Abstract
Occurrence of 24-h rhythms in species apparently lacking functional molecular clockwork indicates that strong circadian mechanisms are not essential prerequisites of robust timing, and that rhythmical patterns may arise instead as passive responses to periodically changing environmental stimuli. Thus, in a new synthesis of grazing in a ruminant (MINDY), crepuscular peaks of activity emerge from interactions between internal and external stimuli that influence motivation to feed, and the influence of the light/dark cycle is mediated through the effect of low nocturnal levels of food intake on gastric function. Drawing on risk allocation theory, we hypothesized that the timing of behavior in ruminants is influenced by the independent effects of light on motivation to feed and perceived risk of predation. We predicted that the antithetical relationship between these 2 drivers would vary with photoperiod, resulting in a systematic shift in the phase of activity relative to the solar cycle across the year. This prediction was formalized in a model in which phase of activity emerges from a photoperiod-dependent trade-off between food and safety. We tested this model using data on the temporal pattern of activity in reindeer/caribou Rangifer tarandus free-living at natural mountain pasture in sub-Arctic Norway. The resulting nonlinear relationship between the phasing of crepuscular activity and photoperiod, consistent with the model, suggests a mechanism for behavioral timing that is independent of the core circadian system. We anticipate that such timing depends on integration of metabolic feedback from the digestive system and the activity of the glucocorticoid axis which modulates the behavioral responses of the animal to environmental hazard. The hypothalamus is the obvious neural substrate to achieve this integration.
Keywords
Robust daily timing synchronized by the solar cycle is a prominent feature of animal behavior. According to the standard model developed half a century ago, such timing is a product of interaction between endogenous, self-sustaining (i.e., circadian) rhythms and the organism’s response(s) to periodically changing environmental stimuli (Pittendrigh, 1960; Aschoff, 1960, 1966). In functional terms, circadian components of temporal organization, entrained by exogenous cues such as the 24-h light-dark (LD) cycle, confer fitness by enabling animals to synchronize their physiological state with rhythmic variation in environmental factors that influence their performance (Daan, 1981; Woods and Strubbe, 1994).
Strong circadian mechanisms are not, however, essential prerequisites of robust timing. Distinct crepuscular (i.e., dawn and dusk) peaks of locomotor activity in reindeer/caribou (Rangifer tarandus; hereafter “Rangifer”), for instance, track the solar cycle across the year (Erriksson et al., 1981) despite the apparent absence of a functional molecular clockwork in this species (Stokkan et al., 2007; Lu et al., 2010). Consistent with a weak circadian drive, these peaks persist neither during the continuous daylight of the northern summer nor, crucially, during the continuous darkness of the high Arctic winter (Svalbard reindeer at 78° N latitude: van Oort et al., 2005, 2007), implying that rhythmical patterns occur only as a passive response to the LD cycle.
The LD cycle has a powerful influence on the temporal pattern of activity in ruminants mediated through its effect on gastric function. Lengthy retention time of digesta (Clauss et al., 2010) generates an ultradian rhythm of activity in which bouts of foraging, typically lasting 0.5 to 4 h, alternate with bouts of rumination/resting of similar duration (Van Soest, 1982; Forbes, 1995; Gregorini et al., 2006). The resulting rhythm has a period of around 4 to 6 h (e.g., Gibb, 2007; van Oort et al., 2007) but is often only weakly expressed owing to the appearance of pronounced crepuscular peaks of activity that superimpose a strong 24-h rhythm on the ultradian component (Gregorini, 2012).
Gregorini and colleagues (2013, 2015) proposed a synthetic model of a grazing ruminant (MINDY) in which the crepuscular peaks of grazing are a product of interaction between internal and external stimuli that influence motivation to feed. Foremost among the latter is light. Ruminants are generally less active and graze little in the dark (e.g., Georgii, 1981; Gibb et al., 1998; Rutter, 2006; Pagon et al., 2013; Sheahan et al., 2013; Owen-Smith and Goodall, 2014) owing to their being 1) prone to attack by nocturnal/crepuscular predators, 2) better able to detect predators in daylight, and 3) more conspicuous and therefore more susceptible to attack when active (e.g., Lima and Dill, 1990; Kie, 1999). The resulting low nocturnal levels of feed intake (Sibbald, 1994; Stimmelmayr, 2001; Graf et al., 2005), in conjunction with a long and intensive period of rumination, diminish both the supply of nutrients from the rumen and ruminal fill, thus increasing the animal’s motivation to feed in the morning (Gregorini, 2012). In MINDY, therefore, the prominent bout of grazing activity at dawn is modeled as a consequence of a drive to alleviate discomfort associated with hunger by redressing the energy deficit accumulated across the preceding night, while the prominent bout at dusk is modeled as a response aimed at promoting a steady release of nutrients at night (Gregorini et al., 2013). Implicit in this model, light is a proxy for risk of predation and influences the behavioral state of the animal for that reason. The timing of grazing events is, thus, a result of a direct effect of 1) the level of light (i.e., the perceived level of risk) on the subject’s behavioral state and 2) an indirect (passive) effect of light on its internal state in terms of rumen function. Perception of light and hunger set the onset of feeding at dawn while perception of light and food intake delimit the offset of feeding at dusk.
MINDY thus embraces 2 dimensions: energy transactions and light. However, while the dynamics of the former in relation to grazing behavior have been examined in some detail (Forbes and Gregorini, 2014; Gregorini, 2012; Gregorini et al., 2013, 2015), the richness of the latter has been little explored in this respect. There are, for instance, few data and no models bearing directly on whether ruminants associate darkness with danger (Gregorini et al., 2006) or on light-dependent alertness in this group. Moreover, MINDY in its present form fails to embrace the situation where behavioral decisions are simultaneously state and risk dependent. Both conditions may alter the priority afforded food and safety and hence the temporal distribution of activity (Gregorini et al., 2006; Ferrari et al., 2009; Kotler et al., 2010; Matassa and Trussell, 2014).
