Abstract
We have recently shown that molecular rhythms in the murine suprachiasmatic nucleus (SCN) are affected by repeated social defeat (SD) during the dark/active phase (social defeat dark [SDD]), while repeated SD during the light/inactive phase (social defeat light [SDL]) had no influence on PERIOD2::LUCIFERASE explant rhythms in the SCN. Here we assessed the effects of the same stress paradigm by in vivo biotelemetry on 2 output rhythms of the circadian clock (i.e., activity and core body temperature) in wild-type (WT) and clock-deficient Period (Per)1/2 double-mutant mice during and following repeated SDL and SDD. In general, stress had more pronounced effects on activity compared to body temperature rhythms. Throughout the SD procedure, activity and body temperature were markedly increased during the 2 h of stressor exposure at zeitgeber time (ZT) 1 to ZT3 (SDL mice) and ZT13 to ZT15 (SDD mice), which was compensated by decreased activity during the remaining dark phase (SDL and SDD mice) and light phase (SDL mice) in both genotypes. Considerable differences in the activity between SDL and SDD mice were seen in the poststress period. SDD mice exhibited a reduced first activity bout at ZT13, delayed activity onset, and, consequently, a more narrow activity bandwidth compared with single-housed control (SHC) and SDL mice. Given that this effect was absent in Per1/2 mutant SDD mice and persisted under constant darkness conditions in SDD WT mice, it suggests an involvement of the endogenous clock. Taken together, the present findings demonstrate that SDD has long-lasting consequences for the functional output of the biological clock that, at least in part, appear to depend on the clock genes Per1 and Per2.
Due to the earth’s rotation around its own axis, most species are exposed to a rhythmic environment. To anticipate these predictable environmental changes, organisms have evolved an internal time-keeping system (i.e., the circadian clock). In mammals, the clock system is composed of a central pacemaker residing in the suprachiasmatic nucleus (SCN) of the hypothalamus and multiple subsidiary clocks in peripheral tissues. Both the SCN and peripheral organs possess an endogenous clock machinery, thus enabling the formation of self-sustained circadian rhythms in the absence of external cues. However, to stay tuned to geophysical time, the circadian system requires daily synchronization of the endogenous oscillation to the solar day. This is accomplished using environmental time signals, whereof the 24-h light/dark (LD) cycle is the most important one (Dibner et al., 2010; Kwon et al., 2011).
The generation of circadian rhythms at the molecular level depends on the activity of so-called clock genes, which form interlocking feedback loops of gene transcription and translation. The positive limb of the mammalian core feedback loop involves the proteins CLOCK and BMAL1, which heterodimerize and act as transcriptional activators of Period (Per1-3) and Cryptochrome (Cry1/2) genes. Their respective protein products, PER and CRY, function as the negative limb by forming heterodimers that interfere with the CLOCK/BMAL1 complex to repress their own transcription. Additional feedback loops involving the nuclear receptor genes RevErbα/β and Rorα stabilize and regulate the resetting properties of the aforementioned core feedback loop (Reppert and Weaver, 2002). Of note, the negative elements of the core clock machinery are characterized by a certain degree of redundance. Although resulting in aberrant circadian periodicity, clock oscillation is sustained when a single gene within the Per or Cry family is mutated (Bae et al., 2001; van der Horst et al., 1999; Zheng et al., 2001). In contrast, Cry1/Cry2 double-mutant mice (van der Horst et al., 1999) and Per1/2 double mutants (Zheng et al., 2001) lose rhythmicity immediately after release into constant darkness (DD), suggesting a complete functional disruption of the clock.
It has been shown that glucocorticoids (GCs)—steroid hormones secreted into the bloodstream upon stressful stimuli and in a circadian fashion under basal conditions—can reset the rhythmic phase of peripheral clocks by a rapid induction of Per1 and Per2 through GC response elements (GREs) in their promoter regions without affecting the central clock in the SCN (Balsalobre et al., 1998). In line with that, various early studies indicated that stress does not appear to have a robust effect on the central circadian pacemaker as assessed by free-running activity rhythms (Meerlo et al., 1997; Moberg and Clark, 1976; Van Reeth et al., 1991). However, stress can have some masking effects on circadian parameters. Decreased amplitudes of body temperature rhythms following social stress (Meerlo et al., 1997) or repeated foot shocks (Kant et al., 1991) have been reported in rats. Furthermore, surgery stress was found to increase the circadian amplitude of heart rate and temperature rhythms in rats (Harper et al., 1996). Intriguingly, recent studies reveal dampened PER2, CLOCK, and BMAL1 protein expression in the SCN upon chronic unpredictable stress (Jiang et al., 2011) and decreased PER2 protein expression following repeated restraint stress (Kinoshita et al., 2012), indicating that certain stressors are capable of perturbing clock core function in the SCN.
In contrast to light, which induces Per gene expression only during the night (Albrecht et al., 1997; Shearman et al., 1997), GC-dependent phase-resetting processes can be induced at any time of the LD cycle (Balsalobre et al., 2000). Yet, exposure of rats to a novel environment or restraint stress, as psychological means to raise GC levels, resulted in stronger hypothalamic-pituitary-adrenal (HPA) axis responses at early day compared with early night. Vice versa, physical stressors, such as hypoglycemic shock, elicit the largest responses at the onset of activity, demonstrating a stressor-specific circadian gating of the stress system (Dickmeis, 2009; Jacobson, 2005). In line, we have previously shown that repeated social defeat (SD) has different effects on the central clock in the SCN and peripheral output rhythms, depending on the time of day of stressor exposure. SD during the early dark phase (zeitgeber time [ZT] 13 to ZT15; social defeat dark [SDD]), but not during the early light phase (ZT1-ZT3; social defeat light [SDL]), resulted in blunted activity rhythms, flattened or shifted GC rhythms (Bartlang et al., 2012), and increased PER2 protein expression and PER2 rhythm amplitudes in the SCN (Bartlang et al., 2014).
Based on these findings, the present study sought to investigate the consequences of repeated SDL and SDD on 2 output rhythms of the clock: activity and body temperature. Therefore, wild-type (WT) and Per1/2 double-mutant mice were implanted subcutaneously with small transmitters, and body temperature and activity were recorded under baseline, stress, and poststress conditions.
Materials and Methods
Animals
Male C57BL/6N and C57BL/6J mice (Charles River, Sulzfeld, Germany) as well as male Per1/2 double-mutant mice were used as experimental animals exposed to SD (see below). The C57BL/6N substrain was used in our previous study (Bartlang et al., 2012; Bartlang et al., 2014), whereas the C57BL/6J substrain served as an adequate congenic control group for the Per1/2 double mutants that carried this genetic background. Per1/Per2 mutant mice were bred from in-house colonies maintained at the Central Animal Facility, University of Lübeck, Germany. Periodic cross-breeding of Per1/Per2 mutant mice with C57BL/6J WT animals directly purchased from the supplier was carried out to ensure a stable and standardized genetic background. All mice were kept under standard laboratory conditions (23 ± 1 °C; 55% ± 5% humidity) and had free access to tap water and standard mouse diet. Unless otherwise noted, they were exposed to a 12:12-h LD cycle (light phase: 250 ± 50 lux) with lights-on at 07:00. The time of lights-on is defined as ZT0 and lights-off as ZT12. Routine cage changing and stressor exposure during the dark phase were performed under dim red light. All animals were individually housed in standard polycarbonate cages (17.5 × 35.5 × 13 cm, experimental mice or 41 × 15 × 25 cm, residents) for 1 week before the start of the experiment. Animals weighed 17 to 22 g when exposed as intruders to male-dominant resident CD1 mice weighing 30 to 35 g (Charles River). All experimental protocols were approved by the Committee on Animal Health and Care of the local government and performed according to the international guidelines given in the Declaration of Helsinki and the U.S. National Institutes of Health’s Guide for the Care and Use of Laboratory Animals. All efforts were made to minimize the number of animals used and their suffering.
Experimental Design
Intra-abdominal telemetry transmitters were surgically implanted subcutaneously. Based on a previous study, baseline body temperature and activity data collection started 9 days after implantation of the transmitters at ZT0 (Vlach et al., 2000). Twenty-one days after implantation, mice were assigned to a single-housed control (SHC), an SDL group, and an SDD group, according to basal activity. SHC mice remained undisturbed except for a weekly change of bedding at the beginning of the dark phase. Based on previous studies, SHC mice were considered the most appropriate controls in this paradigm (Singewald et al., 2009). SDL and SDD mice were exposed to SD for 2 h daily across 19 days (except for days 7 and 14), either at the beginning of the light phase (ZT1-ZT3, SDL mice) or at the beginning of the dark phase (ZT13-ZT15, SDD mice). Subsequent to the last SD on day 19, data collection was continued for 14 days in the poststress period, either in LD or DD conditions.
SDL and SDD Paradigms
The stress paradigm was performed as described before (Bartlang et al., 2012; Bartlang et al., 2014). Briefly, during each SD session, the experimental mouse was taken from its home cage and placed into a male resident’s home cage. The same receiver placed underneath the standard polycarbonate cages of experimental mice was used for collecting the data during the 2 h of SD. After the first attack by the resident that resulted in submissive behavior from the intruder, both mice were separated from each other by a perforated partition wall. This protected the intruder from physical injuries by the resident but allowed visual, olfactory, and auditory contact as described previously (Reber et al., 2006). During the subsequent 2 h of sensory contact, food pellets, but not tap water, were available for all experimental mice, including SHC mice. Following removal of the partition wall, direct social interaction between the resident and the experimental mouse was allowed until the first attack by the resident that resulted in submissive behavior from the intruder. Each experimental mouse was exposed to a different resident every day to avoid habituation. The stress procedure was carried out in the same room in which experimental mice were housed but isolated from SHC mice.
