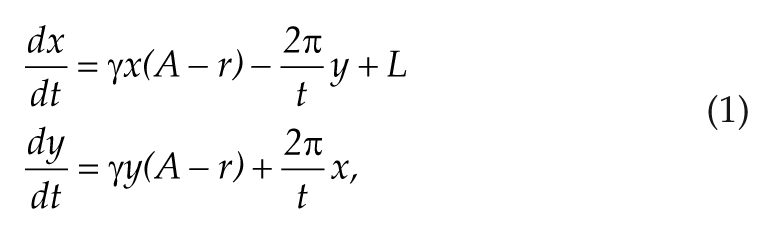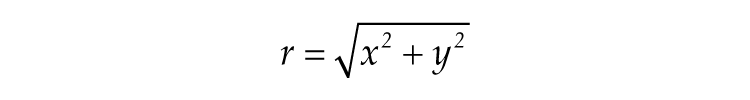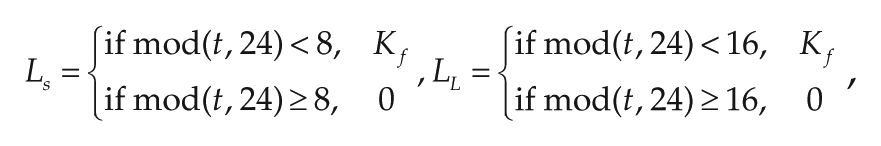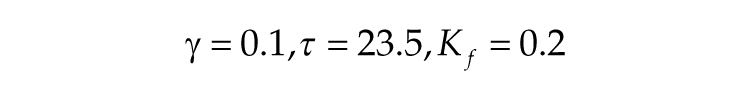Abstract
The suprachiasmatic nucleus (SCN) adapts to both the external light-dark (LD) cycle and seasonal changes in day length. In short photoperiods, single-cell activity patterns are tightly synchronized (i.e., in phase); in long photoperiods, these patterns are relatively dispersed, causing lower amplitude rhythms. The limit cycle oscillator has been used to describe the SCN’s circadian rhythmicity and predicts that following a given perturbation, high-amplitude SCN rhythms will shift less than low-amplitude rhythms. Some studies reported, however, that phase delays are larger when animals are entrained to a short photoperiod. Because phase advances and delays are mediated by partially distinct (i.e., nonoverlapping) biochemical pathways, we investigated the effect of a 4-h phase advance of the LD cycle in mice housed in either short (LD 8:16) or long (LD 16:8) photoperiods. In vitro recordings revealed a significantly larger phase advance in the SCN of mice entrained to short as compared to long photoperiods (4.2 ± 0.3 h v. 1.4 ± 0.9 h, respectively). Surprisingly, in mice with long photoperiods, the behavioral phase shift was larger than the phase shift of the SCN (3.7 ± 0.4 h v. 1.4 ± 0.9 h, respectively). To exclude a confounding influence of running-wheel activity on the magnitude of the shifts of the SCN, we repeated the experiments in the absence of running wheels and found similar shifts in the SCN in vitro in short and long days (3.0 ± 0.5 h v. 0.4 ± 0.9 h, respectively). Interestingly, removal of the running wheel reduced the phase-shifting capacity of mice in long days, leading to similar behavioral shifts in short and long photoperiods (1.0 ± 0.1 h v. 1.0 ± 0.4 h). As the behavioral shifts in the presence of wheels were larger than the shift of the SCN, it is suggested that additional, non-SCN neuronal networks in the brain are involved in regulating the timing of behavioral activity. On the basis of the phase shifts observed in vitro, we conclude that highly synchronized SCN networks with high-amplitude rhythms show a larger phase-shifting capacity than desynchronized networks of low amplitude.