In contrast to MINDY, risk allocation (RA) models focus specifically on how animals allocate time in relation to the temporal distribution of risk and their energy state. The optimal strategy maximizing fitness is modeled in terms of a trade-off between 2 mutually incompatible behavioral states: foraging and not foraging. When foraging (strategy “Food”), prey gain energy but are exposed to increased risk of predation; not foraging (strategy “Safety”) reduces risk of predation but at the cost of zero energy intake (Brown and Kotler, 2004; Bednekoff, 2007; Hebblewhite and Merrill, 2009). High motivation to feed, reflecting high energy demand or low opportunity for or reward of feeding, is assumed to increase tolerance of risk (i.e., the level of risk that prey accept before ceasing to forage). High risk, in terms of the frequency and amplitude of hazard, on the other hand, may drive prey to abandon feeding opportunities (e.g., Fortin et al., 2015). It follows that where energy gain has a higher priority than survival, temporal partitioning between predators and prey is predicted to decrease, reflecting the likelihood that the emergent pattern of activity in prey is driven chiefly by their internal state (energy balance; Brodin, 2007; Cozzi et al., 2012; Lehmann et al., 2012; Bonter et al., 2013). Ultimately, this may result in reversal of the temporal pattern of behavior (“temporal niche switching”; Hut et al., 2012) as, for instance, where normally nocturnal or crepuscular prey adopt daylight or moonlight foraging (e.g., Haftorn, 1989; Duvergé et al., 2000; Rutter et al., 2002; Berger and Gotthard, 2008; Ferrari et al., 2009; Prugh and Golden, 2014). Conversely, where safety has higher priority than energy gain, temporal partitioning between predators and prey is predicted to increase, reflecting the fact that the emergent pattern of activity is chiefly predator driven.
Risk allocation models extend the dynamic range of decision making beyond that of MINDY but are arbitrary with respect to solar time. They consider neither circadian organization nor the influence of the LD cycle of gastric function (but see Bednekoff and Houston, 1994). The RA and MINDY approaches are not, however, mutually exclusive. We hypothesize that behavioral timing is influenced simultaneously by the independent effects of light on motivation to feed and on the perceived level of risk. Specifically, we predict that the antithetical relationship between these 2 drivers varies with photoperiod, resulting, therefore, in a systematic shift in the phase of activity relative to the solar cycle across the year. Here we formalize this prediction in a single model that we test using data on the temporal pattern of activity in Rangifer. Trade-off models formulated to include photodependency in both drivers have good potential for explaining shifts in the timing of daily behavior in natural settings.
Materials and Methods
Animal and Behavioral Activity
This study is based on data on general locomotor activity defined as endogenous movement with a spatial component at the level of the whole organism. For large ungulates, this includes grazing, walking/trotting, and social interactions but not ruminating (whether performed lying down or standing), and >85% of locomotor activity in female Rangifer is associated with grazing (Skogland, 1984; Tyler, 1987; Boertje, 1985).
Among large ungulates, Rangifer is a useful animal model in which to test the interactive effects of photoperiod and predation hazard on behavioral timing for 2 reasons. First, as a boreal to super-boreal species complex, it is naturally exposed to the maximum possible range of photoperiod (PP), extending from PP = 24 h in mid-summer to PP = 0 h in mid-winter. Second, it is preyed on by a variety of typically nocturnal/crepuscular mammal predators, including bear (Ursus species) (Boertje et al., 1988; Mowat and Heard, 2006), cougar (Puma concolor) (Wittmer, 2004), lynx (Lynx lynx) (Andrén et al., 2011; Mattisson et al., 2014), wolf (Canis lupus) (Dale et al., 1994; Kojola et al., 2004; Hebblewhite et al., 2007), and wolverine (Gulo gulo) (Wittmer, 2004; Andrén et al., 2011).
Data
Data consisted of continuous 24-h × 329-day records of general locomotor activity obtained in each of 6 female reindeer Rangifer tarandus tarandus (Figure 1). The animals, aged >31 months at the start of trials, formed part of a herd of approximately 700 semi-domesticated reindeer that belonged to Sámi pastoralists and grazed freely at natural mountain pasture around 70° N, 25° E in northern Norway throughout the year. All data were created and stored in 15-min bins in accelerometer-based loggers worn by the animals from which they were subsequently exported into spreadsheets (for details, see van Oort et al., 2004). Permission to work with the reindeer was granted by the Norwegian Committee on Ethics in Animal Experimentation.
