Abstract
The duper mutation in Syrian hamsters shortens the free-running period (τDD) of locomotor activity by approximately 1 h when expressed on the wild-type background and by 2 h on the tau mutant background (“super duper”). In either case, duper markedly amplifies the phase response curve (PRC) of the light pulse. This work examined whether the duper mutation alters parametric as well as nonparametric properties, intensity thresholds, and noncircadian responses to light. Furthermore, it assessed the effects of duper on the range of entrainment and circadian aftereffects. In the first study, duper mutant and wild-type (wt) hamsters showed a similar intensity threshold for light-induced phase shifts. In the second, wt, tau mutant, and super duper hamsters were exposed to LD cycles whose period (T) progressively shortened. Regardless of whether the light phase was held at 50% of T or fixed at 3 h, super duper mutants entrained to a wider range of T cycles and showed aftereffects upon release into DD. In the third study, τLL was measured in mutant and wt hamsters that were maintained for 30-day intervals in constant light of progressively greater intensities. With increasing light intensity, the circadian period shortened in duper mutants. Circadian rhythms of super duper hamsters were disrupted at light intensities considerably below those that induced arrhythmicity in wt, tau heterozygote, or duper homozygote hamsters. In the fourth study, hamsters that were wt or homozygous for duper received two 15-min light pulses: the first at CT14 to CT16 or CT17 to CT19 and the second 2 h later. As expected, wt and duper mutants showed weak and strong resetting, respectively. Light pulses in early subjective night had an additive effect in mutant but not in wt hamsters, indicating that larger phase shifts of the pacemaker take longer to complete. Finally, super duper hamsters showed slightly but not significantly more negative masking than did wt or duper mutant hamsters. These results indicate that the duper mutation affects the properties of the central circadian pacemaker. The mutant allele affects not only the PRC but also parametric responses to light.
When expressed on a wild-type background, the duper mutation in Syrian hamsters shortens the free-running period of locomotor activity rhythms in constant darkness (τDD) by about 1 h (Monecke et al., 2011). Unlike the better characterized tau mutation, duper is recessive and is not attributable to a change in the coding region of casein kinase 1ϵ or 1δ. In super duper hamsters (homozygous for both duper and tau), τDD is approximately 18 h. Duper mutants entrain quite stably and super dupers less so when maintained in 14L:10D, but their phase angle is more positive than that of wt hamsters. Both duper and super duper hamsters show high-amplitude resetting upon exposure to 15-min light pulses during the subjective night (Krug et al., 2011). The exaggerated phase shifts of hamsters that carry the duper mutation may arise from a change in the sensitivity to light, such that the intensities of light pulses used for resetting are perceived as brighter by mutant than wt hamsters. Agostino et al. (2009) reported that tau mutant hamsters have a lower intensity threshold for phase shifts than wt hamsters, raising interest in determining whether the duper mutation might have a similar effect. On the other hand, the shortened free-running period in constant darkness, high-amplitude resetting curves, and phase angle of activity onset in T24 suggest changes in the pacemaker function of duper mutants and raise the possibility that their range of entrainment is expanded.
The nonparametric model of entrainment stipulates that dawn and dusk transitions rapidly reset the pacemaker (Pittendrigh and Daan, 1976a). This prediction has been validated in wt animals by experiments in which a second light pulse falls relatively soon after the first is presented during the early or late subjective night. When such 2-pulse experiments have been performed in wt hamsters, the results indicate that the circadian oscillator achieves the new phase within an hour of light exposure (Best et al., 1999). Such animals show low-amplitude resetting, however, and it remains to be determined whether phase shifts of 8 to 12 h, such as those induced by light in duper mutants, can be completed as rapidly.
Light also has parametric effects, and it has been argued that an effect of light to alter the speed of the clock may contribute to entrainment (Johnson, 1939; Aschoff, 1960; Comas et al., 2007; Roenneberg et al., 2010; Taylor et al., 2010). Mutations that affect entrainment may provide a tool to discriminate between the genetic basis of responses to acute versus sustained light pulses. Thus, it is of interest to determine whether duper also affects the parametric effects of light intensity on the circadian period. In particular, this study aimed to explore the effects of the duper mutation upon the relationship generalized as Aschoff’s rule in which the circadian period of nocturnal animals is directly proportional, and that of diurnal animals is inversely proportional, to light intensity.
Finally, light may have effects upon activity patterns that are not mediated by the circadian system. Specifically, light has negative or positive masking effects in which activity is reduced or increased in response to illumination even in animals lacking an intact circadian pacemaker (Bunger et al., 2000; Mrosovsky and Hattar, 2003; Li et al., 2005; Guler et al., 2008). The effects of the duper mutation may be restricted to circadian responses to light or may also extend to masking.
Materials and Methods
Syrian hamsters derived from the LVG strain (Lakeview) were bred in our laboratory. All procedures were approved by the University of Massachusetts Animal Care and Use Committee and conform to United States federal animal welfare requirements. Hamsters were raised to adulthood in 14L:10D and allowed ad libitum access to food and water throughout the experiments. The tau mutant genotype was confirmed in the parental lines by restriction digest as described by Monecke et al. (2011). In light of previous findings that sex has no significant effect on τDD in either wt or duper hamsters (Bittman, 2012), both males and females were used in these experiments.
Intensity Threshold for Phase Shifts
To construct an Aschoff type I PRC, hamsters were transferred to DD. Running wheel activity was recorded and analyzed using ClockLab software (Actimetrics, Evanston, IL, USA) as described previously (Krug et al., 2011). At intervals of 10 to 12 days, 28 hamsters (6 super duper, 10 duper, and 12 wt) were exposed to a 15-min pulse of 1) dim, 2) intermediate, or 3) standard intensity (0.002, 0.09, or 4 µE/m2/sec, respectively) at approximately CT15 or CT18 and returned to DD. Steady-state phase shifts (after any transients had subsided) were calculated by linear regression as described by Krug et al. (2011). The results of this experiment indicated that the threshold for duper hamsters lies between the dim and intermediate intensities. An additional experiment was conducted to better identify the intensity threshold for phase shifts in the 2 genotypes during the early subjective and midsubjective night. Accordingly, 44 additional hamsters (16 duper and 28 wt) were presented with a 15-min light pulse of 0.007 µE/m2/sec at either CT15 or CT18 on day 10 of DD, and phase shifts were assessed over the ensuing 11 days.
Range of Entrainment
Two experiments were conducted in a room in which fluorescent lights (approximate intensity of 2 μE/m2/sec) were controlled by Eagle Flexopulse timers (Danaher Industrial Controls, Elizabethtown, NC). The first such study utilized wt (n = 10), tau mutant (n = 11), and super duper (n = 12) hamsters. T was decreased by approximately 30 min at 10- to 14-day intervals; the timers were manually adjusted to hold the duration of both the light and dark phases at approximately 50% of T. Entrainment was assessed by comparison of the period of locomotor onsets to the period of the light cycle as recorded by photocells: when the linear regression fit (±1 SD, generated by Actimetrics ClockLab software) used to assess the period of locomotor onsets over the last 7 days of exposure to the T cycle matched the period of the L:D cycle, the animal was judged to have maintained entrainment. After super duper animals broke entrainment, they were returned to T15 (7.5L:7.5D) for 3 weeks to establish and assess re-entrainment. This was followed by 11 days of DD to confirm that the free run began from the phase of locomotor onset in the light:dark cycle and to assess aftereffects of the T15 cycle. A 15-min light pulse was presented in early (approximately CT15) or late (CT20) subjective night, and phase shifts were assessed over the next 10 days.
In the second study, new groups of tau homozygote (n = 10) and super duper (n = 14) hamsters were transferred from 14L:10D to 3L:21D for 10 days. They were then exposed to a series of light:dark cycles in which the photophase was held constant at 3 h. T was reduced by shortening the duration of the dark phase by approximately 45 min at intervals of approximately 12 days until all animals had broken entrainment, except for intervals of 8 and 21 days during which hamsters were maintained in DD after exposure to T19.5 and T17.3, respectively. Animals were again released into DD following maintenance in T15.5 to assess masking and to confirm entrainment to the prior light:dark cycle.
