Abstract
Transplantation studies have pinpointed the circadian clock of the Madeira cockroach to the accessory medulla (AME) of the brain’s optic lobes. The AME is innervated by approximately 240 adjacent neuropeptidergic neurons, including 12 pigment-dispersing factor (PDF)-expressing neurons anterior to the AME (aPDFMEs). Four of the aPDFMEs project contralaterally, controlling locomotor activity rhythms of the night-active cockroach. The present in vitro Ca2+ imaging analysis focuses on contralaterally projecting AME neurons and their responses to PDF, GABA, and acetylcholine (ACh). First, rhodamine-dextran backfills from the contralateral optic stalk identified contralaterally projecting AME neurons, which were then dispersed in primary cell cultures. After characterization of PDF, GABA, and ACh responses, PDF immunocytochemistry identified ipsilaterally and contralaterally projecting PDFMEs. All PDF-sensitive clock neurons, PDF-immunoreactive clock neurons, and the majority of ipsilaterally and contralaterally projecting cells were excited by ACh. GABA inhibited all PDF-expressing clock neurons, and about half of other ipsilaterally projecting and most contralaterally projecting clock neurons. For the first time, we identified PDF autoreceptors in PDF-secreting cockroach circadian pacemakers. The medium-sized aPDFMEs and all other contralaterally projecting PDF-sensitive clock cells were inhibited by PDF. The ipsilaterally remaining small PDF-sensitive clock cells were activated by PDF. Only the largest aPDFME did not express PDF autoreceptors. We hypothesize that opposing PDF signaling generates 2 different ensembles of clock cells with antiphasic activity, regulating and maintaining a constant phase relationship between rest and activity cycles of the night-active cockroach.
Keywords
Introduction
Neuropeptidergic circadian clocks synchronize the timing of physiology and behavior to geophysical 24-h light–dark cycles in different animal species. Because of its large size and long lifetime, the cockroach Rhyparobia maderae provides an excellent model system to study neuropeptide function in the circadian clock on a cellular and behavioral level. Lesion and transplantation studies have located the cockroach circadian clock in the accessory medulla (AME), ventromedial to the medulla in the brain’s optic lobes (Stengl and Homberg, 1994; Reischig and Stengl, 2003a). The AME is innervated by about 240 neurons from 7 adjacent soma groups that abundantly express colocalized neuropeptides (Petri et al., 1995; Reischig and Stengl, 1996, 2003b; reviews: Stengl et al., 2015; Stengl and Arendt, 2016). Of these neurons, the best characterized are the pigment-dispersing factor–immunoreactive (PDF-ir) neurons next to the AME. One anterior group of 12 PDF-ir neurons, aPDFMEs, and one posterior group of 6 neurons, pPDFMEs, innervate the AME (Fig. 1A and 1B). In addition, 2 PDF-ir groups in the lamina also appear to send inputs to the AME (Fig. 1A) (Reischig and Stengl, 2003b; Wei et al., 2010). The network of PDF pacemaker neurons has been suggested to control rest–activity cycles, to synchronize the bilaterally symmetric clocks, and to take part in light entrainment (reviews: Stengl et al., 2015; Stengl and Arendt, 2016). The 12 aPDFMEs are subdivided into 4 subgroups according to soma size: 1 conspicuously largest, 3 large, 4 medium-sized, and 4 small aPDFMEs (Reischig and Stengl, 2003b). The small aPDFMEs appear to be local neurons of the AME, whereas the others are projection neurons that connect the AME to other optic lobe and midbrain regions (Fig. 1). Backfills from the contralateral AME stain somata of AME cells that connect both bilaterally symmetric clocks. Among them are the largest and 3 medium-sized aPDFMEs that colocalize different neuropeptides (Reischig and Stengl, 2002, 2004; Söhler et al., 2011; Petri et al., 1995; Hofer and Homberg, 2006; review: Stengl and Arendt, 2016).
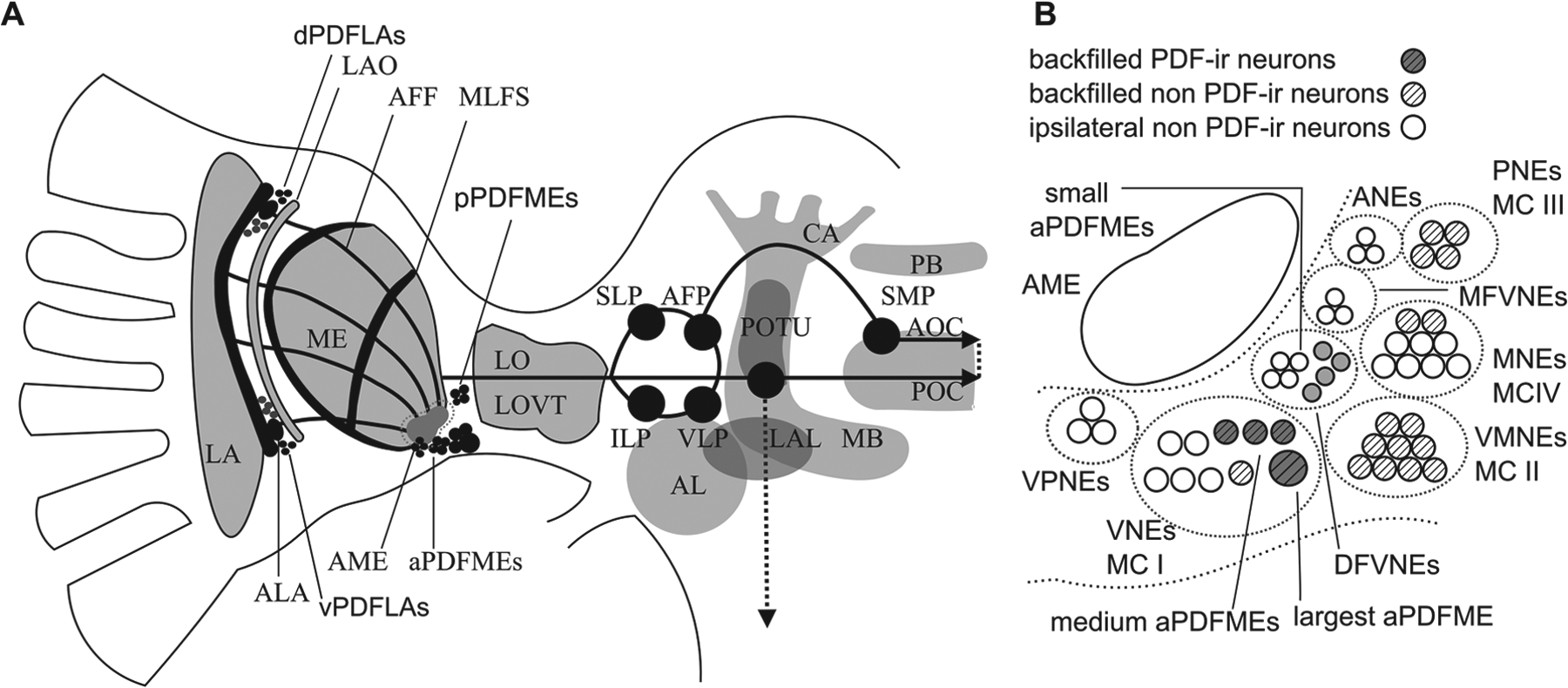
Schematic network of the pigment-dispersing factor–immunoreactive (PDF-ir) circadian clock neurons of the Madeira cockroach. Of the bilaterally symmetric network, only PDF-ir somata and arborizations in the left brain hemisphere are shown (A), with details of the adjacent soma groups in (B). The PDF-ir neurons in dorsal (dPDFLAs) and ventral soma groups (vPDFLAs) in the lamina (LA) innervate the accessory laminae (ALAs) and the proximal LA, and appear to project via the anterior fiber fan (AFF) to the accessory medulla (AME). The AME is the circadian clock of the Madeira cockroach that controls locomotor activity rhythms via contralaterally projecting PDF-ir neurons. Two soma groups of anterior (aPDFMEs) and posterior PDF-ir neurons (pPDFMEs) innervate the AME. The 12 aPDFMEs consist of 4 small, 4 medium-sized, 3 large, and one conspicuously largest clock cell. The 4 small aPDFMEs are local neurons of the AME. Three of the 4 medium-sized aPDFMEs project contralaterally. They send their axons via the lobula valley tract (LOVT) and the anterior optic commissure (AOT) to ipsi- and contralateral midbrain targets (black circles), such as the superior lateral (SLP) and superior medial protocerebrum. They reach finally the contralateral optic lobe, innervating the contralateral AME. The largest aPDFME projects via the posterior optic commissure (POT), innervating the posterior optic tubercles (POTUs). It appears to innervate all midbrain targets of the circadian clock, also the ipsi- and contralateral AME, connecting POT and AOT via the anterior fiber plexus (AFP). (B) Adjacent to the AME (open oval) are 8 neuropeptidergic soma groups: ANEs (anterior neurons), PNEs (posterior neurons), MFVNEs (medial group of frontoventral neurons), DFVNEs (distal group of frontoventral neurons), MNEs (medial neurons), VPNEs (ventroposterior neurons), VNEs (ventral neurons), and VMNEs (ventromedian neurons). The small aPDFMEs belong to the DFVNEs, whereas all other aPDFMEs belong to the VNEs. The pPDFMEs belong to the PNEs. Backfills from the contralateral AME identified contralaterally projecting AME neurons (striped) that were grouped into 4 soma groups, MCI–IV (Söhler et al., 2011), that form subgroups of the above AME soma groups. Four of the aPDFMEs (the largest and 3 medium; dark gray filled circles) belong to MCI neurons, together with one non-PDF-ir VNE (diagonally striped open circle). Non-backfilled neurons (diagonally striped open circles) include the small aPDFMEs and other ipsilaterally remaining non-PDF-ir neurons. AL = antennal lobe; CA = calyx; ILP = inferior lateral protocerebrum; LAL = lateral accessory lobes; LAO = lamina organ; LO = lobula; MB = mushroom body; PB = protocerebral bridge; VLP = ventrolateral protocerebrum.
