Abstract
The active form of vitamin D, 1α,25-(OH)2D3, has been associated with metabolism control, cell growth, differentiation, antiproliferation, apoptosis, and adaptive/innate immune responses, besides its functions in the integrity of bone and calcium homeostasis. The circadian rhythm regulates a variety of biological processes, many of them related to the functions associated with 1α,25-(OH)2D3. In the present study, we determine whether 1α,25-(OH)2D3 alters the expression of circadian genes in adipose-derived stem cells (ADSCs). The effect of 1α,25-(OH)2D3 on the expression of circadian genes BMAL1 and PER2 was measured by qPCR, over a 60-h period every 4 h, in serum shocked ADSCs, serum shocked ADSCs supplemented with 1α,25-(OH)2D3, and ADSCs under the presence of only 1α,25-(OH)2D3. The results showed that 1α,25-(OH)2D3 was able to synchronize circadian clock gene expression in ADSCs. The expression of circadian genes BMAL1 and PER2 in ADSCs that contained only 1α,25-(OH)2D3 has a profile similar to that found in the ADSCs synchronized by a serum shock. The results suggest an important role of 1α,25-(OH)2D3 in the regulation of the molecular clock.
The role of 1α,25-dihydroxyvitamin D3 (1α,25-(OH)2D3), the hormonally active form of vitamin D, in the integrity of bone and calcium homeostasis has been well established. In addition, 1α,25-(OH)2D3 has been associated with many important functions, such as metabolism control, cell growth, differentiation, antiproliferation, apoptosis, and adaptive/innate immune responses. 1α,25-(OH)2D3 regulates gene expression in target cells by binding to the nuclear vitamin D receptor (VDR), which is widely distributed in the body (DeLuca, 2004). Liganded VDR forms a heterodimer with the retinoid X receptor (RXR) and affects gene transcription binding to vitamin D response elements (VDREs) in collaboration with coactivators (Christakos et al., 2003).
A genetic pathway has been identified recently, underlying the observation that vitamin D deficiency alters the expression of the circadian genes NPAS2 and PER2 during osseointegration (Mengatto et al., 2011). The circadian rhythm regulates a variety of biological processes in addition to playing a role in regulating the sleep-wake cycle (Takahashi et al., 2008). The aim of this study was to determine whether 1α,25-(OH)2D3 alters the expression of circadian genes in adipose-derived stem cells (ADSCs). We provide evidence for the rhythmic expression of 2 circadian genes in ADSCs synchronized with 1α,25-(OH)2D3.
To determine whether 1α,25-(OH)2D3 affects the expression of circadian genes, we measured the expression of 2 core clock genes, BMAL1 and PER2, in serum shocked ADSCs, serum shocked ADSCs supplemented with 1α,25-(OH)2D3, ADSCs supplemented with only 1α,25-(OH)2D3, ADSCs spiked with 1α,25-(OH)2D3, and ADSCs under the presence of free-serum medium as control. Under the same conditions, we also measured the expression of 2 clock output genes, DEC1 and BGLAP, which are involved in cell differentiation and bone homeostasis, respectively. ADSCs were isolated and cultured as previously reported (Cuevas-Diaz Duran et al., 2013). ADSCs were grown until they reached confluence and were then exposed to 5 different experimental conditions. ADSCs were serum shocked for 2 h with 50% horse serum as previously reported for other cell lines (Balsalobre et al., 1998). ADSC medium was replaced with serum-free DMEM-F12 or with DMEM-F12 containing 1α,25-(OH)2D3 at a final concentration of 50 nM. In other conditions, ADSCs were treated for 2 h with DMEM/F12 supplemented with 50 nM 1α,25-(OH)2D3, and then media were replaced with serum-free DMEM-F12 or with DMEM-F12 supplemented only with 1α,25-(OH)2D3. The fifth condition included serum-free DMEM-F12 as a control. ADSCs were harvested every 4 h over a 60-h period for the 5 mentioned experimental conditions. Total RNA was isolated for the triplicate cultured ADSCs and reverse-transcribed into cDNA. SYBR Green–based quantitative real-time PCR (qPCR) was used to measure the levels of target genes and GAPDH as an internal control gene. Relative expression ratio was calculated as previously described (Pfaffl, 2001). The detailed procedures are available in the supplementary online material.
