Abstract
In an invariantly rhythmic world, a robust and stable mammalian circadian clock is presumed to confer fitness advantages. In shift-work or after rapid transmeridian travel, however, a stable clock might be maladaptive and a more flexibly resettable clock may have advantages. The rate at which rodents can adjust to simulated time zone travel and the range of entrainment can be markedly increased through simple light manipulations, namely, by exposing animals to extremely dim light (<0.01 lux) at night or by bifurcating rhythms under 24-h light-dark-light-dark (LDLD) cycles. Here we investigated the separate effects of dim light and bifurcation on the ability of mice to entrain to 18-h days (LD 13:5; T18). Incorporating dim light at night, mice in Experiment 1 were exposed either to LD cycles with photophases that were progressively shortened from LD 19:5 to LD 13:5 or to bifurcating LDLD cycles with photophases that were lengthened from LDLD 7:5:7:5 to LDLD 13:5:13:5. In both cases, wheel-running rhythms were robustly synchronized to T18 and the phase of the free-running circadian rhythm was controlled by the timing of release into constant conditions. In Experiment 2, either dimly illuminated nights or a history of bifurcation without continuing dim light was sufficient to allow behavioral entrainment to T18 whereas previously unbifurcated mice under dark nights failed to entrain to T18. Additionally, concurrent measurement of body temperature rhythms in T24 LDLD revealed them to be bimodal. These studies suggest that the circadian system is markedly more flexible than conventionally thought and that this flexibility can be achieved in a noninvasive and nonpharmacological way. Facilitation of behavioral entrainment to extreme light-dark cycles may have translational potential for human shift-workers.
The hypothalamic suprachiasmatic nucleus (SCN) generates an approximately 24-h neural rhythm that synchronizes clocks throughout the body to orchestrate daily rhythms in physiology and behavior (Reppert and Weaver 2002; Welsh et al. 2010). The phase of the SCN is set by light, which synchronizes it with the environmental light-dark cycle. However, the circadian timing system is generally slow to shift, and organs shift at different rates (Kiessling et al. 2010). During shift-work requiring altered rest-activity cycling, or after transmeridian travel, the circadian timing system may be disturbed, inducing a state of temporarily desynchronized oscillators (i.e., jetlag) (Kiessling et al. 2010). Chronic circadian disruption is associated with elevated risk of obesity, impaired immune function, increased cancer risk, and impaired cognition (Costa 1996; Hansen 2001; Karatsoreos et al. 2011; Voigt et al. 2013; Colwell and Matveyenko 2014; Marquié et al. 2014). A more flexible circadian system might be expected to decrease clock disruption in these contexts. The current study explores 2 different light manipulations that increase flexibility of activity rhythms in mice and therefore have potential to reduce negative effects of circadian disruption.
Under traditional laboratory conditions, most mammals have been shown to exhibit stable entrainment to a narrow range (e.g., ±2 h) of periods around 24 h (Daan and Aschoff 2001). Syrian hamsters (Mesocricetus auratus), for example, failed to entrain to T-cycles longer than 26 h (Boulos et al. 2002; Gorman et al. 2005). This range of entrainment, however, can be markedly extended by exposure to dim light at night (compared with complete darkness) at an illuminance comparable to starlight or dim moonlight (Boulos et al. 2002; Gorman et al. 2005; Gorman et al. 2006), even though such low light levels have been reported to be too weak to shift the clock (Brainard et al. 1982; Brainard et al. 1984; Nelson and Takahashi 1991a; Nelson and Takahashi 1991b; Brainard et al. 2001). With the incorporation of dim nighttime illumination, hamsters were successfully entrained to T-cycles up to 30 h (Gorman et al. 2005) or as low as 19 h (Chiesa et al. 2005).
Additionally, in 24-h light-dark-light-dark (LDLD) cycles, a bifurcated entrainment pattern, characterized by nearly equal amounts of running wheel activity in each of 2 nights every 24 h, is facilitated by addition of dim light at night in both mice and hamsters (Gorman 2001; Gorman et al. 2003; Gorman and Elliott 2003; Gorman and Elliott 2004; Evans et al. 2012). Bifurcation itself was recently shown to exert surprising effects on enhanced adaptability of activity rhythms of rodents. Bifurcated hamsters, for example, more than doubled speed of reentrainment to new light cycles after travel across virtual time zones (Harrison and Gorman 2015). Additionally, even without dim light, bifurcated mice were capable of entraining to an LDLD cycle lengthened from 24 h to 30 h, whereas unbifurcated mice free-ran under these conditions (Harrison et al. 2016).
The effects of previous states of entrainment on properties of the biological clock are termed after-effects. Best known are period after-effects—lengthened and shortened free-running period in constant darkness following entrainment to long versus short light cycles (Pittendrigh and Daan 1976) and waveform after-effects—lengthened and shorted active period (alpha) in free-run after short versus long photoperiod. Less well known are after-effects on entrainment—different patterns of entrainment in identical conditions in T22 depending on lighting history (Chiesa et al. 2006) and after-effects on phase resetting—larger light-induced phase shifts in hamsters entrained to short compared with long photoperiods (Pittendrigh et al. 1984; Evans et al. 2004; Glickman et al. 2012; Glickman et al. 2014). The ability to entrain to 30-h LDLD cycles following bifurcation, but not other conditions, represents another type of circadian entrainment after-effect.
In studying modulation of circadian flexibility (Harrison and Gorman 2015; Harrison et al. 2016), our prior work studied the effects of dim light in close conjunction with bifurcation so that the relative contributions of each on entrainment have not been clearly distinguished. The current study, therefore, aimed to separate these contributions on the capacity of mice to entrain behaviorally and physiologically to non-24-h cycles. Further, to test whether extraordinary entrainment plasticity was limited to lighting cycles lengthened from 24 to 30 h, the current study explored behavioral entrainment to short, 18-h, T-cycles and used telemetry to determine whether entrainment extended to daily rhythms in body temperature. Together with earlier work, the current results establish that photic manipulations permit extraordinary entrainment to a diversity of non-24-h conditions and identify dim light, without prior bifurcation, as a sufficient stimulus for this capacity.
Methods
Nomenclature
The study of bifurcation in non-24-h conditions strains conventional circadian nomenclature. In this article, the zeitgeber period is specified by T (e.g., T18 or T24). Zeitgeber cycles are additionally described as unimodal (LD) or bimodal (LDLD). Although T12 LD and T24 LDLD are formally equivalent, the latter description is preferred here, since the bifurcated entrainment state is not generally a 12-h rhythm but rather a 24-h rhythm comprised of 2 active periods. For example, bifurcation was first demonstrated in LDLD cycles that were not equivalent to T12 LD (e.g., LDLD 9:5:5:5) (Gorman and Steele 2006), and bifurcation appears to have a 24-h organizational basis in the SCN (Watanabe et al. 2007; Yan et al. 2010). In contrast, prior entrainment studies under LDLD 10:5:10:5 (Harrison et al. 2016), where T15 LD was formally equivalent to T30 LDLD, suggest either a 15-h oscillation or a driven rhythm. Pending information about SCN rhythmicity, these cycles are referred to as T15/T30 cycles. This paper reports on cycles that could be considered T18 LD cycles or T36 LDLD cycles. Because the native 24-h period is much closer to 18 h than to 36 h, we generally avoid reference to T36 LDLD cycles in favor of the simpler T18 LD designation. Moreover, no analysis revealed any suggestion of behavioral rhythmicity with a period of 36 h.
Animals and Housing
C57BL/6J mice (Jackson, Sacramento, CA), 6 to 9 weeks of age at the start of the experiments, were individually housed in plastic shoebox cages (L × W × H: 28 × 18 × 15 cm) furnished with a running wheel (13 cm diameter). Food (Mouse Diet 5015; Purina, St. Louis, MO) and water were available ad libitum. Cages were placed in light-tight chambers to ensure full control of light exposure. During the photophases, chambers were lit by white fluorescent lamps providing 300 to 350 lux at the cage level. Scotophases were either complete dark (LDark) or illuminated with green LEDs (555 ± 30 nm) mounted approximately 25 cm outside of the cage generating illuminance less than 0.01 lux (irradiance of 3.90 × 10-5 W m-2) measured within the cage (LDim). For details on lighting, see Evans et al (2012). Cages were changed at least every 21 days on a schedule to minimize disruption of activity rhythms. Animals were weighed at the beginning and end of the experiment. Experiments were conducted with prior approval of the University of California San Diego Institutional Animal Care and Use Committee.