It is not understood why an unusually high number of neuropeptides colocalize in single AME neurons. Often, the same combinations of neuropeptides occur in input, local, and projection neurons of the AME. It is possible that they form genetically encoded, neuropeptide-dependently labeled lines that allow for activation of a neuronal circuit that controls a specific behavior (Stengl and Arendt, 2016). The genetically encoded neuropeptide-dependently labeled lines from input to output have been suggested to function in the following way: a stimulus, such as light, activates a neuropeptidergic neuron that releases a neuropeptide such as PDF. PDF then activates all PDF-sensitive cells that express PDF receptors, including PDF-releasing cells with autoreceptors for PDF. The PDF-secreting and PDF-sensitive cells synchronize via positive feed-forward interactions, which generates an ensemble of PDF cells that fire action potentials at the same time. If these synchronized PDF-sensitive cells synapse on the same clock output to locomotor control centers, the synchronously occurring excitations of the output cell will sum to allow the cell to reach spike threshold. Thus, PDF-dependent ensemble formation activates (gates) an output of the clock to control locomotor activity (Schneider and Stengl, 2005; Stengl and Arendt, 2016). A prerequisite for such a neuropeptide-dependently labeled line circuit would be that the neuropeptide X-secreting presynaptic cell can activate itself and the next neuropeptide X-secreting cell via autoreceptors, using positive feed-forward signaling. Autoreceptors are expressed in the plasma membrane of a neurosecretory cell that detects the neuropeptide that the respective cell synthesizes and releases. Therefore, if a PDF autoreceptor is excitatory, the PDF-releasing neuron would excite itself, sustaining its PDF release, even for hours. Thus, even a brief input signal would result in sustained PDF release. If the PDF autoreceptor is inhibitory, the PDF-releasing neuron would inhibit itself, and the negative feedback would maintain the phasic activity of the clock cell (shorter than the input signal), possibly even restraining it to durations of a few milliseconds. Thus, inhibitory autoreceptors are ideally suited for transient gating (on/off signals) of a clock output, or for transient phase shifts of postsynaptic oscillators. In contrast, excitatory autoreceptors are suited to maintain cell activity (and the cell’s output such as PDF release) throughout long periods of time, even after a very transient signal. In Drosophila, autoreceptors for PDF are present in about half of the PDF-expressing neurons (Shafer et al., 2008). In the cockroach, however, it is unknown whether PDF-ir neurons directly innervate themselves and other PDF-sensing and -releasing neurons.
In this study, we used a combination of backfill, ultrananocrystalline diamond (UNCD) films, Ca2+ imaging analysis, and immunocytochemistry to examine PDF sensitivity of ipsilaterally and contralaterally projecting AME neurons. Contralaterally projecting pacemaker cells have been previously predicted to control locomotor activity rhythms via the bilaterally symmetric clocks (Page, 1978). Lesion and transplantation experiments have shown that PDF-ir AME neurons control locomotor activity rhythms in the Madeira cockroach (Stengl and Homberg, 1994; Reischig and Stengl, 2003a). Thus, our focus was to determine whether contralaterally projecting aPDFMEs that control locomotor activity rhythms possess autoreceptors for PDF. In addition, we tested whether PDF-sensitive AME neurons respond to GABA, the most prominent inhibitory neurotransmitter, or to the excitatory neurotransmitter ACh. It has previously been demonstrated that ACh activates almost all of the AME neurons in primary cell culture (Baz et al., 2013). Unexpectedly, we found that all contralaterally projecting PDF-sensitive AME neurons were inhibited by PDF. In contrast, all ipsilaterally remaining PDF-sensitive AME neurons were activated by PDF. PDF autoreceptors were found in all ipsilaterally and contralaterally projecting aPDFMEs recorded, except for the largest aPDFME. All of the aPDFMEs recorded were excited by ACh and inhibited by GABA. These results further support our previously suggested hypothesis of a PDF-dependently labeled line connectivity pattern (Stengl and Arendt, 2016). Our findings provide evidence for 2 antagonistically phase-locked PDF-dependent subnetworks in the AME that maintain a constant phase relationship between rest and activity rhythms in insects.
Materials And Methods
Animals
Adult male Madeira cockroaches (Rhyparobia maderae) were raised in laboratory colonies at the University of Kassel in a temperature of about 26 °C with 60% relative humidity, and maintained on a 12:12-h light–dark cycle with lights on at 08:00 am. All physiological experiments were performed during the day.
Backfill with Rhodamine-Dextran
To identify contralaterally projecting AME neurons and to increase the probability of detecting PDF-ir pacemaker neurons, backfill experiments with dextran-labeled rhodamine from the contralaterally cut optic stalk were performed (n = 117). The rhodamine-dextran backfills were accomplished as reported by Reischig and Stengl (2002). Briefly, about 100 nl of 5% rhodamine-dextran solution in an application capillary with a tip opening of 300–400 µm (Molecular Probes, 3000 MW) was slipped over the stump of the removed optic stalk. After the operation, the head capsule was closed with wax. The animals were kept in the refrigerator (4 °C) overnight to allow intracellular transport of the tracer.
Ultrananocrystalline Diamond (UNCD) Films
To strongly attach the dispersed neurons in primary cell cultures, we developed a new surface. UNCD films prepared by microwave plasma chemical vapor deposition on silicon wafers were used as substrates for the biological tests. To locate physiologically recorded cells during immunocytochemistry on the coverslips, titanium grids with a thickness of 50 nm were integrated beneath the UNCD layer (with a typical thickness of about 700 nm). The films were composed of diamond nanocrystallites with a diameter of up to 10 nm embedded in an amorphous carbon matrix. Their topography is characterized by rounded features with diameters of several hundred nanometers, which themselves possess a substructure. The root-mean-squared roughness of the films was 9–13 nm. The surface of the as-grown UNCD films was H-terminated and hydrophobic (with contact angles against water of 70–80°). UV/O3 treatment for 10 min in air rendered the surface O-terminated and hydrophilic (contact angles of less than 10°). No changes in the topography were observed after the modification. Details about the deposition of UNCD films, their bulk and surface properties before and after the modification, and the technological steps for incorporation of the Ti grid structure are provided in a previous publication (Voss et al., 2016).
Primary Cell Cultures and Calcium Imaging
Primary cell cultures (n = 156 cultures from 117 backfilled animals) of AME neurons from ipsilateral optic lobes were prepared under sterile conditions by mechanical dissociation and enzymatic treatment, as described previously (Petri and Stengl, 1999). The cell suspension was plated on UV/O3-treated UNCD films with integrated grids, which enabled cell adhesion and identification of individual neurons after physiological characterization and immunocytochemistry. The procedure for loading AME cells with Fura-2 and the measurement of their intracellular Ca2+ concentration changes using a Till photonic imaging system were described in Wei and Stengl (2012). Briefly, the primary cell cultures were loaded with 4 µM Fura-2 acetoxymethyl ester (Fura-2-AM, Molecular Probes). The contralaterally projecting neurons labeled with the fluorescent dye rhodamine could be identified using another fluorescence filter set. The cell cultures were continuously superfused with standard Ringer’s solution. The stimulus solutions (500 nM PDF: NSEIINSLLGLPKVLNDA, Iris Biotech, Marktredwitz, Germany; 10 µM GABA and 1 µM ACh) were bath-applied. Stimuli were applied via switching lines between bath/vehicle control solutions and bath/vehicle solutions containing the respective stimuli, such as ACh. We controlled for the specificity of stimulus-dependent responses with repetitive application of the drugs, with different doses to ensure dose dependence of the drug response, with application of a modified PDF neuropeptide with scrambled sequence but the same number of amino acids, and with vehicle controls (Baz et al., 2013; Wei et al., 2014). None of the controls simulated drug effects. Switching lines between 2 lines that both contained bath/vehicle control solutions did not produce any reproducible effects, nor did vehicle applications via switching from stimulus lines to bath application lines (Suppl. Fig. S1) (Wei et al., 2014). The concentrations of the drugs applied were determined in previous studies (Baz et al., 2013; Wei et al., 2014).
