Abstract
Predictable daily activity patterns have been detected repeatedly even in mammals living in stable environments, as is the case for subterranean rodents. Whereas studies on activity of these rodents under laboratory conditions almost exclusively have concerned themselves with the influence of light, many field studies have revealed signs of an association between the activity pattern and daily fluctuations of temperature under the ground. This would assume that behavioral thermoregulation is probably involved. The only exceptions to the relationship between temperature and activity are 2 eusocial mole-rats of the genus
Keywords
Most mammals display circadian rhythms of activity and rest, which ensure that activity takes place during an optimal period of the day in terms of food availability, predation risk, and climatic conditions (Bartness and Albers, 2000). In mammals living in a stable environment with lack of external cues that would potentially reset their circadian clocks, we would expect circadian activity rhythms to be weak or absent. Examples of such mammals are subterranean rodents, that is, rodents that forage underground and rarely emerge from their sealed burrows. The subterranean ecotope is dark and thermally buffered, and neither food availability nor predation risk is thought to substantially vary there during the course of a 24-h cycle (Nevo 1999). It is therefore surprising that many field studies regarding subterranean rodents have exhibited predictable daily activity patterns (e.g., Jarvis, 1973; Gettinger, 1984; Benedix, 1994; Šklíba et al., 2007; Lövy et al., 2013). Some authors have suggested that there is a relationship between activity patterns and daily temperature cycles in the depth of the burrows, even though its amplitude is often very small (typically <4 °C). There are few laboratory data dealing with the effect of temperature on the activity pattern in these animals (cf. Goldman et al., 1997), which contrasts with many studies testing merely the influence of the photoperiod (e.g., Oosthuizen et al., 2003, and citations therein).
The relationship between activity patterns and daily temperature fluctuations that has been detected in some free-living subterranean rodents has the character of either preference or avoidance of high or low temperatures, depending on the species, latitude, or the season, as can be illustrated by the following examples. Whereas blind mole rat
African mole-rats (Bathyergidae) are a family of strictly subterranean rodents of which the majority of the species are highly social, living in multigenerational family groups. This provides us with an opportunity to study the effect of sociality in addition to environmental factors on these animals’ daily activity patterns. Strong social influence on behavioral rhythms is common in social animals living in constraining environments, but it can be expressed in different ways, such as synchronization, desynchronization, or individually variable response to social cues depending, for example, on dominance status (Favreau et al., 2009). In highly cooperative mole-rat family groups, we would expect desynchronized rather than synchronized activity patterns, presumably enabling more effective cooperation in search for food and territory maintenance. This is also in accord with most laboratory observations (Bennett, 1992; Riccio and Goldman, 2000; Schielke et al., 2012; but see Davis-Walton and Sherman, 1994, for signs of activity synchronization in naked mole-rats). Field-based activity data are available from only 2 of the social species, Damara mole-rat
The 2 above-mentioned species,
To better understand the relative importance of social and environmental cues for the mole-rat activity pattern, we chose to study the activity of the Ansell’s mole-rat
Materials and Methods
Studied Animals
The Ansell’s mole-rat is a small-sized social species inhabiting a mesic area close to the Zambian capital Lusaka. In the study population, the body masses of adults (≥50 g) were 72 ± 14 g (maximum 96 g) for males and 62 ± 8 g (maximum 83 g) for females. Family groups of up to 13 individuals inhabited extremely large and complicated burrow systems, with the majority of the tunnels located at a depth (from the bottom of the burrow to the soil surface) of around 11 cm (foraging tunnels). A single communal nest per family group was located 47 ± 17 cm (range, 25-90 cm) underground (Šklíba et al., 2012).
Study Locality
The study was conducted in the Lusaka East Forest Reserve in Zambia (15°28′S, 28°25′E, altitude 1320 m), which is covered by natural open-canopy miombo woodland. The climate in Zambia is characterized by 3 seasons: a rainy season (October/November to March/April), a dry cold season (April to July), and a dry hot season (August to October). Average annual rainfall in Lusaka is 809 mm, and on average 5.4 months per year have more than 25 mm of precipitation (http://www.ncdc.noaa.gov/ghcnm/, accessed May 2012).
Fieldwork
Mole-rats were captured by use of Hickman traps. Captured individuals were weighed, their sex was determined, and they were examined for their reproductive status. We considered breeding males as those with conspicuous head muscularity, pigmented corners of the mouth, and conspicuously large testes; breeding females were recognized by perforated vagina and enlarged teats (the breeding status was later confirmed by use of genetic methods, see Patzenhauerová et al., 2013). All animals weighing more than 45 g were anesthetized for a short time by ketamine and xylazine, fitted with radio-collars (brass collar, Pip transmitter; Biotrack Ltd, Dorset, UK), and released back into the burrows where they were trapped within 48 h from capture. The weight of each radio-collar was 2.3 g (<5% of the body mass of the smallest animals under study). Radio-tracking started 3 days after the release of the last animal.
