Abstract
The mammalian circadian pacemaker is commonly thought to be a rigid oscillator that generates output under a variety of circumstances that differ only in phase, period, and/or amplitude. Yet the pacemaker is composed of many cells that each can respond to varying circumstances in different ways. Computer simulations demonstrate that networks of such pacemaker cells behave differently under a light-dark cycle compared with constant darkness. The differences demonstrate that the circadian pacemaker is plastic: The pacemaker shapes its properties in response to the circumstances. A consequence is that properties of a pacemaker under a light-dark cycle cannot be derived from studies of the same system in constant darkness. In this paper we show that the dispersion of phase in a network of coupled oscillators can influence ensemble period: For the considered type of coupling, it is demonstrated that the more synchronous the cells are, the longer is the ensemble period. This is consistent with various data sets obtained in mammals, and even with a data set from fruit flies, in which circadian variation in behavior is regulated in a distinctly differently way from that in mammals. We conclude that environmental circumstances such as photoperiod and exposure to light pulses in otherwise darkness modify the phase distribution of the network and, thereby, the period of the ensemble. Our study supports the view that such properties as circadian period are not solely determined by clock genes but are also determined by the genes that regulate the communication in cellular networks.
In many animal species, a biological pacemaker in the nervous system regulates circadian variation in a multitude of aspects of physiology and behavior (Ralph et al., 1990; Liu et al., 1997). Much is known about the structure of that pacemaker, at least in some model systems like rodents and fruit flies. In mammals, the pacemaker is composed of many cells of various types that interact and collectively produce the signals that provide other tissues in the body with the information needed to help modulate physiology and behavior in a circadian fashion. This collection of cells is found in the suprachiasmatic nucleus (SCN) of the hypothalamus (Moore and Leak, 2001). The SCN has often been perceived as a single, integrated clock, producing one integrated signal toward the rest of the body (Beersma et al., 1999; Forger et al., 1999; Johnson, 1999). Commonly, it is implicitly assumed that the circadian pacemaker is some kind of wave generator that has its own intrinsic period and that adjusts its phase and, perhaps, its amplitude in response to the external signals it perceives.
Phase shifts of the pacemaker in response to isolated pulses of light during otherwise darkness have been measured in many mammals, the results being summarized in so-called phase response curves (PRCs) (Johnson, 1999). These curves present the phase shifts of the clock as a function of the circadian phase the animal was in at the time of the light pulse. They provide a basic understanding of the process of entrainment, even if quantitative predictions on entrainment are not upheld in detail (Pittendrigh and Daan, 1976b). Adding an amplitude response curve to the PRC will add to the explanatory power of the model, and adding a period response function (describing changes in period in response to light pulses) will do so even more (Beersma et al., 1999). However, such additions are all based on the concept of a clock as being one integrated and rather rigid construction. Such concept does not take into account that the circadian pacemaker is composed of a multitude of cells that each may behave in different ways and that each may receive its own stimuli and be connected in its own way to downstream processes. Several studies show that the latter situation is likely to apply (Welsh et al., 1995; Nagano et al., 2003; Kalsbeek et al., 2006; Evans et al., 2011). A convincing case in this respect stems from the work of Vanderleest et al. (2009), who demonstrated that phase shifts of the pacemaker are largest when the amplitude of the circadian electrical discharge pattern is maximal. This experimental finding is explained by the authors to result from the synchrony between cells: In winter, circadian amplitude of electrical discharge rate is large because the short photoperiod pulls the phases of the cells’ activity patterns together. As a consequence of this enhanced synchrony, the cells become simultaneously sensitive to light. Virtually all cells respond if a light pulse is applied at the right time. This leads to large phase shifts of the ensemble of cells. In summer, cells are more dispersed in the timing of their activity and fewer cells are simultaneously sensitive to light, leading to smaller responses of the ensemble. The phenomenon of the behavior in the network is incompatible with properties of classic (single) oscillators where large amplitudes work against large phase shifts. It was concluded, therefore, that the SCN cannot be adequately modelled by a classical single oscillator (Vanderleest et al., 2009).