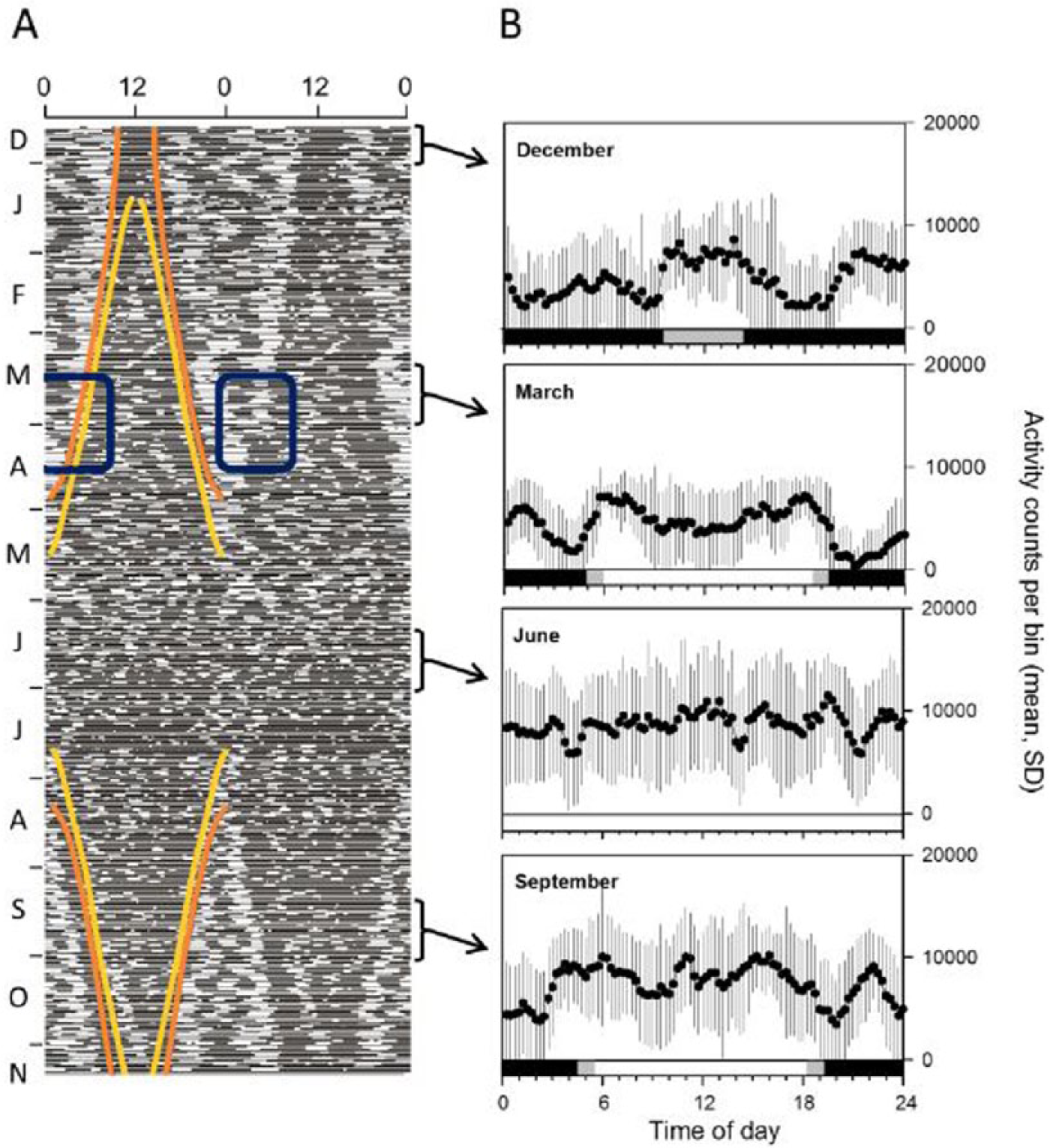
(A) Double-plotted actogram running from December to November for 1 of the 6 free-ranging female reindeer in the present trial (age at start of trial = 31 months). Each line represents 2 consecutive days (days 1-2, days 2-3, etc.); the abscissa is time of day (hours). Bouts of activity (black horizontal bars) are interspersed with bouts of inactivity (open spaces). Colored lines indicate the instant of civil twilight in the morning and evening (orange) and of sunrise and sunset (yellow). Note how from the beginning of March, as the nights grew shorter, the single nocturnal bout of activity first approached and then, in mid-April, fused with the dawn bout (blue rectangles). Figure from van Oort et al. (2007) with permission. (B) Rate of activity in consecutive 15-min intervals (bins) across 24 h × 13 days centered on December 25 and 24 h × 21 days centered on 21 March, 21 June, and 21 September. Data (logger counts per bin; from panel A) are plotted as mean (SD). Photoperiod for the 21st of each month is indicated in a horizontal band at the bottom of each graph (black = sun >6° below the horizon; gray = sun between 0° and 6° below the horizon, i.e., twilight; white = sun above the horizon).
Solar data for the location of the animals were extracted from http://aa.usno.navy.mil/data/docs/RS_OneYear.php.
Terminology
Bout: An uninterrupted sequence of 15-min bins of data of one sort (i.e., either activity or inactivity). n (bins) for a single bout is therefore ≥1.
Crepuscular activity: Activity performed mainly around dawn and/or dusk.
Dawn and dusk bouts of activity: Bouts coincident with the instant of sunrise or sunset, respectively.
Phase angle difference (Ψ): The interval (h) between a given stage of a bout of activity and the instant of sunrise or sunset. Following Daan and Aschoff (1975), values of Ψ onset (= the start of the dawn bout of activity) are by definition positive (precede sunrise) while values of Ψ offset (= the end of the dusk bout of activity) are necessarily negative (follow sunset).
Conceptual Model and Statistical Analysis
Trade-off model (Figure 2A)
We developed a qualitative graphical model of the phasing of dawn and dusk bouts of activity. The model defines the point of onset of activity at dawn and the point of offset of activity at dusk in terms of 1) a trade-off between the motivation to feed at these 2 times (M) and 2) the perceived risk of attack by nocturnal/crepuscular predators (PR) to which animals respond by seeking safety through inactivity (i.e., not feeding). M is a product of hunger dynamics modeled under 5 assumptions: 1) animals are reluctant to feed in the dark, 2) M increases with inactivity (i.e., zero food intake) owing to progressive decline in the supply of nutrients from the rumen and decreases with activity owing to the reverse (i.e., the approach to satiation; Gregorini, 2011; Gregorini et al., 2009b, 2011), 3) the probability of activity increases with M (Gregorini et al., 2007, 2009a; Gregorini, 2011) and decreases with PR (Lima and Bednekoff, 1999; Higginson et al., 2012; Fortin et al., 2015), 4) a bout of activity starts when M > PR and ends when PR > M (Fortin et al., 2015), and 5) PR decreases with light (i.e., predators are nocturnal/crepuscular). From these assumptions, it follows that M increases as PR decreases toward sunrise while it decreases as PR increases after sunset. Abscissa values where M = PR represent the optimal phase angle differences for maximizing energy intake and minimizing risk of predation and, hence, the time of start of the dawn bout of activity prior to sunrise (which defines Ψ onset ) and the time of the end of the dusk bout of activity after sunset (which defines Ψ offset ).
