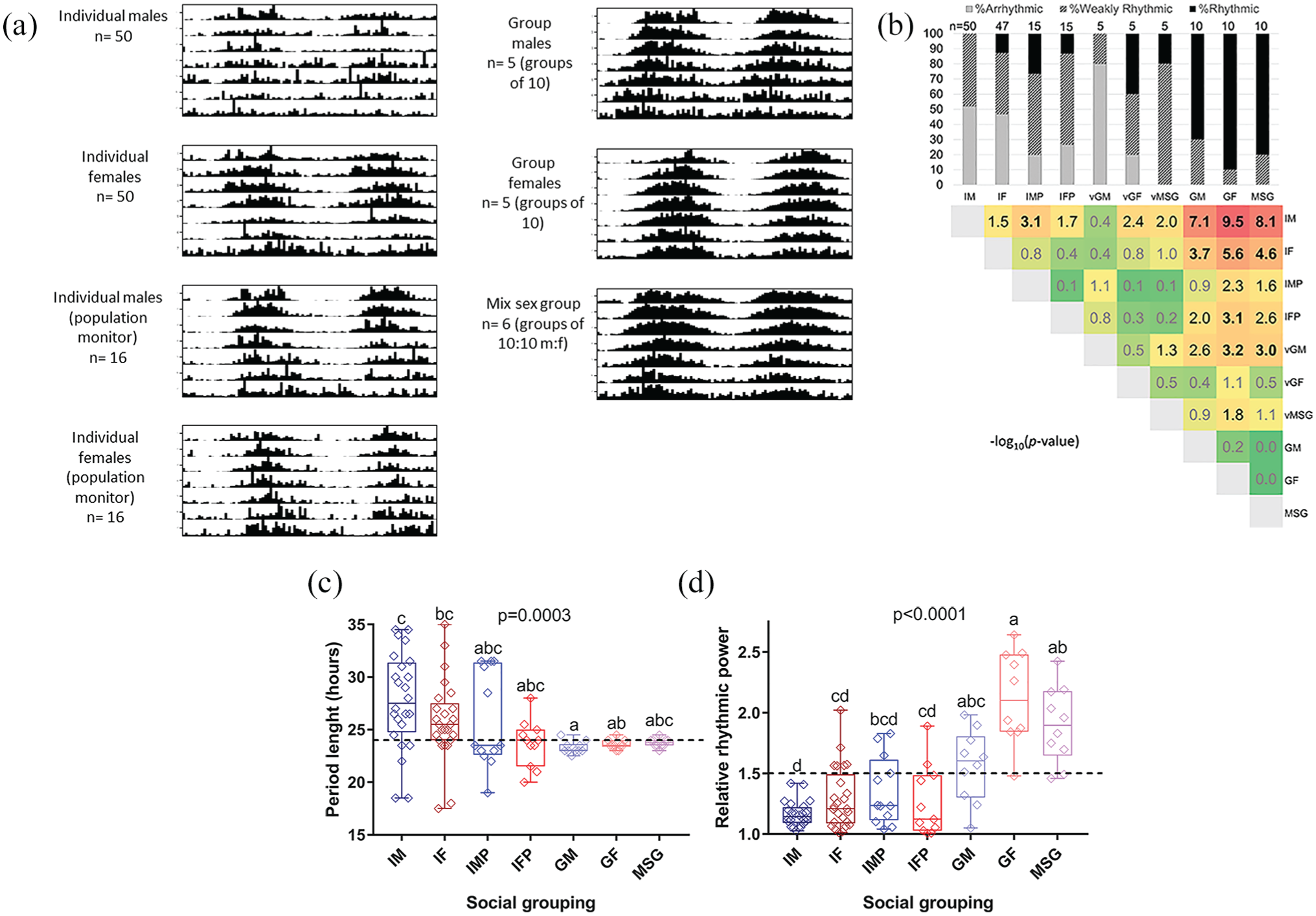
Impact of social setting on D. suzukii circadian behavior under constant conditions. Prior to the analyzed DD 23 °C condition, flies were entrained to a 12:12 LD 23 °C cycle. (a) Double-plotted actograms collected over a 7-day period in 30-min bins. (b) Top panel: distribution of DD rhythmicity for each social setting across 3 categories: arrhythmic, weakly rhythmic, and rhythmic (see the Materials and Methods section). Bottom panel: heat map of –log10 (p value) scores representing the outcome of pairwise 2 × 3 Fisher’s exact tests for differences in distribution across rhythmicity categories between different social settings. Values representing nonsignificant and significant p values are indicated in gray and black typeface, respectively, with values representing significant differences after Bonferroni multiple testing correction indicated in bold. (c, d) Graph of circadian period length and relative rhythmic power, respectively, as a function of different social settings. Dashed line denotes 24-h length and 1.5 relative rhythmic power. Kruskal-Wallis p values for the effect of social setting on each of these parameters are printed on the graphs, and significant pairwise differences in post hoc tests are annotated as well. IM = individual males; IF = individual females; IMP = individual males in population monitors; IFP = individual females in population monitors; VGM = virtual group males; VGF = virtual group females; VMSG = virtual mixed-sex groups; GM = group males; GF = groups of females; MSG = mixed-sex groups. Only summer morph flies were included in this analysis.
