Abstract
The mechanisms that enable mammals to time events that recur at 24-h intervals (circadian timing) and at arbitrary intervals in the seconds-to-minutes range (interval timing) are thought to be distinct at the computational and neurobiological levels. Recent evidence that disruption of circadian rhythmicity by constant light (LL) abolishes interval timing in mice challenges this assumption and suggests a critical role for circadian clocks in short interval timing. We sought to confirm and extend this finding by examining interval timing in rats in which circadian rhythmicity was disrupted by long-term exposure to LL or by chronic intake of 25% D2O. Adult, male Sprague-Dawley rats were housed in a light-dark (LD) cycle or in LL until free-running circadian rhythmicity was markedly disrupted or abolished. The rats were then trained and tested on 15- and 30-sec peak-interval procedures, with water restriction used to motivate task performance. Interval timing was found to be unimpaired in LL rats, but a weak circadian activity rhythm was apparently rescued by the training procedure, possibly due to binge feeding that occurred during the 15-min water access period that followed training each day. A second group of rats in LL were therefore restricted to 6 daily meals scheduled at 4-h intervals. Despite a complete absence of circadian rhythmicity in this group, interval timing was again unaffected. To eliminate all possible temporal cues, we tested a third group of rats in LL by using a pseudo-randomized schedule. Again, interval timing remained accurate. Finally, rats tested in LD received 25% D2O in place of drinking water. This markedly lengthened the circadian period and caused a failure of LD entrainment but did not disrupt interval timing. These results indicate that interval timing in rats is resistant to disruption by manipulations of circadian timekeeping previously shown to impair interval timing in mice.
Mammals have evolved multiple systems to track the passage of time across a wide range of intervals, from milliseconds to seconds, hours, days, and even years (Buonomano, 2007). Two of the most studied and well characterized of these systems govern intervals differing by several orders of magnitude: the circadian system for intervals approximating the 24-h solar day and an interval timing system for shorter, arbitrary intervals in the seconds-to-minutes range. This study explores a hypothesis that timing intervals in the seconds-to-minutes range is critically dependent on circadian organization of behavioral state.
Circadian rhythms in mammals are regulated by a hierarchically organized system of cell-autonomous circadian oscillators located in the brain and most peripheral tissues (Dibner et al., 2010). At the cellular level, circadian oscillations emerge from interlocking transcription-translation feedback loops that result in rhythmic expression of a set of core “clock genes” (Partch et al., 2014). At the systems level, a circadian pacemaker in the suprachiasmatic nucleus (SCN) of the hypothalamus sits at the top of the hierarchy and plays a critical role by synchronizing circadian rhythms to the daily light-dark (LD) cycle (Welsh et al., 2010). Circadian oscillators elsewhere in the brain and body can be synchronized directly by daily cycles of food availability and potentially by other nonphotic cues (Dibner et al., 2010; Mistlberger, 2011).
The set of mechanisms responsible for timing intervals in the seconds-to-minutes range is referred to as the interval timing system and is critically involved in a variety of cognitive processes (Gallistel and Gibbon, 2000). In the most familiar example of short interval timing, the fixed-interval (FI) schedule of reinforcement, a reward is delivered to an animal contingent upon the first operant behavior performed after a target interval following the presentation of a stimulus. When averaged over multiple trials, an anticipatory response pattern is observed, with response rate increasing as a function of time and reaching a maximum at the time of reward presentation (Gibbon and Church, 1990). In the peak interval (PI) procedure, rewarded trials are interspersed with randomly occurring peak trials, in which reinforcement is withheld. In these trials, responding assumes a roughly Gaussian distribution, centered over the normal time of reinforcement. The time at which the maximum response rate is reached can be used as an estimate of when the animal expected reinforcement, providing a measure of the animal’s accuracy and variability in its ability to time the interval.
Multiple theoretical models of interval timing have been proposed. In pacemaker-accumulator models, time perception is based on a pacemaker that generates pulses that are temporarily stored in an accumulator over a given interval. At the time of reward, the number of pulses stored in the accumulator is transferred to reference memory. Later temporal judgments are based on comparing the number of pulses currently stored in the accumulator with the values stored in reference memory (Gallistel and Gibbon, 2000). Pulses generated by the pacemaker are subject to stochastic error, causing variability in time estimation to increase linearly with the interval being timed (i.e., the scalar property) (Gibbon, 1977).
In other models, elapsed time is represented by the phases of multiple oscillators of differing native frequencies (Church and Broadbent, 1990; Gallistel, 1990). Changes in the phase relationships among these oscillators as a given interval elapses are used as a marker for temporal duration. Comparison of the phase relationships among the oscillators at any given moment of the timed interval with the representation stored in reference memory provides a means of temporal discrimination. Support for oscillator phase models comes from reports of small deviations from linearity (i.e., violations of the scalar property) in the ability to time particular short intervals, which have been hypothesized to correspond to the intrinsic periodicity of individual oscillators (Crystal et al., 1997; Crystal, 1999, 2001; Crystal and Baramidze, 2007).
The evidence that both the circadian and interval timing systems rely on one or more oscillators has fueled speculation that the two timing systems may be intrinsically linked (Gallistel, 1990; Agostino, Golombek et al., 2011). One way that two systems can be linked is if one modulates the output of the other. There is evidence that interval timing is modulated by circadian phase. In humans, the speed at which the interval timing clock is thought to tick (inferred from estimations of temporal intervals) is positively correlated with core body temperature (Aschoff, 1984, 1998; Kuriyama et al., 2005; Nakajima et al., 1998; Pfaff, 1968). Circadian modulation of interval timing in rats (Bussi et al., 2015; Meck, 1991; Shurtleff et al., 1990) and mice (Agostino, do Nascimento, et al., 2011) has also been reported, although the timing parameters that were observed to vary with time of day or circadian phase differed across studies (see Discussion).
Another way that two systems can be linked is by sharing a common timing mechanism. Mice harboring a point mutation to the zinc finger homeobox 3 (Zfhx3) gene, a transcription factor highly expressed in the SCN, exhibit both a 5% shortening of free-running circadian periodicity and a 15% to 30% leftward shift in the response distribution to a peak interval task compared with wild-type controls (Balzani et al., 2016). A single gene mutation is thus associated with an apparent acceleration of the mechanism for measuring days and minutes.
A third form of linkage is where timing mechanisms are distinct but one relies on the other to operate normally. For example, if interval timing across a wide range of intervals were based on phase coincidence of multiple ultradian oscillators, a circadian clock might be necessary for establishing and maintaining a common starting phase. Evidence supporting a critical role for circadian clocks in interval timing is provided by studies showing that disruption of circadian rhythms in mice by exposure to constant light (LL) completely eliminates the ability to time short intervals (Agostino, do Nascimento, et al., 2011; Bussi et al., 2014). Interpretation of those observations is challenged by earlier findings that mice with SCN ablation (Lewis et al., 2003) or clock gene mutations (Cordes and Gallistel, 2008; Papachristos et al., 2011) show unimpaired interval timing. Agostino, Golombek, et al. (2011), however, identified limitations to these studies. In the report demonstrating intact interval timing in SCN-ablated mice (Lewis et al., 2003), the majority of mice from both the lesion and the control groups demonstrated very imprecise timing functions, raising the possibility of a “floor effect” that could have obscured lesion-specific differences. Histological verification that the SCN lesions were complete was also not provided in that study. A report that Clock mutant mice show no deficits in interval timing (Cordes and Gallistel, 2008) demonstrates that the Clock gene is not necessary for interval timing, but that report does not rule out a role for circadian rhythmicity, because the mice were housed in 12:12 LD, which sustains rhythmicity in the Clock mutant mouse (Vitaterna et al., 1994). Finally, a report that Cry1/Cry2 double knock-out mice show normal interval timing (Papachristos et al., 2011) is also complicated by study procedures. To motivate performance, the mice in that study were maintained on a restricted feeding schedule. Cry1/Cry2 double knock-out mice, although arrhythmic in constant conditions, can show entrainment of circadian rhythms to a daily feeding schedule (Iijima et al., 2005). Activity was not recorded in task-performing animals in the Papachristos et al. study, but it is likely that behavioral rhythms and circadian oscillators in brain areas important for interval timing, such as the dorsal striatum, were entrained to the daily meal (Wakamatsu et al., 2001; Patton et al., 2014; Verwey and Amir, 2012). These interpretive issues leave open the possibility that interval timing in mice does require circadian timekeeping.