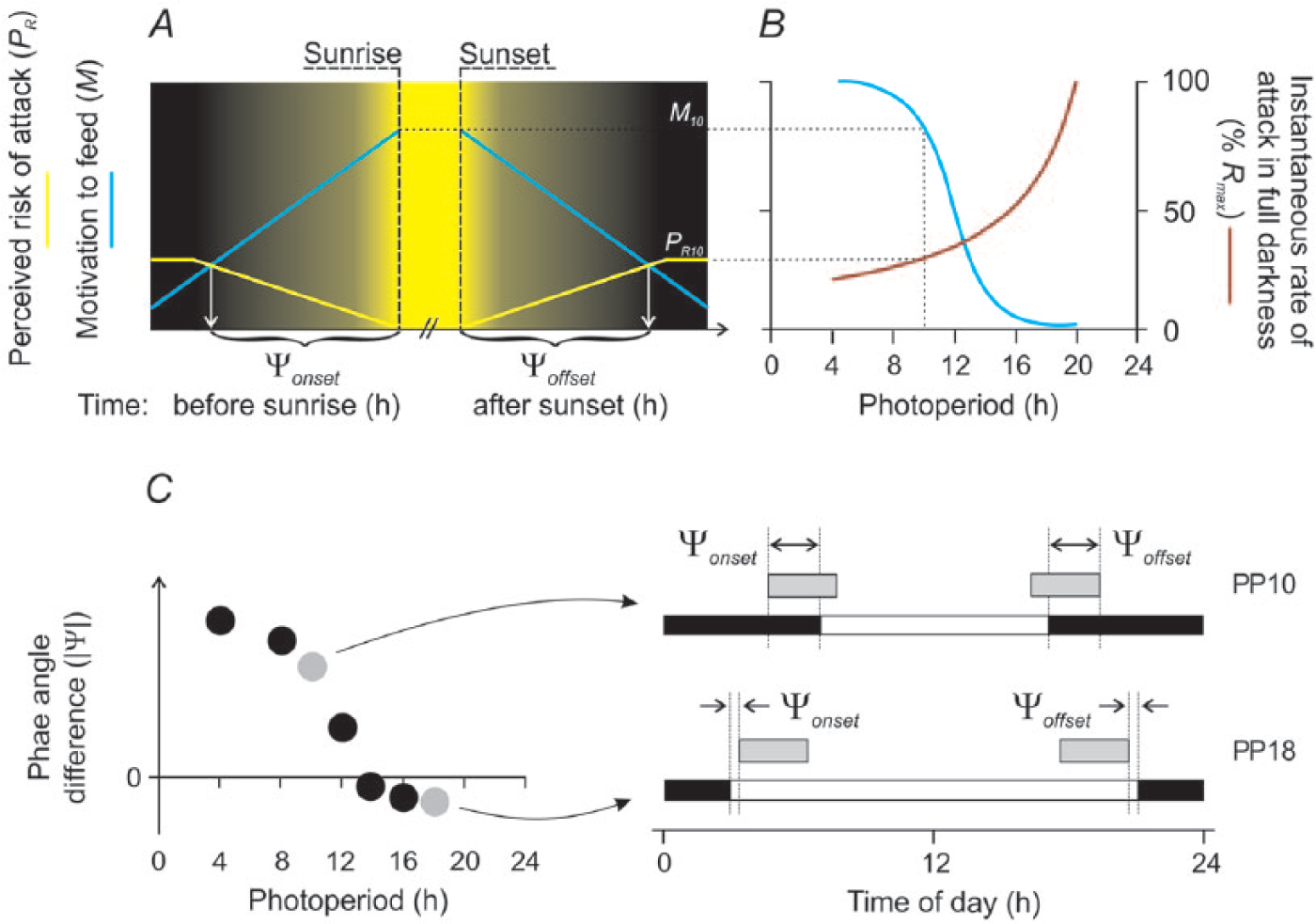
Conceptual model of the effect of photoperiod on the phasing of dawn and dusk bouts of activity in a wild ruminant. (A) Setting the phase angle difference. Trade-off between motivation to feed at dawn and dusk (blue line, M) and the perceived instantaneous risk of attack by nocturnal/crepuscular predators (yellow line, PR), which sets the onset of activity before sunrise and the offset of activity after sunset. We are concerned only with the value of M during the crepuscular phase. Thus, M increases toward sunrise, owing to reduced feeding in darkness, and decreases after sunset owing to feeding at dusk. PR takes a maximum value during full darkness and a minimal value between sunrise and sunset. Its transit from maximum to minimum (dawn) and from minimum to maximum (dusk) is a light-dependent function. PR is a function of the instantaneous rate of attack in full darkness (R), which changes across the year (see B). The abscissa coordinate at the point of intersection (M = PR) represents 1) the duration of activity in twilight which simultaneously maximizes time spent active (i.e., feeding; see Materials and Methods) and minimizes the perceived risk of predation and, therefore, also 2) the optimal phase angle difference for the start of the dawn bout of activity prior to sunrise (Ψ onset ) and the end of the dusk bout of activity after sunset (Ψ offset ). In this diagram the model is set at photoperiod (PP) = 10 h, and the motivation to feed (ordinate M10) is therefore 82% Mmax. For simplicity, the perceived instantaneous rate of attack in full darkness (ordinate PR10) is set equal to R10 and is therefore 29% Rmax (see B). (B) Effect of photoperiod. M is inversely proportional to photoperiod in the range 4 < PP < 20 h. M increases with decreasing photoperiod (owing to increased duration of darkness when the animals are reluctant to feed), but the relationship is sigmoidal (see Materials and Methods). The point of inflexion, set here at PP ≈ 12 h, occurs where nocturnal jejunation exceeds the animals’ fasting tolerance resulting in their having to forage in darkness. From this point, M approaches an asymptote (Mmax) before decreasing as photoperiod tends to 0 h (not shown). The instantaneous rate of attack in full darkness (R) is also proportional to photoperiod. The model assumes that the average number of attacks that a predator makes per 24 h remains constant across the year and R therefore increases with photoperiod to a point beyond which there are insufficient hours of darkness for the predator to meet its energy requirements exclusively through nocturnal/crepuscular hunting. From this point, Rmax, the predator must increasingly hunt in daylight and R will therefore decrease (not shown). Rmax is set here at PP ≈ 20 h. (C) Left-hand panel: Calculated abscissa values for Ψ at PP = 4, 8, 10, 12, 14, 16, and 18 h. The phasing of activity at the abscissa values PP10 and PP18 (gray symbols) is shown in the right-hand panel. Right-hand panel: Two specific scenarios for the effect of the tension between M and PR on the phasing of dawn and dusk bouts of activity. Bouts of activity (gray bars) are shown in relationship to sunrise and sunset, respectively, on 1 short day (PP10) and 1 long day (PP18). Daylight (white) and darkness (black) are indicated in the horizontal bar beneath each actogram. Under short days (PP = 10 h), the animals experience high motivation to feed before dawn/after dusk (M10 = 82%; see B) but are exposed to a low rate of attack (R10 = 29%, see B). The onset of the dawn bout of activity is therefore advanced relative to sunrise (increase in Ψ onset ) while the offset of the dusk bout is delayed relative to sunset (decrease in Ψ offset ) with the