Mean (±SEM) of relative rhythmic power (RRP) and period length (hours) for social groupings held under constant darkness and constant temperature conditions (DD 23 °C).
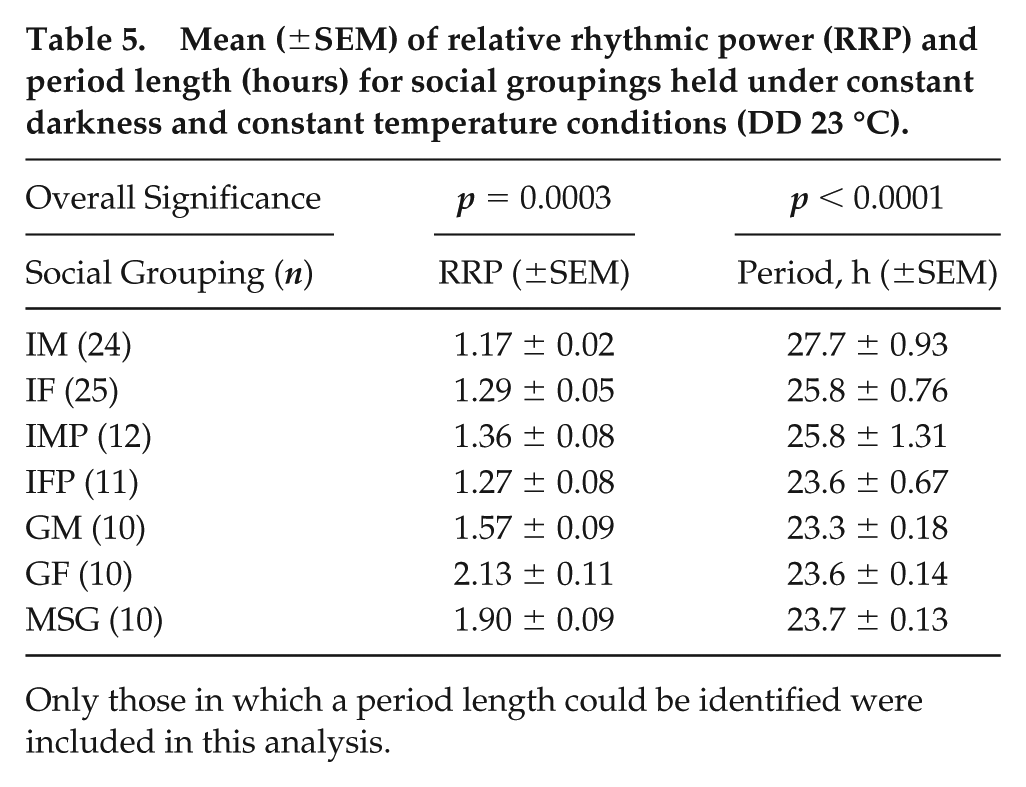
Only those in which a period length could be identified were included in this analysis.