Surgical Implantation of Transmitters
A small G2-E-Mitter implant (16.5 × 6.5 mm, 1.1 g; med-NATIC GmbH, München, Germany), containing a microchip as well as an antenna enclosed in bioglass, was sterilized with 70% ethanol and kept in saline until implantation under semisterile conditions. Before insertion, transmitters were programmed with the ID number of the respective mouse to avoid interference from neighboring mice during data collection. For surgery, experimental mice (intruder) were anesthetized with isofluorane (Baxter GmbH, Unterschleißheim, Germany). After shaving at the back, each transmitter was implanted subcutaneously in the abdominal region via a 1.5-cm-long skin incision at the back of the mouse. The skin of the back was sutured with 2 to 3 stitches, and iodine was applied to the wound. During surgery, body temperature was maintained by a heating pad. To avoid postsurgical infections, mice received 0.1 mL antibiotics (SC, Baytril 2.5%; Bayer Vital GmbH, Leverkusen, Germany). We chose the subcutaneous implantation of the transmitters because the mice were rather small at the beginning of the experiments and we did not want to risk losing them during or after surgery. This was especially true for the double mutants, which were difficult to obtain in larger numbers.
Telemetry System and Telemetric Data Collection
The telemetry monitoring system (Mini Mitter, Bend, OR, provided by med-NATIC GmbH) is composed of a passive (i.e., battery-free) temperature/inertia sensor and telemetry transmitter (G2-E-Mitter), a receiver (model ER-4000, placed underneath the cage), and a data acquisition system with accompanying software (Vital View; Mini Mitter). The temperature sensors were calibrated by the producer in the range from 33 to 41 °C with an accuracy of 0.1 °C. The data acquisition system was programmed to sample body temperature and activity at 1-min time intervals around the clock. Data collection began 9 days after surgery at ZT0 and continued for 8 weeks. Based on the fact that routine control procedures, such as placement into a clean cage, can affect behavior and induce an activation of the stress system (Balcombe et al., 2004; Duke et al., 2001), days on which cages were changed (day 1, 8, and 15) were excluded from the analysis (see Figure 1).
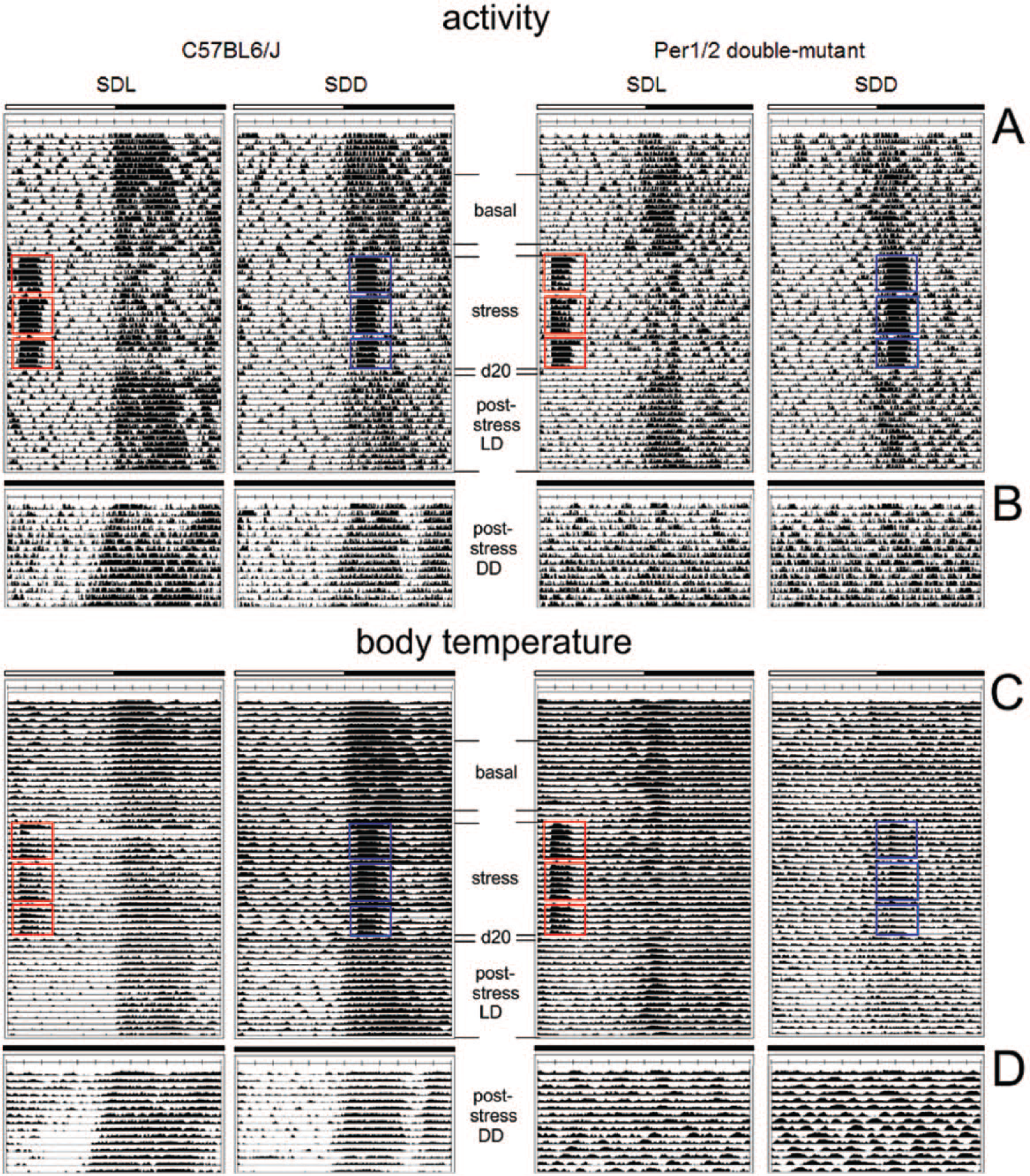
Representative individual activity (A, B) and body temperature profiles (C, D) of C57BL/6J and Per1/2 mutant social defeat light (SDL) and social defeat dark (SDD) mice under LD (A, C) or DD (B, D) conditions. The white bar indicates the light phase (ZT0-ZT12), and the dark bar indicates the dark phase (ZT12-ZT0). For SDL and SDD mice, the different episodes (i.e., basal, stress, d20, poststress under LD or DD) are labeled in the actograms/temperature profiles with the days of social defeat exposure marked in red (SDL) and blue (SDD), respectively.
Data Analysis and Statistics
Analysis of activity and body temperature was performed using Microsoft Excel. Average activity profiles and the phases of the activity peaks were calculated as described in Schlichting et al. (2014). Briefly, for peak determination, we smoothened the activity profiles of single mice over a moving average of 30 min with the help of Excel (Microsoft). This degree of smoothing made the activity profile more compact but preserved the 3 activity bouts as separate peaks. The times of the individual peaks were then determined by manually selecting the respective maxima with the mouse pointer. For calculating average activity profiles in DD, we first plotted each actogram based on the mouse’s individual period (circadian time [CT]); then we calculated an average day for each mouse and, subsequently, normalized it by interpolation to a period of 24 h. These normalized individual average days were used to calculate the average activity profiles of all mice of a given genotype or stress group. Free-running period lengths in DD were determined using χ2-periodogram analysis, and actograms were drawn using the ImageJ plugin ActogramJ (Schmid et al., 2011). Activity onsets and offsets were determined on single actograms with the tool “activity on- and offset” of ActogramJ. The values were then used to calculate the activity bandwidth during LD and DD. For activity bandwidth calculation in DD, we first plotted the actograms in CT (see above) to avoid an influence of the period on the values. Average activity levels were calculated from mean activity counts of single mice during daytime (ZT0-ZT6 [first half] and ZT6-ZT12 [second half]) and nighttime (ZT12-ZT18 [first half] and ZT18-ZT0 [second half]) for basal, stress, day 20, and poststress (LD and DD) periods, respectively. Average body temperature levels for the different periods (basal, stress, day 20, and poststress [LD and DD]) were calculated as described for activity above.
For statistical comparisons, the software package SPSS (version 19; SPSS, Inc., an IBM Company, Chicago, IL) was used. Data were tested for normal distribution applying a 1-sample Kolmogorov-Smirnov test. To test for significant differences in normally distributed data sets, 1-way analysis of variance (ANOVA) or ANOVA for repeated measures (rm ANOVA) was applied, followed by a post hoc pairwise comparison with LSD correction. Data are presented as means ± or + SEM. Significance was set at p ≤ 0.05.
Results
Figure 1 shows typical activity (A, B) and body temperature (C, D) recordings of WT (C57BL/6J) and Per1/Per2 mutant mice that were subjected to SD in the morning (SDL) or evening (SDD), respectively, under LD conditions. The periods of stressor exposure can be easily recognized by the elevated activity and body temperature levels during the acute stress episodes and the reduced levels during the remaining night. Half of the animals were kept in LD after the 19 days of stressor exposure (A, C), while the other half were transferred into DD (B, D) to assess the properties of the free-running circadian clock. For analyzing the effects of repeated stressor exposure on activity and body temperature, we divided the recording period in the following sections: basal, stress, immediately after the stress (day 20), poststress in LD, and poststress in DD (Fig. 1).
