Abstract
Daily rhythms in mammalian physiology and behavior are generated by a central pacemaker located in the hypothalamic suprachiasmatic nuclei (SCN), the timing of which is set by light from the environment. When the ambient light-dark cycle is shifted, as occurs with travel across time zones, the SCN and its output rhythms must reset or re-entrain their phases to match the new schedule—a sluggish process requiring about 1 day per hour shift. Using a global assay of circadian resetting to 6 equidistant time-zone meridians, we document this characteristically slow and distance-dependent resetting of Syrian hamsters under typical laboratory lighting conditions, which mimic summer day lengths. The circadian pacemaker, however, is additionally entrainable with respect to its waveform (i.e., the shape of the 24-h oscillation) allowing for tracking of seasonally varying day lengths. We here demonstrate an unprecedented, light exposure–based acceleration in phase resetting following 2 manipulations of circadian waveform. Adaptation of circadian waveforms to long winter nights (8 h light, 16 h dark) doubled the shift response in the first 3 days after the shift. Moreover, a bifurcated waveform induced by exposure to a novel 24-h light-dark-light-dark cycle permitted nearly instant resetting to phase shifts from 4 to 12 h in magnitude, representing a 71% reduction in the mismatch between the activity rhythm and the new photocycle. Thus, a marked enhancement of phase shifting can be induced via nonpharmacological, noninvasive manipulation of the circadian pacemaker waveform in a model species for mammalian circadian rhythmicity. Given the evidence of conserved flexibility in the human pacemaker waveform, these findings raise the promise of flexible resetting applicable to circadian disruption in shift workers, frequent time-zone travelers, and any individual forced to adjust to challenging schedules.
Light around dawn and dusk entrains the suprachiasmatic nuclei (SCN) by inducing typically small daily advances and delays, respectively, in the phase of the circadian pacemaker by perturbing expression levels of molecular elements of interacting transcription-translation feedback loops in SCN neurons (Kuhlman et al., 2003). Exposure to bright light at other times in the biological night, such as may occur with shiftwork or time-zone travel, can induce disruptive phase shifts and temporary misalignment of components of the circadian time system, with deleterious effects on performance and health (Evans and Davidson, 2013) including increased mortality in animal models (Davidson et al., 2006).
Differential SCN entrainment to winter versus summer photoperiods, as reflected in SCN electrical and molecular rhythms, allows for seasonal variation in activity rhythms, reproduction, and other photoperiodic traits (Schwartz et al., 2001). Under winter photoperiods, moreover, the light sensitivity of the circadian clock is increased 40-fold (Glickman et al., 2012), and light induces larger phase shifts than it does under summer day lengths (Evans et al., 2004; Goldman and Elliott, 1988; Glickman et al., 2014). Whether waveform manipulation could enhance resetting to full photoperiods outside of the laboratory remains to be assessed.
Syrian hamsters were entrained for 9 weeks to summer (long day [LD], 16 h light, 8 h dark) or winter (short day [SD], 8 h light, 16 h dark) conditions. Subsets of each photoperiod group were transferred to summer conditions in 1 of 6 phase shifts varying in magnitude and direction (0 shift, 4- or 8-h advance, 12 shift, 8- or 4-h delay). As seen in representative activity records (Fig. 1), hamsters pre-entrained to long days (LD) adapted to the shifted light cycles very gradually (~1.3 h/day), requiring more than a week, for instance, to realign in the 8-h delay group. If pre-entrained to the short photoperiod (SD), by contrast, hamsters aligned activity with the new light-dark schedule in the first few cycles, regardless of the magnitude and direction of the shift. Group mean activity onsets, which mark the beginning of biological night, deviated by at least several hours in a distance-dependent manner from the new dark onset in the LD cohort (Figs. 2 and 3) and had not uniformly achieved target phase within 2 weeks. In contrast, for all conditions but the 0 shift, SD animals adjusted substantially more rapidly than their LD counterparts and achieved stable entrainment within 1 week. Averaging across all phase shift groups, including those requiring an 8- or 12-h shift, the onset of subjective night of the SD cohort was only 2.3 h from the respective new schedules in the first 3 days after the shift, a 49% reduction of mismatch (Fig. 3). The same enhancement was observed in the other 3 measures of phase: mid-subjective night, onset of subjective day, and the number of days to re-entrain onsets (Suppl. Fig. S1).