Circadian rhythms are intrinsic to most organisms and are controlled by an endogenous pacemaker or clock. In mammals, the circadian clock is located in the suprachiasmatic nucleus (SCN) within the anterior hypothalamus (Ralph et al., 1990). Individual SCN neurons are genetically encoded for rhythm generation (Welsh et al., 2010) and are considered to function as autonomous single-cell oscillators. To achieve robust oscillations and high output amplitude, these oscillators in the SCN are mutually coupled and require sufficient phase synchrony (Welsh et al., 2010). Entrainment to the external light-dark (LD) cycle is mediated by the retinohypothalamic tract, which projects directly from the retina to the SCN via glutamatergic and pituitary adenylate cyclase-activating polypeptide (PACAP)–containing fibers (Morin and Allen, 2006). The influence of light on the clock depends on the clock’s phase: light pulses applied at the beginning of the night produce a phase delay, whereas light pulses applied at the end of the night produce a phase advance (Johnson, 1999). After a phase shift, the rhythm resumes with the previous period (i.e., before a light pulse was applied). Because this behavior is typical of a limit cycle oscillator, the circadian clock has been modeled extensively as such an oscillator (Kronauer et al., 1982; Goldbeter, 1995; Winfree, 2001; Forger and Peskin, 2003; Abraham et al., 2010; Phillips et al., 2013). The phase-shifting capacity of a limit cycle oscillator depends on the amplitude; high-amplitude oscillators have less phase-shifting capacity than low-amplitude oscillators. This limit cycle behavior has been shown in unicellular organisms (Johnson and Hastings, 1989), and for example, in cyanobacterium Synechococcus elongatus, high-amplitude rhythms have a weak phase-shifting capacity, whereas low-amplitude rhythms show large-magnitude shifts (Hosokawa et al., 2013).
The amplitude of the phase-response curve (PRC) (i.e., phase-shifting capacity) is not fixed but is dependent on the photoperiodic history of the organism. After adaptation to a short photoperiod, the amplitude of the PRC is larger than in long photoperiods (Refinetti, 2002; Pittendrigh et al., 2008; vanderLeest et al., 2009). The phase-shifting capacity in long and short days can be mimicked in vitro by application of the glutamate receptor agonist N-methyl-D-aspartate (NMDA), and thus, it seems to be determined at the level of the SCN (vanderLeest et al., 2009). In short photoperiods, the neurons of the SCN become more synchronized, leading to high-amplitude rhythms, while in long photoperiods, the neurons are more desynchronized, leading to low-amplitude rhythms (Schaap et al., 2003; Brown et al., 2005; Rohling et al., 2006; Inagaki et al., 2007). Thus, based on limit cycles, one would have expected to obtain large phase shifts in the SCN from a long photoperiod, which is in contrast to the experimental result. These findings suggest that networks of single-cell limit cycle oscillators may have emergent properties that are not present at the single-cell level.
To date, however, studies have been limited to behavioral experiments, and the response to phase-advancing pulses has not been tested at the level of the SCN (Pittendrigh et al., 1991; Refinetti, 2002; Refinetti, 2003). This lack of data regarding phase advances in the SCN has hampered our ability to generalize the results regarding limit cycle oscillator behavior to the SCN network. Testing the SCN’s phase-advancing behavior is particularly relevant, as the intracellular signaling cascade that is triggered by glutamate release differs between delays and advances (Ding et al., 1998; Gillette and Mitchell, 2002; Schwartz et al., 2011).
Here, we investigated the advancing responses of the SCN and behavior of mice with long and short photoperiods. As C57 mice show only small advancing phase shifts in response to a light pulse, we subjected the mice to an advance of the LD cycle to induce an advanced rhythm (vanderLeest et al., 2009). In vitro recordings of the SCN’s electrical activity revealed that the SCN in mice with short photoperiods phase-advanced significantly more than the SCN from mice entrained to long photoperiods. Surprisingly, the behavioral phase advances observed when a running wheel was available to the mice were significantly larger in long photoperiods as compared to short photoperiods. When the running wheel was removed, the behavioral phase-advancing capacity of the long photoperiod–entrained mice was significantly reduced, but the shifts in the SCN were unaltered, indicating that the occurrence of intense physical activity (e.g., running-wheel activity) can influence the magnitude of behavioral phase advances without affecting the SCN. Based on its phase-response properties, we conclude that unlike typical limit cycle oscillators, the SCN network does not have an inverse phase relationship between amplitude and phase-shifting capacity.
Materials and Methods
Ethics Statement
All experiments were performed in accordance with national animal welfare laws and were approved by the Animal Experiment Ethics Committee of Leiden University Medical Center.