The most important result was that 1α,25-(OH)2D3 was able to synchronize circadian clock gene expression in ADSCs, as observed through the oscillation of the BMAL1 and PER2 genes. The expression profiles of circadian genes BMAL1 (Figure 1) and PER2 (Figure 2) in ADSCs were consistent in each of the experimental conditions. The rhythmic expression of BMAL1 and PER2 genes showed different amplitudes for each condition, while the control had a more stable profile throughout the 60-h period. The 1α,25-(OH)2D3-treated conditions showed higher amplitude in PER2 expression than in the serum shocked cells, while the BMAL1 expression profile from serum shocked cells showed a higher amplitude than the 1α,25-(OH)2D3-treated cells, even though the control showed no amplitude in all the mentioned conditions. A 2-h treatment with spike in the 1α,25-(OH)2D3 condition was enough to synchronize ADSCs, but a lower amplitude was observed for the BMAL1 gene expression profile compared with a constant presence of 1α,25-(OH)2D3 in the medium. Although the serum shock and 1α,25-(OH)2D3 conditions were able to synchronize the expression of BMAL1 and PER2 genes in ADSCs, there was no additive effect on the gene expression profiles when the 2 treatments were used together. Also, the expression profiles of BMAL1 and PER2 were in opposite phases, as expected.