Aschoff Rule and Splitting
To examine whether and how the duper and tau mutations, alone or in combination, influence the response of free-running hamsters to light intensities, τDD was assessed in 6 duper, 6 tau heterozygote, 7 tau homozygote, and 11 super duper hamsters. Animals were next placed in constant dim fluorescent light (0.002 μE/m2/sec) for 30 days, after which they were moved to a higher light intensity (0.09 μE/m2/sec) for an additional 30 days. Finally, these animals were moved to constant light at standard lighting intensity (4.0 μE/m2/sec) for at least 100 days.
Phase Shift Latency
An experiment was performed to determine whether the large phase shifts of the pacemaker induced by 15-min light pulses in duper mutant hamsters take longer to complete than those of wt hamsters. The phase shift responses of wt and super duper hamsters to single 15-min light pulses at about CT15 and CT18, and to 2 pulses at an interval of 2 h beginning at CT15 and CT18, were assessed on 4 occasions separated by at least 11 days in counterbalanced order. For purposes of statistical comparison, phase shifts were plotted monotonically.
Masking
Hamsters that had been in DD for at least 3 days were exposed to alternating 3.5-h intervals of light and darkness for 35.5 h. Included in this experiment were 14 wt, 15 tau homozygote, 7 duper, and 8 super duper hamsters. The number of wheel revolutions was assessed to evaluate the effect of acute light exposure on locomotor activity during subjective night.
Statistical Comparisons
Group differences were evaluated using analysis of variance (ANOVA) (JMP statistical software, SAS Institute, Cary, NC, USA), with corrections for repeated measures where appropriate. When ANOVA indicated significant main effects or interactions, post hoc comparisons were made using the Tukey test. The experiments in which the intensity threshold for light induced phase shifts suggested an all-or-none response. Accordingly, the χ2 test was used to evaluate differences between genotypes in the proportion of animals that experience phase shifts >1 h in response to light pulses of various intensities. χ2 tests were also used to assess the proportion of animals of each genotype that could entrain to shortening T cycles. Locomotor activity in constant light was analyzed using Actimetrics software as previously described (Bittman, 2012), including the use of χ2 periodograms to assess the amplitude of free-running locomotor rhythms.
Results
Intensity Threshold for Phase Shifts
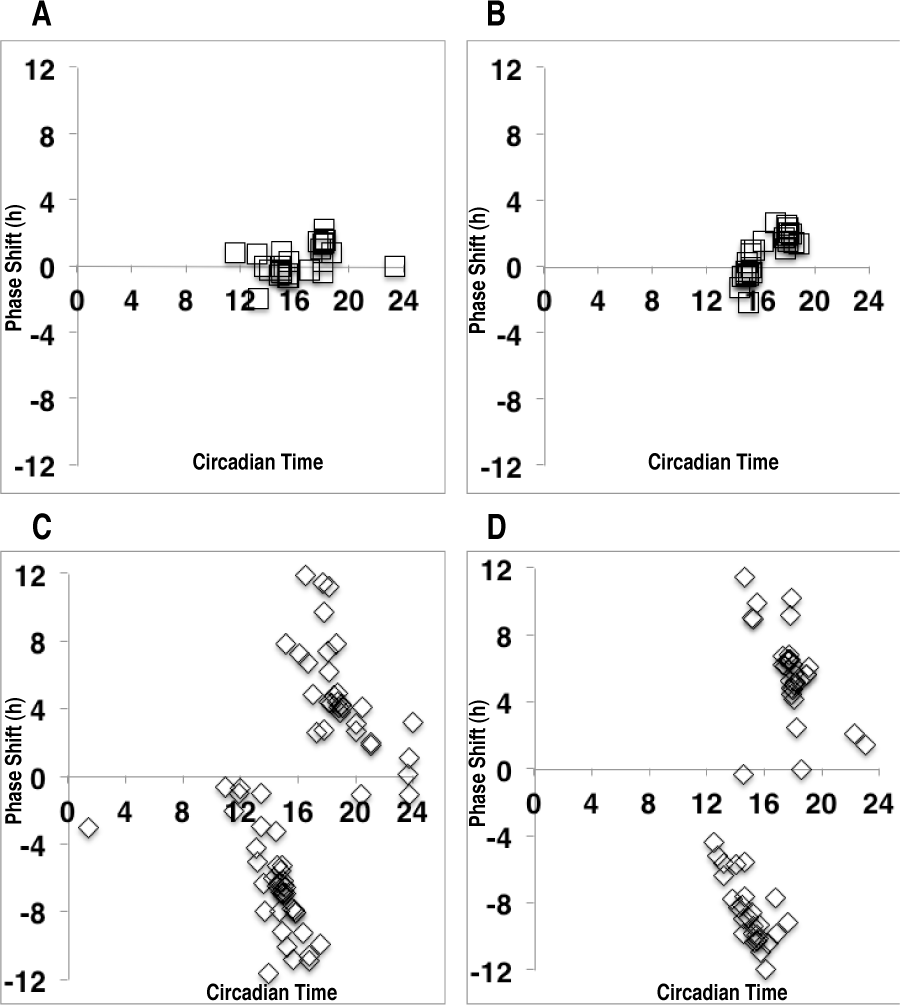
Both light pulse intensity and genotype significantly influenced the amplitude of the phase shift. The interaction between these 2 main factors was statistically significant, and these effects were apparent in both the early subjective and mid subjective night (F3,37 = 156.7 and F3,39 = 20.3, respectively; p < 0.001). Pulses of dim light (intensity of 0.002 μE/m2/sec) presented during the early subjective night (between CT14 and CT16) were below the threshold for inducing phase delays in both wt and duper hamsters (mean ± SEM phase shifts of 0.17 ± 0.19 and −0.26 ± 0.30 h, respectively; only 2/10 wt and 2/15 duper hamsters showed shifts >1 h). These dim pulses elicited small phase advances in wt hamsters but not in mutants when given later in the subjective night (CT17-CT19; mean phase shifts of 1.19 ± 0.24 v. –0.12 ± 0.09 h, respectively, with 4/11 and 1/13 duper hamsters shifting >1 h) (Fig. 1). Thus, for dim pulses presented around CT18, wt hamsters actually showed significantly greater phase advances than did duper mutants (p < 0.001).