Impact of Social Setting and Environment on Activity Rate
There were significant differences in the average activity counts per hour per fly in relation to environmental condition and social housing (Table 6). Group females displayed the highest activity counts per hour per fly in 5 of the 14 environmental conditions, the highest of any social grouping. Individual females in population monitors displayed the highest counts in 3 of the 6 environments in which they were monitored. Social conditions containing females showed, typically, higher than average activity counts per hour per fly. Individual females, groups of females, and mixed-sex groups had higher than average activity counts per hour per fly in 9, 10, and 8 of the 14 conditions, respectively. Individual males and group males displayed activity counts lower than the mean in 11 and 9 of the 14 environmental conditions, respectively. Virtual groups were created to define whether variation in activity counts was attributed to fly density and the result of flies disturbing one another within the assay or whether individual flies could be analyzed as groups and gain the same results as real groups. This would mean that if only individual fly data were available, it could be converted to give an indication of how groups of flies would behave. Different social groupings exhibited significant differences in average activity per hour per fly for almost all environmental conditions, the only exception being DD 23 °C. Further, average activity levels observed for winter versus summer morph flies showed no significant difference in individual male behavior or individual female behavior. However, mixed-sex group activity counts differed between morph in the April semifield conditions, with summer morph flies displaying higher activity counts.
Mean (±SEM) D. suzukii activity counts per hour per fly as a function of social setting (rows) and environmental condition (columns).
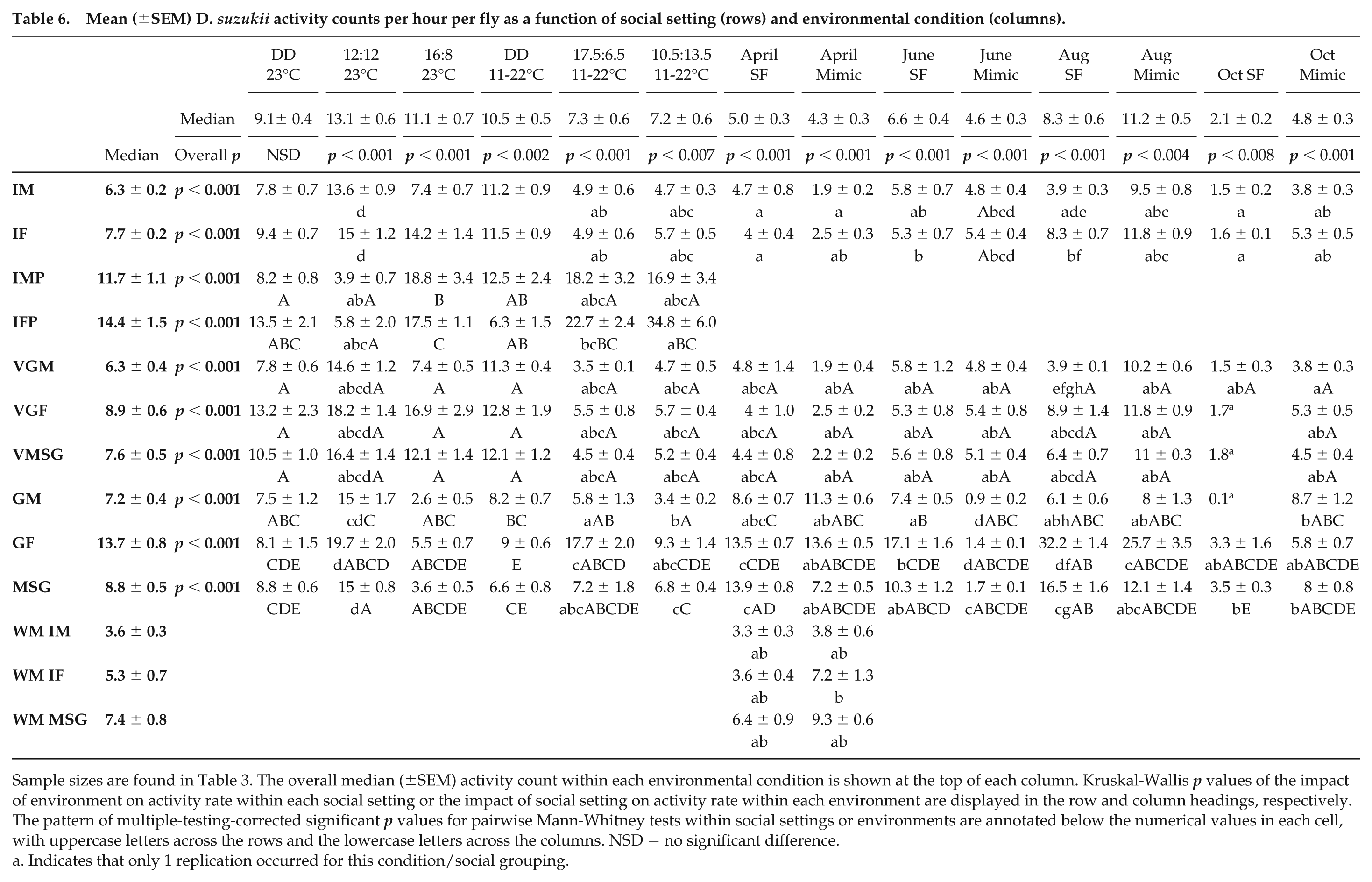
Sample sizes are found in Table 3. The overall median (±SEM) activity count within each environmental condition is shown at the top of each column. Kruskal-Wallis p values of the impact of environment on activity rate within each social setting or the impact of social setting on activity rate within each environment are displayed in the row and column headings, respectively. The pattern of multiple-testing-corrected significant p values for pairwise Mann-Whitney tests within social settings or environments are annotated below the numerical values in each cell, with uppercase letters across the rows and the lowercase letters across the columns. NSD = no significant difference.
