Abstract
Ocular clocks, first identified in the retina, are also found in the retinal pigment epithelium (RPE), cornea, and ciliary body. The retina is a complex tissue of many cell types and considerable effort has gone into determining which cell types exhibit clock properties. Current data suggest that photoreceptors as well as inner retinal neurons exhibit clock properties with photoreceptors dominating in nonmammalian vertebrates and inner retinal neurons dominating in mice. However, these differences may in part reflect the choice of circadian output, and it is likely that clock properties are widely dispersed among many retinal cell types. The phase of the retinal clock can be set directly by light. In nonmammalian vertebrates, direct light sensitivity is commonplace among body clocks, but in mice only the retina and cornea retain direct light-dependent phase regulation. This distinguishes the retina and possibly other ocular clocks from peripheral oscillators whose phase depends on the pace-making properties of the hypothalamic central brain clock, the suprachiasmatic nuclei (SCN). However, in mice, retinal circadian oscillations dampen quickly in isolation due to weak coupling of its individual cell-autonomous oscillators, and there is no evidence that retinal clocks are directly controlled through input from other oscillators. Retinal circadian regulation in both mammals and nonmammalian vertebrates uses melatonin and dopamine as dark- and light-adaptive neuromodulators, respectively, and light can regulate circadian phase indirectly through dopamine signaling. The melatonin/dopamine system appears to have evolved among nonmammalian vertebrates and retained with modification in mammals. Circadian clocks in the eye are critical for optimum visual function where they play a role fine tuning visual sensitivity, and their disruption can affect diseases such as glaucoma or retinal degeneration syndromes.
Ocular Circadian Rhythms
A role for circadian mechanisms in the vertebrate retina was adumbrated in work on retinomotor movements of rods, cones, and melanin pigment granules of the retinal pigment epithelium in the catfish retina (Welsh and Osborn, 1937). These cellular movements in fish and amphibia are thought to be adaptive because they optimize light capture by rods and cones at different times of day. Interestingly, extensive investigation of retinomotor movements in the late 19th and early 20th centuries had led to general conclusion that they were direct responses to light or darkness (Arey, 1915). Although this appears to be correct in some species (Ali, 1975), the analysis of catfish, which itself followed similar findings for pigment granule migration in crustacean eyes (Welsh, 1930), demonstrated that the movements between daytime and nighttime positions were blocked in constant light but persisted in constant darkness with noticeable damping by the second day. The general conclusion that retinomotor movements persist in constant darkness has been extended to many teleost and amphibian species, including Xenopus laevis (Besharse et al., 1982) and zebrafish (Menger et al., 2005). However, the pattern varies greatly among species and is strongly correlated with the animal’s own activity rhythms (Ali, 1975; Besharse, 1982; Douglas and Wagner, 1982).
Early work also identified a strong circadian rhythm of mitotic activity in the corneal epithelium (Scheving and Pauly, 1974; Scheving et al., 1974) and of intraocular pressure (Boyd and McLeod, 1964), suggesting a broader role of circadian regulation in the eye. However, an intense interest in circadian mechanisms began with the finding that turnover of the phototransduction organelle, the outer segment, is controlled through circadian mechanisms in both rat (LaVail, 1976) and in X. laevis (Besharse et al., 1977). Retinomotor movements are for the most part limited to fish and amphibia, but the rapid turnover of the phototransduction organelle through disc shedding occurs in all vertebrates and is critical to visual function (Young, 1976). Thus, the discovery of disc-shedding rhythms was followed by a surge in research on the control mechanisms (Besharse, 1982) and attempts to study the process in vitro (Besharse et al., 1980; Heath and Basinger, 1983).
A Light-Sensitive Retinal Circadian Clock
Use of an in vitro eye cup system for the study of rod disc shedding (Besharse et al., 1980) and cone retinomotor movement (Besharse et al., 1982) in X. laevis led inevitably to the discovery of a light-sensitive circadian timing mechanism within the eye (Besharse and Iuvone, 1983). The histological assays for disc shedding and cone movement were rather cumbersome for around-the-clock analysis of in vitro experiments. However, the finding that a circadian melatonin synthetic pathway similar to that of the pineal gland was present in the vertebrate retinas (Hamm and Menaker, 1980) and that assays for serotonin N-acetyl transferase (AANAT, NAT), the penultimate enzyme of the pathway, could be used to follow rhythmicity in X. laevis (Figure 1) enabled an immediate determination of whether AANAT rhythms persisted in darkness in eye cups (Besharse and Iuvone, 1983).
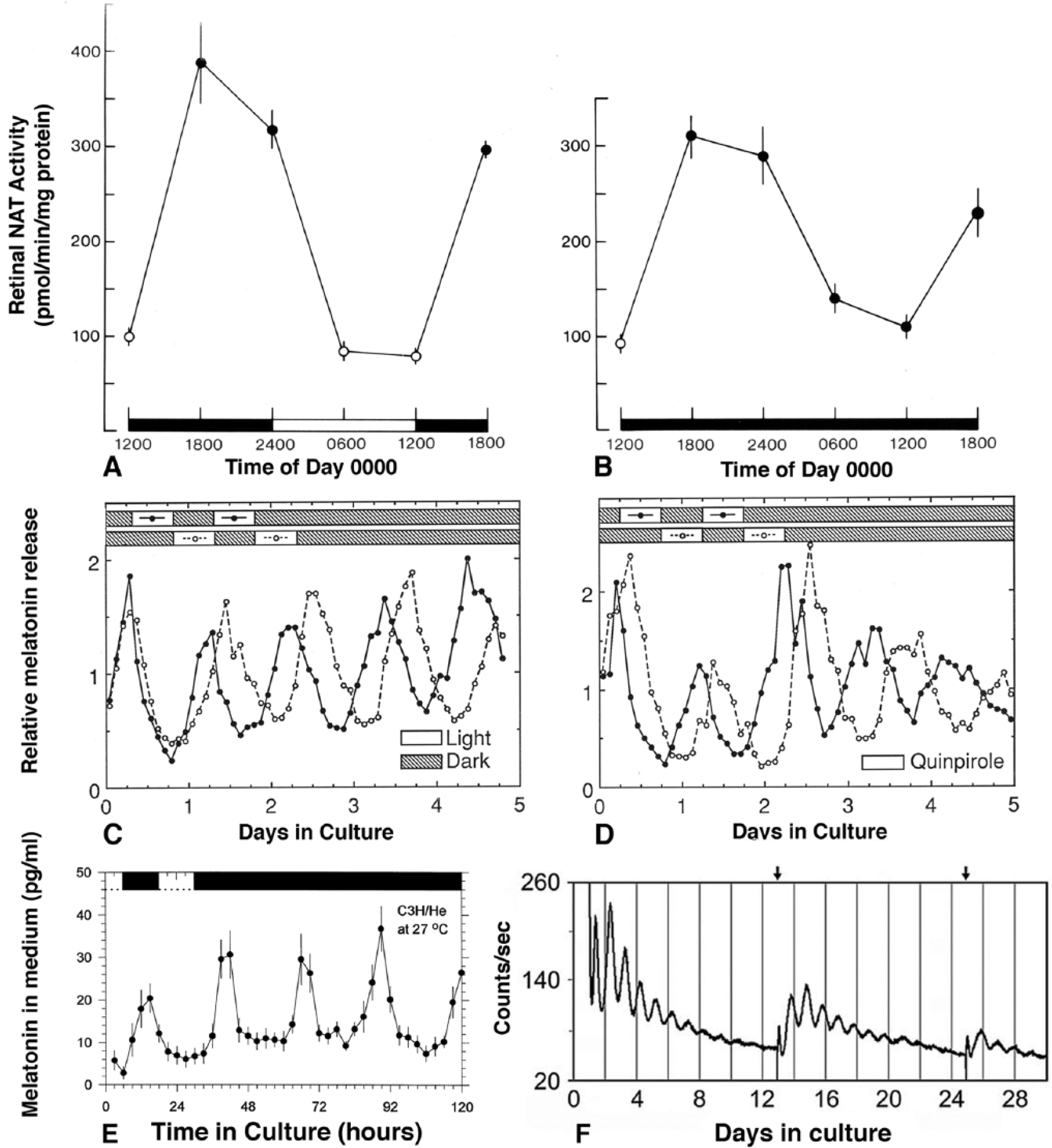
Circadian oscillations in vitro of eyecups from Xenopus laevis (A and B), individual photoreceptor layers from X. laevis (C and D), isolated retinas from melatonin-proficient C3H/He mice (E), and an isolated retina from a melatonin-deficient C57BL/6 mouse (F). A and B are measurements of activity AANAT, a melatonin synthetic enzyme, at different times of day in a light-dark cycle (A) or constant darkness (B) in culture (from Besharse and Iuvone, 1983, with permission of the publisher). C shows light-dependent phasing of melatonin release rhythms (measured every 2 h) from 2 isolated photoreceptor layers maintained in flow through culture on opposite light-dark cycles for 2 days followed by constant darkness. D is similar to C except that the cultures are in constant darkness and are maintained on opposite cycles of quinpirole, a dopamine D2/D4 agonist, instead of light for 2 days before continued culture in darkness (C and D are from Cahill and Besharse, 1993, with permission of the publisher). (E) Average melatonin release from isolated retinas of melatonin proficient mice (from Tosini and Menaker, 1998a, with permission of the publisher). (F) PER2::luc rhythm of a single isolated mouse retina over 29 days in culture with near-continuous monitoring of luciferase activity. Note that the robust luciferase damps over time, but its amplitude is partially restored when medium is changed (arrows at top) on days 12 and 24 (from Ruan et al., 2008, with permission of the publisher).