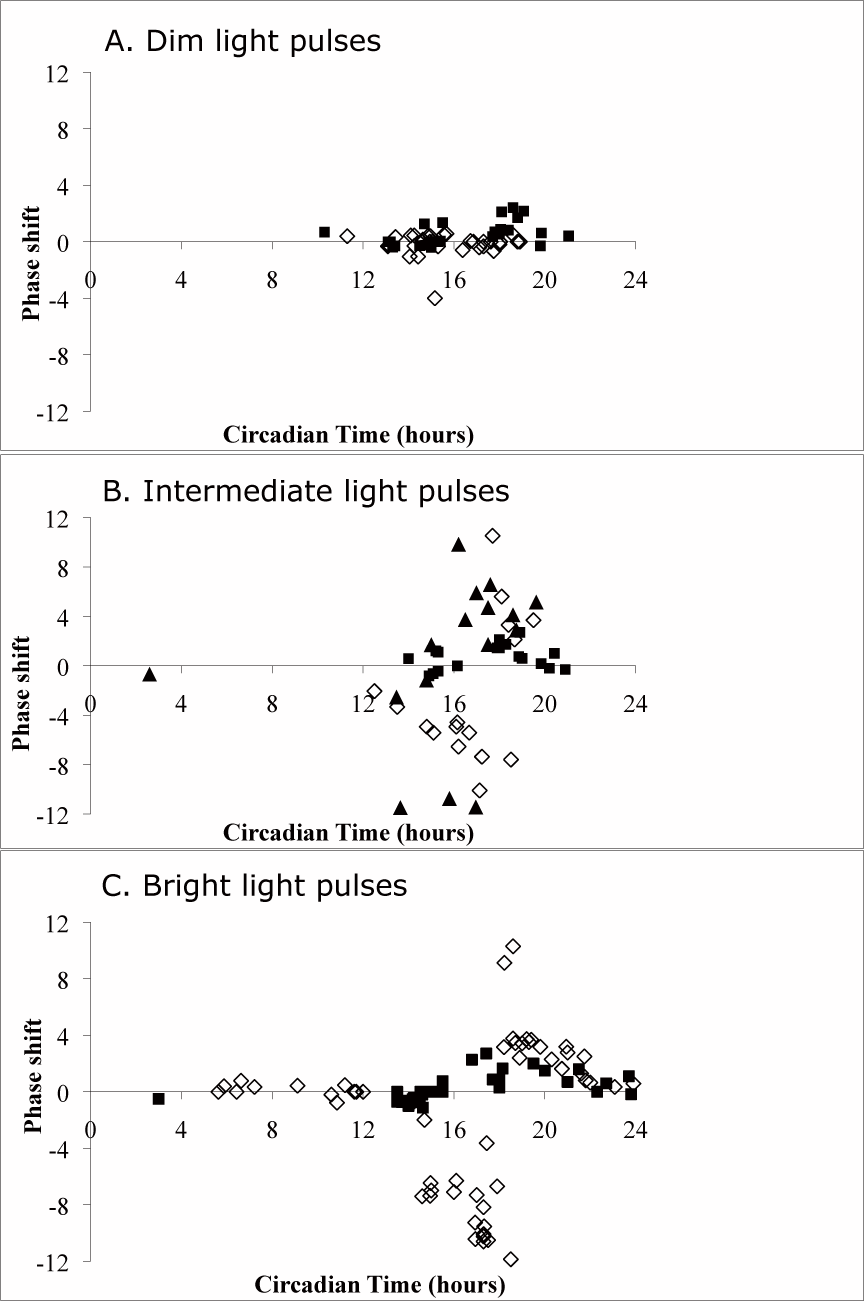
The PRCs of hamsters exposed to 15-min light pulses of (A) 0.002, (B) 0.09, or (C) 4 µE/m2/sec. Filled squares depict responses of wt animals, open diamonds indicate phase shifts of duper mutants, and filled triangles show data from super duper hamsters (intermediate light pulses only). Hamsters were allowed to free run in DD for at least 10 days before administration of the light pulse, and the steady-state phase was determined thereafter. Data were binned for CT14 to CT16 and CT17 to CT19 for statistical comparisons, which indicated significant main effects of both genotype and light intensity, as well as an interaction between these factors, at both circadian phases (p < 0.001).
Light pulses with an intermediate intensity of 0.09 μE/m2/sec were above the threshold for the induction of both delays and advances: all duper and wt hamsters showed phase shifts ≥1 h. Light pulses in both the early subjective (CT14-CT16) and mid subjective (CT17-CT19) night produced larger phase shifts in duper mutants than in wt animals (both p < 0.001). The intermediate light pulses produced phase shifts of the same magnitude as did pulses at the standard intensity of 4 μE/m2/sec.
These observations indicated that the threshold for phase shifts of duper hamsters may lie between the “dim” and “intermediate” intensities. Accordingly, an additional experiment was run using pulses of 0.007 μE/m2/sec, which lies between these intensities. When given at about CT18, the effectiveness of this intensity was close to half-maximal in both genotypes: 6 of 13 wt and 5 of 9 duper mutant hamsters showed advancing phase shifts ≥1 h (Suppl. Fig. S1). Interestingly, phase shifts appeared to be all or none rather than a graded function: duper hamsters either showed very little shift or a shift >5 h. As with the intermediate and brighter intensities, wt hamsters rarely showed a substantial phase shift when a pulse of 0.007 μE/m2/sec was given at about CT15 (1/15 shifted >1 h), but about half (3/7) of the duper hamsters shifted >1h in response to light pulses at CT15.
Range of Entrainment
Entrainment was first examined in wt, tau mutant, and super duper hamsters over a range of T cycles in which the lengths of the light and dark phases were held equal. In each genotype, the phase angle decreased progressively as T was reduced. The range of entrainment, however, differed markedly between groups. Actograms of representative hamsters are shown in Figure 2, and phase angles over the range of T cycles to which animals entrained appear in Figure 3. Moreover, wt hamsters broke entrainment when the photoperiod decreased <23 h (Fig. 2A). Tau homozygote (super short, τss) hamsters were uniformly able to entrain to T19.6, but 4 of 8 broke entrainment when T was reduced to 17.7 (Fig. 2B). Two of the remaining tau homozygotes could entrain to T17.1 but not below (Fig. 3). In contrast, all 7 super duper hamsters in this study were able to entrain to T cycles as short as 15.05 h, and 3 animals entrained to T14.1 (Figs. 2C and 3, left). The proportion of super duper mutant versus tau mutant hamsters that entrained to T cycles of ≤22 h differed significantly (p < 0.001, χ2 test). Immediately after the 2-week period of exposure of super duper hamsters to T14.1, the photoperiod was increased to 7.5L:7.5D to confirm that these mutants could entrain to T15. Each individual established a stable phase angle to this light:dark cycle. After 2 weeks on this regimen, 16 super duper mutant hamsters were returned to DD to obtain a second estimate of τDD. Over the next 10 days, the free-running period of these hamsters averaged 16.64 ± 0.05 h. Every individual exhibited a shorter τDD during the second free run than it had when initially placed in DD. This 6.5% reduction of τDD represents a significant aftereffect when compared to its value of 17.80 ± 0.09 h after release into DD from T24 (τdependent = 10.55, df = 16, p < 0.0001). Light pulses administered to 12 super duper mutants that had been entrained to T15 for 2 weeks and released into DD for 11 days prior produced maximal shifts of 9 h. These data suggest that the PRC was not further amplified in super duper hamsters showing the circadian aftereffect.
