Abstract
Like two dancers, the circadian clock and cell cycle are biological oscillators engaged in bidirectional communication, resulting in circadian clock–gated cell division cycles in species ranging from cyanobacteria to mammals. The identified mechanisms for this phenomenon have expanded beyond intracellular molecular coupling components to include intercellular connections. However, detailed molecular mechanisms, dynamics, and physiological functions of the circadian clock and cell cycle as coupled oscillators remain largely unknown. In this review, we discuss current understanding of this connection in light of recent findings that have uncovered intercellular coupling between the circadian clock in Paneth cells and the cell cycle in intestinal stem cells via WNT signaling. This extends the impact of circadian rhythms regulating the timing of cell divisions beyond the intracellular domain of homogenous cell populations into dynamic, multicellular systems. In-depth understanding of the molecular links and dynamics of these two oscillators will identify potential targets and temporal regimens for effective chronotherapy.
Molecular Mechanisms of Circadian Rhythms
Most organisms exhibit circadian rhythms, reflecting an internal biological clock with an approximate period of 24 h, to anticipate and align their physiological functions with daily environmental changes. Autonomous oscillations of circadian rhythms are generated at single-cell level by transcriptional-translational feedback loops (TTFLs), which consist of positive and negative elements (Hurley et al., 2016; Lowrey and Takahashi, 2011) (Fig. 1A). In mammals, the positive elements consist of heterodimeric basic helix loop helix-Per/Arnt/Sim (bHLH-PAS) transcription factors, Circadian Locomotor Output Cycles Kaput (CLOCK) and Brain and Muscle ARNT-Like 1 (BMAL1), which regulate the expression of negative elements,
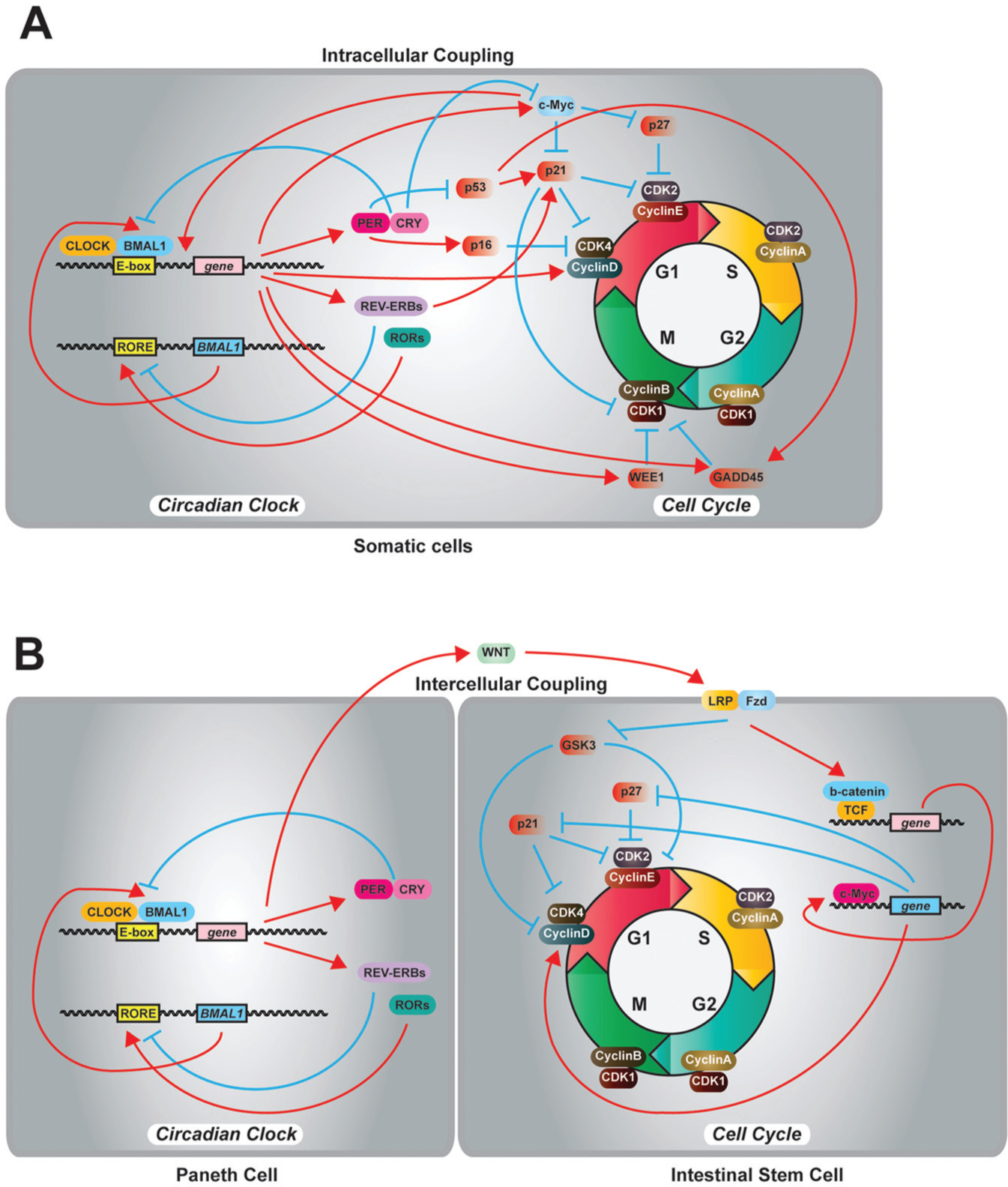
Molecular connections between the circadian clock and the cell cycle. (A) In mammalian somatic cells, the circadian clock gates the cell cycle by intracellular molecular connections. (B) The circadian clock in Paneth cells regulates the timing of divisions in intestinal stem cells and progenitor cells through rhythmic secretion of WNT.