The general thinking at the time was that central clocks such as the suprachiasmatic nucleus (SCN) drove circadian rhythms in tissues such as the eye through neuronal or systemic outputs. This was based on entirely reasonable evidence as the SCN drives circadian melatonin secretion in the rodent pineal gland through neuronal input via norepinephrine receptors (Klein, 1974; Klein and Weller, 1970). Furthermore, a similar mechanism had been identified in the lateral eye of the horseshoe crab, Limulus polyphemus (Barlow, 1983; Kaplan and Barlow, 1980). Here, high-amplitude circadian rhythms of membrane turnover and visual sensitivity are driven by circadian changes in the firing of efferent neurons from a circadian pacemaker in the brain. On the other hand, there was emerging evidence for local clock function in nonmammalian vertebrates, with particularly strong evidence coming from cultured chick pineal gland (Kasal et al., 1979; Takahashi et al., 1980; Zimmerman and Menaker, 1979). The data from Xenopus showed unequivocally that a circadian clock whose phase could be shifted by light was present within the eyecup (Figure 1A,B).
The AANAT activity rhythm in Xenopus eyecups (Besharse and Iuvone, 1983) raised the expectation of a rhythm of melatonin release similar to that in pineal tissue that would enable continuous circadian studies of clock activity in single eyes. However, it proved difficult to detect sustained melatonin release in part because melatonin was rapidly metabolized within the eye to 5-methoxytryptamine (Cahill and Besharse, 1989). In addition, the overall capacity of the retina to make melatonin was limited at the first step of the pathway, tryptophan hydroxylase (Cahill and Besharse, 1990), which itself was transcriptionally regulated in a circadian pattern (Green and Besharse, 1994; Green et al., 1995). Ultimately, an eyecup flow-through system using medium supplemented with hydroxytryptophan was designed that exhibited high-amplitude, rhythmic release of melatonin for the detailed characterization of a retinal circadian clock (Cahill and Besharse, 1991). This study also provided the first evidence for a role of dopamine in clock resetting and led to the demonstration that isolated rod and cone photoreceptor layers contained a clock that could also be reset with light and dopamine acting at D2-like receptors (Figure 1C,D) (Cahill and Besharse, 1992, 1993). These studies were also novel in that they provided the first unequivocal evidence that melatonin was synthesized by photoreceptors that also contained D2-like receptors.
While the Xenopus eyecup provided the formal demonstration of a self-sustained retinal circadian system in vitro, it became clear very quickly that retinal clocks were a general phenomenon. In particular, eye patch experiments combined with optic nerve transection demonstrated the presence of a retinal clock releasing melatonin in Japanese quail (Underwood et al., 1990), and photoreceptor-enriched cultures from chick retina were shown to exhibit a circadian rhythm of iodopsin expression in culture (Pierce et al., 1993). Furthermore, using a flow-through system similar to that in Xenopus, it was shown (Cahill, 1996; Tosini and Menaker, 1996, 1998a) that a retinal clock regulating melatonin release exhibited a circadian pattern in zebrafish, hamsters, and melatonin-proficient mice (Figure 1E) (Cahill, 1996; Tosini and Menaker, 1996, 1998a). Interestingly, the melatonin rhythm in zebrafish retina damped quickly in DD compared to the pineal gland (Cahill, 1996).
The Retinal Clock in Rodents
An important development in the study of peripheral circadian oscillators was the introduction of bioluminescent or fluorescent reporter animals such as the transgenic Per1::luc rat (Wilsbacher et al., 2002), Per1::GFP transgenic mice (Kuhlman et al., 2000; Zhang et al., 2005), PER2::LUC knock-in mice (Yoo et al., 2004), and the Per3::luc zebrafish (Kaneko et al., 2006). In these models, green fluorescent protein (GFP) or luciferase is driven downstream of the clock machinery and is not dependent on tissue-specific pathways such as melatonin secretion. One of their most interesting uses was to reveal (Kaneko et al., 2006; Ruan et al., 2008) circadian oscillation in vitro in many tissues, including retina (Figure 1F). In the retina, however, expression of the Per1::GFP and PER2::luc reporters occurs predominantly in the inner retina where Per1::GFP has been specifically colocalized with nictric oxide synthase (NOS) and GABAergic and dopaminergic amacrine cells (Ruan et al., 2008; Zhang et al., 2005). Thus, unlike the situation in Xenopus where circadian efflux of melatonin reflects clock activity in photoreceptors, Per1::GFP and PER2::luc reporters in the mouse retina primarily reflect the circadian activity of the inner retina.
Photoreceptors in wild mice and laboratory rats synthesize and secrete melatonin rhythmically in a manner similar to X. laevis. However, most commonly used inbred mouse strains such as C57Bl/6j lack the capacity to produce melatonin, perhaps owing to the selection for fecundity in the origin of those strains (Ebihara et al., 1987; Ebihara et al., 1986). Paradoxically, mouse strains that do produce melatonin such as C3H also carry the rd1 (PDE6B) mutation that causes rapid photoreceptor degeneration (Bowes et al., 1990; Pittler and Baehr, 1991). Thus, it has been necessary to use lines derived from C3H mice that are wild type at the rd1 locus to study melatonin rhythms in the retina (Argamaso et al., 1995; Doyle et al., 2002a). The general findings from this approach greatly extend the earlier concept of mutual antagonism of melatonin and dopamine (Besharse et al., 1988). First, melatonin rhythms are not necessary for the production of PER2::luc rhythms in isolated retinas (Ruan et al., 2008), suggesting melatonin does not provide substantial feedback on the inner retinal clock driving PER2::luc. Nonetheless, there is a circadian rhythm of dopamine production in the mouse inner retina that is dependent on the photoreceptor rhythm of the melatonin (Doyle et al., 2002a; Doyle et al., 2002b), and loss of melatonin leads to changes in the circadian pattern of clock gene expression, particularly in the inner retina (Hiragaki et al., 2014). Second, the rodent melatonin rhythm, like that in Xenopus, arises from photoreceptors and is regulated by light, cAMP, and dopamine (Fukuhara et al., 2004; Jackson et al., 2011; Jackson et al., 2009). Analysis of this pathway in mice has led to the demonstration that adenylyl cyclase 1 (ADCY1) itself exhibits a circadian rhythm, and dopamine acts to suppress cAMP levels and melatonin synthesis via D4 (DRD4) receptors. While these findings suggest an important role of the DRD4 receptor in circadian melatonin release by photoreceptors, this is not entirely due to the regulation of melatonin synthesis as significant physiological changes are seen in DRD4-deficient mice that lack the capacity to synthesize melatonin (Nir et al., 2002). Furthermore, the importance of this pathway in retinal physiology is complicated by the finding that a DRD4 ⇒ ADCY1 ⇒ cAMP is also present in retinal ganglion cells, where it affects circadian changes in contrast sensitivity (Hwang et al., 2013).
The Retinal Clock(s) and Expression of “Clock Genes”
The finding of a clock in the mammalian retina was pivotal for the study of retinal rhythmicity because it coincided in time with the initial discovery of the basic molecular components of the mammalian circadian clockwork (Antoch et al., 1997; King et al., 1997; Sun et al., 1997; Tei et al., 1997) and opened the opportunity for the genetic analysis of the retinal oscillator. The early discovery of the period gene (PER) in Drosophila (Konopka and Benzer, 1971) had indicated that circadian mechanisms were genetically controlled, and it was clear that the transcription of target genes such as tryptophan hydroxylase and nocturnin (CCR4NL) was under circadian control in the retina (Green and Besharse, 1994, 1996). The discovery of CLOCK and mammalian homologs of Drosophila PER led quickly to the identification (Bunger et al., 2000; Hogenesch et al., 1998) of a binding partner for CLOCK called BMAL1 (MOP3, ARNTL1) and to the identification (Kume et al., 1999; van der Horst et al., 1999) of the Cryptochromes (Cry1 and Cry2). This, in turn, led to an essential finding relevant to all vertebrate circadian clocks (see Figure 2) that CLOCK/BMAL1 together bind to E-boxes in the promoters of period genes (Per1, Per2, and Per3), Cryptochromes (Cry1 and Cry2), and other “clock-controlled genes” to drive their expression while PER and CRY proteins feed back onto the CLOCK/BMAL1 complex to inhibit transcriptional activity (McMahon et al., 2014; Mohawk et al., 2012; Partch et al., 2014). An additional limb of the clock in which nuclear receptor subfamily 1D1 (Rev-erba, NR1D1) and nuclear orphan receptor A (RORA) control transcription of BMAL1 maintains an antiphasic relationship between BMAL1 and transcripts driven by CLOCK/BMAL1 (Guillaumond et al., 2005; Preitner et al., 2002; Ruan et al., 2006). Many additional gene products, including kinases, ubiquitin ligases, and ubiquitin-specific proteases, modulate the mammalian circadian clockwork through regulation of the activity and degradation of the constituent proteins. However, the essential transcription/translational feedback loops and a set of 6 to 10 core “clock genes” underlie both the central circadian oscillator in the SCN as well as that of the peripheral oscillators, including that in the retina.