The collared animals were radio-tracked in 2 continuous 96-h sessions. The first session (9-13 May 2010) involved 17 mole-rats from 5 family groups (named P01, P02, etc.; Table 1), which was reduced to 13 individuals in the second session (25-29 May 2010) due to the loss of some collars. Both sessions were in the cold dry season. We used the IC-R20 receiver (Icom America Inc., Kirkland, WA) and a 3-element handheld Yagi antenna to locate the animals. Position of the animals was fixed hourly starting at 0600 h. Since the animals of the same group used the same nest, these places were checked first, and then the animals that were not present here were carefully approached to a distance of 1 to 4 m and fixed. After the end of radio-tracking, all members of the examined family groups were recaptured and the complete burrow systems were excavated and mapped as a part of another study (see Šklíba et al., 2012, for complete information about group sizes, burrow system parameters, ecological parameters, etc.). Handling live animals was in accord with the Declaration of Helsinki and the U.S. National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Characteristics of radio-tracked mole-rats.
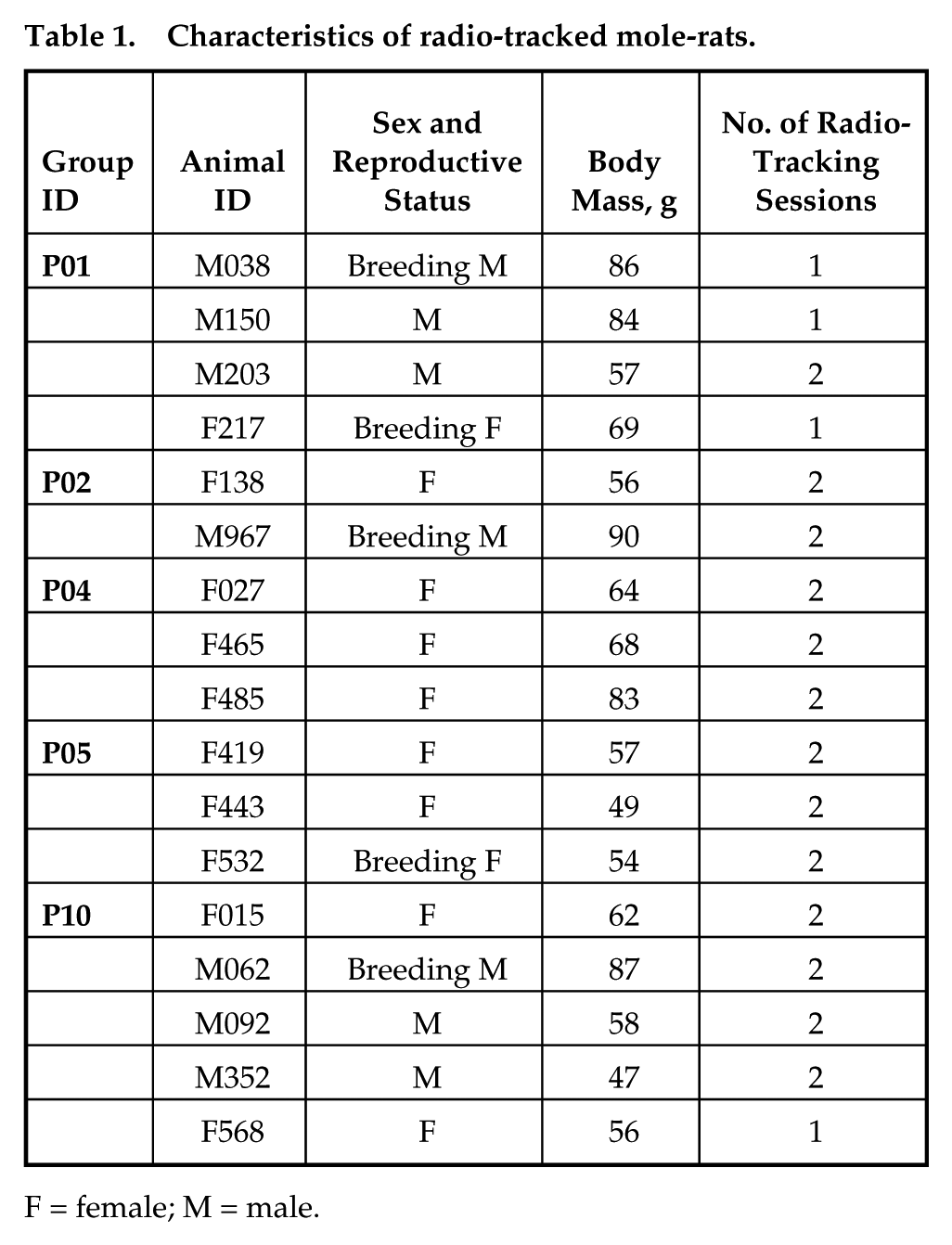
F = female; M = male.
Temperatures of the soil surface and at depths of 10, 20, 30, 60 and 120 cm were recorded every 10 min over the whole period of radio-tracking using temperature loggers (Comet System s.r.o., Rožnov pod Radhošteˇm, Czech Republic). The depth of 10 cm was chosen to approximate the depth of mole-rat foraging tunnels (Šklíba et al., 2012); the other depths were selected to illustrate lag and attenuation of the temperature fluctuation as a function of the distance from the soil surface. We selected a relatively exposed site for measuring the temperatures, because most of the area of the mole-rat territories was also relatively exposed to the sun. For comparison we placed 1 additional temperature logger at a depth of 10 cm at a site shaded by dense shrubs and trees.
