Abstract
Female Drosophila melanogaster, like many other organisms, exhibit different behavioral repertoires after mating with a male. These postmating responses (PMRs) include increased egg production and laying, increased rejection behavior (avoiding further male advances), decreased longevity, altered gustation and decreased sleep. Sex Peptide (SP), a protein transferred from the male during copulation, is largely responsible for many of these behavioral responses, and acts through a specific circuit to induce rejection behavior and alter dietary preference. However, less is known about the mechanisms and neurons that influence sleep in mated females. In this study, we investigated postmating changes in female sleep across strains and ages and on different media, and report that these changes are robust and relatively consistent under a variety of conditions. We find that female sleep is reduced by male-derived SP acting through the canonical sex peptide receptor (SPR) within the same neurons responsible for altering other PMRs. This circuit includes the SPSN-SAG neurons, whose silencing by DREADD induces postmating behaviors including sleep. Our data are consistent with the idea that mating status is communicated to the central brain through a common circuit that diverges in higher brain centers to modify a collection of postmating sensorimotor processes.
Across many insect species, mating alters a variety of behaviors and physiological responses that can influence an organism’s fitness. For example, in female Drosophila melanogaster, copulation induces rejection behavior (Manning, 1967), a decrease in longevity (Chapman et al., 1995; Fowler and Partridge, 1989), increased egg production and laying (Heifetz et al., 2000; Herndon and Wolfner, 1995), alterations in sleep (Isaac et al., 2010), and changes in dietary intake and preference (Carvalho et al., 2006; Walker et al., 2015). Although many of these phenomena are well-established, fundamental questions remain about their molecular and circuit-level underpinnings. For example, what are the sensory cues that stimulate changes in distinct behaviors? What neurons are responsible for receiving and communicating these signals? Do common neuronal circuits modulate multiple behaviors, and if so, where and how do these signals diverge to ultimately influence distinct motor outputs?
Some of the peripheral and central mechanisms triggering post-mating behavioral switches have recently emerged (Feng et al., 2014; Haussmann et al., 2013; Rezával et al., 2012; Rezaval et al., 2014; Soller et al., 2006). For example, it is known that Sex Peptide (SP), transferred via male-derived seminal fluid during copulation, is critical for changes in several behaviors (Aigaki et al., 1991; Feng et al., 2014; Gillott et al., 2003; Rezával et al., 2012; Walker et al., 2015); however, other ejaculatory peptides also play minor roles (Heifetz et al., 2000; Herndon and Wolfner, 1995; Rexhepaj et al., 2002). SP induces changes in rejection behavior and dietary preference by silencing the neuronal activity of a group of SP-sensitive sensory neurons (SPSN) in the female reproductive tract (RT) that project to the Abdominal Ganglion (Abg) (Feng et al., 2014; Walker et al., 2015). From the Abg, a majority of this mating information is transmitted to higher brain centers via the Sex Peptide Abdominal Ganglion (SAG) neurons, which appear to be silenced upon receipt of SP (Feng et al., 2014; Walker et al., 2015). While the SPSNs and SAG neurons play a large role in communicating mating status, other neurons and mechanisms also contribute to these and other postmating responses (Haussmann et al., 2013; Rezával et al., 2012, 2014).
Genetic and neural principles underlying sleep-wake regulation are conserved between Drosophila and mammals (Sehgal and Mignot, 2011), underscoring the significance of addressing basic mechanisms in flies. The decrease in sleep in female flies after mating (Isaac et al., 2010) provides a model to investigate the basis of environment-induced plasticity in sleep. Similar to other postmating responses, sleep appears to be modulated mainly by SP (Isaac et al., 2010). However, it is unknown whether SP is received through the same SPSNs within the RT and if it this signal is transmitted via the same second-order neurons to change sleep. Here, we first provide an in-depth characterization of the postmating sleep response in female flies and demonstrate that decreases in sleep after copulation occur in many strains and under different conditions. We then confirm that SP is largely responsible for inducing this decrease, and demonstrate that the canonical sex peptide receptor (SPR) is necessary and acts in specific neurons to alter postmating sleep. Using a pharmacogenetic approach (Becnel et al., 2013), we establish that silencing the “canonical” SPSN-SAG postmating circuit is sufficient to decrease female sleep. Finally, experiments ablating the SPSN neurons suggest mechanistic differences in some PMRs. Together, these data support the idea that postmating changes in sleep are driven through male-derived SP acting on the SPR within a common subset of sensory neurons in the female RT. This information is then communicated via second-order interneurons and subsequently uncoupled in higher-order brain regions to affect various sensorimotor systems.
Materials and Methods
Fly Maintenance and Stocks
Flies were raised and maintained on a standard cornmeal-based diet and reared in approximately 12:12-h light-dark (LD) conditions at 40-60% humidity. Except when noted, flies were entrained to strict 12:12 LD conditions for at least 2-3 days prior to the start of each experiment. The following fly stocks were used: In Fig. 1A-D, iso31, w1118, OregonR (OreR), Canton-S (CS, White Berlin (WB)); In Fig. 2E, spermless males were obtained by crossing ProtB males to tud1 homozygous mothers; male offspring from this cross lack sperm. In Fig. 3A, ProtB (GFP:sperm) flies were a gift from Scott Pitnick (Manier et al., 2010) and the DTA-E flies were a gift from Mariana Wolfner (Kalb et al., 1993). In Fig. 3B, the UAS-Kir2.1 line was used (Baines et al., 2001) and the corazonin-Gal4 (crz-Gal4; second chromosome) driver was obtained from Bloomington [stock# 51976; (Tayler et al., 2012)]. In Fig. 4A, sp0 flies were a gift from the laboratories of Barry Dickson and Irene Miguel-Allaga; the spΔ130 deficiency line was a gift from Irene Miguel-Allaga and Mariana Wolfner. Allelic combinations using stocks from each individual laboratory yielded similar results. In Fig. 4B, the spr deficiency line was obtained from Bloomington (stock #7708) and the second and third chromosomes were replaced with isogenized chromosomes from iso31. In Fig. 5, the spr-RNAi lines were a gift from Barry Dickson (Yapici et al., 2008) and were outcrossed to iso31 four times before building them into the isogenized spr mutant background. The VT3280-Gal4 line was a gift from Barry Dickson and ppk-Gal4 line was a gift from Wes Grueber (Grueber et al., 2007). Each driver was outcrossed twice to iso31 before being built into the isogenized spr mutant background. In Fig. 6, the UAS-SPR line was a gift from Barry Dickson (Yapici et al., 2008) and was outcrossed to iso31 four times before building it into the isogenized spr mutant background. In Fig. 7, an isogenized version of UAS-Kir2.1 was used in combination with the VT3280-Gal4 driver, as in Fig. 5. The split Gal4 driver, SAG-1, was a gift from Barry Dickson (Feng et al., 2014). In Fig. 8, the DREADD flies were a gift from Charles Nichols (Becnel et al., 2013). In Fig. 9, UAS-Reaper was used as reported by others (Cavanaugh et al., 2014), and UAS-NachBac was isogenized into the iso31 background.
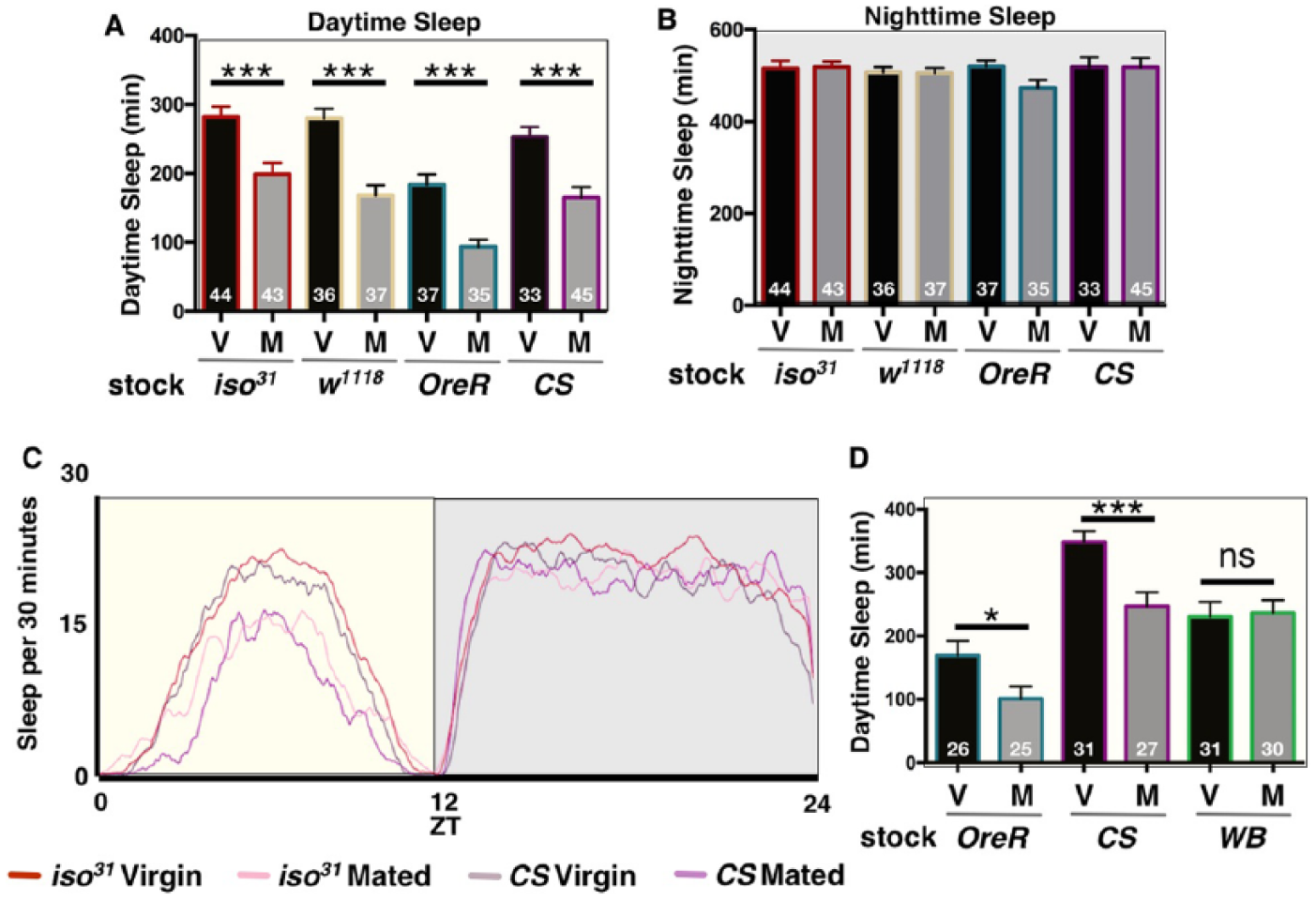
Postmating decreases in female sleep are largely independent of genetic background. (A) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) female flies of the indicated fly stock. (B) Total nighttime sleep (ZT12-24) for virgin (V) and mated (M) female flies of the indicated fly stock. Note, no significant differences in nighttime sleep were detected after mating. (C) Sleep is plotted as a function of time across the first full day of the experiment (ZT0-24) for the indicated fly stocks and mating status. For simplicity, indicated genotypes were used as representative examples. (D) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) female flies of the indicated fly stocks. White Berlin (WB) was the only line that did not exhibit postmating changes in sleep. Total experimental size is indicated within each bar of the graph. ***p ≤ 0.0001; *p < 0.01.