Basal Activity and Body Temperature
Activity of both WT strains showed 3 activity bouts, a short one after lights-off, a longer one in the first half of the night, and one of intermediate duration toward the end of the night (Figs. 1A, 2A, 4A). This trimodal pattern was principally reflected in the temperature profile, although it was less clear (Figs. 1C, 3A, 5A). In contrast, Per1/Per2 mutant mice only showed 1 activity bout that coincided with the first dark phase activity bout of WT mice, but it was much broader (Figs. 1A, 6A). The same was evident for the body temperature profile (Figs. 1C, 7A).
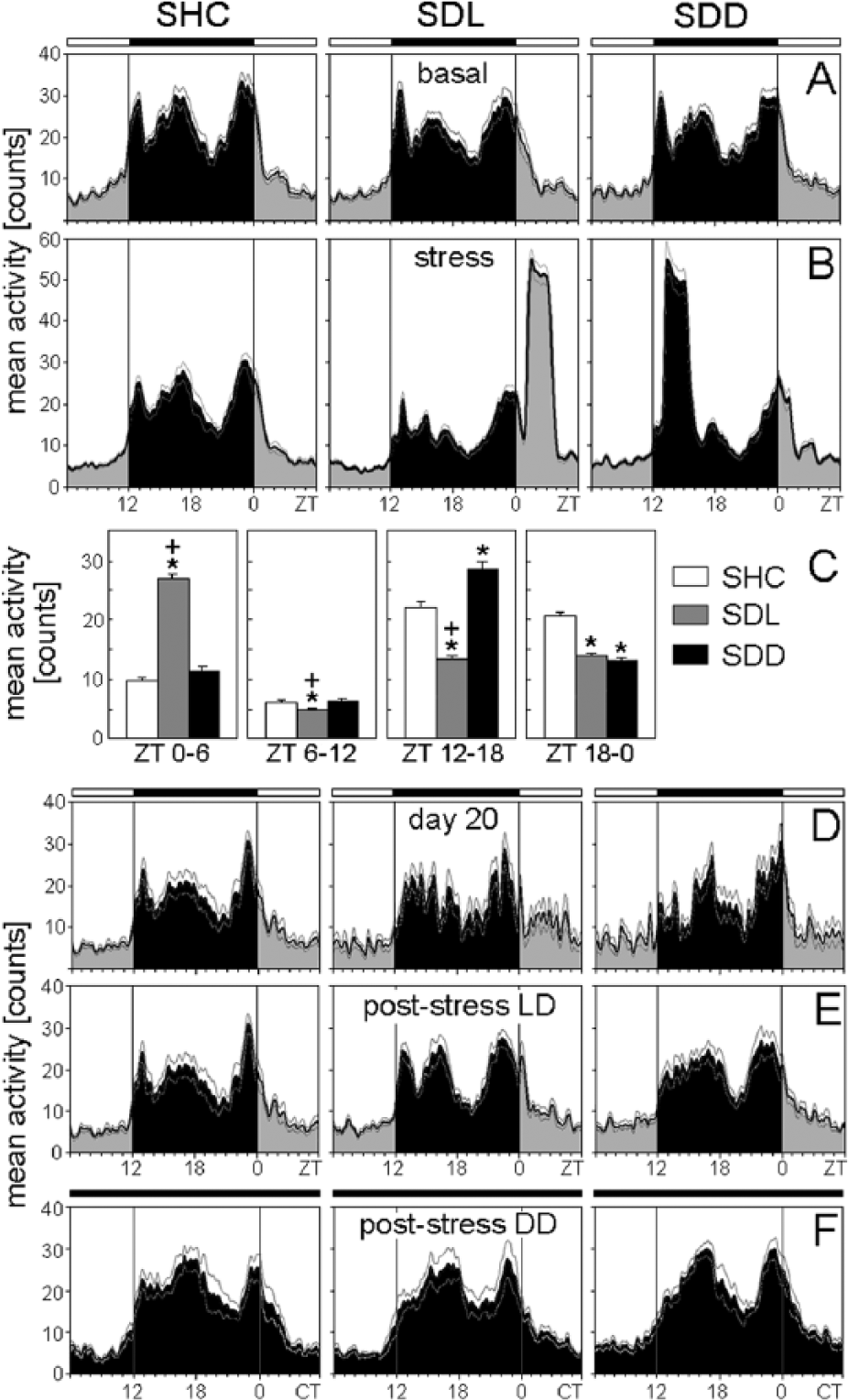
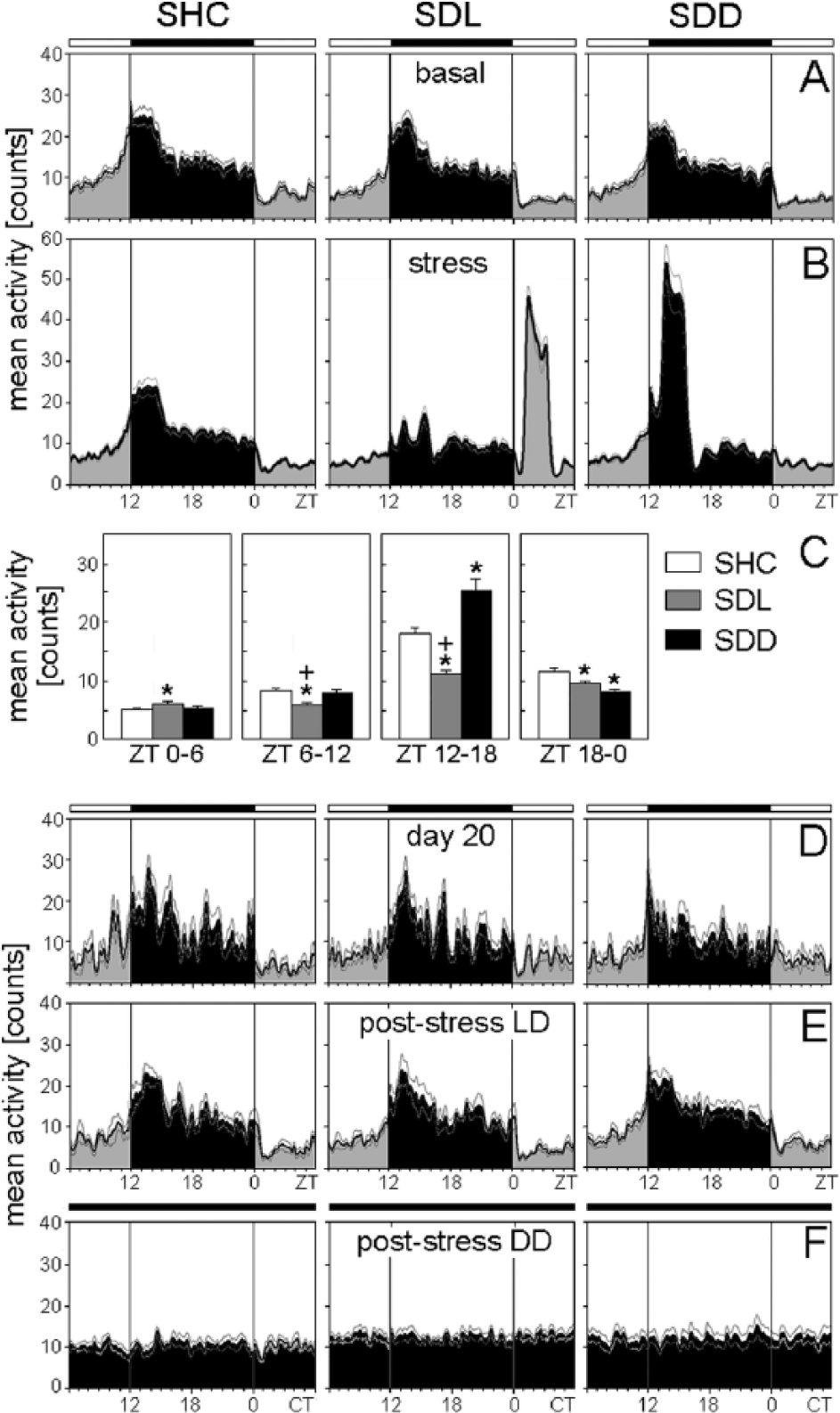
Average activity profiles of C57BL/6N single-housed control (SHC), social defeat light (SDL), and social defeat dark (SDD) mice during the basal (A), stress (B), immediately after stress (D), poststress in LD (E), and poststress in DD (F) periods. (C) Average activity profiles were calculated for each treatment group: SHC (A-D: n = 19; E: n = 10; F: n = 9), SDL (A-D: n = 18; E: n = 9; F: n = 9), and SDD (A-D: n = 17; E: n = 8; F: n = 9). The black bar indicates the dark phase (ZT12-ZT0); the white