Data Processing
To determine daily patterns of activity of the radio-tracked mole-rats, each radio-fix was designated as either inside or outside the nest. From our previous telemetry studies of mole-rats (Šklíba et al, 2007, 2009), we estimate the accuracy of our fixes at ±0.5 m; thus, all fixes within a 0.5-m radius of the nest were treated as inside the nest. For each animal, an actogram of the outside nest activity was created based on the primary dataset containing the hourly presence or absence of an individual inside the nest. This dataset was also used to test the relationship of outside-nest activity with environmental factors and to test the activity synchronization of individuals within the same group. Subsequently, radio-fixes of each individual from each 96-h radio-tracking session were grouped into twelve 2-h blocks, and the proportion of fixes outside the nest was determined for each of the blocks. This dataset was used to compare activity patterns between individual mole-rats using a multivariate method.
Statistical Analyses
Data throughout the text are provided as mean ± SD. Generalized linear mixed models (GLMMs) were used to test the effect of various environmental factors to activity—that is, presence outside the nest. Presence outside the nest was the dependent variable with binomial distribution. The factors tested (explanatory variables) were light-dark 24-h cycle (categorical predictor: the light hours [0600-1800 h] marked as 1, dark hours [1800-0600 h] marked as 0) and temperatures at various depths (continual predictors; surface, 10, 20, 30, and 60 cm). The temperature at 120 cm underground was not included since its daily amplitude was close to the resolution of the temperature loggers. The 17 radio-tracked individuals were grouped into the following 9 subsets reflecting their sex, reproductive status, and group affiliation: (1) breeding males, (2) breeding females, (3) nonbreeding males, (4) nonbreeding females, and (5-9) members of the respective family groups. Due to the unequal sample size in the categories and their overlap, GLMMs were performed separately for each of the categories. In all GLMMs, both the individuality of a given animal and the radio-tracking day (1-8) were treated as random factors to avoid pseudoreplications. We used Bonferroni procedure to correct the significance level of the tests since we tested the 6 factors separately (α = 0.05 / 6). All the tests were performed using R statistical software (R Development Core Team, 2013) extended with the lmer4 package (Bates et al., 2013), which is designed for models with incorporated random factors.