Experiment 1: Entrainment to T18 Cycles
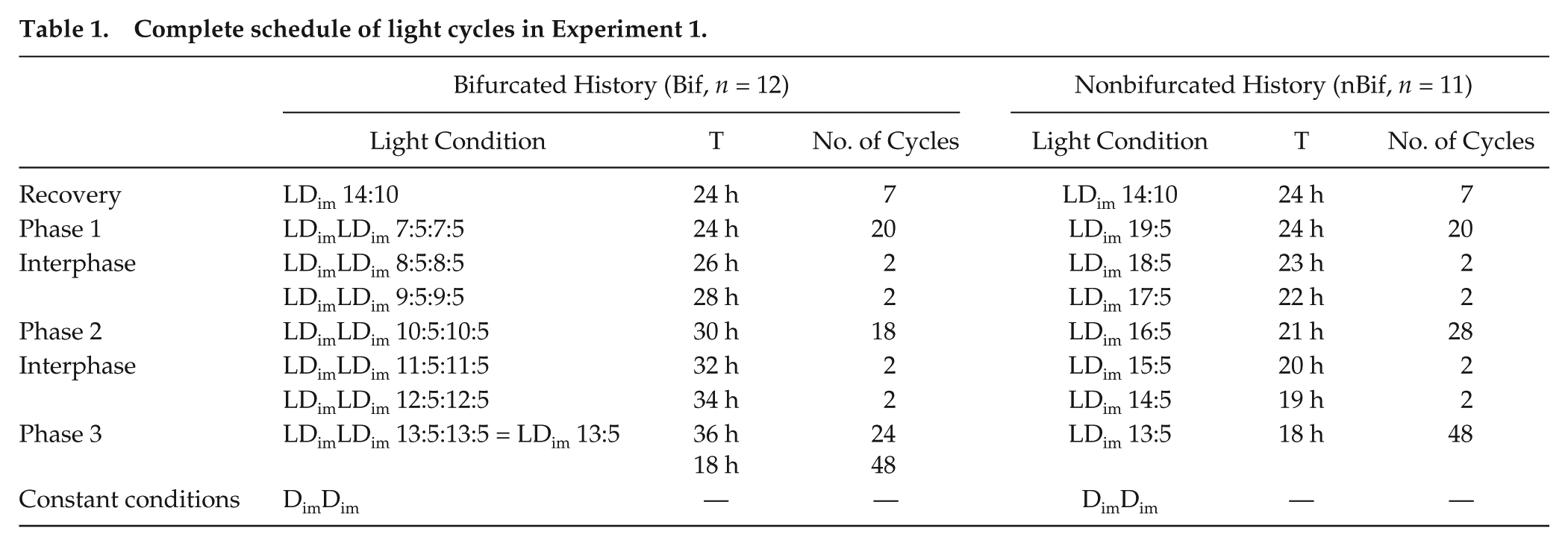
To test the hypothesis that prior bifurcation would uniquely enable entrainment to short T-cycles, mice from the colony (LDark 14:10) were assigned to 1 of 2 groups. One group was housed in LDimLDim 7:5:7:5 to induce bifurcation (Bif; n = 12, 6 females, 6 males), while a second group was housed in LDim 19:5, which does not allow rhythm bifurcation (nBif; n = 11, 6 females, 5 males). In 3 phases the photoperiods were progressively lengthened from 7 to 10 to 13 h in the Bif group and shortened from 19 to 16 to 13 h in the nBif group (Table 1; Suppl. Fig. S1). In both groups, scotophase duration was fixed at 5 h. Fifty days after the start of the experiment, light conditions were identical between groups, LDim 13:5, but are denoted with regard to whether mice had a bifurcated history (LDimLDim 13:5:13:5—Bif) or not (LDim 13:5—nBif) (Table 1).
Complete schedule of light cycles in Experiment 1.
After 48 unchanged T18 LD cycles, animals were released in constant condition (DimDim) at 1 of 2 different time points. For half the animals (n = 12), photophase lighting was extinguished permanently at the beginning of a scheduled scotophase; for the other half (n=11), photophase lighting was extinguished 9 h later, after an abbreviated 4-h photophase. After 20 days in constant conditions, the experiment was ended.
Body temperature and general locomotor activity were continuously recorded using surgically implanted telemeters. To permit characterization of the endogenous body temperature rhythm independent of masking-effects of running wheel activity, wheels were blocked with a pin for 3 full LDLD cycles during all 3 phases with at least 3 full cycles between each time being blocked. With the exception of the days of wheel-blocking, wheel running was continuously recorded.
Experiment 2: Cues for Induction and Maintenance of T18 LD Entrainment
Because Experiment 1 yielded unexpected robust entrainment to LDim 13:5 in both previously bifurcated and nonbifurcated groups, Experiment 2 assessed the roles of dim light and bifurcation history for their separate contributions to T18 entrainment. Adult mice from a separate cohort were assigned to each of 4 groups (n = 8, 4 females per condition). All groups were first entrained to T24 cycles for 3 weeks before exposure to T18 cycles. The first group was bifurcated in LDimLDim 7:5:7:5 and subsequently transferred directly to LDim 13:5 (Bif-Dim). A second group bifurcated in LDimLDim 7:5:7:5 was moved to LDark 13:5 (Bif-Dark). A third group went straight from nonbifurcating LDim 14:10 into LDim 13:5 (nBif-Dim), while the last group was exposed initially to LDark 14:10 and then to LDark 13:5 (nBif-Dark). Running wheel behavior was continuously recorded.
To assay T18 entrainment under potentially weaker zeitgebers (i.e., with lower L-D ratio), after 3 weeks in LDim 13:5 or LDark 13:5, all groups were exposed to days that were shortened and nights lengthened by 2-h increments every 15 days (i.e., twenty 18-h cycles), resulting in the following light-dark or light-dim cycles: LDim 11:7, LDim 9:9, LDim 7:11, LDim 5:13, LDim 3:15, LDim 1:17, and constant conditions. Light offsets were kept constant, and light onsets were delayed 2 h for every new phase.
Data Collection and Analysis
Running wheel activity (RW; half revolutions, Experiments 1 and 2), general locomotor activity (GLA, Experiment 1), and body temperature (Tb, Experiment 1) were collected using VitalView (MiniMitter, Sun River, OR). All data were stored in 6-min bins. In Experiment 2, 2 of 8 animals in both Bif-Dark and Bif-Dim groups were excluded from analyses because they failed to meet objective and subjective criteria for bifurcation in the initial LDLD 7:5:7:5.
Entrainment quotients
To quantitatively assess entrainment to T18 light cycles, Lomb-Scargle periodograms were generated, and power at 18 h and the peak power value in the circadian range (22-26 h) were each determined. Entrainment quotients (EQs) were calculated as the ratio between power at 18 h and ~24 h [power18h / (power18h + power~24h)] and logit-transformed using base e {log[EQ/(1-EQ)]}. EQs were separately calculated for wheel running (EQWheel) and body temperature (EQTb) data.
Phase angles of entrainment
Activity onsets were eye-fitted. Phase angles in LDimLDim 7:5:7:5 were calculated separately for each of the alternate dark episodes. For all other light cycles, a single phase angle was calculated averaging onsets from all dark phases. Only animals that met criteria of entrainment (i.e., EQ > 0.75) were included in the analysis.
Period of free-running animals
For animals that were poorly entrained to T18 (EQWheel < 0.75), periods of the free-running components in T18 were determined from periodograms calculated over 20 cycles as the peak power in the range from 22 to 26 h.