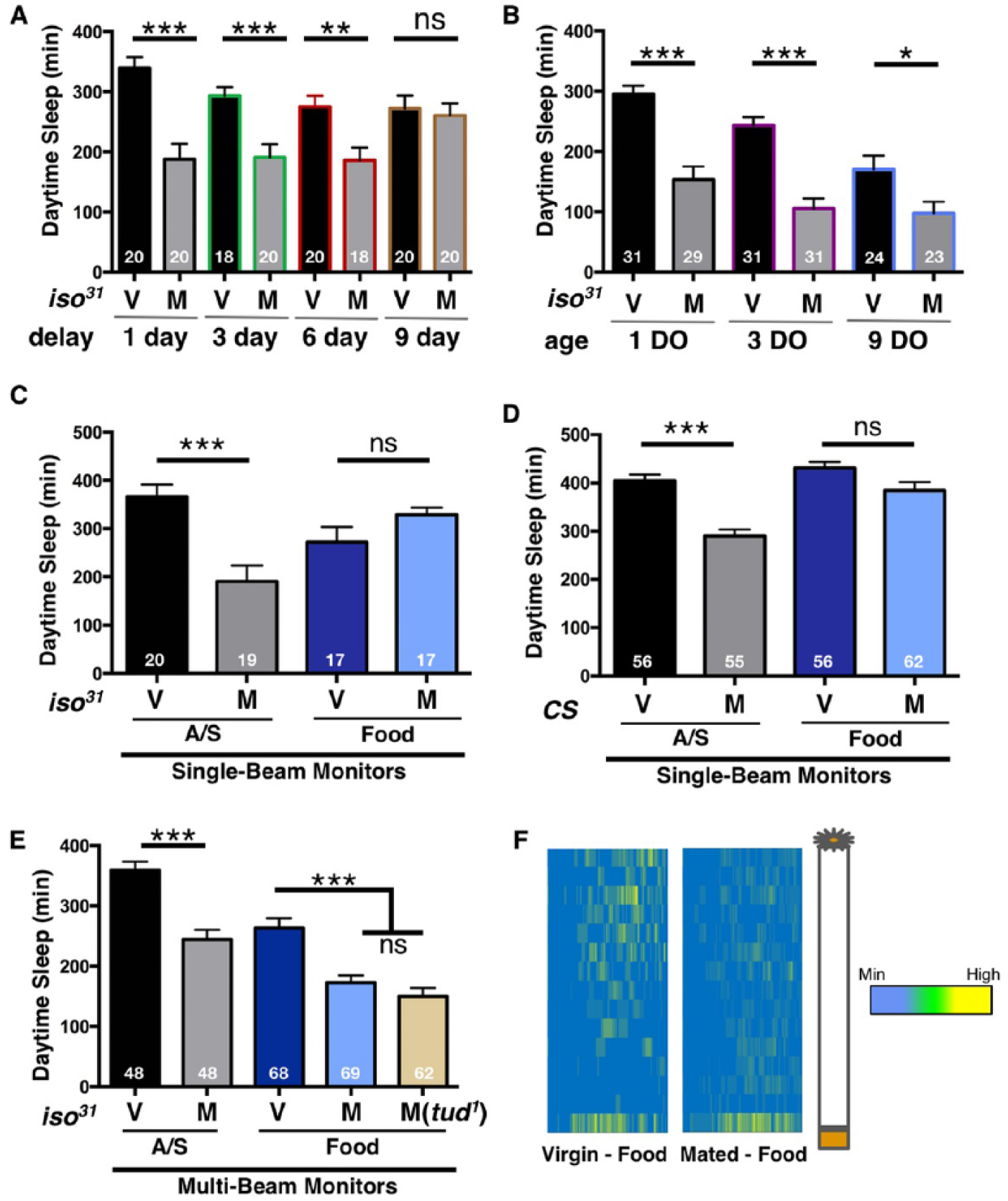
Postmating decreases in female sleep are long lasting and independent of age and nutrient availability. (A) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) iso31 female flies. Sleep experiments were performed following the indicated time delay after copulation. (B) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) iso31 female flies of the indicated age (DO, days old). (C) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) iso31 female flies loaded into DAM tubes containing the indicated nutrient source and detected using the traditional single-beam infrared monitoring system. (D) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) CS female flies loaded into DAM tubes containing the indicated nutrient source and detected using the traditional single-beam infrared monitoring system. (E) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) iso31 female flies loaded into DAM tubes containing the indicated nutrient source and detected using the multibeam (MB) infrared monitoring system. Notice that the MB system is able to detect postmating decreases in sleep while the single beam does not. Total experimental size is indicated within each bar of the graph. A/S = 5% agarose containing 1% sucrose. Food = standard yeast-containing laboratory food. tud1 males do not generate sperm and thus do not produce fertilized eggs in the mated females. (F) Sleep heat maps illustrating average time spent sleeping in each individual beam across the first full day (ZT0-12) after mating. Each row represents sleep data from an individual infrared beam and each column designates sleep. The color scale reflects the average time flies spent sleeping in each beam. To the right of the plots is a schematic demonstrating the orientation of the locomotor tube, including the nutrient source at the bottom. ***p ≤ 0.0001; **p ≤ 0.001; ns, not significant.
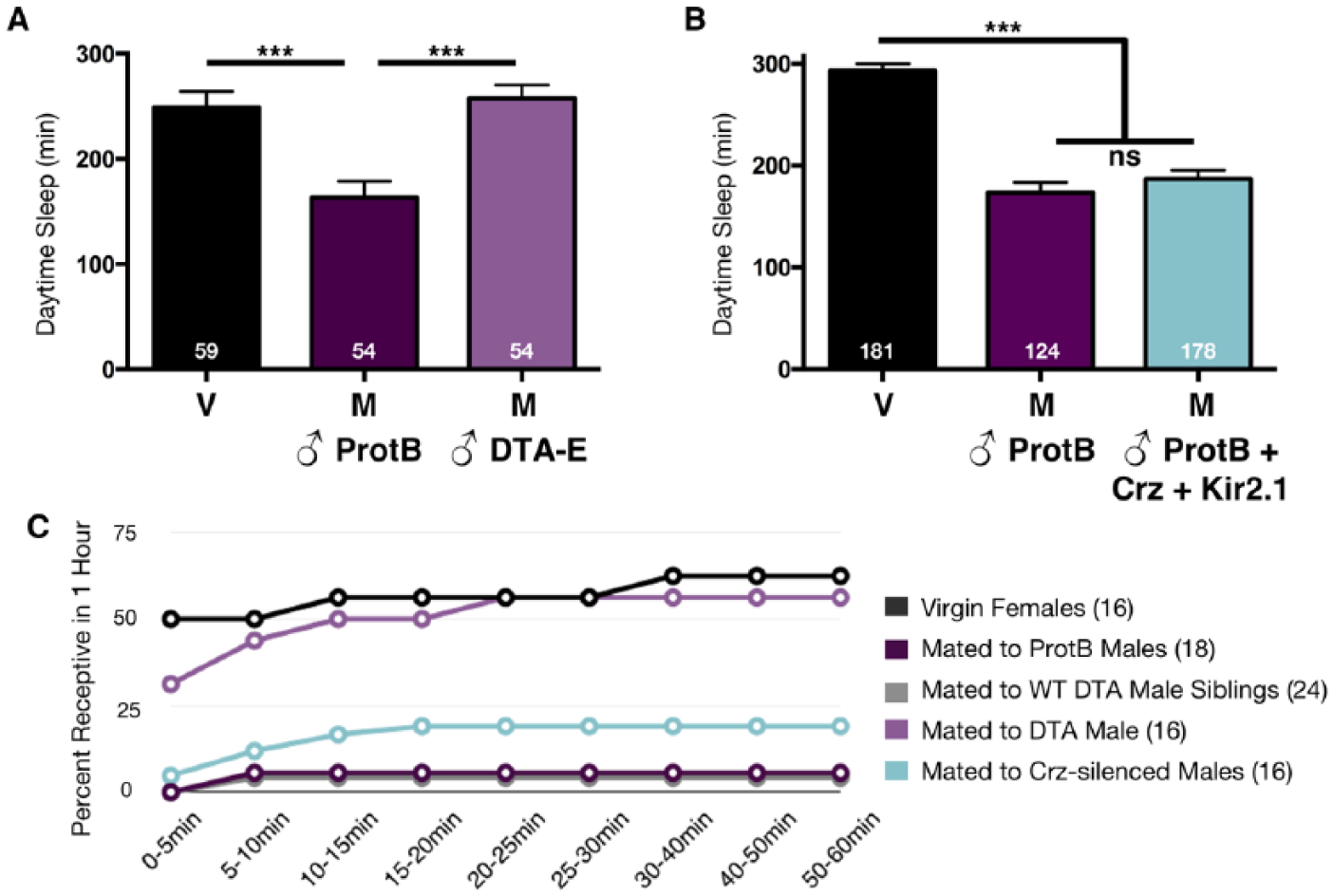
The main cells of the accessory gland are essential for inducing postmating changes in female sleep. (A) Total daytime sleep (ZT0-12) for virgin (V) flies or females mated (M) to the indicated male genotype. DTA-E males fail to produce seminal fluids derived from the main cells of the accessory gland and do not induce postmating decreases in female sleep. (B) Total daytime sleep (ZT0-12) for virgin (V) flies or females mated (M) to the indicated male genotype. Corazonin (crz)-silenced males do not transfer sperm during copulation, as evidenced by the lack of GFP-sperm in the female reproductive tract [data not shown; (Tayler et al., 2012)]. Interestingly, these males are still able to induce postmating decreases in female sleep, suggesting some seminal fluids are transferred to the female. Total experimental size is indicated within each bar of the graph. ***p ≤ 0.0001. (C) Data represent the percentage of females, who after 24 h of initially copulating with the indicated male genotype, were able to re-mate with a naïve male within 1 h. Accessory gland ablated males (DTA) did not induce rejection behavior and remained as receptive as virgin females, while crz-silenced males induced postmating decreases in receptivity similar to ProtB and DTA-E sibling males. Total numbers of females tested are listed in parentheses.