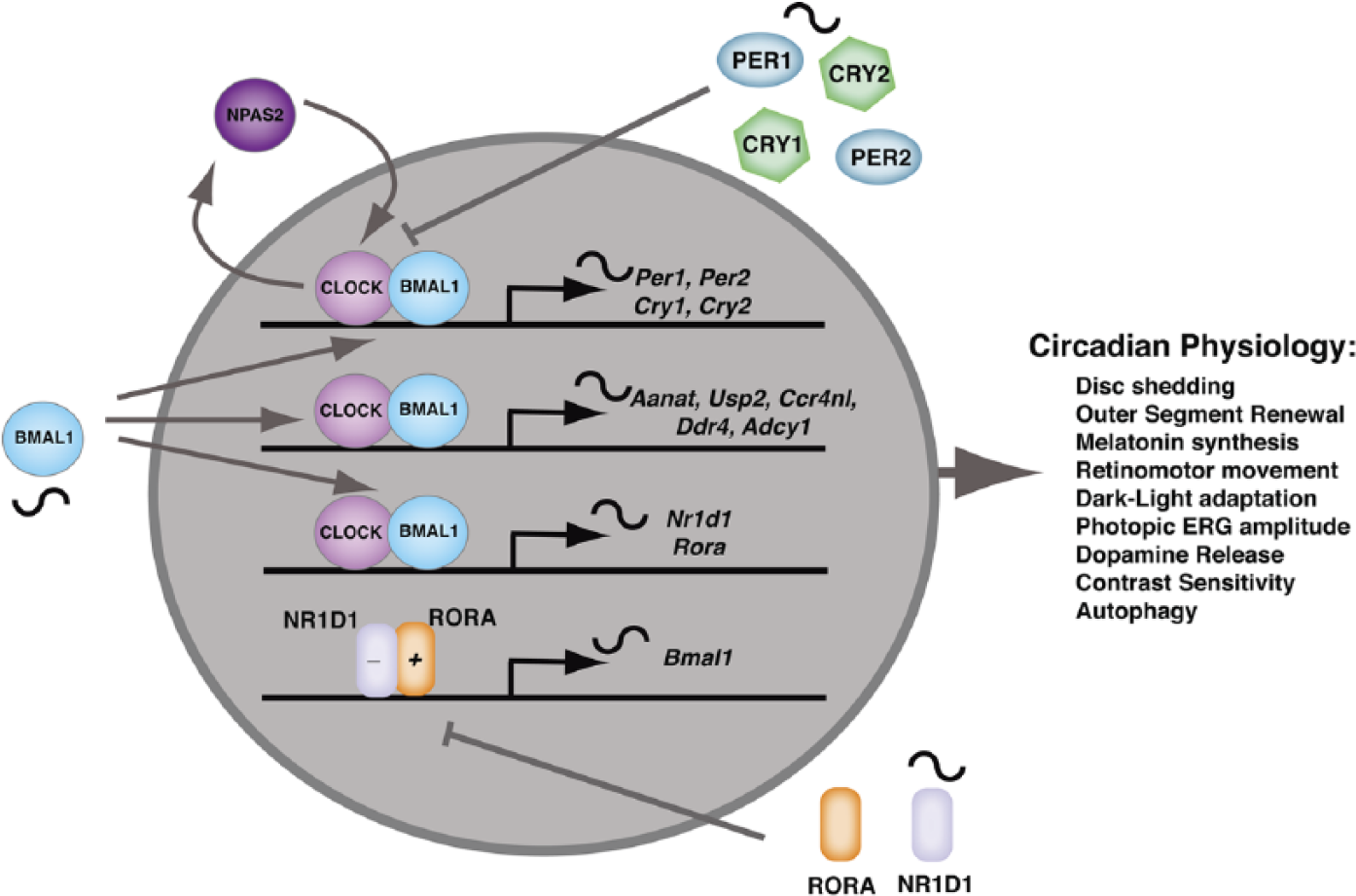
In the retina, the molecular clockwork has features like that in most cells of the body along with retina-specific features. CLOCK/BMAL1 heterodimers drive circadian expression of clock component genes such as Per1, Per2, Cry1, and Cry2. They also drive many “clock-controlled genes” such as Aanat and Usp2 that serve as retina-specific circadian effectors. PER and CRY proteins provide negative feedback to inhibit CLOCK/BMAL1-regulated transcription. CLOCK/BMAL1 also drives expression of NR1D1, a nuclear receptor, which in association with RORA regulates BMAL1 expression in a pattern that is antiphasic to Per and Cry genes as well as “clock-regulated genes.” This regulatory loop reinforces the circadian oscillation initiated through PER and CRY negative feedback. NPAS2 can replace CLOCK within the molecular clockwork. This occurs in the retina within a subset of retinal ganglion cells that are important in the circadian regulation of contrast sensitivity.
The study of “clock gene” expression in the rodent retina has led to the question of whether photoreceptors in mice even have intrinsic clock activity. PER1 expression has been highly associated with cells of the inner retina (Witkovsky et al., 2003), and single-cell polymerase chain reaction (PCR) of steady-state messenger RNA (mRNA) of a set of 6 “core clock genes” showed that all 6 were detectible in 30% of the dopaminergic amacrine cells but in none of the photoreceptors. This provided evidence for functionally autonomous clocks in inner retinal cells but not photoreceptors (Ruan et al., 2006). Although this conclusion has been somewhat controversial (reviewed in McMahon et al., 2014), it is at least consistent with the general finding of very low expression of “clock genes” in rod cells, which has been replicated more recently using immunocytochemistry with the addition that cones, which represent only about 3% of the mouse photoreceptors, do express a full complement of “core clock genes” (Liu et al., 2012). Laser capture microdissection of the rat outer retina, although not at the single-cell resolution, also reveals that most of the “clock genes” are expressed in the outer retina (Tosini et al., 2007). In addition, both rhythmic melatonin release and Per1::luc expression by rat photoreceptor layers (Tosini et al., 2007), using ablation methods similar to those in the original studies on Xenopus (Cahill and Besharse, 1992, 1993), indicate that rat photoreceptor layers exhibit clock activity. Thus, current evidence suggests that in mice, cone cells along with many cell types in the inner retina, including intrinsically photosensitive ganglion cells, express a full complement of “clock genes” (Liu et al., 2012; Ruan et al., 2006), while rod cells, which make up 95% to 97% of the photoreceptors, either lack the molecular machinery for sustained clock activity altogether or express those genes at the limits of detection by current technology. Although the results with rats contrast with those in mice, a simple interpretation is that in rats, expression of “core clock genes” and a Per1::luc rhythm in photoreceptor layers reflects the circadian activity of cones. Finally, circadian clock function in the retina is not limited to neurons, as a recent study has demonstrated that mouse and human Müller glia also express circadian clock genes and express circadian gene expression rhythms in cell culture (Xu et al., 2016).
Although the CLOCK/BMAL1 heterodimer is at the core of the molecular clockwork, NPAS2, a CLOCK homologue, can also dimerize with BMAL1 to drive circadian gene expression (Bertolucci et al., 2008; Reick et al., 2001). Interestingly, one of the places that NPAS2 can substitute for CLOCK (see Figure 2) is in a subset of retinal ganglion cells that are important in the circadian control of contrast sensitivity in mice (Hwang et al., 2013). NPAS2 expression in NPAS2-lacZ reporter mice is found in a subset of BRN3a-positive retinal ganglion cells, and NPAS2 mRNA is expressed rhythmically in ganglion cell layers (GCLs) isolated by laser capture microdissection. Furthermore, daytime contrast sensitivity is significantly reduced in NPAS2–/– mice, suggesting NPAS2-dependent clock activity at the level of ganglion cells plays a major role in the control of contrast sensitivity. Remarkably, similar reduction in daytime contrast sensitivity is seen in mice lacking the D4 dopamine receptor (DRD4) as well as in mice lacking the type 1 Ca2+/calmodulin-sensitive adenylyl cyclase (ADCY1). DRD4 receptors are known to regulate the circadian expression of ADCY1 (Jackson et al., 2011), and the circadian rhythm of ADCY1 expression is abolished in the GCLs of both DRD4 and NPAS2 knockout mice (Hwang et al., 2013). Together these studies identify a subset of retinal cells and a molecular mechanism involving DRD4 ⇒ NPAS2 ⇒ cAMP signaling for the circadian regulation of contrast sensitivity. However, the fact that deletion of DRD4, NPAS2, and ADCY1 only partially reduces the amplitude of the rhythm but does not abolish it implies that other circadian clock mechanisms involving CLOCK/BMAL1 also affect contrast sensitivity.