Behavioral Experiments
C57BL/6 mice were entrained to either a long (LD 16:8) or short (LD 8:16) photoperiod for at least 30 days prior to experimentation. The mice were housed in clear plastic cages, and locomotor activity was monitored by either passive infrared detectors or a running wheel and recorded using Actimetrics software (Wilmette, IL, USA). The animal cabinets contained 9 cages each and were lighttight and illuminated using white fluorescent tubes. The light intensity at the bottom of each cage was approximately 85 µW/cm2. After 30 days of entrainment, the mice were subjected to a phase-advancing light stimulus. Because C57 mice show only small advancing phase shifts in response to a light pulse, the mice were subjected to a phase advance of the LD regime. The advance was accomplished by advancing the time of light onset. We chose to apply a 4-h phase advance in the LD cycle (rather than a 6-h advance) to ensure that the advanced light cycle would exclusively cover the advance part of the PRC (vanderLeest et al., 2009). This 4-h phase advance in the LD regime was then followed by 1 fully shifted LD cycle. After receiving this phase-advancing protocol, the mice were housed in continuous darkness (DD) for at least 10 days. The behavioral phase shift was calculated as the time difference between the activity onset (e.g., running-wheel activity) before the advance in the LD regime and the time of activity onset in DD. Activity onset was determined using ClockLab analysis software (Actimetrics, Wilmette, IL, USA).
In Vitro Experiments
Mice were entrained to either long or short photoperiod days as described above. The SCN slices were prepared just before light offset in the second cycle following the 4-h advance in the LD cycle, at external time (ExT, with ExT 12 defined as middle of the day [Daan et al., 2002]) 15.5 h and 19.5 h, for the short and long photoperiod groups, respectively. Control experiments were performed using mice that were not subjected to a 4-h phase advance in the LD regime. Brain slices (~450 µm thick) were prepared using a tissue chopper, and the slice containing the SCN was transferred to a laminar flow chamber within 6 minutes after decapitation (Schaap et al., 2003). The tissue was bathed in bicarbonate-buffered artificial cerebrospinal fluid that was gassed by continuously blowing a warmed, humidified mixture of O2 (95%) and CO2 (5%) over the solution. The slice was submerged in the solution and stabilized using an insulated tungsten fork. The slice settled in the recording chamber for approximately 1 h before the electrodes were placed in the center of the SCN. Of note, there are no consistent phase differences between SCN regions in electrical activity rhythms. In the intact SCN, electrical activity patterns are integrated, as opposed to patterns in gene expression (Meijer et al., 2010). Action potentials were recorded using 50-µm or 75-µm 90% platinum/10% iridium electrodes. The signals were amplified 10,000 times and band-pass filtered (0.3 Hz low pass, 3 kHz high pass). The action potentials that exceeded a predetermined threshold well above noise (~5 µV) were counted in 10-second bins using a custom-made automated computer program.
Data Analysis
The electrophysiological data were analyzed using a custom-made program in MATLAB (The Mathworks, Natick, MA, USA) as described previously (VanderLeest et al., 2007). The time of maximum activity was used as a marker of the phase of the SCN and was determined as the first peak in multiunit activity (both for the control and phase-advanced groups). Multiunit recordings of at least 24 h in duration that expressed a clear peak in multiunit activity were moderately smoothed using a least-squares algorithm (Eilers, 2003). Subsequently, the SCN peak time, the peak width, and the relative peak amplitude (peak-to-trough ratio) of the first cycle in vitro were determined. Statistical analyses were performed using Origin 7 (OriginLab Corp., Northampton, MA, USA). All summary data are reported as the mean ± the standard error of the mean (SEM). p values were calculated using the 2-tailed Student t test, and differences with p < 0.05 were considered to be statistically significant.
Model
We examined the difference in amplitude and phase-shifting capacity between mice entrained to long and short photoperiods using a mathematical model based on limit cycle oscillator theory. We refer to high- and low-amplitude rhythms when we discuss the biological system, whereas we refer to high- and low-amplitude oscillations when we discuss the mathematical oscillator. The Poincaré model is a model for describing limit cycle oscillators and is used to describe the 24-h oscillations in the SCN (Abraham et al., 2010). This model is a generic model with 2 variables (x and y) in Cartesian coordinates and is described as follows:
where γ represents the relaxation parameter, A and τ represent the amplitude and free running period of the SCN, respectively, L is the light term, and r is the amplitude of the SCN oscillator. r is defined as follows:
Experimental data showed that the amplitude of the SCN rhythm is higher in short photoperiods than in long photoperiods (vanderLeest et al., 2009; Ramkisoensing et al., unpublished data). Without loss of generality, the amplitude A is defined as AS = 2 for short days and AL = 1 for long days. The light term L also differs between photoperiods; LS represents the light term under a short photoperiod, and LL represents the light term under a long photoperiod. LS and LL are defined as follows:
where Kf represents the light intensity. The other parameters are the same for the short and long photoperiods and are defined as follows:
We used the fourth-order Runge-Kutta method with a time increment of 0.01 h to numerically simulate this system. The initial 100,000 time increments were excluded from the analysis to avoid the effect of transients. The initial conditions were selected randomly from a uniform distribution in the range of 0 to 1 for the variables x and y in the Poincaré model.