Immunocytochemistry for Primary Cell Cultures
After the physiological analysis, the PDF-ir AME neurons were identified with immunocytochemistry using antisera against the neuropeptide PDF (Dr. H. Dircksen, Stockholm, Sweden; n = 156 experiments). A necessary prerequisite for immunocytochemistry with the primary cell cultures was the strong attachment of the single cells on the UNCD films. The primary cell cultures were fixed after Ca2+ imaging analysis with 4% paraformaldehyde in 0.1 M phosphate-buffered saline (PBS) for 30 min. They were subsequently washed 3 times for 10 min with 0.1 M PBS and preincubated at room temperature with 5% normal goat serum (NGS, Jackson ImmunoResearch, West Grove, PA, USA) for 30 min. Afterward, the primary cell cultures were incubated overnight with anti-PDF (1:80,000) in PBS containing 1% NGS at 4 °C. After washing (3 times, 10 min each), the primary cell cultures were incubated with Alexa 647-tagged secondary antibody (1:500, Jackson ImmunoResearch) and DAPI (0.1 µg/ml) for 3 h. After staining, the primary cell cultures were washed for 10 min and analyzed with fluorescence microscopy.
Results
The PDF-ir networks of the cockroach circadian pacemaker center in the AME consist of ipsilateral and contralateral PDF-ir inputs, ipsilateral PDF-ir local neurons, and ipsilateral and contralateral PDF-ir outputs (Fig. 1A and 1B). We determined whether contralaterally projecting (referred to as contralateral) and ipsilaterally remaining (referred to as ipsilateral) PDF-expressing neurons express PDF autoreceptors. In addition, we examined which other types of AME neurons respond to PDF, ACh, or GABA. To accomplish these tasks, neuroanatomical and physiological studies were combined. First, with rhodamine-dextran backfills (n = 117) from the contralateral optic stalk, all AME cells that project to the contralateral optic lobe were labeled (Fig. 2A1). Theoretically, the backfills increase the probability to identify PDF-ir pacemaker cells to about 16%, because among all contralateral AME neurons are 4 PDF-ir aPDFMEs (Söhler et al., 2011). In contrast, among the many non-rhodamine-labeled ipsilateral medulla neurons are 4 small, 1 medium-sized, and 3 large aPDFMEs (Petri et al., 1995; Reischig and Stengl, 2002, 2004; Söhler et al., 2011; reviews: Stengl et al., 2015; Stengl and Arendt, 2016). After the backfills, the ipsilateral AME with surrounding neurons was excised and dispersed into single cells. Among the excised AME neurons were also the rhodamine-dextran-labeled contralateral aPDFMEs and other contralaterally projecting pacemaker neurons. The primary cell cultures (n = 156 experiments) were plated on coverslips with UNCD surfaces with integrated grids (Fig. 2A1–D3). These specially developed coverslips allowed for stronger adhesion of the cells. In addition, the grids allowed us to identify and track individual cells during the different experimental settings of Ca2+ imaging and subsequent immunocytochemistry (Fig. 2A–D). Approximately 10 AME neurons per cell culture could be recorded, including backfilled neurons. Neither rhodamine-dextran labeling (Fig. 2A1), nor loading of dispersed AME neurons with Ca2+ indicator Fura-2 (Fig. 2A2) for Ca2+ imaging experiments, affected cell viability or physiology (not shown). To identify and physiologically characterize PDF-responsive AME neurons, the cultured pacemaker neurons were stimulated with PDF, GABA, and ACh in Ca2+ imaging experiments (n = 1557 cells stimulated in 156 experiments). Then, immunocytochemistry with antisera against PDF was performed (n = 156 experiments; Fig. 2A3; 2B1,3; 2C1,3; and 2D1,3). Of the 156 primary cell cultures with approximately 1557 AME neurons, a total of 98 (6%) backfilled contralateral neurons were physiologically and immunocytochemically examined (Fig. 2A1 and Table 1). Of the neurons examined, 16% were contralateral PDF-ir neurons: 3 large aPDFMEs (Figs. 1 and 2B1–3) and 13 medium-sized aPDFMEs (Figs. 1 and 2C1–3). Of the 1459 non-backfilled neurons, 7 small aPDFMEs (0.5%) were characterized (Figs. 1 and 2D1–3). About 10% (n = 149 of 1459) of ipsilateral and 44% (n = 43 of 98) of contralateral AME neurons responded to PDF. Of the 1557 clock cells, all of the PDF-ir neurons also responded to GABA and ACh. Of all non-PDF-ir AMEs, 60% (n = 928) responded to GABA and 92% (n = 1409) to ACh (Table 1).
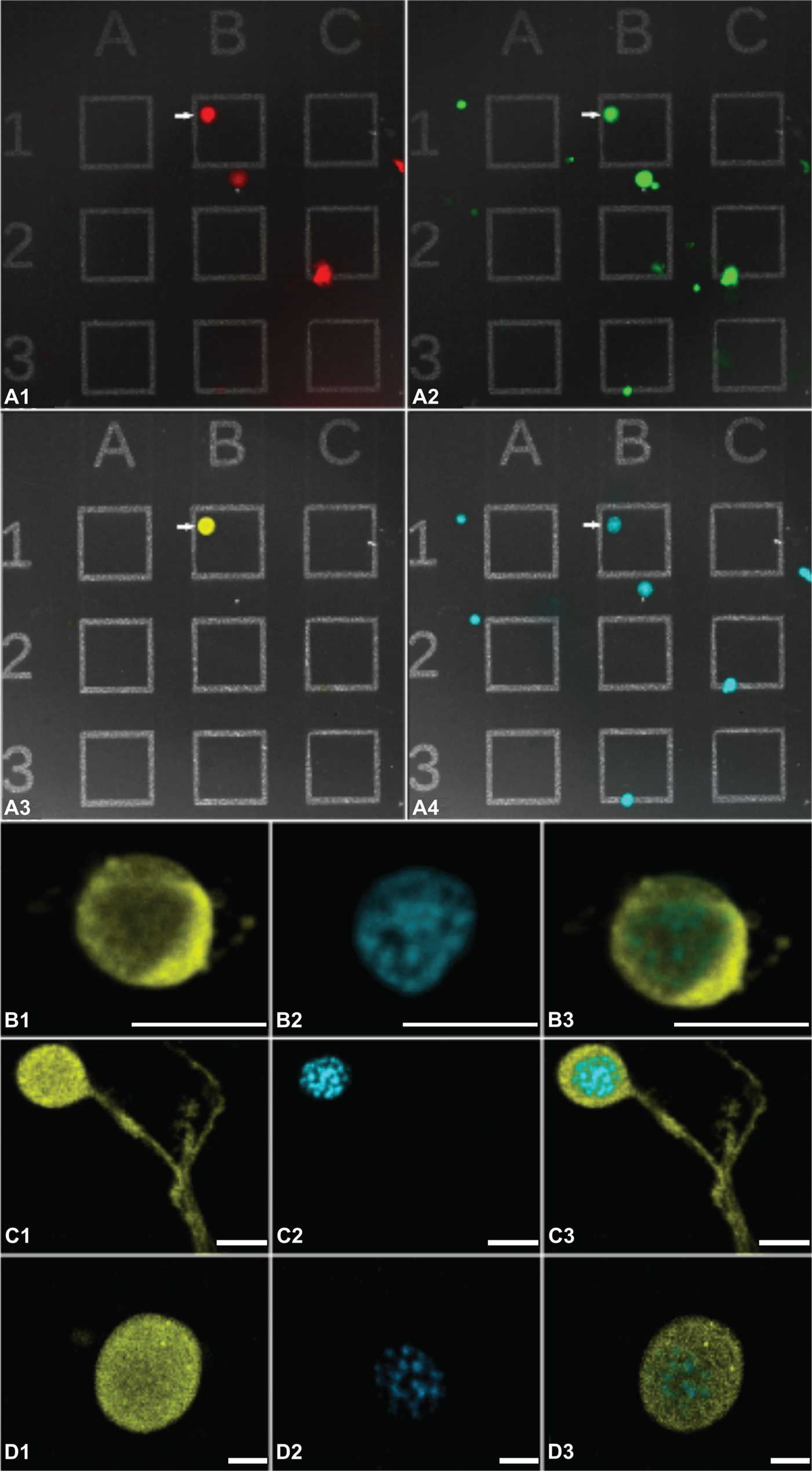
Backfills and immunocytochemistry combined with Ca2+ imaging identified and characterized contralateral anterior pigment-dispersing factor (PDF)-expressing neurons (aPDFMEs) of the accessory medulla (AME) in primary tissue culture (n = 1557). Before the ipsilateral AME was dispersed into single cells, rhodamine-dextran backfills from the contralateral optic stalk labeled all neurons next to the AME that project into the contralateral optic lobe (A1, red somata). To allow for identification of single cells, the primary cell cultures of the ipsilateral AME were plated on an ultrananocrystalline diamond (UNCD) surface with integrated grids. Next, they were loaded with Ca2+ indicator Fura-2 (A2, green) for physiological characterization. After Ca2+ imaging, anti-PDF immunocytochemistry was performed to identify PDF-ir neurons in the primary cell cultures (A3, yellow), and DAPI nuclear staining (A4, cyan) labeled all cells on the grids for quantitative analysis. The arrow marks the contralateral largest aPDFME that was identified according to its characteristic size, rhodamine labeling, and PDF labeling. Confocal images (B–D) show enlargements of one largest (B1–3), one medium-sized (C1–3), and one small aPDFME (D1–3) that were identified with anti-PDF immunocytochemistry (yellow) and stained with DAPI (cyan) on the grids, after physiological analysis. Double labeling is shown in the overlay (D3). Scale bars = 50 µm in B1–3, 10 µm in C1–3, and 5 µm in D1–3.