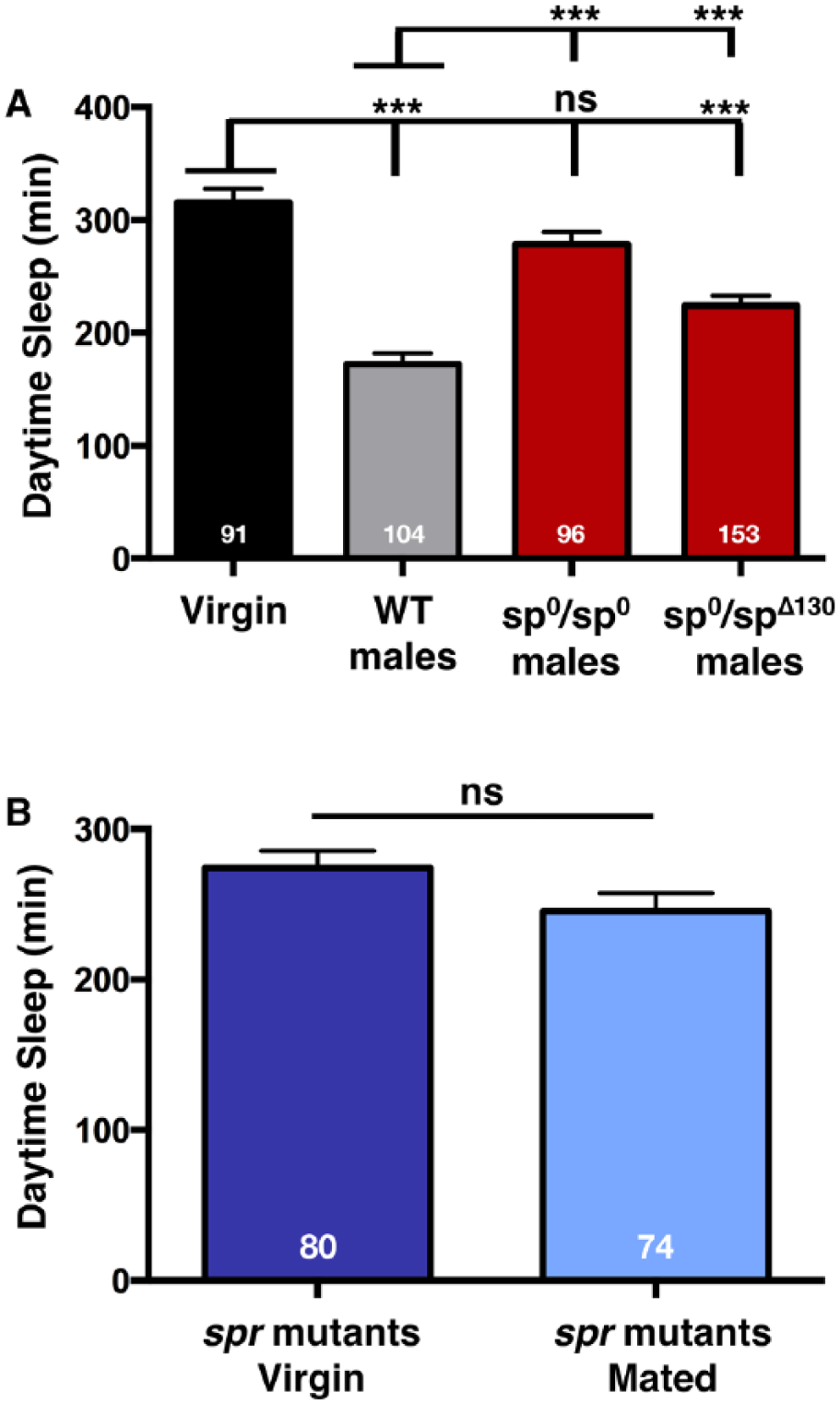
Sex peptide and its receptor are required to induce postmating decreases in sleep. (A) Total daytime sleep (ZT0-12) for virgin (V) flies or females mated (M) to the indicated male genotype. Note, the significantly different effect on postmating sleep between the homoallelic and heteroallelic sp mutant males. Mating was confirmed in all experimental flies by the presence of GFP-sperm in the female abdomen. (B) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) spr female flies. Total experimental size is indicated within each bar of the graph. *** p ≤ 0.0001; ns, not significant. See also Suppl. Fig. S3.
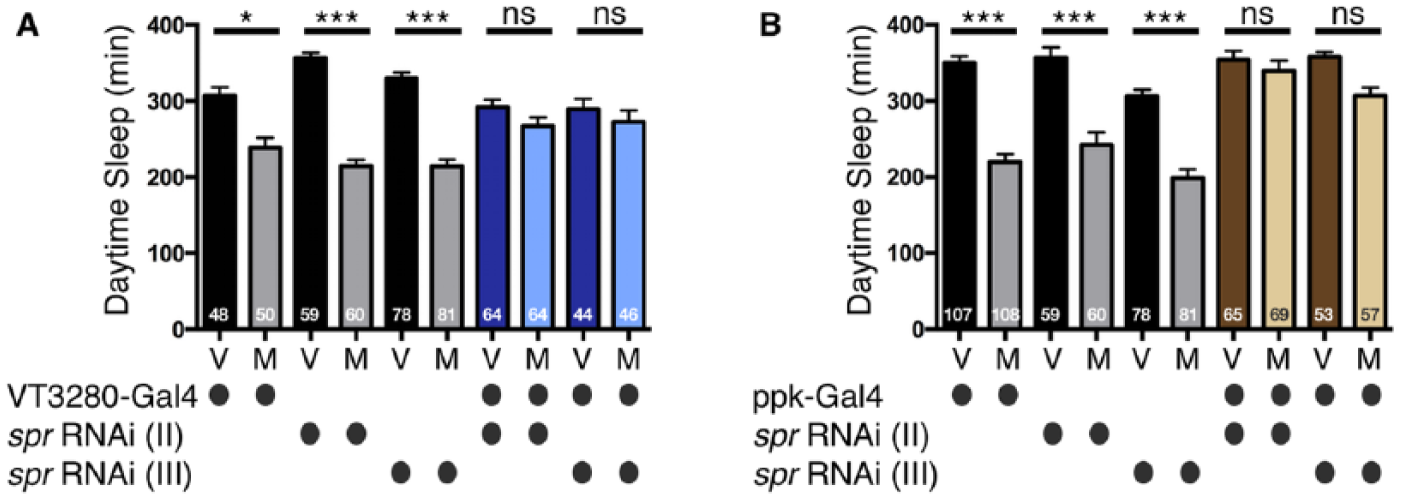
Sex peptide receptor (SPR) is necessary in sensory neurons of the female reproductive tract to alter postmating sleep. (A) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) female flies of the indicated genotypes. Note, females with reduced SPR expression in VT3280 neurons are unable to decrease sleep after mating. (B) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) female flies of the indicated genotypes. Note, females with reduced SPR expression in ppk neurons are unable to decrease sleep after mating. Values are combined from at least two separate experiments and total experimental size is indicated within each bar of the graph ***p ≤ 0.0001; *p ≤ 0.01; ns, not significant.
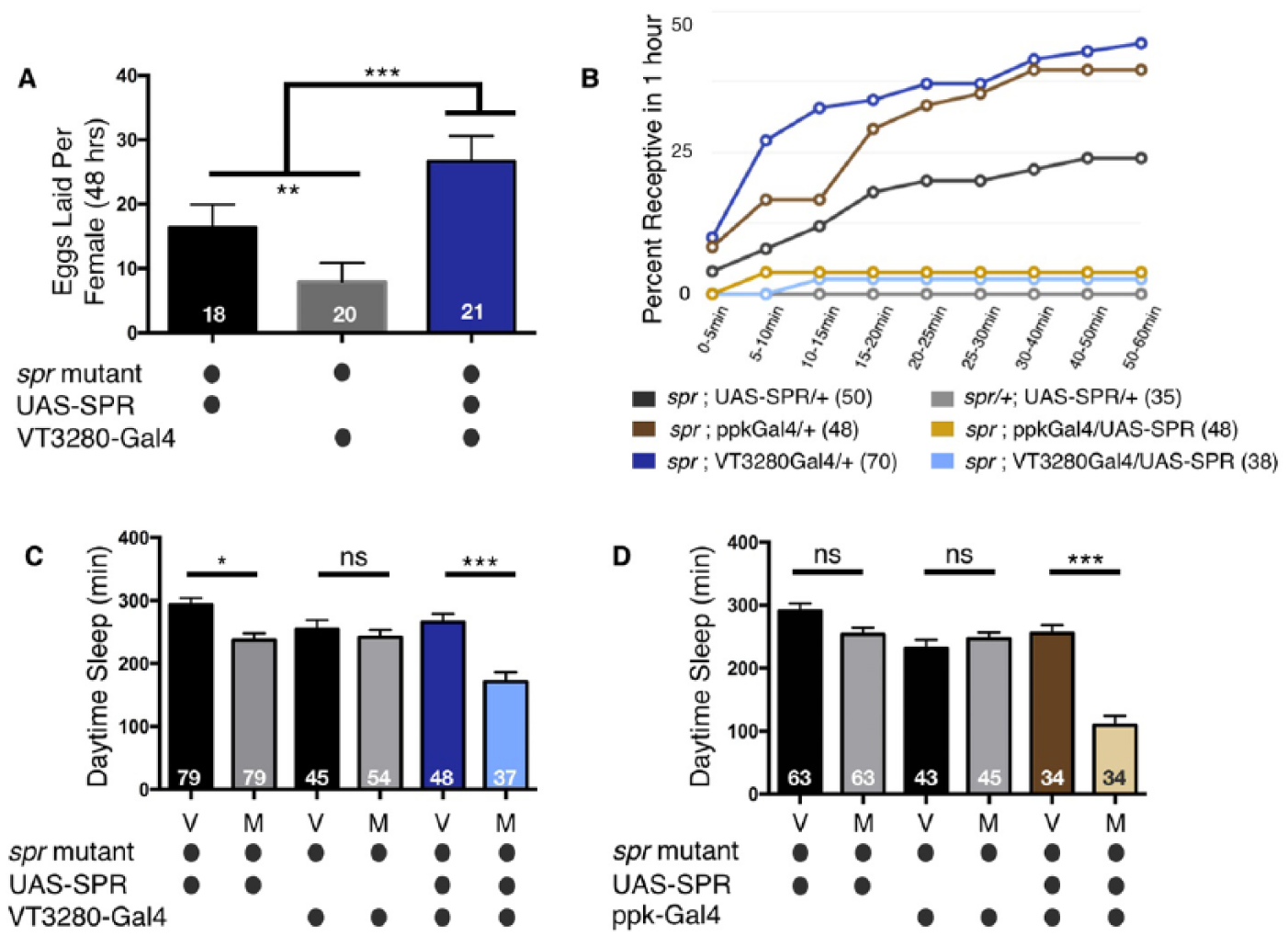
Sex peptide receptor (SPR) in sensory neurons of the female reproductive tract is sufficient to alter postmating sleep. (A) Specifically re-expressing SPR in VT3280 neurons is able to rescue egg laying defects observed in an otherwise spr mutant background. (B) Specifically re-expressing SPR in VT3280 or ppk neurons is able to restore appropriate rejection behaviors in an otherwise spr mutant background. Data represents the percentage of mated females that were able to re-mate with a naive wild type male 24 h after the initial copulation event. While spr mutant animals re-mate within 1 h (dark grey, brown, and dark blue lines), SPR expression in ppk or VT3280 neurons (light yellow and light blue lines respectively) elicits appropriate postmating rejection behaviors similar to mated control heterozygous animals (grey line). Total numbers of females tested are listed in parentheses. (C) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) female flies of the indicated genotypes. Normal postmating decreases in sleep are observed when SPR expression is restored specifically in VT3280 neurons. Note, in panels A-C, spr mutants containing the UAS-SPR transgene alone exhibit small but significant postmating responses in the three independent assays, suggesting the possibility of low-level leaky expression. (D) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) female flies of the indicated genotypes. Note, normal postmating decreases in sleep are observed when SPR expression is restored specifically in ppk neurons. Similar to panel C, the spr mutant containing the UAS-SPR transgene alone trends toward slightly lower postmating sleep; however, this decrease was not significant. In panels C and D, values are combined from three separate experiments and total experimental size is indicated within each bar of the graph. ***p < 0.0001; **p < 0.001; *p < 0.01; ns, not significant.