result that the animals spend more time active in twilight. Under long days (PP = 18 h), the animals experience low motivation to feed before dawn/after dusk (M18 = 2%, see B) but are exposed to a high rate of attack (R18 = 67%, see B). The onset of the dawn bout of activity is therefore delayed relative to sunrise (decrease in Ψ onset ) while the offset of the dusk bout is advanced relative to sunset (increase in Ψ offset ) with the result that the animals spend less time active in twilight. Thus, under long days, the dawn bout of activity starts earlier by solar time but later relative to sunrise than under short days, while the dusk bout ends later by solar time but earlier relative to sunset.
Effect of photoperiod (Figure 2B)
Both M and PR are inversely proportional to light level. The influence of light on M arises from the effect of reduced activity (i.e., predator avoidance in low light conditions) on gastric function (see “Trade-off model,” above). The effect of light on PR is a function of light level and the photoperiod-dependent instantaneous rate of attack by predators in darkness (R). In addition, both M and R are considered nonlinear functions of photoperiod. M increases under short days (long nights) to a point beyond which nocturnal jejunation exceeds the animals’ fasting tolerance. From this point, animals have increasingly to feed in the dark and M, while continuing to increase, approaches an asymptote. For R, the model assumes the average number of attacks that a predator makes per 24 h remains constant across the year. This simplifying assumption is consistent with observations by Stahler et al. (2006) who found only small seasonal differences in the daily rate of kill of ungulates by gray wolves (Canis lupus) in most years. R therefore increases under long days (short nights) so long as there are sufficient hours of darkness for the predator to meet its energy requirements exclusively through nocturnal/crepuscular hunting. Beyond that point (Rmax), it must increasingly hunt in daylight (e.g., Frafjord, 2013), and R therefore decreases (not shown).
Prediction (Figure 2C)
The model predicts that 1) Ψ onset and Ψ offset vary with photoperiod, and 2) the relationship will be curvilinear, reflecting the shifting relationship between relative magnitude of Mx and Rx.
Statistical analysis
The most parsimonious description of phase angle difference as a function of photoperiod was determined by confronting general linear models (glm) with second- and third-order polynomial models and general additive models (gam). The gam were estimated with nonparametrically smoothing splines. The back-fitting algorithm is a Gauss-Seidel method for fitting additive models by iteratively smoothing partial residuals (Venables and Ripley, 2002). All analyses were performed in R version 3.2.3 (R Core Team, 2015).
Results
Dawn bouts of activity in pooled data from 6 animals lasted on average (median) 3.9 h (quartile range, 3.9-5.0 h, n = 908); the corresponding values for dusk bouts of activity were 4.0 h (range, 2.8-5.3 h, n = 891).
Ψ onset and Ψ offset varied systematically across the year (Figure 3A,B), and confrontation of linear with nonlinear models (Table 1) confirmed the appropriateness of a curvilinear conceptual model for explaining temporal variation in phase angle difference in relation to photoperiod (Figure 2C). Specifically, the relationship between Ψ onset and photoperiod was best described by general additive models during both the first (January-May) and the second half of the year (June-December; Table 1) while the relationship between Ψ offset and photoperiod was best described by second- or third-polynomial models in the same 2 periods (Table 1). No linear glm was significant (Table 1).