Summary of pigment-dispersing factor (PDF)-immunoreactive (-ir) neurons (aPDFMEs) stimulated with PDF, GABA, and acetylcholine (ACh).

To summarize, with backfills from the contralateral optic lobe, we could distinguish ipsilateral and contralateral clock neurons. The backfills significantly increased the probability to detect contralateral PDF-ir neurons that control locomotor activity rhythms. We found that many more contralateral AME neurons responded to PDF than ipsilateral neurons. In addition, whereas nearly all AME cells were activated by ACh, only about half were sensitive to GABA. In contrast, all PDF-expressing clock neurons were inhibited by GABA and excited by ACh.
Application of PDF Inhibited Contralateral Non-PDF-ir Neurons and Excited Ipsilateral Non-PDF-ir Neurons
In 156 cultures, 82 (5%) of the 1557 AME neurons recorded were labeled with rhodamine, but lacked PDF-ir (Figs. 2A1 and 3A–G). Spontaneous Ca2+ transients, indicative of action potential bursts (Wei and Stengl, 2012), occurred in 68% of the non-PDF-ir contralateral AME neurons (Table 1; 56 of 82). Application of PDF (500 nM) blocked 38% of all spontaneously active contralateral clock neurons (21 of 56; Fig. 3A). PDF also decreased the baseline Ca2+ level in 16% (n = 9) of the silent (not spontaneously active) contralateral clock neurons (Fig. 3B). None of the contralateral non-PDF-ir but PDF-sensitive neurons were activated by PDF application (n = 82). Among the non-PDF-ir but PDF-sensitive contralateral clock cells, 87% were inhibited by GABA and 94% were activated by ACh (n = 82; Fig. 3E–G). Statistical analysis confirmed the significance of PDF, GABA, and ACh effects in PDF-sensitive, contralateral non-PDF-ir neurons (Fig. 3E–G and Table 1).
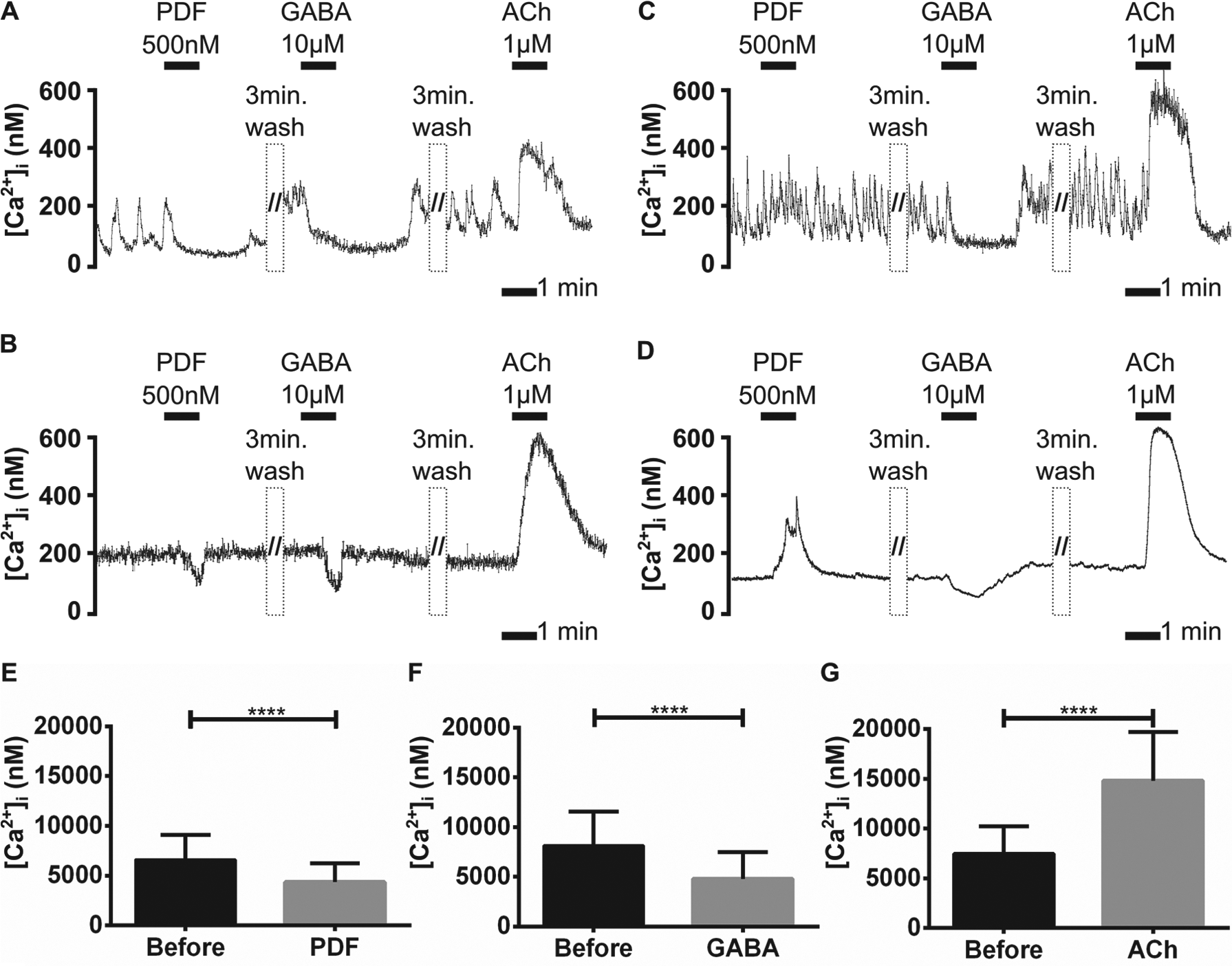
The contralateral non-PDF-ir accessory medulla (AME) neurons were blocked by pigment-dispersing factor (PDF) application, whereas the ipsilateral non-PDF-ir AME neurons were activated via PDF. The rhodamine-labeled contralateral AME neurons (n = 82) (A,B) and the ipsilateral AME cells (n = 1452) (C,D) that both lacked PDF immunoreactivity were grouped into spontaneously active (n = 270) (A,C) and silent cells (n = 1264) (B,D) that did not express Ca2+ transients. In a Ca2+ imaging experiment, application of 500 nM PDF and also 10 µM GABA blocked spontaneous Ca2+ transients in contralateral active clock cells (n = 56) (A), or decreased baseline Ca2+ levels in contralateral silent clock cells (n = 26) (B). The ipsilateral clock cells were blocked by GABA and never displayed an inhibition after PDF treatment. Rather, baseline Ca2+ levels increased PDF-dependently in both the active (C) and the silent (D) ipsilateral clock cells. Thus, PDF affected AME neurons cell-type specifically, and acetylcholine (ACh) elevated intracellular Ca2+ levels in all cell types alike. The intracellular Ca2+ baseline levels of contralateral non-PDF-ir AME neurons were significantly reduced with PDF and GABA application, but they were increased with addition of ACh (n = 30) (E–F). Bars represent the mean (±SD) of Ca2+ baseline changes resulting from (E) PDF 500 nM, (F) GABA 10 µM, and (G) ACh 1 µM applications in contrast to saline controls applied before (asterisks; paired t test; <0.0001; GraphPad Prism6 Software).
Ipsilateral non-PDF-ir neurons lacked rhodamine and PDF-ir (n = 1452). Only 15% (n = 214 of 1452) were spontaneously active. In 10% of the ipsilateral, non-PDF-ir neurons (142 of 1452), the frequency of oscillating Ca2+ transients or the Ca2+ baseline concentration increased after PDF application (Fig. 3C and 3D and Table 1) (Wei et al., 2014). PDF-dependent inhibition of ipsilateral non-PDF-ir but PDF-sensitive neurons was not observed in any of the experiments. About 59% of the ipsilateral non-PDF-ir but PDF-sensitive clock cells were inhibited by GABA, and 92% were activated by ACh (n = 1452) (Fig. 3C and 3D; Baz et al., 2013). Statistical analysis (Fig. 4A and 4B) confirmed the significance of GABA and ACh effects in the contralateral non-PDF-sensitive clock cells.