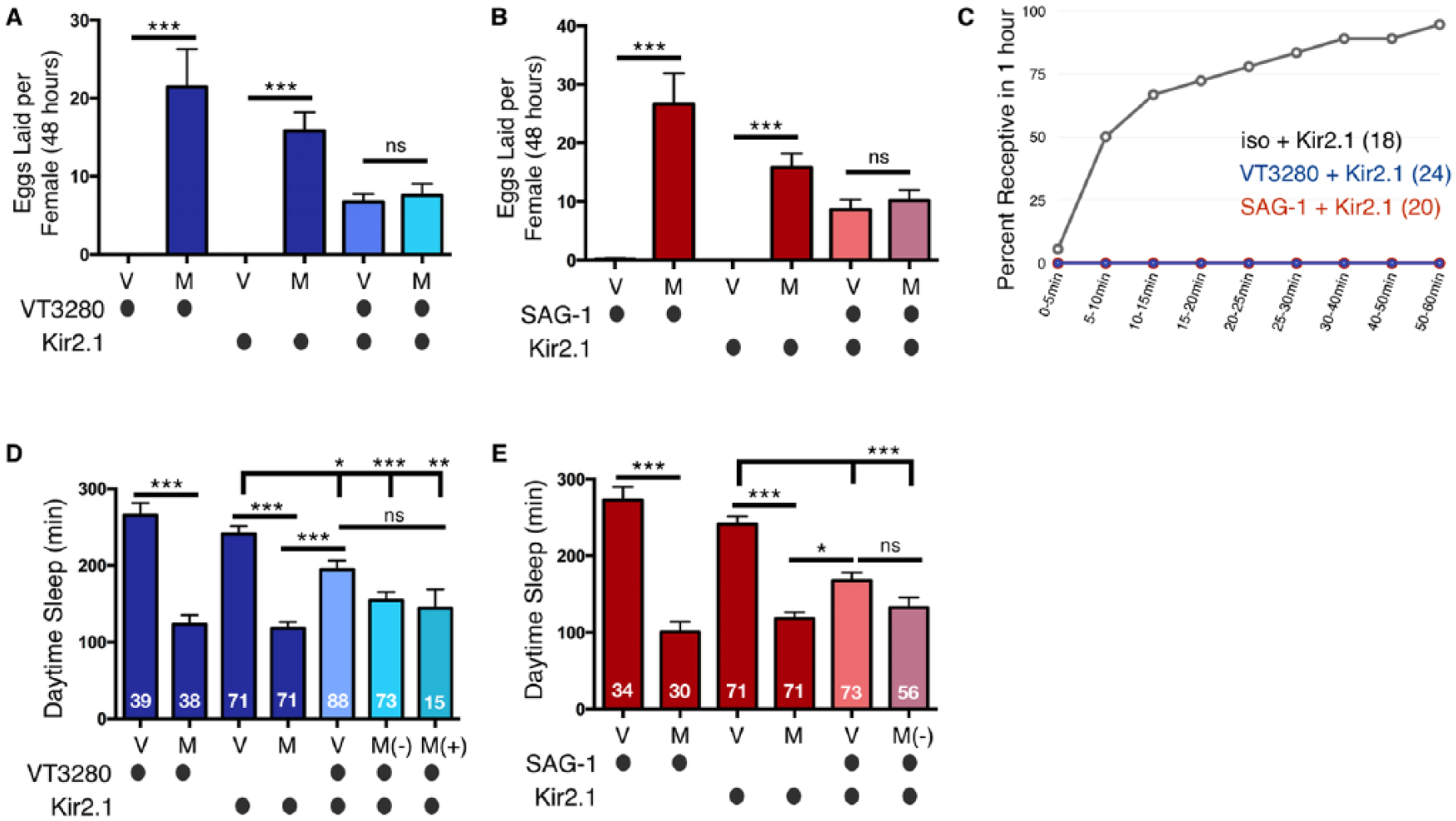
Chronically silencing the SPSN-SAG postmating circuit decreases female daytime sleep. (A) Chronically silencing VT3280 neurons with Kir2.1 is able to partially induce egg laying in virgin females; however, egg laying is lower than that in mated control flies. (B) Chronically silencing SAG-1 neurons with Kir2.1 is able to partially induce egg laying in virgin females; however, similar to panel A, egg laying is lower than that observed in mated control flies. In panels A and B, mating does not significantly increase egg laying of VT3280-silenced females, most likely because these animals are better able to reject courtship advances from male files (see panel C). (C) Chronically silencing VT3280 or SAG-1 neurons with Kir2.1 induces rejection behaviors in virgin females. After 1 h, all Kir2.1-silenced virgins are unreceptive to male courtship (note, the red and blue lines overlap at 0%), while wild type virgin controls (grey line) show normal levels of copulation. Total numbers of females tested of each genotype are listed in parentheses. (D) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) female flies of the indicated genotypes. A significant decrease in daytime sleep is observed in virgin flies when VT3280 neurons are silenced with Kir2.1; however, we note that sleep levels are still significantly higher than that seen in mated controls. (E) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) female flies of the indicated genotypes. A significant decrease in daytime sleep is observed in virgin flies when SAG-1 neurons are silenced with Kir2.1; however, as in panel D, sleep levels are still significantly higher than that in mated controls. (Panels D and E) M(-) = paired with a male but did not mate, as evidenced by a lack of GFP-sperm transfer; M(+) = GFP-sperm was transferred into female abdomen. Pairing with male flies in the absence of mating, M(-), does not significantly decrease sleep further from virgins who have had their neurons silenced. Note, in panel D, in contrast to the copulation assay in panel C, after being paired with male flies overnight, some of the VT3280-silenced females were able to mate; in panel E, none of the SAG-1-silenced females mated. Values are combined from three separate experiments and total experimental size is indicated within each bar of the graph. ***p < 0.0001; **p < 0.001; *p < 0.01; ns, not significant.