In this paper we investigate the behavior of the mammalian circadian pacemaker from the perspective that the SCN is an ensemble of interacting cells with slightly different properties. Each has the capacity of endogenous self-sustained circadian oscillations (Welsh et al., 1995; Honma et al., 1998). We use simulations obtained from a computer model of such a network, building on earlier work by Enright (1980b) and Beersma et al. (2008). One of the results is that the period of the ensemble signal depends on the phase distribution of the pacemaker cells: The narrower the phase distribution of the cells, the longer the period. Similar mechanisms may underlie variations in free running period induced by exposure to different photoperiods in several species of mammals (Pittendrigh and Daan, 1976a; Myung et al., 2015) and insects (Tomioka et al., 1997). While the data of those studies already implied differences in pacemaker constellation between summer and winter, simulations suggest that these differences are caused by the plasticity of the network of interacting cells, not necessarily by changes within the cells themselves.
The Circadian Pacemaker as an Ensemble of Interacting Pacers
Jim Enright was the first to extensively and successfully tackle the problem of modeling a circadian pacemaker as a network of many interacting pacemaker cells (Enright, 1980b). He considered the class of models in which each cell was attributed a value for each relevant parameter drawn randomly from a normal distribution. Thus, he characterized each parameter by its intercell average and standard deviation.
In his book (Enright, 1980b) and simultaneously published generalization on precision (Enright, 1980a), Jim Enright characterized the model as follows. First, he assumed that each cell in the ensemble would oscillate in a circadian fashion, as was later demonstrated to be correct (Welsh et al., 1995; Honma et al., 1998). The oscillations were supposed to be periodic switches between two states: an electrically active state in which the cell fires action potentials and is capable of activating other cells and an inactive state in which the cell is silent. The interaction between cells was postulated to be such that each cell can initiate its active state earlier if many other cells are active at that time. The effects of light were introduced in the model through the assumption that light can activate cells in a similar way. With this model Enright could simulate a series of phenomena that resemble observations on animal behavior. These include spontaneous development of circadian rhythmicity in the absence of a light-dark cycle; persistent rhythmicity in constant darkness; loss of circadian rhythmicity in pacemaker output in constant light; entrainment to light-dark cycles; aftereffects of zeitgeber cycles with different periods; and generation of realistic phase response curves to light pulses. Unfortunately, the model could not simulate observed changes in animal behavior in response to changes in photoperiod. In 2008, we published an extension of Enright’s model that overcomes this problem (Beersma et al., 2008). The change is that the cells in the extended model are responding to the activity of other cells not only by starting their activity earlier but also by sustaining their activity for a longer time when many other cells are active at that time. This new model accommodates such changes as observed under different photoperiods. The dispersion of phase of the pacemaker cells is wider under long photoperiods than under short photoperiods. This fact results from the pacer responses to light and is sufficient to explain the experimental observation that responses to light pulses applied in summer are smaller than in winter (vanderLeest et al., 2009).
The extended model is defined by specifying the various parameters, which resulted in the simulations shown by Beersma et al. (2008). Some of the mathematical underpinnings are more extensively discussed by Beersma et al. (2011). We concluded that a model of this kind could be considered to represent a daily clock for all seasons: Not only does it generate a pattern of activity of the ensemble, but some cells are consistently active in the morning and others in the afternoon, and they change their timing in the course of the seasons. Linking up downstream processes to specific cells in the SCN can induce different behaviors in the morning or at the end of the day. Such a clock is not just triggering sleep or wakefulness: It can regulate any behavior of which the circadian timing is beneficial to the animal.
Effects of Photoperiod on Behavioral Patterns
The period of the circadian rhythm of locomotor activity in darkness depends on the duration of the photoperiod to which the animal was exposed in the 24-h light-dark cycle before the transfer to constant darkness. In laboratory mice and deer mice (less so in hamsters), the free running period in darkness is shorter after exposure to summer days compared with winter days (Figures 12, 13, 15 and Table 5 in Pittendrigh and Daan, 1976a; Myung et al., 2015). We speculate that functionally, this is almost a necessary requirement for entrainment: Data of phase response curves in mice (Comas et al., 2006) show that the longer the light pulse, the smaller the advances and the larger the delays induced by such pulses. Such a phenomenon is expected from a theoretical point of view: Compare the effects of a light pulse of a few hours with the effects of a much longer light pulse. Both pulses can only briefly hit the region of the PRC where advances are generated, because the response rapidly advances the system to a later phase. Depending on the duration of the light pulse, the residual duration of exposure can hit the delay zone of the PRC, thereby reducing that advancing effect. Under long photoperiods, this response of the system will tend to induce a delayed phase of entrainment. Under extreme conditions, such as close to the arctic circle, in midsummer this response may even cause the system to lose entrainment, as demonstrated, for instance, in the Yellow-necked mouse (Erkinaro, 1969). Both of these types of responses may be undesirable, depending on the behavioral requirements of the animal. One mechanism by which an animal can compensate for the delayed phase induced by cumulative light effects in summer is by having the system generate a short intrinsic period in summer (i.e., during a darkened day in summer, the observed period is shorter than during a darkened day in winter). This is exactly what has been observed in the studies published (Pittendrigh and Daan, 1976a; Myung et al., 2015): In constant darkness, the period of a system previously entrained to long photoperiods is shorter than when previously entrained to short day lengths.