The simulations produced a 3.0-h phase advance for long photoperiods and a 1.5-h phase advance for short photoperiods following a 4-h advance in the LD cycle (Suppl. Fig. S1). The limit cycle oscillator theory predicts that, in line with the outcomes of the simulation, the phase shift will be larger for small-amplitude rhythms than for large-amplitude rhythms. Therefore, we consider our protocol to be valid for testing the phase-advancing properties of the SCN for short and long photoperiods, and we consider that deviations from the predicted outcome cannot be attributed to our protocol and should therefore be considered meaningful.
Results
Large Advances in the SCN on Short Days
The SCN of mice that were entrained to either a short or long photoperiod with access to a running wheel were prepared following a 4-h phase advance in the LD regime. Electrical recordings (Fig. 1A and 1B) revealed a narrow waveform in the SCN of mice entrained to short photoperiods (8.91 ± 0.64 h; n = 5 mice) and a significantly broader peak in the SCN of mice entrained to long photoperiods (15.53 ± 1.93 h; n = 6 mice) (p < 0.01). The circadian phase of the SCN was defined as the maximum SCN activity (SCNTmax), and the phase advance was defined as the difference between the subjective time of the SCNTmax in the phase-advanced mice and the SCNTmax of the control (i.e., non–phase-advanced) mice. In the short-day and long-day control SCN groups, the SCNTmax occurred at ExT 11.7 and 12.5, respectively (i.e., midday). Mice entrained to short days showed a phase advance of 4.2 ± 0.3 h (n = 8) in the SCNTmax after a 4-h advance in the LD regime (Fig. 1A and 1C). In contrast, the mice with long days showed an advance of 1.4 ± 0.9 h (n = 7) in the SCNTmax (Fig. 1B and 1C). The SCN’s phase-advancing capacity of mice that were entrained to a short day in the presence of a running wheel was significantly larger than the SCN’s phase-advancing capacity of mice entrained to a long day (p < 0.001) (Fig. 1C and Table 1).