Phase of free-run in DimDim
Skipping the first 3 days in DimDim, 8 consecutive eye-fitted onsets of activity were used for a least-squares regression to project the phase of activity onset at the day of release (Suppl. Fig. S2A). Watson-Williams test was used for differences in mean phase between the 2 times of release, and the Rayleigh test was used to test for uniformity in the distribution of projected activity onsets relative to onsets of DimDim.
Activity-independent temperature
To calculate body temperature independent of activity, a nonlinear regression method was adapted from previously described techniques (Weinert and Waterhouse 1998; Damaggio and Gorman 2014) using only data collected during blocking of the running wheels. General locomotor data were convolved with a delay function (Equation 1) to create a smoothed activity measure that takes into account the delayed effect it has on temperature. Parameters were fit to get the highest correlation with GLA data. Next, the nonlinear relationship between temperature and activity was estimated (Equation 2). All parameters were fit using the nonlinear least squares function in R (Rstudio, Version 0.98.1062, Boston, MA; R, version 3.1.1, Vienna, Austria). To estimate endogenous body temperature (Tb′), the activity-induced temperature was subtracted from the measured temperature signal with Equation 2 fitted parameters.

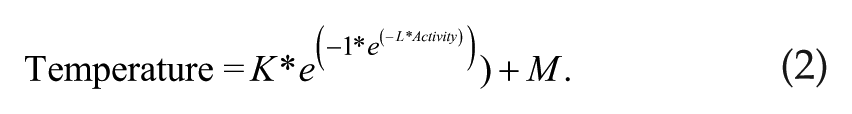
Surgery/Telemetry
Miniature radio telemeters (<2.0 g; G2 E-Mitter; Respironics, Inc., Bend, OR) were implanted intraperitoneally under isoflurane anesthesia. Animals received a single subcutaneous dose of 0.05 mg/kg buprenorphine interoperatively for pain relief. After the animals were ambulatory, they were returned to colony conditions (LDark 14:10) and allowed 7 days for recovery before experiments started. Prior to surgery and during recovery, mice were housed in same-sex groups of 3 or 4. Cages were placed on an ER-4000 Energizer/Receiver (Respironics, Inc.) to communicate with the telemeters. Over the course of the experiment, 4 telemeters gave unusable data, so that body temperature (Tb) and general locomotor activity (GLA) analyses were conducted with n = 9 and n = 10 for Bif and nBif, respectively.
Results
Experiment 1: Entrainment to T18 Cycles
Figure 1 depicts representative patterns of running wheel activity (RW) over 12 weeks of mice initially bifurcated in T24 LDimLDim 7:5:7:5 and exposed to cycles lengthening to T36 LDimLDim 13:5:13:5 (Fig. 1A) and of mice initially nonbifurcated in T24 LDim 19:5 and exposed to cycles shortening to T18 LDim 13:5 (Fig. 1B). In both cases and in all phases of the experiment, most activity occurred during the dimly lit nights, and there was no visually salient free-running periodicity. Qualitatively, GLA and Tb showed similar patterns as RW, with greater activity and higher temperatures in the scotophases (Suppl. Fig. S3). When uninterrupted wheel running of the same 2 mice is plotted modulo 18 (Fig. 1, C and D), both animals showed activity strictly aligned with the dark periods with activity onsets nearly coincident with lights-off.
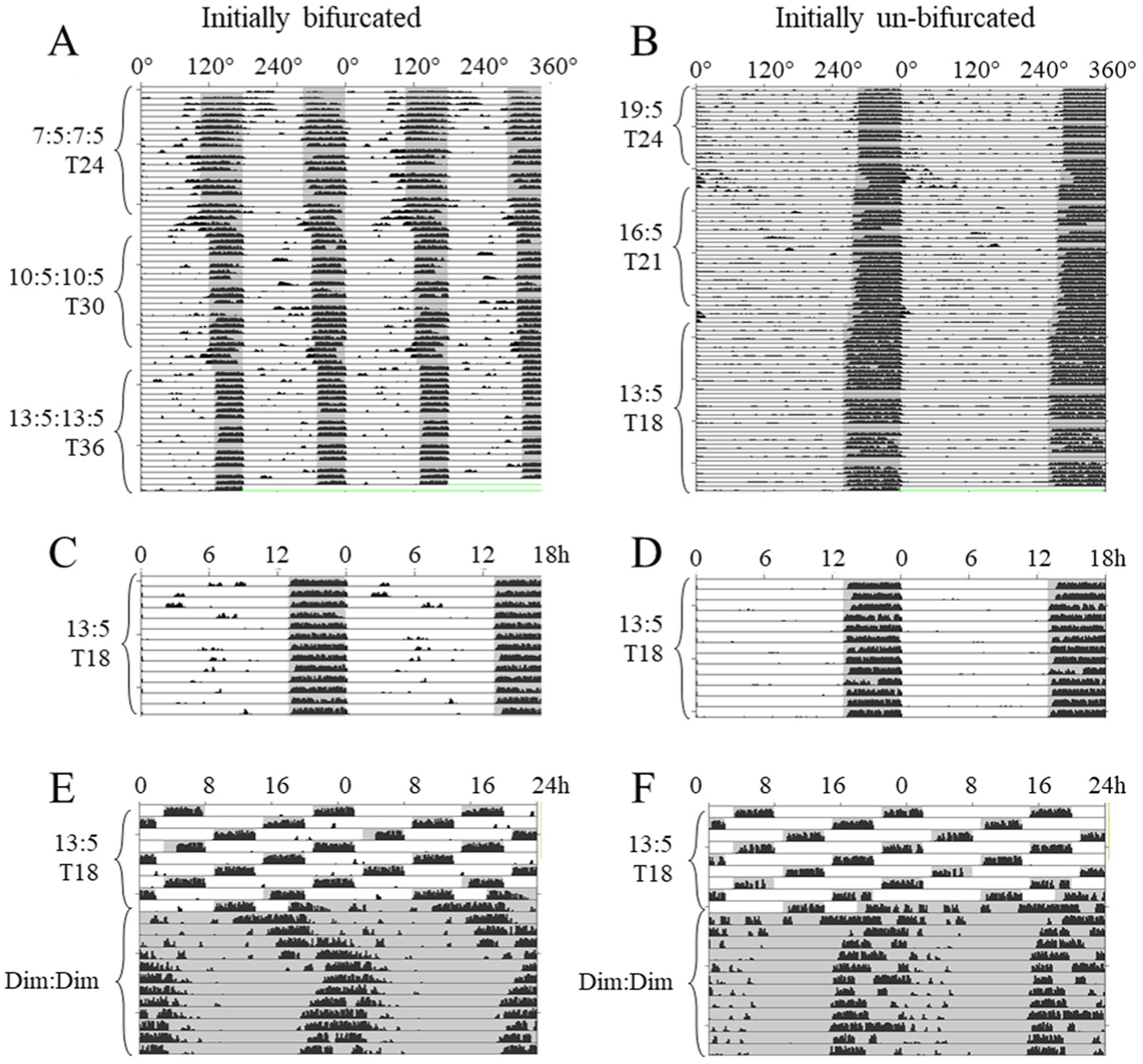
Representative double-plotted wheel-running actograms from Experiment 1 of individual mice that were (A) initially bifurcated and (B) not bifurcated in LDimLDim 7:5:7:5 and LDim 19:5, respectively, and gradually transitioned to LDim 13:5. Data are plotted modulo 360 angular degrees. Gray shading indicates times of relative darkness. Gaps in data represent lack of wheel running activity while the wheels were blocked. Transitional photoperiods are not labeled. From the same animals, parts C and D replot, modulo 18 h, the same 14 uninterrupted cycles from the final experimental phase. Parts E and F show the transition from LDim 13:5 to DimDim, from the same animals. Y axes are scaled from 0 to 200 half revolutions per minute.