How do animals achieve this functional adjustment? Is it an active process, for instance through differential expression of specific genes, or could this be a passive response built into the network of interacting cells? We will address this question through model simulations. (For a description of the model and the parameter values used in this paper, see the supplementary online information.)
Dispersion of Phase in an Ensemble of Interconnected Pacers
Simulations with the extended model showed that summer days lead to a wider distribution of phase in the ensemble than winter days (Beersma et al., 2008). Under the short days of winter, the active phases of most cells, through their response to light, are more or less compressed in the short light interval and thereby are roughly simultaneous. In the long days of summer, some cells begin to be active near dawn and stop being active halfway through the day, while other cells begin later, some extending their activity up to dusk (Vanderleest et al., 2009). Simulating after-effects of prolonged exposure to different photoperiods is of course possible, but the results are difficult to interpret. If differences emerge, they may have several causes. The effects could be due to the dispersion of phase, or they may have resulted from direct effects of prior light on specific cells. To circumvent this difficulty of interpreting the results, we performed a set of simulations in constant darkness, in which we varied the size of day-to-day fluctuations in period of each individual pacer. In fact, we thereby induced dispersion of phase of the pacers in the absence of light, so without modifying photoperiod. The results of these simulations are presented in Figure 1a. With increased day-to-day variation in pacer period (from left to right in the figure), the period of the ensemble shortens. In Figure 1b we present the results of another way to modify the phase dispersion in the ensemble, again in darkness. Here we decreased the strength of mutual interactions between cells. This also reduces synchrony and leads to shorter ensemble period, just as is the case with increased noise in the ensemble. Qualitatively, such response is exactly of the type needed to overcome the delays that accumulate during long summer days. The mechanisms that lead to this change can be understood from considering the simplest network one can think of: a network of just two identical pacers.

Double plotted model simulations. From top to bottom, subsequent 24-h intervals are plotted beneath each other. Each line shows 48-h of the simulation. (A) From left to right: increasing day-to-day variation in cycle duration (hence decreasing mutual synchrony between the cells) shortens tau. (B) From left to right: decreasing mutual coupling (and hence decreasing mutual synchrony) between cells shortens tau.
For such a simple network, compare two situations, one in which (in the absence of light) the two cells are active simultaneously, and one in which one cell is 1 h earlier than the other one. Consider the response of each cell as described above. In the situation in which the cells are simultaneously active, no other cells are active in the interval before each cell starts its activity. Hence both cells do not experience any stimulation by other cells to start their activity early, so they will start their activity at the time at which they would have started when no interaction with other cells existed. Near the intended end of activity, both cells experience the activating influence of the (active) other cell; hence, both cells will stay active for a longer time than without interaction, causing a longer period in the ensemble than based on individual cell properties alone. This differs from the situation in which one cell is earlier than the other. Now the first cell will simply start at its intended time, but the second pacer, in response to the activation by the first cell, will start earlier than intended. This reduces the difference in timing of activity between the cells and advances half of the ensemble. At the end of the activity interval, one cell senses the activity of the other cell and will continue to be active a little longer. Depending on the relative timing, however, the other cell may or may not experience a similar situation. Therefore, it is likely that the delay induced at the end of the activity intervals is smaller when the cells are not simultaneously active, but—depending on the difference in timing—there also might be no difference between the two situations. More advance and less delay in the nonsimultaneous timing model both lead to a shorter period. So, for the simple model of two identical coupled oscillators, ensemble period is longer when the cells are simultaneously active than when they are not. It is of interest to note here that such an extremely simple system of two coupled phase–only oscillators has been studied before (Fig. 7 in Daan and Berde, 1978) and produced the aftereffects of photoperiod on system period observed in animals.