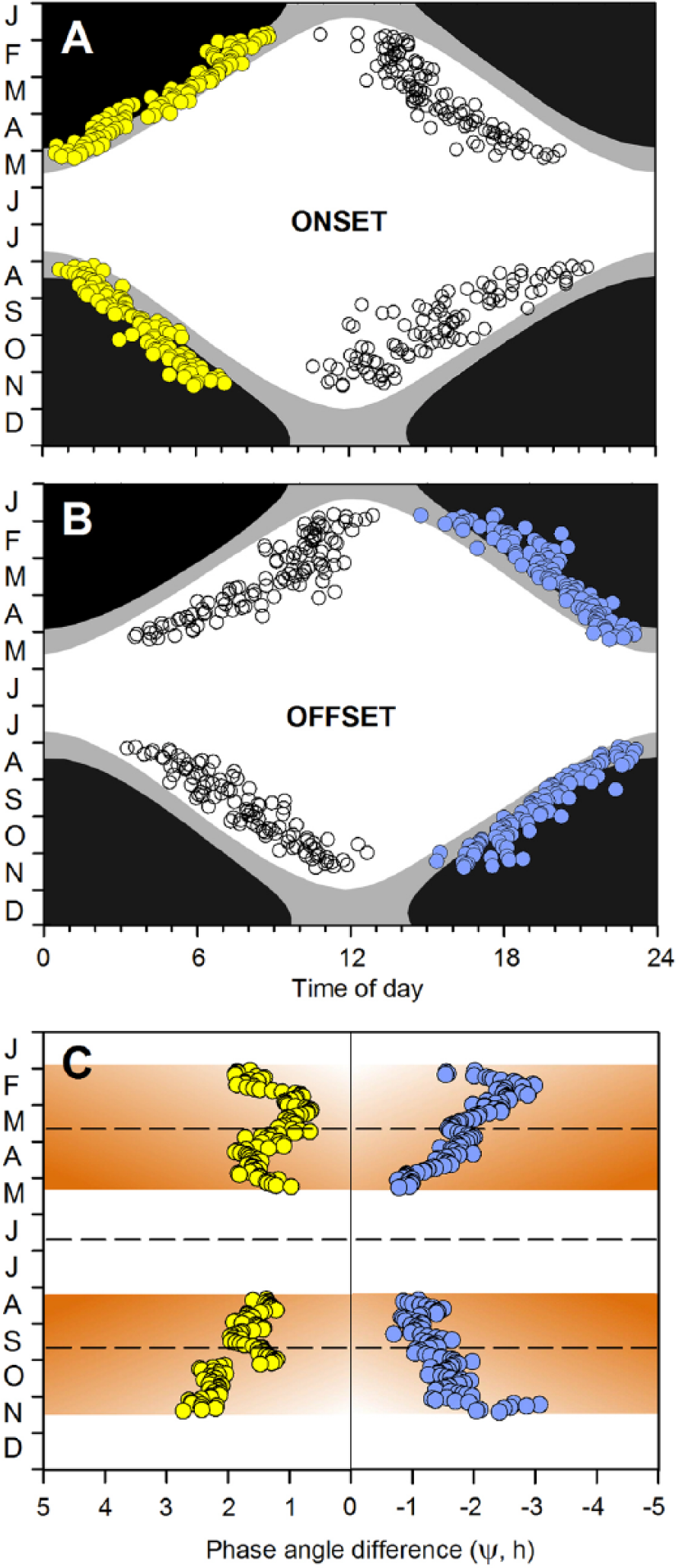
(A) Mean time of onset of each dawn (yellow circles) and each dusk (white circles) bout of activity (see Terminology) under photoperiods ranging from 4 to 20 h (25 January to 9 May and 4 August to 17 November at the latitude at which the study was undertaken) in free-ranging female reindeer (n = 6, age at start of trial = 31 months). Solar data: black = sun >6° below the horizon; gray = sun between 0° and 6° below the horizon, i.e., twilight; white = sun above the horizon. (B) Mean time of offset of each dawn (white circles) and each dusk (blue circles) bout of activity, respectively. Data and shading as in panel A. (C) Phase angle difference (Ψ h; 5-day running means) for the onset of dawn bouts of activity (yellow circles, from A) and for the offset of dusk bouts of activity (blue circles, from B). The abscissa value of zero = the instant of sunrise (for dawn bouts) and sunset (for dusk bouts). Phase angle differences are indicated positive for dawn bouts because these start before sunrise and are indicated negative for dusk bouts because these end after sunset. Overlapping points are not distinguished. The brown shading indicates relative perceived risk of attack by nocturnal/crepuscular predators (dark = high, light = low; see Figure 2B) during the hours prior to sunrise (positive abscissa values) or following sunset (negative abscissa values). Dashed horizontal lines indicate the vernal (21 March) and autumnal (21 September) equinox and mid-summer (21 June), respectively.
Linear and Nonlinear Analyses of the Response in Phase Angle Difference (Ψ s ) to Temporal Changes in Photoperiod (Predictor).
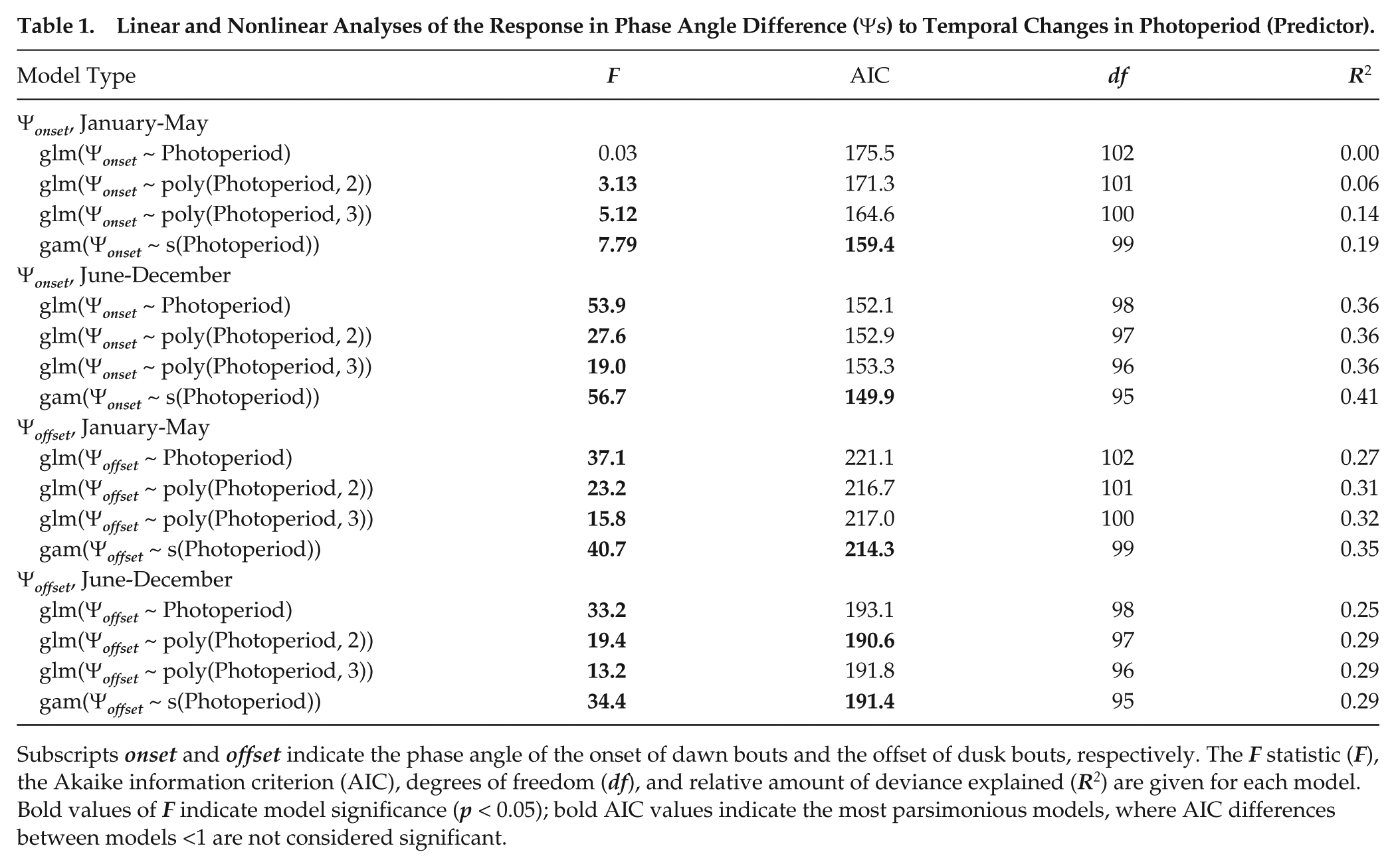
Subscripts onset and offset indicate the phase angle of the onset of dawn bouts and the offset of dusk bouts, respectively. The F statistic (F), the Akaike information criterion (AIC), degrees of freedom (df), and relative amount of deviance explained (R2) are given for each model. Bold values of F indicate model significance (p < 0.05); bold AIC values indicate the most parsimonious models, where AIC differences between models <1 are not considered significant.
Ψ onset decreased from 1.53 (3.53 to 0.12, n = 31) h (median, quartile range) under short days (January and November) to 1.28 (2.32 to 0.12, n = 32) h under long days (May and August) while Ψ offset increased (i.e., reduced in size) from −1.54 (–0.10 to −4.67, n = 31) h to −0.93 (–0.10 to −2.05, n = 46) h across the same periods (Figure 3C).
The relationships between the phase angle difference and both photoperiod (Table 1) and day of year (DOY: a proxy for photoperiod; Figure 3C, Suppl. Table S1) were highly nonlinear. The nonlinearity of the relationship between Ψ onset and DOY was exaggerated in the first half of the year, owing to reversal of its sign early in March and again in mid-April (Figure 3C), and consequently, Ψ onset approached its seasonal maximum under both short (end of January, PP ≈ 5 h) and long (mid-April, PP ≈ 16 h) photoperiods.
Discussion
We have identified a nonlinear relationship between the phasing of crepuscular activity and photoperiod in free-living Rangifer (Figure 3C; Table 1). The temporal pattern of behavior in these animals, which apparently lack a functional molecular clockwork (Lu et al., 2010), is, moreover, consistent with a model in which the phase of activity emerges from implementation of a photoperiod-dependent trade-off between food and safety. This suggests, in particular, that nonlinear adaptation of behavior to shifting photoperiod may emerge even where circadian organization is weak or absent.
