Abstract
Although it is well known that body temperature (Tb) is higher during the day in diurnal birds than at night, no data are available regarding exactly how Tb varies during a 24-h period, how this differs under different photoperiods, and how it responds to a change in photoperiod. This study used implanted temperature loggers in starlings (
Birds and mammals are endothermic and heterothermic; although deep body temperature (Tb) is routinely maintained above ambient and is fairly constant, it does vary. Mean Tb for all birds in the active phase of their daily cycle is approximately 2.5 °C higher than during the resting phase (Prinzinger et al., 1991). So it is higher during the day in diurnal birds and during the night in nocturnal birds. However, few data are available to show exactly how Tb varies during a 24-h period or how this differs under different photoperiods. Early studies were handicapped by the inability to record Tb in unrestrained birds (Hudson and Kimzey, 1966). This was first made possible by radiotelemetry, in which the sensor/transmitter unit is implanted into the body cavity (Refinetti and Menaker, 1992). More recently, implantable data loggers have been developed. Data loggers allow the acquisition of temporal sequences of digital data so that, for the first time, changes in Tb can be quantified and statistically analyzed. The first aim of this study was to use data loggers to characterize the 24-h profiles of Tb in unrestrained birds (starlings—
The second aim was to assess the degree to which the daily cycle in Tb is circadian. This was achieved in two ways: first by investigating how the daily cycle in Tb changes following an acute increase or a decrease in photoperiod, and second by transferring birds into constant darkness for 24 h. Previous studies have demonstrated a circadian cycle of Tb in mammals (Refinetti and Menaker, 1992) and in birds held in constant darkness (Hudson and Kimzey, 1966; Binkley et al., 1971; Underwood, 1994) but the data are somewhat qualitative.
Materials and Methods
Juvenile starlings were caught at Berwick upon Tweed, UK, 55.8 °N, during August 2013. Only males were kept; females were released. Birds were initially kept in an indoor aviary but were exposed to natural light and temperatures. They were provided with turkey starter crumbs ad libitum. All birds were treated with a single dose of anthelminthic (Flubenvet) in food to eliminate gut parasites and 1% Ivermectin on the skin on the back to remove ectoparasites.
In October, birds were divided into 5 groups and each group was put into an environmentally controlled chamber (2.5 m × 2.0 m and 2.2 m high) in which birds were allowed to fly freely. Temperature was kept at 14 °C (mean summer temperature where the birds were caught) with 5 changes of air per hour. The time that lights came on and went off each day was controlled by a Lutron GRAFIK Eye QS system, Coopersburg, PA, USA. Light intensity increased from 0 to 1000 lux during a 1-min period at dawn and decreased during 1 min at dusk. Photoperiod for all groups was changed daily and simulated the natural decrease at 56 °N until the winter solstice at 7L:17D. One group was then held on 7L:17D (
Daily Tb Cycles under Constant Photoperiods
After several weeks on stable photoperiods, the daily cycles in Tb were assessed using DST nano-T temperature data loggers (Star-Oddi Ltd, Gardabaer, Iceland) in which recorded data are stored in the logger’s internal memory with a real-time clock reference for each measurement. The loggers were synchronized with the same computer as the GRAFIK Eye lighting controls. The loggers were 17 mm × 6 mm, weighing 1 g. Each bird was anesthetized with isoflurane, a 10-mm incision was made in the skin of the nape of the neck, and a logger was inserted subdermally. The incision was closed with tissue adhesive (Vetbond, The Vet Store, Bradfod, UK). The loggers were programmed to start recording 5 days after implantation. Temperature was recorded every 5 min for 6 days. Birds were again anesthetized, the loggers were removed, and data were downloaded using the SeaStar program (Star-Oddi Ltd). The procedures were licensed by the UK Home Office (project license PPL 60/4176).