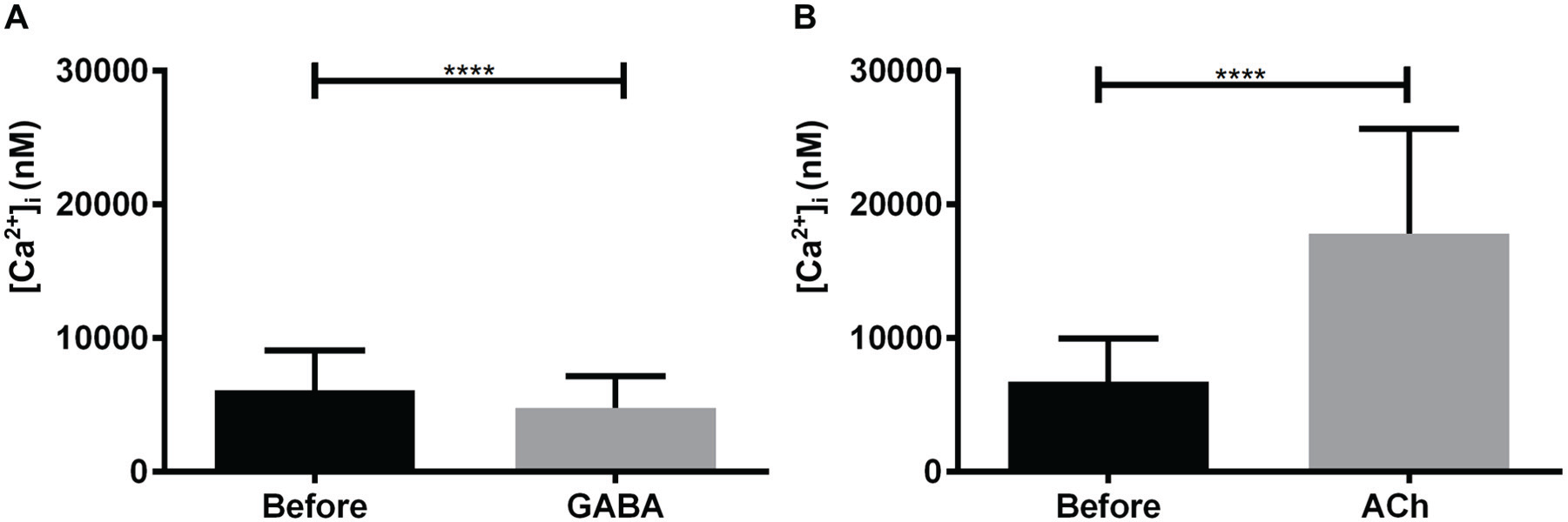
The intracellular Ca2+ baseline levels of contralateral non-PDF-ir, non-PDF-sensitive accessory medulla (AME) neurons were significantly reduced via GABA application, but they were increased with addition of acetylcholine (ACh) (n = 71). Bars represent the mean (±SD) of Ca2+ baseline changes resulting from (A) GABA 10 µM and (B) ACh 1 µM application in contrast to saline control before (asterisks; paired t test; <0.0001; GraphPad Prism6 Software).
To summarize, the orientation of the non-PDF-synthesizing AME neurons determined their PDF sensitivity; all of the contralateral PDF-sensitive clock cells were inhibited by PDF, whereas all ipsilateral cells were activated by PDF. In addition, most contralateral non-PDF-ir clock cells expressed spontaneous activity, and 4 times as many were PDF-sensitive compared with the respective ipsilateral clock cells. Furthermore, more than twice as many of the PDF-sensitive clock neurons were spontaneously active compared with all non-PDF-sensitive clock cells.
The Largest PDF-ir Neuron Does Not Contain PDF Autoreceptors
In 3 (3%) of 98 contralateral-projecting neurons of the AME, the largest aPDFME was found. It was identified according to its characteristically large soma with a diameter of up to 50 µm and its rhodamine-dextran labeling, which identified it as a contralateral AME neuron (Figs. 2A1–4, 2B1–3, and 5A–C). The largest aPDFME spontaneously generated regular Ca2+ transients with large amplitudes (≥100 nM) (Fig. 5A–C). Bath application of PDF (500 nM) did not affect Ca2+ transients (Fig. 5A–C and Table 1). Although Ca2+ transients were blocked after application of GABA (10 µM), the Ca2+ baseline was not largely affected. In contrast, ACh (1 µM) increased the Ca2+ baseline (Fig. 5A–C) and the frequency of the Ca2+ transients.
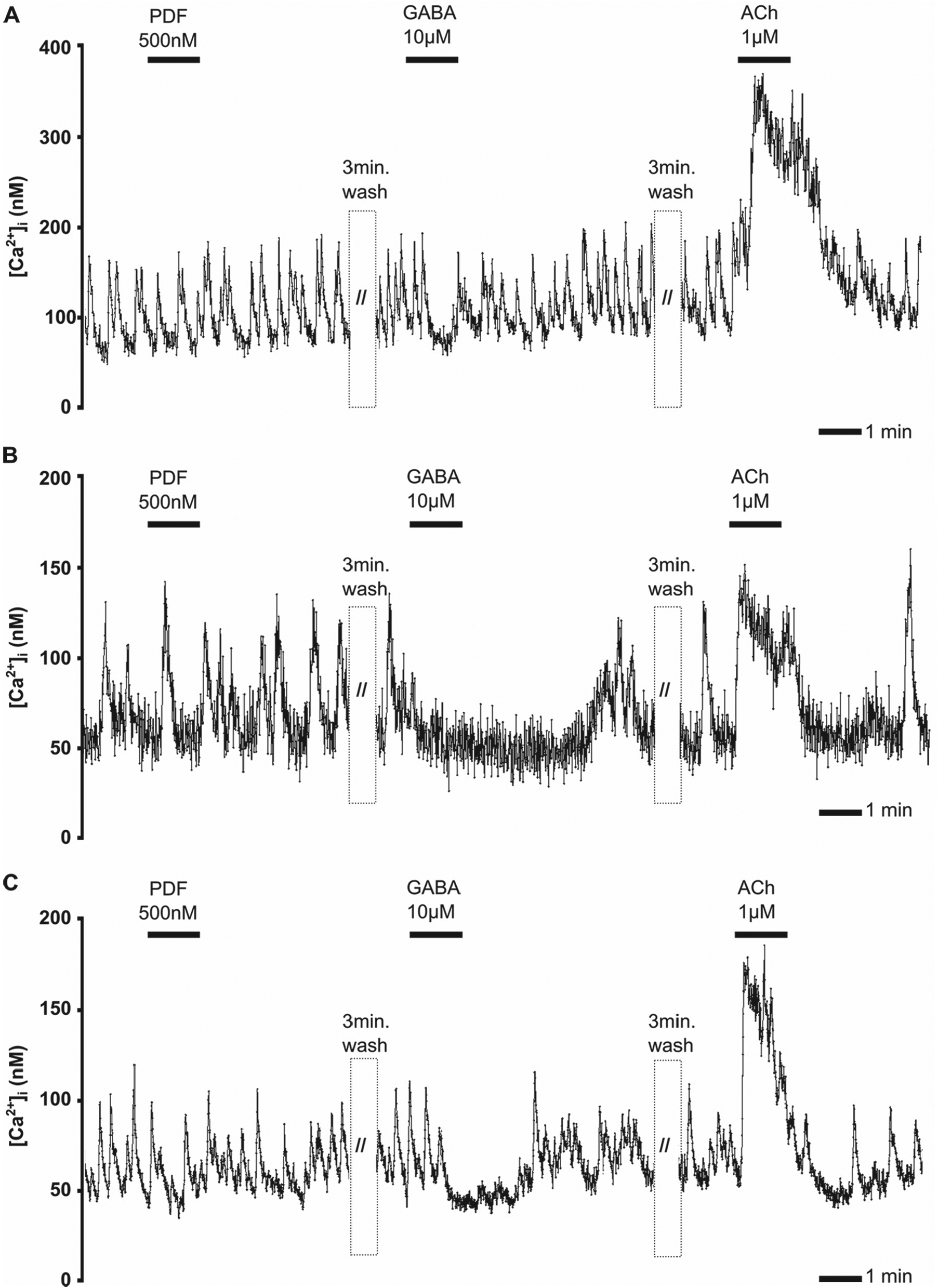
The largest pigment-dispersing factor (PDF)-expressing neurons anterior to the accessory medulla (aPDFMEs) do not express PDF autoreceptors. In 3 of 156 experiments, the backfilled largest aPDFMe was identified with anti-PDF antisera after physiological characterization (Fig. 2A and 2C). Spontaneous regular Ca2+ transients of the 3 largest aPDFMEs (A–C) were not affected by PDF (500 nM) but were blocked by GABA (10 µM), transiently (A) or over several minutes (B,C). All 3 cells were strongly activated by acetylcholine (ACh) (1 µM).
To summarize, the largest aPDFME, which projects to all ipsilateral and contralateral output targets of the circadian clock, produces very regular spontaneous bursts of activity. It expresses receptors for the neurotransmitters GABA and ACh, but is not sensitive to PDF.
Contralaterally Projecting Medium-Sized aPDFMEs Are Inhibited by PDF
Rhodamine- and PDF-labeled medium-sized aPDFMEs (10–20 µm soma diameter; Fig. 2C1–3) were identified in the primary cell cultures (n = 13 of 98 rhodamine-labeled cells; Table 1). Most showed spontaneous Ca2+ transients throughout the recording (Fig. 6A and 6C and Table 1). The other cells recorded showed little activity before PDF application and became more active with an increased baseline level after washing off PDF (Fig. 6B). Application of 500 nM PDF abolished the Ca2+ transients and transiently decreased the Ca2+ baseline in most cells (Fig. 6A and 6C). In other medium-sized aPDFMEs, PDF caused a sustained decrease of the Ca2+ baseline (Fig. 6B). Thus, medium-sized aPDFMEs contain inhibitory but not excitatory PDF autoreceptors (Fig. 6D and Table 1). Application of GABA blocked the Ca2+ transients and reduced the Ca2+ baseline in all contralateral medium-sized aPDFMEs recorded (Fig. 6 A–C and 6E). Perfusion with ACh rapidly increased the Ca2+ baseline level of all medium-sized aPDFMEs (n = 13; Fig. 6A–C and 6F).