Activity profiles averaged across subjects are depicted in Figure 2, demonstrating that in each experimental phase including the initial bifurcation, activity was almost exclusively restricted to hours of darkness. EQ values approached 1 and activity onsets shortly followed light offsets (Table 2, Suppl. Fig. S4). Under identical lighting conditions of T18 (LDim 13:5), quantitative analyses revealed no differences between Bif and nBif in LDim 13:5 in EQWheel (Suppl. Fig. S4), total amount of running wheel activity in day or night, or in phase angles between light offsets and activity onsets (Table 2, Fig. 2). To distinguish between a 12-h and 24-h rhythm in phase 1 (LDLD 7:5:7:5), phase angles of wheel-running activity were compared between alternate scotophases. In 60% of animals there was a significant difference in phase angle in the odd nights compared with the even nights, indicative of a 24-h basis to the entrainment. Under LDLD 10:5:10:5 and LDLD 13:5:13:5, however, no animals showed systematic differences between alternating nights, indicating 15/18 h rather than 30/36 h bases, respectively, of their rhythmicity (Fig. 2). Therefore we averaged activity data from all scotophases to report a single phase angle under non-24-h conditions.
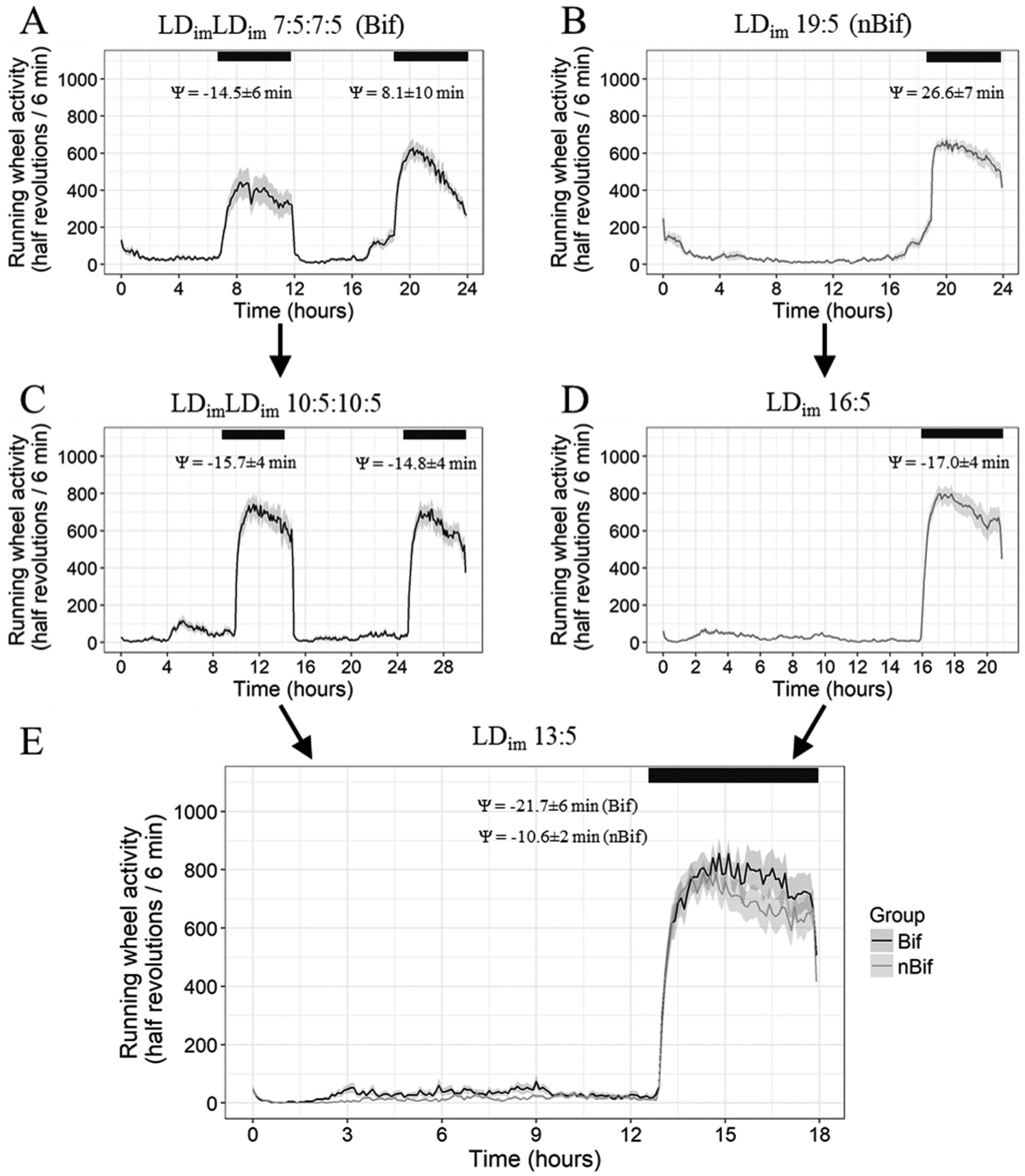
Averaged time series of running wheel activity (half wheel revolution) in (A) LDimLDim 7:5:7:5, (C) LDimLDim 10:5:10:5, and (E) LDim 13:5 for initially bifurcated animals and (B) LDim 19:5, (D) LDim 16:5, and (E) LDim 13:5 for unbifurcated animals from Experiment 1 across all days with wheel. Shading represents between-subject SE of the activity profiles. Black bars represent dark phases. Phase angles are shown as mean ± SEM (min), with positive values indicating that activity phase-leads onset of darkness. Y axes represent half revolutions per 6-min bin.
Entrainment measures in LDim 13:5 from Experiment 1.
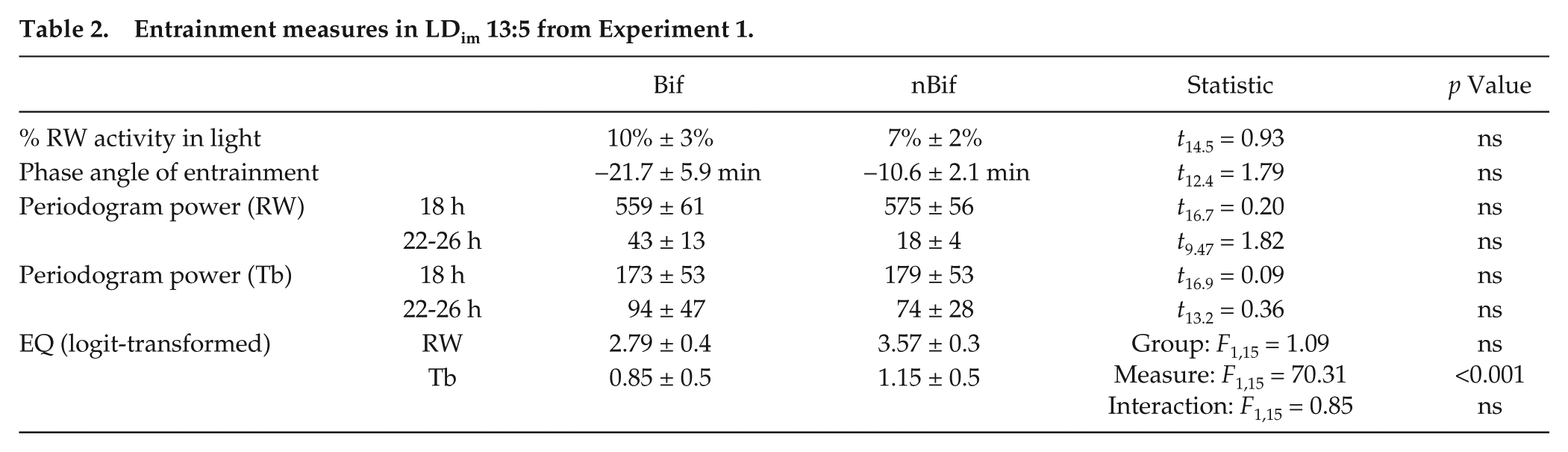
In all animals, the EQTb was lower than the EQWheel (F1,15 = 70.31, p < 0.001, Suppl. Fig. S4), independent of Bif/nBif (interaction, F1,15 = 0.9, p > 0.05). Lower EQTb was reflected not only by lower T18-power but also statistically greater power in the free-running, circadian (22-26 h) range (Suppl. Fig. S5). Entrainment quotients were significantly lower in males than in females (F1,15 = 4.88, p < 0.05). A more comprehensive analysis of sex differences in this and other studies will be published separately.