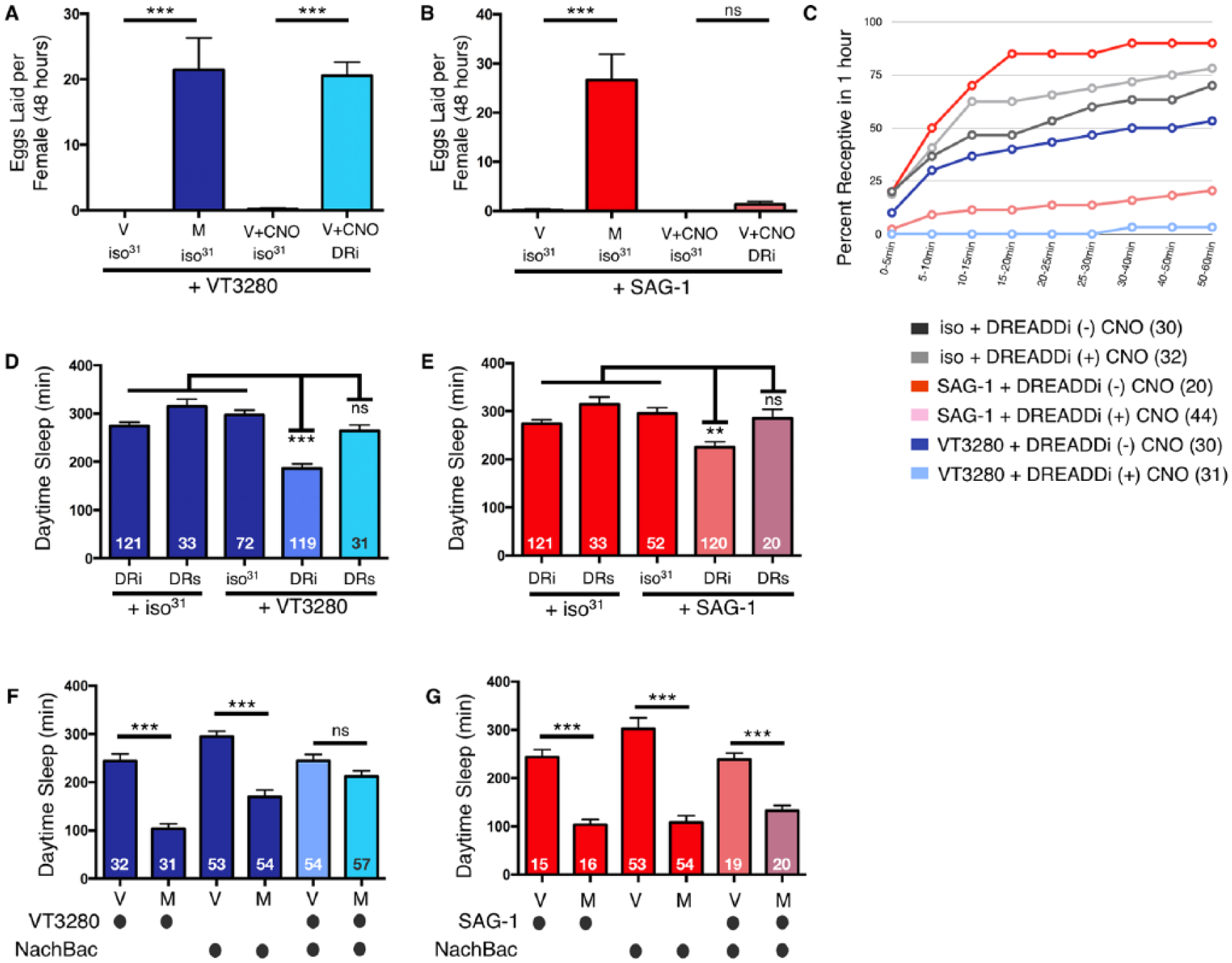
Acutely silencing the SPSN-SAG postmating circuit decreases daytime female sleep. (A) Acutely silencing VT3280 neurons by stimulating the DREADDi receptor (DRi) with 0.5 mM clozapine-N-oxide (CNO) induces egg laying in virgin (V) females comparable to that of mated female controls (M). (B) Acutely silencing SAG-1 neurons by stimulating the DRi receptor with CNO does not induce egg laying in virgin females as compared with mated female controls. Virgin (V) and mated (M) flies of the indicated genotypes. (C) Acutely silencing VT3280 or SAG-1 neurons with DRi [(+) CNO; light blue and pink lines, respectively] causes decreases in receptivity of virgin females within 1 h as compared with the same flies without DRi activation [(-) CNO; dark blue and red lines, respectively]. Control flies show comparable levels of receptivity in the absence or presence of CNO (dark and light and gray lines, respectively). Total numbers of females tested of each genotype are listed in parentheses. (D) Total daytime sleep (ZT0-12) for female flies of the indicated genotypes in the presence of CNO. A significant decrease in daytime sleep is observed in virgin flies when VT3280 neurons are silenced with DRi but not when activated by DREADDs (DRs). (E) Total daytime sleep (ZT0-12) for female flies of the indicated genotypes in the presence of CNO. A significant decrease in daytime sleep is observed in virgin flies when SAG-1 neurons are silenced with DRi but not when activated by DRs. In panels D and E, values are combined from three separate experiments (except for DRs, which was run twice) and total experimental size is indicated within each bar of the graph. (F) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) female flies of the indicated genotypes. Activation of VT3280 neurons with NachBac blocks postmating decreases in daytime sleep. (G) Total daytime sleep (ZT0-12) for virgin (V) and mated (M) female flies of the indicated genotypes. Note, activation of SAG-1 neurons with NachBac does not block postmating decreases in daytime sleep. In panels F and G, values are combined from two separate experiments and total experimental size is indicated within each bar of the graph. ***p < 0.0001; **p < 0.001; ns, not significant.
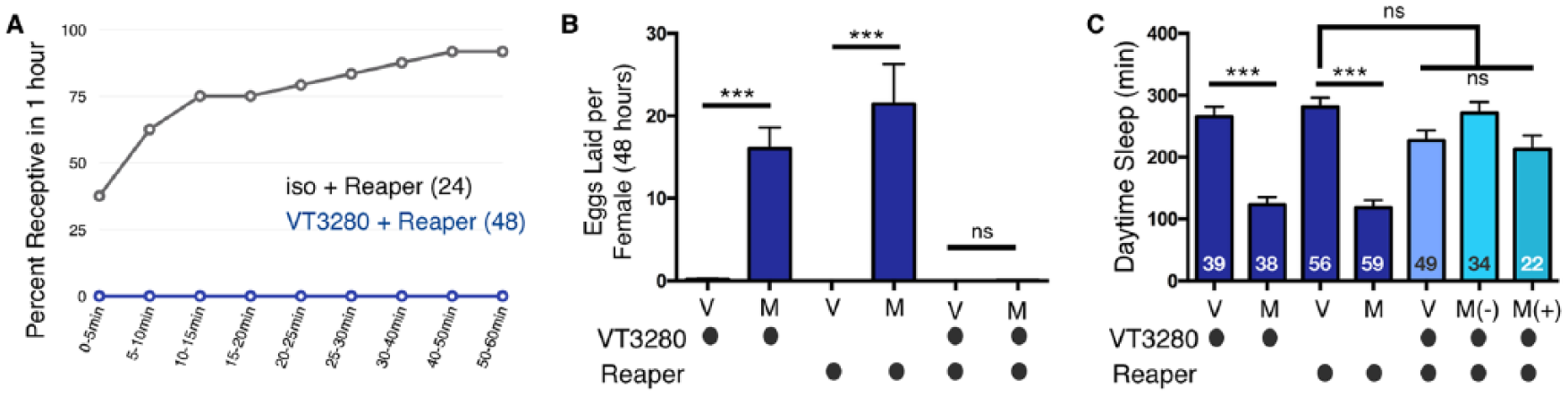
Ablation of VT3280 neurons induces rejection but not egg laying or decreases in daytime sleep. (A) Ablation of VT3280 neurons with Reaper induces rejection behavior in virgin females within 1 h as compared with control flies. (B) Ablation of VT3280 neurons with Reaper abolishes mating-induced egg laying, presumably because the flies are no longer able to sense the SP transferred from the male. Virgin (V) and mated (M) flies of the indicated genotypes. (C) Ablation of VT3280 neurons with Reaper inhibits mating-induced decreases in daytime sleep, presumably because the flies are no longer able to sense the SP transferred from the male. M(-), paired with a male but did not mate, as evidenced by lack of GFP-sperm transfer; M(+), GFP-sperm was transferred into female abdomen. Note, in panel C, in contrast to the copulation assay in panel A, after being paired with male flies overnight, some of the VT3280-ablated females were able to mate; however, this still did not lead to decreases in daytime sleep, most likely because the flies are no longer able to sense the SP transferred from the male. Values are combined from two separate experiments and total experimental size is indicated within each bar of the graph. ***p < 0.0001; ns, not significant.
Behavioral Assays
Standard Postmating Sleep Assays
For experiments performed at the University of Pennsylvania, flies were collected and entrained for 2-3 days within temperature- and light-controlled incubators (Percival; I22VL or I36VL; 12-h light-dark, 25 °C, ~40-60% humidity) prior to the start of each experiment. The day before the experiment, 3- to 5-day-old virgin female flies were either group-housed (virgin) or mated overnight with ProtB males (mated) containing GFP:sperm. To control for fly density, virgin vials contained twice as many females as the mated vials. Mated vials contained half the number of females plus an equal number of males to even out the total fly population per vial in both groups. The following day, flies were anesthetized using CO2, loaded into Drosophila Activity Monitors (DAM5) within 5 × 65mm glass pyrex tubes (Trikinetics, Waltham, MA) containing 5% sucrose in 1% agar (A/S), and put into incubators under the same environmental conditions. At the end of most experiments, female abdomens were dissected and analyzed for GFP-sperm as a marker of successful copulation. We note that ProtB males elicited the same postmating effects on sleep as wild type iso31 males (see Figure 4A). Postmating sleep was analyzed on the first full day of LD, starting at ZT0, only from females that had GFP-sperm present in their abdomens. Post-acquisition sleep analysis was mainly accomplished using Pysolo (Gilestro and Cirelli, 2009). For experiments performed at Swarthmore College, sleep analysis was performed using SCAMP (Donelson et al., 2012), and data from postmating days 2-4 were averaged together (Fig. 1D, 2D, and Suppl. Fig. S3).
Modifications to Standard Sleep Assay
Modifications were performed for different experiments. In Fig. 2A, after an overnight mating period, male flies were removed and females were group-housed for the designated time periods before running the sleep experiment. In Fig. 2B, female flies were aged for the designated time periods before running in a standard sleep assay. In Fig. 2C and D, iso31 was loaded in DAM tubes containing A/S or standard yeast-containing fly food while CS flies were loaded into DAM tubes containing A/S or cornmeal/dextrose food. In Fig. 8, the day before the experiment, 3-5 day old virgin females of the designated genotypes were group-housed in vials containing standard fly food plus 0.5 mM clozapine-N-oxide (CNO) (#4936, Tocris Bioscience, Bristol, UK; dissolved in water). Treated flies were subsequently loaded into DAM tubes containing A/S plus 0.5 mM CNO for the duration of the experiment. In Suppl. Fig. S1, naïve (isolated), group-housed, or mated male flies were run in the experiment; in Suppl. Fig. S2, Pysolo was used to determine the bout length and bout number for each experiment.
Multibeam Analysis
In Fig. 2, MB5-MB Drosophila Activity Monitors (Trikinetics, Waltham, MA), and 5 × 80-mm glass pyrex tubes containing either A/S or normal yeast-containing food were used when performing multibeam recordings. Sleep was determined as immobility bouts lasting ≥5 min using the “Movement” detection setting (Garbe et al., 2015). Post-acquisition sleep analysis was done using custom Excel macros and formulas that calculate sleep duration and architecture across 30-min time bins (Garbe et al., 2015). Sleep heat maps were generated by averaging the sleep time in each of the 17 beams for all flies on a minute-by-minute basis, and these average sleep times were plotted using three color scale conditional formatting in Excel. The color scale reflects the average time spent asleep in each beam; yellow designates high sleep and blue indicates that flies spent no time sleeping in the beam (Cavanaugh et al., 2016).