Indicates that only 1 replication occurred for this condition/social grouping.
Discussion
We examined the impact of environmental and social parameters on the daily locomotor activity profile of the horticultural pest species D. suzukii with a particular interest in re-creating and predicting behavioral patterns encountered in the field. The behavior of mated female flies was of particular interest given their role in mediating damage to fruit and the relative scarcity of virgin females in the field (Revadi et al., 2015). Thus, despite the fact that the mating state of D. suzukii influences patterns in locomotor behavior in D. suzukii (Ferguson et al., 2015), all observations made in this study used mated flies, in both individual and social housing groups. Prior analyses of D. suzukii locomotor activity have been reported for laboratory and seminatural light conditions (Hamby et al., 2013; Ferguson et al., 2015), while 2 more studies inferred diurnal activity in the field based on trapping results (Evans et al., 2017) or visual detection (Van Timmeren et al., 2017). However, the present study is, to our knowledge, the first for this species to both document high-resolution daily activity profiles in a semifield setting as well as systematically address the contributions of temperature, light, and social groupings on daily locomotor activity.
In both laboratory- and field-collected profiles, most activity occurred during the daytime, and very little occurred during the night. This also matches observations for oviposition rhythms in the laboratory and field (Shaw et al., 2018b). Moreover, subjective day-active patterns were found under constant dark conditions. However, it is important to note that the temperature cycles applied in this study did not include noxiously warm temperatures. In a study that did include high (>30 °C) temperature maxima, Ferguson et al. (2015) reported crepuscular behavior for D. suzukii faced with simulated summer conditions in the laboratory, while field observations made on similarly warm days likewise suggested a crepuscular activity pattern (Evans et al., 2017; Van Timmeren et al., 2017). Therefore, we hypothesize that D. suzukii daily locomotor activity patterns may seasonally switch from unimodal diurnal profiles to crepuscular, as temperature maxima reach noxiously high levels. Indeed, this hypothesis is consistent with previously observed temperature-disrupted, D. suzukii oviposition patterns (Shaw et al., 2018b), and seasonal variation of activity peaks in Drosophila melanogaster Meigen under seminatural conditions (Menegazzi et al., 2013).
Although anticipatory behavior is seen as a key indicator of the presence of a functioning circadian clock (Stoleru et al., 2004), in our assays we found that social settings that favored increased evening anticipation under a 12-h or longer photoperiod (individual males or females in population vials) showed weaker rhythms under constant conditions than groups of females and mixed-sex groups that showed very little to no anticipation of lighting evenings in laboratory conditions.
The impact of daily temperature cycles on D. suzukii locomotor activity was apparent from the increased amplitude in rhythm upon the introduction of a temperature gradient in constant darkness. Moreover, the daily activity profiles of flies under June, August, and October semifield conditions appeared to track the daily temperature profiles. However, the April temperature cycle, which featured a longer interval at or near peak temperature, resulted in a delay of maximal daily activity by an average of 4 h across all social groups. The same was also true for the winter morph flies under the April semifield conditions. In addition, as Drosophila are ectothermic, it may be a form of behavioral thermoregulation used to heighten physiological performance in colder conditions (Dillon et al., 2009). These features were recapitulated relatively well when the semifield temperature and light cycles were mimicked in the laboratory, although the artificial lighting conditions triggered acute bursts of activity following the light/dark transitions. This hyperactivity at light transitions, termed masking or startle response, has been observed previously in D. melanogaster (e.g., Sheeba et al., 2010) and is thought to represent behavioral responses to changes in illuminance that shroud or mask underlying circadian behavior (Mrosovsky, 1999; Allada and Chung, 2010). We found that the hyperactivity responses observed in lab assays of D. suzukii were clearly influenced by the social housing condition, with female and mixed-sex groups displaying reduced hyperactivity and showing the most accurate behavioral representations of June and August semifield conditions when subjected to the laboratory mimic conditions. The absence of similar bouts of acute hyperactivity in semifield conditions suggests that the abrupt changes in illuminance in the lab are responsible for this phenomenon. While both lab and semifield conditions featured comparable maximum illuminance levels (2670 lux in the lab versus 2746 lux in the August semifield setting), the changes in illuminance were much more gradual in the field.