We conducted 2 experimental manipulations to determine whether interval timing in rats is also dependent on circadian organization of behavior. We first attempted to extend to rats the evidence obtained in mice that interval timing is abolished by suppression of circadian rhythms in LL. We then assessed whether slowing of the circadian clock and subsequent desynchrony from LD, by intake of 25% D2O in place of water (Pittendrigh and Daan, 1976), might slow or disrupt interval timing measured concurrently. We could find no evidence for disruption of interval timing by procedures that markedly disrupted circadian timing.
Methods
Animals and Ethics Statement
All procedures were approved by the University Animal Care Committee at Simon Fraser University (Permit 1065p-12) and conformed to the Canadian Council on Animal Care’s Guide to the Care and Use of Experimental Animals and Canada’s Animals for Research Act. twenty-eight adult male Sprague-Dawley rats (Charles River Laboratories, Quebec, Canada) were housed individually in a lighting-, humidity-, and temperature-controlled (~22 °C) vivarium. Animal health was checked once daily. Cages were cleaned and rats weighed once weekly.
Activity Recording and Analysis
Locomotor activity was continuously monitored using infrared motion sensors located above the cage and stored at 1-min intervals using the Clocklab data acquisition system (Actimetrics, Wilmette, IL). For visual inspection, activity data were plotted in 10-min bins in standard raster plot format using Clocklab or Circadia (Dr. T. A. Houpt, Florida State University) and as 24-h wave-forms averaging 1 or more days within and across rats using Prism 5.0 (Graphpad Software Inc., La Jolla, CA). Presence of rhythmicity was determined by Lomb-Scargle periodogram analysis using Clocklab.
Apparatus
Behavioral training and testing were conducted in operant chambers (ENV-008; Med Associates, St. Albans, VT) housed in individual sound-attenuated cubicles (ENV-018MD; Med Associates). Each chamber was equipped with a retractable lever, stimulus light, and liquid receptacle located on the front panel. Sound-attenuated cubicles remained illuminated (~75 lux, white LED) for the duration of the training sessions. An IBM-PC computer running MED-PC Version IV Research Control and Data Acquisition System software (Med Associates) attached to an electronic interface (DIG-700P2-R2 and SG-6510D; Med Associates) was used to control the experimental equipment and record the operant data. The time of each lever press was recorded to an accuracy of 10 msec and placed into 1-sec time bins for analysis of mean response rates.
During food restriction schedules, food was accessed via an automated feeding system designed and constructed in the investigators’ laboratory. Food access times were scheduled and controlled remotely using custom software. Automated feeders were attached to the outside of “windows” cut into the sides of the rats’ cages. Access to food was controlled via a sliding plastic door situated between the cage window and a food bin. Metal bars spaced 1 cm apart were installed over the cage window to prevent rats from hoarding large food pellets during food access times. Door position was monitored, and investigators were immediately alerted to any failure to open or close as scheduled. Automated feeding systems were visually inspected daily at the time of operant training.
Operant Training
Operant shaping
Rats were trained to press a lever for a 0.3% saccharin solution reward (~50 µL) via a continuous reinforcement schedule in which each lever press results in reward. Additionally, a “free” reward (~35 µL of saccharin solution) was automatically delivered every 3 min. Each training session lasted for 60 min or 60 reinforcements, whichever occurred first. Rats received operant shaping until at least 3 sessions of 60 reinforcements were obtained in less than 60 min.
Fixed-interval training
Rats were next trained on 15-sec and 30-sec FI schedules of reinforcement. Interval initiation was signaled by illumination of the stimulus light located above the liquid receptacle and extension of the lever. The first lever press to occur after the fixed interval resulted in delivery of a liquid reward (~50 µL of 0.3% saccharin solution). Delivery of the reward resulted in termination of the stimulus light, retraction of the lever, and initiation of a 25 ± 5-sec variable intertrial interval (ITI). If no response was made after 3× the duration of the target interval (i.e., 45 sec or 90 sec, for 15-sec and 30-sec interval schedules, respectively), the trial was terminated and scored as an omission. Each session consisted of 60 trials. All rats were given at least 5 FI sessions for each interval.
Peak interval training
In the 15-sec and 30-sec PI procedures, FI trials were intermixed with unreinforced PI trials (described below) with an FI:PI trial ratio of 3:2, semirandomly distributed. In PI trials, trial initiation was signaled by illumination of the stimulus light and extension of the lever (as in FI trials); however, in PI trials, the signals remained active for 3× the duration of the usual interval (i.e., 45 sec or 90 sec) with no reinforcements given. At the end of the PI interval, the trial was terminated independent of response, and a 25 ± 5-sec variable ITI was initiated. If no response was given during the trial time, the trial was scored as an omission. Each session consisted of 60 trials.
Experimental Timeline
LD 12:12, heavy water administration (LD)
Rats in the LD group (n = 7) were housed in a 12-h light, 12-h dark (12:12 LD) cycle with food and water available ad libitum for 25 days before water access was gradually restricted. The rats were then trained on the FI and PI tasks 6 h after lights-on (1100 h local time) and given approximately 15 min of water access immediately following training. After 12 training sessions on the 30-sec PI task, the rats were given 30 min of food and water access and then were food deprived and left undisturbed in constant dark (DD) for 80 h. Following food deprivation, the rats were given 6 days of ad libitum food and water availability before resumption of the water restriction schedule and 30-sec PI training. Performance on the PI 30-sec task was allowed to stabilize, at which point the rats had daily water rations replaced with 25% D2O for 2 days with no operant training sessions. The rats were then again tested on the 30-sec peak procedure (while still receiving 25% D2O for water rations). Following 6 sessions of training, the rats were given an additional 8 days of ad libitum 25% D2O access before water restriction and testing on the 30-sec peak procedure resumed. Following training, the rats were given ad libitum 25% D2O access in DD for 3 days. The rats were then switched to ad libitum water (no D2O) access for 8 days, were given sufficient time for circadian rhythms disrupted by D2O to reentrain to LD, and then were water restricted and tested on the 30-sec peak procedure.
Constant bright light, ad libitum food access (LL-AdLib)
Rats in the LL-AdLib group (n = 7) were maintained in bright LL (~250 lux maximum cage exposure), with food and water available ad libitum, for 44 days, a sufficient length of time to eliminate free-running, SCN-mediated circadian rhythms in all but 1 rat. Lighting was provided by an external bank of cool white fluorescent tubes supplementing the LED cage lights. Over a period of 5 days, the rats were gradually water restricted to a single 1-h access period and then trained on the 15-sec FI and PI tasks. Task-performing rats received approximately 15 min of water access immediately following operant training. Immediately following the final 15-sec PI session, the rats received 3-h unrestricted access to food and water and were then food deprived and left undisturbed in LL for 73 h. After a 6-day ad libitum food and water recovery period, the rats were once again water restricted and trained on the 30-sec FI and PI tasks.
Constant bright light, 4-h ultradian feeding schedule (LL-RF)
Rats in the LL-RF group (n = 7) were maintained in LL (~250 lux) with food and water available ad libitum for 100 days, a sufficient length of time to eliminate free-running, SCN-mediated circadian rhythms in all rats. To prevent the rats from voluntarily concentrating their food intake to the time of day when water would be available, food access was then restricted to 30-min food access periods, 1 period every 4 h (0300, 0700, 1100, 1500, 1900, and 2300 h local time). After 5 days of gradual water restriction, the rats were trained on the 15-sec FI and PI tasks. The rats were given 15-min daily water access during the first food access window after operant training (i.e., at 1500 h local time). This food access window was reduced to 10 min in length to discourage binge feeding. After the final 15-sec PI session, the rats were given 30 min of food and water access and then were food deprived and left undisturbed for 80 h. After a 4-day ad libitum food and water recovery period, the rats were food and water restricted as before and trained on the 30-sec FI and PI tasks.