bars represent the light phase (ZT0-ZT12). For days of social defeat exposure, activity levels for both halves of the light phase and dark phase were calculated as mean activity, respectively (C). Data are presented as mean ± or + SEM. *p ≤ 0.05 versus respective SHC mice;
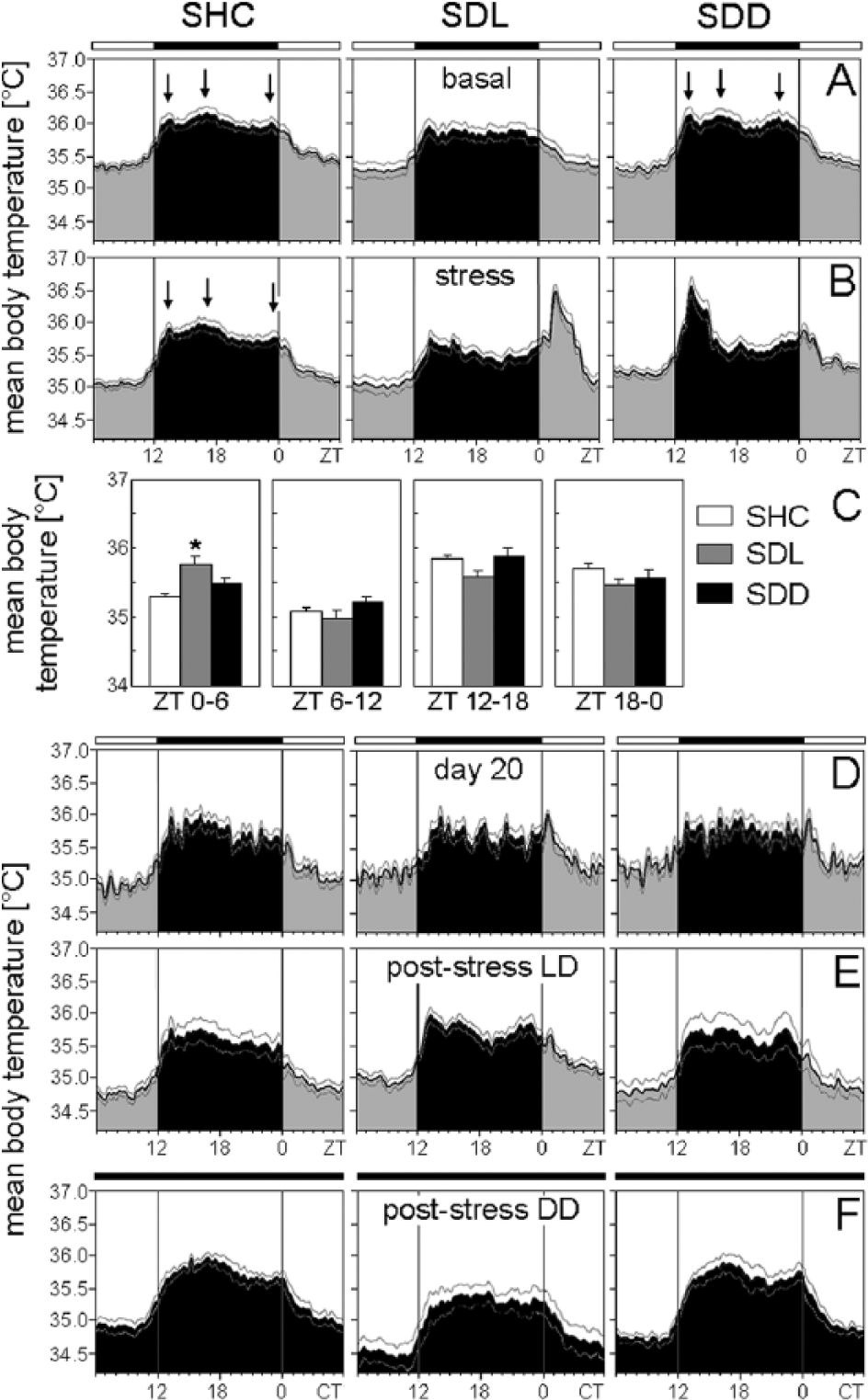
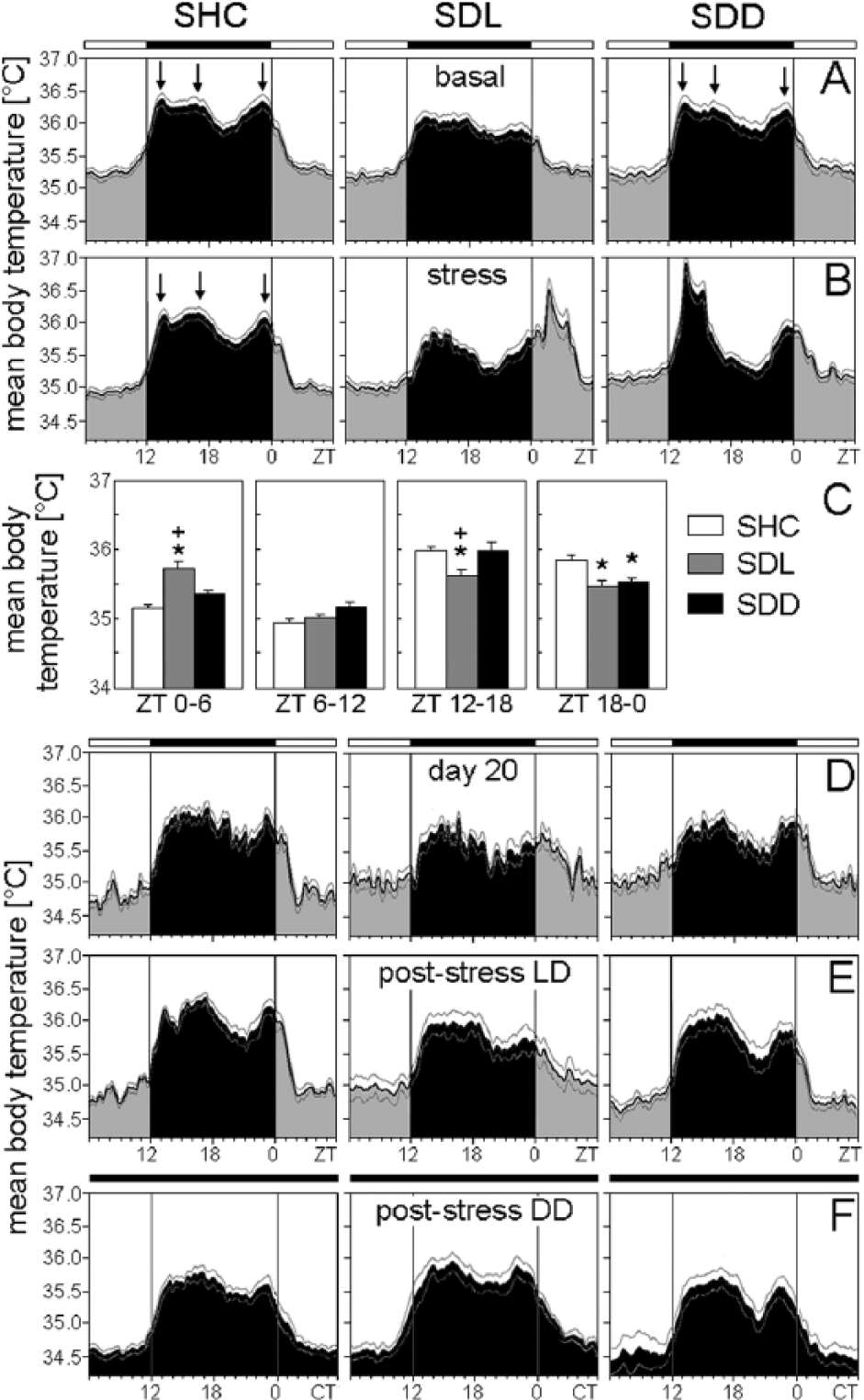
Mean body temperature of C57BL/6N single-housed control (SHC), social defeat light (SDL), and social defeat dark (SDD) mice during the basal (A), stress (B), immediately after stress (D), poststress in LD (E), and poststress in DD (F) periods. (C) Average body temperature profiles were calculated for each treatment group: SHC (A-D: n = 19; E: n = 10; F: n = 9), SDL (A-D: n = 18; E: n = 9; F: n = 9), and SDD (A-D: n = 17; E: n = 8; F: n = 9). Arrows point to the 3 temperature peaks whenever clearly visible. The black bar indicates the dark phase (ZT12-ZT0); the white bar represents the light phase (ZT0-ZT12). For days of SD exposure, activity levels for both halves of the light phase and dark phase were calculated as mean activity, respectively (C). Data are presented as mean ± or + SEM. *p ≤ 0.05 versus respective SHC mice.