Our analyses across environmental conditions provided evidence that sex, space constraints, and social interactions all influenced locomotor behavior. Apart from their impact on hyperactivity at light/dark transitions, social settings also affected behavioral rhythmicity under constant conditions for group-housed flies. Within this project, individual males and females had the highest percentage of arrhythmic counts in constant conditions and had a much lower RRP than group-housed flies. The impact of social interactions on rhythm maintenance may be related to prior findings in D. melanogaster (Bloch et al., 2013), in which social olfactory cues were found to be capable of regulating or resetting circadian rhythms in the same way environmental cues do (Levine et al., 2002). In D. melanogaster, Lone and Sharma (2011) concluded that social grouping resulted in an increase of phase synchrony in comparison with individual flies or pairs of flies under constant darkness after prior entrainment to an L:D cycle. They also found that under constant darkness, housing mutant flies with loss of olfaction function in groups did not result in rhythm maintenance, supporting the idea that it is an olfactory signal that is driving this response. It will be interesting to probe the relevant sensory input and neural pathways in this regard. The broader relevance of observing behaviors under social conditions has been promoted by several groups who observed that interaction between individuals will affect behavior, not only in species that have a complex social structure, such as the honeybee, but also in less socially complex systems, including D. melanogaster (Bloch et al., 2013; Fujii et al., 2007). In particular, prior studies have established an impact of social interactions on activity rhythms in the honey bee (Beer et al., 2016; Moore et al., 1998) and both activity rhythms and mating behavior in D. melanogaster (Fujii et al., 2007; Krupp et al., 2008; Levine et al., 2002). Mating and courtship in D. suzukii involve mobile rituals of wing flashing and circling and would surely increase the activity levels in populations of flies (Revadi et al., 2015). De et al. (2013) modified lighting factors “under otherwise semi-natural” conditions and monitored the locomotor behavior of D. melanogaster, to determine how activity was affected by changes in light. They also hypothesized that there would be changes in activity due to courtship and concluded that the morning peak of activity in D. melanogaster is due to “courtship-related locomotion.” Within our assay, individual flies housed in the narrow cuvettes frequently displayed a different locomotor profile than groups in the large population vials. Although, unlike De et al. (2013), we do not see variation in morning activity peaks between individual and group-housed flies under the semifield collected locomotor profiles in D. suzukii. However, we did see this variation in the laboratory collected profiles.
Comparison of the behavior of individual flies housed in narrow tubes versus population vials in the lab revealed a number of differences. These included changes in the balance between hyperactivity at lights-on versus lights-off. Moreover, circadian rhythmicity under constant conditions was reduced for males in narrow tubes. Virtual groups were also created from matching numbers of individual fly data sets to demonstrate that the increased circadian rhythmicity of group-housed flies was not simply due to an increased signal-to-noise ratio in multifly records. Instead, social interactions rather than vial size or pooled activity levels were implicated as driving increased circadian rhythmicity in D. suzukii.
For each social grouping, changes in environmental conditions triggered changes in the activity rate per fly. Conversely, social groupings affected the activity rate per fly for all environmental conditions, save one: constant 23 °C in DD. When evaluating social housing alone, typically individual males and groups of males had lower than average counts per hour per fly than any housing that contained females, both as individuals or in single- and mixed-sex groups. We considered that having a higher density of flies in mixed-sex groups (20 individuals) would result in disturbances and therefore higher activity counts than in the single-sex groups (10 individuals) or the individual flies. This hypothesis was investigated in an extensive examination of social interactions and its effect on sleep in D. melanogaster, conducted by Liu et al. (2015). Patterns of individual mated flies were compared with single-sex groups of various densities, and variations in sleep habits were detected. The researchers found that total sleep decreased drastically as population density increased. In our assay, groups of females, which contained 10 individuals, displayed the highest activity counts per hour per fly in 5 of the 14 conditions. This was higher than any other social grouping, including the mixed-sex groups containing 20 individuals per vial, which, based on the interpretation by Liu et al. (2015), would have been expected to exhibit the highest activity counts. The increased activity per fly per hour in group females may be due to females searching for additional egg-laying sites, as D. suzukii prefer less dense sites compared with D. melanogaster (Mitsui et al., 2006). This increase in activity could also be driven by D. suzukii females trying to reduce oviposition competition between themselves and conspecifics. (Shaw et al., 2018a).