Efficient operation of the above interlocked feedback loops that sustain autonomous circadian oscillations requires posttranslational regulation of the core clock proteins. Casein Kinase 2α (CK2α) rhythmically phosphorylates BMAL1, which determines the rhythmic accumulation of CLOCK/BMAL1 in the nucleus (Kondratov et al., 2003; Tamaru et al., 2009). Casein Kinase 1ε (CK1ε) phosphorylates CRY1, CRY2, and BMAL1, and CKIε-dependent phosphorylation of BMAL1 positively regulates its transcriptional activity (Eide et al., 2002). Importantly, the negative elements undergo extensive phosphorylation that regulates the function and degradation of these proteins. PER2 in mammals and its analogous protein, FREQUENCY (FRQ) in
Intracellular Molecular Links between the Cell Cycle and the Circadian Clock
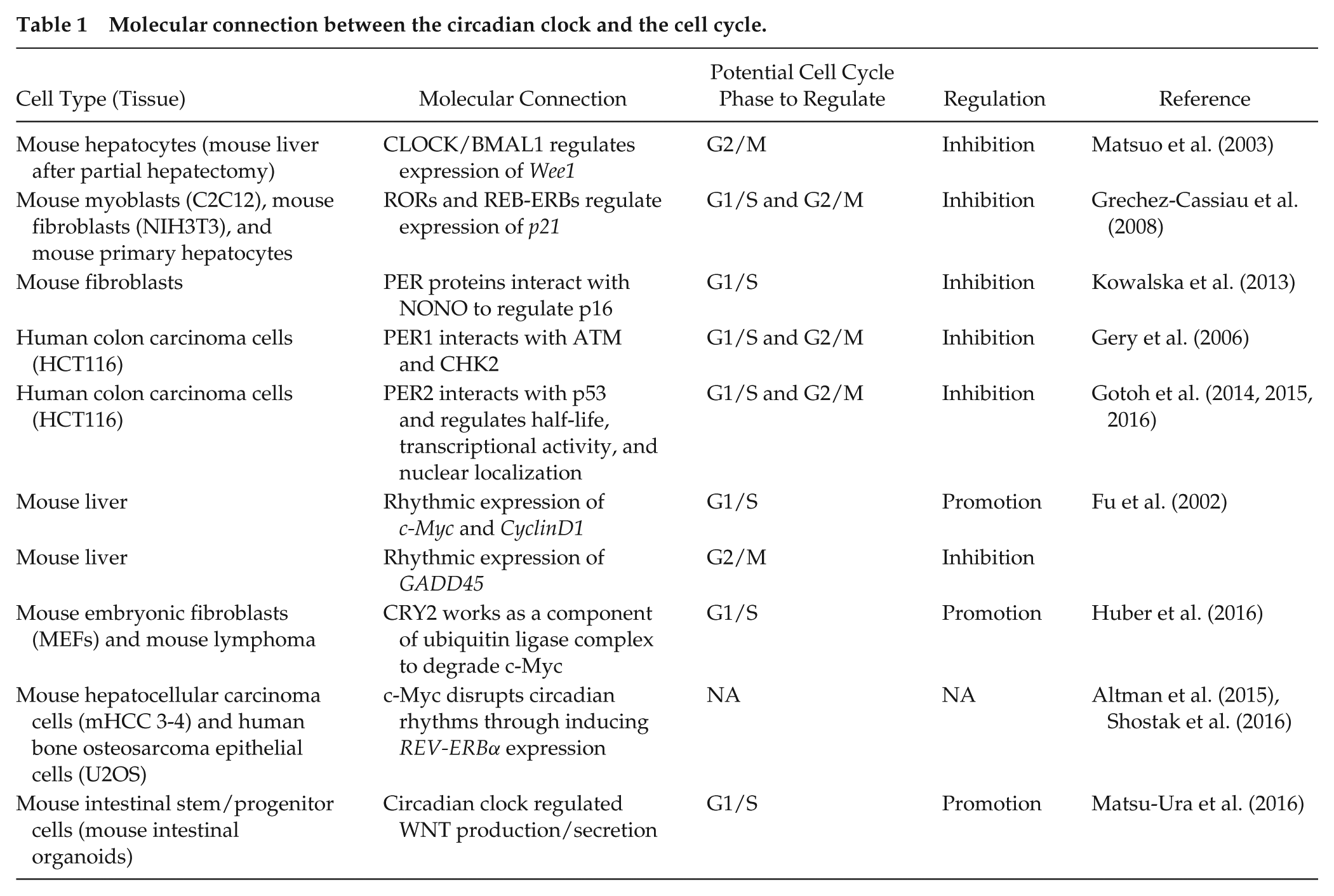
Previously, several circadian clock–regulated cell cycle components have been identified. Matsuo et al. (2003) reported that the expression of a G2/M checkpoint kinase,
Importantly, core circadian clock elements regulate key components that control cell proliferation and tumorigenesis. Gotoh et al. reported a series of studies of PER2 interaction with a tumor suppressor, p53. The studies revealed that the physical interaction between PER2 and p53 results in (1) stabilization of p53 from Murine Double Minute-2 (MDM2)–mediated ubiquitination and degradation (Gotoh et al., 2014), (2) inactivation of the transcriptional activity of p53 (Gotoh et al., 2015), and (3) nuclear translocation of p53 in human colon cancer HCT116 cells (Gotoh et al., 2016). A proto-oncogene, c-Myc, and its downstream genes,
Molecular connection between the circadian clock and the cell cycle.
Dysregulated cellular proliferation is a characteristic property of cancer. Oscillations of circadian clock genes were reported in cancer cell lines including osteosarcoma cells (U2OS) (Hughes et al., 2009), breast cancer cells (MCF10A) (Xiang et al., 2012), and colorectal cancer cells (HCT116 and Caco2) (Gotoh et al., 2016; Moore et al., 2014). In contrast, it has been shown that circadian clock–related genes are impaired in most human cancers, suggesting that cancer cells target the circadian clock machinery to achieve uncontrolled growth and proliferation (Davidson et al., 2006). In fact, the number of rhythmic genes is dramatically reduced in cancers and immortalized cell lines cultured in vitro (percentage of rhythmic genes: 1.5% in U2OS [Krishnaiah et al., 2017]; 2.6% in