Partial effect of 3 variables (body mass, sex × reproductive status, and group affiliation) on the mole-rat activity patterns (defined by proportions of outside-nest fixes in the twelve 2-h blocks of the 24-h day) was computed by variance partitioning technique (Lepš and Šmilauer, 2003) using the redundancy analysis (RDA). Sex × reproductive status and group affiliation were expressed as sets of 4 and 5 binary (“dummy”) variables, respectively. The RDA was performed with the software package CANOCO for Windows, version 4.52 (Microcomputer Power, Ithaca, NY) (ter Braak and Šmilauer, 2002), with the proportions of outside-nest fixes arcsine transformed and all variables entering the analyses centered (to have zero mean). To avoid pseudoreplications (treating 2 sessions of the same individual as independent), the variance partitioning was performed with data from the first radio-tracking session only.
To test whether the outside-nest activity of individuals of the same group is synchronized, we developed a simple permutation test. For each group and radio-tracking session we computed score
Results
Characteristics of the radio-tracked mole-rats are presented in Table 1; their actograms are depicted in Figure 1. All individuals except 1 breeding male (M967) performed a single daily peak of activity, usually between 1100 and 1600 h. There was no apparent temporal shift of the activity peak between the 2 radio-telemetry sessions. In both of them, the activity was culminating (i.e., the most animals were active) around 1400 h (Figs. 1 and 2a). Besides demonstrating a peak at about 1400 h, the breeding male M967 performed 2 additional daily activity peaks—in both radio-tracking sessions around 0100 h and 0600 h.
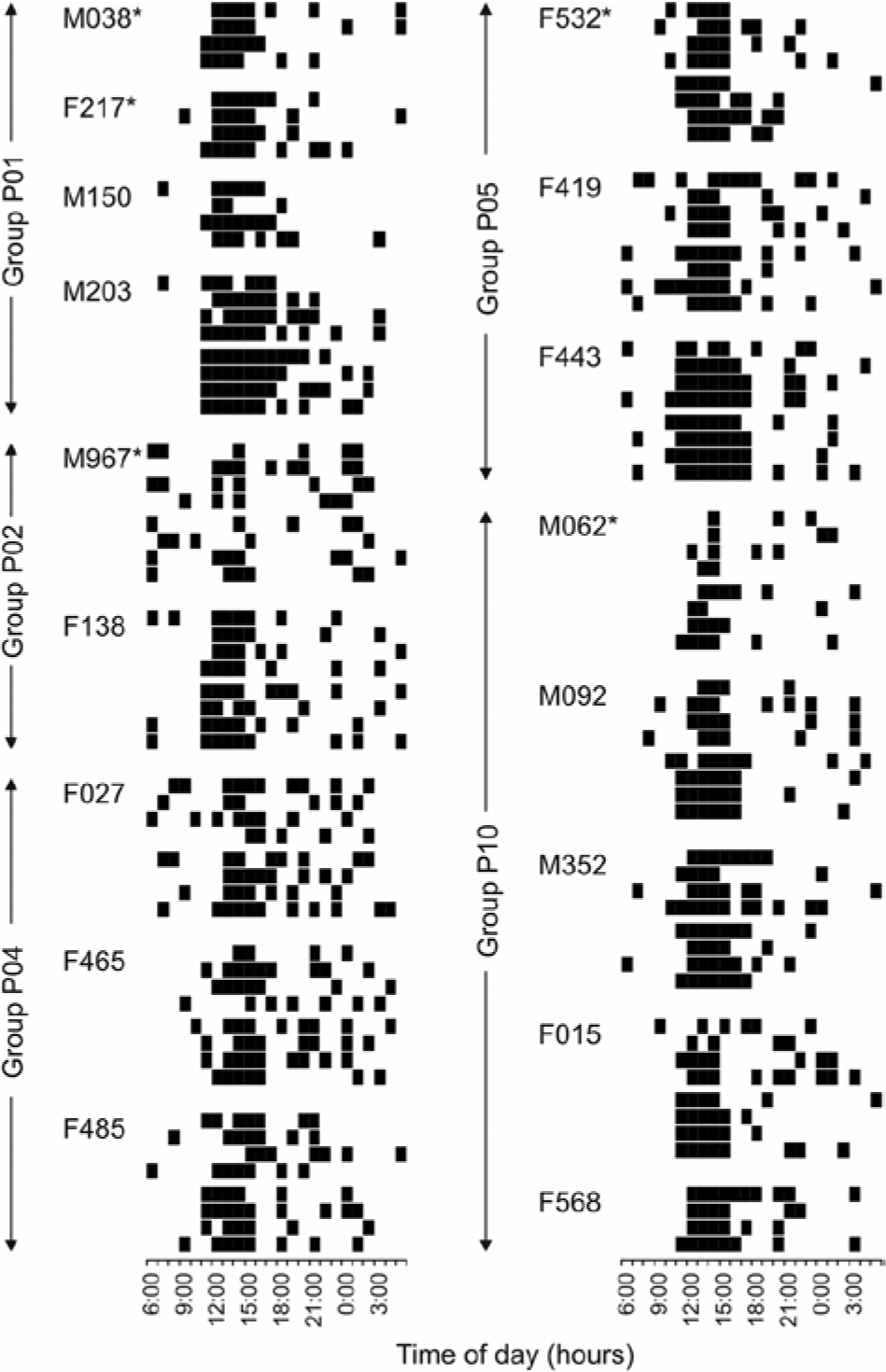
Actograms of 17 radio-tracked mole-rats. Dark bars represent activity (occurrence outside the nest). Every block of 4 lines represents 1 continuous 96-h radio-tracking session of a particular individual. Asterisks mark breeding individuals.
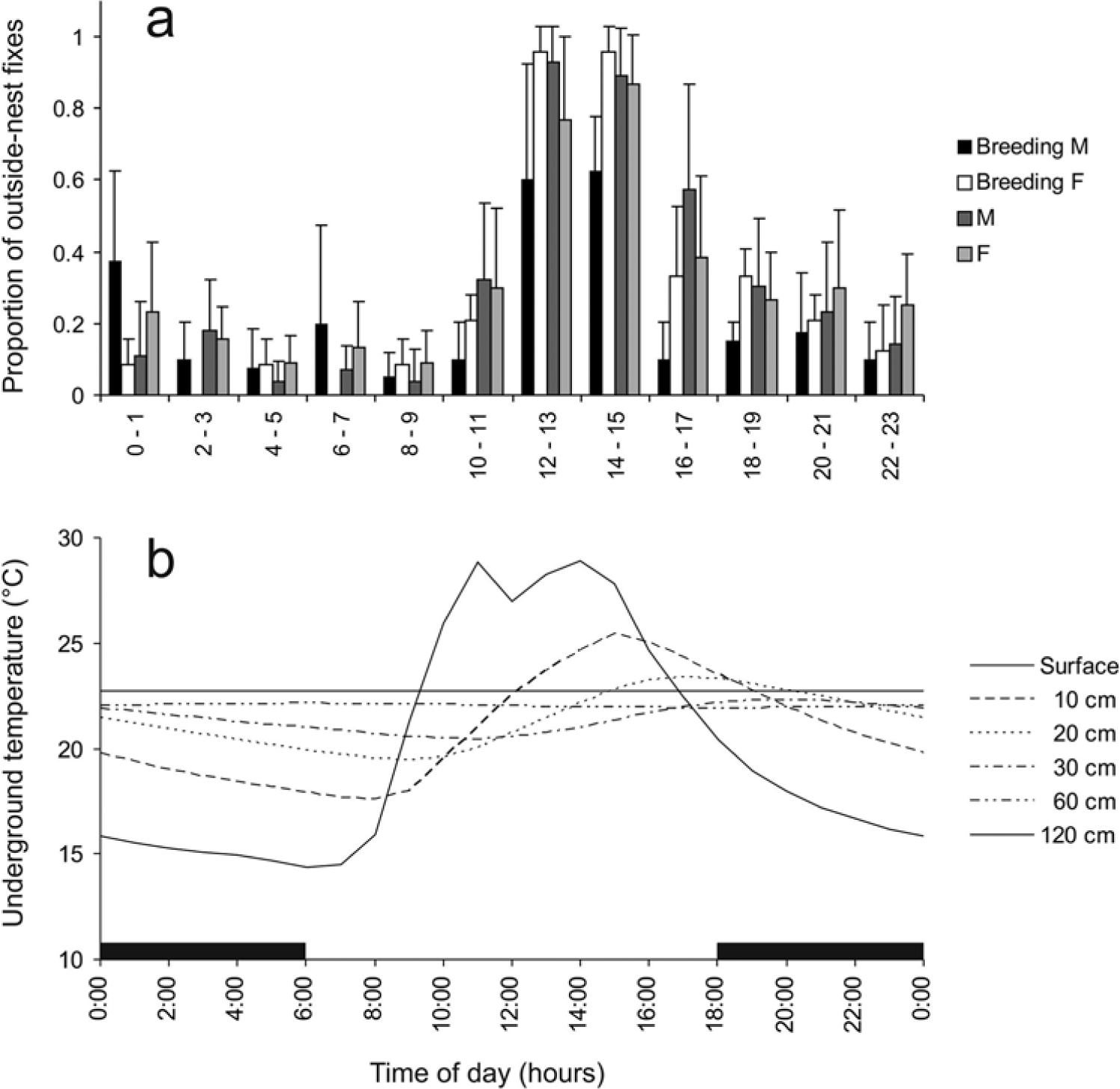
Outside-nest activity of the radio-tracked mole-rats (a) and underground temperature fluctuations (b) during a 24-h day. Means ± standard deviations are presented for the proportions of outside-nest fixes. Black horizontal bars indicate the dark phase of the day. F = female; M = male.