Constant bright light, randomized training schedule (LL-Random)
Rats in the LL-Random group (n = 7) were maintained in brighter LL (~750 lux) with food and water available ad libitum for 60 days, a sufficient length of time to eliminate free-running, SCN-mediated circadian rhythms in all rats. Following 5 days of gradual water restriction, the rats were trained and tested on the 15-sec FI and PI tasks. To prevent entrainment to the interval timing tasks or to water availability, the interval between operant sessions was pseudo-randomized, with a minimum of 12 h and a maximum of 36 h between sessions. After the final 15-sec PI session, the rats were given ad libitum access to food and water and were left undisturbed for 7 days to check for the presence of circadian rhythmicity. The rats were once again water restricted and trained on the 30-sec FI and PI tasks using the same pseudo-randomized schedule. Following the final 30-sec PI session, the rats were given 1 h of food and water access and then were food deprived and left undisturbed for 80 h.
Data Analysis
Peak interval performance was analyzed using Excel (Microsoft, Redmond, WA) and Prism 5.0 (Graphpad Software). Mean response rate is expressed as responses per minute from the last 5 sessions of PI training. Omitted trials were not considered in this analysis. Mean response rates for each rat were fit using the Marquardt-Levenberg iterative algorithm to find the coefficients of a Gaussian + linear equation that gave the best fit via square-root minimization between the equation and the data. The following generalized model was used:
where t is the current 1-sec bin and R(t) is the mean number of responses at time t (Buhusi et al., 2005). Using this equation, we obtained for each rat the following parameters: peak time (t0), peak width (b), and peak rate (a + d).
Start and stop rate indices were calculated as defined by Cheng and Meck (2007). Briefly, for the 15-sec PI procedure, the start rate index and the stop rate index were defined by the response rate over the 3 sec immediately prior to or after the criterion time, respectively, divided by the total response rate over the 15 sec before (for starts) or after (for stops) the criterion time. When applied to the 30-sec PI procedure, the same method was used except that the lengths of the intervals defining the numerators and denominators were proportionally increased to 6 sec and 30 sec, respectively.
To analyze the effects of D2O on interval timing performance, training in the LD group was divided into 4 conditions: the last 5 sessions on the PI-30 task (H2O-1), the first 5 sessions of the first round of D2O training (D2O-1), the 5 sessions of the second round of D2O training (D2O-2), and the 5 sessions of training after return to H2O (H2O-2). During this portion of training, curves could not be fitted to the average response distributions of 2 rats (one for the D2O-2 condition only, another for both D2O-2 and H2O-2). Thus, these rats were excluded from all analyses of response distribution parameters. For peak-by-session analyses, there were 3 sessions in which the response distributions for individual rats could not be fit with a curve (2 sessions in the D2O-2 condition and 1 session in the H2O-2 condition, all with different rats). In these cases, the missing data point was replaced with the mean peak time for the individual rat in that particular condition for the purpose of running the 2-way analysis of variance (ANOVA). These data points were excluded from regression analyses. All means are reported with standard errors. Statistical analyses were carried out as described in the text using Prism 5.0 (Graphpad Software) with alpha level set at p < 0.05.
Results
Assessment of Circadian Rhythmicity in Constant Light
The objective of this study was to assess interval timing in neurologically intact rats in which circadian rhythms mediated by the SCN pacemaker were abolished. We therefor paid special attention to possible residual rhythmicity prior to, during, and after operant training. Rats in the LL-AdLib group were maintained in bright LL for 44 days prior to water restriction and training. Visual inspection of activity data in double-plotted actogram format revealed the expected gradual loss of circadian rhythmicity. Nonetheless, an impression of rhythmicity was evident in most actograms due to obligatory health inspections that occurred once daily between 0900 h and 1500 h (Fig. 1A; Suppl. Fig. S1A). Although the inspections were purely visual (no physical contact), rats usually did awaken and orient to the research staff. The impression of rhythmicity was confirmed quantitatively by Lomb-Scargle periodogram, which revealed broad, low-amplitude 24-h components in the power spectra. Consequently, the rats were left undisturbed for 3 days over a weekend, with no entry into the vivarium room. The data for this 3-day block were inspected visually using 72-h bivariate waveforms and were analyzed using the Lomb-Scargle periodogram (Suppl. Fig. S2). During this 3-day time block, circadian rhythmicity was detected in only 1 rat. The rats then received operant training to test interval timing.
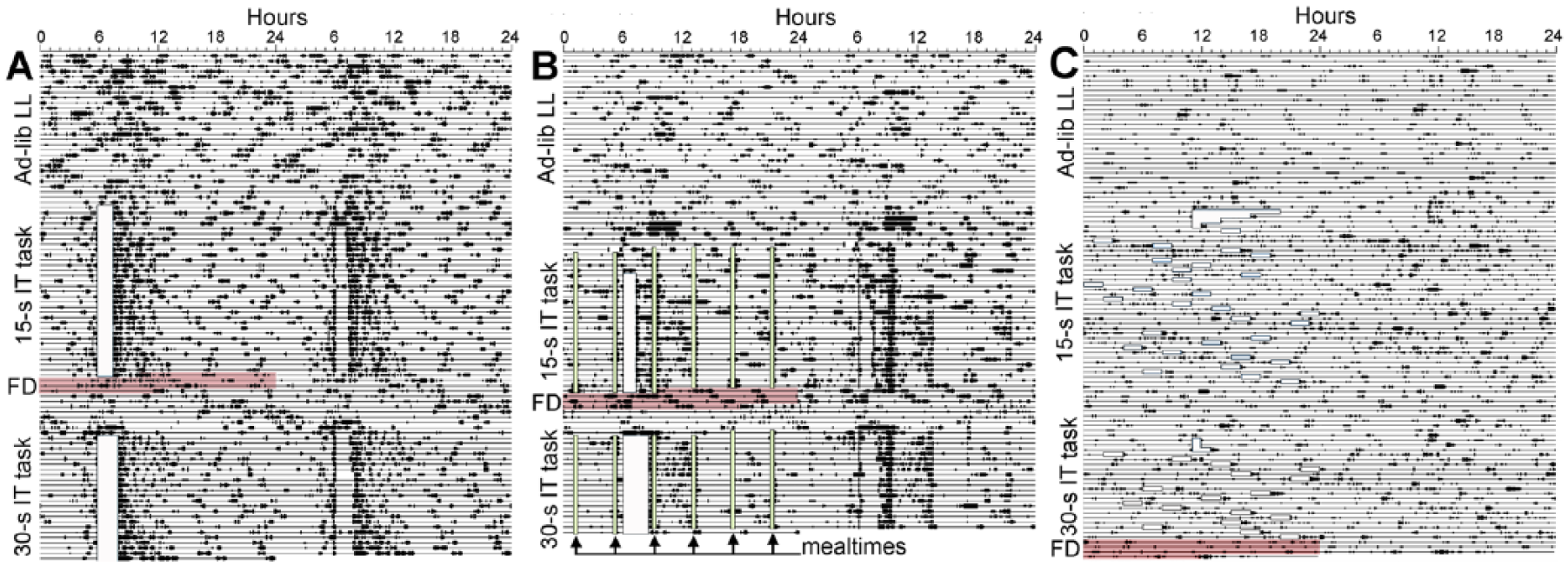
Double-plotted activity records from 3 representative rats maintained in LL with food available either ad libitum (A, C) or restricted to 6 daily 30-min food access periods (B). Data are plotted in 10-min bins from left to right. Consecutive days are plotted left to right and top to bottom. Training and testing time is denoted by opaque white boxes. Food access periods (in B) are denoted by vertical light-green shaded boxes, and by the arrows on the lower left of the panel. Food deprivation following peak interval testing is denoted by red shading.