The present study also included a limited comparison between the behavior of summer and winter morph flies. The locomotor behavior of flies exposed to April semifield or April laboratory mimic conditions displayed minor shifts in average activity level between winter and summer morph flies, with winter morph flies relatively less active in the field but more active in the laboratory. Perhaps an environmental difference between the 2 settings, such as the gradual light/dark cycle or the presence of a relative humidity cycle in the field condition, might explain these relatively modest differences. Nevertheless, the wave forms of daily activity patterns observed for summer morph versus winter morph flies were similar. Thus, under the conditions tested, summer morph behavior was also representative of winter morph behavior. From a practical approach, this is a welcome result, as culturing significant numbers of winter morph flies is time-consuming and logistically challenging. As winter morph flies are in reproductive diapause (Rossi-Stacconi et al., 2016; Toxopeus et al., 2016; Wallingford and Loeb, 2016), cultures are not continuous and need frequent replenishment.
Within this research, we have investigated locomotion under both semifield and laboratory settings. Ideally, to predict natural behaviors, observations should be made under (semi-)field conditions (Kannan et al., 2012). However, there are obvious limitations and restrictions to working in natural conditions. The reproducible control environment provided by the laboratory enables researchers to replicate parameters and provides second chances, which would not occur in the field as no 2 days are the same. Also, being able to manipulate conditions enables investigations at any time opposed to waiting for environmental conditions to occur naturally in the field. Where semifield-based assays are not appropriate, controlled environment chambers with gradual changes in light and temperature are a good substitute for investigating physiological behaviors, such as diapause (Nagy et al., 2018). We did not attempt to manipulate relative humidity within the semifield environments, whereas in the laboratory, it was maintained at 65%. As a result, we were able to dissociate the effects of light and temperature from those of humidity while promoting a high level of survival (Tochen et al., 2016; Hamby et al., 2016). Yet, despite this artificial feature, and the use of fluorescent rather than dimmable lights, we were able to successfully mimic locomotor behavior found for summer semifield conditions in the laboratory.
Conclusion
The main aim of this article was to identify the determinants of daily rhythms of D. suzukii activity that may help predict field behavior. We demonstrated clear separable impacts of the circadian clock, light, temperature, and social housing on D. suzukii behavioral rhythms. By comparing laboratory-based and semifield locomotor assays, we have investigated a wide range of environmental conditions to identify those that would be most appropriate for making behavioral predictions in the laboratory. Drosophila locomotor activity assays are most commonly conducted for individual male flies in laboratory conditions. However, in our analyses, mixed-sex or mated female groups showed stronger circadian rhythmicity and were more amenable to the re-creation of semifield behavior in the laboratory. Using these social settings, summer semifield conditions were successfully reproduced in incubators with fluorescent lights programmed to provide a realistic daily temperature cycle and photoperiod at constant humidity. As the behavior of mixed-gender populations is also of more direct concern to integrated pest management strategies aimed at minimizing harm to soft and stone fruit cultures by wild D. suzukii populations, we propose that future studies of natural locomotor behavior should preferentially focus on flies in this social setting.
Supplemental Material
Supplemental_Figure_S1_300dpi_1 – Supplemental material for Control of Daily Locomotor Activity Patterns in Drosophila suzukii by the Circadian Clock, Light, Temperature and Social Interactions
Supplemental material, Supplemental_Figure_S1_300dpi_1 for Control of Daily Locomotor Activity Patterns in Drosophila suzukii by the Circadian Clock, Light, Temperature and Social Interactions by Bethan Shaw, Michelle Fountain and Herman Wijnen in Journal of Biological Rhythms
Footnotes
Acknowledgements
We thank the University of Southampton and the Agricultural and Horticultural Development Board for funding this project. We also thank Charlie Hurdle and Miguel Ramirez Moreno for technical advice and support.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
