Abstract
Hibernation is a naturally occurring model for studying diseases such as obesity and osteoporosis. Hibernators, marmots (Marmota flaviventris) among them, are able to nearly double their body mass by increasing fat stores prior to hibernation without the negative consequences of obesity. They are also physically inactive for extended periods of time without experiencing negative effects on the skeleton. The endocannabinoid system is involved in modulating neural signaling, circannual rhythms, behavior, appetite, thermogenesis, and bone and energy metabolism. These systems are also altered to maintain homeostasis during hibernation. This study aims to better understand the involvement of the endocannabinoid system in the regulation of physiological processes during hibernation by quantifying the seasonal variation of endocannabinoids and endocannabinoid-like ligands in both active and hibernating marmots. We hypothesized that there would be significant changes in endocannabinoid concentrations at the tissue level in marmots between active and hibernating states. Concentrations were measured in brain, serum, brown adipose tissue, white adipose tissue, bone marrow, cortical bone, and trabecular bone using microflow chromatography coupled with tandem quadrupole mass spectrometry. Significant changes were found, such as a 30-fold decrease in 2-arachidonoyl glycerol (2-AG) in cortical bone during hibernation. Many endocannabinoid and endocannabinoid-like ligands decreased in brown adipose tissue, white adipose tissue, and cortical bone, while several ligands increased in bone marrow. This result supports our hypothesis and suggests the possibility of a peripherally controlled shift in energy metabolism, reduction in bone metabolism, and suppression of the immune system during hibernation.
The endocannabinoid system includes numerous endogenous, lipid-derived ligands and their g-protein coupled receptors. The two dominant endocannabinoids, 2-arachidonoyl glycerol (2-AG) and anandamide (AEA), are synthesized and degraded on demand by intracellular enzymes (Battista et al., 2012). Endocannabinoids are synthesized from membrane glycerophospholipids and are largely a product of dietary fatty acids, such as the polyunsaturated fatty acid (PUFA) linoleic acid (Wang and Ueda, 2009; Naughton et al., 2013). These endocannabinoids primarily bind to cannabinoid receptors CB1 and CB2 located in various tissues but also interact with peroxisome proliferator-activated receptor (PPAR) transcription regulators and other receptors (Hansen et al., 2006; Ross, 2009; Di Marzo and De Petrocellis, 2010). The molecule 1-arachidonoyl glycerol (1-AG) is also produced endogenously and is the inactive isomer of 2-AG (van der Stelt et al., 2002). Endocannabinoid-like compounds N-palmitoylethanolamine (PEA) and N-oleoylethanolamine (OEA) are structurally similar to AEA and follow the same pathway of synthesis and degradation. PEA and OEA are not able to bind directly to cannabinoid receptors but are thought to interfere with the degradation process of true endocannabinoids like 2-AG and AEA (De Petrocellis and Di Marzo, 2009). PEA and OEA lead to appetite reduction and have anti-inflammatory effects, respectively, through activation of PPAR-α (De Petrocellis and Di Marzo, 2009). Just as with their receptors, endocannabinoid and endocannabinoid-like ligands themselves are found and synthesized in numerous tissues throughout the body.
The endocannabinoid signaling system is known to be involved in the modulation of diverse physiological processes and rhythms such as neural signaling, circannual rhythms, thermoregulation, appetite, and bone and energy metabolism and diseases such as cancer (Carey et al., 2003; Bellocchio et al., 2008a; Pagano et al., 2008; Maccarrone et al., 2015; Bogren et al., 2016; Prospero-Garcia et al., 2016; Korf, 2017). The endocannabinoid system influences regulation of bone remodeling through various avenues. Activation of CB2 receptors located on bone cells has been found to promote bone formation by increasing osteoblast proliferation and activity (Maccarrone et al., 2015). However, thoughts on the role of CB2 in the regulation of osteoclast activity remain highly conflicting (Idris and Ralston, 2012). CB1 receptors interface between bone and the central nervous system. Sympathetic nerve endings express CB1 receptors that inhibit norepinephrine release when activated by 2-AG synthesized by local osteoblasts (Maccarrone et al., 2015). Norepinephrine is responsible for both decreasing bone formation and increasing resorption (Maccarrone et al., 2015). CB1 receptors are the most abundant g-coupled protein receptor in the brain and are able to promote increased appetite and hypothermia when stimulated in the hypothalamus (Bellocchio et al., 2008a; Pagano et al., 2008). Endocannabinoids are involved in the sleep cycle as well. 2-AG and AEA promote non-rapid-eye movement sleep and, through interaction with melanin-concentrating hormone neurons, also promote rapid-eye movement sleep (Prospero-Garcia et al., 2016). Alternatively, OEA and PEA promote wakefulness (Prospero-Garcia et al., 2016). 2-AG has even been shown to play a crucial role in seasonal reproductive cycles (Korf, 2017). Evidence of regulation of energy metabolism by the endocannabinoid system is also shown in adipose tissue. Interaction of endocannabinoids with PPAR-γ is thought to drive an increase in adipogenesis (Bellocchio et al., 2008a; Pagano et al., 2008). The formation of adipocytes expands energy storage capacity. Therefore, endocannabinoids promote not only the intake of energy through increased appetite but also the storage of energy in adipose tissue. CB1 receptors have been found in differentiated adipocytes, but the expression of CB2 is still debatable (Pagano et al., 2008). Stimulation of CB1 by CB1-selective agonists on fat cells has shown increased lipoprotein lipase activity, accumulation of intracellular lipid droplets, and glucose uptake (Pagano et al., 2008). These factors contribute to the production and storage of fat. Much like the opposing effects on sleep cycle in the brain, OEA induces a response opposite to that of true endocannabinoids in adipose tissue. OEA diminishes desire for food intake and promotes lipolysis (Pagano et al., 2008). OEA and PEA both have high affinity for PPAR-α (Pagano et al., 2008). In brown adipose tissue (BAT) specifically, endocannabinoids promote the decrease of thermogenesis (Bellocchio et al., 2008a). Although these systems outwardly demonstrate routine functions, they may also have a role in driving other specialized physiological processes such as hibernation.