Interval timing in this study was measured using lever pressing rewarded by sweetened water. To motivate rats to perform the lever pressing response, all rats were restricted to approximately 15 min of water access each day after the operant training session. Thirsty rats may restrict their own food intake to coincide with the time of water access. This could restore circadian organization in LL-exposed rats by nonphotic inputs to the SCN pacemaker or to food-entrainable circadian oscillators elsewhere in the brain (Mistlberger, 1992, 1993; Lamont et al., 2005). This concern was validated in LL-AdLib rats by weighing food consumption during the 3 h immediately following the 15-min drinking period each day. Rats in this group were found to consume 44.2% ± 2.3% of their total daily caloric intake in this 3-h time block. Following training on the 15-sec interval timing task, LL-AdLib rats were food deprived for 3 days to determine whether the binge feeding might have induced a 24-h food-anticipatory activity rhythm. This did not seem to be the case, as there was no evidence for a persisting anticipation rhythm. However, when food and water were then provided ad libitum for the next week, all but 1 rat exhibited a low-amplitude, free-running circadian activity rhythm, with the onset of the daily active period extrapolating back to the previous water access time (Fig. 1A; Suppl. Fig. S1A). Periodogram analysis confirmed significant circadian rhythmicity in 6 of the 7 rats. These results suggest that a daily schedule of operant training, drinking, and/or eating might have rescued circadian rhythms controlled by the SCN. This could in turn support interval timing in this group if circadian organization is a prerequisite for short interval timing.
To prevent a rescue of circadian rhythmicity by the test procedure, the procedures were modified to eliminate potential nonphotic circadian time cues. The first modification was to implement a noncircadian feeding schedule (group LL-RF). A second modification was to schedule training and testing sessions at pseudo-random times of day (group LL-Random). Prior to operant training, rats in the LL-RF group were maintained in LL with food and water access ad libitum for 100 days. This included a 72-h time block during which no person entered the vivarium room. Actograms, 72-h bivariate waveforms, and periodogram analyses confirmed no significant 24-h rhythmicity in all 7 rats (Fig. 1B; Suppl. Figs. S1B, S2). When the rats in this group began water restriction and operant training, binge feeding following the training sessions was prevented by restricting food intake to 6 small meals per day, spaced at 4-h intervals. With food restriction in place, the amount of food consumed during the 3 h after water access as a percentage of total daily intake was significantly reduced (23.4% ± 1.7%, t12 = 7.24, p < 0.001), and there was no evidence of circadian rhythmicity during either food deprivation or subsequent ad libitum food access (Fig. 1B; Suppl. Fig. S1B).
To eliminate all potential 24-h time cues, rats in the LL-Random group were maintained in bright LL (~750 lux) with food and water available ad libitum for 60 days, a sufficient length of time to severely disrupt free-running, SCN-mediated circadian rhythms in all rats. This included a 10-day time block during which the vivarium room was not entered. To prevent entrainment to the interval timing task or water or food intake, the interval between operant sessions was pseudo-randomized, with a minimum of 12 h and a maximum of 36 h between sessions. With water access provided immediately after operant testing, this random testing schedule eliminated all 24-h time cues in both the task itself and any water-associated binge feeding. Visual inspection of actograms confirmed severe disruption of circadian rhythmicity in all of the rats across the approximately 70 days of experimental procedures (Fig. 1C; Suppl. Fig. S1C). Using data from the 10-day block during which the vivarium room was not entered, the Lomb-Scargle periodogram detected significant rhythmic components in the ultradian (5-7 h) range in all 7 rats. Very low amplitude rhythmic components in the 20 to 30 h range were also evident (Suppl. Fig. S3). Lomb-Scargle analyses of 7-day blocks immediately following the 15-sec and 30-sec PI tests detected a low-amplitude but significant circadian periodicity in both blocks in 2 of 7 rats. All statistical analyses reported in the next session were therefor analyzed twice, once including all 7 rats and again after excluding the 2 weakly rhythmic rats. Using the smaller subset of 5 rats did not change any of the inferential statistical test outcomes, and so the full sample of 7 rats is presented, to show all of the data, and descriptive statistics for the subset are provided in the supplementary materials (Suppl. Table S1).
Interval Timing Is Not Impaired Despite Loss or Severe Disruption of Circadian Rhythms in Long-term LL
After extended time in LD or LL as described above, rats in all 4 groups were gradually water restricted and subsequently trained and tested on 15- and 30-sec PI procedures. To analyze the parameters of the response distributions during the peak interval tests, response rates per 1-sec bin, averaged over the last 5 sessions of peak training for each interval, were fitted with Gaussian + linear curves, and the parameters peak time, peak width, and peak rate were obtained for each rat (see Fig. 2A for response distributions). All curves had significantly better fits, as measured by extra sums-of-squares F tests, than simple linear models applied to the same data (all p < 0.001). Three separate 4 × 2 mixed-factor ANOVAs were used to compare differences in peak time, peak width, and peak rate both between groups of rats (between-subjects factor) and between target interval durations (within-subjects factor). For all 3 parameters, there was a significant effect of target interval (peak time: F1,24 = 6.31, p = 0.019; peak width: F1,24 = 11.16, p = 0.003; peak rate: F1,24 = 46.52, p < 0.001), with later relative peak times, smaller relative peak widths, and lower peak rates for the 30-sec interval compared with the 15-sec interval. There was no significant main effect of group (peak time: F3,24 = 0.31, p = 0.820; peak width: F3,24 = 1.51, p = 0.236; peak rate: F3,24 = 0.50, p = 0.686) or group by target interval interaction (peak time: F3,24 = 0.06, p = 0.979; peak width: F3,24 = 0.25, p = 0.863; peak rate: F3,24 = 2.20, p = 0.114) for any of the parameters tested (Fig. 2, B- D).
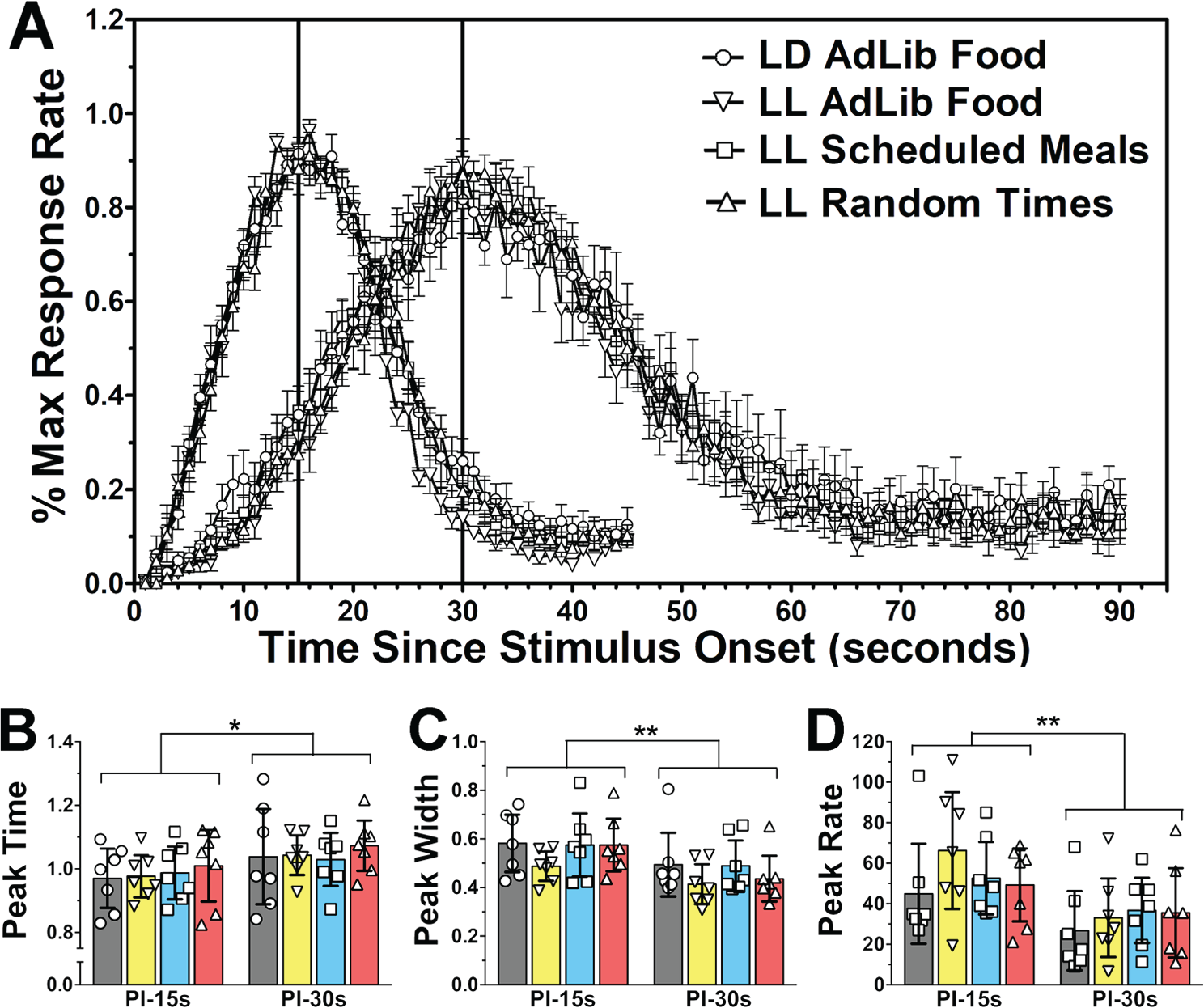
Performance on 15- and 30-sec PI tasks in rats housed in LD 12:12 (circles in all panels), LL with ad libitum food access (inverted triangles), LL with 6 daily meals scheduled at 4h intervals (squares), and LL with operant training done at pseudo-random intervals (triangles). (A) Group mean (±SEM) response rates for the last 5 sessions of the 15- and 30-sec PI procedure expressed as percentage of the maximum response rate for each rat at each 2-sec bin following the stimulus onset (beginning of trial). (B-D) Dot-plots and group mean (bars with 95% confidence intervals) of parameters obtained from curves fitted to the response distributions of each rat for the last five PI sessions for 15- and 30-sec targets. (B) Relative peak time. (C) Relative peak width. (D) Peak Response Rate. **p<0.01, *p<0.05 (mixed-factor ANOVAs).