Egg-Laying Assays
Groups of virgin females, aged 3-5 days, were placed in vials (n = 3-8 females per vial) containing normal food and mated with an equal number of wild type males. For the next three days, flies were flipped approximately every 24 h into a new vial and eggs laid from the previous day were counted manually. The average number of eggs per fly was determined for each vial (total number of eggs / total number of flies). These values were then averaged across all vials for an individual genotype within an experiment. Each genotype was tested 2-4 times and data was combined to make the graph. Statistics and significance were calculated in GraphPad based on the number of groups analyzed across all experiments.
Copulation Assays
Males and females were collected after eclosion and group-housed separately for 3-5 days. For each assay, a single pair of male and female flies were introduced to an individual well of a spot plate (Cole Palmer, EW-17824-04) via aspiration and covered with cover glass. Each assay was run for 1 h and time of mating was scored manually in 5-min bins within a temperature- and humidity-controlled room. For re-mating experiments, female flies were first mated overnight to wild type males. The next day, males were removed and mated females were group-housed for an additional ~24 h in vials containing standard food. Copulation assays were performed the following day at ZT1-2 within a temperature- and humidity-controlled room.
Statistical Analysis
Statistical analysis was performed using the GraphPad 6.0 Prism (GraphPad Software, La Jolla, CA) software package. For data shown in Fig. 1A, B, and D, an unpaired two-tailed Student’s t-test with analysis type (virgin vs mated) for each wild type fly stock was run. In Figure 2A and B, an unpaired two-tailed Student’s t-test with analysis type (virgin vs mated) as a factor was run individually for each condition (time of delay or age). For data shown in Figure 2C and 2D, an unpaired two-tailed Student t test with analysis type (virgin versus mated) as a factor was run for each nutrient source (A/S versus Food). In Fig. 2E, an unpaired two-tailed Student t test with analysis type (virgin versus mated) as a factor was run for flies on A/S, while a one-way ANOVA was run for each analysis type (virgin versus mated) for flies on food. Tukey multiple comparison post-hoc tests were performed based on the ANOVA results to determine which pairs were significantly different. For data shown in Fig. 3, a one-way ANOVA was run for each analysis type (virgin versus mated) for each experiment. Tukey multiple comparison post-hoc tests were performed based on the ANOVA results to determine which pairs were significantly different. For data shown in Fig. 4A, a one-way ANOVA was run for each analysis type (virgin versus mated) for each experimental genotype. Tukey multiple comparison post-hoc tests were performed based on the ANOVA results to determine which pairs were significantly different. For data shown in Fig. 4B, an unpaired two-tailed Student t test was run with analysis type as a factor (genotype). For sleep experiments shown in Figs. 5-9, a one-way ANOVA for each individual experiment was run, including genotype and mating status. Tukey multiple comparison post-hoc tests were performed based on the ANOVA results to determine which pairs were significantly different. See “Egg Laying” methods above for a statistical analysis of these experiments.
Results
Changes in postmating sleep occur across many genetic backgrounds, are female-specific, long lasting, and independent of age, nutrition, and egg laying.
Drosophila sleep patterns are sexually dimorphic, with female flies sleeping less than males during the day (Andretic and Shaw, 2005; Garbe et al., 2015; Huber et al., 2004). Previous research has shown that, after copulating with a male, mated females of the Oregon-R (OreR) wild type laboratory strain increase daytime locomotor activity and consequently decrease total amounts of daytime sleep (Isaac et al., 2010). We compared daytime sleep of virgin and mated females from OreR and three additional commonly used wild type laboratory lines, iso31, w1118, and Canton-S (CS), and found that all four wild type laboratory strains showed a post-mating decrease in sleep, which was specific to the daytime (Fig. 1A-C), supporting the idea that this effect occurs across many genetic backgrounds. However, one commonly used stock, White Berlin (WB), did not exhibit changes in daytime sleep following copulation (Fig. 1D; see Material and Methods). Examination of sleep architecture in lines that showed decreases revealed that reduced sleep was mainly due to changes in daytime bout length; although, minor changes in daytime bout number were also observed in the OreR line (Suppl. Fig. S1). Moreover, none of the isolated, naive, or mated male flies exhibited modified daytime or nighttime sleep following copulation, confirming that postmating changes in sleep are female-specific [Suppl. Fig. S2; (Isaac et al., 2010)].
We next sought to determine the duration of this postmating effect on sleep by delaying our analysis to discrete time points following copulation. A previous longitudinal study demonstrated that postmating decreases in sleep persist for up to 10 days (Isaac et al., 2010). However, one caveat to that longitudinal study was that flies remained individually confined to activity-monitoring glass tubes throughout the duration of the experiment, which could have independent effects on sleep. Thus, we avoided starting the sleep analysis in mated females immediately after copulation. Instead, after pairing males and females overnight for ~12-16 hours, we removed the males and subsequently group-housed mated females in standard food-containing vials for 1, 3, 6, or 9 days. After this time, mated females were loaded into glass tubes and analyzed for sleep. These experiments confirm that postmating effects on sleep are long-lasting; decreases in postmating sleep are still observed after delaying the experiment for at least 6 days. However, our results suggest that postmating sleep returns to baseline levels between 6 and 9 days after copulation (Fig. 2A), in slightly less time than previously reported (Isaac et al., 2010).
Young flies have higher amounts of daytime sleep than older flies (Kayser et al., 2014). To investigate whether age impacts postmating effects on sleep, we compared daytime sleep from virgin and mated female flies at 1, 3, and 9 days. As reported, we found that daytime sleep generally decreases with age (Fig. 2B); however, mating was able to further reduce daytime sleep even when flies were aged for 9 days (Fig. 2B), indicating that the postmating sleep response is independent of other age-related decreases in sleep.
The experiments described thus far were performed under standard conditions using Drosophila Activity Monitors (DAM) tubes containing 5% agar + 1% sucrose (A/S). It is well established that female flies do not lay eggs on A/S (Yang et al., 2008), suggesting that postmating changes in sleep are independent of egg laying. Moreover, the observed PMR in sleep could also be a byproduct of being tested in a nutrient-poor environment. Thus, we sought to investigate the PMR in sleep when flies were placed in DAM tubes containing standard laboratory food. While virgin daytime sleep was generally less on standard yeast-containing food using the traditional single-beam (SB) infrared (IR) DAM system, we did not detect further postmating decreases in sleep under these conditions in either the iso31 or the CS wild type backgrounds (Fig. 2C and D). This is consistent with previous results demonstrating that socially housed female flies (previously grouped with males and presumably mated) do not reduce daytime sleep on A/S+yeast using the SB monitoring system (Catterson et al., 2010). As the pattern of sleep:activity within the tube may change on standard laboratory food, we sought to obtain higher resolution data and so employed the more sensitive multibeam (MB) IR DAM system (Garbe et al., 2015). As with the SB monitors, the MB system detected overall less sleep in virgin flies on normal food than on A/S (Fig. 2E). However, in contrast to SB IR data, the MB system detected copulation-dependent decreases in sleep regardless of the nutrient source (Fig. 2E). Heat maps displaying the average time spent sleeping within individual beams of food-containing tubes within the MB system confirmed that mated, wild type females exhibited lower levels of sleep on regular food compared to virgins (Figure 2F). Differences in sleep levels between the SB and MB data suggest that the SB IR beam misses a portion of total activity when flies are placed in standard food-containing DAM tubes, thereby overestimating sleep duration; this reinforces the benefits of MB acquisition under specific contexts (Garbe et al., 2015). Together, these data demonstrate that postmating changes in sleep occur regardless of nutrient composition.
One obstacle of using mated females in food-containing DAM tubes is that egg laying ensues and larvae emerge, possibly complicating the results. Indeed, after 24 h, we observed larval movement by eye within the tube when mated females were housed in food-containing DAM tubes. Increased larval activity could potentially be mistaken as fly activity in the MB system, perhaps inappropriately lowering total sleep duration after mating. Thus, to verify that the observed postmating decrease in female sleep on standard yeast-containing food was a true behavioral change and not a result of misinterpreted larval activity, we restricted our analysis to the first day after mating and also mated females to tud1 males. tud1 mutants do not produce sperm and thus cannot generate fertilized eggs in females (Boswell and Mahowald, 1985). Similar to the MB data acquired using wild type males, tud1 males also produced postmating changes in female sleep within the first day of being placed on normal food (Fig. 2E). These data confirm that larval activity does not interfere with our analysis on the first day following copulation. Together, these data indicate that postmating decreases in daytime sleep are female-specific, long lasting, and independent of background, age, nutrition, and egg laying.
Ejaculatory Duct Proteins Do Not Influence Changes in Postmating Sleep
Products of the accessory glands, specifically Sex peptide (SP), and of the ejaculatory duct, particularly Dup99B, are important for many PMRs (Gillott, 2003; Saudan et al., 2002). While SP has been implicated in the postmating decrease in sleep (Isaac et al., 2010), a role for ejaculatory duct secretions has not been tested. To assess the function of each of these separately, we first used males in which the accessory gland main cells were specifically disrupted by targeted expression of diphtheria toxin subunit A [DTA-E; (Kalb et al., 1993)]. These males fail to produce main cell accessory gland proteins (Acps), including SP, but court/mate normally and retain ejaculatory duct secretions, including Dup99B (Kalb et al., 1993). Main cell accessory gland-ablated males failed to decrease female sleep after mating (Fig. 3A), demonstrating that neither ejaculatory duct peptides nor mechanical stimulation during the physical act of mating are sufficient for the observed postmating effects on sleep.
We performed another set of experiments using male flies genetically engineered to presumably block ejaculation during copulation. These males express the inward rectifying potassium channel, Kir2.1, in four male-specific abdominal ganglion (Abg) corazonin (crz) interneurons, thereby silencing their activity and supposedly blocking male ejaculation (Tayler et al., 2012). For our studies, the ProtB transgene [GFP-sperm; (Manier et al., 2010)] was incorporated into these crz-silenced flies to monitor sperm transfer into the female during copulation. Consistent with previous data, we found that males expressing Kir2.1 in crz neurons failed to transfer GFP-sperm into the female (Tayler et al., 2012). However, these males were still able to decrease female daytime sleep after mating (Fig. 3B). Moreover, crz-silenced males induced additional postmating responses including increases in rejection behavior, while DTA-E males did not (Fig. 3C). These data suggest that silencing crz neurons specifically blocks sperm transfer but other ejaculate secretions (presumably from the main accessory cells) are likely transferred to the female to alter PMRs. Combined, the DTA-E and crz-silencing experiments demonstrate that seminal fluid from the main accessory cell, but not sperm or ejaculatory duct proteins (see above), are required for modifying female sleep (Isaac et al., 2010). In addition, our data suggest that some secretions are indeed transferred in crz-silenced males to induce postmating responses (but see Tayler et al., 2012).