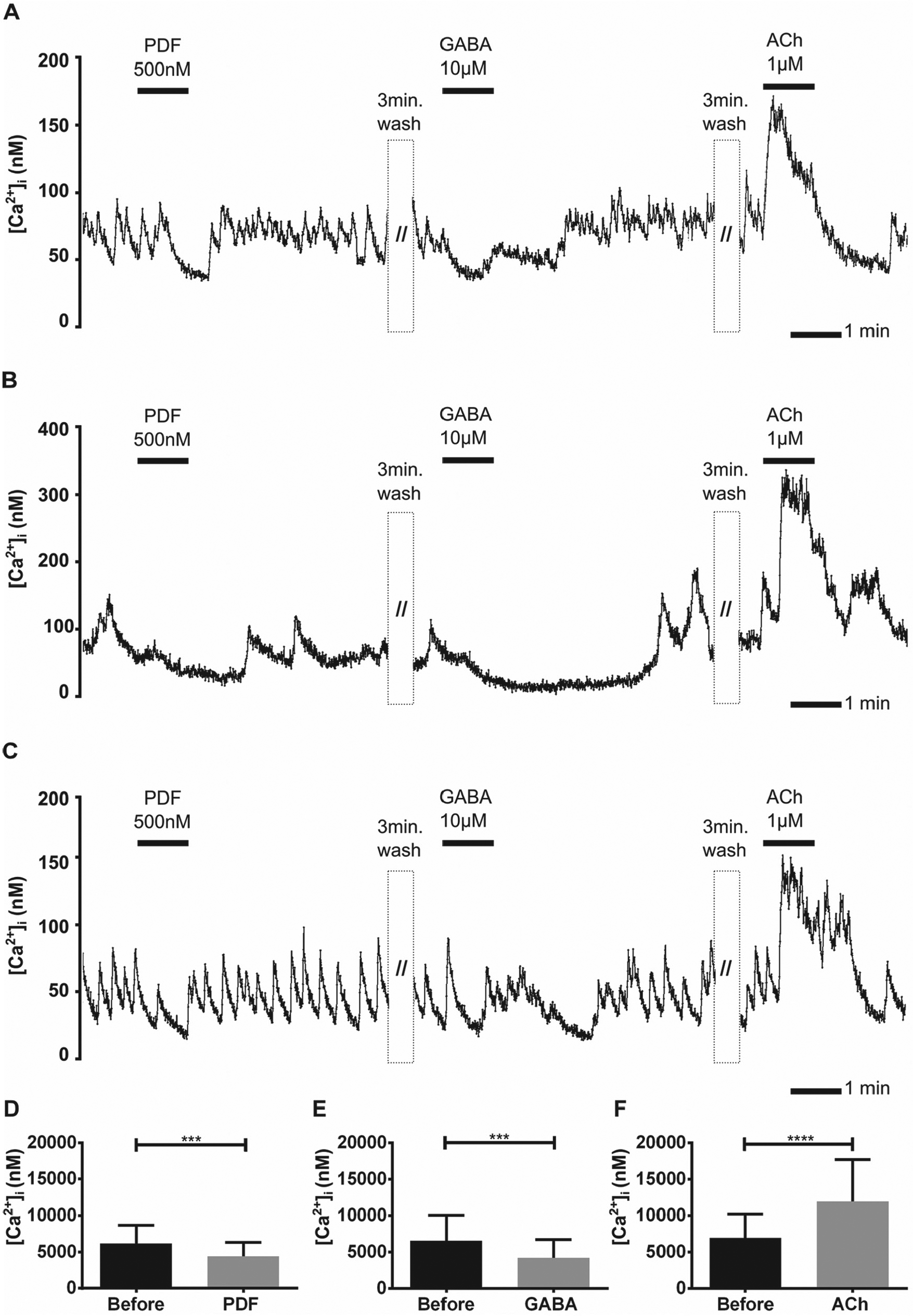
The contralateral medium-sized aPDFMEs (pigment-dispersing factor [PDF]-expressing neurons anterior to the accessory medulla) were inhibited via PDF autoreceptors. (A–F) The Ca2+ baseline was reduced and the Ca2+ transients of all contralateral medium-sized aPDFMEs were blocked by PDF and GABA applications, whereas the cells were activated by acetylcholine (ACh) applications (n = 13). All cells were spontaneously active, and most of the cells (A,C) expressed regular Ca2+ transients. Bars represent the mean (±SD) of Ca2+ baseline changes resulting from (D) PDF 500 nM, (E) GABA 10 µM, and (F) ACh 1 µM applications in comparison to saline control applications (asterisks; paired t test; <0.0002, 0.0002, 0.0001; GraphPad Prism6 Software).
To summarize, contralateral medium-sized aPDFMEs express inhibitory autoreceptors for PDF and were not activated by PDF. In addition, all of these neurons expressed inhibitory receptors for GABA and excitatory receptors for ACh (Fig. 6A–C and Table 1).
Small Local aPDFMEs Are Activated by PDF Application
Small aPDFMEs (5–10 µm soma diameter) were identified (n = 7 of 156 preparations) with PDF immunocytochemistry (Fig. 7A–C). As expected for local interneurons of the AME, the small aPDFMEs were not labeled with rhodamine-dextran (n = 7, not shown). They showed no regular spontaneous activity (Fig. 7A–C); however, PDF application rapidly increased their intracellular Ca2+ baseline levels and generated oscillating Ca2+ transients (Fig. 7A–D and Table 1). In addition, the small aPDFMEs were inhibited by GABA to different extents (Fig. 7A–C and 7E), and all were more uniformly, strongly activated by ACh application (Fig. 7A–C and 7F).
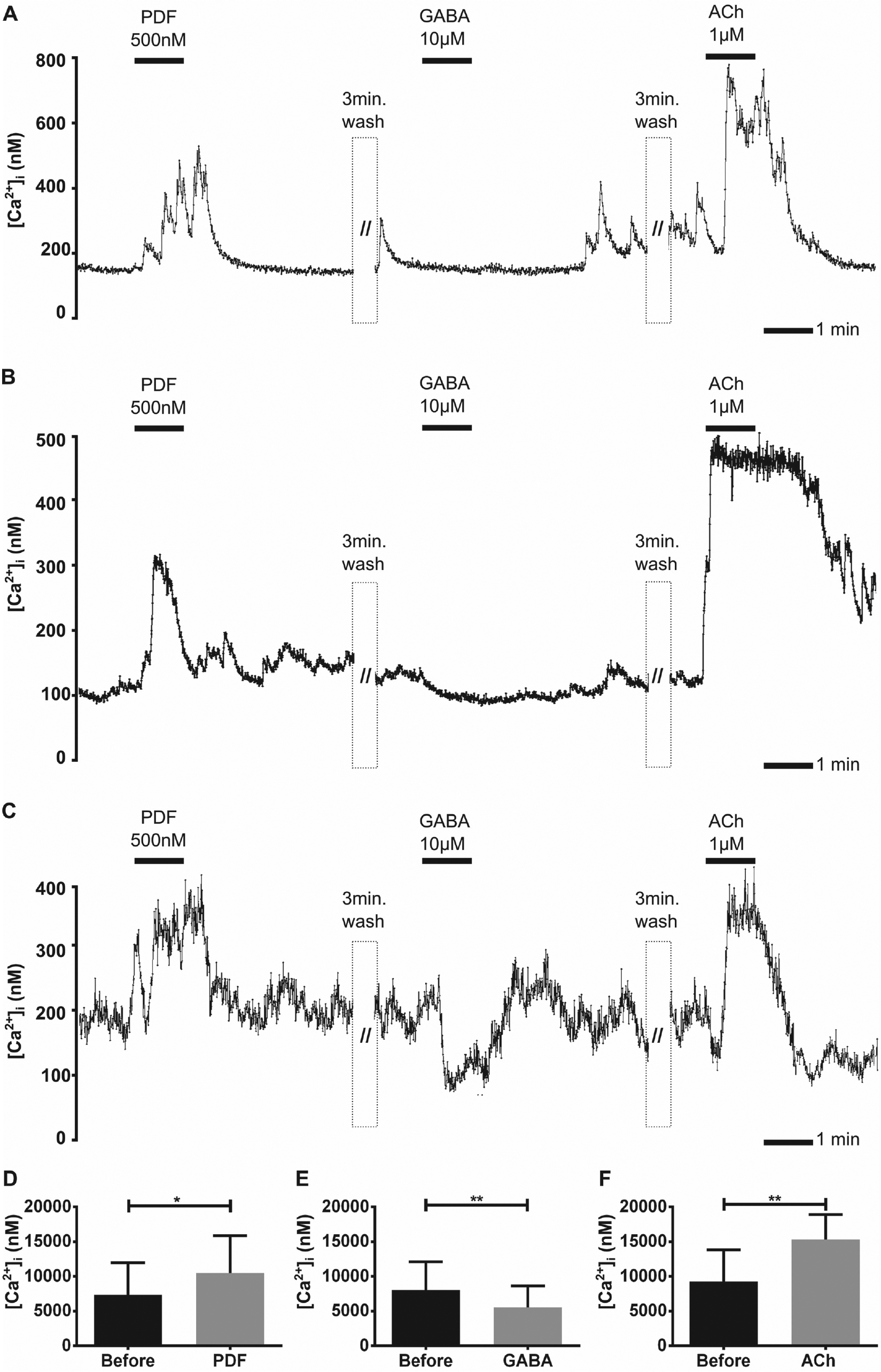
The ipsilateral small aPDFMEs (pigment-dispersing factor [PDF]-expressing neurons anterior to the accessory medulla) expressed excitatory PDF autoreceptors. (A–F) Small PDF-immunoreactive aPDFMEs that were not rhodamine-labeled were examined in Ca2+ imaging experiments (n = 7). Application of PDF (500 nM) and acetylcholine (ACh) applications (1 µM) increased the intracellular Ca2+ baseline level in all local small aPDFMEs. In contrast, they expressed different sensitivity to GABA (10 µM) application. Although the first (A) showed no clear response, the second (B) expressed a small decline of the Ca2+ baseline level, and the third (C) with the highest intracellular Ca2+ level showed a very strong GABA-dependent decline of the intracellular Ca2+ concentration. Bars represent the mean (±SD) of Ca2+ baseline changes resulting from (D) PDF 500 nM, (E) GABA 10 µM, and (F) ACh 1 µM applications in comparison to saline control applications (asterisks; paired t test; <0.0148, 0.0076, 0.0082; GraphPad Prism6 Software).