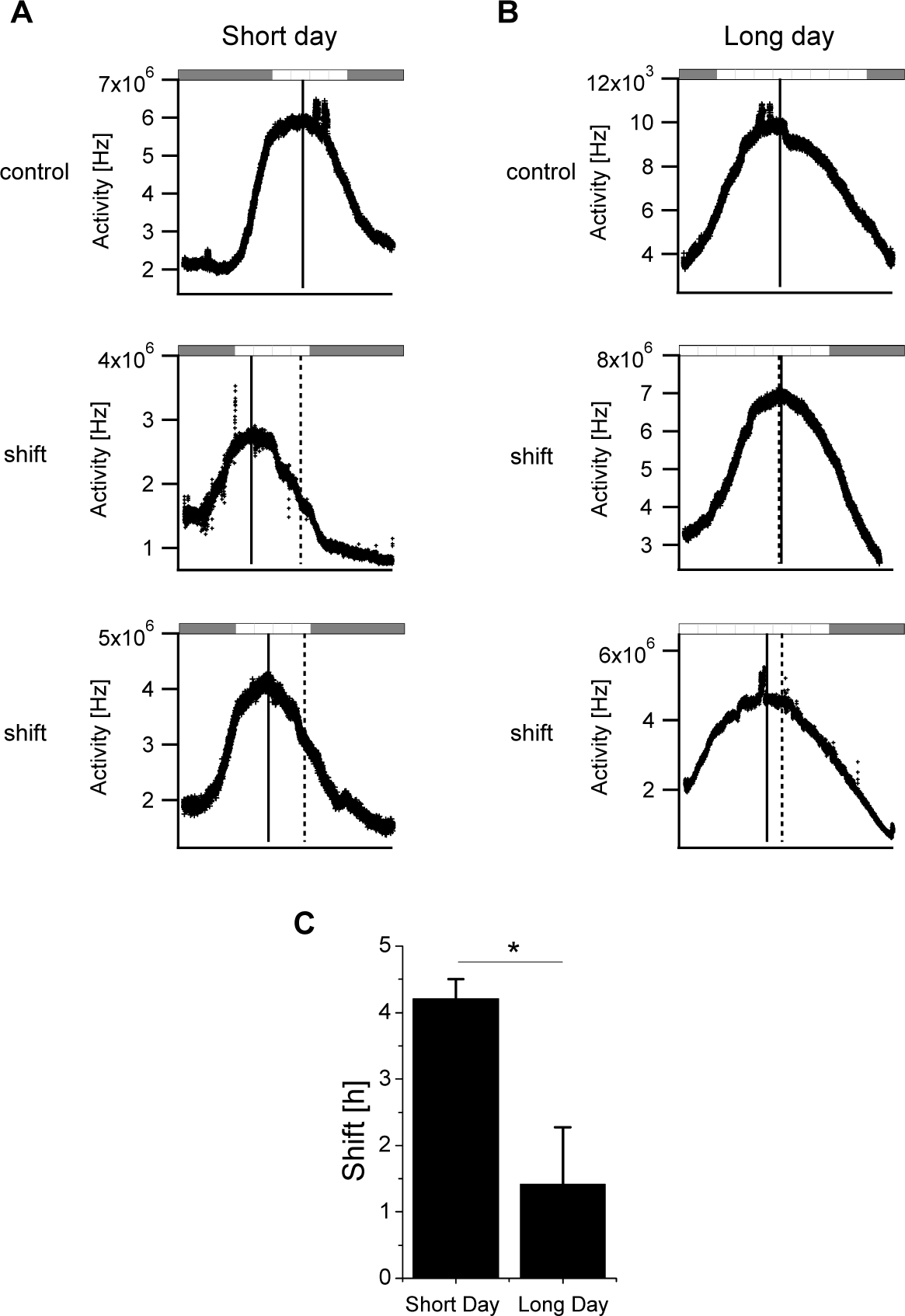
Phase advances in electrical activity rhythms recorded in the SCN isolated from mice entrained to a short or long photoperiod in the presence of a running wheel. (A, B) Examples of electrical activity recorded from the SCN of mice entrained to either a short-day (LD 8:16) protocol (A) or a long-day (LD 16:8) protocol (B). The plotted LD regimes were extrapolated from the LD schedules to which the mice were entrained prior to the preparation of the SCN slices. The upper panels show the representative electrical activity measured from the SCN isolated from mice that did not experience a 4-h advance in the LD regime (control). The middle and lower panels show the electrical activity recorded from the SCN of mice with short and long days following a 4-h phase advance in the LD regime. The dashed vertical lines indicate the mean SCNTmax of the control groups, and the solid vertical lines indicate the SCNTmax of each example recording. The magnitude of the phase advance of the SCN was calculated as the difference between the mean control SCNTmax and the shifted SCNTmax. (C) Summary of the advance in the activity rhythm of the SCN prepared from mice with short days (n = 8) and long days (n = 7). *p < 0.05.
Phase Advances in the SCN and Behavior of Mice Previously Entrained to Short Days or Long Days in the Presence or Absence of a Running Wheel
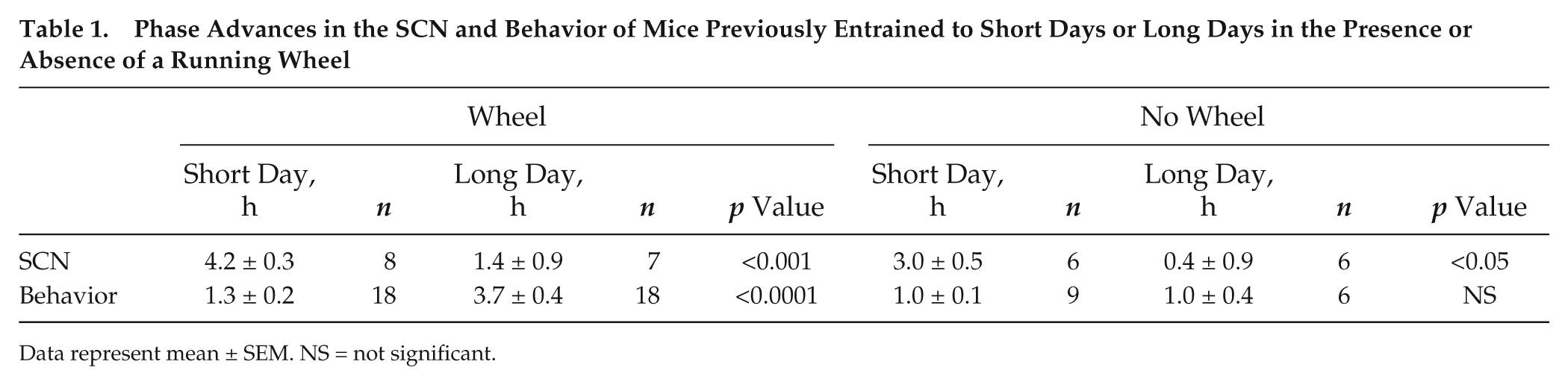
Data represent mean ± SEM. NS = not significant.