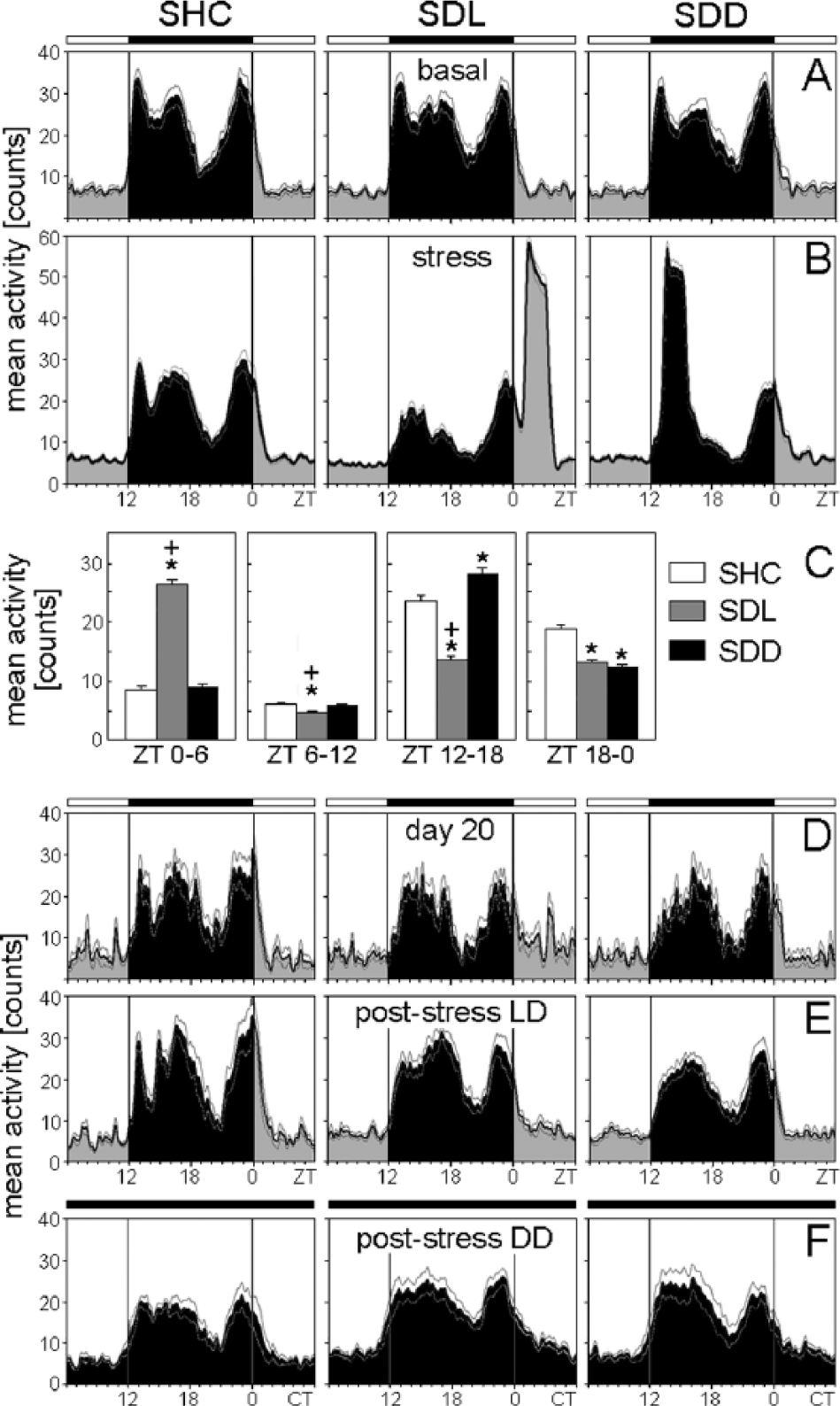
Average activity profiles of C57BL/6J single-housed control (SHC), social defeat light (SDL), and social defeat dark (SDD) mice during the basal (A), stress (B), immediately after stress (D), poststress in LD (E), and poststress in DD (F) periods. Average activity profiles were calculated for each treatment group: SHC (A-D: n = 20; E: n = 10; F: n = 10), SDL (A-D: n = 17; E: n = 8; F: n = 9), and SDD (A-D: n = 17; E: n = 8; F: n = 9). For days of social defeat exposure, activity levels for both halves of the light phase and dark phase were calculated as mean activity, respectively (C). Data are presented as mean ± or + SEM. *p ≤ 0.05 versus respective SHC mice;
Mean body temperature of C57BL/6J single-housed control (SHC), social defeat light (SDL), and social defeat dark (SDD) mice during the basal (A), stress (B), immediately after stress (D), poststress in LD (E), and poststress in DD (F) periods. Average body temperature profiles were calculated for each treatment group: SHC (A-D: n = 20; E: n = 10; F: n = 10), SDL (A-D: n = 17; E: n = 8; F: n = 9), and SDD (A-D: n = 17; E: n = 8; F: n = 9). For days of social defeat exposure, mean temperatures for both halves of the light phase and dark phase were calculated, respectively (C). Data are presented as mean ± or + SEM. *p ≤ 0.05 versus respective SHC mice;
Average activity profiles of Per1/2 double-mutant single-housed control (SHC), social defeat light (SDL), and social defeat dark (SDD) mice during the basal (A), stress (B), immediately after stress (D), poststress in LD (E), and poststress in DD (F) periods. Average activity profiles were calculated for each treatment group: SHC (A-D: n = 15; E: n = 7; F: n = 8), SDL (A-D: n = 12; E: n = 4; F: n = 8), and SDD (A-D: n = 15; E: n = 7; F: n = 8). For days of social defeat exposure, activity levels for both halves of the light phase and dark phase were calculated as mean activity, respectively (C). Data are presented as mean ± or + SEM. *p ≤ 0.05 versus respective SHC mice;
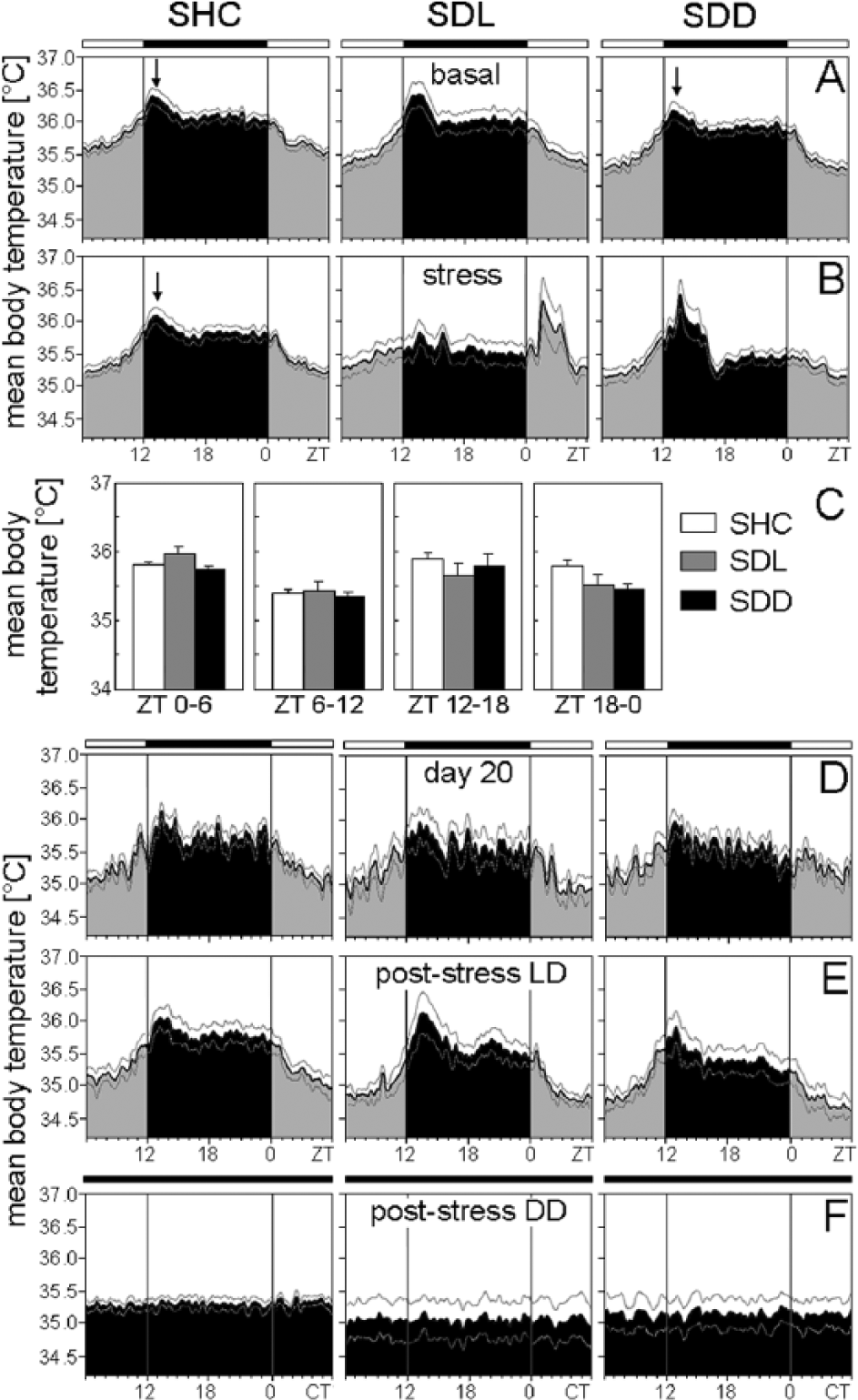
Mean body temperature of Per1/2 double-mutant single-housed control (SHC), social defeat light (SDL), and social defeat dark (SDD) mice during the basal (A), stress (B), immediately after stress (D), poststress in LD (E), and poststress in DD (F) periods. Average body temperature profiles were calculated for each treatment group: SHC (A-D: n = 15; E: n = 7; F: n = 8), SDL (A-D: n = 12; E: n = 4; F: n = 8), and SDD (A-D: n = 15; E: n = 7; F: n = 8). For days of social defeat exposure, mean temperatures for both halves of the light phase and dark phase were calculated, respectively (C). Data are presented as mean ± or + SEM. Labeling as in Figure 3.