The principal clock genes have also been identified in Xenopus, and the results largely confirm the early conclusions from melatonin release assays (Cahill and Besharse, 1993). They also validate the idea that “core clock gene” expression can be used to identify cellular oscillators (Liu et al., 2012; Ruan et al., 2006). Homologues of CLOCK, CRY1, CRY2, and PER1 and PER2 (see Figure 2) are all expressed in both rod and cone photoreceptors in Xenopus (Zhu and Green, 2001; Zhu et al., 2000; Zhuang et al., 2000), and both PER genes are also expressed at lower levels in the inner retina and RPE (Zhuang et al., 2000). Furthermore, overexpression of a dominant CLOCK transgene (CLΔQ-GFP), similar to the mouse “CLOCK” mutation (Antoch et al, 1997), specifically in photoreceptors, ablates the melatonin rhythm in eye cups without suppressing total melatonin production (Hayasaka et al., 2002). This suggests a basic photoreceptor clock organization similar to that in the mouse SCN and other peripheral oscillators. Subsequent work using different promoters to drive the CLΔQ-GFP separately in rods and cones reported loss of melatonin rhythms when the rod promoter was used, but results with the cone-specific promoter were more ambiguous in that most eye cups remained rhythmic but with lower amplitude (Hayasaka et al., 2010). This work identifies rods as clock cells but since rods by volume contribute a large portion of photoreceptor mass, despite their near 1:1 ratio in numbers, one cannot rule out the possibility that loss of clock activity in cones was masked by that from rods. Additional, molecular evidence for the X. laevis photoreceptors as clock cells has also come from studies of light and dopamine induction of PER2 mRNA specifically in photoreceptors under conditions that also led to phase shifts of the melatonin rhythm (Besharse et al., 2004; Steenhard and Besharse, 2000). These studies are consistent with the idea that increased PER2 in photoreceptors is part of the transduction pathway for both light- and dopamine-induced phase shifts (see below).
Rodent models have provided an opportunity for systematic analysis of “clock gene” mutations in the retina. The tau mutation in casein kinase epsilon shortens the period of the retinal clock similar to its effect on the central SCN clock and partially disrupts temperature compensation of the retina clock as well (Tosini and Menaker, 1998b). Furthermore, the circadian rhythm of disc shedding persists in free-running hamsters in constant darkness, and its period is shortened in tau mutant animals (Grace et al., 1996). Loss of BMAL1, which has been shown to be essential for circadian rhythm generation (see Figure 2), also disrupts circadian gene expression profiles as well as circadian changes in the B-wave of the electroretinogram (ERG) in mice (Storch et al., 2007). In addition, the conditional deletion of BMAL1 in the retina has essentially the same effects as the global mutation, implying that the retinal clock, rather than another extrinsic clock such as the SCN, autonomously controls retinal rhythms of gene expression and changes in the photopic ERG (Storch et al., 2007). Other clock gene mutations have been analyzed in PER2::luc mice with particularly strong disruptive effects on retinal rhythms in CRY1/CRY2 double mutants, which are arrhythmic (Cameron et al., 2008), as well as PER1, CRY1, and CLOCK mutants (Ruan et al., 2012). A particularly interesting aspect of the latter work is that direct comparison of PER2::luc rhythms in retina and SCN revealed that individual “clock gene” mutations had a much stronger effect in retina than in the SCN. This is thought to be due to a higher degree of coupling of cell-autonomous oscillators in the SCN compared to those in the retina (Ruan et al., 2012). This high degree of coupling of the SCN makes it less susceptible to disruption and particularly suitable as a pacemaker for peripheral oscillators of the circadian system (Liu et al., 2007). The retinal oscillator, which damps quickly and exhibits weak coupling compared to the SCN, appears to be similar to other peripheral oscillators. Although coupling in the retina appears weak compared to the SCN, period dispersion among isolated inner nuclear, ganglion cell, and photoreceptor layers compared to that in intact retinas implies a significant degree of coupling among oscillators within the layers, with a particularly strong link between the ganglion cell layer and inner nuclear layer (Jaeger et al., 2015). The retinal oscillator appears to function independently of the SCN and other peripheral oscillators owing to the fact that its phase can be set directly by the ambient light dark cycle (see below). Nonetheless, enucleation in mice leads to alteration in period and amplitude of behavioral rhythms, suggesting that retina and SCN interact in a way that affects state variables of the pacemaker system (Yamazaki et al., 2002).
Entrainment of Retinal Clocks by Light and Dopamine
Entrainment of the rhythm of serotonin N-actyltransferase (NAT) by light in vitro was one of the criteria in the initial identification of a retinal circadian clock (Besharse and Iuvone, 1983). Contemporaneous work showed that cone retinomotor movement, a circadian mechanism in Xenopus, was regulated by melatonin, cAMP, and dopamine, acting at D2-like receptors (Besharse et al., 1982; Pierce and Besharse, 1985). These effectors were also found to play a role in the regulation of AANAT (Iuvone and Besharse, 1983, 1986a, 1986b). Light and dopamine caused acute light-adaptive cone contraction and AANAT suppression, while cAMP caused dark-adaptive cone elongation and increased AANAT. It was these acute effects that led to the model (Figure 3) in which dopamine and melatonin, under diurnal or circadian control, serve as light- and dark-adaptive effectors in retina and to the concept of reciprocal inhibition in which melatonin suppressed dopamine release and dopamine inhibited melatonin synthesis (Besharse et al., 1988; Pierce and Besharse, 1985). This model has broad relevance and has been elegantly evaluated in pigeon retina, where the antiphasic melatonin and dopamine rhythms are particularly prominent (Adachi et al., 1998). A signature feature of the data supporting this model was that dopamine receptor antagonists blocked acute light effects, indicating that light acted via dopamine through cAMP suppression (see Figure 3). Among the modern D2-like receptors (DRD2, DRD3, and DRD4), it is likely that DRD4 receptors on photoreceptor cells mediate light effects on retinomotor movement, melatonin synthesis, and other features of the pathway within photoreceptors (Hillman et al., 1995; Jackson et al., 2011; Pozdeyev et al., 2008).
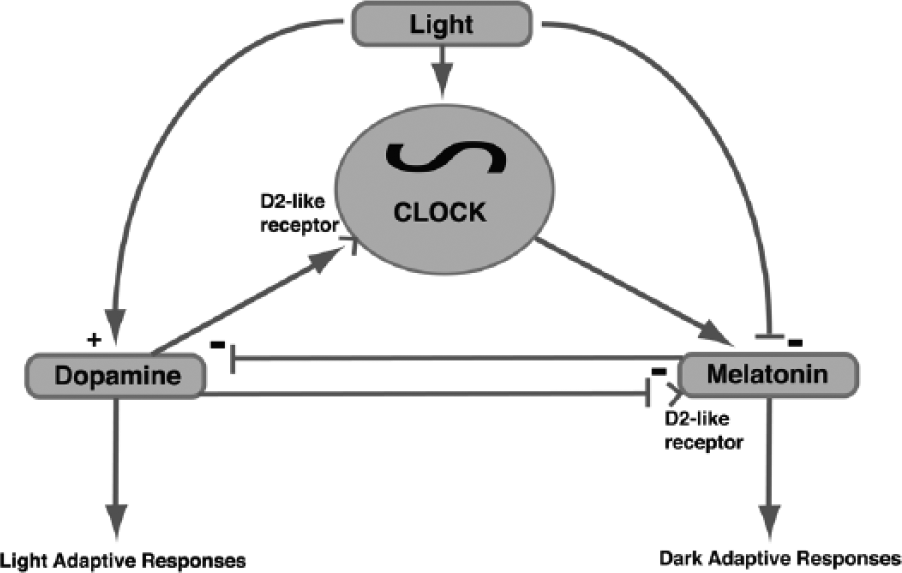
In the Xenopus laevis retina, there is a complex interplay of the light-dark cycle, the retinal circadian clock, and dopamine and melatonin, which are regarded as light- and dark-adaptive effectors, respectively (redrawn with modifications and updated from Figure 1 in Besharse and Green, 1996). Light directly regulates the phase of a retinal clock and also directly upregulates dopamine release and downregulates melatonin synthesis. Dopamine also modulates the phase of the clock via D2-like receptors, which in turn drives a rhythm of melatonin synthesis and release. There is also mutual antagonism in that melatonin inhibits dopamine release in circadian night, and dopamine via D2-like receptors inhibits melatonin synthesis in the day. Of the D2-like receptor types in mice, DRD4 plays a prominent role. However, dopamine phase shifts the PER2::luc oscillator in mice via a DRD1 instead of a D2-like receptor.
Light and dopamine were also found to phase shift the retinal clock in Xenopus driving melatonin synthesis with early evening delays and late-night advances (Cahill and Besharse, 1991). Since this effect was observed in photoreceptor layers, the results demonstrated the existence of a light- and dopamine-sensitive clock or clocks within photoreceptors (Cahill and Besharse, 1993). It is important to distinguish between acute effects on a circadian rhythm such a melatonin synthesis (i.e., inhibition) or cone movement (i.e., contraction) and phase shifts on the underlying clock because the details of the signaling pathways are not the same (Figure 4). In contrast to the acute effects, dopamine antagonists did not block the effect of light on phase shifts, indicating the existence of a dopamine-independent, light-signaling pathway to a photoreceptor clock (Cahill and Besharse, 1991). Subsequent studies using photoreceptor layers demonstrated that cAMP analogues also caused phase shifts that were the inverse (dark phase shifts) of those caused by light and dopamine (Hasegawa and Cahill, 1998). Furthermore, dopamine and quinpirole, a DRD2 agonist, suppress cAMP and phase shift the oscillator in the photoreceptor layers, while light causes phase shifts without suppressing cAMP (Hasegawa and Cahill, 1999). This clarifies the original finding that a D2-like antagonist does not block phase shifts and demonstrates a dopamine and cAMP-independent signaling pathway for light. In a parallel set of studies, it was found that both light and a D2-like agonist increased PER2 expression in photoreceptors (Besharse et al., 2004; Steenhard and Besharse, 2000). As with phase shifts, light induction of PER2 was not blocked by a D2-like antagonist (Steenhard and Besharse, 2000). Since increases in PER2 are thought to play a role in phase shifts at the level of the clockwork itself, the data suggest that the dopamine and light pathways converge at the level of molecular clockwork (see Figure 4).