We analyzed the temporal dynamics of behavior in terms of rumen function, based on the concept of a whole-animal grazing model (Gregorini et al., 2013, 2015), implemented within the context of RA theory (Lima and Bednekoff, 1999; Higginson et al., 2012). The resulting model, although cast as a trade-off between food and safety, differs from conventional RA models insofar as its output is behavioral timing rather than behavior. We are interested in when the animal is active rather than what proportion of time it allocates to activity. Indeed, the proportion of time spent active is set at unity in our model. The predictions of the 2 approaches are nevertheless similar: in each case, the allocation of time to activity in a high-risk state increases with exposure to risk (i.e., Ψ ∝ [1 − P] in our model) but decreases with level of risk (R, which is equivalent to the attack rate ratio [αH/αL] in the RA model [Lima and Bednekoff, 1999]) and internal state (motivation to feed [M ] or “energetic condition” [Lima and Bednekoff, 1999], respectively). The likeness extends to activity under constant risk where the ratio of activity under high and low risk converges to unity (i.e., αH = αL in RA models). This is not explicit in our model only because we restricted analysis to the period of year with strong light-dark cycles (i.e., 4 < PP < 20). Temporally invariant risk pertains, however, under continuous light (PP = 24) or dark (PP = 0) under which conditions Rangifer is active around the clock (van Oort et al., 2005, 2007) as our model predicts (i.e., M = Ψ = 0). The key difference in our approach, however, is the central assumption that animals use the intensity of light as a proxy for risk. Thus, in our model, light changes the way the animal perceives its feeding environment with regard to potential (or imminent) risk of predation and modulates its functional response accordingly.
Consistent with our model, Ψ onset was generally proportional and Ψ offset inversely proportional to photoperiod in Rangifer (Figure 3C; Table 1, Suppl. Table S1). The onset of the dawn bout of activity and the offset of the dusk bout both shifted further into twilight as day length decreased in winter and were withdrawn from twilight as day length increased at the approach of summer (Figure 3C). In functional terms, the animals displayed increased tolerance of risk and hence began activity progressively earlier before sunrise and remained active progressively later after sunset as motivation to feed at dawn and dusk increased in winter. Conversely, they displayed reduced tolerance of risk and hence delayed the onset of activity before sunrise and ended activity earlier after sunset as motivation to feed at dawn and dusk decreased in summer. The timing of activity was, thus, in effect driven chiefly by motivation to feed in winter when, under short days, the animals were unable to meet their energy requirements by daylight feeding alone and chiefly by risk avoidance in summer when, under long days, feeding opportunities were not light limited.