NIH3T3 [Menger et al., 2007]; and 1.9% in Rat-1 [Duffield et al., 2002]) compared with liver and other organs (10%-40%) (Panda et al., 2002; Vollmers et al., 2009; Zhang et al., 2014). Although the difference in the number of rhythmic genes between cell lines and mouse organs may be due to differences in conditions in vitro and in vivo, these results suggest a disruption of the molecular clockworks in cancer and immortalized cells. In 2007, the International Agency for Research on Cancer (IARC) categorized shiftwork that involves circadian disruption as carcinogenic to humans (Straif et al., 2007).
The Circadian Clock in Adult Stem Cells and Its Connection to the Cell Cycle
Many of the previous studies used cancer or immortalized cell lines to investigate molecular coupling between the circadian clock and cell cycle/proliferation in the mammalian system. However, it is important to note that adult stem cells are one of the major proliferating cell types in our body, and the circadian clock appears to operate differently depending on the context of stem cells. Therefore, it is critical to investigate the roles of circadian rhythms in stem cell regeneration and proliferation. Yagita et al. (2010) reported the lack of circadian rhythms in mouse embryonic stem cells and demonstrated the initiation of circadian rhythms during differentiation. In contrast, human epidermal stem cells possess canonical TTFL of clock components with similar robustness as differentiated keratinocytes, with stemness, differentiation, epidermal aging, and tumorization regulated by the circadian clock (Janich et al., 2011; Janich et al., 2013). The circadian clock gates the cell cycle in epidermal stem cells, which controls the timing of S-phase at around the late afternoon to provide protection against DNA damage from UV irradiation (Geyfman et al., 2012; Plikus et al., 2013). In addition, quiescent neural stem/progenitor cells in the subgranular zone of adult mouse hippocampus possess rhythmic
In contrast to the aforementioned direct/intracellular coupling and regulation of stem/progenitor cell proliferation by the circadian clock, adult stem cells are also regulated indirectly by circadian rhythms. Hematopoietic stem cells and progenitor cells (HSPCs) circulate in the bloodstream, and the release of HSPCs from bone marrow is rhythmically regulated by noradrenaline secretion from the sympathetic nervous system that is under the control of the master clock (Mendez-Ferrer et al., 2008).