Daily Tb Cycles during Change in Photoperiod
The birds held under 7L:17D were moved to 14L:7D and those under 17L:7D were moved to 7L:17D. Temperature loggers were implanted into birds as above and programmed to start recording 5 days later. Temperature was logged every 5 min for 3 days under the original photoperiod. Photoperiod was then changed by changing both dawn and dusk equally, so that the middle of the photoperiod remained at the same time. Temperature was logged during the following 3 days. The birds originally held under 14L:7D were moved to 17L:7D and, some weeks later, implanted with loggers programmed to start recording 5 days later. Temperature was recorded for 3 days under 17L:7D. During the fourth day birds remained in total darkness, and then they returned to 17L:7D on the next day.
Data Analysis
To make the statistical analysis more manageable, each group of three 5-min recordings was averaged to provide a figure for each 15 min. Under constant photoperiods, the mean for each bird for each 15-min point was averaged over the 6 days. These data were then used in a repeated-measures ANOVA (Graph Pad Prism, San Diego, CA, USA) followed by Tukey’s multiple comparison test. For the change in photoperiod studies, each single 15-min point was used (i.e., not the means over several days).
Results
Constant Photoperiods
Starlings under 5 different constant photoperiods all showed dramatic daily cycles in Tb, but the pattern differed between photoperiods (Fig. 1). In all groups, body temperature began to increase before dawn and continued to increase after dawn. There was no sudden change at dawn. Under shorter photoperiods, the increase before dawn exceeded that after dawn. In contrast to the gradual changes around dawn, at dusk there was a sudden rapid decrease in temperature under all photoperiods.
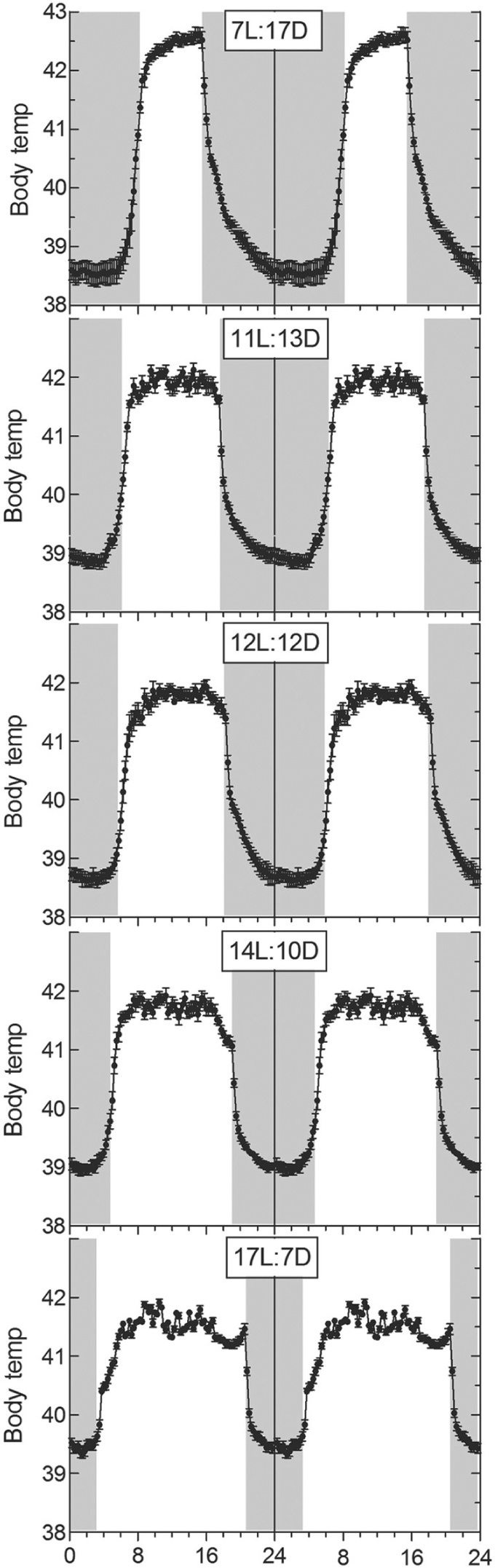
The diurnal cycle in body temperature (Tb) in starlings held on 5 different photoperiods: 7 h of light and 17 h of darkness each day (7L:17D) (
7L:17D
The lowest Tb was recorded 4 h before dawn. Values rose significantly (
11L:13D
The lowest Tb was recorded 3.5 h before dawn. Values rose significantly (
12L:12D
The lowest Tb was recorded 3 h before dawn. Values rose significantly during the hour before dawn (
14L:10D
The lowest Tb was recorded 3 h before dawn. Values rose significantly during the hour before dawn (
17L:7D
The lowest Tb was recorded 2 h before dawn. Values rose significantly during the hour before dawn (
Mean Tb over 24 h increased (
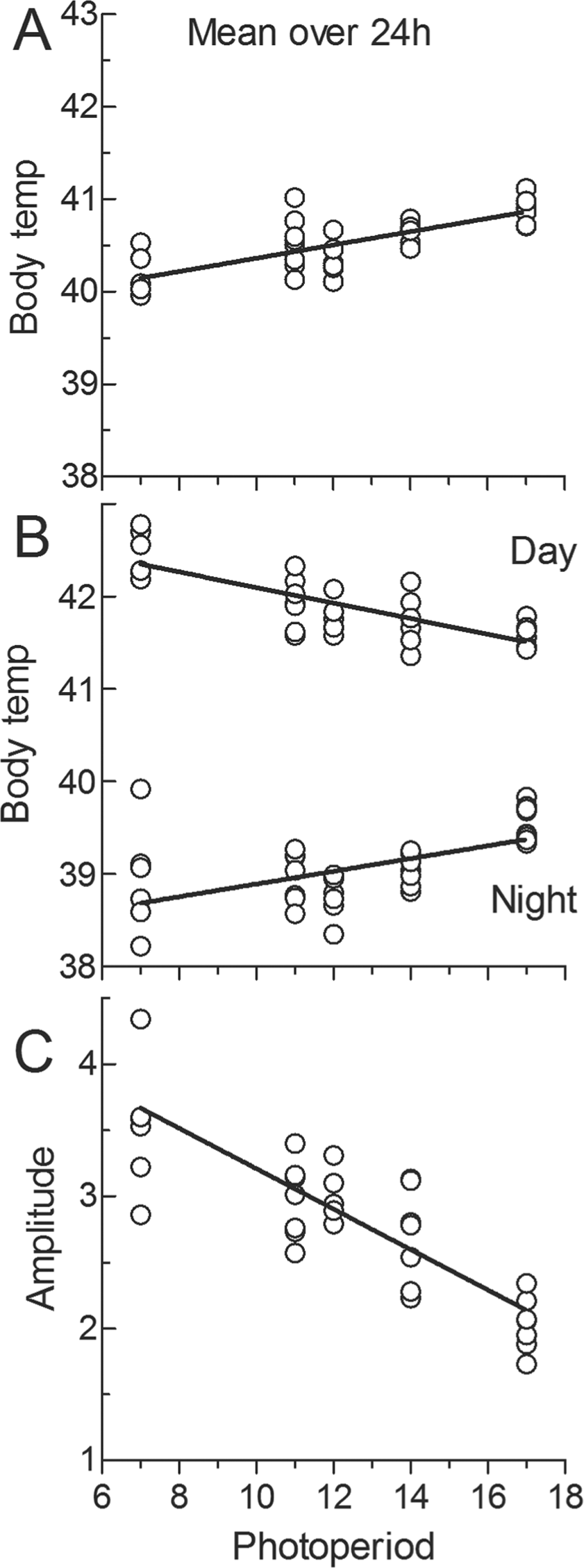
(A) Mean body temperature (Tb) over 24 h in starlings under constant photoperiods ranging from 7L:17D to 17L:7D. Each point represents an individual bird. (B) Mean daytime (0900-1500 h) Tb (upper points) and mean nighttime (2100-0300 h) Tb (lower points) for birds under different photoperiods. (C) The daily amplitude in Tb (i.e., the difference between daytime and nighttime Tb in B) for birds under different photoperiods. In all cases the lines are linear regressions.