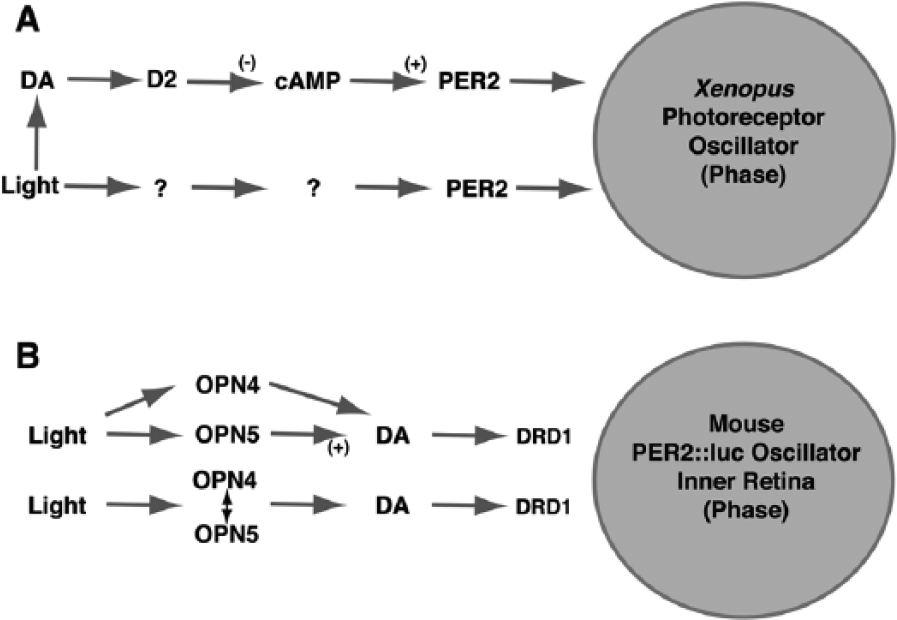
There are similarities and significant differences in light- and dopamine-mediated phase regulation of the retinal clock in Xenopus laevis driving melatonin release and that in mice driving PER2::luc expression. (A) In Xenopus, dopamine acting at D2-like receptors suppresses cAMP and increases PER2 expression in a pathway leading to both phase advances and phase delays. However, there is a dopamine and cAMP-independent pathway through which light directly increases PER2 and phase shifts the clock. (B) In mice, dopamine acting at D1 receptors (DRD1) phase shifts the clock. In addition, dopamine mediates the phasing effect of light as DRD1 antagonists block the phasing effect of light. There is evidence that retrograde signaling through melanopsin (OPN4) in intrinsically photosensitive retinal ganglion cells (ipRGCs) modulates the effects of light on dopamine release from amacrine cells. However, OPN4 is not required for light-induced phasing of the PER2::luc rhythm, and neuropsin (OPN5), also in a set retinal ganglion cells, is required. Current data imply that retrograde signaling from ipRGCs phase shifts the clock through a pathway involving dopamine, but it is unclear whether OPN5 is in a class of cells that is distinct from those expressing OPN4.
Despite involvement of dopamine in both Xenopus and mice, there are major differences in entrainment of in vitro PER2::luc of rhythms in mice compared to the melatonin rhythms in Xenopus that likely reflect differences in the cells exhibiting circadian oscillations in the 2 systems. While the melatonin rhythm is present in photoreceptors (above), inner retinal bioluminescence dominates the PER2:luc rhythms in mice (Ruan et al., 2008). Nonetheless, dopamine plays a dominant role in phase shifts of the retinal PER2::luc rhythm but in this case via DRD1 receptors (Figure 4). Furthermore, dopamine appears to contribute to light-induced phase shifts as they are reduced by DRD1 antagonists (Figure 4). The phase-shifting effects of dopamine on the PER2::luc rhythm also contrasts with dopamine’s direct regulatory drive of circadian rhythms of ADCY1, cAMP, and melatonin synthesis in photoreceptors (Fukuhara et al., 2004; Jackson et al., 2011; Jackson et al., 2009). Here the rhythms appear to be driven tonically by dopamine occupancy of DRD4 receptors. Thus, while the photoreceptor rhythms are robust, they may be driven by a circadian system in the inner retina via dopamine. From a functional point of view, circadian drive from a clock in the inner retina would be equivalent to cell-autonomous circadian drive in photoreceptors.
Light-induced phase shifts in photoreceptor layers from either Xenopus or rat are presumed to involve phototransduction events within rods and cones because other cell types were removed in those preparations, and in Xenopus, the phase shifts occur independently of dopamine. However, light-induced phase shifts of the PER2::luc rhythm in mice are mediated by dopamine (Ruan et al., 2008), and mechanisms that regulate light responses of dopaminergic amacrine cells are likely relevant in phase shifting. Dopaminergic neurons in isolated retina respond with transient (Dkhissi-Benyahya et al., 2013) or sustained electrical activity (Zhang et al., 2012). The sustained responses remain in retinas lacking rods and when bipolar cell activity is blocked, but under these conditions, the transient responses are blocked (Zhang et al., 2008). Furthermore, mice lacking melanopsin (OPN4), which is found in intrinsically photosensitive retinal ganglion cells (ipRGCs), also lacked the sustained responses (Zhang et al., 2012). This has led to the idea that melanopsin-containing ipRGCs provide a retrograde pathway in the retina to control dopaminergic cells (see Figure 4). Indeed, loss of melanopsin leads to loss in the rhythms of some clock genes in the outer and inner retina, blunting of rhythmicity, and light induction of tyrosine hydroxylase, a key synthetic enzyme for dopamine (Dkhissi-Benyahya et al., 2013). Although loss of melanopsin has been shown to affect diurnal and circadian patterns in the cone ERG (Barnard 2006), it is not required for light-induced phase shifting of the PER2::luc rhythm in mice (Buhr et al., 2015). Instead, recent work shows that an additional “orphan opsin” called neuropsin (OPN5) is required (Buhr et al., 2015). Early results indicate that neuropsin is also localized to ganglion cells, and OPN5–/– mice lack the ability to phase shift in response to light (see Figure 4). This implies that a retrograde pathway is indeed required for phase shifting of the PER2::luc rhythm in mice but raises important questions about the relationship of neuropsin and melanopsin expression in ipRGCs and the impact of loss of neuropsin on dopaminergic amacrine cells.
While it is clear that light directly phase shifts retinal clocks in vitro, it has been reported that phase shifts of the disc-shedding rhythm in rats require an intact optic nerve (Teirstein et al., 1980). Consistent with a role for a local retinal clock, eye patches were used to establish distinct shedding rhythms in the 2 eyes of single animals, but the expected peaks of disc shedding were not phase shifted after optic nerve transection. This experiment has been widely interpreted to mean that retrograde signaling from the SCN or another brain region via the optic nerve is required for entrainment of the rat disc-shedding rhythm. Nonetheless, a role for the SCN pacemaker seems unlikely since entrainment of the morning peak of disc shedding is maintained in rats long after SCN lesion (Terman et al., 1993). Although it is possible that retrograde signaling from another brain region is involved in retinal entrainment, an alternative interpretation is that optic nerve lesions disrupt the behavior of melanopsin (Zhang et al., 2012) and neuropsin (Buhr et al., 2015) containing ipRGCs that are required for local retinal entrainment.
Relationship of the Retinal Clock to the SCN Pacemaker
Today, the general consensus in mammals is that the SCN is a central pacemaker in the circadian system that receives light signals from retinal ganglion cells and in turn coordinates phasing of a plethora of light-insensitive peripheral oscillators (Balsalobre et al., 1998; Nagoshi et al., 2004; Pando et al., 2001; Schibler and Sassone-Corsi, 2002; Yoo et al., 2004). While the concept of a local ocular clock initially seemed somewhat at odds with the primacy of a central pacemaker in the mammalian model, the data on retinal clocks in the 1980s were from nonmammalian vertebrates in which a broader pacemaker role for tissues such as the pineal gland or retina seemed plausible. For example, in the Japanese quail, a prominent ocular clock driving melatonin release to the blood is at least part of the broader circadian pacemaker system (Steele et al., 2003; Underwood et al., 1990). In avian systems, unlike mice, melatonin seems necessary for sustained activity rhythms, and in Japanese quail, a substantial portion of the serum melatonin comes from the eye, and the animals become arrhythmic when the eyes are removed. In addition, light-sensitive circadian oscillation is even more widely dispersed in both Drosophila (Giebultowicz et al., 2000; Plautz et al., 1997) and zebrafish (Kaneko et al., 2006). In zebrafish, even tissue-cultured cell lines exhibit light-sensitive circadian clocks (Cermakian et al., 2002; Kaneko et al., 2006; Pando et al., 2001). Such data suggest that among vertebrates, circadian organization evolved from a system of widely dispersed, photosensitive clocks with phasing controlled by the light-dark cycle to the mammalian hierarchical system with a strong central pacemaker and a system of light-insensitive clocks. Even in zebrafish, however, there is likely a need for central coordination within the intact organism because the individual light-sensitive clocks in peripheral tissues exhibit an array of diverse properties (Kaneko et al., 2006).