Sex Peptide and Its Receptor Contribute to Postmating Decreases in Sleep
Our data demonstrate that products of the main cells of the accessory gland are required for the postmating drop in sleep. The best candidate for this effect is SP, which was previously shown to be required for alterations in OreR female daytime sleep after mating (Isaac et al., 2010). This prior study, while convincing, was limited by: 1) a lack of confirmation that copulation occurred, and 2) the use of only a single homoallelic sp mutant (sp0/sp0). Thus, while it appeared that SP is required to produce changes in postmating sleep behavior, the phenotype was not actually mapped to the sp locus. We confirmed the relevance of SP by combining the sp null allele (sp0), containing a targeted mutation in the sp gene, and the sp deficiency allele (spΔ130) (Liu and Kubli, 2003), with ProtB [GFP-sperm; (Manier et al., 2010)]. This strategy allowed us to both investigate heteroallelic combinations of sp mutants as well as verify that all experimental females mated with sp mutant males as evidenced by GFP-sperm transfer. Wild type ProtB males produced the typical postmating decrease in female daytime sleep, while females mated to sp0/sp0 males (confirmed by GFP sperm transfer into the female abdomen) maintained near virgin-like sleep levels (Fig. 4A). These data confirm that homoallelic sp0/sp0 males mate normally but are unable to induce postmating changes in female sleep. Interestingly, heteroallelic sp0/spΔ130 males produced an intermediate effect: sp0/spΔ130 males consistently and significantly induced changes in female sleep but to a lesser extent than females mated to wild type males (Fig. 4A). Similar observations were made with respect to the effect of SP on receptivity (Feng et al., 2014), and may reflect the contribution of factors in the spΔ130 background. These data suggest that other proteins, in addition to SP, play a role in regulating female postmating sleep. Moreover, when female CS flies were mated to either homoallelic or heteroallelic sp mutant males (see Material and Methods), they, too, decreased sleep but to a lesser extent than wild type CS males (Suppl. Fig. S3). These data support the idea that SP contributes to PMRs but suggest that other factors are also involved.
Previously, it was shown that SP is able to induce oviposition via doublesex (dsx) neurons even in sex peptide receptor (spr) mutants, suggesting SPR-independent control of SP-regulated postmating behaviors (Haussmann et al., 2013). To investigate this possibility in regards to sleep, we next examined postmating sleep in spr mutants. As shown in Fig. 4B, females mutant for spr did not exhibit postmating decreases in sleep, maintaining similar levels of daytime sleep as virgins. Combined with the SP results above, these data demonstrate that both the peptide and its canonical receptor are at least partially responsible for decreasing female sleep immediately following copulation. Given that the SPR, via its other ligand, the myoinhibitory protein, was recently shown to function as part of a homeostatic sleep-stabilizing pathway in Drosophila (Oh et al., 2014), our data establish a multifaceted role for the receptor in controlling various aspects of sleep.
SPR Is Necessary and Sufficient in Sensory Neurons of the Female Reproductive Tract to Decrease Postmating Sleep
SP and SPR are partially required to induce changes in female postmating sleep; however, in what neurons is the receptor required to induce this response? For other PMRs, such as receptivity and dietary preference, SPR is required in a specific subset of SP-responsive sensory neurons (the SPSNs) in the female RT (Feng et al., 2014; Walker et al., 2015). However, SP and other accessory gland proteins (Acps) have been detected in the female hemolymph (after being deposited from the male), suggesting the possibility of action more distal to the source of transfer (Ding et al., 2003; Haussmann et al., 2013; Kubli, 2003; Lung and Wolfner, 1999). To address where the SP signal is received to modulate sleep, we tested whether SPR was specifically required in female RT neurons, using two distinct spr RNAi constructs that target different regions of the gene (Yapici et al., 2008) driven by the SPSN drivers, VT3280- and pickpocket (ppk)-Gal4. These two Gal4 drivers were previously shown to be expressed in neurons that modulate additional postmating behaviors (Feng et al., 2014; Häsemeyer et al., 2009; Rezával et al., 2012; Ribeiro and Dickson, 2010; Yang et al., 2009). Females in which SPR levels were reduced in VT3280 or ppk neurons failed to exhibit significant changes in sleep after mating with a male (Fig. 5A and B). Thus, SPR expressed in ppk and VT3280 neurons within the female RT is required for the decrease of postmating sleep.
Next, we asked whether SPR is sufficient in VT3280 and ppk neurons to regulate sleep after mating by re-expressing it in these neurons in spr mutants. We first confirmed transgenic rescue by comparing egg laying and receptivity of SPR-rescued flies to spr mutants without SPR re-expression (Yapici et al., 2008). As previously reported, expression of SPR in VT3280 neurons rescued postmating responses of egg laying and receptivity in spr mutants (Fig. 6A and B) (Häsemeyer et al., 2009; Ribeiro and Dickson, 2010; Walker et al., 2015; Yang et al., 2009). We then tested these flies for sleep levels following mating. Control spr mutant females containing the ppk-Gal4, VT3280-Gal, or UAS-SPR transgene alone exhibited near wild type levels of sleep (Fig. 6C and D). We noted that the spr mutant line containing UAS-SPR alone could be “leaky”, as low levels of rescue were observed for all three behaviors, even in the absence of Gal4 (Fig. 6A-C). However, expression of UAS-SPR specifically in the SPSNs, using ppk- or VT3280-Gal4, markedly increased the ability of spr mutant females to respond appropriately to the mating cue for all three behaviors, including sleep (Fig. 6A-D). These data demonstrate that SPR expression in VT3280 and ppk neurons is sufficient to restore the female postmating sleep response.
Silencing the SPSN and SAG Circuit Decreases Daytime Female Sleep
SP has been shown to induce postmating behaviors such as receptivity and dietary preference via the silencing of SPSN neuronal activity (Feng et al., 2014; Walker et al., 2015; Yang et al., 2009). SPSNs subsequently silence downstream second-order SAG neurons, presumably to modulate their communication with higher, behaviorally relevant brain centers (Feng et al., 2014; Walker et al., 2015). Therefore, we sought to determine if female sleep is also regulated via silencing of the same SPSN-SAG neuronal circuit. Consistent with the idea that postmating effects on sleep are female-specific (Fig. 1), we initially noticed that SAG-1 projections to the pars intercerebralis (PI) in adult brains are absent in male flies (Suppl. Fig. 4). These data extend the findings from another study demonstrating sexually dimorphic SAG-1 projections within the Abg (Feng et al., 2014). To address whether silencing the SPSN-SAG circuit affects female sleep, we used two different strategies to reduce neuronal activity of the SPSNs and SAG-1 neurons. First, we chronically silenced both sets of neurons using the Kir2.1 inward rectifying potassium channel. We initially confirmed this approach was effective by monitoring rejection and egg laying behaviors. As expected, chronically silencing the SPSN-SAG circuit by expressing Kir2.1 with VT3280-Gal4 and SAG-1-Gal4 moderately increased egg laying and induced rejection behaviors in virgin flies (Fig. 7A-C). Note, while this manipulation only partially increased egg laying compared with control flies mated to males, it fully reduced virgin receptivity (Fig. 7A-C), suggesting the possibility of mechanistic differences. We next asked whether chronically silencing this postmating circuit changes female sleep. Driving Kir2.1 in either of these Abg neurons decreased daytime sleep independent of mating status, demonstrating that neuronal silencing within this circuit modulates sleep. Similar to the egg laying response, sleep was only partially decreased as compared with that of control flies mated to males (Fig. 7D and E). Mating eventually occurred in some females after pairing them overnight with males, as evidenced by the presence (M+) or absence (M-) of GFP-sperm, but it failed to further decrease sleep. Interestingly, when paired with a male fly, female sleep trends lowered; however, this difference was not significant [compare VT3280>Kir2.1 virgin (v) to (M+) or (M-)]. Taken together, these data demonstrate that silencing neuronal activity with Kir2.1 in the SPSN/SAG-1 neuronal pathway induces postmating-like changes in egg laying, rejection, and sleep behaviors; however, given that egg laying and sleep are not affected to the same level as that seen when females are mated with a male, it is possible that receptivity is more sensitive to perturbations to, rather than silencing of, these neurons. Alternatively, other neurons may influence post-mating egg laying and sleep behaviors.
Since constitutive manipulations with Kir2.1 could cause developmental defects that indirectly affect adult behaviors, we next acutely silenced SPSNs and SAG neurons in adults using DREADDs [Designer Receptors Exclusively Activated by Designer Drugs; (Becnel et al., 2013)]. In this approach, G-protein-coupled receptors were engineered to be biologically inactive except in the presence of their exogenous, artificial ligand, CNO. Upon activation with CNO, the DREADDi (DRi) receptor specifically couples with inhibitory Drosophila G protein effector pathways (Gαi) to alter behaviors and physiological function in larvae or adults (Becnel et al., 2013). To confirm this technique worked in our assay, we initially investigated egg laying and rejection behavior following the expression of the DRi receptor in SPSNs and treatment with CNO. As expected, DRi receptor expression in VT3280 neurons induced both egg laying and rejection behaviors upon the addition of CNO (Fig. 8A and C). Activation of DRi receptor in VT3280 neurons by CNO also decreased female sleep; however, activating the DRs (Gαs) receptor had no effect (Fig. 8D). Likewise, silencing ppk neurons with DRi decreased sleep in virgin females (Suppl. Fig. 5). These data indicate that, like other postmating behaviors, acutely silencing neuronal SPSN activity in adulthood contributes to decreases in postmating sleep.
In the SAG neurons, induction of DRi with CNO also induced changes in sleep and rejection behaviors but it did not significantly enhance egg laying (Fig. 8B, C, and E). These data are consistent with the idea that egg laying is regulated largely by neurons found locally in the female reproductive system, potentially with less influence from higher brain regions (Monastirioti, 2003). However, since silencing SAG neurons with Kir2.1 marginally induced egg laying, we cannot rule out the possibility that DRi is expressed at lower levels when driven by SAG-1-Gal4.