Given the observation that separate thresholds may be used to start and stop responding around a criterion time (Church et al., 1994), start and stop rate indices were also calculated. Two separate 4 × 2 mixed-factor ANOVAs were used to compare differences in start and stop rate indices both between groups of rats (between-subjects factor) and between target interval durations (within-subjects factor; Fig. 3, A and B). For the start rate index, there was a significant main effect of target interval (F1,24 = 31.16, p < 0.001), with indices being higher for the 30-sec than 15-sec target interval. There was no main effect of group (F3,24 = 0.26, p = 0.852) or group by target interval interaction (F3,24 = 0.16, p = 0.924). For the stop rate index, there was no significant effect of target interval (F2,24 = 0.16, p = 0.691), group (F3,24 = 0.81, p = 0.499), or group by target interval interaction (F3,24 = 0.69, p = 0.565).
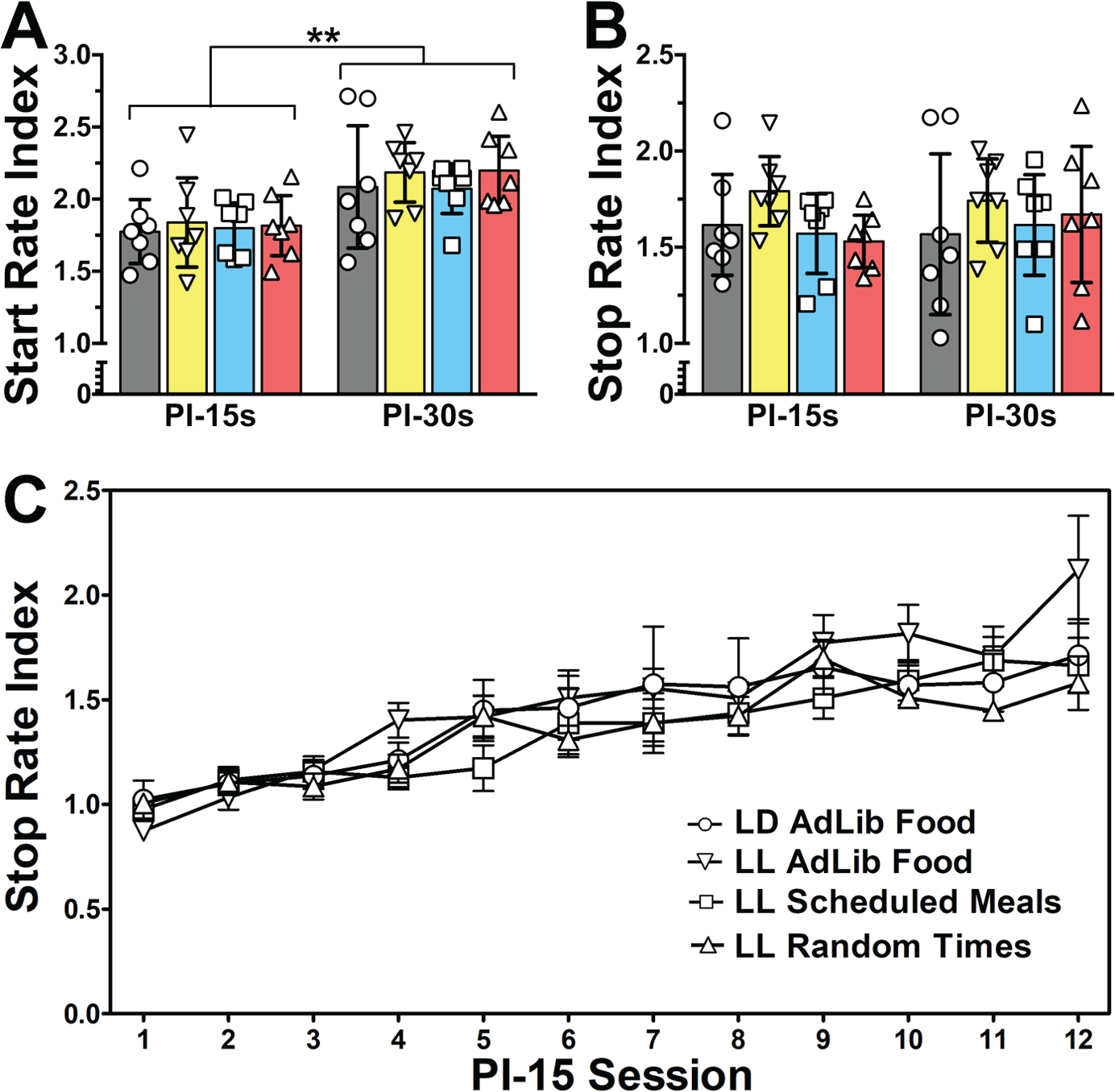
Performance on 15- and 30-sec PI tasks in rats housed in LD 12:12 (circles in all panels), LL with ad libitum food access (inverted triangles), LL with 6 daily meals scheduled at 4-h intervals (squares), and LL with operant training done at pseudo-random intervals (triangles). (A-B). Dot-plots and group means (bars with 95% confidence intervals) for (A) start and (B) stop rate indices. (C) Group mean (± SEM) stop rate index for each 15-sec PI session. **p<0.01, *p<0.05 (mixed-factor ANOVAs).
Stop rate indices for each session of the 15-sec peak procedure were used to examine potential group differences in the rate at which the rats learned to stop responding after the target interval (Fig. 3C). A 2-way mixed-factor ANOVA revealed a significant main effect of session (within-subjects factor; F11,264 = 24.65, p < 0.001) but no significant effect of group (between-subjects factor; F3,264 = 1.06, p = 0.385) or group by session interaction (F33,264 = 0.96, p = 0.527). Furthermore, regression lines fitted to stop rate indices over session revealed that all groups displayed significant, positive nonzero slopes (LD: F1,82 = 26.17, p < 0.001; LL-AdLib: F1,82 = 84.88, p < 0.001; LL-RF: F1,82 = 58.75, p < 0.001; LL-Random: F1,82 = 58.85, p < 0.001), none of which differed from LD controls (LD vs. LL-AL: F1,164 = 3.93, p = 0.148; LL-RF: F1,164 = 0.04, p > 0.999; LL-Random: F1,164 = 0.42, p > 0.999).