We attribute the replacement of a monotonic (sigmodal) by a polyphasic response curve for Ψ onset in the first half of the year—negative to March, positive from March to mid-April, and negative from mid-April (Figure 3C)—to a predictable shift in the temporal dynamics of activity. Thus, as nights grew shorter toward summer, the nocturnal bout of activity approached and eventually fused with the advancing dawn bout (blue rectangle in Figure 1A). Consistent with the model, Ψ onset decreased with photoperiod for the dawn bout prior to fusion and for the composite bout after fusion. Deviation from prediction (reversal of sign) around the spring equinox coincided with, and is probably a consequence of, the process of fusion itself. We note that the transition was achieved in just a few days in a single animal (Figure 1A), whereas the sign of the relationship was reversed for several weeks in the combined sample (Figure 3C): presumably, the individual responses were not tightly synchronized in this respect.
Our model focuses only on the influence of photoperiod on the extension of activity into the crepuscular phase and considers the behavioral state of the animal (active, nonactive) only in terms of a trade-off between motivation to feed and instantaneous perceived risk of attack. These are key points. Features of the daily pattern of behavior, such as the apparent anticipation of dusk evident in data for March (Figure 1B), potentially demand more complex explanations. The point here, however, is not whether models involving anticipatory timers of one form or other may also account for the data, but rather that a model that invokes no such timer(s) accounts for observations that have elsewhere been attributed them (e.g., Pittendrigh and Minis, 1964; Pittendrigh, 1981). Moreover, Ψ onset only weakly predicts Ψ offset (Suppl. Figure S1), and potentially anticipatory facets of the daytime pattern therefore have no bearing on the crepuscular pattern.
Both the occurrence of a seasonal shift in the timing of activity in relation to sunrise and sunset and its functional significance may be quite general. Thus, photoperiod-dependent shifts in Ψ onset and Ψ offset have been observed in Svalbard ptarmigan, Lagopus muta hyperborea (Reierth and Stokkan, 1998), and in a variety of non-boreal species of songbirds and small mammals exposed to a high-latitude photoperiod (Daan and Aschoff, 1975). The phasing of activity in these latter, in particular, varied with the photic regime to which the animals were exposed. Thus, seasonal shifts in Ψ onset and Ψ offset , which were barely discernible at 47° N, increased spontaneously, becoming prominent when the animals were transported and exposed to ambient photoperiod at 66° N (Daan and Aschoff, 1975). Crucially, none of the species tested were Arctic residents—indeed, the northern treeshrew (Tupaia belangeri) is a tropical species that normally never experiences large variation in photoperiod. Daan and Aschoff’s (1975) data indicate that photoperiod-dependent phasing of crepuscular activity relative to sunrise and sunset is a highly plastic, acute response to darkness and one that potentially reflects a widespread adaptive response through which diurnal ecotypes minimize the risk of nocturnal predation.
In apparent contrast with the present results, Ensing and colleagues (2014) reported no seasonal shifts in Ψ onset and Ψ offset in 2 populations of red deer (Cervus elaphus). However, the low resolution of their data (1 sample every 2 h) was not sufficient to detect values of Ψ of the magnitude observed in the present study and elsewhere (Daan and Aschoff, 1975; Reierth and Stokkan, 1998) which would therefore have precluded detection of similar responses in their deer.
In our model the effect of darkness on the timing of behavior is a consequence of reduced feed intake. In small birds and mammals, by contrast, the effect of darkness on the timing of activity/rest cycles is generally interpreted in terms of changes in energy expenditure (e.g., Brodin, 2007; Lehmann et al., 2012; Hut et al., 2011). The difference in perspective reflects the fact that we analyzed patterns of activity in a well-insulated mammal that remains comfortably within its thermoneutral zone under all but the most extreme weather conditions (Moote, 1955; Nilssen et al., 1984). This is not the case for small, relatively poorly insulated animals (Scholander et al., 1950; Hut et al., 2012). In these, night commonly represents a period of increased metabolic demand and entering a phase of nocturnal inactivity, whether through roosting or resting in insulated burrows, therefore represents an effective way of reducing energy costs. Indeed, nocturnal mice can be made to shift their activity into the light phase, or even to enter torpor, by reducing ambient temperature to 10 °C, which is some 40 °C above the lower critical temperature of winter-insulated reindeer (Nilssen et al., 1984; Hut et al., 2011; van der Vinne et al., 2014). Thermal defense has also been invoked to account for the persistence of daily activity rhythms through the polar summer in Arctic ground squirrels (Urocitellus parryii; Williams et al., 2012). The key point, however, is that shifts in metabolic balance, whether in reindeer or rodents, can alter behavioral timing and this may occur independent of central clock function. Indeed, shifts in the activity patterns of mice can occur without any corresponding changes in the profiles of circadian gene expression in the suprachiasmatic nucleus (van der Vinne et al., 2014).
The photoperiod-dependent trade-off we have developed here, like the thermoenergetic model (Hut et al., 2011; van der Vinne et al., 2015), provides a mechanism for behavioral timing that is independent of the core circadian system. We anticipate that such timing depends on integration of metabolic feedback from the digestive system with the activity of the glucocorticoid axis. The latter shapes the psychological state of the animal and, hence, its behavioral responses to environmental hazard (Rodrigues et al., 2009). The hypothalamus is the obvious neural substrate to achieve this integration. A full synthesis must also take into account seasonal—as opposed to purely light-dependent—changes in the physiology and ecology of species under consideration. Seasonal changes in energy demand associated with rhythms of growth and reproduction (Loudon et al., 1989; Loudon, 1994; Tyler et al., 1999; Rhind et al., 2002), for instance, are likely to contribute to the phase-angle response function. Exploration of these effects in wild species such as Rangifer, for which extensive behavioral data can be coupled with good understanding of seasonal physiology, is an exciting prospect.
Footnotes
Acknowledgements
We are most grateful to Lars Folkow, who kindly provided data on activity patterns in reindeer upon which this study is based, and to Mike Forbes, Roelof Hut, and 2 anonymous referees who kindly commented on an earlier version of the manuscript.
Conflict of Interest Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