Because silencing of either SPSNs or SAG neurons was able to decrease female sleep, we reasoned that artificially activating these neurons should block circuit silencing that occurs naturally after mating, thereby preventing a postmating response. To test this, we drove expression of the bacterial sodium channel NaChBac (Ren et al., 2001) in both VT3280 and SAG neurons. This genetic perturbation is predicted to elevate the resting membrane potential of the neurons. Indeed, when NaChBac was expressed in VT3280 neurons, mating no longer decreased female sleep (Fig. 8F). On the other hand, elevating the resting membrane potential of SAG-1 neurons did not block this effect; mated females still exhibited a reduction in daytime sleep (Fig. 8G). One interpretation of this latter result is that additional neurons downstream of the SPSNs contribute to the regulation of postmating sleep; however, similar to the DRi experiments above, we cannot rule out the alternative possibility that NachBac expression is weaker when driven by SAG-1-Gal4 and thus unable to prevent neuronal silencing after SPR activation.
SPSN Ablation Induces Rejection Behaviors in Virgin Flies
During the course of our investigations, in an attempt to block SPSN activity, we also killed these cells by expressing UAS-Reaper with VT3280-Gal4. Interestingly, SPSN-ablated virgin females completely rejected advances from males in our copulation assay (Fig. 9A). These data suggest that the loss of VT3280 neurons mimics the effect of neuronal silencing in virgin flies: both manipulations increase rejection behavior. However, ablation did not mimic silencing with respect to the other PMRs tested. Neither virgin sleep nor egg laying were affected by ablation of SPSN neurons, perhaps because other neurons compensate for the loss of SPSN neurons in virgin flies to regulate these behaviors. As in Figure 7, mating eventually occurred in some females after pairing them overnight with males, as evidenced by the presence (M+) or absence (M-) of GFP-sperm, but it failed to further decrease sleep (Fig. 9B and C). We surmise that VT3280-ablated females are unable to detect SP due to the lack of these sensory cells in the RT, thereby exhibiting egg laying and sleep responses that are similar to virgins and spr mutants. Thus, while the loss of SPSNs is compensated in virgins, it is not in mated females, where they are essential for postmating responses. These data also demonstrate that, in certain contexts, mechanisms controlling rejection behavior are separable from other PMRs and support the idea that multiple pathways mediate mating-regulated behaviors (Haussmann et al., 2013).
Discussion
In this study, we investigated postmating changes in Drosophila sleep and addressed the mechanisms underlying these changes. We show that postmating decreases in female daytime sleep are 1) to a large extent, independent of the genetic background of the fly, 2) observed in animals that are up to 9 days old, 3) persist at least six days after mating, and return to baseline levels by day 9, 4) not dependent upon nutrient content, and 5) independent of oviposition. Given the growing appreciation of background-dependent variations in sleep:wake (Faville et al., 2015; Zimmerman et al., 2012), it was critical to demonstrate that most common laboratory stocks exhibit this postmating response. Supporting the data in this manuscript, it was recently shown that mating also impacts sleep in another Drosophila species, Drosophila suzukii (Ferguson et al., 2015). Likewise, since flies decrease sleep naturally with age (Kayser et al., 2014), it was crucial to establish that mating decreases sleep independent of age-related effects. Lastly, it has been proposed that daytime sleep decreases after mating due to the female’s urge to find appropriate nutrient sources (Isaac et al., 2010; Riberio et al., 2015). Our multibeam data show that, even in the presence of standard laboratory yeast-containing food, postmating changes in sleep occur, suggesting that decreases in sleep are independent of nutrient availability.
Peptides and Receptors Regulating Postmating Behaviors
The list of identified behaviors and physiological processes altered by mating is growing. Currently, it includes changes in egg production and laying, rejection behaviors, locomotor activity, sleep/wake cycles, food preference, immune response, and longevity. Although the majority of the ~100 male-derived proteins transferred to the female during copulation have been identified (Avila et al., 2011), SP is largely responsible for most PMR changes. Our data agree with previous reports demonstrating that SP plays a major role in regulating postmating sleep (Isaac et al., 2010). However, our results using heteroallelic sp mutant males (to eliminate background modifiers) suggest potential contributions from other factors. Indeed, additional ejaculate proteins have been shown to subtly influence PMRs (Herndon and Wolfner, 1995; Ram and Wolfner, 2009; Rexhepaj et al., 2003). Moreover, another study investigating SPSN-SAG neurophysiology within individual flies reached similar conclusions, demonstrating a small but significant difference in receptivity between virgins and sp0-mated females (Feng et al., 2014, Suppl. Fig. S4E). Thus, while SP contributes to most observed postmating changes in behavior, other proteins may provide “accessory” functions to coordinately, or perhaps independently, regulate postmating switches. Interestingly, male flies that have been genetically engineered to block ejaculation still exhibit changes in PMRs (Fig. 3). While it is clear that GFP-sperm are not transferred during copulation, it seems likely that other seminal fluids are transmitted, given that multiple postmating behaviors are induced. We speculate that ejaculation is not entirely blocked in crz-silenced males, and transfer of seminal proteins, although not sperm, still occurs. However, it is formally possible that additional sensory cues can also induce these behavioral responses.
We show also that the canonical SPR is necessary for the postmating response and acts in VT3280 and ppk neurons. While a role for SPR may be considered a given, we note that SP induces oviposition via dsx neurons even in spr mutants, suggesting SPR-independent control of some SP-regulated postmating behaviors (Haussmann et al., 2013). In addition, SPR may have functions beyond those indicated here. For instance, it has been implicated in sleep homeostasis independently of the postmating switch, via an alternative ligand, myoinhibitory protein (MiP) (Oh et al., 2014). It would be interesting to determine if MIP affects postmating sleep.
Neuronal Circuits Receiving, Relaying, and Integrating Mating Status to Modulate PMRs
The majority of SP-dependent responses are mediated by a small group of sensory neurons within the female genital tract (Feng et al., 2014; Häsemeyer et al., 2009; Rezával et al., 2012, 2014; Walker et al., 2015; Yang et al., 2009). Within the proximal reproductive tract, relevant neuronal populations include doublesex (dsx), fruitless (fru), pickpocket (ppk), and VT3280 neurons. While partial overlap exists, unique populations of these neurons are anatomically distinct, suggesting functional differences. Accordingly, egg laying and receptivity are differentially affected by manipulations of subsets of these neurons (Feng et al., 2014; Häsemeyer et al., 2009; Rezával et al., 2012, 2014; Rideout et al., 2010; Walker et al., 2015; Yang et al., 2009). We also note that, since SP can be detected in fly hemolymph and SPR is expressed broadly in neuronal cells, behavioral regulation due to SPR activity at a distance from the point of SP transfer is also possible.
Here, we demonstrate that SPR is necessary and sufficient in VT3280 and ppk neurons to modulate postmating sleep. Thus, these SPSNs appear to receive male-derived SP to influence postmating daytime sleep duration. Silencing VT3280 neuronal activity induces postmating changes in sleep, egg laying, and rejection behavior in virgins, supporting mechanistic similarities. On the other hand, our VT3280 cell ablation experiments indicate that silencing and ablation similarly affect rejection behavior but that ablation does not induce changes in sleep and egg laying. We suggest that, in virgin flies, additional neurons compensate for the loss of VT3280 cells by keeping sleep high and egg laying low. However, VT3280 cells are essential for mating-induced decreases in sleep and increases in egg laying, as these changes do not occur in the absence of these cells.
The female’s nervous system must not only have the capacity to receive male-derived mating cues in the seminal fluid, but also have the ability to communicate and interpret these cues to direct relevant physiological and behavioral changes. What neurons are responsible for communicating mating status once it has been received? In addition to their roles in receiving the SP cue, populations of dsx, fru, and ppk neurons were shown to relay the mating status so as to control higher-order behaviors (Häsemeyer et al., 2009; Rezával et al., 2012; Yang et al., 2009). However, previous experiments were complicated by the fact that many of the Gal4 drivers used were broadly expressed, making it difficult to identify and manipulate distinct subpopulations of neurons (Rideout et al., 2010; Stockinger et al., 2005; Zhou et al., 2014). In an attempt to further delineate the relevant postmating neurons, both stochastic labeling and intersectional approaches were used to target a small subset of SAG neurons in the adult nervous system. These data showed that silencing the SPSNs propagates the mating signal from the reproductive tract to the Abg. In this context, the SPSNs synapse with and silence the SAG neurons, relaying the mating information to the brain and inducing rejection behaviors (Feng et al., 2014). In a recent study, it was demonstrated that SP signaling through the SPR within SPSNs also silences SAG activity to drive postmating changes in appetite (Walker et al., 2015). Together, these previously published data strongly indicate that silencing of the SPSN-SAG circuitry robustly induces at least two distinct postmating responses— receptivity and nutrient sensing.
Our results indicate that SP transferred during copulation also influences sleep through a shared SPSN/SAG circuit. This conclusion is drawn from two complementary sets of observations. First, as discussed above, SPR is necessary and sufficient in VT3280 neurons to respond to the mating cue. Moreover, silencing these VT3280 sensory neurons with either Kir2.1 or pharmacogenetically using the DREADDi receptor, decreases sleep. Second, we demonstrate that silencing downstream SAG neuronal activity also significantly decreases levels of postmating sleep. Thus, it appears that silencing neuronal activity of SAG neurons regulates postmating switches in receptivity, dietary preference, and sleep. Interestingly, the SAG neurons innervate regions of the pars intercerebralis (PI) (Feng et al., 2014), a functional homolog of the mammalian hypothalamus that was recently shown to be part of the circadian output circuit that drives rest:wake behavior (Foltenyi et al., 2007; Park et al., 2014; Cavanaugh et al., 2014). Neurons of the PI also mediate effects of octopamine on fly sleep:wake behavior (Crocker et al., 2010), raising the intriguing possibility that postmating status is communicated to the PI to regulate sleep.
While it is clear that the SPSN/SAG circuit mediates a large majority of the postmating behaviors, our SAG-1>NachBach data (Fig. 8) along with data from previous reports (Feng et al., 2014; Haussmann et al., 2013; Rezával et al., 2012, 2014), demonstrate that other neurons also influence many PMRs. In the future, high-resolution mapping of circuits and their associated activity changes upon SP stimulation will provide insight into the circuitry governing the SP-induced postmating switch of reproductive and sleep behaviors. Given the significance of sleep for an animal’s overall fitness, it is interesting that mating generates a situation in which a female evidently decreases her own fitness potentially for the good of her progeny. The fly model provides a unique opportunity to study how environmental cues impact an organism’s internal state.
Footnotes
Acknowledgements
We are very grateful to all the generous laboratories that contributed reagents and flies to these studies. See the Materials and Methods section of the manuscript for individual contributions. We also thank the Bloomington Stock Center for fly lines.
Conflict of Interest Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