Within more extended networks similar mechanisms will apply, which explains the observed simulation results of Figure 1 (see supplementary online information). Generally, for networks like the ones we investigate here, a wider dispersion of the phase distribution leads to a faster oscillator.
These are the results of computer simulations, based on networks with specifically defined properties. The question arises whether real SCN cells behave in similar ways. It is quite likely that they do. SCN cells fire action potentials, and action potentials are triggered in response to appropriate changes in membrane potential. If other cells deliver stimulating currents to an SCN cell, the cell will fire action potentials earlier. At the other end of the activity interval, the opposite will happen: Inputs from neighboring activating cells will postpone the time before action potentials are shut down, because the membrane potential will stay above the action potential level for a longer time. For these reasons, we extrapolate from the simulations that also in real SCNs the width of the dispersion of phase in the ensemble of cells influences the period of the ensemble: The broader the phase distribution, the shorter the intrinsic period. This network property serves as an automatic built-in method to regulate intrinsic pacemaker period in adjustment to the photoperiod to which the animal is exposed: a relatively long period in winter and a shorter intrinsic period in summer. The network property also subserves other responses of the system to specific stimuli. These are discussed below.
Transient Responses to Single Light Pulses
If the width of the dispersion of phase determines the intrinsic period of the network, then it does not matter much how the width of the distribution was generated. A light pulse at the beginning of the subjective day would lead to a broadening of the phase distribution, because it would advance a subset of the pacers, while none are delayed. A light pulse at the end of the subjective day would also broaden the distribution, but now because a subset of the pacers is delayed by the light pulse, while none are advanced. While the two pulses elicit opposite immediate phase shifts, the period of the ensemble will initially shorten in both cases. In the dark interval that follows, the pacers will influence each other, which eventually will lead to a return to a phase distribution similar to the initial pattern (except for the phase shift). Hence, the shorter period of the ensemble will be a transient situation, fading away after some time. The expected response patterns are as follows: A light pulse near subjective dawn will induce an advance, followed by additional advances of diminishing size due to the transient shortening of period. Taken together, this will look like a phase shift that needs a couple of days to come to full expression. A light pulse near subjective dusk, in contrast, will look like an immediate shift. The pulse will induce an immediate delay of (part of) the ensemble, but this will be followed by a temporary shortening of the period, generating the impression that the complete phase shift is present after less than 1 day. Sometimes there may even be an overshoot (i.e., a delay shift in the first cycle that is a little larger than the steady-state phase shift observed during subsequent days). Several studies have been published that clearly demonstrate the prolonged transient phase upon advancing light pulses, versus a 1-day overshoot of the shift in the timing of locomotor activity induced by delaying light pulses (Fig. 1 in Daan and Pittendrigh, II, 1976; Fig. 4 in Pittendrigh, 1981). Figure 2 provides an example.