Interestingly, the physiological processes regulated by the endocannabinoid system are altered to maintain homeostasis during hibernation. The seasonal rhythm of hibernation in rodents is characterized by torpor bouts lasting several days to approximately 5 weeks in certain species, drastically lowering body temperature to 4 to 6 °C and metabolism to 2% to 5% of summer basal metabolic rates, interrupted by interbout arousal periods lasting 12 to 24 h (Carey et al., 2003; Bogren et al., 2016). Some species of hibernators, such as marmots (Marmota flaviventris), must rely on lipolysis of white adipose tissue (WAT) as a sole energy source during the hibernating season since feeding does not occur during this period (Carey et al., 2003; Humphries et al., 2003). This means that there would no longer be dietary intake of PUFAs as a source for endocannabinoids. PUFAs are also preferentially retained in lipid stores during hibernation over saturated and monounsaturated fatty acids (Florant et al., 1990). The summer season is spent undergoing extreme hyperphagia to increase WAT stores for hibernation (Carey et al., 2003). BAT is used by some species of hibernators in nonshivering thermogenesis to aid in the onset of rewarming during interbout arousals but is metabolically suppressed during deep torpor (Milner et al., 1989; Biggar and Storey, 2014). Another proposed function of BAT is to stimulate bone anabolism (Rahman et al., 2013; Lidell and Enerback, 2015). Mammals that hibernate have adapted physiological mechanisms to prevent the negative effects of inactivity such as osteoporosis, hypercalcemia, and obesity-related issues. Studies have found that humans and other animals experience bone loss due to an unbalanced remodeling process that degrades bone with disuse (Leblanc et al., 1990; Kaneps et al., 1997). In contrast, hibernators have evolved unique physiological mechanisms to survive extreme environmental conditions (Doherty et al., 2015), such as suppressing bone remodeling and preserving bone structural, mineral, and mechanical properties during periods of physical inactivity lasting up to 8 months (Donahue et al., 2006; McGee-Lawrence et al., 2008; McGee-Lawrence et al., 2015; Doherty et al., 2016). Reduced bone turnover also allows hibernators to conserve energy by reducing the metabolically expensive processes of bone remodeling. This unique trait of suppressed bone remodeling with physical inactivity has sparked investigation of changes in the endocrine system (Doherty et al., 2014) and the bone proteome (Doherty et al., 2016) with respect to hibernation.
Of 506 identified proteins in marmot bone, monoacylglycerol lipase (MAGL) was found to show the greatest difference in abundance between summer active and hibernating marmots; MAGL was 98-fold higher in the bones of hibernating marmots compared with summer marmots (Doherty et al., 2016). MAGL is the primary enzyme that degrades 2-AG. In addition to being found in bone, MAGL has been observed in many other tissues, including brain, adipose tissue, kidney, ovary, testis, adrenal gland, and heart (Karlsson et al., 1997). The current study quantified the concentrations of endocannabinoid system-affiliated ligands AEA, 2-AG, 1-AG, PEA, and OEA in hibernating and active marmots to help elucidate the role that the endocannabinoid system plays in regulating seasonal physiological changes associated with hibernation. Concentrations were measured using microflow chromatography coupled with tandem quadrupole mass spectrometry (LC-QQQ-MS) in bone, bone marrow, serum, brain, and adipose tissues. We hypothesized that there would be significant changes in endocannabinoid concentrations at the tissue level of marmots during hibernation due to the known degradative role of MAGL in brain and peripheral tissues (Karlsson et al., 1997), as well as the need to regulate an array of physiological processes during the extreme metabolic conditions of hibernation.
Materials and Methods
Sample Collection
Eight wild yellow-bellied marmots (M. flaviventris) were captured in the Colorado Rocky Mountains during the summer and housed at Colorado State University, under IACUC approved protocol No. 15-5721A, in a specialized facility that provided natural light and temperature conditions similar to those in the wild. During the summer (May-September), housing temperature was kept at approximately 20 °C, and then it was gradually transitioned to 4 °C and continuous darkness for the winter. The animals were provided food and water ad libitum. The marmots entered hibernation by late October. The torpor bouts of the hibernating marmots were synchronized 6 weeks prior to the sacrifice by handling the animals at room temperature for multiple hours. Body temperatures were taken every few days after synchronization to follow torpor bouts. An infrared thermometer (Cen-tech, model #61894; Harbor Freight Tools Co., Calabasas, CA) with an accuracy of ±4.5% was used to measure temperature between the shoulder blades. The hibernating marmot group was sacrificed on 27 February 2015 starting approximately 1½ h after sunrise, from 0800 h to 1100 h. The hibernating group contained 2 males and 2 females with weights ranging from 1.32 to 2.90 kg and body temperatures ranging from 7.8 to 8.6 °C. The active marmot group was sacrificed during prehibernation on 2 October 2015 starting approximately 1 h after sunrise, from 0800 h to 1100 h. The active group was made up of 2 males and 2 females, with weights ranging from 2.30 to 5.35 kg and temperatures near 37 °C. Prior to sacrifice, the prehibernation group was anesthetized with isoflurane. All animals were euthanized by exsanguination via cardiac puncture.
During necropsy, samples of BAT, WAT, brain, serum, fourth metatarsal bone, and bone marrow were collected. The tissue samples and respective collection locations are summarized in Table 1. The bone marrow was separated from the bone by first cutting off both bone ends, leaving only the diaphysis. Then, a water pick was inserted into one end of the diaphysis, and methanol was used to flush the bone marrow through the other end of the diaphysis for collection. Bone collected from the diaphysis served as samples of pure cortical bone. Both the metaphysis and epiphysis were included in the bone ends. The bone ends were too small to separate tissue types for collection of pure trabecular bone samples. Therefore, the bone end samples analyzed included a mixture of trabecular, marrow, and cortical tissues. Blood was centrifuged to isolate the serum. All samples were flash frozen in liquid nitrogen and stored at −80 °C until sample preparation for mass spectrometry analysis.
Collected samples.

Endocannabinoids were measured in multiple tissues. Here, the specific sampling location for each tissue is summarized.
Sample Preparation for LC-MS
BAT, WAT, brain, and bone samples
The samples were processed for LC-MS using our established protocols (Kirkwood et al., 2016). Frozen samples were homogenized by mortar and pestle in the presence of liquid N2. Powdered homogenate was weighed in a 2-mL glass extraction vial (VWR Scientific, Radnor, PA). Tissue sample masses were as follows: 30 to 60 mg for adipose tissue, 25 to 50 mg for brain, 80 to 120 mg for bone. All homogenized tissue samples were stored at −80 °C until endocannabinoid extraction. Exact masses were recorded for each tissue sample, and the endocannabinoid levels are reported as endocannabinoid mass divided by tissue sample mass in units of pg/mg for adipose, brain, and bone samples.