Response to Change in Photoperiod
An Increase in Photoperiod from 7L:17D to 14L:10D
The daily Tb profile changed rapidly so that by day 3 it closely resembled that of birds fully adapted to 14L:10D (Fig. 3). On day 0 there was no increase in Tb before 5 h (the time of dawn on the following day), but between 5 h and dawn (8.5 h) Tb increased by 3.1 °C (
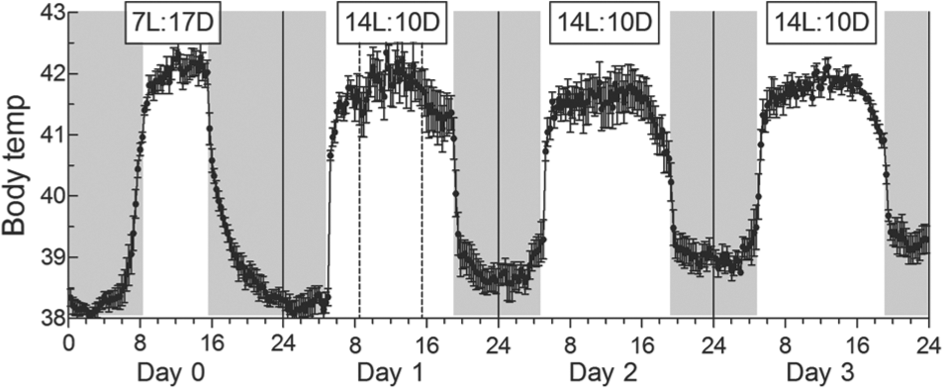
Change in the daily cycle of Tb following an increase in photoperiod from 7L:17D (day 0) to 14L:7D (days 1, 2, and 3). Periods of darkness are shown by shaded areas. Each point represents mean ± SE (
A Decrease in Photoperiod from 17L:7D to 7L:17D
Again, the daily Tb profile changed rapidly so that by day 3 it closely resembled that of birds fully adapted to 7L:17D (Fig. 4). On day 0, under 17L:7D, there was an increase in Tb of 0.5 °C before dawn (
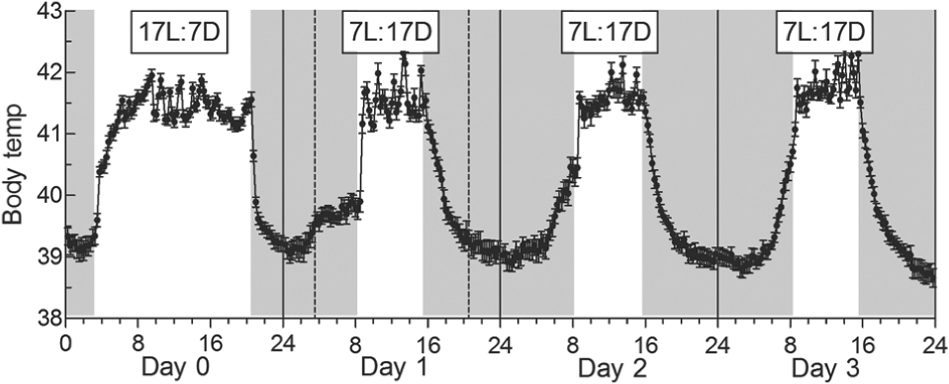
Change in the daily cycle of Tb following an decrease in photoperiod from 17L:7D (day 0) to 7L:17D (days 1, 2, and 3). Periods of darkness are shown by shaded areas. Each point represents mean ± SE (
17L:7D to DD and Then Back to 17L:7D
During the first 6 h of day 0, under 17L:7D, Tb increased by 1.9 °C (
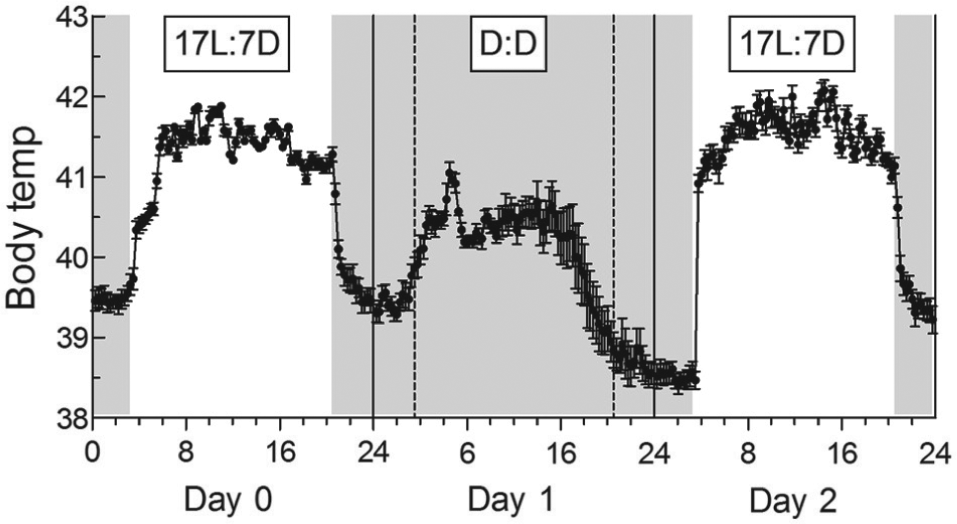
Change in the daily cycle of Tb following transfer from 17L:7D (day 0) to D:D (day 1) and then back to 17L:7D (day 2). Periods of darkness are shown by shaded areas. Each point represents mean ± SE (
Discussion
Mean Tb in starlings under different photoperiods was just over 40 °C, which is similar to that found in other bird species (Yahav, 2015). However, birds, as mammals, are heterothermic; Tb varies with environmental change. In this case, the data show that there is a dramatic daily cycle in body temperature in starlings. It has been known for some time that nighttime Tb in diurnal birds, as in mammals, is lower than daytime Tb (Prinzinger et al., 1991). The present data also show for the first time that the amplitude of the daily cycle was closely related to photoperiod. Under long photoperiods it was about 2 °C, but this increased to 3.5 °C under short photoperiods. Under