WNT Signaling in Adult Stem Cells and Its Interaction with the Circadian Clock
WNT signaling is known to regulate the self-renewal and differentiation of various adult stem cells. In the small intestine, secreted WNT from Paneth cells promotes self-renewal of ISCs (Clevers et al., 2014). The activity of WNT is meticulously controlled for tissue patterning in the stomach. The homeodomain transcription factor, BarH-Like Homeobox 1 (BARX1), is abundantly expressed in embryonic mesenchymal cells in the stomach and regulates the expression of WNT inhibitor, Secreted Frizzled Related Protein 1 (SFRP), resulting in the development of gastric epithelium (Kim et al., 2005). In addition, gastric and colon cancers are associated with abnormality of canonical WNT signaling (Polakis, 2000; Vogiatzi et al., 2007). The importance of WNT signaling for self-renewal of adult stem cells in the gastrointestinal tract is also highlighted by the requirement of exogenous WNT for the growth and maintenance of organoid cultures from mammalian taste bud (Aihara et al., 2015), stomach (Barker et al., 2010), intestine (Sato et al., 2011), and colon (Sato et al., 2011). As well, WNT signaling is critical for the niche of adult stem cells in other tissues. Adult rat hippocampal neural progenitor cells (AHPs) express various types of
Interactions of the circadian clock and WNT signaling have been described in other cells, tissues, and organisms. Numerous genes in WNT signaling are under circadian regulation, which highlights the impact of circadian rhythms on WNT signaling. Approximately 50 genes that are involved in WNT signaling demonstrate rhythmic expression profiles in 39 microarray data sets from 19 distinct mammalian tissues (Sotak et al., 2014). ChIP-Seq analysis using mouse liver (Rey et al., 2011) revealed rhythmic occupancy of BMAL1 on the promoters of the canonical WNT signaling pathway including
Genetic and physiological disturbances of circadian rhythms affect the expression of genes in WNT signaling. Xenografted cancer cells proliferate faster in nude mice exposed to a “circadian disrupted” constant light condition compared with nude mice exposed to a “normal” 12-h light-dark cycle, secondary to increased expression of WNT10A in dermal fibroblasts due to disruption of the circadian clock (Yasuniwa et al., 2010). Furthermore, overexpression of
Conclusion
Three and a half centuries following Christiaan Huygens’ classic description of coupled oscillations between two pendulum clocks in 1665 (Huygens, 1893), coupled oscillators continue to be identified throughout the natural world. In biology, coupled oscillators generate diverse dynamic behaviors such as the synchronous flashing of fireflies (Blair, 1915), circadian rhythms in the SCN (Barker et al., 2007), frog choruses (Aihara et al., 2014), myocardial cell contractions (Mirollo and Strogatz, 1990), polyrhythmic interactions among critical brain areas during working memory maintenance (Fujisawa and Buzsaki, 2011), and time-of-day proliferation of somatic cells including adult stem/progenitor cells. These observations indicate the ubiquitous nature of coupled oscillators, but molecular mechanisms that determine diverse dynamic behaviors remain largely unexplored. The circadian clock and the cell cycle use both intra- and intercellular coupling mechanisms to establish their connections, which may vary depending on the cell types and multicellular environment.
We recently demonstrated that the circadian clock in Paneth cells regulates rhythmic expression and secretion of WNT, which coordinates the timing of cell divisions of adult stem and progenitor cells resulting in different coupling ratios depending on the inherent frequency of cell cycle times (Matsu-Ura et al., 2016). Importantly, we identified rhythmic intestinal crypt formation following the coordinated cell division cycles, as well as a reduced number of crypts in the small intestine of circadian arrhythmic,
Unintended damage to healthy stem and progenitor cells remains a major limitation of cancer chemotherapy (Davidson et al., 2006; Savvidis and Koutsilieris, 2012). Further, disruption of circadian rhythms is associated with a higher risk of various human cancers (Savvidis and Koutsilieris, 2012), and the circadian clock is dysregulated in most human neoplasms (Davidson et al., 2006). Therefore, it will be important to characterize cell cycle profiles of different adult stem cells and cancer cells with respect to the circadian cycle and determine whether those cells show time-of-day-specific responses to chemotherapy. This information could then be exploited to design novel treatment regimens to administer chemotherapeutic drugs at the trough of cell divisions of healthy stem/progenitor cells to reduce the risk of unintended damage to healthy cells, without reducing the efficacy of drugs to eliminate cancerous cells. Further investigation of the molecular coupling components and the intimate dance between the cell cycle and the circadian clock in both normal and cancer cells will be critical to effectively use circadian molecular signatures to treat cancer.
Footnotes
Acknowledgements
This work was supported by Department of Interior grant D12AP00005 (to S.R.M. and C.I.H.) and NIH grant 1U19AI116491 (to S.R.M. and C.I.H.). All authors contributed to writing this manuscript and generating figures.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