In the Presence of a Running Wheel, the Behavioral Phase Advance Is Larger in Mice with Long Days
In both the short and long day groups, the mice were physically active almost exclusively during the dark phase (Fig. 2A and 2B). The behavioral phase advance following a 4-h advance in the LD regime was measured using wheel-running onset in DD. Mice with short days had a behavioral phase advance of 1.3 ± 0.2 h (n = 18) (Fig. 2A and 2C), and mice with long days had a phase advance of 3.7 ± 0.4 h (n = 18) (Fig. 2B and 2C). Thus, when given free access to a running wheel, the phase-advancing capacity of mice in long photoperiods was significantly larger than in mice in short photoperiods (p < 0.0001) (Table 1 and Fig. 2C).
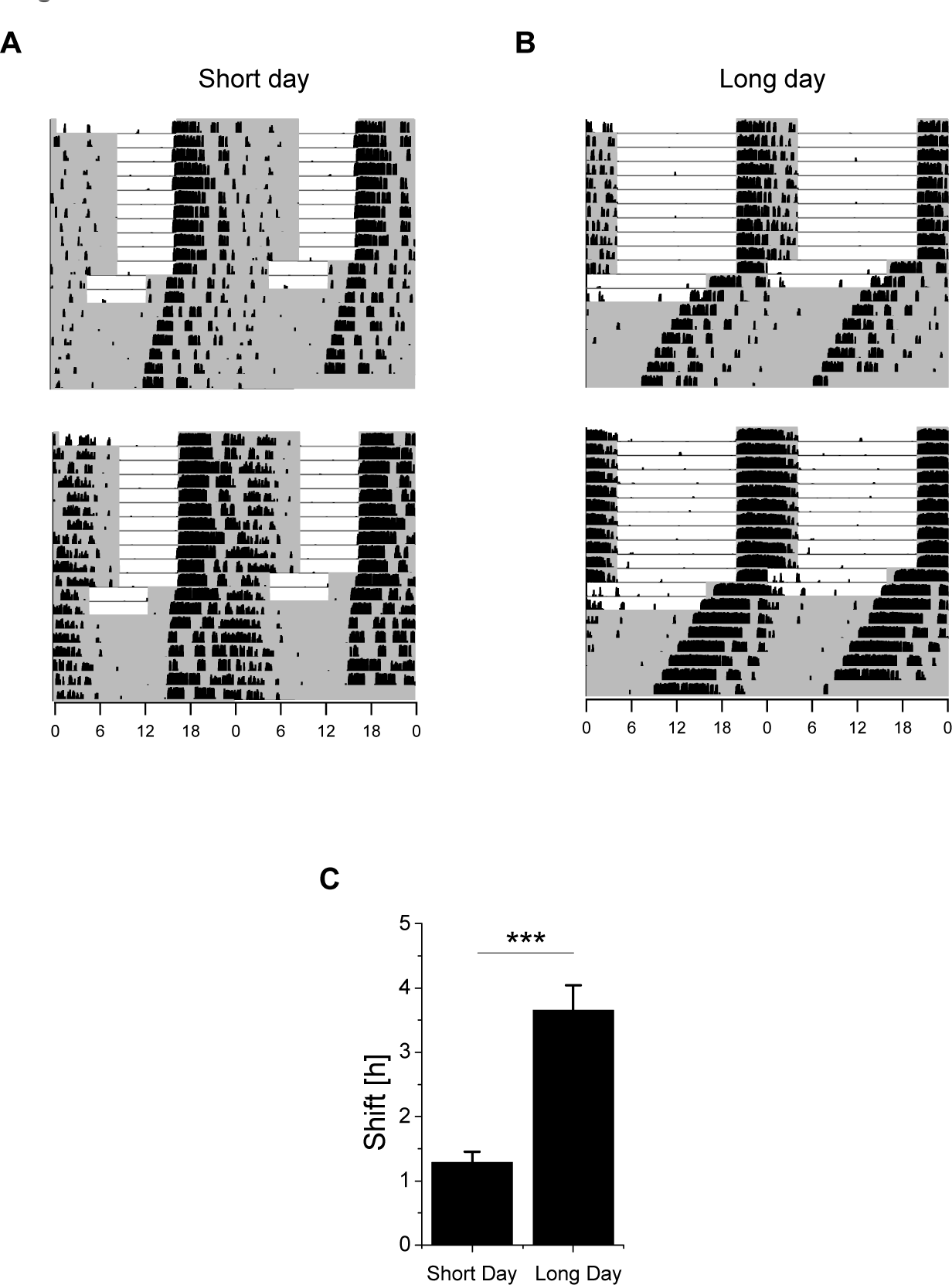
Phase advance in the running-wheel activity of mice following a 4-h phase advance in the LD cycle. (A, B) Examples of running-wheel activity in 4 representative mice entrained to either a short (A) or long (B) photoperiod. Each actogram is double plotted and shows consecutive days on successive lines; the white areas are the light periods, and the shaded gray areas are the dark periods. The vertical black upticks indicate running-wheel activity. After 30 days of entrainment to a short or long photoperiod, the mice were exposed to a 4-h phase advance. The animals were then kept in the shifted LD regime for 1 additional complete cycle and afterward kept in DD. The phase advance was measured as the difference between the time of wheel-running onset before the phase shift and the time of wheel-running onset in DD after the phase shift (transients were excluded). (C) Summary of the advances in the wheel-running behavior of mice in short days (n = 18) and long days (n = 18). *p < 0.0001.