Mean daily temperatures at different depths are depicted in Figure 2b. At a depth of 10 cm, which corresponds with the depth of the Ansell’s mole-rat foraging tunnels, mean daily temperature was 20.9 °C; mean daily minimum and maximum temperatures were 17.5 and 25.5 °C, respectively; and mean daily temperature span was 8.0 °C. In the shaded location, the mean daily temperature at a depth of 10 cm dropped to 18.2 °C and the daily temperature span decreased to 1.8 °C. The outside-nest activity in Ansell’s mole-rats was best explained by the temperature cycle at a depth of 10 cm. Of all 6 explanatory variables, this variable explained the largest proportion of variability in the activity in each of the 9 subsets of individuals defined by sex, reproductive status, and group affiliation (Table 2). The effect of the light-dark cycle, although also significant, had markedly less explanatory power. In reproductive males, a markedly lower proportion of explained variability in the activity data indicates a less pronounced effect of all variables examined.
Relationship of outside-nest activity of the radio-tracked Ansell’s mole-rats to environmental variables as revealed by the generalized linear mixed model with binomial distribution.
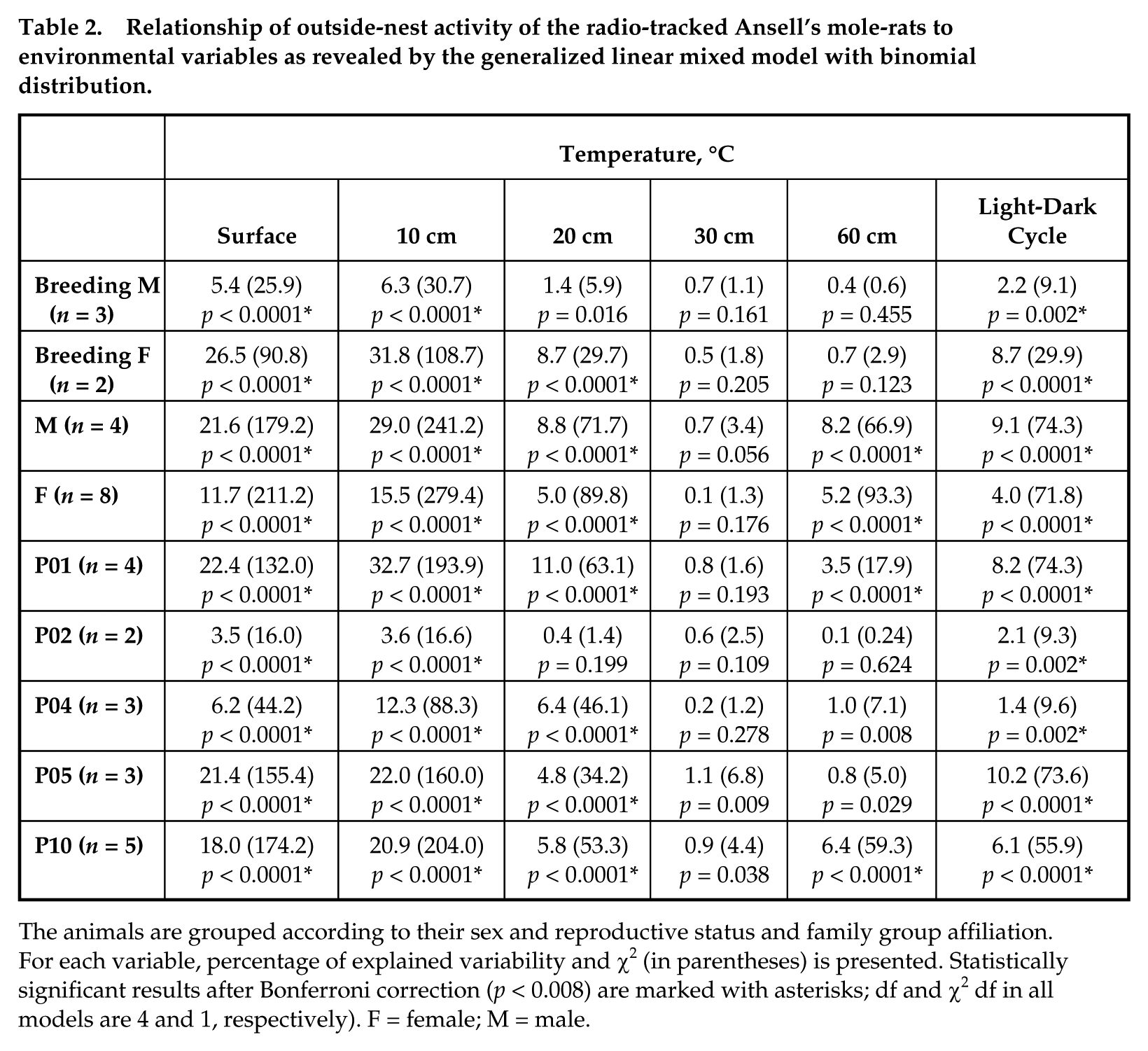
The animals are grouped according to their sex and reproductive status and family group affiliation. For each variable, percentage of explained variability and χ2 (in parentheses) is presented. Statistically significant results after Bonferroni correction (
The amount of variability in the activity patterns explained by sex × reproductive status, body mass, and group affiliation was significantly higher than zero only for the dataset where both radio-tracking sessions were combined—that is, including pseudoreplications (Table 3). Group affiliation explained the largest part of the variability of the data, which again was significant only for the dataset containing both radio-tracking sessions.