Start rate indices for each session of the 15- and 30-sec FI procedure were used to determine whether there were differences in the rate at which the rats acquired the start response threshold. Two separate 2-way mixed-factor ANOVAs revealed no significant effect of group (between-subjects factor; 15-sec FI: F3,96 = 0.41, p = 0.744; 30-sec FI: F3,96 = 2.23, p = 0.111) but a significant main effect of training session (within-subject factor; 15-sec FI: F4,96 = 15.84, p < 0.001; 30-sec FI: F4,96 = 102.40, p < 0.001), with start rate indices increasing across sessions, and a group by session interaction (15-sec FI: F12,96 = 2.31, p = 0.012; 30-sec FI: F12,96 = 2.84, p = 0.002; data not shown). Bonferroni-corrected post hoc comparisons showed that this interaction was driven by start rate indices that differed significantly between the LD and LL-Random groups on just 1 of the 5 training sessions at each interval (session 4 for the 15-sec interval, t = 2.63, p < 0.05, and session 1 for the 30-sec interval, t = 2.96, p < 0.05, with indices being higher in the LD condition in both cases).
Interval Timing Is Not Significantly Disrupted by Deuteriation (25% D2O)
Consumption of 25% D2O in place of drinking water robustly lengthens the circadian period of SCN-dependent rhythms in constant conditions and phase delays or disrupts entrainment in the presence of an LD cycle, in mice, Syrian hamsters (Pittendrigh and Daan, 1976), and rats (Mistlberger et al., 2001). If interval timing and circadian rhythms share common processes, D2O consumption within a few days should slow or disrupt interval timing. To explore this, rats initially tested in LD were retested under 4 additional conditions. In condition H2O-1, the rats were retested on the 30-sec peak procedure in LD. In condition D2O-1, rats were maintained on 25% D2O for 2 days and were then tested again on the 30-sec peak procedure (receiving 25% D2O water rations throughout). Following 5 sessions of training, only 1 rat exhibited a clear delay in LD-entrained nocturnal activity onsets, suggesting an inadequate dose of D2O. Therefore, in condition D2O-2, the rats were given ad libitum 25% D2O for an additional 8 days before water restriction and testing on the 30-sec peak procedure resumed. Finally, to test for a “rebound effect” (a temporary leftward shift in the timing function; Meck, 1996) following withdrawal of D2O in condition H2O-2, the rats were given water (no D2O) ad libitum for 8 days to normalize circadian rhythms and were then water restricted and retested on the 30-sec peak procedure (see Fig. 4B for training sequence).
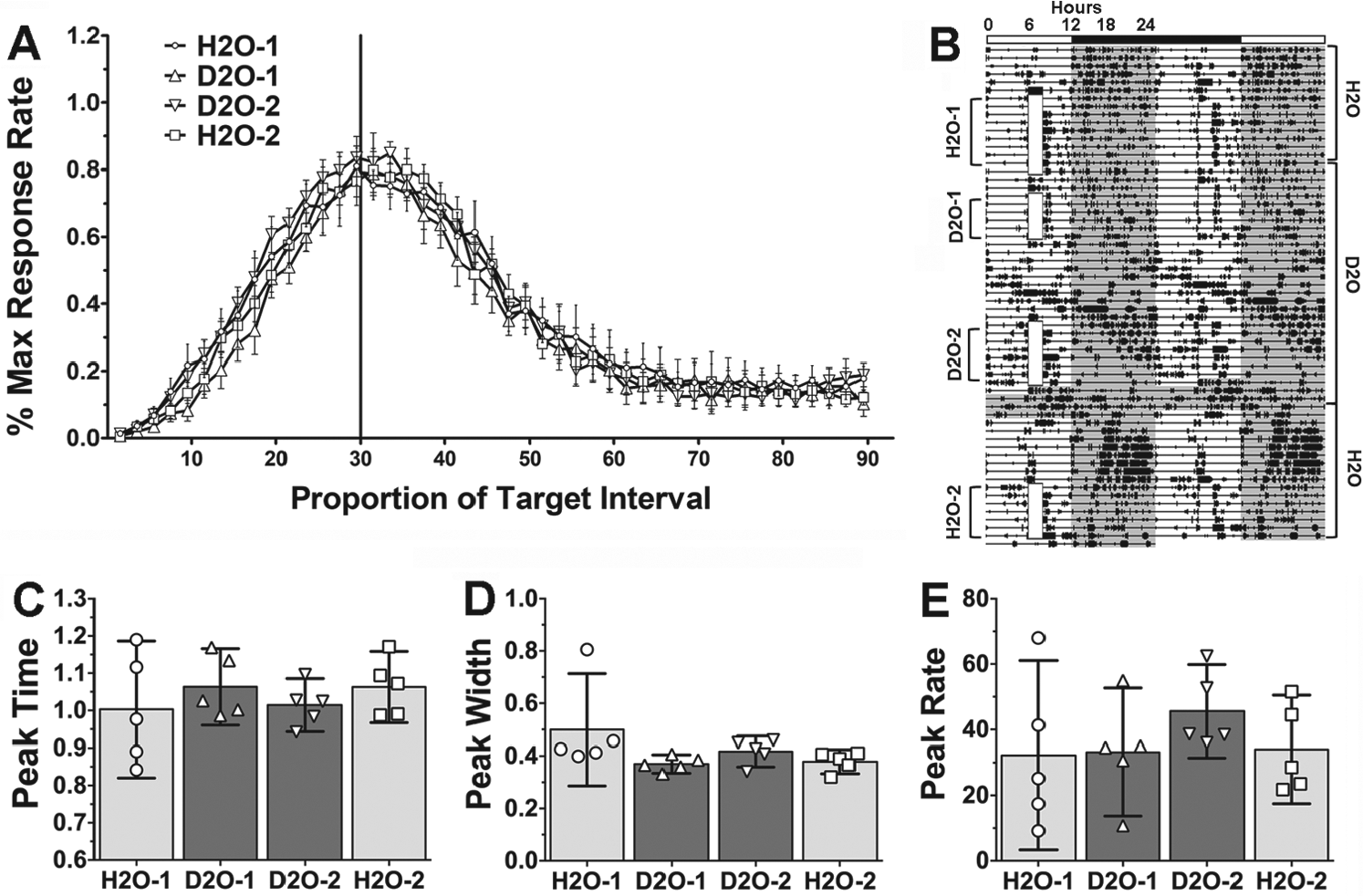
Performance on 30-sec PI task in rats housed in LD 12:12 during initial H2O baseline (H2O-1; circles in panels A,C-E and light grey bars in panels C-E), initial 25% D2O administration (D2O-1; triangles and dark grey bars), following an additional 8 days of ad libitum D2O access (D2O-2; inverted triangles and dark grey bars), and return to H2O (H2O-2; squares and light grey bars). (A) Mean (±SEM) response rates for the 5 sessions of the 30-s PI procedure expressed as percentage of the maximum response rate for each rat at each 2-second bin following the stimulus onset (beginning of the trial). (B) Representative activity record (plotting conventions as in Fig. 1). Training time is denoted by opaque white vertical boxes. Testing condition is indicated on the left of the activity record. Times that the animal was given H2O or D2O is indicated to the right. (C-E) Dot plots and group means (bars with 95% confidence intervals) obtained from curves fitted to the response distributions of each rat for the five PI sessions in each condition. (C) Relative peak time. (D) Relative peak width. (E) Peak Response Rate. No significant differences were found for any of the measures (repeated-measures ANOVAs).
Three separate repeated-measures ANOVAs revealed no significant differences in peak time (F2,12 = 1.74, p = 0.213), peak width (F2,12 = 2.55, p = 0.105), or peak rate (F2,12 = 2.44, p = 0.114) between the 4 conditions (Fig. 4, C and D; n = 5). Furthermore, another 2 repeated-measures ANOVAs revealed no significant differences in the start rate index (F3,18 = 1.31, p = 0.301) or stop rate index (F3,18 = 0.35, p = 0.791) between the conditions (Fig. 5, A and B; n = 7).