Phase Advance in the SCN In Vitro Is Not Affected by Running-Wheel Activity
To exclude any potential effect of running-wheel activity on the behavioral advances, we subjected mice to a 4-h phase advance in the absence of a running wheel; the SCN slices were then prepared immediately following the phase advance and recorded. The electrical recordings revealed a narrow waveform for the SCN of mice with short days (9.15 ± 0.77 h; n = 6 mice) and a significantly broader waveform for the SCN of mice with long days (12.84 ± 1.44 h; n = 5) (p < 0.05). Following the phase advance, the SCN of mice with short days had a phase advance of 3.0 ± 0.5 h (n = 6) in the SCNTmax (Fig. 3A and 3C), whereas the SCN of mice with long days had a phase advance of only 0.4 ± 0.9 h (n = 6) (Fig. 3B and 3C). Thus, the SCN’s phase-advancing capacity of mice with short photoperiods was significantly larger than that of mice with long photoperiods (p < 0.05) (Fig. 3C and Table 1). Importantly, these results did not differ significantly from the results obtained from SCN recordings from mice that had access to a running wheel (p = 0.06 for short-day mice and p = 0.46 for long-day mice). We pooled the data from the SCN recordings both with and without a running wheel and found a significantly higher normalized amplitude in the SCN on short days (0.58 ± 0.04) as compared to long days (0.45 ± 0.03) (p < 0.05).