Quantification of the effects of the 3 variables on the temporal activity pattern of the Ansell’s mole-rat as computed by the variance partitioning technique (Lepš and Šmilauer, 2003) using the redundancy analysis with the data set containing only the first radio-tracking session.
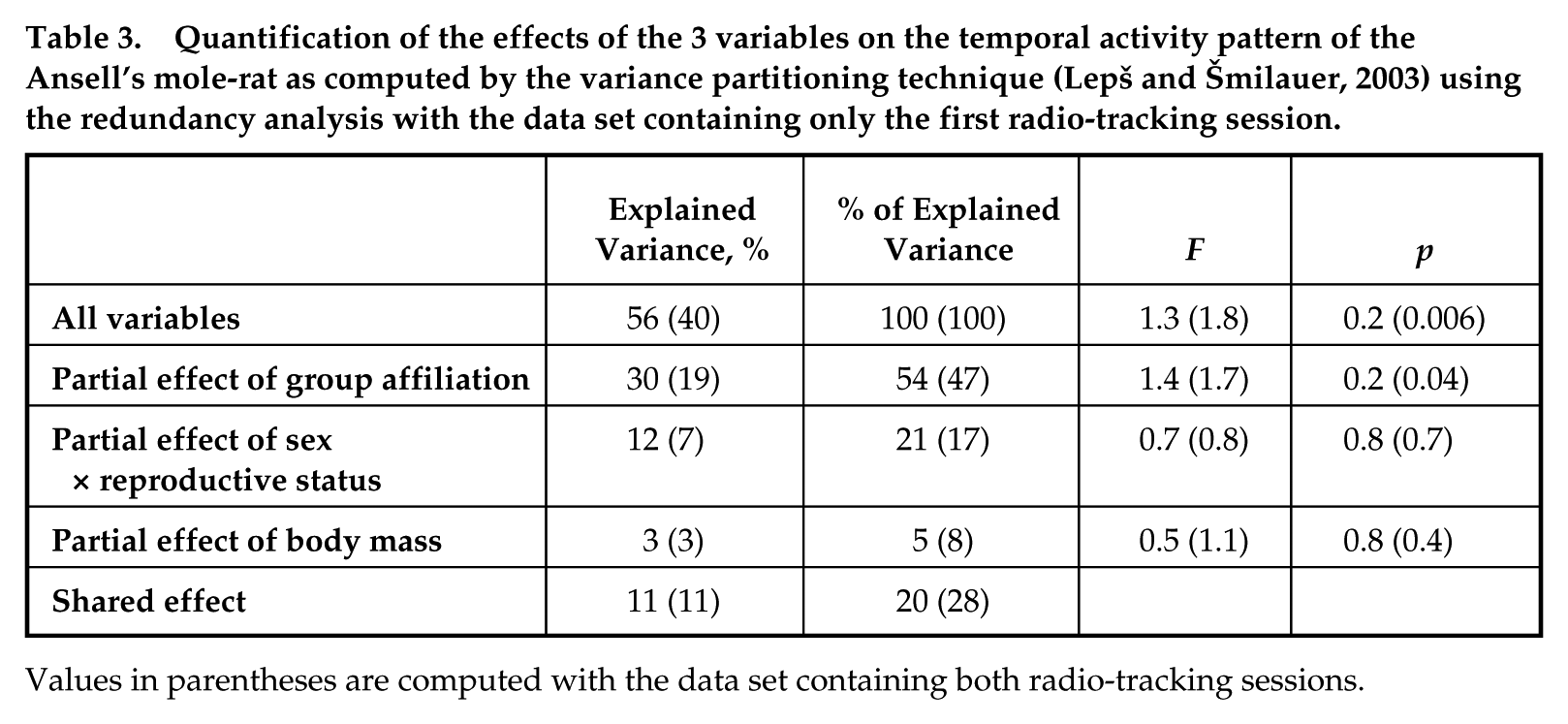
Values in parentheses are computed with the data set containing both radio-tracking sessions.