Serum and bone marrow samples
Serum and bone marrow exudates were thawed on ice. For each sample, 0.05 mL of serum and 0.50 mL of bone marrow exudate was placed into 2-mL glass extraction vials. Endocannabinoid levels for serum and bone marrow were reported in units of pg/mL.
All samples were subjected to biphasic extraction as previously described by Kirkwood et al. (2016). Briefly, 1 mL of cold toluene (–20 °C) containing 100 pg/mL of internal standards was added to each tissue, serum, and bone marrow sample. Samples were vortexed for 15 min at medium speed at room temperature. Biphasic extraction was induced with the addition of 0.4 mL of 3% formic acid in water. After protein precipitation overnight at −80 °C, the organic top layer was transferred to a new vial and concentrated to dryness under N2 (g). The dried extract was resuspended in 0.04 mL of 50:50 acetonitrile-methanol followed by 0.06 mL of water with 0.2% formic acid. For adipose tissue samples, the resuspended extract was centrifuged (2000 rpm, 4 °C, 10 min) to pellet residual lipids. The top layer of the resuspension was removed and placed into a new glass vial insert (Waters Corporation, Milford, MA) for LC-MS injection.
Calibration curve
A master mix solution of AEA, OEA, PEA, and 2-AG was mixed at a concentration of 10 mg/mL in 100% acetonitrile (ACN). The master mix was serially diluted by 2.5× in 100% ACN to form a set of standard samples with known concentrations; 50 mL of each standard dilution was extracted with 1 mL of toluene extraction solution as outlined above. The standards were used to map the linear relationship between peak area and concentration so that the unknown concentrations of the samples could be determined using peak area measurements.
Microflow liquid chromatography
All analytical standards and internal standards (AEA, OEA, PEA, AEA-d4, and 2-AG-d5) were purchased from Cayman Chemical (Ann Arbor, MI). Toluene, LC-MS grade acetonitrile, and LC-MS grade water were from Fisher Scientific (Lafayette, CO). LC-MS grade formic acid and ammonium acetate were from Sigma (St. Louis, MO) (Kirkwood et al., 2016).
LC-QQQ-MS was performed on a Waters M-Class Acquity UPLC coupled to a Waters Xevo TQ-S triple quadrupole mass spectrometer. Chromatographic separations were carried out on a Waters iKey separation device (150 µm × 50 µm, 1.7 µm BEH C18). Mobile phases were 99.9% acetonitrile, 0.1% formic acid (B), and water with 0.1% formic acid (A); 10 µL of sample was injected onto a C8 trapping column (Waters Atlantis C8, 300 mm × 50 mm, 5 mm) at 10% B and held for 2 min. The analytical gradient was as follows: time 0 min, 60% B; time 7 min, 70% B; time 8 min, 100% B; time 9 min, 100% B; time 10-14 min, 100% B; time 20 min, 60% B. Flow rate was time 0-8 min, 2 µL/min; time 9 min, 4 µL/min; time 10-14 min, 7 µL/min; time 15-20 min, 4 µL/min. Temperature of the iKey device was held at 55 °C from 0 to 9 min and then increased to 80 °C from 9 to 20 min. The iKey was cooled for 3.4 min before next injection. Total run time injection to injection was 26 min. Samples were held at 6 °C in the autosampler.
Tandem mass spectrometry
The MS was operated in selected reaction monitoring (SRM) mode, where a parent ion is selected by the first quadrupole and is fragmented in the collision cell, and then a fragment ion is selected by the third quadrupole. Product ions, collision energies, and cone voltages were optimized for each analyte by direct injection of individual synthetic standards. Interchannel delay was set to 3 msec. The MS was operated in positive ionization mode with the capillary voltage set to 3.6 kV. Source temperature was 120 °C, and desolvation temperature was 350 °C. Desolvation gas flow was 1000 L/h, cone gas flow was 150 L/h, and collision gas flow was 0.2 mL/min. Nebulizer pressure (nitrogen) was set to 7 bar. Argon was used as the collision gas.
LC-MS data analysis
All raw data files were imported into the Skyline open source software package (MacLean et al., 2010). Each target analyte was visually inspected for retention time and peak area integration. Peak areas were extracted for target compounds detected in biological samples and normalized to the peak area of the appropriate internal standard or surrogate in each sample. Normalized peak areas were exported to Excel, and absolute quantitation was obtained by using the linear regression equation generated for each compound from the calibration curve.
Limits of detection and quantitation
Limits of detection (LOD) and limits of quantitation (LOQ) were determined as previously described (Shrivastava, 2011; Broccardo et al., 2013) and normalized to tissue sample mass or serum volume. For those cases that fell below the LOD, the chromatograms were visually inspected for the presence of clearly defined peaks to determine whether the values were still within the sensitivity of the instrument. All values below the LOD had clearly defined peaks in the chromatograms and were therefore included in statistical analysis. Tissues with sample values below the LOD/LOQ are indicated in Tables 2 through 6.
Anandamide concentrations for tissues of active and hibernating marmots.
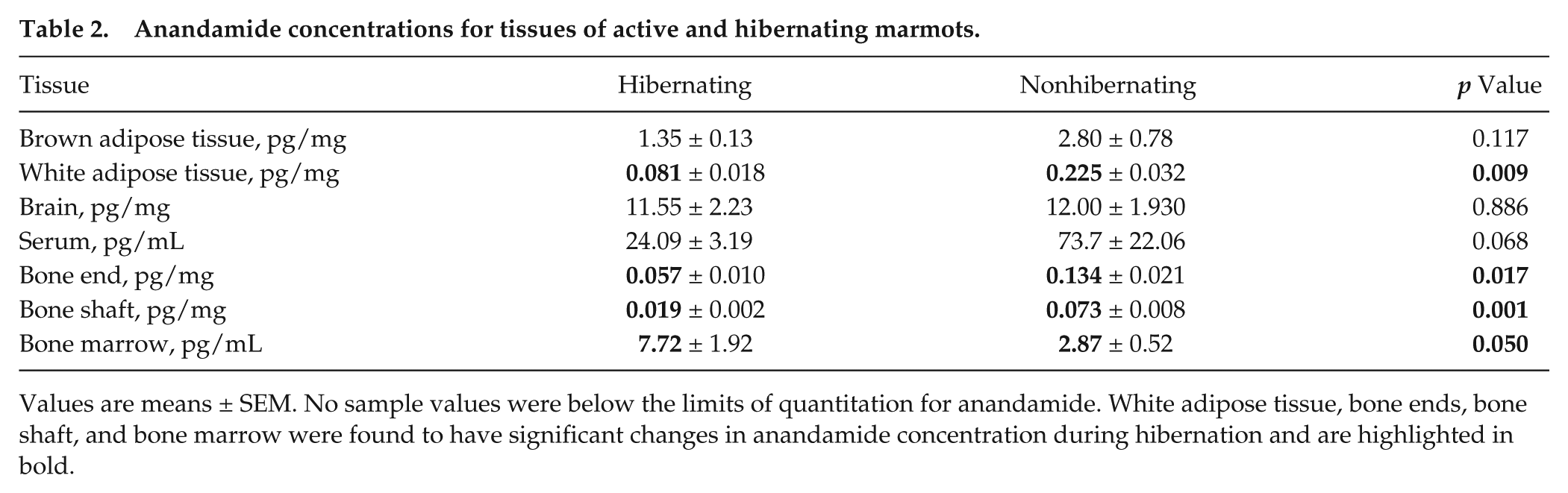
Values are means ± SEM. No sample values were below the limits of quantitation for anandamide. White adipose tissue, bone ends, bone shaft, and bone marrow were found to have significant changes in anandamide concentration during hibernation and are highlighted in bold.