Activity and Body Temperature during the Stress Episodes
During SD exposure, the trimodal activity pattern of WT mice was profoundly disrupted (Figs. 1A, 2B, 4B). Furthermore, as already observable in the actograms (Fig. 1A), SD exposure affected the activity levels of WT and Per1/Per2 mutant mice in a similar way. Activity levels in the first half of the light phase (ZT0-ZT6) on days of SD exposure were dependent on the employed SD paradigm (SHC, SDL, or SDD) for C57BL/6N (F2,51 = 191.04; p ≤ 0.001), C57BL/6J (F2,51 = 175.67; p ≤ 0.001), and Per1/Per2 mutant (F2,39 = 3.30; p ≤ 0.05) mice. Post hoc analysis revealed increased activity of WT SDL mice between ZT0 and ZT6 (p ≤ 0.001) compared with both SHC and SDD mice (Figs. 2B+C, 4B+C), while Per1/2 double-mutant SDL mice showed increased activity (p ≤ 0.05) compared with SHC mice only (Fig. 6B+C). The activity level in the second half of the light phase (ZT6-ZT12) on days of SD exposure was again dependent on the SD paradigm for C57BL/6N (F2,51 = 8.71; p ≤ 0.001), C57BL/6J (F2,51 = 8.19; p ≤ 0.001), and Per1/Per2 mutant (F2,39 = 6.48; p ≤ 0.01) mice. Post hoc tests showed that SDL mice exhibited decreased activity compared with SHC (p ≤ 0.01) and SDD mice (p ≤ 0.05) (Figs. 2B+C, 4B+C, 6B+C). Activity in the first half of the dark phase (ZT12-ZT18) was similarly dependent on the SD paradigm for C57BL/6N (F2,51 = 58.42; p ≤ 0.001), C57BL/6J (F2,51 = 52.84; p ≤ 0.001), and Per1/Per2 mutant (F2,39 = 25.56; p ≤ 0.001) mice. Post hoc analysis revealed reduced active behavior of SDL mice compared with SHC (p ≤ 0.01) and SDD (p ≤ 0.001) mice and increased activity of SDD mice compared with SHC animals (p ≤ 0.01) (Figs. 2B+C, 4B+C, 6B+C).
Finally, activity in the second half of the dark phase (ZT18-ZT0) was dependent on the SD paradigm for C57BL/6N (F2,51 = 20.09; p ≤ 0.001), C57BL/6J (F2,51 = 13.96; p ≤ 0.001), and Per1/Per2 mutant (F2,39 = 9.85; p ≤ 0.001) mice. Post hoc tests revealed that both SDL (p ≤ 0.05) and SDD (p ≤ 0.001) mice showed less active behavior compared with SHC mice (Figs. 2B+C, 4B+C, 6B+C).
Body temperature was affected by SD in a similar manner as activity, although the effects were less pronounced and not always significant, especially not in the mutants. Body temperature of C57BL/6N mice in the first half of the light phase (ZT0-ZT6) was dependent on the SD paradigm (F2,51 = 6.85; p ≤ 0.01). Post hoc analysis revealed increased body temperature of SDL mice in comparison to SHC animals (p ≤ 0.001) (Fig. 3B+C). Body temperature of C57BL/6J mice was dependent on the SD paradigm in the first half of the light phase (ZT0-ZT6) (F2,51 = 11.48; p ≤ 0.001), in the first half of the dark phase (ZT12-ZT18) (F2,51 = 5.86; p ≤ 0.01), and in the second half of the dark phase (ZT18-ZT0) (F2,51 = 5.83; p ≤ 0.01). Post hoc tests revealed increased body temperature of SDL mice between ZT0 and ZT6 compared with SHC (p ≤ 0.001) and SDD (p ≤ 0.01) mice, while body temperature of SDL mice was decreased between ZT12 and ZT18 compared with SHC (p ≤ 0.01) and SDD (p ≤ 0.01) mice. Body temperature between ZT18 and ZT0 was decreased in SDL (p ≤ 0.01) and SDD (p ≤ 0.01) mice compared with SHC mice, as revealed by post hoc analysis (Fig. 5B+C). Body temperature of Per1/2 double-mutant mice was not dependent on the SD paradigm, regardless of the time of assessment (first half of the light phase, second half of the light phase, first half of the dark phase, second half of the dark phase; Fig. 7B+C). Overall, no habituation regarding the acute stress-induced rises in activity and body temperature was observed across the 19 days of SD.
Activity and Body Temperature Immediately after SD Stress (Day 20)
Already on the first day following SD exposure, the animals returned to more pre-stress-like activity patterns. The above-described differences in activity levels between SHC, SDL, and SDD mice disappeared in the 2 WT strains and in the Per1/2 double-mutants (Fig. 2D, 4D, 6D). Activity peaks in the mutants after lights-off were not dependent on the SD paradigm or on the time of assessment (basal period, day 20, and poststress; Fig. 8). In contrast, the activity pattern of the 2 WT strains on day 20 remained different from that during the basal period (Figs. 2D, 4D). Most WT SDL and SDD mice lacked the first activity bout, and if it was present, it occurred significantly later than before the 19 days of stressor exposure (Fig. 8). rmANOVA revealed that the first activity peak was dependent on both the SD paradigm and the time of assessment (F4,28 = 12.55; p ≤ 0.001). Post hoc analysis indicated that both SDL and SDD C57BL/6N mice had a significantly delayed first activity peak on day 20 compared with SHC mice (p ≤ 0.01) and in comparison to the basal period (p ≤ 0.001). The same was evident for C57BL/6J mice (F4,26 = 6.74; p ≤ 0.001). In addition, the second activity peak was affected by the SD paradigm and the time of assessment in C57BL/6N mice (F4,48 = 5.30; p ≤ 0.001) and by the time of assessment in C57BL/6J mice (F4,48 = 5.30; p ≤ 0.001). Post hoc analysis showed that C57BL/6N SDD mice exhibited a delayed second activity peak on day 20 compared with SHC (p ≤ 0.01) and SDL (p ≤ 0.001) mice and compared with the basal period (p ≤ 0.001), while C57BL/6J SDD mice showed a delayed second activity peak on day 20 compared with the basal period (p ≤ 0.01).
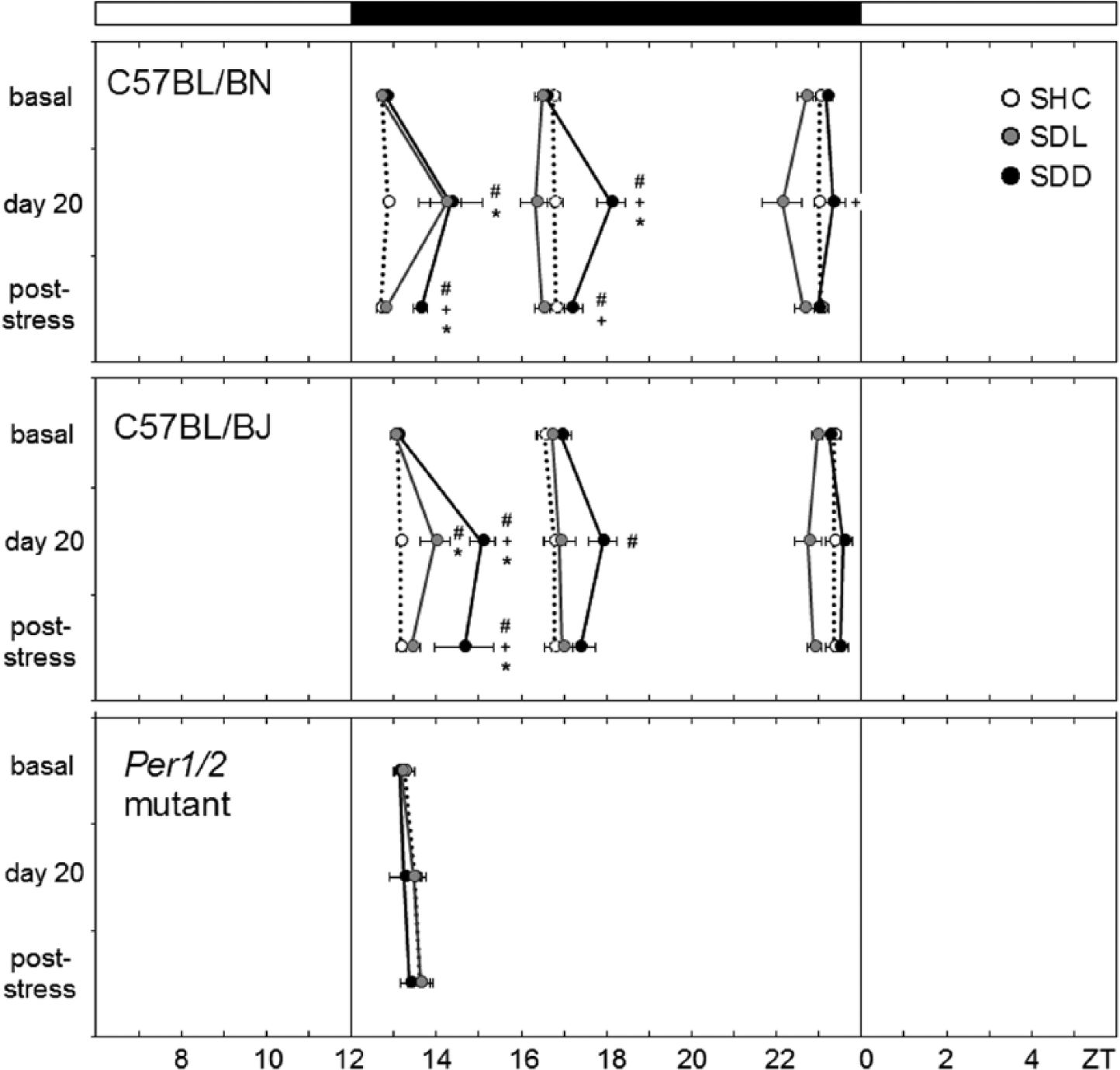
Activity peak phases of stressed (social defeat light [SDL], social defeat dark [SDD]) and control (single-housed control [SHC]) wild-type (WT) (C57BL/6N, C57BL/6J) and Per1/2 double-mutant mice. WT mice usually showed 3 activity bouts, the average phases of which were calculated before social defeat (SD) stress treatment (basal), directly after SD stress (day 20), and during the post-stress period in LD. In contrast, Per1/2 double-mutant mice showed only 1 activity bout for which the peak average phases could be calculated. The black bar on top indicates the dark phase (ZT12-ZT0). The white bars represent the light phases (ZT0-ZT6 and ZT6-ZT12). Data are presented as mean ± SEM; *p ≤ 0.05 versus respective SHC mice; +p ≤ 0.05 versus respective SDL mice; #p ≤ 0.05 versus respective basal levels. Please note that after stress (day 20 and poststress), not all WT animals showed the first and second activity bout. The following numbers of animals are included in the calculation: Basal C57BL/6N: SHC: n = 10, SDL: n = 9, SDD: n = 8; basal C57BL/6J: SHC: n = 10, SDL: n = 8, SDD: n = 8; basal Per1/2 double-mutants: SHC: n = 7, SDL: n = 4, SDD: n = 7. Day 20 C57BL/6N: SHC: n = 10, SDL: n = 4 (peak 1)/9 (peaks 2+3), SDD: n = 3 (peak 1)/8 (peaks 2+3). Day 20 C57BL/6J: SHC: n = 10, SDL: n = 4 (peak 1)/8 (peaks 2+3), SDD: n = 3 (peak 1)/8 (peaks 2+3). Day 20 Per1/2 double-mutants: SHC: n = 7, SDL: n = 4, SDD: n = 7. Poststress C57BL/6N: SHC: n = 10, SDL: n = 9, SDD: n = 3 (peak 1)/8 (peaks 2+3). Poststress C57BL/6J: SHC: n = 10, SDL: n = 7-8, SDD: n = 2 (peak 1)/8 (peaks 2+3). Poststress Per1/2 double mutants: SHC: n = 7, SDL: n = 4, SDD: n = 7.