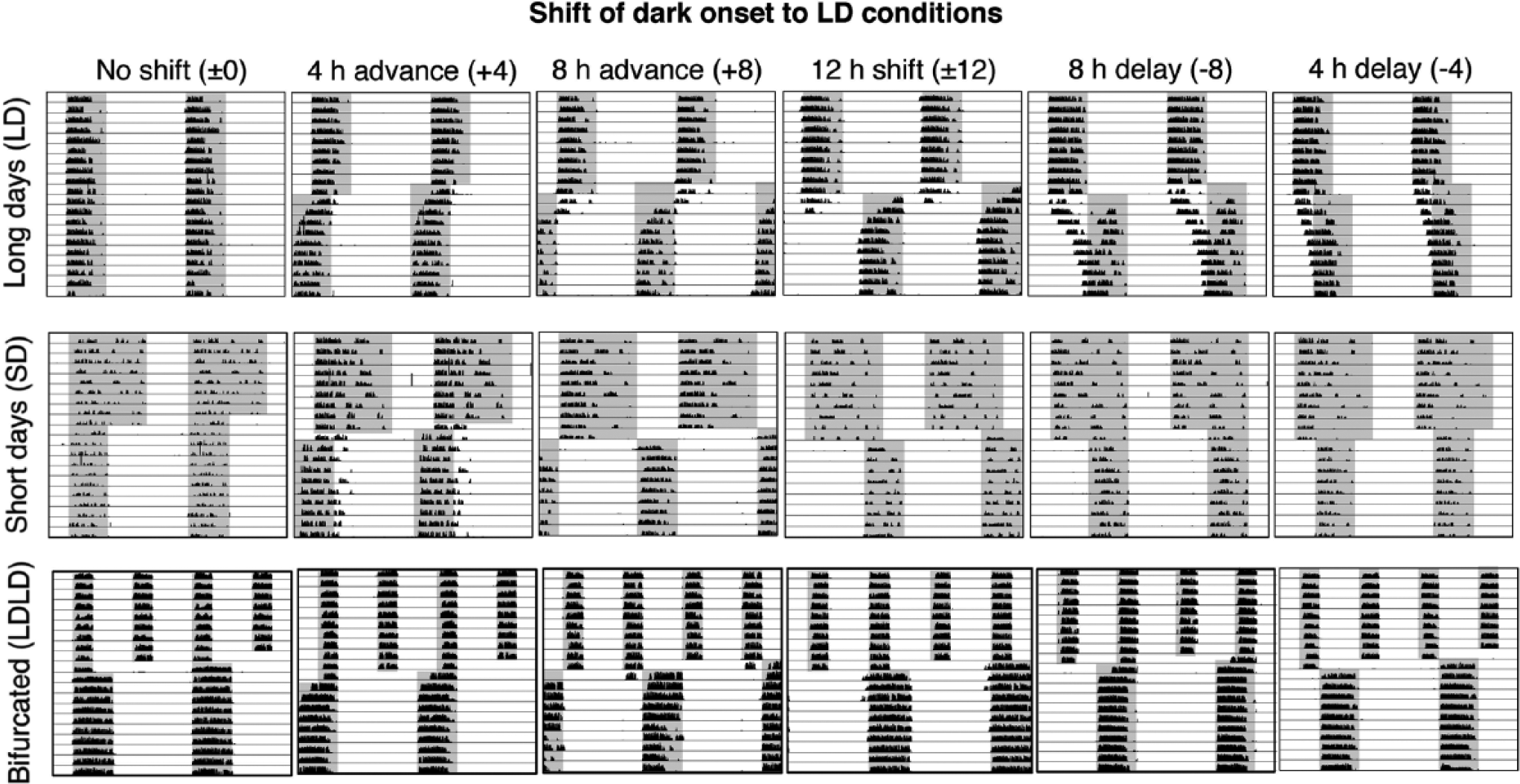
Sluggish re-entrainment observed under long-day (LD) 16:8 summer photoperiods (top row) is accelerated following entrainment to LD 8:16 winter (middle row) and light-dark-light-dark (LDLD) 8:4:8:4 photoperiods (bottom row). Representative double-plotted actograms (i.e., each horizontal line plots 48 h of data, and horizontal lines begin at 24-h intervals, so that all data are plotted doubly) of wheel-running activity in Syrian hamsters exposed to each of 6 phase shifts in this assay of global resetting. For each actogram, 10 days of baseline entrainment and 10 days after the shift are shown. The size of the photoperiod shift is denoted with respect to change in lights-off (+4 h represents a 4-h advance in lights-off in both LD and short-day [SD] animals. Because of the change in waveform, SD animals would simultaneously experience a 12-h change in lights-on). For LDLD animals, this lights-off reference point was derived from the original scotophases in LD 20:4, pre-bifurcation.