N-palmitoylethanolamine concentrations for tissues of active and hibernating marmots.

Values are means ± SEM. N-palmitoylethanolamine had 7 sample values below the limits of quantitation: 1 in white adipose tissue, 3 in bone shaft, and 3 in serum. Of the N-palmitoylethanolamine sample values below the limits of quantitation, 4 were below the limits of detection. Two of the values below the limits of detection were in bone shaft and the remaining 2 were in serum. Brown adipose tissue, white adipose tissue, serum, and bone shaft were found to have significant changes in N-palmitoylethanolamine concentration during hibernation and are highlighted in bold.
N-oleoylethanolamine concentrations for tissues of active and hibernating marmots.
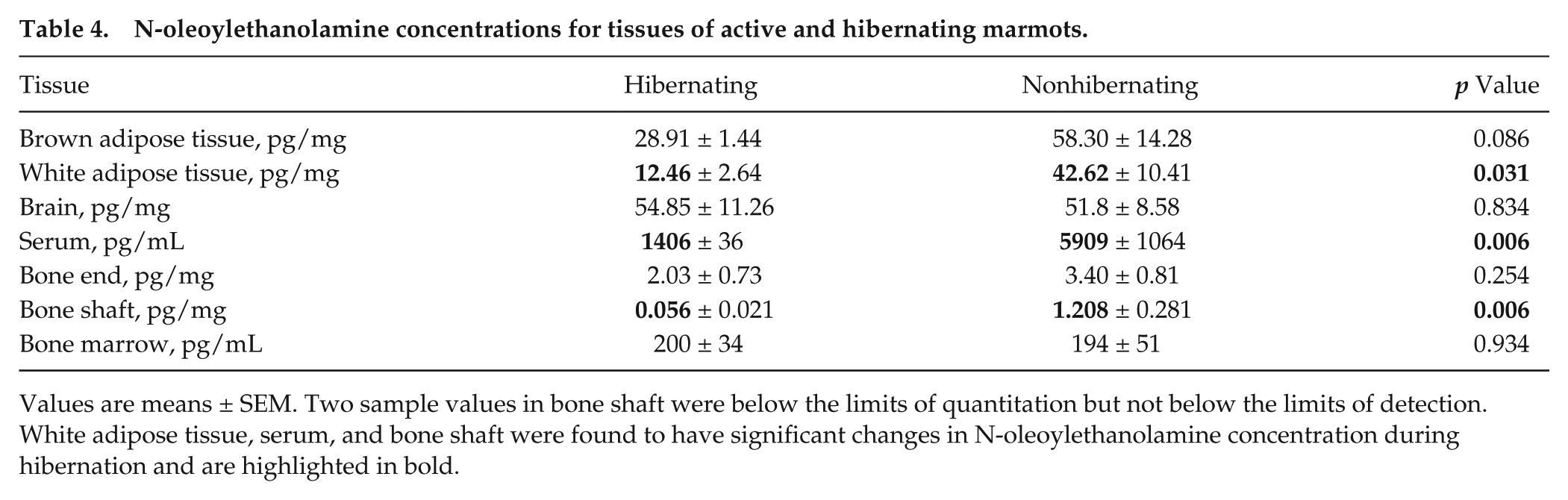
Values are means ± SEM. Two sample values in bone shaft were below the limits of quantitation but not below the limits of detection. White adipose tissue, serum, and bone shaft were found to have significant changes in N-oleoylethanolamine concentration during hibernation and are highlighted in bold.
Concentrations of 2-arachidonoyl glycerol for tissues of active and hibernating marmots.
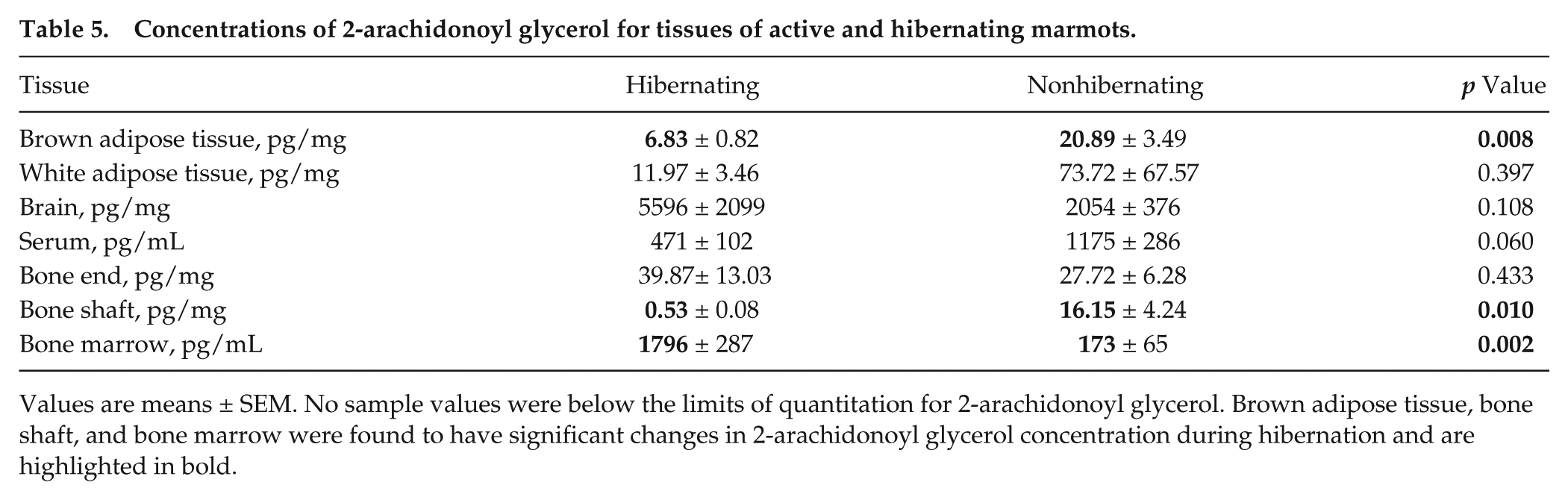
Values are means ± SEM. No sample values were below the limits of quantitation for 2-arachidonoyl glycerol. Brown adipose tissue, bone shaft, and bone marrow were found to have significant changes in 2-arachidonoyl glycerol concentration during hibernation and are highlighted in bold.