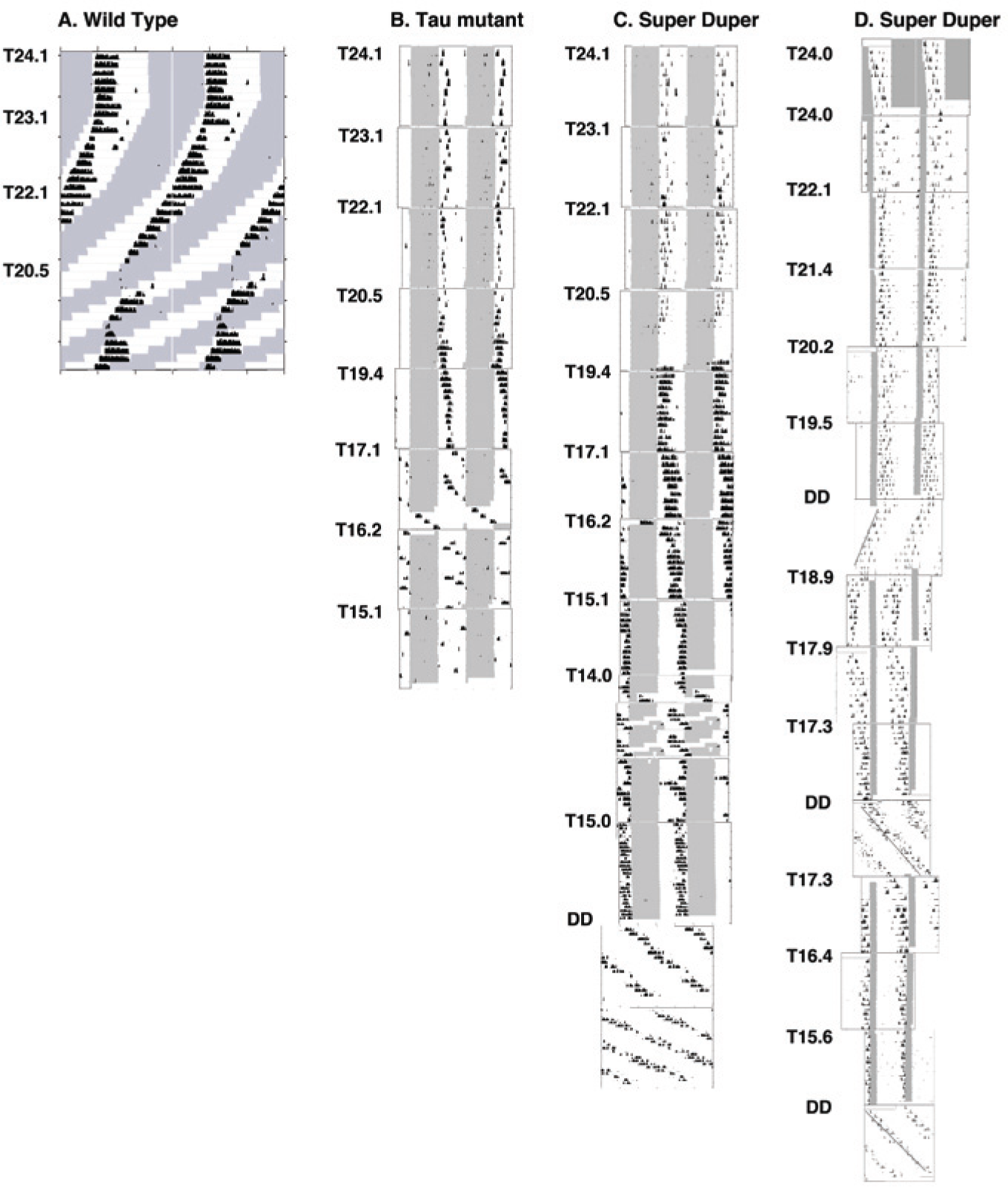
Double-plotted actogram records of locomotor activity of 4 representative hamsters during exposure to T cycles. Shading indicates the light phase. Timers were adjusted every 10 days to 2 weeks to achieve progressive shortening of the period of the light cycle; T values are indicated to the left of the actograms of mutant hamsters. (A) Record of a wt hamster held in T cycles in which light and dark phases were of equal duration. The hamster broke entrainment when the period of the LD cycle was reduced to T22.1. (B) A tau homozygote hamster exposed to progressive shortening of T in a regimen in which the durations of the light and dark phases were held equal. The hamster entrained to T19.4 but not to T17.1. (C) Record of a super duper hamster exposed to the same series of T cycles. This hamster entrained to T15.1 but showed relative coordination in T14. Note the change in the phase angle as T’s duration transitions from greater than to less than τDD (T19.4-T15.1). This hamster re-entrained when returned to T15.0; note that the final free run in DD begins from the entrained phase. (D) Record of a super duper hamster exposed to a series of T cycles in which the duration of the light phase was held constant at 3 h. The linear regression line fit to activity onsets is shown for intervals of constant darkness after T19.5, T17.3, and T15.6 to allow assessment of the correspondence of the phase of the free run to the phase of locomotor onset in the prior T cycle and thus confirm entrainment. Note that actograms are plotted modulo 24.1 h (A) and modulo T (B-D).
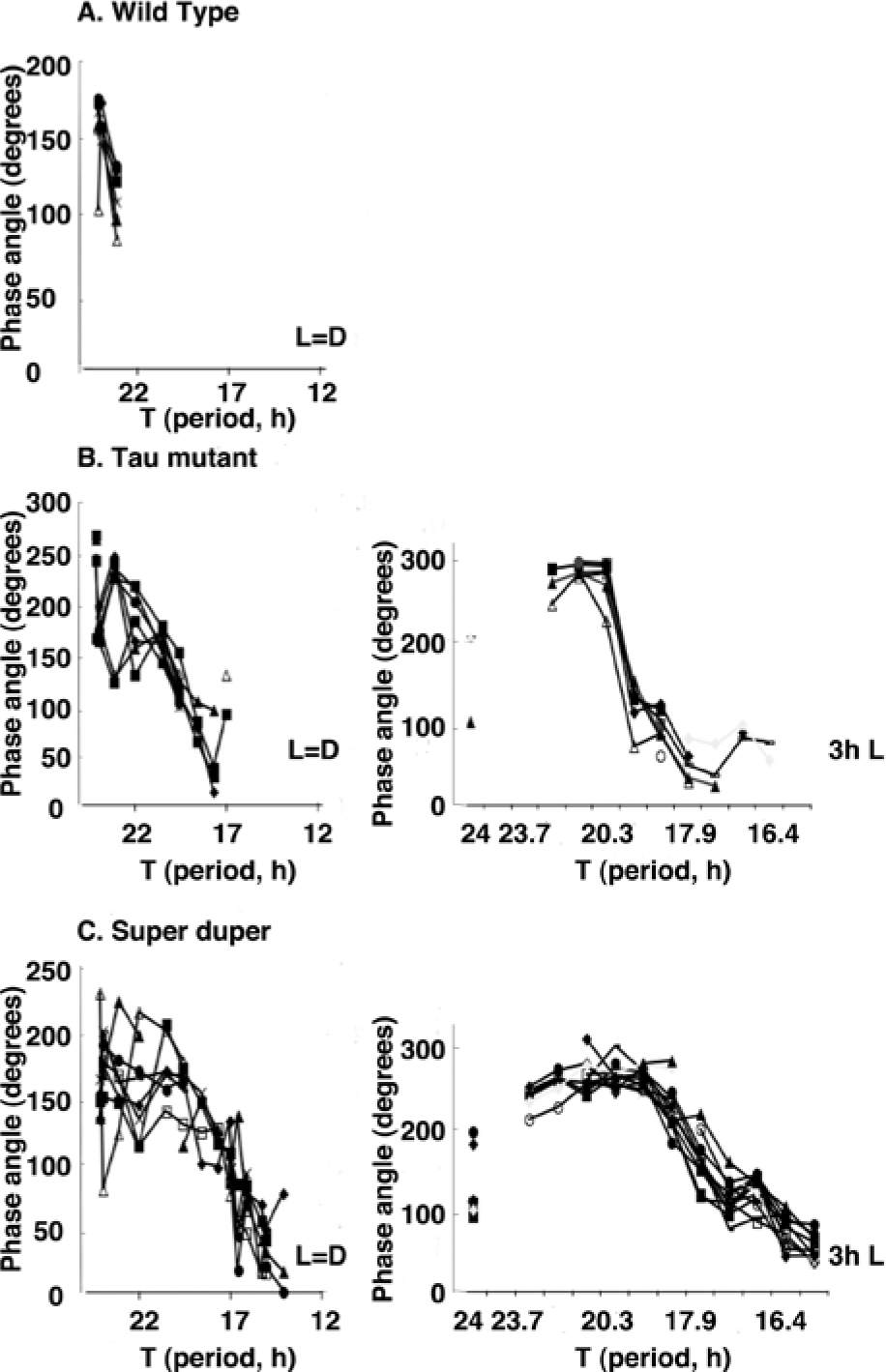
Entrained phase angle of (A) wt, (B) tau mutant, and (C) super duper hamsters that were exposed to progressively shortening T cycles in which the duration of the light interval was held approximately equal (L = D, left) or was held constant (3-h L, right). The phase angle of each individual hamster is plotted and linked by a line as it was exposed to successively shorter T cycles; when a hamster did not entrain, no data point is shown. The range of entrainment was wider in tau homozygote than in wt hamsters and greater in super duper than in tau mutant hamsters. Note that super duper hamsters entrained to the widest range of T cycles. For all 3 genotypes, the phase angle (interval between activity onset and light onset) decreased systematically over the range of entrainment as expected (Aschoff and Pohl, 1978).