Bifurcated Body Temperature in LDLD 7:5:7:5
Wheel blockage altered the Tb rhythm, as evident in the generally lower body temperatures during dark periods when wheels were blocked (Suppl. Fig. S6, A and C). Moreover, Tb closely tracked GLA suggestive of additional acute effects of nonrunning activity on Tb (Fig. 3, A and C). As would be expected, nBif mice showed generally unimodal Tb with elevation during the night. Rhythms of bifurcated mice exhibited bimodality with elevations of Tb in both nights (Fig. 3, A and C). Similar to Tb, Tb′ was elevated during the dimly lit nights in both Bif and nBif. That is, when controlling for levels of GLA, Tb′ is still bimodal in Bif (Suppl. Fig. S7). In N1 (shared night), Tb′ trended lower in Bif than in nBif (t12.5 = 2.1, p = 0.056; Fig. 3), yielding a lower amplitude rhythm. Averaged across the 5-h dim nights, Tb′ during N2 (additional night) was significantly higher in Bif mice compared with nBif mice (t16.0 = 4.91, p < 0.001). There was no difference between N1 and N2 in Bif animals (t10.6 = 1.30, p > 0.05), whereas for nBif mice, Tb′ was significantly higher in the night than at antiphase (t17.7 = 9.0, p < 0.001).
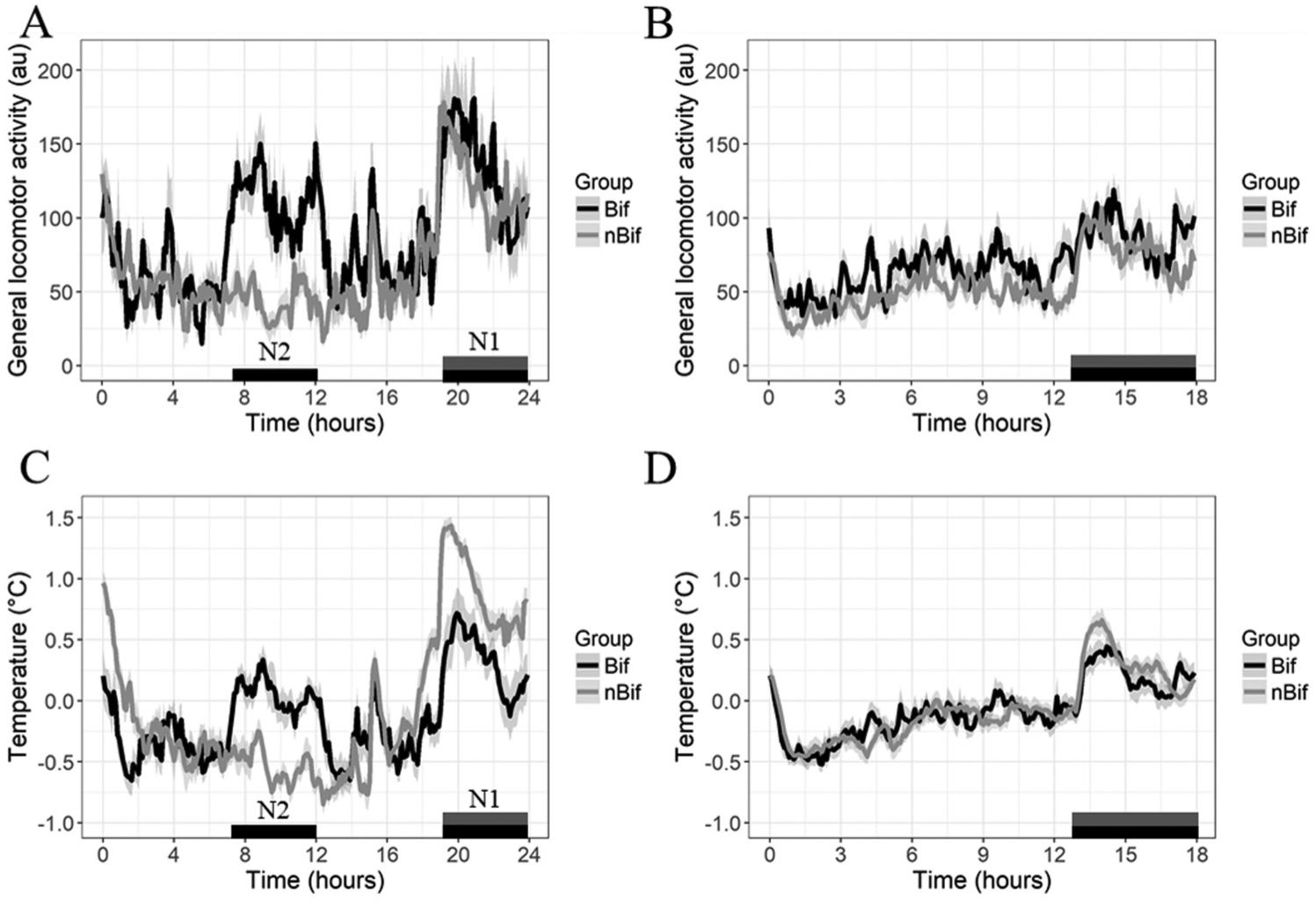
(A, B) GLA and (C, D) Tb averaged across 72 h of wheel blocking in Experiment 1 during 24-h conditions and 18-h conditions, respectively. Temperature is plotted in degrees above or below the daily mean. Bars at the bottom of the panels represent the light-dark cycle, with black-gray being dim and white being light. Black and gray represent Bif and nBif groups, respectively. Shading represents SE. N1 = 19-24 h and N2 = 7-12 h.
Body Temperature in T18 LD 13:5
Also in T18 (LD 13:5), Tb closely tracked activity (Suppl. Fig. S6, B and D), with higher levels of activity and body temperature during the night (F1,16 = 88, p < 0.0001; Fig. 3, B and D). However, lower EQTb (i.e., higher power in circadian range) did not allow for similar methods used in T24 to control for levels GLA.
Release into Constant Conditions
When released into DimDim, animals reverted to free-run close to 24 h within a few cycles (Fig. 1, E and F). The projected phases of activity onset were significantly clustered with a mean phase angle of −0.09 h (R19 = 0.43; p < 0.05, Suppl. Fig. S2) relative to dark onset. Individually, neither release group was significantly clustered (R9 = 0.49, p < 0.09, and R9 = 0.37, p < 0.25, for 0 and +9, respectively), but small sample sizes compromised the statistical power of these tests. When the time of release into DimDim was delayed by 9 h, the projected onsets were delayed 8.5 h compared with those mice beginning DimDim at the beginning of a previously scheduled scotophase (F1,18 = 14.3; p = 0.001).
Experiment 2: Cues for Induction and Maintenance of T18 Entrainment
Inductions
As in Experiment 1, representative mice that were bifurcated in LDimLDim 7:5:7:5 entrained readily, as evident from alignment of activity with shaded dark periods, when transferred directly to LDim 13:5 (Fig. 4, A, C, and E). The same was true for mice bifurcated in LDimLDim 7:5:7:5 and transferred to LDark 13:5 and mice transferred from LDim 14:10 to LDim 13:5 (actograms not shown). In contrast, mice transferred from LDark 14:10 to LDark 13:5 showed unambiguous evidence of free-running activity, recognized as a lack of alignment with a clear free-running period greater than 24 h (Fig. 4, B, D, and F).