Outside-nest activity of group members was significantly synchronized only in the first session of group P01 and second session of group P04 (permutation test,
Discussion
The Ansell’s mole-rat was previously reported to have “a weak circadian clock” based on a study of singly-housed captive individuals under artificial light regime (De Vries et al., 2008) and only a “tendency towards rhythmicity” in locomotory activity based on monitoring of captive family groups under natural (European) light cycle (Schielke et al., 2012). On the contrary, the activity patterns recorded under natural conditions in the present study showed a clear 24-h periodicity in most individuals (Fig. 1). Since the pattern was similar in all 5 family groups studied and all individuals irrespective of body mass, sex, and reproductive status, we assume that it was determined by an external cue. As expected, of the external factors tested, the best predictor of outside-nest activity appeared to be temperature fluctuations at depth of the foraging tunnels.
An animal activity pattern under natural conditions can differ considerably from the one observed in a laboratory, as illustrated by an example of the coruro
Activity of subterranean rodents includes active digging, which, especially in hard soils, requires a manifold increase in the metabolic rate. This suggests a high potential for heat production and presumably a risk of hyperthermia (Vleck, 1979). We would therefore expect that subterranean rodents would prefer to be active during parts of day when the ambient temperature is lower than average. Exactly the opposite was found in our study of the Ansell’s mole-rat. What could be the reason for this behavior?
It is clear that the energetically costly activities, such as excavation of burrows, pushing soil within the burrows, and pushing soil aboveground, are probably not performed throughout the total time an individual spends outside the nest. Exploration or patrolling the burrow systems, as well as activities such as feeding and carrying small food items into food stores, could also take considerable time. Nevertheless, most digging and soil pushing activities still occurred during a period of high daily temperature at depths of the surface tunnels (J. Šklíba, unpublished data, 2010).
Activity patterns with higher activity in the warmer part of the day and/or lower activity in the colder part are surprisingly common in so far studied rodents with dominant subterranean activity (see Table 4 for data from the available literature). The burrow temperatures given in the corresponding studies (including the present one) are usually well below the lower critical temperature of the thermoneutral zone of the studied animals (Table 4), which means that activities at these temperatures require an increased energy expenditure, and thus the most plausible explanation for the temperature-related activity patterns seems to be energetic savings on thermoregulation (cf. Benedix, 1994). Comparable laboratory data on strictly subterranean rodents kept under artificial temperature cycles are, to our knowledge, limited to
Relationship of activity patterns of free-living subterranean rodents with temperature fluctuation and selected physiological characteristics of studied species connected to their thermoregulatory capabilities.
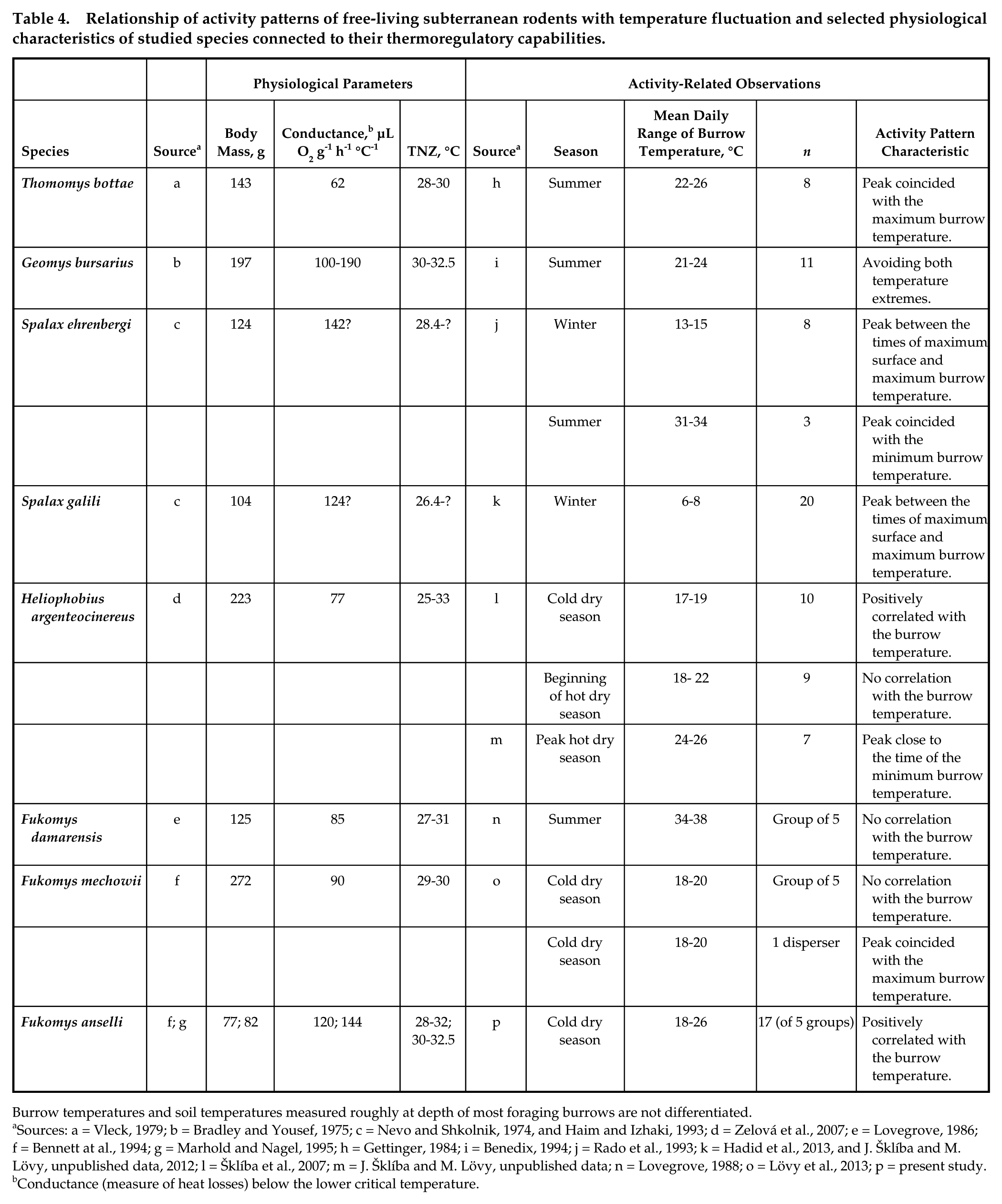
Burrow temperatures and soil temperatures measured roughly at depth of most foraging burrows are not differentiated.