In a second study of the range of entrainment in tau homozygote and super duper hamsters, the photoperiod was held constant at 3 h as T was reduced. This ensured that entraining effects were attributable to a coincidence of light with only the early or the late subjective night. As in the first experiment, stable entrainment to short T cycles was evident in both strains, and the phase angle of entrainment decreased monotonically as T was shortened (Fig. 2D). Nevertheless, the super duper animals exhibited a wider range of entrainment than the tau homozygotes: a majority of the double mutants entrained to all T cycles between 15.56 and 22.11 h (Fig. 3, right). In contrast, less than half of the tau homozygotes entrained to T cycles longer than 21.39 h or shorter than 18.85 h. For example, 14 of 14 super duper and 9 of 10 tau mutant hamsters held steady phase angles in T19.54. In contrast, 12 of 13 super duper but only 3 of 10 tau mutant hamsters entrained to T17.31, and 11 of 12 super duper but 0 of 10 tau mutant hamsters entrained to T15.56. The difference in the proportion of tau homozygote versus super duper hamsters entraining to these short T cycles was statistically significant (p < 0.001, χ2 test). Assessment of the phase of free-running rhythms after release into DD from each of these T cycles confirmed that the control of activity phase by the 3-h photophase reflected entrainment rather than masking. The final free-running period after release into DD from T15.56 was significantly shorter than that before T cycles commenced (τDD = 17.20 ± 0.09 v. 17.91 ± 0.11 h in super duper hamsters and 19.61 ± 0.16 v. 20.41 ± 0.13 h for tau mutant hamsters, respectively; both p < 0.01).
Aschoff’s Rule and Splitting
Wild type tau heterozygote and homozygote, duper, and super duper hamsters were allowed to free run at each light intensity for 30 days. Overall, ANOVA showed significant main effects of genotype and light intensity on period as well as an interaction between these factors (all p < 0.001). Changes in the free-running period generally stabilized in the second or third 10-day interval of maintenance in each light intensity, and the τ values between days 20 to 30 were used to assess the steady-state period. Among wt hamsters, the effect of light intensity on the free-running period did not reach significance when all intensities were included (p = 0.059, dependent t test). This was due to variable responses to transfer from DD to dim light: t increased upon transfer from DD to dim light in only 1 wt hamster, while in 2 others, it decreased, and in 2, it did not change. In contrast, t lengthened significantly when wt hamsters were transferred between dim and brighter light intensities (p = 0.04). Free-running period did not change significantly as tau heterozygotes were exposed to progressively brighter intensities of constant light. In contrast, t shortened as tau homozygotes and as duper and super duper mutants were transferred to brighter intensities of constant light (Figs. 4B-E and 5A). In each genotype, the duration of activity (α) decreased as light intensity was raised (Fig. 5B). The amplitude of the χ2 periodogram also decreased as light intensity increased, and genotypes differed in the intensity of constant light at which rhythmicity deteriorated (Fig. 5C): super duper and tau homozygote hamsters became arrhythmic at lower LL intensities than did wt or duper hamsters.
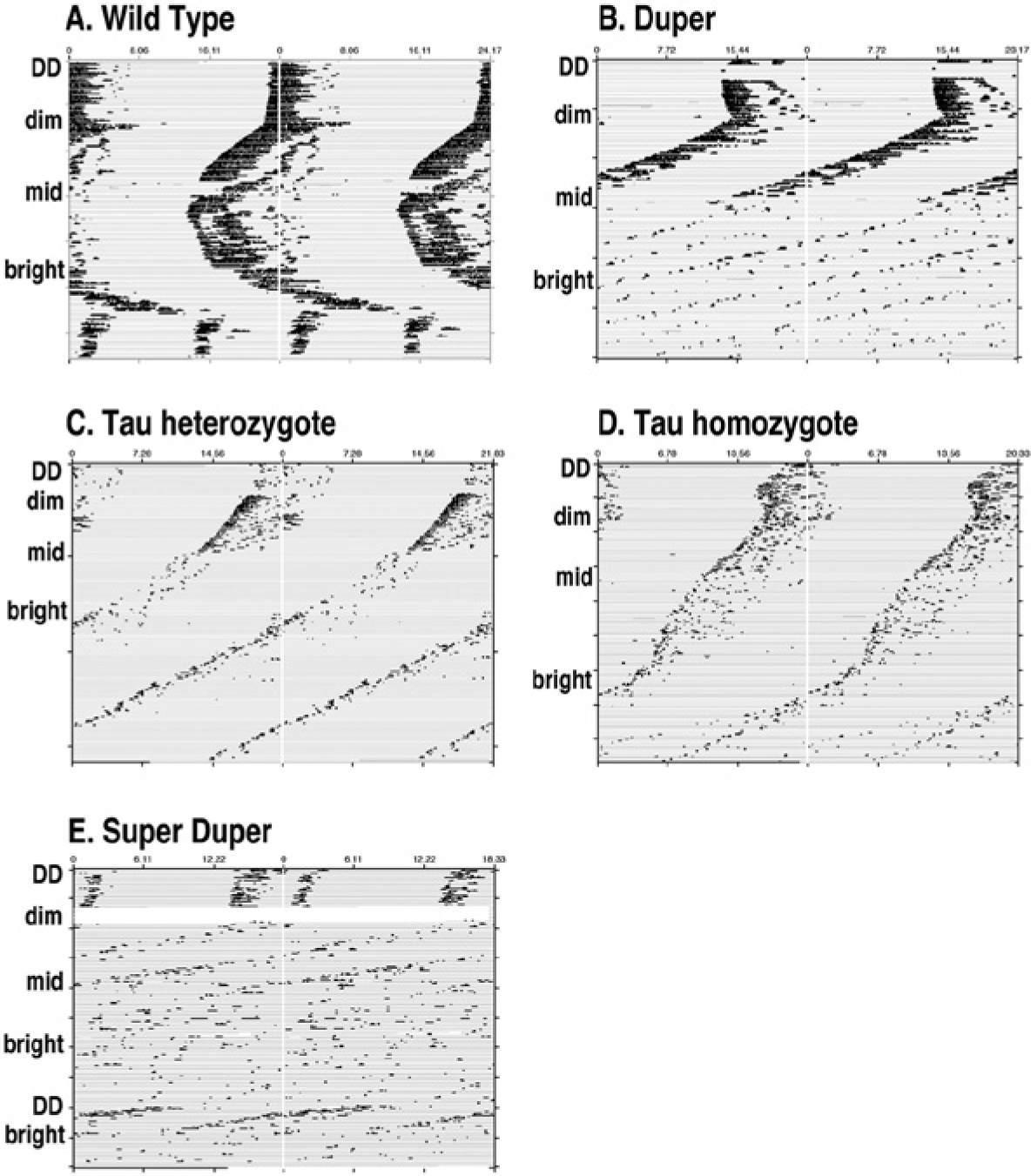
Aschoff’s rule and splitting in 5 different hamster genotypes. Double-plotted actograms for each animal are, plotted modulo τDD, as determined during the first 2 weeks of the experiment. Representative records of (A) wt (modulo 24.17 h), (B) duper (modulo 23.17 h), (C) tau heterozygote (modulo 21.83 h), (D) tau homozygote (modulo 20.33 h), and (E) super duper (modulo 18.33 h) hamsters exposed to various intensities of constant light are shown. Animals were shifted from DD to LL of dim (0.002), intermediate (0.09), and bright (4 μE/m2/sec) intensities at intervals of 1 month. Super duper hamsters became arrhythmic in mid-LL and bright LL. Upon return to DD, short period circadian rhythms returned immediately, only to stop soon after the return to bright LL. Note that circadian rhythms split in wt animals exposed to bright LL but not in hamsters carrying the tau mutation.