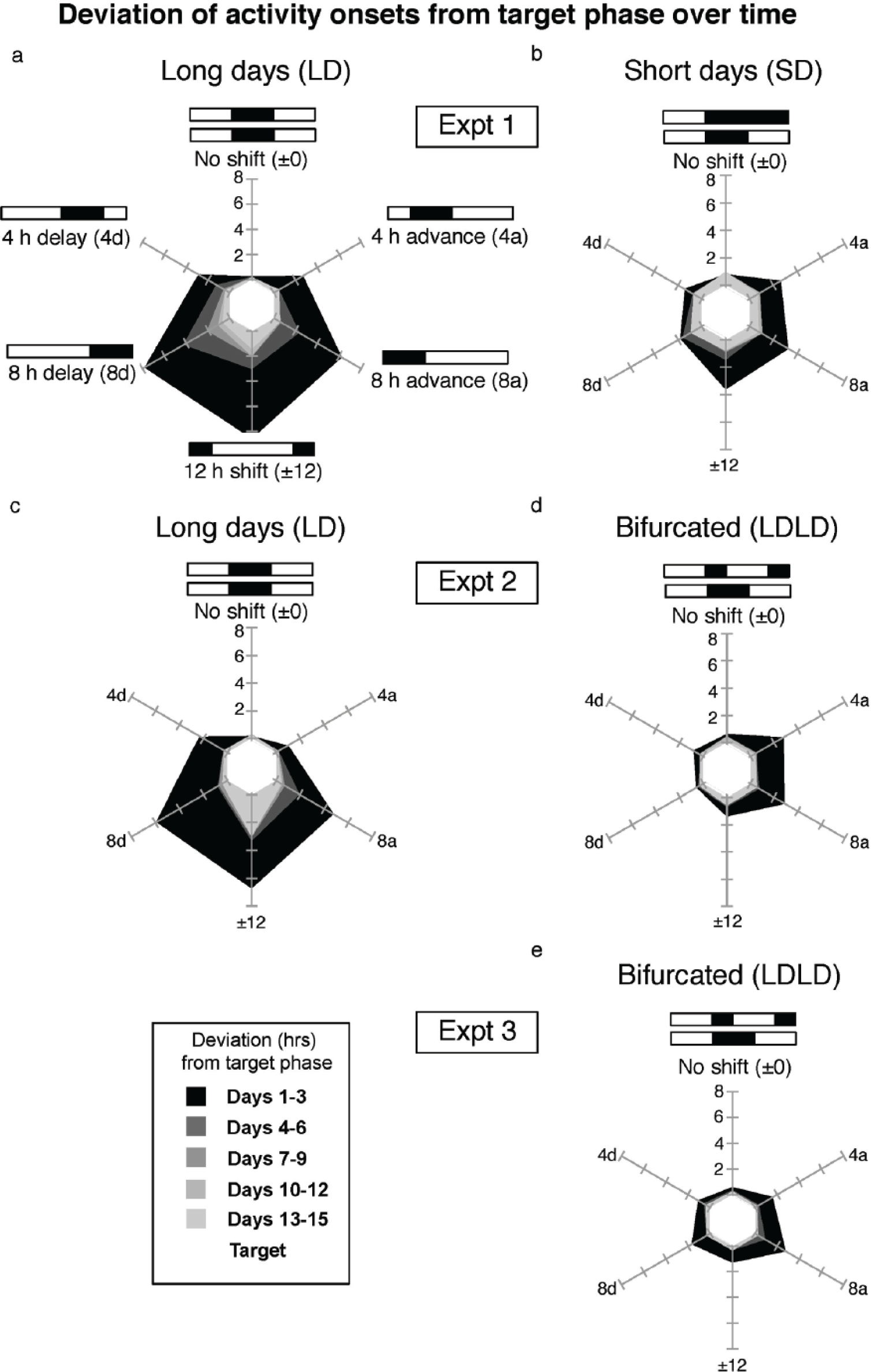
Compared with hamsters entrained to long-day (LD) 16:8 photoperiods (a and c), mean phase shifts are more rapid after LD 8:16 (b) or light-dark-light-dark (LDLD) 8:4:8:4 entrainment (d and e). Shown are radar plots of phase mismatch at progressive 3-day intervals. Each axis of the radar plot represents a shift of varying magnitude to a new LD 16:8 schedule, represented in white and black bars for light and dark intervals, respectively. The white hexagon in the center of each plot represents the “target” time of lights-off in the new schedule. Each shaded polygon represents the absolute value of the average 3-day deviation of the mean onset from the new lights-off in hours. As in Figure 1, photoperiod shifts are designated with respect to lights-off. Assignment to other reference points (e.g., mid-dark, or lights-on) would rotate the plotted polygons but not change their area.