To summarize, all small ipsilateral PDFMEs were silent local neurons of the circadian clock. All exhibited excitatory autoreceptors for PDF and excitatory receptors for ACh, and they displayed different sensitivities to GABA (Fig. 7A–F).
Discussion
In this study, we examined PDF autoreceptors in a further effort to test our hypothesis of neuropeptide-dependent labeled line encoding (Stengl et al., 2015; Stengl and Arendt, 2016). For the first time, our combination of physiological and anatomical techniques allowed us to distinguish the different aPDFMEs according to their ipsilateral or contralateral branching patterns. We focused on the contralateral clock cells and found that almost half of them responded to PDF, and most were inhibited by GABA and activated by ACh. Among the ipsilateral AME clock cells, however, much fewer (10%) were PDF sensitive, and fewer responded to GABA, but about as many were activated by ACh. Thus, significantly more contralateral AME neurons were more PDF and GABA sensitive than ipsilateral neurons. In contrast to Wei et al. (2014), who identified only 5% PDF-sensitive AME neurons in primary cell cultures of the unlabeled AME, labeling with contralateral backfills significantly increased the probability of identifying PDF-sensitive neurons. The unexpected finding that the opposing PDF sensitivity correlated with the different branching pattern of the PDF-sensitive cells indicates that there are at least 2 PDF-dependent ensembles with different functions in the AME. Both PDF-dependent ensembles would appear to cycle in antiphase: while one is active, the other is inhibited during the time of PDF release. Furthermore, the largest aPDFME appears to be unique among the PDF-ir neurons, because it generated the most regular spontaneous Ca2+ transients and did not express PDF autoreceptors.
Nearly All Circadian Clock Neurons Receive Cholinergic Excitatory Synaptic Inputs
Previous studies have shown that ACh acts as the most prominent excitatory neurotransmitter in the circadian clock, as 96% of all cockroach AME neurons express ionotropic nicotinic ACh receptors (Baz et al., 2013). Among them are apparently all PDF-ir neurons and all contralateral AME outputs. It remains unknown which neurons in the cockroach brain express ACh. Current histochemical studies using acetylcholinesterase staining suggest that there are many different cholinergic neurons innervating the clock (Azar Massah, Julia Schendzielorz, Sebastian Korek, and Monika Stengl, unpublished).
GABAergic Tracts Connect Glomeruli of the AME to the Optic Lobe and Midbrain Neuropil
In the cockroach AME, as in the mammalian suprachiasmatic nucleus, GABA innervation is abundant and is present in local clock neurons and in input and output pathways (Van den Pol and Tsujimoto, 1985; Moore and Speh, 1993; Wagner et al., 1997; Strecker et al., 1997; Liu and Reppert, 2000; Shinohara et al., 2000; Michel and Colwell, 2001; De Jeu and Pennartz, 2002; Petri et al., 1995, 2002). In the Madeira cockroach, the most prominent is the GABA-ir distal tract that connects the medulla to the glomerular neuropil of the cockroach AME (Reischig and Stengl, 1996; Petri et al., 2002). Its function is not known, but it is thought to be involved in homeostatic plasticity, such as gain control mechanisms, and possibly photic entrainment.
Next to the distal tract, there are additional GABA-ir tracts and many GABA-ir local neurons (Petri et al., 2002). GABA, thus, likely plays many different roles, most of which remain unknown. An important function of GABA is, however, synchronization of pacemakers that spike spontaneously in the gamma frequency range (20–80 Hz). Thereby, GABA release forms ensembles of phase-synchronized neurons via GABAA receptor-dependent inhibitions. These ensembles are assumed to activate (gate) specific clock outputs (Schneider and Stengl, 2005). Ongoing research is investigating GABA signaling of specific AME neurons to further characterize GABA functions in the cockroach clock (unpublished).
PDF Release Forms 2 Antagonistic Ensembles with Opposite Activity via Either PDF-Dependent Inhibition or Excitation
In contrast to neurotransmitters, which are copiously distributed in the brain, neuropeptides are typically expressed in smaller clusters of neurons. They are thought to influence specific physiological and behavioral contexts (Nässel and Winther, 2010). Because circadian clock neurons abundantly express colocalized neuropeptides, they appear to be crucial for clock functions. The best characterized neuropeptide is PDF, the most important insect circadian coupling factor that in many ways resembles the mammalian circadian coupling signal vasoactive intestinal polypeptide (Aton et al., 2005; Vosko et al., 2007). In Drosophila, PDF controls circadian locomotor activity rhythms and adapts rest–activity cycles to long-day photoperiods via Zeitgeber time-dependent phase shifts (reviews: Helfrich-Förster, 2014; Taghert and Nitabach, 2012). In the Madeira cockroach, PDF controls circadian rest–activity rhythms, apparently via PDF-dependent phase synchronization, resulting in PDF-dependent ensemble formation (Stengl and Homberg, 1994; Reischig and Stengl, 2003a; Schneider and Stengl, 2005; Stengl et al., 2015). Our current study hints at 2 PDF-dependent ensembles in antiphase. During PDF release, the ipsilateral aPDFMEs are activated via their excitatory PDF autoreceptors. They synchronize and activate each other via positive feed-forward interactions, resulting in continuous PDF release. Thus, even a transient input signal to the ipsilateral aPDFMEs could lead to long-duration PDF release. In contrast to this sustained activation of the ipsilateral PDF cells, the activity of contralateral PDF-sensitive cells is suppressed when PDF is released in the AME (Fig. 8).