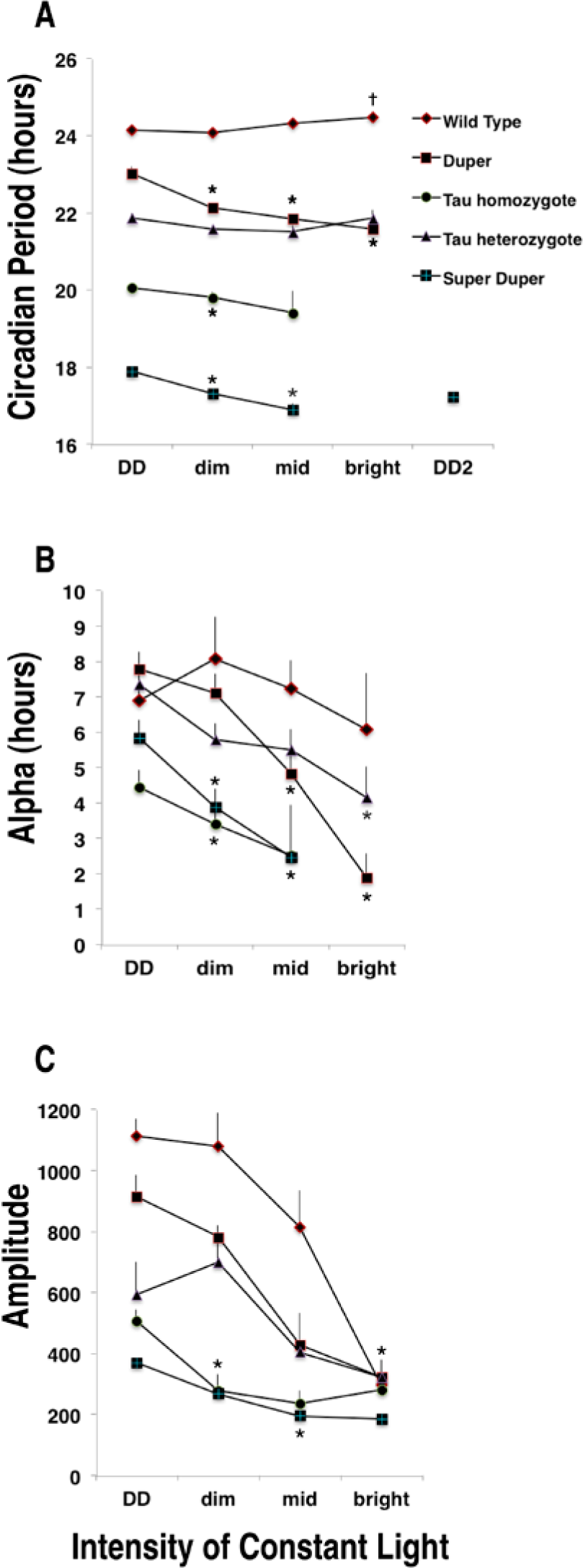
Response of wt (diamonds), duper (squares), tau heterozygote (triangles), tau homozygote (circles), and super duper [+] hamsters exposed to constant light of dim (0.002), intermediate (0.09), or bright (4 µE/m2/sec) intensities for successive 30-day intervals. Values (mean ± SEM) of the final 10 days of maintenance in each intensity are plotted. (A) Free-running period, (B) active period (a), and (C) amplitude of the χ2 periodogram. Most tau homozygote and super duper hamsters became arrhythmic at the brightest intensity. Super duper hamsters rapidly regained rhythmicity when returned to constant darkness (DD2). *Significant difference from the DD condition. †Significant difference from dim intensity (τdependent, p < 0.05).
The tau and duper alleles had different effects upon the incidence of splitting and arrhythmicity in LL (Fig. 4). In both the wt and duper homozygote groups, 4 of 6 animals split in either the middle light intensity or within 10 days of transfer from the intermediate to the brighter intensity of LL. χ2 analysis indicated that the period of the split component was approximately half the circadian period (12.06 ± 0.06 h in wt and 10.89 ± 0.07 h in duper hamsters). In contrast, none of the tau heterozygotes or the super duper hamsters, and only 1 of the tau homozygotes, split in LL. All of the wt hamsters remained rhythmic throughout the 90-day exposure to constant light (showing significant peaks in the χ2 analysis with amplitudes >200 and χ2 >170 throughout their 30 days of exposure to each intensity). With 2 exceptions, the duper hamsters also remained rhythmic even throughout the exposure to the brightest intensity. In contrast, most tau homozygote and all super duper hamsters became arrhythmic at intermediate light intensities. Locomotor rhythmicity resumed immediately upon the return of super duper hamsters to DD with a period of about 17 h (Fig. 4E). Upon return to the bright LL, however, arrhythmic locomotor patterns returned without apparent transients.
Phase Shift Latency
The nonparametric model stipulates that light pulses rapidly reset the circadian clock and that transient cycles reflect events that occur distal to the pacemaker. This prediction is validated in wt hamsters by 2-pulse experiments (Best et al., 1999), but such rapid resetting may only be possible for an oscillator with a low-amplitude PRC. The aim was to test the hypothesis that the large phase shifts of dupers take longer to complete than the 2- to 3-h shifts of wt hamsters. This experiment required that the light pulses be presented at the phase of the maximal phase delay or advance of mutant hamsters and at the same CT to control animals. Thus, wt and duper (on a tau heterozygote background) hamsters were subjected both to single 15-min light pulses at about CT15 and CT18 and to 2 pulses at an interval of 2 h beginning at CT15 and CT18, on 4 occasions separated by at least 11 days, in counterbalanced order. The reasoning was that if the phase shift induced by the first pulse had not been completed within 2 h, the effects of the 2 pulses would be additive. In contrast, if the shift induced by the first light pulse was completed within 2 h in duper mutants, the second pulse would strike the dead zone of the PRC.
In wt hamsters, the phase advance elicited by double pulses given at a 2-h interval exceeded the response to a single light pulse if given during the midsubjective to late subjective night (F = 12.53, df = 9, p < 0.01) but not during the early subjective night (F = 7.86, df = 6, p > 0.1). This may reflect the fact that an advance triggered by the first pulse at approximately CT18, even if immediate, would reset the oscillator by only 2 h, such that the second pulse 2 h later would occur in late subjective night and thus strike the advance portion of the PRC. In contrast, the phase-shifting effect of 2 light pulses administered during the midsubjective to late subjective night to hamsters carrying the duper mutation did not significantly differ from that of a single pulse (F = 332.0, df = 14, p = 0.42) (Fig. 6A and 7A). This is consistent with the prediction that the first pulse had elicited a large phase shift in mutants than in the wt hamster, throwing the oscillator to the subjective day (dead zone) within 2 h. However, double pulses given to mutant hamsters during the early subjective night elicited slightly but significantly greater phase delays than did single pulses (F = 98.8, df = 15, p = 0.004) (Fig. 6B and 7B). This outcome is consistent with the hypothesis that the large phase shifts had not been completed within 2 h.
Responses of wt (squares) and duper (tau heterozygote background) (diamonds) hamsters to (A, C) single 15-min light pulses or to (B, D) two 15-min light pulses given at an interval of 2 h.