Despite the plausibility of independent pacemaking activity of a retinal clock as demonstrated in Japanese quail, this does not seem to be the case in the retina of either the X. laevis or mice. In X. laevis, for example, blinding does not affect circadian activity rhythms (Harada et al., 1998). Furthermore, melatonin synthesis by photoreceptors is locally limited because it is rapidly degraded to 5-methoxytryamine (Cahill and Besharse, 1989), and its synthesis is rate limited in capacity at the first enzymatic step, tryptophan hydroxylase (Cahill and Besharse, 1990). Thus, even the detection of robust rhythms of melatonin in the Xenopus flow-through culture required supplementation of the medium with hydroxytryptophan. It has been argued that the design of the retinal melatonin system in Xenopus is to limit its availability to the eye, and it is unlikely that the eye in Xenopus could serve as an endocrine organ secreting melatonin. In mice, on the other hand, behavioral activity driven by the SCN does not require melatonin as most strains of mice used for circadian studies lack melatonin synthetic capacity (Ebihara et al., 1986; Goto et al., 1989). Virtually all of the data support the idea that the retinal oscillator functions locally. We know that the SCN requires the retina for its photic input, and it is possible this input is gated by a retinal clock, but the retinal-hypothalamic tract seems to be designed primarily to ensure adjustment of the circadian phase of the SCN. Since the retina can be synchronized by daily light cycles independent of the phase of the SCN (Buhr et al., 2015), it is likely that the retinal clock functions to control local retinal physiology independently.
There is evidence that retinal circadian signaling to the brain may occur without light-dark stimulation. In constant darkness, subregions of the SCN clock nuclei express retina-driven circadian rhythms in the phosphorylation of the extracellular regulated kinase, ERK, a molecule normally activated by retinal ganglion cell input to the SCN during light resetting of the clock (Lee et al., 2003). SCN ERK activation rhythms in constant darkness are ablated upon enucleation, suggesting that they originate in the retina, rather than in the SCN itself. In addition, retinal signals in darkness also contribute to the maturation and functional organization of the brain’s biological clock, as enucleation in darkness during the first few weeks of life causes changes in the free-running period of locomotor rhythms in hamsters (Yamazaki et al., 2002). Interestingly, ipRGCs have been shown to express clock genes (Liu et al., 2012), and their light responses are modulated with a circadian rhythm and by dopamine (Van Hook et al., 2012; Weng et al., 2009), suggesting the possibility that the retina-driven rhythms observed in the SCN may originate in the ipRGCs themselves or in retinal neurons with synaptic input to the melanopsin-expressing ipRGCs (e.g., dopamine amacrine cells). In addition, it has been also reported that ipRGCs are the first cells in the retina to respond to light during development (Sekaran et al., 2005), that light pulses can induce c-fos in the ganglion cells at postnatal day 1 (Mateju et al., 2010), and that light transduction may occur in utero by day 17 of gestation in the mouse (Rao et al., 2013). Therefore, ipRGGs may play a role in the entrainment of the circadian rhythm in the perinatal period of development in rodents.
Visual Function and Retinal Clocks
Why did retinal clocks evolve and what is their global effect on visual function? One view is that a clock permits anticipation of the regular alternation of photopic and scotopic conditions of the daily light-dark cycle (as surely as night follows day) by presetting the retina to partially light and dark adapted states. Indeed, this provides an explanation for circadian retinomotor movements (Welsh and Osborn, 1937), which preposition the phototransduction machinery of rods and cones for optimum photon absorbance. The size and number of cone synaptic ribbons also fluctuate in a circadian pattern with increases in daytime in at least some teleost species (Wagner, 1973, 1975; Wagner and Ali, 1977). This plasticity parallels function of the cone visual system and is regulated by dopamine (Weiler et al., 1988). A more extreme example (Emran et al., 2010) is the case of young zebrafish fry in which cone-mediated neurotransmission is lost entirely at night due to rhythmic nighttime disassembly of cone synaptic ribbons (Emran et al., 2010). These newly hatched fish are functionally blind at night during the first few days of free-swimming life as their rod photoreceptors have not fully developed. Regardless of whether retinomotor movements or ribbon reorganization is involved, the retinal circadian clock in a number of vertebrate species, including fish, amphibians, birds, and mammals, has been shown to drive shifts in the rod-cone balance such that cone vision is favored in the day and rod vision is favored at night. Furthermore, circadian dopaminergic regulation of rod-cone coupling is a key mechanism (Krizaj et al., 1998; Li et al., 2013; Manglapus et al., 1999; Ribelayga et al., 2008; Wang and Mangel, 1996). At night, rod-cone coupling is strong, allowing cones to receive rod signals in very dim light (Barnard et al., 2006), but in the day, dopamine acting at D2-like receptors or DRD4 receptors reduces this coupling to promote photopic vision.
The impact of the retinal circadian clock is also seen in the light-adapted, photopic ERG (Cameron et al., 2008; Jackson et al., 2012; Storch et al., 2007), which have higher amplitudes during the subjective day than the subjective night. These circadian changes are lost when the retinal circadian clock is ablated using retina-specific BMAL1 knockout mice (Storch et al., 2007). In those studies, the clock in the SCN remained intact, showing unequivocally that circadian rhythmicity in the photopic ERG depends on the circadian clocks located within the retina. The use of CRY1 and CRY2 double knockout mice as an alternate strategy for clock ablation gave a similar loss of rhythmicity on the photopic ERG. Melatonin signaling through the MT1 receptor is necessary to maintain rhythmicity in the photopic ERG, although not the dopamine rhythm (Sengupta et al., 2011). Interestingly, melanopsin (OPN4) is also required for maintenance of rhythms in the photopic ERG as OPN4–/– mice lack these changes and exhibit photopic ERGs resembling a midpoint between “daytime” and “nighttime” at all times of day (Barnard et al., 2006). This suggests that melanopsin and ipRGCs are required to maintain ERG rhythmicity under circadian conditions. A possible explanation is that ipRGCs are required for proper entrainment of retinal clocks and that the individual cellular oscillators are desynchronized in OPN4–/– mice. However, this idea is not consistent with more recent in vitro data showing that neuropsin (OPN5) is required for entrainment of the retinal PER2::luc rhythm while melanopsin (OPN4) is dispensable (Buhr et al., 2015). Understanding the role of ipRGCs in retinal and SCN entrainment will require better insight into the different roles of these 2 photopigments.
Genetic depletion of retinal dopamine by retina-specific knockout of the key dopamine synthesizing enzyme, tyrosine hydroxylase (rTHKO), strongly reinforces the view that dopamine is a key circadian output signal of the retinal clock to reconfigure retinal circuits (Jackson et al., 2012). This is most readily seen in measurements of the photopic ERG and in behavioral contrast sensitivity, which is also clock regulated (Hwang et al., 2013). Depletion of retinal dopamine blunted the circadian rhythm in the light-adapted ERG, a cone-driven response, and at the behavioral level reduced daytime contrast sensitivity. Both of these effects are mediated by DRD4 receptors as they are rescued by a specific DRD4 agonist and are phenocopied in DRD4–/– mice (Hwang et al., 2013). Dopamine is important for daytime vision, and the rTHKO mice appear to be locked in the nighttime state with low-amplitude photopic ERGs and reduced contrast sensitivity. The results with rTHKO mice are reminiscent of human Parkinsonian degeneration of dopaminergic neurons in humans, where patients exhibit deficits in the ERG and in contrast sensitivity. It seems likely that at least some of the visual symptoms of this neurodegenerative disease are due to loss of retinal dopamine (Haug et al., 1994; Ikeda et al., 1994; Ingster-Moati et al., 1996).
Overall retinal sensitivity to photopic light, contrast sensitivity, and the function of specific classes of ganglion cells is under circadian control via dopamine signaling. This occurs either through elevation of dopamine secretion driven by the retinal circadian clock during circadian day or as a result of diurnal or circadian rhythms in DRD4 expression (Bai et al., 2008; Jackson et al., 2011; Klitten et al., 2008; Storch et al., 2007). In the light-dark cycles experienced in the natural environment, light-induced dopamine release would further enhance dopamine signaling during the day. While most functional studies, including those on X. laevis and the rTHKO mouse noted above, have identified effects primarily on cone-mediated vision, this is likely an oversimplification as dopamine affects the retina through multiple receptors. As a case in point, dopamine also acts at D1 receptors to extend the operating range of rod signaling in the presence of background illumination (Herrmann et al., 2011). It also acts through D2-like receptors to regulate rod electrical coupling leading to increased signal-to-noise ratios for dim light responses at night (Jin et al., 2015). Thus, from the most general perspective, dopamine signaling acts to enhance the performance of both the rod and cone systems in the presence of background lights appropriate for their operating ranges.