The same tendency appeared to be present in body temperature, but most patterns were too irregular to allow clear determination of temperature peak phases.
Poststress Activity and Body Temperature in LD
The above-described differences in the presence and timing of the first activity peak persisted during the entire poststress period in SDD, but not in SDL, WT mice. Evaluation of actograms from SDD animals yielded a complete loss of the first activity bout in 62.5% (C57BL/6N) and 75% (C57BL/6J) of the animals, resulting in a bimodal nocturnal activity pattern. Furthermore, ANOVA with post hoc analysis revealed that the remaining 25% to 37.5% of C57BL/6J (F4,26 = 6.74; p ≤ 0.001) and C57BL/6N (F4,28 = 12.55; p ≤ 0.001) SDD mice showed a delayed first activity peak compared with both SHC (p ≤ 0.001) and SDL (p ≤ 0.01) mice as well as compared with basal levels (p ≤ 0.01) (Fig. 8). Concerning the second activity peak at around ZT17, a significant dependence on the SD paradigm and the time of assessment was observed for C57BL/6N mice (F4,48 = 5.30; p ≤ 0.001). Post hoc analysis revealed that SDD C57BL/6N mice showed a delayed activity peak compared to SDL mice (p ≤ 0.05) and to baseline conditions (p ≤ 0.01).
In contrast, SDL mice did not differ from SHC mice with respect to the nocturnal trimodal activity pattern and the phase of the activity peaks. Once more, the same tendency appeared to be present in body temperature, but it was more difficult to assess.
Poststress Activity and Body Temperature in DD
Directly upon transfer into DD conditions, Per1/2 double mutants lost their circadian behavioral rhythmicity, as expected (Zheng et al., 2001), whereas the 2 WT strains free-ran with a period slightly shorter than 24 h (Fig. 1B+D; Fig. 9A). During free-run, the trimodal activity pattern of the WT mice was retained in C57BL/6N mice (Fig. 2F; Fig. 9A) but became less obvious in C57BL/6J mice (Fig. 4F). Most important, the first activity bout remained absent in most SDD mice (Fig. 2F; Fig. 9A), which significantly shortened their activity bandwidth in DD (C57BL/6J: F2,25 = 10.11; p ≤ 0.001; C57BL/6N: F2,24 = 37.43; p ≤ 0.001), in comparison to SHC (p ≤ 0.001) and SDL (C57BL/6J: p ≤ 0.05; C57BL/6N: p ≤ 0.001) mice (Fig. 9C). This indicates that repeated SDD had long-term effects on the circadian clock of male WT mice. Furthermore, repeated SDL had significant effects on activity bandwidth and free-running period of the mice. Whereas SHC did not change the activity bandwidth in DD compared with LD (Fig. 9C), SDL and SDD exposure narrowed it. ANOVA revealed that the activity bandwidth was dependent on the light condition in the poststress period in both C57BL/6J (F1,25 = 34.76; p ≤ 0.001) and C57BL/6N (F1,24 = 64.35; p ≤ 0.001) mice. Post hoc analysis showed that SDL (p ≤ 0.01) and SDD (p ≤ 0.001) mice showed a significantly shorter activity bandwidth in DD compared with LD conditions (Fig. 9C). Furthermore, slight period differences were found in stressed C57BL/6N mice compared with unstressed SHC mice (Fig. 9B). Period was dependent on the SD paradigm (F2,24 = 22.01; p ≤ 0.001), and post hoc analysis revealed that SDL (p ≤ 0.001) (23.73 ± 0.02 h) and SDD (p ≤ 0.001) (23.73 ± 0.02 h) mice exhibited a significantly shorter period in comparison to SHC (23.91 ± 0.02 h) mice. No significant effect of SD stress on period was observed in C57BL/6J mice, but SDD mice showed at least a tendency to shorten period in response to stress (Fig. 9B).
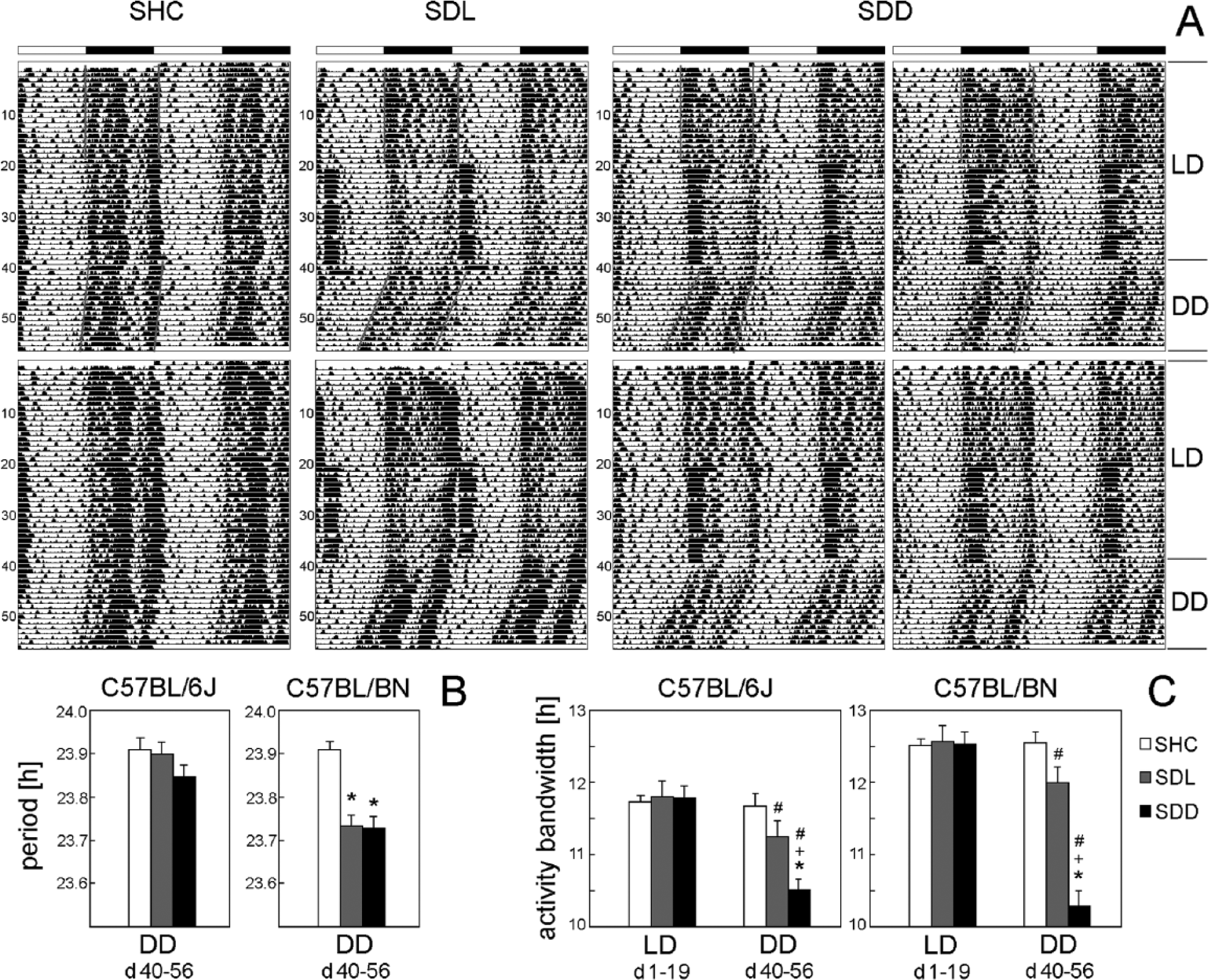
Actogram examples of C57BL/6N single-housed control (SHC), social defeat light (SDL), and social defeat dark (SDD) mice kept under DD in the poststress period (A); calculated free-running periods (B); and activity bandwidths (C) of C57BL/6J (SHC: n = 10, SDL: n = 9, SDD: n = 9) and C57BL/6N mice (n = 9 per group). Two actograms are shown for SHC and SDL mice, and 4 actograms are shown for SDD mice. Most SDD mice lost the first activity peak under DD and, consequently, showed a delayed activity onset (A). The beginning and end of activity was determined by the tool “activity on- and offsets” in ActogramJ (Schmid et al., 2011) and is indicated in the upper actograms. Activity bandwidth was calculated as the difference between activity onset and offset. Please note that activity bandwidth was narrowed also in the few SDD mice that retained the first activity peak (e.g., lower left actogram of a SDD mouse). C57BL/6J and C57BL/6N mice differed in their response to SDD stress with respect to changes in free-running period (B) and activity bandwidth (C). In general, C57BL/6N mice showed a stronger stress response than did C57BL/6J mice. Data are presented as mean + SEM. *p ≤ 0.05 versus respective SHC mice; +p ≤ 0.05 versus respective SDL mice; #p ≤ 0.05 versus LD conditions.