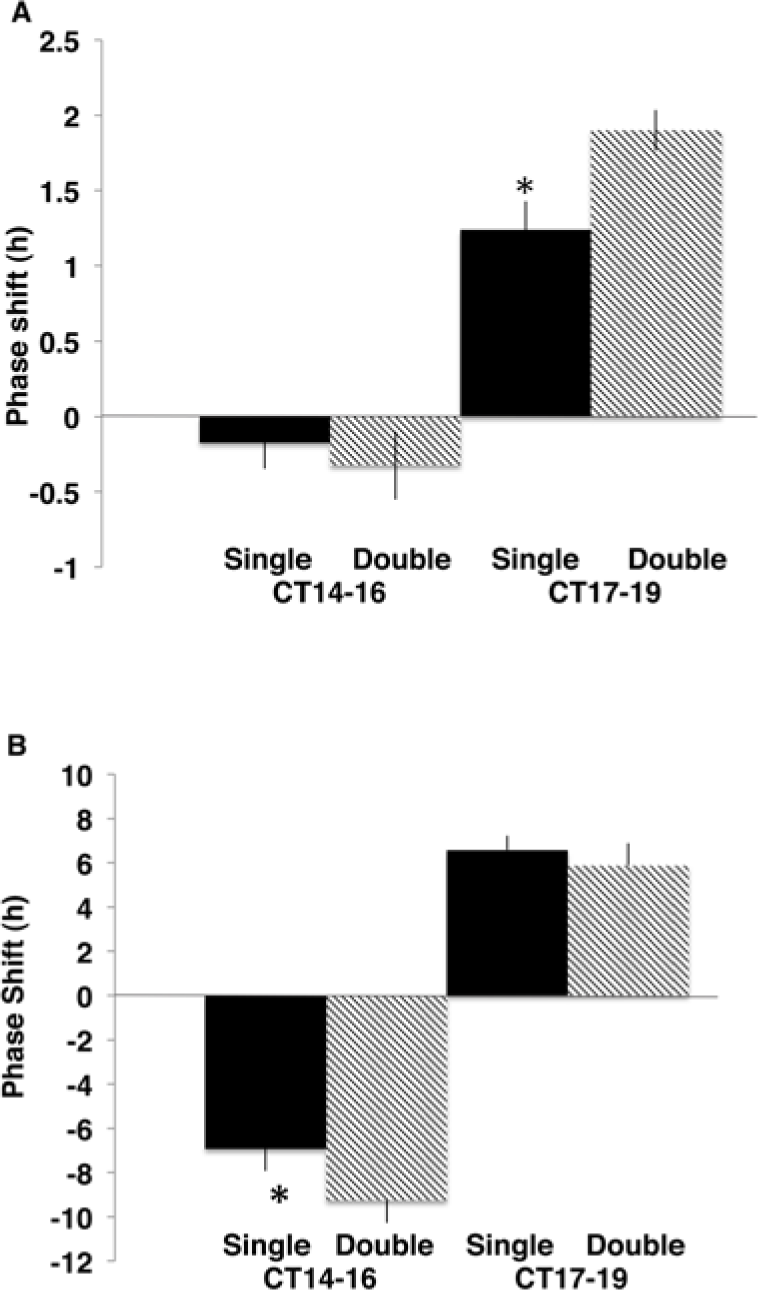
Mean ± SEM phase shifts of (A) wt or (B) duper (tau heterozygote background) (bottom) hamsters to single (black bars) or double (hatched) light pulses given at CT14 to CT16 or CT17 to CT19. *Significant difference in the amplitude of the shift compared with hamsters of the same genotype receiving double pulses beginning at the same phase (p < 0.01).
Masking
Exposure to light during the subjective night elicited comparable suppression of wheel revolutions in wt and duper mutant hamsters (71.2% ± 9.3% and 63.1% ± 18.3%, respectively). Super duper hamsters showed a slightly more substantial masking effect (84.6% ± 2.6%), but this difference was not statistically significant (p > 0.10).
Discussion
The present results extend the characterization of the effects of 2 mutations that shorten period—tau and duper—on the hamster circadian system and its response to light. The effects of these mutations upon parametric responses show some similarity in that both mutants shorten period as light intensity increases. Nevertheless, hamsters carrying the tau mutation, alone or in combination with duper, are more susceptible to arrhythmicity in constant illumination. In addition, hamsters carrying either 1 or 2 copies of tau are resistant to splitting in LL. The tau and duper mutations also differ in their effects on nonparametric responses to light, as evidenced by the amplified PRC of the duper hamsters, and the effect of this mutation seems not to result from a decrease in threshold light intensity. Furthermore, the duper mutation substantially increases the range of entrainment to an extent much greater than the tau mutation. Finally, at least some of the larger phase shifts of the duper mutant may take longer to complete than those of wt hamsters.
Intensity Threshold for Phase Shifts
The amplitude of phase shifts of wt hamsters is linearly related to irradiance (Nelson and Takahashi, 1991). Differences in protocols make it difficult to compare studies in detail, but the intensity threshold of tau mutants is below that of the wt animal (Agostino et al., 2009) and decreases during prolonged maintenance in DD (Shimomura et al., 1998). Results of the present study do not indicate that duper confers a comparable sensitization to phase-shifting effects of light. Under the conditions used here, the threshold appears to lie close to 0.002 μE/m2/sec for wt hamsters as more than one third of these animals exhibited phase advances >1 h when such pulses were administered around CT18. If anything, the duper mutants have a higher intensity threshold for light-induced phase shifts than do wt hamsters as only 1 of 13 animals experienced a phase advance in response to a pulse of 0.002 μE/m2/sec. A light pulse of 0.007 μE/m2/sec induced a phase advance >1 h in about half the wt and duper hamsters, indicating that the mutation has little effect on the intensity threshold. The slope of the function of shift magnitude versus intensity may be affected by the mutation: rather than appearing to be a graded response, duper mutants appeared to show either no shift or a full-fledged (>5 h) phase shift in response to a pulse of 0.007 μE/m2/sec at CT18. The results suggest that the duper mutation affects entrainment at a central rather than a retinal level and further distinguish the duper from the tau mutation. The effect of tau to lower the intensity threshold for phase resetting may reflect its basis of a gain of function of casein kinase 1ϵ, which may participate in glutamatergic transmission (Li et al., 2011) as well as core clock function and thus regulate light input to the pacemaker at multiple levels. Changes not only in the retina but also in the SCN can affect phase-shifting responses (Butler et al., 2011) and PRC amplitude (Gu et al., 2012). The response of duper animals may saturate at lower light intensities than that of the wt animal. While it is not likely that the duper mutation amplifies resetting by altering the sensitivity of photoreceptors, the relationship between light pulse intensity and molecular events in the pacemaker that determine the final amplitude of the phase shift remain to be explored.
Range of Entrainment
Does the duper mutation increase the range of circadian entrainment? The present estimate of the lower range of entrainment in wt animals is similar to those of Pittendrigh and Daan (1976b) and Carmichael et al. (1981), although light intensity, photophase duration, and other aspects of the procedure may affect this value (Boulos et al., 2002; Chiesa et al., 2005 and 2007; Gorman et al., 2005). A strength of the nonparametric model of entrainment is that the amplitude of the PRC to brief light pulses predicts the range of entrainment of the circadian clock in full photoperiods. In agreement with this prediction, super duper hamsters were able to entrain to a wider range of T cycles than were hamsters carrying the tau mutation alone. It is noteworthy that even though τDD was shorter in the double mutant than the tau homozygote, these super duper hamsters were able to entrain to T cycles that were both longer and shorter than those to which τss hamsters could entrain. In both strains, the progressive shortening of the entraining cycle produced a monotonic change in the phase angle of the onset of locomotor activity, and “probe” intervals in which hamsters were released into DD confirmed that their free runs commenced from the entrained phase. In addition, the plasticity of the circadian clock (as assessed by the magnitude of aftereffects) was similar when each of these hamster genotypes was entrained to T cycles near their lower range of entrainment.
It is of interest to compare these findings on mutant hamsters with earlier reports of effects of deletion of core clock components and modifying enzymes on parametric and nonparametric effects of light in mice. Studies of phase responses to light and entrainment of mice deficient in Per1, Per2, Cry1, or Cry2 show some alterations, but all PRCs are type I and of similar waveform to the wt animal (Spoelstra et al., 2008; Pendergast et al., 2010). Consistent with the amplification of the delay portion of the PRC in Per1–/– mice, Pendergast et al. (2010) observed an expansion of the lower limit of the range of entrainment. In this respect, Per-deficient mice are similar to duper mutants. Mutation or deletion of Clock produces an effect on PRCs that is somewhat similar to duper, although light pulses as long as 6 h must be used to explore resetting responses (Vitaterna et al., 2006). The effects of the dominant-negative ClockD19 mutation to induce type 0 resetting indicate a decrease in oscillator amplitude, and Clock–/– mice are similar in this regard (Dallmann et al. 2011). Examination of light-induced core clock gene expression in super duper hamsters likewise suggests that duper reduces the amplitude of the circadian pacemaker (Krug et al., 2011). Unlike duper, however, Clock mutants show a lengthening of τDD. After-hours mice, which show a lengthening of period, also show a reduction of oscillator amplitude and type 0 resetting (Guilding et al., 2013). While none of the existing mouse mutants or knockouts replicates the phenotype of duper hamsters, Rev-Erbα–/–/Per1Brdm1 mice show shortened circadian periods and amplified phase-shifting responses to light (Jud et al., 2010). This suggests that the duper allele may directly or indirectly regulate the expression of multiple core clock genes. Identification of the genetic basis of duper may provide a more efficient means of discovering important regulatory domains than construction of mouse knockouts or mutants that contain multiple targeted substitutions or deletions.