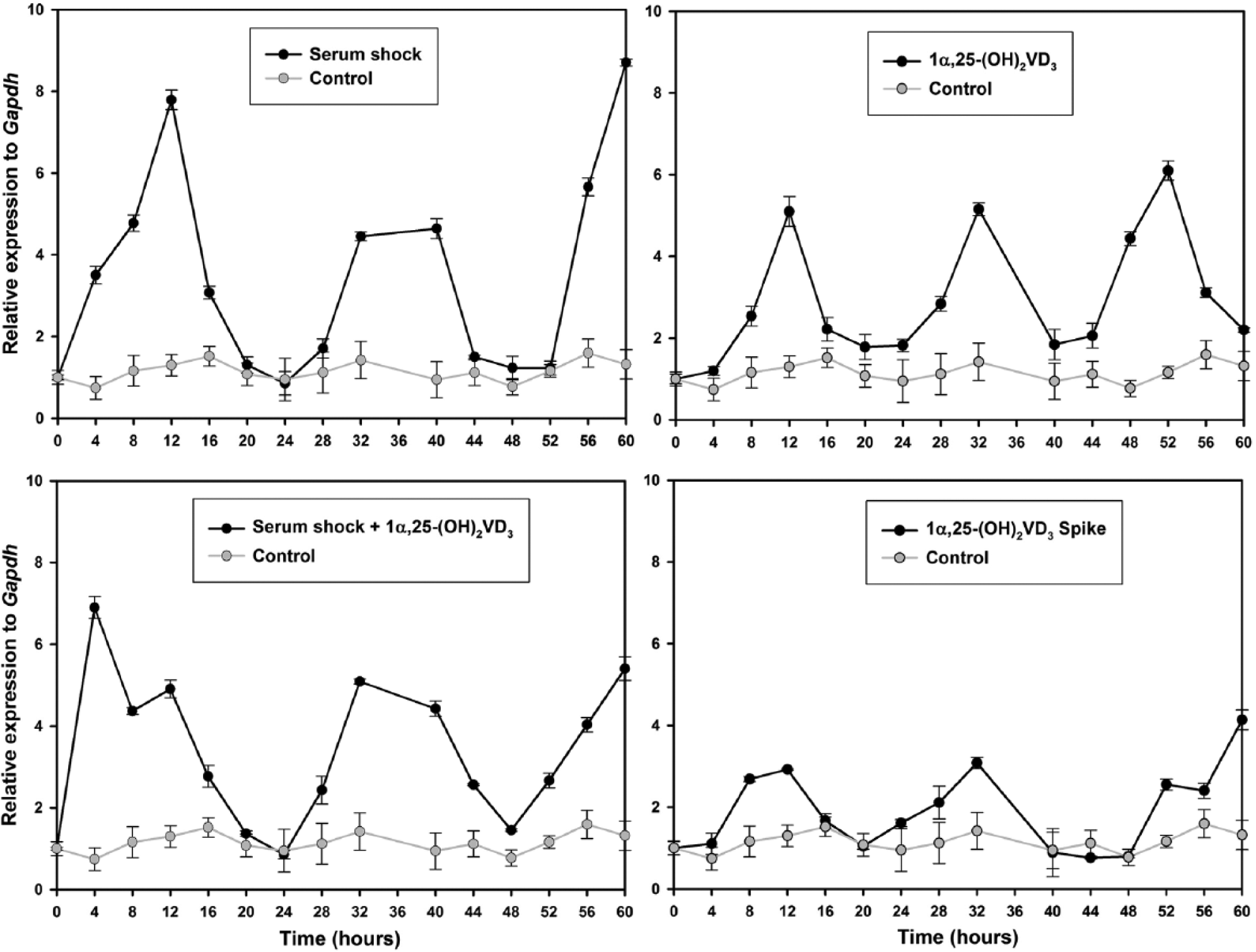
Temporal expression profile of BMAL1 gene in synchronized ADSCs. The expression of BMAL1 gene in serum shocked ADSCs (upper left), serum shocked ADSCs supplemented with 1α,25-(OH)2D3 (lower left), ADSCs under the constant presence of 1α,25-(OH)2D3 (upper right), and ADSCs exposed to 1α,25-(OH)2D3 for 2 h (lower right) was measured by qPCR. Samples were collected every 4 h for 60 h. Time point 36 was lost. Data points (mean of triplicates ± standard derivation) were normalized using GAPDH as internal control gene and to the first time point (t = 0). Fold expressions from the control condition (gray) were repeated in each treatment condition (black) for comparative purposes.
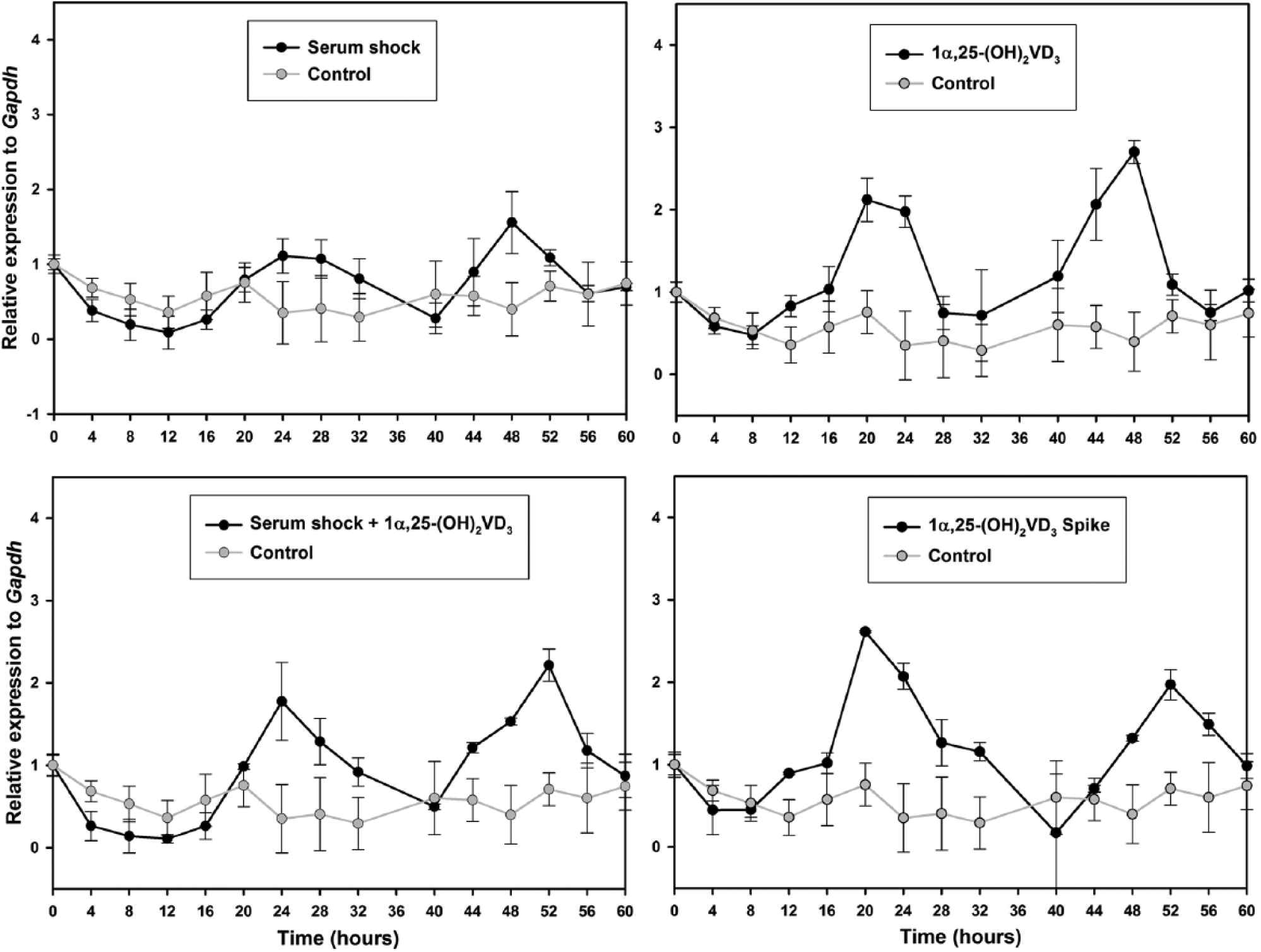
Temporal expression profile of PER2 gene in synchronized ADSCs. The expression of PER2 gene in serum shocked ADSCs (upper left), serum shocked ADSCs supplemented with 1α,25-(OH)2D3 (lower left), ADSCs under the constant presence of 1α,25-(OH)2D3 (upper right), and ADSCs exposed to 1α,25-(OH)2D3 for 2 h (lower right) was measured by qPCR. Samples were collected every 4 h for 60 h. Time point 36 was lost. Data points (mean of triplicates ± standard derivation) were normalized using GAPDH as internal control gene and to the first time point (t = 0). Fold expression from the control condition (gray) were repeated in each treatment condition (black) for comparative purposes.
PER2 and BMAL1 work in opposition to each other. Their expression is regulated by an autoregulatory transcription-translation feedback loop. The heterodimer CLOCK:BMAL1 binds to E-box elements in the promoter region of PERs and CRYs genes. PERs and CRYs proteins heterodimerize to inhibit the CLOCK:BMAL1 complex (Takahashi et al., 2008). The 1α,25-(OH)2D3 ligand promotes VDR:RXR heterodimerization and the three together adjust the activation or repression of genes (Orlov et al., 2012). It is known that RXR can create a negative regulatory mechanism of the circadian feedback loop by inhibiting CLOCK:BMAL1-dependent transcriptional activation of an E box–containing circadian enhancer element (McNamara et al., 2001). This receptor-mediated negative regulation of the circadian clock might explain the mechanism through which 1α,25-(OH)2D3 regulates circadian gene oscillation. These findings establish a model whereby circadian gene expression mediated by nuclear receptors may be influenced by other steroids and vitamins.