short photoperiods, nighttime Tb was lower than under long photoperiods, and also daytime Tb was higher than under long photoperiods. Why is this? It is presumably an adaptive mechanism in that during long winter nights, environmental temperature is likely to be lower, and lowering Tb more will reduce energy loss. Hypothermia reduces energy expenditure and increases the chances of surviving winter (Waite, 1991; Maddocks and Geiser, 1997; Clark and Dukas, 2000), and birds with reduced food intake show a lower daily nadir in body temperature (Thouzeau et al., 1999). The greater decrease in starlings’ Tb was not caused by lower environmental temperature since temperature was kept constant during day-night and was the same for all photoperiods. Lower Tb under shorter photoperiods may be, at least in part, a consequence of the long night. Under all photoperiods, Tb began to decrease immediately after dusk. Under short photoperiods, there is a longer time for Tb to decrease further. In DD, Tb fell to lower values than at any other time. However, this passive effect cannot be the whole explanation since Tb actively increases before dawn.
The daily cycle of electrophysiological activity recorded in vitro in the SCN of mice shows a similar difference in amplitude with photoperiod (Van der Leest et al., 2009). The daily nadir of electrical activity was much lower in mice from a short photoperiod (8L:16D) than in mice from a long photoperiod (16L:8D). This may explain why the phase shifting effect of a light pulse is much greater in animals on shorter photoperiods.
Not only did the daily amplitude in Tb vary with photoperiod, but so did the pattern. Under all photoperiods, Tb increased before dawn. This was more pronounced under shorter photoperiods, where Tb had increased almost fully before dawn. The longer the photoperiod, the less Tb increased before dawn and the more it increased after dawn. Again, presumably this is adaptive. It is important that Tb increases before dawn so that birds can be active as soon as it gets light, and this is even more important when this is followed by a short day.