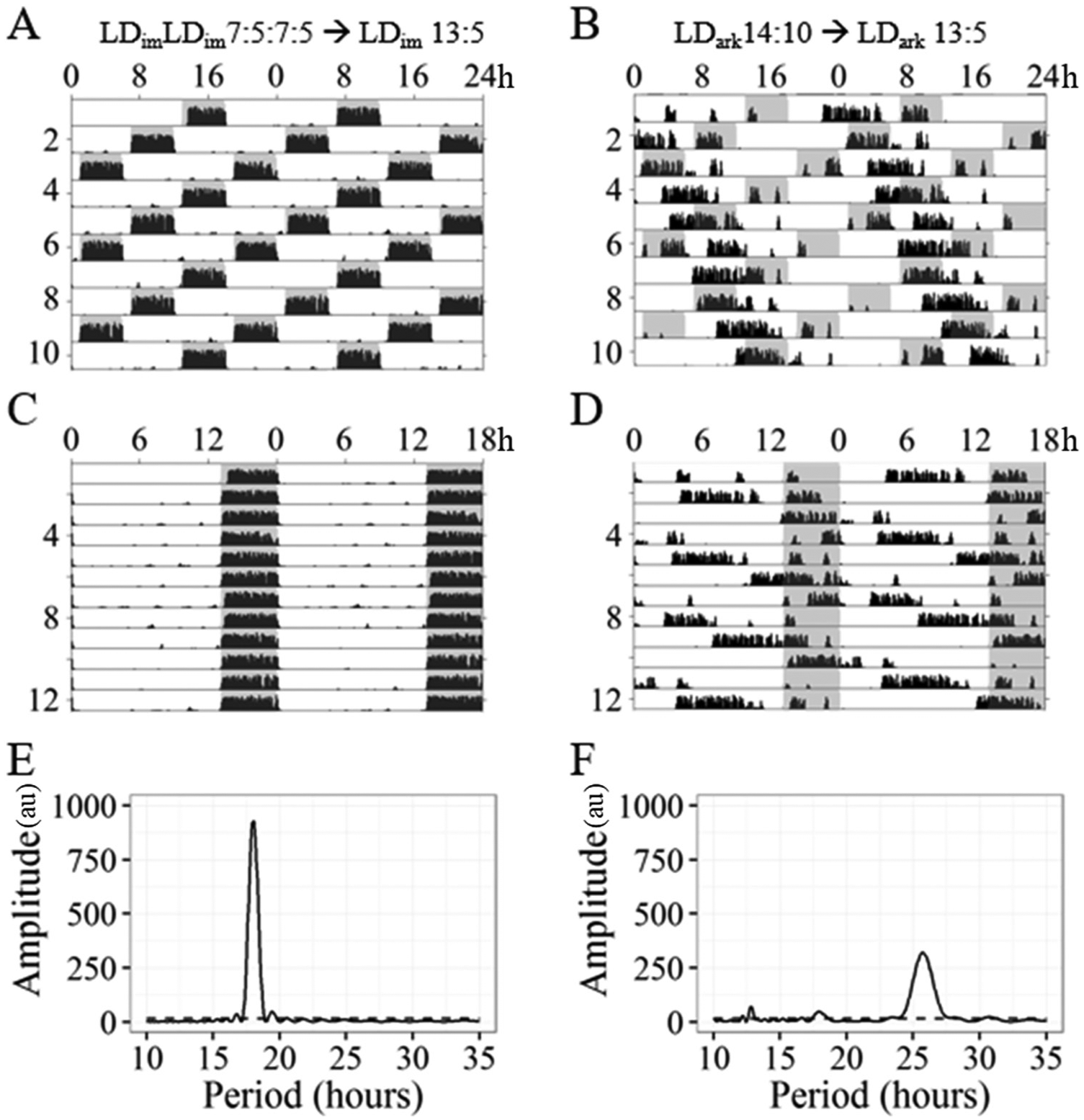
Representative wheel running data from Experiment 2 with respective periodogram for (A, C, E) a successfully entrained animal from Bif-Dim and (B, D, F) a free-running animal from nBif-Dark. A and B show the same data as C and D in modulo 24 h and 18 h, respectively. Conventions as in Figure 1.
Considering all subjects, entrainment failed only in the group without exposure to dim light or a history of bifurcation (nBif-Dark) as reflected in multiple measures (Table 3; Suppl. Fig. S8). Periodogram power at 18 h was significantly different across groups (F3,24 = 5.931, p < 0.01) and lowest in nBif-Dark compared with all other groups (Tukey HSD adjusted p values < 0.05). Power in the circadian range differed significantly between groups (F3,24 = 3.661, p < 0.05). Both Dim groups showed significantly lower power than nBif-Dark (Tukey HSD adjusted p values < 0.05). The EQWheel was lowest in the nBif-Dark group and significantly different from all other groups (F3,24 = 9.18, p < 0.001; all Tukey HSD adjusted p values < 0.05).
Entrainment measures in LDim 13:5 from Experiment 2.
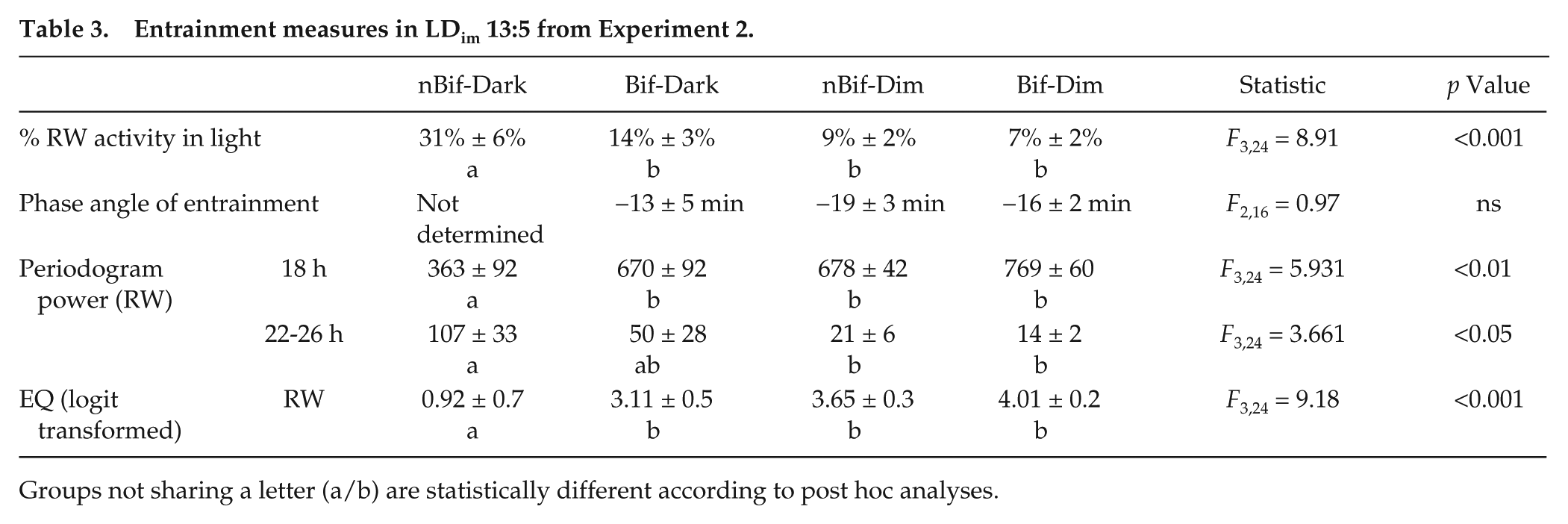
Groups not sharing a letter (a/b) are statistically different according to post hoc analyses.
Maintenance
As the light to dark ratio in T18 was progressively reduced by shortening photophases over the course of the experiment, all animals eventually lost entrainment and started to free-run in or before reaching LD 1:17 (Fig. 5, A and B). Values of EQWheel significantly decreased over the different phases within the experiment (Fig. 5C; F1,23 = 728.48, p < 0.001) and eventually approached zero (i.e., periodogram power exclusively in the circadian range). The pattern of entrainment loss differed by group as indicated by repeated-measures ANOVA (group, F3,23 = 5.51, p < 0.01; interaction with phase, F3,23 = 5.67, p < 0.01). Group differences based on 1-way ANOVA within each phase are indicated in Figure 5C. Phase angles did not change between photoperiods (b < −0.002, SE < 0.001, t = 1.80; 95% CI = −0.004 to 0.0003).