Concentrations of 1-arachidonoyl glycerol for tissues of active and hibernating marmots.

Values are means ± SEM. One sample value was found in serum that was below the limits of quantitation but not below the limits of detection. Serum, bone ends, and bone marrow were found to have significant changes in 1-arachidonoyl glycerol concentration during hibernation and are highlighted in bold.
Statistics
For each tissue, the null hypothesis of equivalent concentration of each endocannabinoid in hibernating and active groups was tested using a t test in SPSS. Due to the understanding that 2-AG spontaneously isomerizes into its inactive form, 1-AG, by acyl migration, Zoerner et al. (2011) recommended the summation of 2-AG and 1-AG mass spectroscopy peaks to yield a more accurate representation of endogenous 2-AG levels. The 2-AG + 1-AG was analyzed using a t test in addition to endocannabinoids AEA, PEA, OEA, 2-AG, and 1-AG. A p value of 0.05 or less was considered statistically significant for all tests used. The data are presented as unadjusted mean ± SEM.
Results
Significant Changes in Endocannabinoid and Endocannabinoid-like Ligand Concentrations of Tissues during Hibernation
Significant differences between hibernating and active concentrations were found to be compound and tissue specific. As shown in Table 2, AEA in hibernating marmots was found to be significantly decreased in WAT (p = 0.009), bone ends (p = 0.017), and bone shaft (p = 0.001) and significantly increased in bone marrow (p = 0.050). Furthermore, in hibernating marmots, PEA was significantly decreased in WAT (p = 0.034), BAT (p = 0.048), serum (p = 0.012), and bone shaft (p = 0.027) (Table 3). OEA showed significant decreases during hibernation in WAT (p = 0.031), serum (p = 0.006), and bone shaft (p = 0.006) (Table 4). 2-AG, shown in Table 5, was significantly decreased in BAT (p = 0.008) and bone shaft (p = 0.010). However, a significant increase in 2-AG was found in bone marrow (p = 0.002) during hibernation. In Table 6, 1-AG showed a similar increase in bone marrow (p ≤ 0.001) as well as bone ends (p = 0.010) but was significantly decreased in serum (p = 0.008). Combined, 2-AG + 1-AG showed significant decreases in hibernating BAT (p = 0.017), serum (p = 0.019), and bone shaft (p = 0.016) and significant increases in hibernating bone marrow (p ≤ 0.001) and bone ends (p = 0.031) (Table 7).
Concentrations of 2-arachidonoyl glycerol + 1-arachidonoyl glycerol for tissues of active and hibernating marmots.

Values are means ± SEM. Brown adipose tissue, serum, bone end, bone shaft, and bone marrow were found to have significant changes in concentrations of 2-arachidonoyl glycerol + 1-arachidonoyl glycerol during hibernation and are highlighted in bold.
Fold-changes in Endocannabinoid and Endocannabinoid-like Ligand Concentrations
Bone shaft displayed the largest differences in AEA, OEA, and 2-AG with 3.8-, 21.6-, and 30.5-fold decreases during hibernation, respectively. PEA showed the most change in serum with a 10.4-fold decrease during hibernation. In bone marrow during hibernation, 1-AG increased drastically by 78.4-fold and 2-AG + 1-AG increased 23.7-fold.
Discussion
The findings presented here are steps toward discovering how hibernators are able to maintain control over metabolic processes under extreme physiological conditions of disuse, obesity, and restricted food intake. Understanding how hibernating mammals seasonally combat the negative consequences of anorexia, disuse-induced bone loss, and obesity could lead to novel therapies for nonhibernating animals and humans. The endocannabinoid system is a known regulator of appetite, weight gain, and energy homeostasis (Bellocchio et al., 2008a; Pagano et al., 2008). Endocannabinoids can be synthesized from dietary PUFAs, including linoleic acid and arachidonic acid (Watkins and Kim, 2014). There is a well-established link between high dietary PUFAs and aspects of hibernation, including promotion of lower body temperature, longer torpor bouts, and slower metabolic rate (Ruf and Arnold, 2008). It is possible that endocannabinoid signaling could serve as the unknown mechanism by which PUFAs affect hibernation. During hibernation, there is no dietary intake of PUFAs, and PUFAs are preferentially retained in WAT over saturated and monounsaturated fatty acids (Florant et al., 1990). Lack of PUFAs available as a source for endocannabinoid synthesis during hibernation could explain the significant decreases in many of the tissues sampled. The seasonal rhythm of endocannabinoid levels during hibernation, as a possible underlying mechanism for precise and flexible control of metabolic processes, has not yet been investigated in multiple tissues. To date, endocannabinoids of hibernating animals have been quantified in only serum and brain (Stewart et al., 2002; Vaughn et al., 2010; Kirkwood et al., 2016). The present study found significant differences in endocannabinoid concentrations in multiple tissues during hibernation. The endocannabinoids that were affected by hibernation and the known tissue-specific actions of endocannabinoids are summarized in Table 8.
Summary of significant changes with respect to hibernation and known actions of activated receptors.