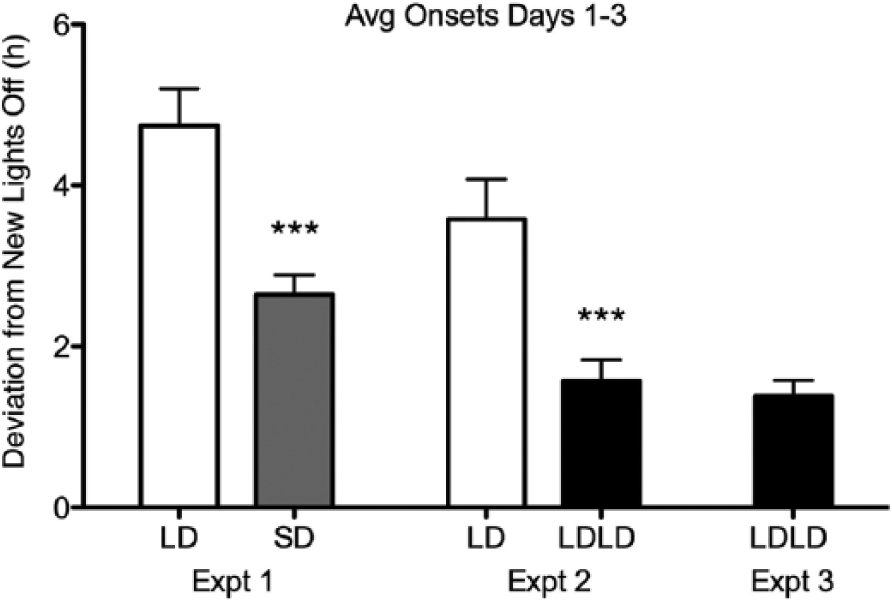
Compared with long-day (LD) (unbifurcated) hamsters, short photoperiod (Expt. 1, 8:16) and bifurcated (Expt. 2, 8:4:8:4) hamsters shifted twice as much in the first 3 days. For the first 3 days following phase shifts, the deviation of activity onset from lights-off was averaged for each animal. Mean ± SEM data are presented collapsed across all photoperiod shifts for each waveform. Asterisks indicate statistically significant difference from LD values in the same experiment (Welch-corrected t, p < 0.001). Post hoc comparison of light-dark-light-dark (LDLD) Expt. 3 hamsters likewise differed significantly from LD hamsters in both experiments (Welch-corrected t, both p < 0.001).
Thus, we here demonstrate that beyond the well-documented enhanced phase-shifting response to brief (nonparametric) light pulses, short photoperiod entrainment enhances resetting to full photoperiods. Following manipulations of arginine vasopressin (AVP) signaling (Yamaguchi et al., 2013) and addition of dim light at night (Evans et al., 2007, 2009), enhanced resetting is observed under full photoperiods but not in response to short light pulses, suggesting dissociable mechanisms of nonparametric and parametric re-entrainment. Given this, the enhanced resetting to full photoperiods after short-photoperiod entrainment is novel.
We subsequently tested whether entrainment could likewise be accelerated following an alternative waveform manipulation, termed “bifurcation,” that requires neither a lengthy adaptation interval nor a reduction in daily light exposure (Gorman and Elliott, 2004). Following permissive 24-h light-dark-light-dark conditions (LDLD), hamsters and mice rapidly and reliably re-entrain their circadian clock to program 2 subjective days and 2 intervening subjective nights in each 24-h period. This bifurcated waveform results in bimodal expression of activity, melatonin, gene expression, and other measures of subjective day and night (Gorman et al., 2001; Raiewski et al., 2012; Yan et al., 2010). Here, LD (unbifurcated) and LDLD (bifurcated) hamsters were challenged with the global phase-resetting assay described above. Bifurcated animals entrained in less than half of the time required by the slow, LD hamsters (Suppl. Fig. S2). Importantly, 4 animals under LDLD conditions failed to bifurcate, and resetting rates for those animals resembled those of LD animals (Suppl. Fig. S4). Across all phase shift magnitudes, including those requiring an 8- or 12-h shift, the onset of subjective night of the bifurcated animals was only 1.6 h from the new light-dark schedule in the first 3 days after the shift and fully entrained within 3 days (Fig. 3 and Suppl. Fig. S2). This same pattern was seen for mid-subjective night and for the onset of subjective day (Suppl. Fig. S2).