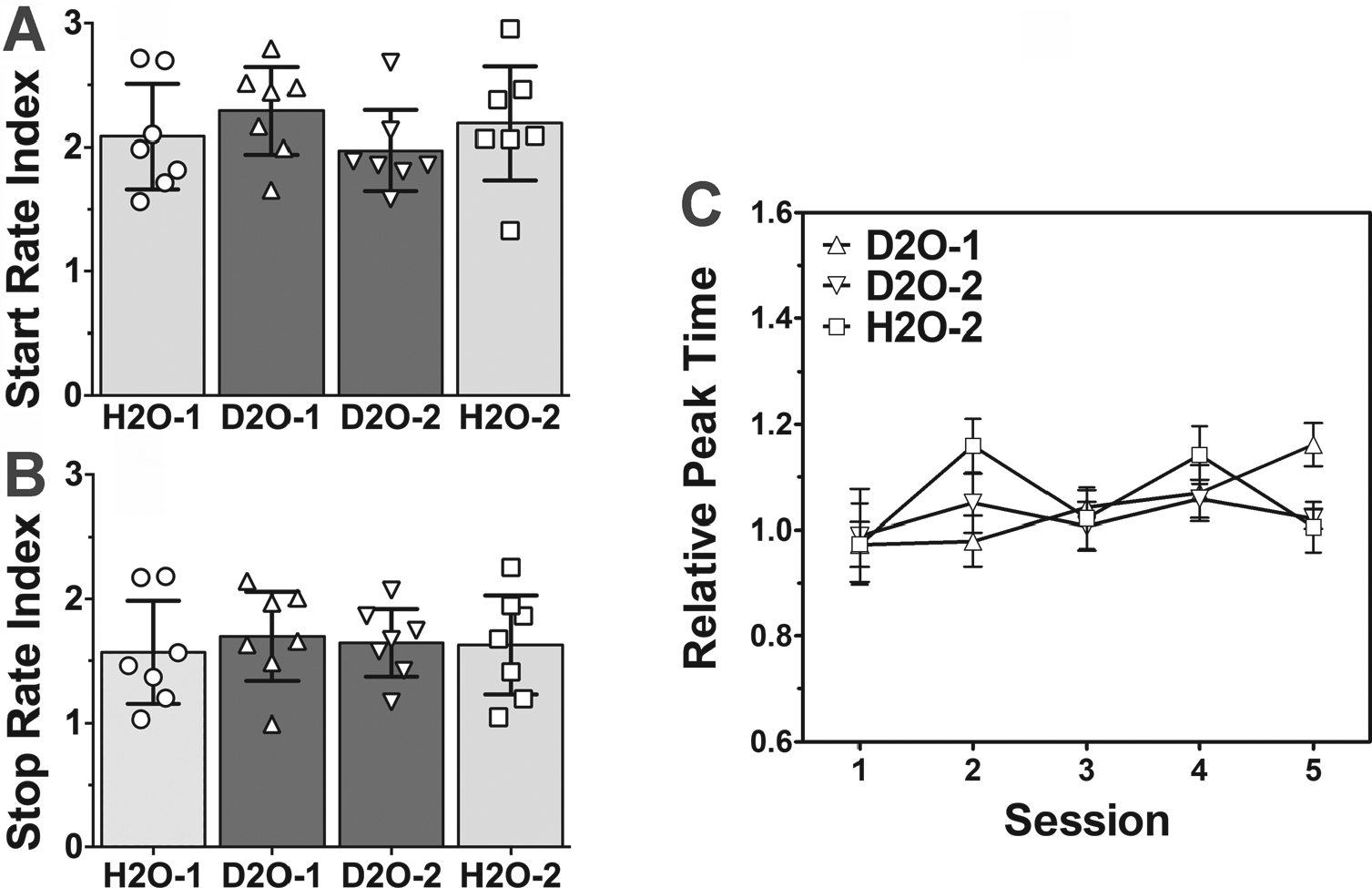
Performance on 30-sec PI task in rats housed in LD 12:12 during initial H2O baseline (H2O-1; circles in all panels and light grey bars in panels A,B), initial 25% D2O administration (D2O-1; triangles and dark grey bars), following an additional 8 days of ad libitum D2O access (D2O-2; inverted triangles and dark grey bars), and return to H2O (H2O-2; squares and light grey bars). (A, B) Dot plots and group mean (bars with 95% confidence intervals) for (A) start and (B) stop rate indices in each condition. No significant differences were found for any of the measures (repeated-measures ANOVAs). (C) Mean (± SEM) relative peak time across the 5 15-sec PI sessions for each condition. No significant main effect was found for condition or PI session (two-way mixed-factor ANOVA).
To examine whether there were changes in the response distributions across the 5 days of training in each of the 4 conditions, curves were fitted to each session and peak times were determined (Fig. 5C). A 2-way repeated-measures ANOVA revealed a significant interaction between condition and session (F8,48 = 2.34, p = 0.033, n = 5) but no significant main effects (condition: F2,48 = 0.44, p = 0.652; session: F4,48 = 2.25, p = 0.078; both n = 5). Bonferroni-corrected post hoc comparisons revealed that peak times for session 5 were significantly longer than for session 1 in the D2O-1 condition (t4 = 6.13, p = 0.004) but did not differ in the D2O-2 (t4 = 0.02, p = 0.988) or H2O-2 (t4 = 0.28, p = 0.790) conditions. Also, session 1 peak times did not differ between the D2O-1 and H2O-2 conditions (t4 = 0.01, p = 0.995). Although a regression line fit to peak times over session displayed a significant positive slope in the D2O-1 condition (F1,23 = 12.49, p = 0.002, r2 = 0.35), goodness-of-fit measures for the other 2 conditions were too low (both r2 < 0.01) to allow for meaningful comparisons.
Periodogram analyses were run on the approximately 1 week of D2O intake between conditions D2O-1 and D2O-2 (no interval timing training). Significant peaks in the circadian range were present in all but 1 rat. On average, the rats (excluding the one with no significant peak in the circadian range) showed a significant rhythm of 26.87 ± 0.49 h (range, 24.6-28.05 h). This is a 2.87-h lengthening of tau from 24 h, representing an 11.9% increase.
To further examine changes in response distributions, 2-way repeated-measures ANOVAs were conducted on start and stop rate indices for each session of interval training (data not shown). There was a significant main effect of session for the start rate index (F4,72 = 4.03, p = 0.005, n = 7) but no significant effect of condition (F2,72 = 1.09, p = 0.357, n = 7) or condition by session interaction (F8,72 = 0.73, p = 0.667, n = 7). Bonferroni-corrected post hoc comparisons revealed no significant differences between session 1 and session 5 start rate indices for any of the conditions (D2O-1: t6 = 1.38, p = 0.218; D2O-2: t6 = 1.39, p = 0.214; H2O-2: t6 = 2.22, p = 0.068), although there was an observable tendency for start rate indices to increase across sessions regardless of condition. For stop rate indices there was no significant effect of condition (F2,72 = 0.07, p = 0.937, n = 7), session (F4,72 = 2.00, p = 0.104, n = 7), or condition by session interaction (F8,72 = 1.34, p = 0.238, n = 7). One would expect a rightward-shifted response distribution to have an increased start rate index and a decreased stop rate index; the reverse would be true for a leftward-shifted response distribution.
Discussion
Circadian timing and interval timing in the seconds-to-minutes range have been conceptualized as distinct processes, based on evidence that interval timing is normal in mice with complete SCN ablations (Lewis et al., 2003), Clock gene mutation (Cordes and Gallistel, 2008), and Cry1/Cry2 double knock-out (Papachristos et al., 2011). Modulation of interval timing by time of day has been detected in humans (Aschoff, 1984; 1998; Kuriyama et al., 2005; Nakajima et al., 1998; Pfaff, 1968) and rodents (Agostino, do Nascimento, et al., 2011; Bussi et al., 2015; Meck, 1991; Shurtleff et al., 1990), but a critical role for circadian timing was not suspected until recent observations that disruption of circadian activity rhythms in mice housed in LL completely abrogated interval timing (Agostino, do Nascimento, et al., 2011; Bussi et al., 2014).