Discussion
To date, only a few rodent studies have addressed long-lasting effects of repeated/chronic stress on circadian functionality. Rats exposed to a chronic mild stress procedure for 4 weeks showed distinct and prolonged disturbances of diurnal and circadian activity rhythms (Gorka et al., 1996). Long-lasting decreases in the amplitude of circadian rhythms for heart rate and core body temperature have been reported following repeated SD in rats (Tornatzky and Miczek, 1993). Strikingly, although it is well known that stress effects are determined by the time of day of assessment (Janssens et al., 1995; Verma et al., 2010), it remains largely unknown whether stress-induced changes in circadian rhythms also depend on the time of day of stressor presentation. In line with a previous hamster study reporting a phase delay in the circadian activity rhythm following immobilization stress at CT12, but little or no effects at any other CT (Van Reeth et al., 1991), our data provide further evidence that the circadian system is influenced in the long term by repeated SD in such a way that the time of day at which the stressor is applied determines stress effects on the circadian system.
Consistent with previous reports (Sgoifo et al., 2001; Sgoifo et al., 2002), no habituation in stress-induced activity and body temperature rises in response to a repeatedly applied psychosocial stressor was observed in the present study. This allowed us to pool all days of SD exposure (except days 1, 8, and 15, where cages were changed). While SDL mice showed increased activity between ZT0 and ZT6 in response to stressor exposure, activity was significantly decreased in this group in the second half of the light phase (ZT6-ZT12) and during the whole dark phase. In contrast, increased active behavior of SDD mice between ZT12 and ZT18, coinciding with stressor exposure at that time, was balanced by reduced activity during the second half of the dark phase (ZT18-ZT0). Interestingly, the activity patterns on days of SD exposure resemble those of rodents on restricted feeding schedules, except that animals with food restriction actively anticipate a rewarding event, whereas in the present study, mice actively respond to a stressor. Both conditions affect clock gene rhythms in the brain (Al-Safadi et al., 2014; Minana-Solis et al., 2009; Verwey and Amir, 2009), indicating a similar effect of stress and restricted feeding on the circadian system throughout exposure time. The considerable additional activity decrease in SDL mice during the second half of the light phase (ZT6-ZT12) may compensate the elevated activity—and thus, likely, loss of sleep—in the first half of their normally inactive period. Given that SDL and SDD Per1/2 mutants exhibited identical changes with respect to activity compared with WT animals, the acute alterations throughout the stress procedure are most likely not a result of an altered internal clock system but rather a direct response to the social stressor. In contrast to earlier studies (Meerlo et al., 1996; Meerlo et al., 1997), body temperature was generally less affected by the stress procedure than activity. This effect was also visible in the poststress period and might be explained by the fact that transmitters were implanted subcutaneously in the present study, whereas in the studies mentioned above, animals were equipped with intraperitoneal transmitters. Furthermore, the average body temperature during the resting phase dropped continuously in the course of the experiment. Again, this seemed to be due to the subcutaneous implantation of the transmitters, because the mice moved the transmitters actively toward their periphery, and this might have led to a drop in the recorded body temperature.
During the poststress period under LD conditions, SDL and SDD mice quickly returned to their normal activity level. Nevertheless, the activity pattern of SDD mice was considerably different in both substrains compared with SDL and SHC mice: Most SDD animals showed a bimodal, instead of a trimodal, nocturnal activity pattern. For the remaining (still trimodal) WT SDD mice, the first activity bout after lights-off was significantly delayed compared with SHC and SDL animals in the entrained poststress period. C57BL/6N SDD mice further showed a delayed second activity bout. The latter effect was not visible in C57BL/6J animals, suggesting that the C57BL/6N substrain is more susceptible to SD stress than the C57BL/6J substrain. The stress-induced loss of the first activity bout might result from the fact that SDD mice develop a conditioned fear-related suppression of their nocturnal activity. Because the stressor exposure occurred at the same time every day, the circadian clock may have provided a time stamp that allowed the mice to anticipate this stressful event. Such a clock-induced time stamp may persist for many days even under DD, when the stressor is no longer taking place. An alternative explanation is that repeated SD affects the circadian clock per se. In favor of the second possibility is the finding that even the free-running period was affected by the repeated stressor exposure in one of the strains. Although we cannot elucidate why SD exposure shortens period and narrows activity bandwidth, our results suggest that repeated SD exposure disturbs the circadian clock. Importantly, in our previous study, though not significant, the phase of the PERIOD2::LUCIFERASE (PER2::LUC) rhythm in SCN explants of SDD mice tended toward a phase delay (Bartlang et al., 2014). The lack of significance may be due to resetting effects during the cultivation procedure (Yoshikawa et al., 2005) or the fact that PER2::LUC mice are bred on a C57BL/6J background, which, as this study suggests, are slightly less responsive to repetitive SD compared with the C57BL/6N substrain.
Following our hypothesis of an altered internal clock in WT SDD mice, we predicted a lack of circadian gating of SD effects in clock-deficient Per1/2 double-mutant mice. Indeed, Per1/Per2 mutant SDD mice showed a normal activity rhythm in the poststress period under LD conditions that was comparable to SHC and SDL mice. As is known from the literature (Zheng et al., 2001), Per1/Per2 double-mutant mice completely lost their rhythm when exposed to the poststress period under DD conditions in the present study.
Based on the fact that rhythmic activity is a reliable indicator of circadian pacemaker function, changes in the circadian phase due to repeated SD can be inferred to reflect SCN activity. Although early reports by Meerlo et al. indicated that changes in circadian output rhythms following stressor exposure do not reflect changes in the central oscillator in the SCN (Meerlo and Daan, 1998; Meerlo et al., 1997), more recent studies have provided evidence that certain stressors are capable of perturbing core clock components in the SCN. In support, we have previously reported increased PER2::LUC SCN explant rhythm amplitude and increased PER2 protein expression in the SCN of C57BL/6J and C57BL/6N mice, respectively (Bartlang et al., 2014). Moreover, a recent study revealed increased Per1 and Per2 expression in the SCN of rats in a model of posttraumatic stress disorder. These changes were reversed by treatment with the melatonin-1/2 receptor agonist/serotonin-2C receptor antagonist agomelatine (Koresh et al., 2012). Furthermore, suppressed PER2 levels in the SCN following chronic restraint stress could be normalized by the mood stabilizer lithium (Kinoshita et al., 2012). Variations in these results may be assignable to the different animal models used, and SCN clock effects appear to critically depend on the type of stressor as well as duration of stressor exposure. In summary, however, these recent studies point toward a role of the circadian Per2 gene/protein in the SCN in mood-related disorders. Interestingly, well-accepted hallmarks of affective disorders such as decreased interest in an unfamiliar conspecific, loss of general behavioral activity, and flattened diurnal corticosterone rhythms have become evident following repeated SDD (Bartlang et al., 2012). In support, polymorphisms in the Per2 gene and abnormal circadian rhythms have been associated with increased vulnerability to major depressive disorder (MDD) and seasonal affective disorder (Lavebratt et al., 2010; McClung, 2007; Partonen et al., 2007). Thus, it can be hypothesized that rhythm disturbances at least sensitize an individual to mood disorders.
Possible mechanisms underlying the more negative outcome of SD exposure during the dark versus the light phase might include the increased sensitivity of the adrenal cortex to adrenocorticotropic hormone in the active compared with the inactive phase (Buijs et al., 1999; Kalsbeek et al., 2011). In addition, based on the phase response curve (Dibner et al., 2010), resetting processes of the biological clock are likely to happen during early night, at a time that interferes with the time of SDD exposure. Thus, a resetting of the circadian clock, required for the appropriate timing of physiological and behavioral processes, might not be possible in mice repeatedly exposed to the stressor at the beginning of the dark phase.
Conclusion
In summary, our results indicate that psychosocial stressor exposure during the dark (i.e., active) phase has long-lasting consequences for the functional output of the biological clock. It is reasonable to speculate that these rhythm disturbances are ultimately linked to the maladaptive consequences of SDD (Bartlang et al., 2012). Although our data point toward an involvement of the circadian gene Per2, and possibly Per1, in the time-of-day-dependent response to stress, more detailed studies, including the analysis of other clock genes mutants, are required to further clarify this point.
Footnotes
Acknowledgements
The authors are grateful to Dr. S. Peters, L. Lucke, M. Stieglitz, L. Onderka, and A. Kühn for excellent technical help. This study was supported by the Deutsche Forschungsgemeinschaft (DFG FO-207/13-1). HO is a Lichtenberg fellow of the Volkswagen Foundation. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