In a third experiment, hamsters were bifurcated under optimized conditions and subjected to the same protocol, resulting in only a 1.4-h mismatch from respective destinations in the first 3 days (Fig. 3 and Suppl. Fig. S3). Notably, while exposure to either SDs or LDLD light cycles induces profound changes in circadian waveform, animals in all groups rapidly adopted a long-day waveform after transfer to their destination light schedule (Suppl. Fig. S5). Thus, bifurcation of the circadian waveform, a scarcely characterized pattern of entrainment, markedly speeds re-entrainment in the absence of any pharmacological or genetic manipulation, but with the same total light exposure as in LD 16:8.
We next verified that the rapid adjustment of locomotor activity to new schedules seen after bifurcation reflects entrainment of the endogenous circadian pacemaker, and not an acute masking response to the new lighting conditions. Six hamsters were bifurcated for 2 weeks and challenged with shifts to 16:8 in anti-phase (equivalent to the 0 shift and 12-h shift conditions). As above, wheel-running closely matched both of the anti-phase conditions by the second complete LD cycle. In constant darkness initiated after the third complete LD cycle, activity onsets of the free-running, endogenously generated activity rhythm projected to a phase that in no case deviated more than 90 min from the prior time of lights-out, establishing that the pacemaker phase had been reset by the 3 days in LD (Fig. 4). Moreover, waveforms of the free-running rhythms were indistinguishable from those of LD animals, indicating that the bifurcated state is readily reversible. Using an analogous protocol, we further determined that no more than 72 h in a bifurcated state is required to achieve this effect.

Only 3 LDLD cycles are required for rapid resetting to anti-phase schedules. Representative actograms illustrating wheel-running activity in constant darkness (DD) after shifts to photoperiods 12 h in anti-phase. When behavioral rhythms are unmasked in DD, it is revealed that animals have been entrained by 3 light-dark cycles to phases 12 h apart. Furthermore, this rapid resetting can be accomplished with as few as 3 days of activity bifurcation prior to the shift in schedule. Phase was determined via a line fit to 7 days of free-running activity, skipping 3 days to allow for transients, then projected back to day 1 of DD. Activity onsets of the free-running, endogenously generated activity rhythm projected to a phase that in no case deviated more than 90 min from the prior time of lights-out in all 6 animals.
While the mechanisms for enhanced rates of resetting are unknown, we speculate the anti-phasic gene expression in the core and shell of SCN neurons found under bifurcation (Yan et al., 2010; Watanabe et al., 2007) reflects a dissociation of oscillators within the SCN that potentially facilitates rapid readjustment to changing light-dark schedules. An et al. (2013) demonstrated that manipulations of vasoactive intestinal peptide (VIP) cause the rhythms of individual cells to desynchronize, or “phase tumble,” resulting in enhanced phase-resetting responses. While this may be predicted by limit-cycle considerations, it contrasts with SCN network findings wherein greater phase coherence in short photoperiods is associated with increased phase-shift responses to light (Meijer et al., 2010). The mechanisms for enhanced resetting after entrainment to short photoperiods and after bifurcation in LDLD conditions need not be the same.
In summary, here we apply a novel assay using full photoperiods to phase shifts of varying magnitude and direction up to 12 h and find that 2 manipulations of circadian waveform—1 natural and 1 highly artificial—markedly enhance circadian adjustment. We show that the mammalian pacemaker is capable of adjusting to LD photoperiods 12 h apart within 3 cycles using a nonpharmacological, noninvasive manipulation. The novel global assay and results described here illustrate the remarkable potential of this waveform manipulation. Unlike long-night adaptation, bifurcation is a manipulation of waveform that can be experimentally-induced in hamsters within a single day and allows for conservation of 8 h of darkness, in a 24 h interval. Moreover, despite the plasticity induced by bifurcation, it is relatively stable as an entrainment state and is resilient to light schedule perturbation (Gorman and Steele, 2006; Harrison and Gorman, 2012). It is as yet unclear whether these studies, conducted in seasonally responsive nocturnal rodents, generalize to diurnal animals such as humans (Harrison and Gorman, 2012). Regardless, experiments conducted in nocturnal rodents will facilitate mechanistic understanding of how resetting flexibility may be modulated. Clearly, apparent constraints of phase-shifting can be markedly reduced with waveform manipulations. Increased phase resetting without genetic or pharmacological manipulation may have practical utility for adjustment to challenging schedules.
Footnotes
Acknowledgements
The authors acknowledge Jeff Elliott and Gena Glickman for comments on an earlier version and Antonio Mora for animal care. Research was supported by NICHD 36460 (MRG) and the Office of Naval Research N00014-13-1-0285 (MRG).
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