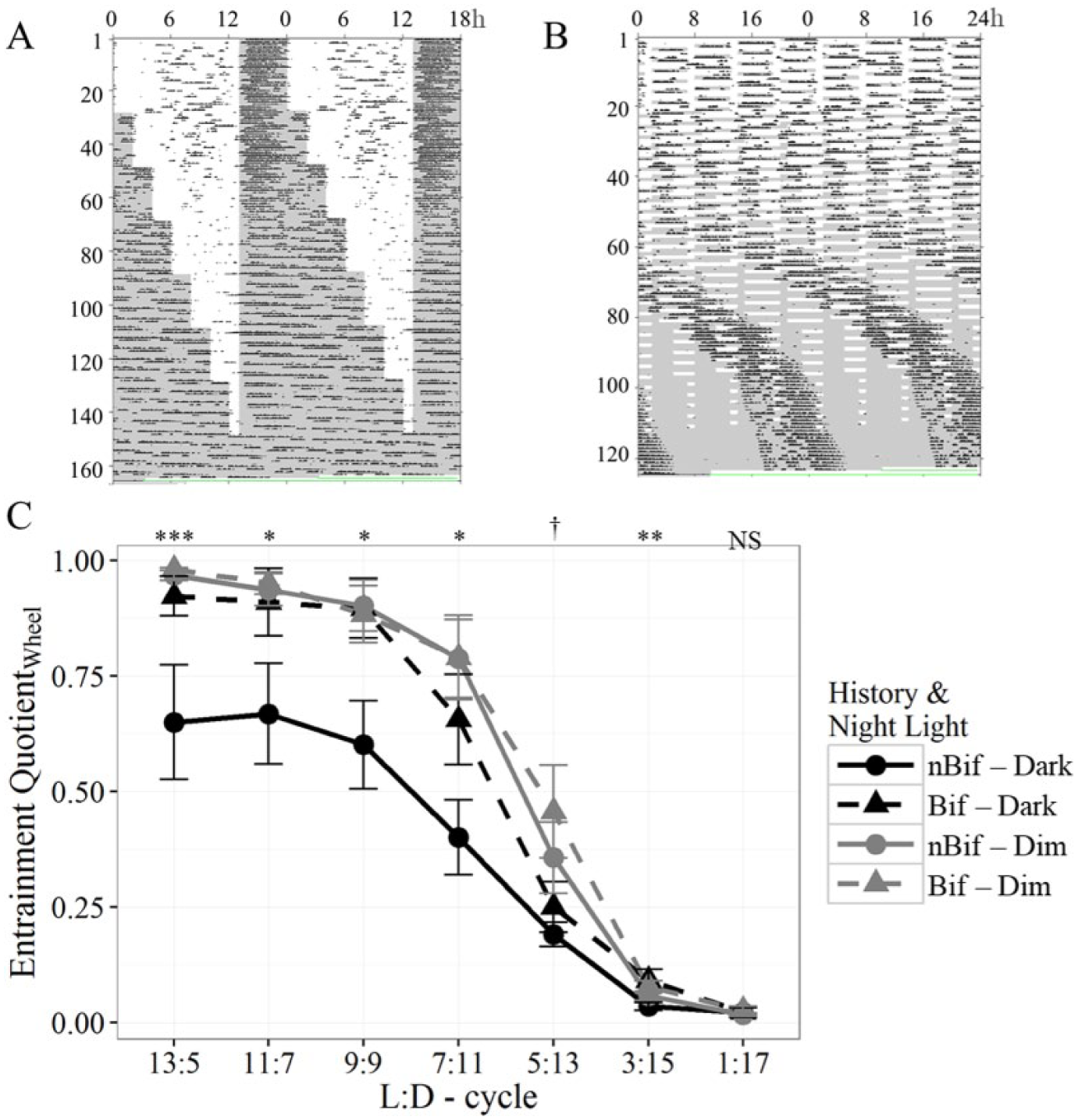
Representative actograms from Experiment 2 plotted (A) modulo 18 h and (B) modulo 24 h of 1 animal across all different T18 conditions. (C) EQWheel in all T18 conditions represented as mean ± SE. Significance symbols show 1-way ANOVA results for group differences in each phase: 0 < *** < 0.001 < ** < 0.01 < * < 0.05 < † < 0.1 < ns < 1.
Period of free-running animals
Tau significantly decreased as the light to dark ratio decreased (b = −0.014, SE = 0.001, t = −12.80), such that for every 1% decrement in light duration, tau was reduced by 0.014 h (95% CI = −0.016 to −0.012). Tau was not significantly different between groups (b = −0.076, SE = 0.082, t = −0.93 and b = −0.099, SE = 0.082, t = −1.19 for history and dim light, respectively).
Body Weight
Relative weight gain over the full duration of the experiment negatively correlated with the ratio of entrainment in both Experiment 1 (b = −0.05, SE = 0.01, t = −5.28) and Experiment 2 (b = −0.07, SE = 0.02, t = −3.57) (Suppl. Fig. S9).
Discussion
The present study demonstrates that under permissive conditions, neurologically intact wild-type mice are capable of robust behavioral entrainment to 18-h LD cycles; that exposure to dim light at night, an immediate history of rhythm bifurcation, and scotophases 9 h or shorter each facilitate or permit this behavioral entrainment; that body temperature rhythms adapt also to 18-h days but in some animals exhibit larger nonentrained rhythmic components than are apparent in wheel-running data; and that demasking of body temperature rhythms for effects of activity establishes a bifurcated but reduced amplitude rhythm of endogenous body temperature.
In Experiment 1, the most unanticipated result was that regardless of the T-cycle (LD 7:5:7:5, 10:5:10:5, 13:5, 16:5, and 19:5) mice almost uniformly adopted activity rhythms with activity closely aligned with the scotophases, minimal activity in the photophases, and rare or absent evidence of free-running rhythmicity. The strong apparent behavioral entrainment to T18 stands in marked contrast to previous reports from C57 mice or other rodents. As examples, 17% of mice failed to entrain wheel-running activity to T21 (Molyneux et al. 2008), and, lacking a wheel, 80% failed to entrain to T21 (Casiraghi et al. 2012). Housed in groups of 5 without wheels, no mice entrained to T20 (Karatsoreos et al. 2011). In a paradigm that shortened T by 5 min daily, wheel-running mice maintained apparent entrainment as far as 20 h, although mice were never exposed to more than 1 cycle of T20 (Pittendrigh and Daan 1976; Aton et al. 2004). Finally, when various durations of light pulses were used, the maximum phase advance shown in C57 mice never exceeded 2 to 3 h (Schwartz and Zimmerman 1990; Comas et al. 2006), leading to the prediction that 22 h would be the lower limit of the entrainment range. Combined, the summarized experiments conclude that entrainment to T18, which requires a daily decrease of nearly 6 h from the free-running period, even when animals are housed in apparent optimal conditions (i.e., singly housed with wheel; Cambras et al. 2000; Chiesa et al. 2005), is unprecedented.
In previous studies, a history of rhythm bifurcation in T24 LDLD was shown to confer extraordinary flexibility of entrainment and reentrainment in rodents. Specifically, bifurcated hamsters exhibited nearly instantaneous entrainment to LD 16:8 photoperiods of any phase, whereas unbifurcated hamsters maintained under conventional LD 16:8 photocycles entrained more slowly and in proportion to the size of the required phase shift. Parenthetically, animals with a history of short day-length exposure (LD 8:16) reentrained to these same phase shifts at intermediate rates (Harrison and Gorman 2015). Additionally, bifurcated mice exhibited robust behavioral entrainment to an LDLD 10:5:10:5 (T15/T30) schedule (Harrison et al. 2016). Knowing already that bifurcation allows entrainment to the phase 2 (T15/T30) conditions of Experiment 1, we predicted that further extension of the photophase to LD 13:5 (T18/T36) would continue to permit behavioral entrainment. In contrast, after entrainment to long day-lengths such as LD 19:5 (T24) that preclude bifurcation, we expected failed entrainment upon reduction of the photophase to LD 13:5 (T18). Contrary to expectation, mice adapted well and quantitatively equivalently to LD 13:5 after these entrainment histories. Thus, with respect to multiple indices of wheel-running behavior and Tb, we see no evidence for a bifurcation after-effect in the present study, although we cannot exclude the possibility that underlying clock mechanisms differ following these histories. Moreover, because such extreme entrainment outside of a bifurcation context was unprecedented, we hypothesized post hoc that dim light alone may be sufficient to significantly extend the range of entrainment in the same manner as does rhythm bifurcation. Notably, dim light was previously shown to extend the limit of entrainment in hamsters (Boulos et al. 2002; Chiesa et al. 2005; Gorman et al. 2005) but was without a substantial effect in enhancing entrainment of mice to a different T30 photocycle (LD 20:10, Harrison et al. 2016).