Disruption of interval timing by circadian desynchrony has important implications for conceptual models of time perception and for identifying possible environmental antecedents and mechanisms of thought and mood disorders in which time perception is disrupted. We therefore sought to confirm and extend the observations of Agostino and colleagues. We initially attempted to use mice but were not satisfied with the degree of circadian disruption that we could achieve with 3 months of bright LL (unpublished observations). We therefore switched to rats, which in our experience are more susceptible to rapid and complete desynchrony of circadian rhythms in LL (e.g., Mistlberger et al., 1990; Mueller et al., 2011). Three groups of rats were housed in bright LL sufficient to severely disrupt or abolish circadian rhythmicity and were then trained and tested on 15- and 30-sec PI procedures. Interval timing was found to be intact and unimpaired in animals housed in LL (LL-AdLib), even when we eliminated potential circadian time cues related to daily feeding (LL-RF) or both daily feeding and daily testing (LL-Random). Furthermore, deuteriation, which lengthens the period of SCN-dependent rhythms and disrupts entrainment to LD cycles, also failed to disrupt interval timing in rats housed in LD, despite marked circadian disruption. Taken together, these results indicate that in rats, interval timing is not dependent on SCN-mediated circadian output. In this respect, interval timing is similar to ultradian rhythms in voles and to food-anticipatory circadian rhythms in rats, neither of which are eliminated by LL or disrupted by D2O (Gerkema et al., 1993; Mistlberger et al., 2001; Prendergast et al., 2012).
The 95% confidence intervals for the peak times in our D2O and H2O-2 conditions ranged from 11% to 15% of the 30-sec interval. D2O lengthened the 24-h entrained circadian period in our rats by 11.9%. If D2O slowed the speed of an interval timing mechanism by a similar percentage, then this effect would likely be too small to detect in the interval timing data. There is reason, however, to suspect that changes in circadian periodicity would not scale linearly to the interval timing domain. In the only study to date to observe concurrent changes in circadian periodicity and interval timing, mice harboring a point mutation to the zinc finger homeobox 3 (Zfhx3) gene, a transcription factor highly expressed in the SCN, displayed an approximate 5% difference in free-running circadian period but an approximate 15% to 30% change in peak times when tested on a 10-sec PI procedure (Balzani et al., 2016). Change in period, as a result of D2O administration in our animals, was more than 2-fold greater than that observed in the mutant Zfhx3sci/– mice. Assuming a similar relationship between circadian periodicity and interval timing as that observed by Balzani et al. (2015), we would have expected to see as much as a 30% to 60% change in peak time. Despite this, we cannot rule out the possibility that changes in timing as a result of D2O administration were present but not detected with the methods used.
A novel element of our study was the focus on residual rhythmicity and possible rescue of rhythmicity in rats maintained in LL for many weeks. Although rats in the LL-AdLib group were maintained in LL for 7 weeks prior to operant training and were confirmed to lack circadian rhythmicity in all but 1 case, the operant training procedure induced a daily rhythm of food intake, secondary to restriction of water access to 15 min per day. During a subsequent week of ad libitum food and water access, 6 of 7 rats exhibited a significant free-running rhythm, despite continued exposure to LL. Daily restricted feeding has previously been shown to rescue circadian activity rhythms in hamsters made arrhythmic by exposure to LL (Mistlberger, 1993) and to induce rhythmic expression of PER2 in the SCN of rats maintained in LL (Lamont et al., 2005). Conceivably, even weak circadian organization could potentially support interval timing. Because our interval timing results differed from those of Agostino and colleagues, we chose to repeat the LL experiment, keeping the rats (LL-RF) in LL for 100 days prior to training, and then restricting food to 6 daily meals at 4-h intervals during the operant training and testing. When food access was spaced equally across the day, no behavioral circadian rhythmicity was observed during testing or subsequent ad libitum food and water access. This confirms that rescue of rhythmicity in LL-AdLib rats was mediated by the feeding binge that followed the time of water access. This is the first explicit demonstration, to our knowledge, of the potential circadian confound that is embedded in operant training protocols that use food or water restriction. Such protocols are widely used in behavioral neuroscience (reviewed by Mistlberger, 1990). In our case, the potential confound present in LL-AdLib was ruled out as relevant by the results of the LL-RF group. The results of this group indicate that interval timing is not dependent on a synchronized SCN circadian clock and also cannot be dependent on circadian oscillators elsewhere in the brain or body that are entrained by circadian feeding schedules.
Although no behavioral circadian rhythmicity could be detected in the LL-RF group, we nonetheless tested one final group of rats in LL, with the training and testing sessions scheduled pseudo-randomly. This schedule contained no nonphotic circadian time cues to potentially rescue even covert circadian rhythmicity. Despite the complete absence of circadian time cues, interval timing in the LL-Random group was virtually indistinguishable from the other groups. The only difference detected in multiple post hoc statistical tests was an interaction between test session and group, with the LL-Random group showing a lower start rate index compared with the LD-AdLib group, in 1 of 5 sessions of 15-sec FI training and 1 of 5 sessions of 30-sec FI training. The groups did not differ on the other 8 sessions and did not differ when averaged over all sessions. A direct comparison of group mean (and 95% confidence intervals) response distributions for the LD group and the LL-Random group shows the remarkable similarity of the 2 groups on both the 15-sec and 30-sec peak interval sessions (Suppl. Fig. S4), despite severe disruption of circadian timing in the LL-Random group.
Using a random test schedule to eliminate circadian cues in LL rats could have been confounded by variability in the motivation to respond from session to session, due to the differences in the duration of water deprivation preceding each session. This could have artificially deflated the start rate index. However, any differences in motivational state across sessions would have been averaged out over the course of training. The results, in aggregate, indicate that interval timing in the LL-Random group was as robust and accurate as in the other groups, including the LD-AdLib rats.
The control rats in the present study were trained and tested only during the light period; therefore, the results do not provide evidence for or against circadian modulation of interval timing. The commonly cited evidence for circadian modulation of interval timing is complicated by inconsistencies across studies in the parameters of interval timing reported to be modulated. One study in rats reported an earlier peak, suggesting a faster clock speed, during the dark period, when rats are active and body temperature is at the circadian maximum (Shurtleff et al. 1990). This is consistent with the positive correlation between inferred clock speed and body temperature in humans. However, another study reported that clock speed did not vary with time of day in rats, although modality-specific memory and attentional components of interval timing did vary (Meck, 1991). A third study, using mice, reported that interval timing accuracy, but again not clock speed, varied with circadian phase (Agostino, do Nascimiento, et al., 2011). This range of findings indicates that the manner in which the circadian system modulates interval timing is not yet settled.
A widely observed feature of time perception is the “scalar property” (Gibbon, 1977), which refers to the observation that the precision in the response distribution scales linearly across different target intervals. Our data show a small, but significant, violation of this property; although absolute peak widths were broader at the 30-sec interval compared with the 15-sec interval (i.e., timing is less precise at longer intervals), the relative peak widths were slightly narrower for the 30-sec peak procedure (where scalar timing predicts no difference). This feature was evident in all 4 groups of rats, indicating that it was not related to circadian rhythmicity, which differed markedly across groups. Violations of the scalar property have been reported and discussed in the literature (Bizo et al., 2006; Crystal et al., 1997; Crystal, 1999; Gallistel et al., 2004; Lewis and Miall, 2009). Violation of scalar timing may reflect improved temporal discrimination when target intervals match the native frequency of ultradian oscillators hypothesized to mediate short-interval time perception (Crystal 1999). Alternatively, because we did not counterbalance the training sequence, the observed violations may be due to aftereffects from training on the 15-sec task first. However, if this were due to memory interference from prior training, one would expect the violation to be in the opposite direction. In any case, the same pattern was observed in all 4 of our animal groups, again demonstrating that circadian disruption failed to affect a characteristic of interval timing that was evident using our procedure.
In conclusion, the results of this study demonstrate that short interval timing in rats does not require coherent circadian output from the SCN circadian pacemaker or from food-entrainable oscillators elsewhere in the brain and does not reflect the periodicity of the SCN pacemaker when chemically forced out of entrainment to LD. These results support and extend previous findings suggesting that circadian timing and interval timing are based on distinct neurobiological processes. The results do not rule out the possibility that there are species differences in how interval timing is affected by chronic bright light or circadian disruption, but they do rule out the possibility that such effects as previously reported represent a general property of interval timing in mammals.
Footnotes
Acknowledgements
Supported by research grants from NSERC (Canada).
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