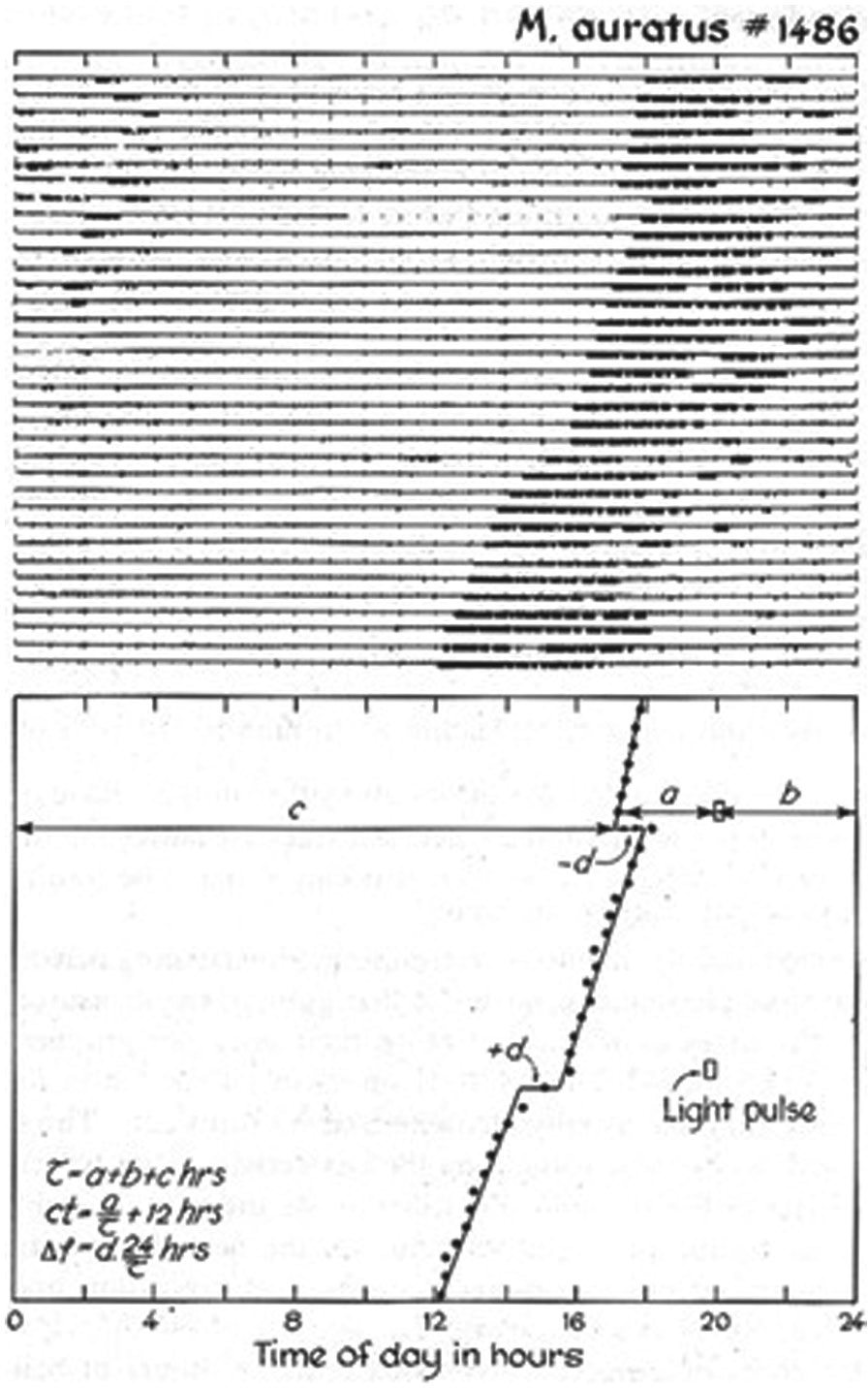
Transient responses of activity-rest patterns to 15-min light pulses in otherwise darkness, applied to a hamster. Note that both acute advances and delays are followed by a few days of shortened free-running period. This figure is reprinted from the Journal of Comparative Physiology A, 106, 253-266, 1976, “A functional analysis of circadian pacemakers in nocturnal rodents, II: the variability of phase responses,” by S Daan and C Pittendrigh, with permission of Springer.
The Influence of Photoperiod on Free Running Period
If the period of the ensemble of pacers is indeed dependent on the width of the dispersion of phases among them, with wider distributions leading to shorter periods, it is possible to estimate how the intrinsic period depends on photoperiod. In long-lasting constant darkness, the pacemaker will show a certain free running period that is determined by the interaction between the pacer cells. Differences in characteristics between those pacers will lead to a certain width of the distribution of their relative phase positions. Under entrainment to a light-dark cycle with a period close to the free running period in DD, the system is likely to entrain in such a way that the center of the phase distribution occurs near the middle of the photoperiod, most pacers being active in the day. At that position in time, a short photoperiod will tend to delay early pacers and tend to advance late pacer cells. As a result, the ensemble will show increased synchrony and, hence, compared with DD, a longer intrinsic tau is expected under short photoperiods. Under a long photoperiod, the light interval may become larger than the width of the phase distribution of the pacers in DD. In that situation, in response to the light, early pacers will be advanced, while late pacers are delayed, leading to a wider dispersion of phase than in DD. Hence, with increasing photoperiod, intrinsic tau will shorten relative to short photoperiods. This effect will become stronger with increasing photoperiod because increasing photoperiods will widen the phase distribution. The decrease in tau, however, will not continue up to the LL situation. In constant light, light-dark transitions do not occur. Hence the activity onsets of all pacers are advanced by the light and activity offsets are delayed, but the absence of light-dark transitions removes the forces exerted by the light-dark cycle to widen the phase distribution of the pacers. Therefore, the LL situation will result in a narrower phase distribution than observed under a very long photoperiod. Hence, it is expected that free running period is larger in DD after exposure to LL than in DD after exposure to a very long photoperiod. We found no data in the mammalian literature to verify these predictions. However, there are data available in fruit flies. Tomioka and colleagues (1997) performed a study in which they systematically varied photoperiod during early development and measured free running period during darkness (DD) afterward. Figure 3 presents their results for the Canton-S Drosophila wild-type strain. Qualitatively, the results follow the predictions. This suggests that also in flies, the distribution of phase in the pacer network modifies the period of the ensemble.