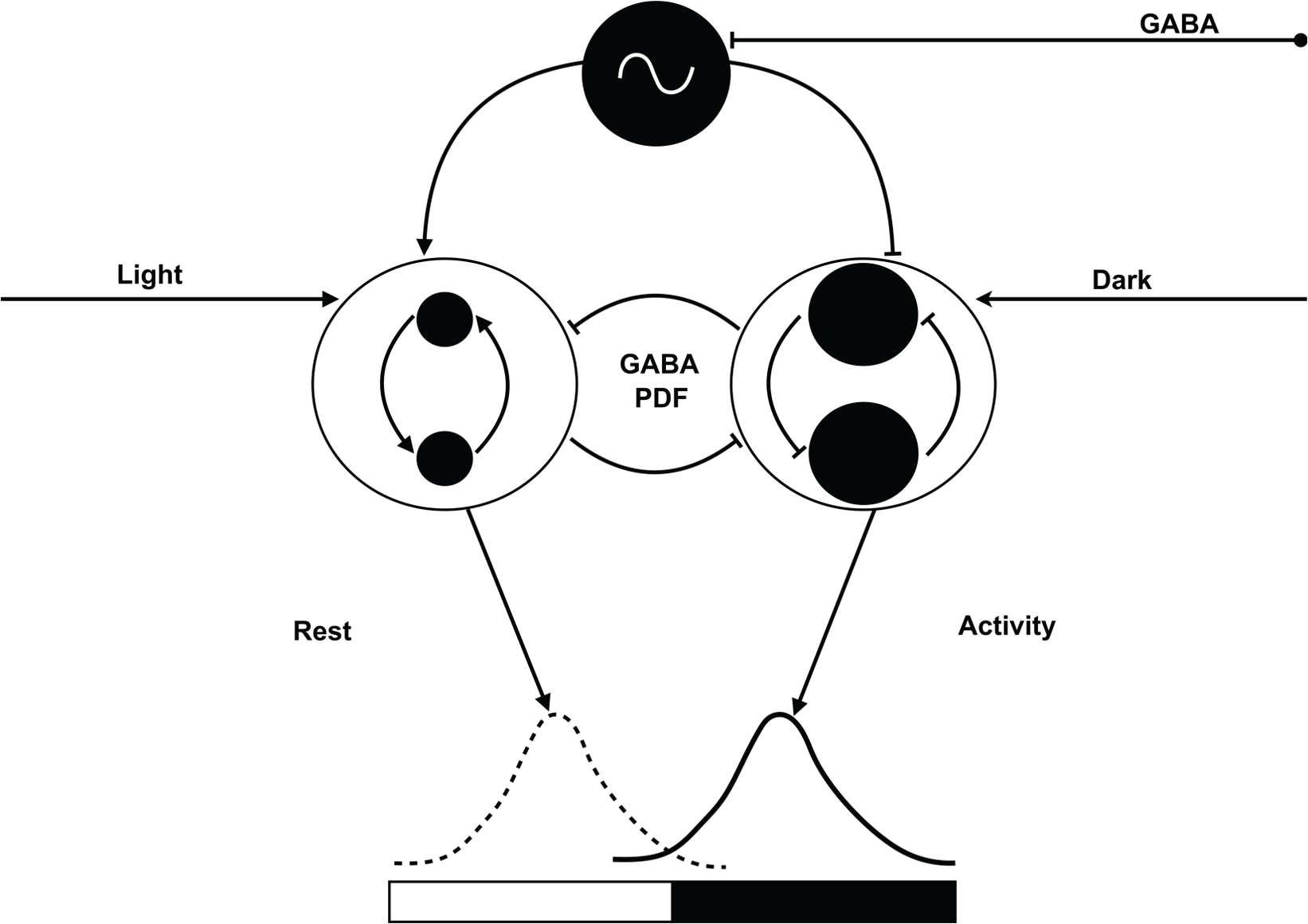
Our new hypothesis explains the unexpected finding of cell-type-specific antagonistic PDF sensitivity of PDF-releasing circadian clock neurons (aPDFMEs = black) of the Madeira cockroach. The largest aPDFME (on top; black with oscillator) expresses regular endogenous circadian action potential rhythms. It is suggested to keep all PDF-sensitive ensembles that are either activated or inhibited via PDF at stable antiphase-relation. The largest aPDFME itself can be phase-shifted GABA- and ACh-dependently, possibly via indirect light input to keep a stable phase to the light–dark cycle. During the day, PDF expression and release are upregulated light-dependently in ipsilateral small aPDFMEs that activate each other via positive feed-forward (left side). They recruit an ensemble of ipsilateral PDF-sensitive neurons that are sleep-promoting during the day. During the day, the small ipsilateral aPDFMEs inhibit (middle) contralaterally projecting medium-sized aPDFMEs that drive an activity-promoting ensemble (right side). The contralateral medium-sized aPDFMEs express inhibitory PDF autoreceptors and inhibit its respective counterparts in the bilaterally symmetric circadian clocks. At dusk, when light levels decline, the contralateral medium-sized aPDFMEs become disinhibited and are activated in darkness. They are locomotor activity promoting in the night-active Madeira cockroach.
Light Stimulates PDF Expression in Specific PDF Neurons, Apparently Recruiting One PDF-Dependent Ensemble during the Day
The time of day that PDF is released remains unknown. It has been shown previously, however, that exposure to long or short photoperiods, or days of different lengths, changes the number of PDF-expressing neurons (Wei and Stengl, 2011). Thus, light differentially regulates PDF synthesis in aPDFMEs, upregulating PDF expression and possibly also release in only some of the clock cells. We do not yet know the exact morphology of the light-dependent aPDFMEs. We hypothesize, however, that increasing light levels during the day will increase PDF release in the largest aPDFME and in ipsilateral aPDFMEs only. Then, PDF-dependent feed-forward activation will recruit a PDF-dependent ensemble during the day. We suggest that this PDF-dependent ipsilaterally remaining ensemble is sleep-promoting for the night-active Madeira cockroach (Stengl and Arendt, 2016).
In turn, during the day, PDF release by the ipsilateral ensemble will inhibit the contralateral PDF-sensitive clock neurons, such as 3 of the medium-sized aPDFMEs identified before (Söhler et al., 2011). Page (1978, 1984) has shown with microsurgery that both bilaterally symmetric circadian clocks control locomotor activity rhythms via inhibitory interactions with projections to the contralateral clock. Thus, contralateral clock outputs control locomotor activity and are also able to inhibit the contralateral clock’s output. This is consistent with our hypothesis that the second, contralateral PDF-sensitive ensemble is activity promoting but can also block the contralateral clock’s output via PDF. This contralateral PDF ensemble would be blocked during the day, when the ipsilateral sleep-promoting ensemble is active. It would regain activity when light levels decline at the end of the day, correlating with locomotor activity onset of the Madeira cockroach (Fig. 8). Current enzyme-linked immunosorbent assays (ELISAs) test our hypothesis that PDF release is high during the day and low during the night (unpublished).
The PDF-dependent biphasic phase-response curve supports our hypothesis that PDF is released during the day. PDF injections delay onset of circadian locomotor activity rhythms late in the day, apparently delaying decline of endogenous PDF levels. In addition, PDF injections advanced locomotor activity onset at the end of the subjective night, apparently advancing the increase of endogenous PDF levels (Petri and Stengl, 1997; Schendzielorz and Stengl, 2014; Stengl et al., 2015). Thus, the different, antagonistic PDF signaling allows for daytime-dependent separation of 2 ensembles, the day-active ipsilateral and the night-active contralateral PDF ensembles. In addition, PDF release in the cockroach clock, possibly also driven by the largest aPDFME, maintains a stable phase difference of these neuronal ensembles via antagonistic PDF actions that have also been reported for the fruit fly (Liang et al., 2016; review: Helfrich-Förster, 2014). Current physiological studies in vivo test our hypothesis (Fig. 8).
The Largest aPDFME Appears to Control the Phase of All PDF-Sensitive Clock Circuits
The function of the largest aPDFME that is not PDF-sensitive but appears to branch to all PDF-dependent target neuropils in the midbrain and optic lobe (Wei et al., 2010) remains unknown. Its unusual morphology and physiology would allow for phase control of all PDF-dependent ensembles via a stimulus other than PDF. In accordance, Page (1978, 1982, 1984) has previously predicted that circadian clock outputs to locomotor control areas are independent of pathways controlling the phase of clock circuits. In Drosophila, at least some of the PDF-ir large lateral-ventral neurons (l-LNvs) do not express PDF autoreceptors (Shafer et al., 2008), and they control rest–arousal rhythms via connections to the upper division of the central complex (Chung et al., 2009; Parisky et al., 2008; Sheeba et al., 2008a). In particular, they regulate sleep levels and sleep latency (Liu et al., 2014). The general morphology of the largest aPDFME does not resemble branching patterns of the l-LNvs of the fruit fly, because it projects via both the posterior and anterior optic commissure. Only its posterior commissure projections arborize in the posterior optic tubercles that are closely connected to the protocerebral bridge of the central complex. Its anterior commissure projections appear to contact mushroom body outputs in the superior medial protocerebrum and neurosecretory control centers in the superior lateral protocerebrum (Fig. 1) (Wei et al., 2010). Thus, the largest aPDFME might control the phase of both sleep- and activity-promoting neuropils in the Madeira cockroach, keeping both at a stable phase difference.
It remains to be examined whether the largest aPDFME is controlled primarily by light or by an endogenous rhythm. It may express endogenous circadian activity that is phase-controlled by both GABA- and ACh-dependent innervations, as both the largest aPDFME of the Madeira cockroach and the l-LNvs of fruit flies receive excitatory nicotinic cholinergic input and GABAergic inhibitory input through GABAA receptors (McCarthy et al., 2011; Muraro and Ceriani, 2015; Chung et al., 2009; reviews: Helfrich-Förster, 1998, 2014). Also, both the largest aPDFME in the Madeira cockroach and the l-LNvs in D. melanogaster are spontaneously bursting neurons during the day (Julia Gestrich, unpublished; Gao et al., 2008; Sheeba et al., 2008b; Muraro and Ceriani, 2015). It remains to be determined whether the largest aPDFME in the cockroach switches its bursting mode to a more tonic activity pattern during the night, as has been suggested for fruit fly l-LNvs (Muraro and Ceriani, 2015). Previous pharmacological experiments in AME neurons have shown that bursts are accompanied by regular Ca2+ transients that are blocked by hyperpolarization, by the HCN-channel blocker DK-AH 269, and by the sodium channel blocker TTX (Wei and Stengl, 2012). Thus, the presence of HCN pacemaker channels and the cells’ regular activity, even when isolated in primary tissue culture, indicate that they are spontaneously, endogenously oscillating clock neurons. Future studies will investigate whether the activity pattern of the largest aPDFME in the Madeira cockroach cycles not only with an ultradian but also with a circadian period.
In summary, using a combination of anatomical and physiological techniques, we characterized for the first time the PDF sensitivity of ipsilateral and contralateral AME neurons, and reported for the first time PDF autoreceptors in PDF-expressing cockroach clock neurons. Based on the unexpected finding of antagonistic PDF sensitivity correlating with different branching patterns of clock cells, we formulated a new hypothesis to explain PDF-dependent control of rest–activity rhythms at a stable phase difference (Fig. 8). Future studies in different insects are needed to test this new hypothesis of PDF function in the insect circadian clock.
Footnotes
Acknowledgements
We are grateful to Dr. H. Dircksen for the generous supply of antibodies. We thank Karin Grosse-Mohr for technical assistance and Andre Arand for animal maintenance.
Grant sponsor: Deutsche Forschungsgemeinschaft (DFG) grants STE531/18-1,2,3 and STE 531/25-1 to MS
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