Since the T18 cycle represents an extreme with respect to expected limits of entrainment, it raises the possibility that the apparently entrained behavioral rhythms reflect masking rather than true entrainment. Experiment 2 excludes the possibility of acute positive masking effects of dim light by demonstrating that under identical lighting schedules with dark nights, wheel-running rhythms match the T-cycle or free-run depending only on their prior entrainment history. Thus, concurrent dim light cannot explain behavioral entrainment. Rather, prior bifurcation, enabled by dim light, is sufficient to permit entrainment to T18. Prior bifurcation, moreover, is not necessary for entrainment to T18, as dim light throughout the experiment yields the same strong entrainment in the absence of bifurcation. The uniformly robust entrainment in Experiment 1, thus, may be interpreted as resulting from the facilitative effects of dim light in the initially unbifurcated group. Likewise, positive masking was shown to be an inadequate explanation for apparent entrainment to T15/T30, as it too persisted following bifurcation without continuation of dim light (Harrison et al. 2016).
Strong negative masking by bright light could additionally contribute to a misleading impression of entrainment of rhythms that were, in fact, free-running. Such an interpretation is discounted by the EQ values obtained as the photophases were progressively shortened in T18. Despite the reduced opportunity for negative masking by bright light, EQs were unchanged in each group as the fraction of light in the cycle decreased from 72% to 50%. In rats, rhythmicity in the circadian range has been shown to increase with increasing scotophase length in T22 and T23 LD cycles (Cambras et al. 2004). Only when scotophases were lengthened to 11 h did our mice begin to show stronger free-running components and lower EQWheel. As the zeitgeber was further weakened, all mice eventually showed visually obvious free-running rhythms and correspondingly low EQ scores.
Additionally, a masking interpretation is discounted by demonstration of phase control, another important entrainment criterion, in DimDim. In Experiment 1, the phase of activity onsets in constant dim was significantly clustered around and controlled by onsets of constant conditions, and the mean phase difference between the 2 groups was close to the difference in time of release. This phase control is evident despite the fact that free-running rhythms quickly go back to species-typical periods near 24 h. If apparent entrainment in LD 13:5 had been a masked free-running activity pattern, random phases with respect to onset of DimDim would be expected, as phase would be predicted by the phase of the masked free-running rhythm (Harrison et al. 2016). Instead, circadian free-running rhythms with a phase controlled by time of release are indicative of a complete reset at time of release, discounting the possible explanation of a masked free-running rhythm for apparent entrainment in T18. These findings corroborate similar findings on phase of activity onsets after release in DD following entrainment in T15/T30 (Harrison et al. 2016).
Thus, multiple criteria indicate that behavioral entrainment to T18 is genuine and stable but needs a sufficiently strong zeitgeber to be maintained. Notably, however, T18 entrainment did not show the changes in activity onset phase angle as predicted by classic nonparametric entrainment theory (Daan and Aschoff 2001; Granada et al. 2013; Schmal et al. 2015) or reported in rodents under less extreme values of T (Molyneux et al. 2008; Schwartz et al. 2011). Rather, activity onset consistently occurred close to the time of lights-off. This same pattern was previously observed in mice entrained to T30 LDLD cycles (Harrison et al. 2016). Thus, entrainment under these conditions likely does not derive from nonparametric resetting mechanisms. Indeed, even with single light pulses as long as 9 h, C57 mice show only minimal (e.g., 1-2 h) phase advances (Comas et al. 2006) that fall far short of the ~6-h advances required for nonparametric entrainment. Perhaps these long-pulse phase response curves were collected against a background of complete darkness instead of dim illumination. Mechanistically, we speculate that this flexible behavioral entrainment is facilitated by altered SCN function, although whether this involves changes in phase relationships and/or amplitudes of component oscillators awaits time series analysis of SCN clock gene expression.
Because wheel-running increases body temperature, we measured rhythms over 3 separate cycles when access to wheels was blocked. Tb rhythms, as well as GLA, during intervals of no wheel access were clearly bifurcated in T24 and showed nighttime elevations in T18 (Fig. 3). In both cases, however, the amplitude was markedly reduced compared with mice in LD 19:5. This is the first report of Tb in rodents entrained to T18. Body temperature is an important cue for synchronizing peripheral organs (Brown et al. 2002), as temperature cycles with 1.5 °C amplitude have been shown sufficient to entrain isolated lung tissue to non-24-h T-cycles (Abraham et al. 2010). Therefore, these findings are potentially important for functional rhythmicity of peripheral organs such as liver or kidney in extreme T-cycles.
While behavioral entrainment to T18 was extremely robust, as reflected in EQWheel scores approaching 1, periodogram analysis of Tb data yielded discernable rhythmicity in the circadian range, thereby reducing EQTb values relative to EQWheel (Suppl. Fig. S4). Simultaneous expression of multiple periods—some entrained and others free-running—has been noted previously. Activity rhythms dissociate in mice in T21 (Casiraghi et al. 2012), and both activity and body temperature show multiple rhythmic components in rats in T22 and T23 (Anglès-Pujolràs et al. 2007). Additionally, in rats, 2 motor activity components can be forced to decouple in T22 and are associated with subdivisions of the SCN (de la Iglesia et al. 2004). Decoupling of behavioral and Tb rhythms could rely on reorganization of SCN subunits and/or decoupling of SCN and extra-SCN oscillators that differentially contribute to regulation of activity and body temperature.
Even in the absence of a wheel, however, measures of Tb reflect the joint influences of any endogenous Tb rhythm and activity-induced temperature. Through correlational analyses, we further estimated general activity-independent rhythms in body temperature. The additive effect of activity on temperature has a clear ceiling effect: Above a certain level of activity, more activity does not increase temperature any further. Therefore, our nonlinear model to estimate circadian body temperature may more accurately characterize endogenous Tb rhythms than would prior linear models (Weinert and Waterhouse 1998). Bimodality of body temperature (linearly demasked with the Weinert and Waterhouse model) in Siberian hamsters during bifurcation has been previously established (Rosenthal et al. 2005). However, in mice, it is novel that not only running wheel behavior but also activity-independent body temperature is bifurcated in LDLD 7:5:7:5.
Both human shift-work and multiple experimental models of circadian disruption in animal models are associated with weight gain and/or markers of metabolic disease (De Bacquer et al. 2009; Barclay et al. 2012). The critical dimensions of these complex exposure scenarios for metabolic risk are not yet fully defined. Although our experiments were not designed with a focus on body weight regulation, consistent negative correlations between weight gain and EQWheel in LD 13:5 suggest that poor entrainment leads to greater weight gain than does stable entrainment to non-24-h days. This association was obtained despite the fact that interval of weight gain included many different T-cycles (Experiment 1) and different photoperiods in T18 (Experiment 2).
In summary, rhythm bifurcation, very dim light at night, or both permit robust running wheel entrainment to an 18-h light cycle that, to our knowledge, exceeds the shortest demonstrated limit of entrainment in mice. As body temperature exhibited greater periodicity ~24 h, adaptation to T18 is not complete and may reflect decoupling of multiple circadian oscillators, as suggested in other experimental contexts (de la Iglesia et al. 2004; Anglès-Pujolràs et al. 2007; Casiraghi et al. 2012). These findings extend previous work establishing hyperplastic control of rest-activity cycles that, regardless of their mechanistic basis, may have translational utility for behavioral adaptation of humans to challenging shift-work or jet-lag exposures.
Footnotes
Acknowledgements
The authors thank Elizabeth Harrison for comments on an early draft of the manuscript and Antonio Mora and Gil Sanchez for their excellent animal care. This work was supported by the Office of Naval Research N000141310285.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