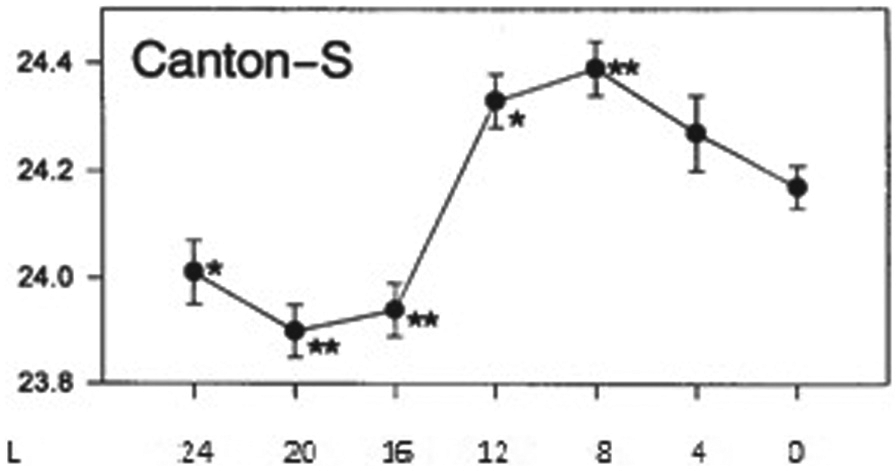
Free running period of Canton-S Drosophila flies after prior exposure to different photoperiods, each in a T-cycle of 24 h. The abscissa indicates the duration of photoperiod. L = 24 refers to continuous light and L = 0 to continuous darkness. L = 24 and L = 0 differ distinctly from the other conditions because they lack light-dark transitions. This figure is reprinted from the Journal of Insect Physiology, 43(3), 297-305, 1997, “Light cycles given during development affect free running period of circadian locomotor rhythm of period mutants in Drosophila melanogaster,” by K Tomioka, K Uwozumi, and N Matsumoto, with permission of Elsevier.
Conclusion and Discussion
The phenomenon of aftereffects of photoperiod, even of single light pulses, on circadian periods can be understood from the interaction between individual oscillating components and by the notion that the SCN is not a rigid pacemaker that generates the same circadian signal under a wide variety of circumstances. Simulations demonstrate that general patterns in pacemaker flexibility can be predicted in considerable detail by a network system. Each cell may well vary its phase position relative to other cells, both as a result of its own intrinsic characteristics and in response to environmental conditions to which this cell is sensitive. The behavior of the cell will influence the behavior of other cells to which it is connected. The result is a plastic pacemaker that sets its characteristics in interaction with its environment. These considerations make clear that the term intrinsic period needs further specification. The intrinsic period (i.e., the period during a day of darkness) of a system previously exposed to prolonged darkness differs from the intrinsic period of a system previously exposed to a light-dark cycle, and the system entrained to a light-dark cycle with a short photoperiod has an intrinsic period that differs from the system entrained to a long photoperiod. We speculate that under real-life conditions, virtually every individual has a pacemaker with an intrinsic period which is very close to 24 h. The system may achieve this by adjusting the phase distribution of the pacemaker cells in response to light exposure. It is entirely possible that there are many days at which the system barely changes its phase in response to the light-dark cycle, even though the system would have a period in long-term constant darkness that deviates from 24 h (see, e.g., Beersma et al., 1999; Hut et al., 1999; Daan, 2000). The period estimates of circadian systems entrained to long photoperiods resemble transitory states, and the rate by which the system returns to the dark-adapted state influences the result. This may explain why some species show substantial differences in free running period in darkness after entrainment to short versus long photoperiods (house mouse, deer mouse) while other species do not (hamster) (Pittendrigh and Daan, 1976a).