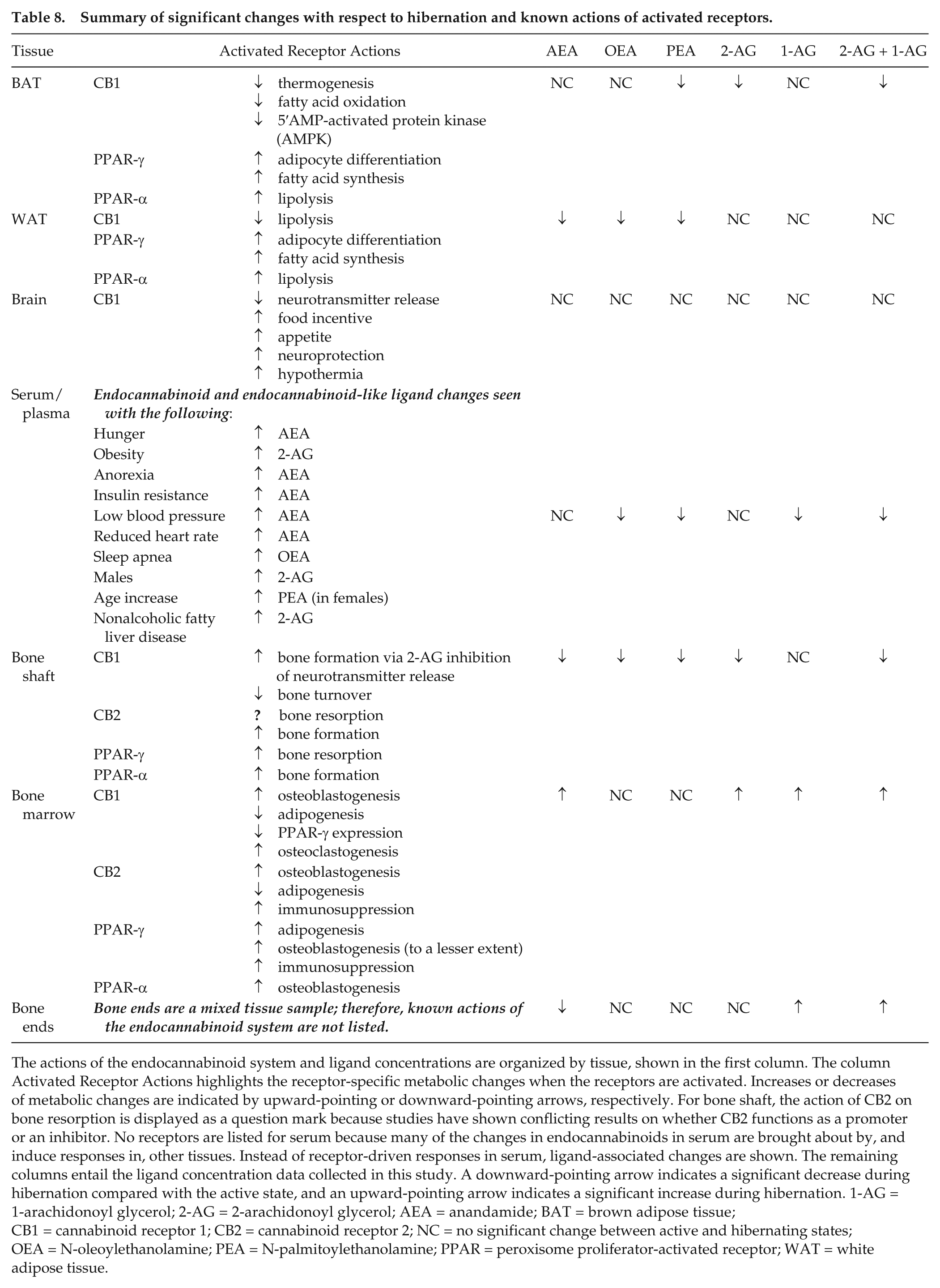
The actions of the endocannabinoid system and ligand concentrations are organized by tissue, shown in the first column. The column Activated Receptor Actions highlights the receptor-specific metabolic changes when the receptors are activated. Increases or decreases of metabolic changes are indicated by upward-pointing or downward-pointing arrows, respectively. For bone shaft, the action of CB2 on bone resorption is displayed as a question mark because studies have shown conflicting results on whether CB2 functions as a promoter or an inhibitor. No receptors are listed for serum because many of the changes in endocannabinoids in serum are brought about by, and induce responses in, other tissues. Instead of receptor-driven responses in serum, ligand-associated changes are shown. The remaining columns entail the ligand concentration data collected in this study. A downward-pointing arrow indicates a significant decrease during hibernation compared with the active state, and an upward-pointing arrow indicates a significant increase during hibernation. 1-AG = 1-arachidonoyl glycerol; 2-AG = 2-arachidonoyl glycerol; AEA = anandamide; BAT = brown adipose tissue; CB1 = cannabinoid receptor 1; CB2 = cannabinoid receptor 2; NC = no significant change between active and hibernating states; OEA = N-oleoylethanolamine; PEA = N-palmitoylethanolamine; PPAR = peroxisome proliferator-activated receptor; WAT = white adipose tissue.
A possible limitation of this study is that the synchronized torpor bout cycles of the marmots became desynchronized due to variation in cycle length between animals. We tried to limit the opportunity for desynchronization by synchronizing the torpor bouts near the time of sacrifice and recording body temperatures with an infrared thermometer. Knowing and controlling for the stage of torpor at sacrifice is important because physiological changes occur during the torpor bout, especially during entrance and exit from deep torpor (Carey et al., 2003). Another limitation of the study is that all 4 seasonal phases were not measured. The 4 phases include active (summer), entrance into hibernation (fall), hibernation (winter), and exit from hibernation (spring). Our “active” sample was collected during the prehibernation, or “entrance into hibernation,” phase. At this time point, the marmots had begun to suppress metabolism but had not yet started hibernating. In October, marmots weigh 149% of July summer values and have food intake 33% of July summer values, while in February they weigh 126% of July summer values and have food intake 0% of July values (Florant et al., 1993). Additionally, the brain samples were taken from the hypothalamus. This limits the interpretation of results from the brain because the actions of ligands, and their relative concentrations, are specific to particular regions of the brain (Bellocchio et al., 2008a). Some values found were below the LOQ in PEA, OEA, and 1-AG. Notably, all the values below the LOQ were found only in hibernating samples. PEA was the only ligand with values below the calculated LOD. These sample values, found in serum and bone shaft, were kept in statistical analyses as their normalized peak areas were at least 1.67× and 2.49× above background, respectively. The summation of 2-AG and 1-AG was conducted in an effort to obtain a more complete picture of the level of 2-AG in vivo because the rate of isomerization of 2-AG to 1-AG is unknown. However, since 2-AG is also degraded into 1-AG and glycerol by MAGL in vivo (Battista et al., 2012), the actual level of 2-AG is likely somewhere between the value for 2-AG and the combined 2-AG and 1-AG value.