In general, circadian dopamine signaling through DRD4 or D2-family receptors can be distinguished from light-adaptive dopamine signaling, which is mediated through DRD1 receptors (Jackson et al., 2012). For example, in rTHKO mice with retina-specific depletion of dopamine, reduced photopic ERG amplitude and contrast sensitivity, which are under circadian control, are restored by DRD4 agonists, while the noncircadian effects on visual acuity are restored by DRD1 agonists (Jackson et al., 2012). Similarly, circadian changes in rod-cone coupling are mediated by D2-family receptors while light-driven changes in horizontal cell coupling are mediated by DRD1 receptors (Ribelayga and Mangel, 2003). Tracer labeling in the mouse retina revealed a circadian rhythm in the extent and strength of rod-cone coupling with weak coupling during the day and strong coupling at night. Dopamine acting at D2-like receptors negatively regulates coupling during the day (Ribelayga et al., 2008) while horizontal cell coupling, which exhibits a constant, noncircadian profile, is greatly reduced by light adaptation mediated by DRD1 receptors (Ribelayga and Mangel, 2003). Key elements of this dichotomy in which D2-family receptors, particularly DRD4 receptors, mediate a “circadian signaling channel” for dopamine in the retina while DRD1 receptors mediate a “light-adaptation signaling channel” have been reported widely among vertebrate species, including X. laevis (Pierce and Besharse, 1985; Witkovsky et al., 1988), Japanese quail (Manglapus et al., 1999), and teleosts (Dearry and Burnside, 1986; Witkovsky and Dearry, 1992). The involvement of D2-family receptors in “circadian signaling” likely reflects their very high affinity for dopamine diffusing at low concentration in the retinal extracellular space (Witkovsky et al., 1993). In addition, a circadian clock drives DRD4 but not DRD1 expression in mice, further enhancing the circadian features of the DRD4 signaling channel (Bai et al., 2008; Jackson et al., 2011; Klitten et al., 2008).
Circadian Disc-Shedding Rhythms
While the realization that photoreceptor outer segment turnover is under circadian control led to a surge in studies of ocular clocks, the molecular details linking disc shedding and clock activity until recently have remained rather murky. Disc shedding and phagocytosis by RPE, which has been investigated most extensively in rods, can occur constitutively throughout the day as well as in a peak after light onset each day. The peak persists with damp-ing in constant darkness but accounts for only a portion of daily turnover in rodents (LaVail, 1976). As a consequence, loss of clock activity or the early morning burst of disc shedding has little immediate impact on photoreceptor morphology (Storch et al., 2007), although it can lead to age-related photoreceptor degeneration (Nandrot et al., 2004). Melatonin can activate (Besharse and Dunis, 1983; Besharse et al., 1982) and dopamine and cAMP can inhibit (Besharse et al., 1982; Pierce and Besharse, 1986; Reme et al., 1983) disc shedding, suggesting that both can play a role in circadian regulation. Nonetheless, it was recognized early that the melatonin/dopamine regulatory system fails to explain key features of circadian disc shedding (Besharse et al., 1988). A possible explanation is that disc shedding involves the coordinated interaction of the photoreceptor outer segment and the RPE (Besharse and Defoe, 1998) and that additional clock mechanisms within the RPE are involved. Consistent with this, it has recently been shown using explants from PER2::luc mice that an independent clock operates within the RPE (Baba et al., 2010). Furthermore, direct analysis of the disc phagocytic mechanism within RPE has shown that knockout of beta 5 integrin (ITBG5) or its ligand, milk fat globule-EGF factor 8 (MFGE8), in mice results in loss of the early morning burst in disc shedding and phagocytosis (Nandrot et al., 2007; Nandrot and Finnemann, 2008; Nandrot et al., 2004). Disc shedding continues in a constitutive manner in ITBG5–/– mice and sustains relatively normal function in the short term, but these mice eventually exhibit photoreceptor degeneration. Interestingly, the outer segment tips of rods also exhibit an early morning peak of surface-exposed phosphatidyl serine, a phagocytic signal, that appears to anticipate the early burst of shedding (Ruggiero et al., 2012). This phagocytic signal is attenuated in ITBG5- and MFGE8-deficient mice, suggesting that the RPE plays a major role in controlling changes at the photoreceptor tip. Determining the connection of ITBG5 signaling and the appearance of outer segment surface phosphatidylserine to the circadian clockwork in either retina or RPE will greatly enhance our understanding circadian disc shedding.
Regulation of Gene Expression by the Retinal Circadian Clock
The molecular circadian clockwork functions to drive a multitude of clock-controlled genes that in turn drive circadian physiology. Early work in the retina demonstrated light-induced as well as circadian clock control of opsin and other gene expression in the photoreceptors of multiple vertebrate species (Farber et al., 1991; Green and Besharse, 1994, 1996; Korenbrot and Fernald, 1989; Pierce et al., 1993). In particular, those early studies demonstrated that circadian melatonin synthesis began with circadian changes in mRNA levels of 2 of its enzymes, tryptophan hydroxylase (Green and Besharse, 1994; Green et al., 1995) and AANAT (Bernard et al., 1997; Chong et al., 1998). Those early studies also provided the first direct evidence that circadian changes in steady-state mRNA levels can be driven at the transcriptional level (Green and Besharse, 1994, 1996). Recently, gene arrays have been used on a genomic scale to define a steady-state circadian transcriptome of the mouse retina (Storch et al., 2007). Here it was demonstrated that about 3000 genes were rhythmic in a light-dark cycle and about 300 of those continued to cycle in constant darkness. The data show that, as is true in other peripheral oscillators (Panda et al., 2002), a large portion of the transcriptome is under circadian control. This includes genes from classes that contribute to basic neural and cellular functions such as synaptic transmission, metabolism, photoreceptor signaling, intercellular communication, regulation of the cytoskeleton, and chromatin remodeling. Interestingly, the retinal molecular clockwork also contributes to the regulation of those genes that were rhythmic only in a light-dark cycle as ablation of the clock by knockout of BMAL1 also disrupted rhythmic gene expression in light-dark cycles (Storch et al., 2007). More recently, RNAseq analysis at 2 critical periods during the circadian cycle has been used to identify genes that are involved in the circadian regulation of the photoreceptor phagocytosis in retinal pigment epithelium (Mustafi et al., 2013). This study identified 365 cycling protein-coding transcripts. Interestingly, several of these oscillating proteins implicate the phosphoinositide lipid signaling pathway in the regulation of photoreceptor phagocytosis (Mustafi et al., 2013). Finally, a circadian proteomic analysis of the mouse retina has identified several proteins with circadian patterns of expression that function in vesicular transport, calcium binding, protein degradation, metabolism, RNA binding, and protein folding (Tsuji et al., 2007), suggesting that disrupted transcriptional and posttranscriptional control by the retinal circadian clock is likely to be involved in the pathogenesis of eye diseases.
Multiple Ocular Clocks
The emphasis so far has been on the endogenous system of clocks within the retina. Yet, the fundamental finding that even fibroblasts in culture exhibit clock activity (Balsalobre et al., 1998; Nagoshi et al., 2004) and that multiple isolated tissues from PER2::luc mice exhibit sustained rhythms in culture (Yoo et al., 2004) suggests that the multiple tissue components within the eye are composed of cell-autonomous clocks. This idea has been confirmed for the retina (Ruan et al., 2008), cornea (Baba et al., 2015; Yoo et al., 2004), and retinal pigment epithelium (Baba et al., 2010) using PER2::luc mice. This more general property is illustrated in Figure 5, which shows self-sustained circadian oscillation of PER2::luc in the retina, RPE-choroid, ciliary body, iris, and cornea. An important point from this comparison is that both the phase and the period of these self-sustained oscillations were different despite the fact that the tissues were isolated at the same time of day. This finding indicates that the different cellular clocks within these tissues exhibit subtly different properties in vitro, which likely reflect differences in clock organization or in its regulation. In this regard, it is interesting that the corneal PER2::luc rhythm can be phase-shifted by light and that this requires neuropsin (Buhr et al., 2015).