The data show the daily cycle in Tb in starlings is weakly circadian. Under all photoperiods, Tb began to increase before dawn, in the absence of external cues, and therefore must be circadian. In addition, in birds transferred from a long photoperiod to DD, Tb not only increased before the time that they had experienced dawn on the previous day but continued to increase for a time after that. However, when photoperiods were changed, light or darkness rapidly increased or decreased Tb, respectively, regardless of time in relation to the cycle in the previous day. For example, when birds were transferred from 7L:14D to 14L:10D, Tb increased immediately at the new earlier dawn and decreased rapidly at the new later dusk. Although these immediate responses may be masking, (i.e., an immediate response to environmental change overriding a circadian rhythm), birds did become fully entrained to new photoperiods by the third day. In birds transferred to DD, although there was an increase in Tb around the “missing” dawn, Tb decreased well before the subsequent “missing” dusk. Although there is evidence for circadian involvement at dawn, there is no evidence for circadian involvement at dusk. Thus, the daily cycle in Tb in starlings is only weakly circadian and appears to be entrained by dusk rather than dawn. Conversely, it is possible that dawn acts as the zeitgeber and that the sudden decrease in Tb at dusk under constant photoperiodic conditions is masking caused by dusk, but that would mean that masking occurs every day (i.e., not just in response to a novel environmental change). Had it been possible to keep the birds for longer than 1 day under DD, and had they shown circadian cycles in Tb, then the free-running period (tau) could have been assessed. Tau longer or shorter than 24 h would have a bearing on whether dawn or dusk acts as the zeitgeber.
Since birds have well-defined circadian cycles in activity under constant darkness (Cassone and Kumar, 2015) and Tb is higher when birds are active than resting (Prinzinger et al., 1991), it is likely that there would be persistent circadian cycles in Tb under constant darkness, and indeed this has been shown (Hudson and Kimzey, 1966; Binkley et al., 1971; Underwood, 1994). In mammals there is a close relationship between circadian cycles in activity and Tb such that in tau mutant-hamsters, which have a circadian period of 20 h, cycles in both Tb and activity are repeated every 20 h (Refinetti and Menaker, 1992). However, in starlings transferred from 17L:7D to 1 day of constant darkness, the pattern of Tb during darkness did not resemble that of the preceding day. Following a curtailed increase at the subjective dawn, Tb declined to lower values than seen in any other group. The decline started before the subjective dusk, and Tb did not start to increase before the following dawn as it had during the preceding LD cycle; values only increased after dawn. That is not to say that if these birds had been kept in continuous darkness for several days (they were not, for ethical reasons), a circadian cycle would not have established, but it does suggest that such a cycle would not have been similar to a cycle under LD. However, birds in this study under DD were in total darkness as opposed to constant dim light, and this may have prevented the expression of a circadian pattern of activity as well as of Tb. This would still be evidence that the daily cycle in Tb is only weakly circadian.
The daily cycle in melatonin has been implicated in the control of cycles in Tb (Binkley et al., 1971; Oshima et al., 1989; Underwood, 1994). Daily melatonin cycles in young starlings do not develop fully until after they become homeothermic (Dawson and Van’t Hof, 2002). However, melatonin rhythms in adult starlings are highly circadian and maintain the profile of the previous day when birds are transferred to DD (Dawson and King, 1994), unlike cycles in Tb. Moreover, under LD cycles, dusk always induced an immediate and rapid decrease in Tb irrespective of photoperiod or recent change in photoperiod. This has not been shown before. Similarly, solar eclipses are noted for causing a rapid decrease in activity among birds. The rapidity suggests a direct involvement of photoreceptors rather than an effect mediated by melatonin. Furthermore, many diurnal birds often migrate at night and presumably maintain high Tb. During the migratory seasons, caged warblers develop intense nocturnal activity yet the daily pattern of melatonin changes little from the nonmigratory nocturnally inactive period (Gwinner et al., 1993). Barn owls (
In conclusion, the daily cycle in Tb is closely related to photoperiod, the amplitude is greater under shorter photoperiods, and birds rapidly entrain to a new photoperiod. The results suggest that the daily cycle in Tb is weakly circadian and is entrained by dusk rather than dawn. Further studies will use this information to assess the involvement of energetics (food availability and ambient temperature) on photoperiodic responses.
Footnotes
Acknowledgements
This work was supported by the Natural Environment Research Council.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