There may be additional reasons. Our modeling approach is based on very simple assumptions. All cells of the ensemble are assumed to have identical characteristics: they all signal to all other cells; they are all responsive to light; and they all show responses to lights-on and also to lights-off. In real SCNs, a variety of cell types occur. It may well be that some species evolved subsystems of cells that specifically respond to lights-on and other subsystems that respond primarily to lights-off (Meijer et al., 1986; Inagaki et al., 2007). Such systems may behave differently than predicted in this paper, depending on the specific interactions between the subsets. Recent studies demonstrate the importance of GABA and VIP in this respect (Farajnia et al., 2014; Evans et al., 2013; Freeman et al., 2013; DeWoskin et al., 2015; Azzi et al., 2017). These studies show differences in GABAergic mechanisms between short and long photoperiods or in response to differences in the period of the applied light-dark cycle, especially between ventral and dorsal SCN. The studies do not report, however, that phase dispersion of cells is sufficient to modify ensemble period between summer and winter, as is demonstrated in this paper. Gu et al. (2016a, 2016b) investigated the relationship between network properties (especially the coupling between ventral and dorsal SCN) and ensemble period. In their model, they consider only the situation of constant darkness and conclude that weaker coupling relates to a longer period of the ensemble, which is inconsistent with the observed differences between the seasons (Pittendrigh and Daan, 1976a; Myung et al., 2015). It remains to be investigated whether inclusion of exposure to long photoperiods can induce an opposite response: weaker coupling in summer leading to a shorter free running period instead of longer, as observed in experiments. On top of that, the work by Gu et al. (2016a, 2016b) suggests that light pulses applied in otherwise darkness would increase the free running period of the ensemble as a consequence of reduced coupling. This would lead to transients that are opposite to observations (see section Transient Responses to Single Light Pulses).
Obviously our model is an extreme oversimplification of the SCN, as opposed to, for instance, the models by DeWoskin et al. (2015) and Gu et al. (2016a, 2016b). While this has the disadvantage of not being able to describe the full range of SCN characteristics, it has the advantage of revealing global mechanisms, which are harder to link to specific aspects of more complex models (see Beersma, 2005).
Our simulations support the notion that the classic view of a rigid pacemaker that tells the animal when to sleep and when to be awake is too simple. The pacemaker is composed of many cells in a network. The network by itself regulates the behavior of the system, leading to entrainment under a wide range of external conditions. The entrained states likely differ between conditions. The notion that the dispersion of phase influences the intrinsic period of the system in that specific state helps to explain a range of observations.
The specific dispersion of phase in a specific situation depends on the strength of the mutual couplings between pacer cells, and on the strength of the responses to light, and it sets the intrinsic period of the ensemble. If long summer days require a certain width of the phase distribution in order to let the animal entrain at a certain phase, the sensitivity to light of the pacer cells must be such that the required phase distribution emerges. Sensitivity to the intensity of light, therefore, plays a crucial role. Some species show latitudinal clines in their circadian period in DD (Hut et al., 2013). In view of our reasoning, such clines will play a role in the phase dispersion processes. Species with a relatively low circadian sensitivity to light are expected to have shorter intrinsic period at higher latitudes, whereas species with high circadian sensitivity to light are expected to increase tau with latitude.
Our simulations make clear that the coupling between cells contributes to free running period in the interval after entrainment and, hence, to the phase of entrainment under the light exposure pattern. Clock genes are thought to regulate the circadian pattern generated within single cells. Hence, clock genes are not likely to be involved in the modifications of circadian period and phase discussed in this paper. Instead, coupling between cells will be controlled by those genes that regulate intercellular communication, such as GABA and VIP (Farajnia et al., 2014; Myung et al., 2015; Azzi et al., 2017). Such genes likely will have their influence in all neuronal networks of the organism. They are probably not specific to the SCN.
Footnotes
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