Serum and Brain
Blood circulates through all tissues. Thus, serum endocannabinoid concentrations indicate overall organismal endocannabinoid levels. Of the 5 ligands measured, only OEA, PEA, 1-AG, and 2-AG + 1-AG were found to be significantly different in serum between seasons. In woodchucks, a close relative of marmots, OEA showed a similar significant decrease during hibernation and relatively constant AEA in hibernating and active states, while PEA and 2-OG (2-oleoylglycerol) showed significant increases during hibernation, and 2-AG could not be detected in either state (Vaughn et al., 2010). In contrast, only 2-AG showed a significant seasonal change in black bear serum (Kirkwood et al., 2016). The highest levels of 2-AG in black bears occurred during the fall transition into hibernation. Serum results in this study on marmots showed significant decreases in 1-AG and 2-AG + 1-AG, but not 2-AG, during hibernation. The contradictory increase of PEA in woodchucks compared with the significant decrease found in this study warrants further investigation. It is possible that the role of PEA as a modulator of inflammation (De Petrocellis and Di Marzo, 2009) does not require tight regulation with respect to the hibernating state. In plasma, AEA has been associated with meal-related changes where AEA concentrations diminish after eating (Matias et al., 2012). In serum, increased levels of 2-AG and arachidonic acid are present with nonalcoholic fatty liver disease (Zelber-Sagi et al., 2017). Increased plasma endocannabinoid and endocannabinoid-like ligand levels have likewise been correlated with obesity, anorexia, insulin resistance, heart rate, blood pressure, sleep apnea, gender, and age (Pacher et al., 2006; Matias et al., 2012). The lack of change in AEA agrees with the signs of decreased appetite shown in late fall and winter marmots but contrasts with the increased AEA seen with insulin resistance in other animals (Matias et al., 2012). This might be a factor of the circannual reversibility of insulin resistance seen in hibernators (Wu et al., 2013; Rigano et al., 2017). Since an increase in circulating OEA is associated with sleep apnea and promoting wakefulness (Matias et al., 2012; Prospero-Garcia et al., 2016), it is logical that OEA would be significantly decreased during hibernation to promote sleep. Decreased 1-AG and 2-AG + 1-AG during hibernation support the promotion of obesity in the fall. Blood also contains immune cells (Yatim and Lakkis, 2015). Endocannabinoid and endocannabinoid-like ligands can exert an immunosuppressive response on immune cell activity and proliferation primarily through activation of CB2 and also through PPAR-γ (Pandey et al., 2009). Hibernators exhibit a suppressed immune response (Carey et al., 2003) that can be explained by the connection between the endocannabinoid system and the immune system in bone marrow discussed below.
In the brain, endocannabinoids and endocannabinoid-like ligands play a strong role in influencing behavior and metabolic processes. The hypothalamus is responsible for maintaining homeostasis of physiological mechanisms such as body temperature, heart rate, autonomic behavior, metabolism, appetite, sleep, bone remodeling, and hormone release. Thus, it was surprising that significant seasonal changes in endocannabinoid levels were not detected in marmot hypothalamus even though the endocannabinoid system has known roles in regulating homeostasis of these mechanisms. The significant seasonal changes in serum endocannabinoid levels, but not brain levels, raise the possibility that hibernation-related physiological changes that are driven by the endocannabinoid system are primarily peripherally regulated. However, the lack of changes in the hypothalamus may also have been due to the timing of sample collection. Samples of the active group were collected in late fall, close to the onset of hibernation. By late fall, it is possible that neurochemical changes related to shifting of appetite regulation and energy metabolism in preparation for hibernation had already begun. This would mean that hypothalamic endocannabinoid concentrations of active fall marmots would look very similar to the concentrations in hibernating winter marmots, whereas concentrations in active summer marmots may not appear similar. Notably, a previous study found drastic changes in the CB1 agonist, oleamide, between whole brains of hibernating (deep torpor) and euthermic (interbout arousal) Richardson’s ground squirrels (Stewart et al., 2002).
Adipose Tissue
The significant decreases in 2-AG, 2-AG + 1-AG, and PEA in BAT and AEA, OEA, and PEA in WAT exhibited during hibernation corroborate the shift in energy metabolism between hibernating and nonhibernating states. The higher levels of endocannabinoid and endocannabinoid-like ligands in adipose tissue during nonhibernation could potentially act as a driver of the adipogenesis and energy storage needed for hibernation, whereas the decreases during hibernation would accurately reflect a diminished appetite and reduced lipogenesis (Kabine et al., 2004; Bellocchio et al., 2008a). For example, hyperactivation of the endocannabinoid system has been linked to obesity. CB1 and 2-AG were found to be upregulated in the adipose tissue of obese animals (Bellocchio et al., 2008b). In BAT, CB1 activation is thought to decrease thermogenesis and fatty acid oxidation (Korf, 2017). Therefore, lower concentrations of 2-AG and 2-AG + 1-AG in BAT would likely promote thermogenesis through reduced CB1 activation during hibernation. Both endocannabinoid and endocannabinoid-like ligands have been shown to directly and indirectly activate PPAR-γ to promote adipocyte differentiation (Sun and Bennett, 2007). PPAR-γ also promotes fatty acid synthesis (Varga et al., 2011). The decrease in endocannabinoid concentrations in BAT and WAT agrees with reduced PPAR-γ expression and increase of mRNA of key PPAR-mediated enzymes used in lipid metabolism and thermogenesis found in BAT and WAT during hibernation in jerboa (Kabine et al., 2004). Many hibernators rely on lipid metabolism as a sole energy source during the winter (Carey et al., 2003; Dark, 2005). Thus, endocannabinoid and PPAR-γ regulation are tied to increased fat deposition in hibernators during the summer and fall and the promotion of fatty acid degradation during the winter. Another mode of action is through PPAR-α. Both OEA and AEA have high affinity for PPAR-α as well as probable indirect activation by PEA (Pagano et al., 2008). OEA acts through PPAR-α to promote lipolysis, while PEA has been shown to decrease inflammation (Sun and Bennett, 2007; Pagano et al., 2008). The significant decreases, during hibernation, of ligands acting on PPAR-α in BAT and WAT support a reduced metabolism and correlate with PPAR-α expression that was decreased in BAT and became undetectable in WAT during hibernation (Kabine et al., 2004). As expected, the decreased concentrations of endocannabinoids and endocannabinoid-like ligands during hibernation in adipose tissue indicate an overall reduction of metabolism during hibernation.