Sources: a = Vleck, 1979; b = Bradley and Yousef, 1975; c = Nevo and Shkolnik, 1974, and Haim and Izhaki, 1993; d = Zelová et al., 2007; e = Lovegrove, 1986; f = Bennett at al., 1994; g = Marhold and Nagel, 1995; h = Gettinger, 1984; i = Benedix, 1994; j = Rado et al., 1993; k = Hadid et al., 2013, and J. Šklíba and M. Lövy, unpublished data, 2012; l = Šklíba et al., 2007; m = J. Šklíba and M. Lövy, unpublished data; n = Lovegrove, 1988; o = Lövy et al., 2013; p = present study.
Conductance (measure of heat losses) below the lower critical temperature.
Since the high relative humidity in the burrows of subterranean rodents complicates evaporative cooling, it might be necessary for these animals to be better physiologically adapted to cope with high burrow temperatures than with low temperatures, as they need to spend some time per day digging and pushing soil in surface tunnels. (Subterranean rodents were rarely reported to build food caches large enough to span extended periods of time such as the hot dry season in tropical Africa; Šklíba et al., 2011, and references therein.) The lack of an effective cooling mechanism makes higher temperatures potentially more fatal than lower ones, which can be coped with by torpor (a mechanism also known in
The herein described activity patterns in Ansell’s mole-rats come from the cold period of the year. Whereas the temperature inside the nests is probably constant year-round, because of depth, communal nesting, and a large amount of bedding (Šklíba et al., 2012), seasonal variation of temperature at shallower depths can be prominent (e.g., Šumbera et al., 2004). Seasonal changes in daily activity pattern of mole-rats would thus be a very effective tool for saving energy on thermoregulation while reducing the risk of overheating. This kind of seasonal change in the activity pattern was described in
Daily temperature fluctuations are substantially buffered, even in top layers of the soil (Bennett et al., 1988), so it might be surprising that even small temperature differences would still affect activity patterns of subterranean rodents. Gettinger (1984) found that peak activity of the pocket gopher
A question remains as to whether the daily activity pattern detected in this species under natural conditions indicates entrainment to a temperature change as a nonphotic zeitgeber or represents masking. This is a challenge for future laboratory studies, but one point should be taken into account—the communal nest of social mole-rats is probably temperature-stable (Šklíba et al., 2012; Šumbera et al., 2012). This means that a change of ambient temperature would hardly trigger activity of an individual that is resting inside. The entrainment of circadian rhythms by temperature changes in the tunnels near the surface thus seems to be more plausible explanation for the observed pattern than masking. Impact of temperature changes on resetting circadian clock was also proposed in
In the present study, the group affiliation explained a larger part of the interindividual variability in the activity pattern than did sex × reproductive status and body mass. Members of the same family group thus tended to be more similar in their activity patterns, as visible even in their actograms: For example, group P01 started to be active slightly earlier than group P04 (Fig. 1). Therefore, we would assume that individuals within a group are rather synchronized in their outside-nest activity. Surprisingly, a significant synchronization of the individuals’ outside-nest activity was detected only in 2 of the 5 family groups. We would need more frequent activity sampling to address this problem in greater detail. Only an indistinctive synchrony of sleep and wakefulness was detected in captive family groups of
The activity pattern of
Footnotes
Acknowledgements
We thank the Zambian Wildlife Authority for permission to conduct our research in Zambia. We thank the Department of Forestry, Lusaka, for permission to work in the Lusaka East Forest Reserve. We acknowledge the great help of Laurence Griffioen and Simon and Cherryl Burgess, who kindly provided us with accommodations and helped us with the logistics. Our thanks go also to Richard Klee for correcting the English and Martin Zatz and 3 anonymous reviewers for useful comments on the manuscript. The study was funded by GAAV (KJB601410826 for J. Šklíba) and GACˇR (P506/11/1512 for R. Šumbera).
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