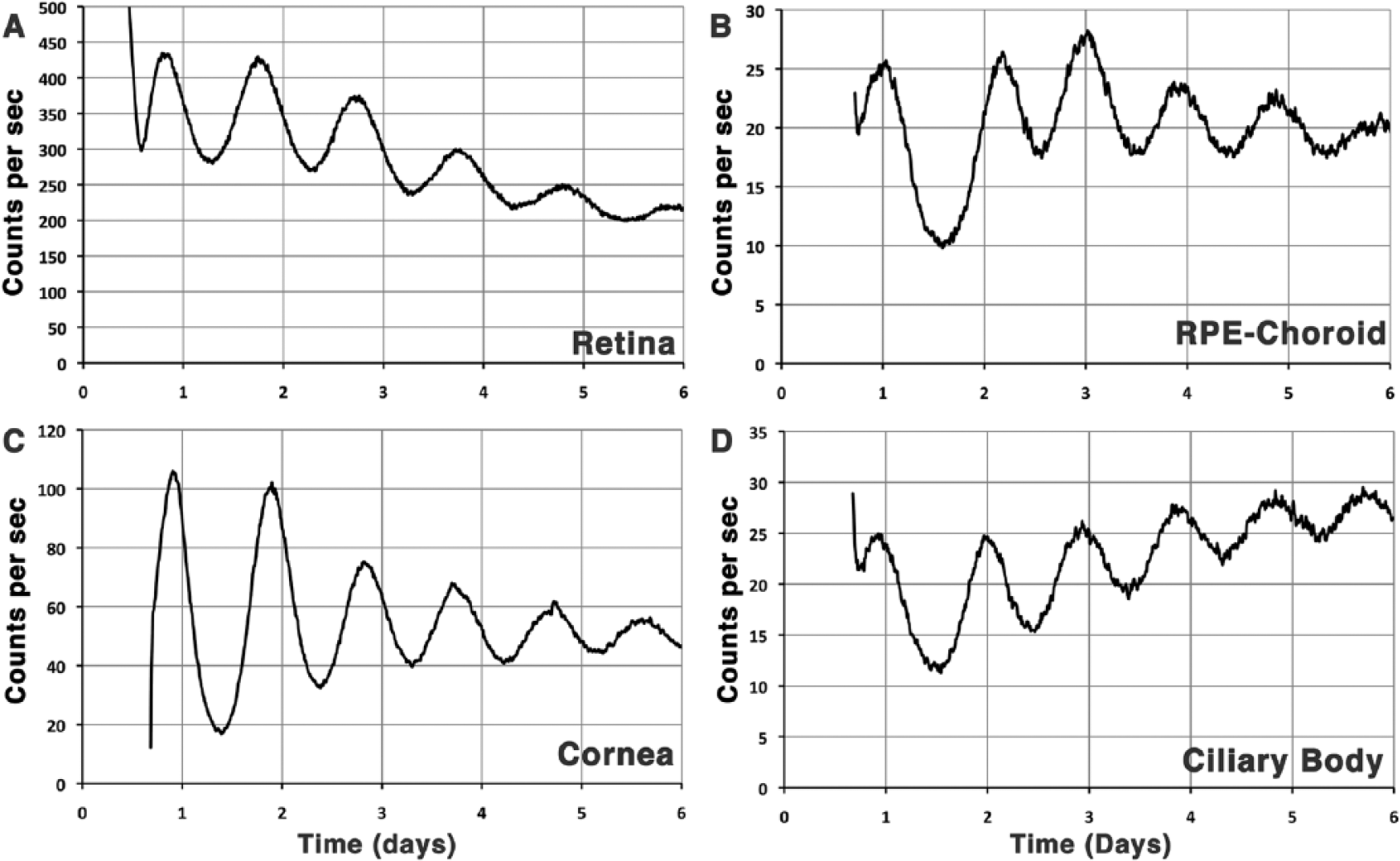
The eye is composed of multiple oscillators or clocks. The retina (A), retinal pigment epithelium (RPE)–choroid (B), cornea (C), and ciliary body (D) dissected from PER2::luc mouse eyes on day 1 and cultured in darkness through 6 days in a Lumicycle device all exhibit sustained rhythms of luciferase activity with a period less than 24 h and only moderate damping. The highly sensitive photomultiplier tubes permit detection of rhythmicity in each explant despite large differences in total photon counts. The plots are from new data from the senior author’s laboratory using methods reported previously (Baba et al., 2010; Ruan et al., 2008; Yoo et al., 2004). Although not surprising, this is the first report of such rhythmicity in the ciliary body.
Another implication of the concept that the eye is a multioscillator system relates to downstream ocular physiology that is under circadian control. For example, it has long been known that the corneal epithelium exhibits high-amplitude rhythms of mitotic activity (Scheving and Pauly, 1974; Scheving et al., 1974) that are likely controlled locally. The same can be said for the ciliary body and the findings that intraocular pressure, aqueous humor flow, and aqueous cAMP levels all exhibit circadian rhythms (Boyd and McLeod, 1964; Rowland et al., 1986; Smith and Gregory, 1989) that are regulated through a noradrenergic system (Eakins and Eakins, 1964; Gianesini et al., 2016; Yoshitomi and Gregory, 1991). It is possible, for example, that such regulation involves synchronization of a local clock in the ciliary body that in turn regulates secretion of aqueous humor.
Retinal Clock and Eye Disease
The widespread role of circadian clocks in retinal signaling, metabolism, and gene expression implies that altered clock function would likely affect retinal disease and pathology. Loss of the daily burst of disc shedding in ITGB5–/– mice is accompanied by age-dependent photoreceptor degeneration (Nandrot et al., 2007). However, this degeneration likely involves factors in addition to loss of circadian disc shedding, because deficiency in MFGE8, the ITGB5 ligand, also results in loss of the diurnal peak in disc shedding but does not result in retinal degeneration (Nandrot et al., 2007; Nandrot and Finnemann, 2008). Photoreceptors are more vulnerable to light damage in subjective night (Organisciak et al., 2000; Vaughan et al., 2002), and this likely reflects circadian changes in the expression and distribution of crystallins that serve a chaperone function in the photosensitive membrane system (Organisciak et al., 2011). In addition, melatonin, which is normally high at night, potentiates light damage (Wiechmann and O’Steen, 1992), and its antagonist, luzindole, is neuroprotective for photoreceptors (Sugawara et al., 1998). Furthermore, loss of both the MT1 and MT2 melatonin receptors (MTNR1A and MTNR1B) results in attrition of cones in older mice (Gianesini et al., 2016). In contrast to its impact on photoreceptors, melatonin and its type 1 receptor (MT1, MTNR1A) are important in maintaining ganglion cells as loss of either melatonin or MT1 receptors results in an age-related decline in ganglion cell numbers and an increase in intraocular pressure (Alcantara-Contreras et al., 2011; Baba et al., 2009). N-acetylserotonin (NAS), melatonin’s immediate precursor, is also high at night and strongly neuroprotective in the retina (Iuvone et al., 2014; Tosini et al., 2012). This neuroprotective effect appears to operate through the type 2 neurotrophic kinase receptor (Trkb, NTRK2) and brain-derived neurotrophic factor (BDNF). The TrkB receptor shows circadian activation in mice capable of making NAS, and TrkB is rapidly activated by NAS in a BDNF-independent manner (Iuvone et al., 2014; Jang et al., 2010).
The retinal clock and its outputs play a major role in the regulation of eye growth and refractive error as disruption of retinal dopamine rhythms causes myopic changes in chicken eyes (Bartmann et al., 1994; Feldkaemper et al., 1999; Guo et al., 1995; Rohrer et al., 1993; Weiss and Schaeffel, 1993), and similar dopamine- and circadian clock–dependent eye growth regulatory mechanisms likely operate in the mammalian retina (Stone et al., 2013). Furthermore, form deprivation, which induces myopia, decreases retinal dopamine (Stone et al., 1989) and disrupts daily rhythms of eye growth (Weiss and Schaeffel, 1993). Gene expression profiling of chicken retina/RPE has also shown altered circadian clock gene and melatonin receptor expression in lens-induced myopia (Stone et al., 2011).
Emerging evidence also suggests that the clock, and Period genes in particular, may play a role in the regulation of vascularization signals in the retina and the specification of photoreceptors. For example, PER2 mutant mice exhibit age-related changes in expression of both endothelial nitric oxide synthase (ENOS) and vascular endothelial growth factor (VEGF), increased vascular damage, and increased vascular permeability (Bhatwadekar et al., 2013). In addition, disruption of melanopsin signaling in OPN4–/– mice affects developmental vascularization of the retina (Rao et al., 2013). Here, light signaling via melanopsin is required for both breakdown of the fetal hyaloid vasculature and proper patterning of the retinal vasculature and may be linked to early PER2 expression in the postnatal retina (Bhatwadekar et al., 2013). In addition, Per1/Per2 knockout mice exhibit sparse retinal deformations and reduced expression of blue cone opsin (Ait-Hmyed et al., 2013). Diabetic retinopathy in experimental models is strongly correlated with suppressed circadian clock gene expression, including strong downregulation of PER2 and BMAL1, suggesting that the clockwork may be an important target for therapeutic intervention in proliferative vascular disease (Busik et al., 2009). Furthermore, decreased retinal dopamine contributes to the reduced spatial frequency and contrast sensitivity seen in a rat model of type 1 diabetes, as visual function improves on treatment with the dopamine precursor, L-DOPA, and by either dopamine DRD1 or DRD4 agonists (Aung et al., 2014).
Summary
Circadian clocks in the retina exhibit many features that are common to peripheral oscillators more generally. For example, retinal clocks control gene expression and key features of retinal function that optimize vision for conditions that vary over the course of a 24-h cycle. In addition, retinal clocks exhibit relative rapid damping compared to the pacemaker in the SCN, which likely reflects relatively weak coupling among cellular oscillators. Unlike most peripheral clocks in mammals, however, the phasing of the retina and the cornea can be controlled directly by light. In addition, light detected in the retina controls the phase of the SCN via light-sensitive ganglion cells that project to the SCN. These light-sensitive ganglion cells appear to provide both anterograde signals to control phasing of the SCN pacemaker as well as retrograde signals to control retinal phasing. This sets the retina apart from other mammalian peripheral oscillators that are light insensitive and require synchronizing signals coordinated by the SCN.
Footnotes
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