Bone Marrow, Bone Ends, and Bone Shaft
Levels of endocannabinoid and endocannabinoid-like ligands in bone support the known suppression of bone remodeling (Doherty et al., 2016) and possible preferential promotion of osteoblastogenesis over adipogenesis during hibernation. Adipose tissue is also found in bone marrow. Contrary to the decreases in BAT and WAT during hibernation, bone marrow showed increases in 2-AG, 1-AG, 2-AG + 1-AG, and AEA. Bone marrow serves as an endocrine organ in energy homeostasis, contains a variety of cell types, and is the site where both hematopoietic and mesenchymal stem cell differentiation takes place (Marquis et al., 2009; Suchacki et al., 2016). There is an inverse correlation between marrow adiposity and bone anabolism (Veldhuis-Vlug and Rosen, 2017). Activation of CB1 and CB2 receptors by endocannabinoids favors osteoblastogenesis instead of adipogenesis (Idris et al., 2009; Sophocleous et al., 2011). Activation of CB1 is also thought to promote osteoclastogenesis, although the influence of CB2 on osteoclastogenesis remains controversial (Idris and Ralston, 2012). While the significant increases of 2-AG, 1-AG, and AEA suggest that osteoblastogenesis should be increased during hibernation, this has not been shown to be the case. Hibernating marmots display a significant increase in marrow adiposity relative to nonhibernators (from 66.9% to 88.7%) in femoral metaphyses and a reduction in the number of osteoblasts in trabecular bone (Doherty et al., 2016). Many signals are integrated for the regulation of bone metabolism. It is possible that decreased mechanical signals during hibernation that would normally promote bone resorption and inhibit formation are counteracted by increased endocannabinoid signals favoring bone formation, so that bone integrity and organismal calcium homeostasis are maintained. This inconsistency might also be explained by the extremely high level of MAGL in bone degrading 2-AG to 1-AG before 2-AG is able to act on cannabinoid receptors (Doherty et al., 2016). The increase in derivatives of 2-AG, in addition to the increase in AEA, could possibly support the phenotype of hibernating marmots when the actions of PPARs are taken into account. In CB1 knockout mice, expression of PPAR-γ was increased (Idris et al., 2009). PPAR-γ promotes adipogenesis (an action contradictory to that of CB1 and CB2) and to a lesser extent promotes osteoblastogenesis (Rosen and Bouxsein, 2006; Takano et al., 2012). PPAR-α stimulates osteoblastogenesis (Rosen and Bouxsein, 2006; Takano et al., 2012). Bone marrow is also the site of immune cell proliferation and maturation (Yatim and Lakkis, 2015). As stated previously, endocannabinoids suppress immune cell proliferation and activity (Pandey et al., 2009). Thus, combined actions of endocannabinoids and PPARs could potentially explain the bone observed in hibernating marmots.
The bone shaft, consisting of pure cortical bone, exhibited significant decreases in all ligands measured except 1-AG. Both CB1 and CB2 are found on osteoclasts and osteoblasts; however, CB2 is in much higher abundance on osteoclasts and osteoblasts (Idris and Ralston, 2012). Although findings on actions of CB2 in bone remain highly controversial (Idris and Ralston, 2012), one study found that CB2 knockout mice had reduced numbers of osteoclasts and had partial protection from ovariectomy-induced bone loss (Idris et al., 2008). The knockout of CB1 results in fewer osteoclasts and reduced bone turnover as well as higher adipogenesis and lower osteoblastogenesis inducing osteoporosis with age (Idris and Ralston, 2012). The main action of CB1 receptors on bone is thought to be through adrenergic signaling. 2-AG binds to CB1 receptors on sympathetic nerve terminals and inhibits release of norepinephrine, thereby promoting bone formation (Tam et al., 2008). Yet neurectomy as a model of forced disuse does not induce bone loss in hibernating arctic ground squirrels (Bogren et al., 2016). It is therefore unlikely that CB1 on sympathetic nerve terminals is a key component of the mechanism for maintaining bone during hibernation. The ligands and their byproducts from degradation could also possibly influence change on bone metabolism by binding and activating PPARs that perform as transcription factors to regulate differential expression of genes. PPAR-γ supports the resorption of bone, while PPAR-α promotes the formation of bone (Stunes et al., 2011). In a study on ovariectomized rats, PPAR-γ exaggerated bone loss while PPAR-α maintained bone mass (Nallamshetty et al., 2014). However, the main influence of PPAR-γ is through regulating differentiation. PPAR-γ was found to have no influence on fully differentiated osteoblasts and osteoclasts (Akune et al., 2004). Therefore, significantly reduced levels of endocannabinoids and endocannabinoid-like ligands indicate suppression in bone turnover during hibernation via CB2, PPARs, and possibly CB1 pathways.
The bone ends, which include marrow, cortical bone, and trabecular bone, had significant increases in 1-AG and 2-AG + 1-AG and a significant decrease in AEA during hibernation. For both 1-AG and 2-AG + 1-AG, bone marrow had a much higher fold change than bone shaft and is likely the dominating tissue, whereas for AEA, bone shaft expressed a higher fold change than bone marrow. This agrees with the significant increase of 1-AG in bone marrow and significant decrease of AEA in bone shaft. All concentrations of endocannabinoids and endocannabinoid-like ligands in bone ends remained between the concentrations for bone marrow and bone shaft. However, changes in pure trabecular bone with respect to hibernation are still unknown.
Summary and Conclusions
This study presents data supporting a significant seasonal variation in endocannabinoid and endocannabinoid-like ligand concentrations, between hibernation and active states, in various tissues of marmots. The wide range of tissue sampling gives the advantage of a more accurate interpretation of the data by enabling us to examine interactions and processes involving multiple tissues for a big-picture understanding of the physiological state. These data provide a step toward understanding the mechanisms regulating hibernation, as the endocannabinoid system is a highly influential regulator of physiological conditions such as appetite, thermoregulation, and bone and energy metabolism. The lack of changes in hypothalamic endocannabinoid concentrations during hibernation possibly indicate peripheral regulation of the shift in energy metabolism, suppression of the immune system, promotion of osteoblastogenesis over adipogenesis in bone marrow, and suppressed bone turnover to prevent bone loss. The role of the endocannabinoid system during hibernation as a possible preventive mechanism for the negative effects of inactivity is worthy of further investigation, and the resulting knowledge could potentially have therapeutic applications in diseases such as osteoporosis and obesity.
Footnotes
Acknowledgements
We thank the Florant Lab for participation in animal trapping and care, as well as sample collection at necropsy. We also thank Fox Acres Country Club for permission to trap on their grounds. This work was supported by NSF (grant number 1555196).
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