The genes DEC1 and BGLAP (Suppl. Figs. S1 and S2, respectively) showed no circadian fluctuation in any of the experimental conditions. The expression profiles were similar to the basal expression found in the control condition. These results showed that the expression of DEC1 and BGLAP genes was not affected by the treatments in ADSCs. DEC1 suppresses CLOCK:BMAL1-enhanced promoter activity, but its role in the circadian system of mammals remains unclear. It is suggested that DEC1 plays a role in the finer regulation and robustness of the molecular clock (Nakashima et al., 2008). Also, it is reported that 1α,25-(OH)2D3 might regulate expression of circadian genes by the VDR-dynamic regulation of the DEC1 gene (Seuter et al., 2013). In this study, the DEC1 gene showed no circadian expression in the 60-h period, suggesting that this indirect mechanism is not responsible for BMAL1 and PER2 circadian fashion expression in ADSCs. To our knowledge, there are no other studies showing the fluctuation of the DEC1 gene in ADSCs.
It is known that 1α,25-(OH)2D3 induces osteogenic differentiation of ADSCs (Jing et al., 2010) and is able to enhance their osteogenic potential by acting via VDR. The regulation of BGLAP, which encodes for osteocalcin, is mediated by the VDR complex, along with other bone markers such as RUNX2, BMP2, and BMP6. The proteins encoded by these genes are rhythmically expressed in serum (Witt-Enderby et al., 2012) and are important for bone homeostasis and osteogenesis, and their expression in ADSCs treated with 1α,25-(OH)2D3 long-term indicates that it plays a role in osteogenic differentiation. These findings suggest that vitamin D may require the circadian machinery to start the process of osteogenic cell differentiation. In this study, BGLAP expression was not affected by the 1α,25-(OH)2D3 treatments, and this could be because the time of 1α,25-(OH)2D3 exposure was shorter than the time used for induction of osteogenic cell differentiation. Furthermore, other well-used cell differentiation inductors, such as forskolin or dexamethasone, are used to synchronize the molecular clock in cell cultures.
Entrainment of circadian rhythms in cell cultures by 1α,25-(OH)2D3 could explain the protective effects of vitamin D in the skin. Direct exposure to UVB light can disrupt the circadian rhythm in normal skin, leading to pathologies such as skin cancer, due to a disruption of cellular circadian-related processes. Also, direct exposure to UVB light has the protective effect of photoproducing vitamin D3 (Desotelle et al., 2012). These 2 phenomena initiated by UVB light may interact to maintain homeostasis of the skin. These findings highlight the role of 1α,25-(OH)2D3 in the regulation of circadian rhythm.
In conclusion, gene expression profiles of BMAL1 and PER2 over a 60-h period in synchronized ADSCs using 1α,25-(OH)2D3 suggest an important role of this hormone in the maintenance of circadian rhythms at the cellular level. Since circadian rhythm influences many physiological processes in all living organisms, this could be the key to better understand the mechanisms by which 1α,25-(OH)2D3 performs many of its assigned functions. Furthermore, our findings demonstrate that adult mesenchymal stem cells may be synchronized with either serum shock or 1α,25-(OH)2D3, suggesting relationships between circadian oscillations and stem cell properties such as pluripotency and proliferation. It might be beneficial to synchronize circadian clock gene expression in stem cells before using a differentiation protocol in order to synchronize the expression of transcription factors that could regulate the fate of stem cells.
Footnotes
Acknowledgements
Gutierrez-Monreal and Cuevas-Diaz express their gratitude to the Mexican National Council for Science and Technology (CONACYT) for the PhD grants scholarship CVU269963 and CVU359186, respectively. This work was funded by Catedra de Hematologia y Cancer and Catedra de Terapia Celular from the Tecnologico de Monterrey.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