Aschoff’s Rule and Splitting
The observation, originally reported in 1960 and enshrined as Aschoff’s rule (Aschoff, 1960, 1979), that τLL increases in wt hamsters as the intensity of constant light increases, was generally confirmed. This effect was not consistent, however, in wt hamsters transferred from DD to the lowest intensities of constant light (Fig. 4A). We previously observed that tau homozygotes violate Aschoff’s rule: their free-running period is shorter in LL than in DD (Bittman et al., 2007). In the present experiment, tau mutants were subjected to a graded progression of intensities. Tau homozygotes had a shorter period when maintained in the middle intensity than when in DD and generally lost rhythmicity in brighter LL. Tau heterozygotes showed no change in period with increasing light intensity. Although the free-running period of duper mutants was similar to that of tau heterozygotes, their period decreased significantly as the intensity brightened. This dissociates the parametric response to light intensity from the free-running period in DD and represents another difference between the phenotypic effects of the tau and duper mutations. Aschoff (1960) found that a and t showed opposite changes as LL intensity rises not only in mice and hamsters but also in finches. Not only was this relationship absent in wt hamsters in the present study, but both a and t also decreased with light intensity in duper mutants.
Effects of mutations on parametric effects of light have been examined in mouse mutants. The free-running period of Per1–/– animals lengthened in LL compared with DD (Pendergast et al., 2010). Per2–/– mice, in which the delay/advance ratio of the PRC was closer to 1.0, did not show a change in period when moved from DD to LL. Steinlechner et al. (2010) also found that Per1–/– and wt mice lengthen period as LL intensity brightens but that Per2-deficient mice shorten period under these circumstances. The findings in the present study of duper hamsters are consistent with the observation that mutations affecting the shape of the PRC may predict parametric responses. However, the increased delay/advance ratio of the duper PRC led us to expect that constant light would lengthen period, most likely through an influence on velocity responses upon exposure to constant light (Taylor et al., 2010). Using the proposed interpretation of the circadian integrated response characteristic (Roenneberg et al., 2010), dupers may show a more pronounced acceleration of the oscillator in the early subjective night than deceleration in the late subjective night. The present data indicate that mutations that shorten period can have widely different effects on circadian organization and entrainment.
Upon release into DD from entraining cycles as short as T15, super duper hamsters showed an aftereffect that resulted in a mean period of well under 17 h. Super duper hamsters can also show periods <17 h during maintenance in LL, but free runs in such conditions are less stable and persistent than those that occur in DD as an aftereffect of short T cycles. Nevertheless, a stable free-running period of about 17 h emerged upon return from bright LL to DD (Fig. 4E). This suggests that the stability of circadian rhythmicity can be dissociated from the free-running period and depends upon the environmental circumstances. Proportionally, the size of the aftereffects in duper hamsters is comparable to that shown by wt and tau homozygote hamsters in other studies (Bittman et al., 2007). Although the basis of such aftereffects remains difficult to explain at the level of the SCN pacemaker (Aton et al., 2004; Molyneaux et al., 2008), it seems most likely that they are epigenetic responses (Azzi et al., 2014). Whatever generates such aftereffects, the data suggest that they are unaffected by either mutation.
The relationship of the effects of constant light to alter period, induce splitting, and provoke arrhythmicity is poorly understood. It was found that as light intensity increased, α and the statistical power (as indicated by the amplitude of the χ2 analysis) decreased in all strains of hamsters. The tau mutation markedly decreased χ2 amplitude even in DD and did so to a much greater degree than duper. In the super duper mutants, the reduction in χ2 amplitude may reflect an additive effect of the 2 mutations. It could be that reductions in oscillator amplitude and coupling of cellular populations that occur in bright light serve as the common basis for both effects. Alternatively, the induction of arrhythmicity and the onset of splitting may represent 2 separate processes. The finding that tau heterozygotes fail to split and often become arrhythmic in the light intensities used here replicates earlier observations (Bittman et al., 2007) and supports the hypothesis that these effects of constant light reflect 2 separate processes. While it is unknown what accounts for the latency to the onset of splitting, typically about 7 weeks in wt hamsters (Pittendrigh and Daan, 1976c; De la Iglesia et al., 2000; Bittman et al., 2007), the tau mutation may accelerate the onset of arrhythmicity so that it occurs before splitting can occur.
Latency of Phase Shifts
The nonparametric model of entrainment specifies that light acts rapidly to shift the pacemaker. Two-pulse experiments validate this not only in nocturnal rodents with low-amplitude PRCs (Best et al., 1999; Comas et al., 2007) but also in Drosophila melanogaster, which has a type 0 PRC (Chandrashekaran, 1967). The present study examined whether the large phase shifts of hamsters carrying the duper mutation might take longer to complete than those of wt animals. The design of such studies requires careful selection of the timing of the first pulse. Best et al. (1999) examined the effects of paired pulses on 3 groups of hamsters, beginning at the maximal delay (CT14) and during the plateau region at which advances are greatest (CT18 and CT20). They found CT15 to be close to the crossover point of the hamster PRC (i.e., eliciting no significant shifts). In consideration of the waveform of the PRC of the duper mutants (Krug et al., 2011) (Fig. 1), the first light pulse was administered at CT15 or CT18 in the present study so that the first pulse would elicit maximal phase delays and advances, respectively.
The response of the wt animals to 2 pulses starting at CT15 was consistent with the findings of Best et al. (1999): there was only a small shift to either single or double pulses as the PRC was of low amplitude during this portion of the subjective night. Double pulses elicited larger mean phase delays, but the difference from single pulses was not statistically significant. In mutants, however, the second pulse given 2 h after CT15 added to the phase-delaying effect of the first. This indicates that the first pulse did not delay the pacemaker a full 7 h within 2 h: had it done so, the second pulse would have struck the dead zone in the late subjective day. Thus, the large phase delays of the duper hamsters take longer to complete than the shorter delays of wt hamsters. Although the result shows that the phase delay is not completed within 2 h, it may well be initiated within that time frame.
The response of the wt animal to paired pulses beginning at CT18 was again in good agreement with previous findings (Best et al., 1999; Comas et al., 2007). The outcome was consistent with the prediction that an advance triggered by the first pulse around CT18, even if immediate, would reset the oscillator by only 2 h, such that the second pulse would occur in late subjective night and thus strike the advance portion of the PRC (Best et al., 1999; Comas et al., 2007). This contrasts with the present results obtained in the duper mutants in which a second pulse given 2 h after CT18 had no additive effect. This indicates that an advance of at least 6 h is completed within 2 h in the mutant, so that the second pulse fell in the dead zone of the PRC (Krug et al., 2011). It is possible that the full shift, which is as large as 12 h, takes more than 2 h to complete. Thus, the possibility cannot be ruled out that the large phase advances initiated at CT18, like the large delays started at CT15, take longer to complete than the smaller shifts of the wt hamster. Further experiments in which the interval between light pulses is varied would be required to determine how long it takes to initiate or complete phase shifts and how the speed of such shifts is affected by mutations that amplify the PRC.
In summary, the duper mutation alters not only the free-running period but also phase-resetting responses to acute light pulses. The amplification of the PRC is not attributable to changes in light intensity thresholds, and the large phase delays of the circadian pacemaker elicited by light pulses during the early subjective night take longer to complete than the more modest shifts of wt hamsters. In addition, duper influences the effects of constant light upon the circadian pacemaker. Although duper resembles tau in its influence on the circadian period, its effects on nonparametric and parametric responses (including splitting) differ considerably. The duper mutation promises to be a useful tool in the analysis of genetic and molecular determinants of circadian organization.
Footnotes
Acknowledgements
This study was supported by National Institute of Mental Health grant RO1-MH070019 and by generous funding from the Department of Biology and the College of Natural Sciences of the University of Massachusetts at Amherst. Portions of this work were presented at the annual meetings of the Society for Research on Biological Rhythms in 2010 and 2012. The author thanks Emily N. Manoogian for thoughtful comments on the article.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