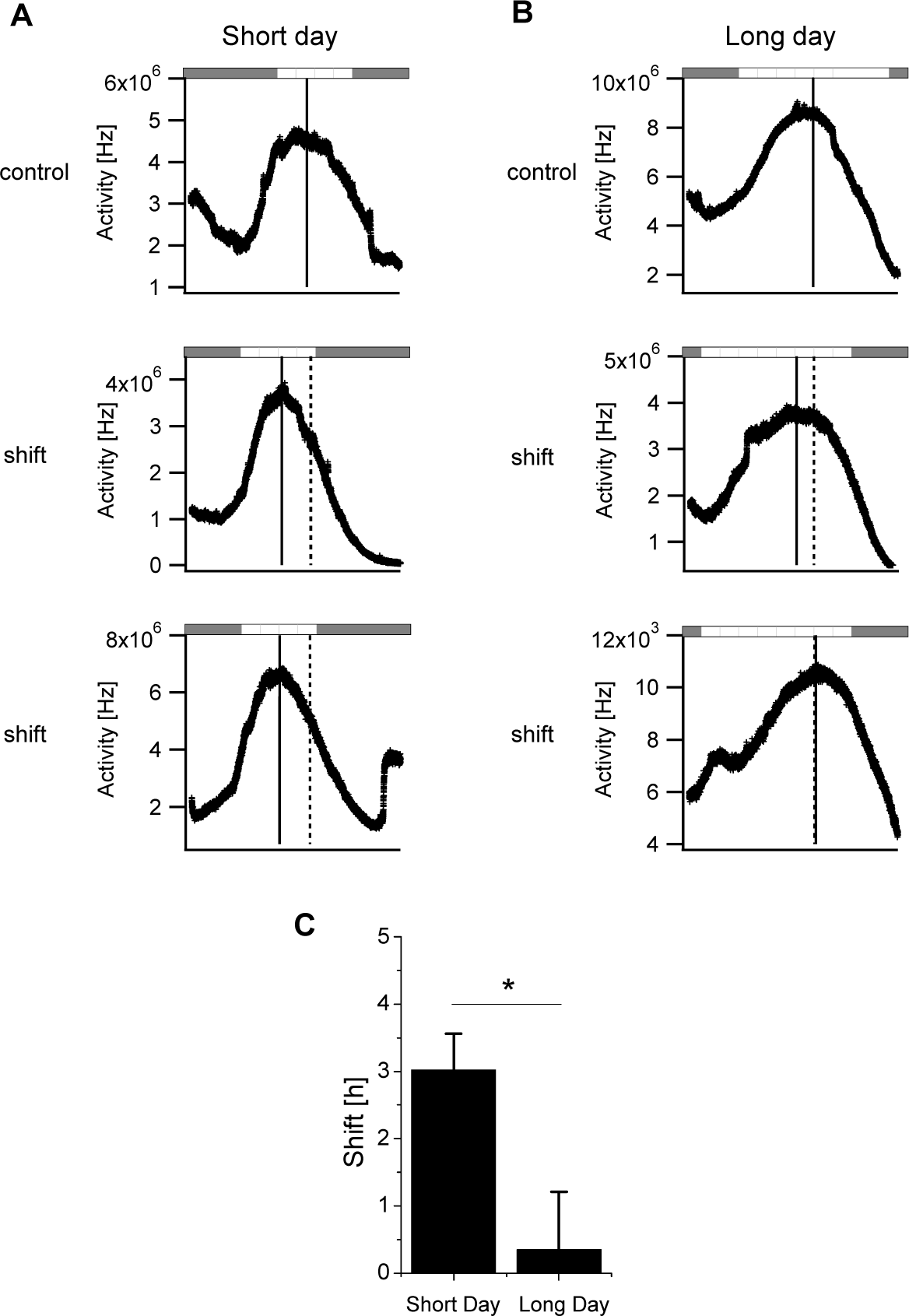
Phase advances in the electrical activity rhythm recorded in the SCN of mice entrained to a short or long photoperiod in the absence of a running wheel. (A, B) Examples of electrical activity recorded from the SCN of mice entrained to either a short-day (A) or a long-day (B) protocol; see Figure 1 for details. (C) Summary of the advance in the activity rhythm of the SCN from mice entrained to short days (n = 6) and long days (n = 5). *p < 0.05.
In the Absence of a Running Wheel, the Behavioral Phase Advance Is Similar in Mice with Short and Long Days
We next compared the effect of a 4-h phase advance on the behavioral shift in mice that were entrained to short or long days in the absence of a running wheel. Following a 4-h advance in the LD regime, the resulting behavioral phase advance was determined in DD by recording the mouse’s activity using passive infrared detectors (Fig. 4A and 4B). The behavioral phase advance was similar between the mice with short days (1.0 ± 0.1 h; n = 9) and long days (1.0 ± 0.4 h; n = 6) (p = 0.93) (Fig. 4C and Table 1). Interestingly, entraining the mice in the absence of a running wheel had no effect on the behavioral phase advance in the short day–entrained mice; in contrast, entraining the mice in the absence of a running wheel significantly reduced the phase-advancing capacity of the long day–entrained mice (1.0 ± 0.4 h v. 3.7 ± 0.4 h in the presence and absence of a running wheel, respectively; p < 0.005) (compare Fig. 4C with Fig. 2C).
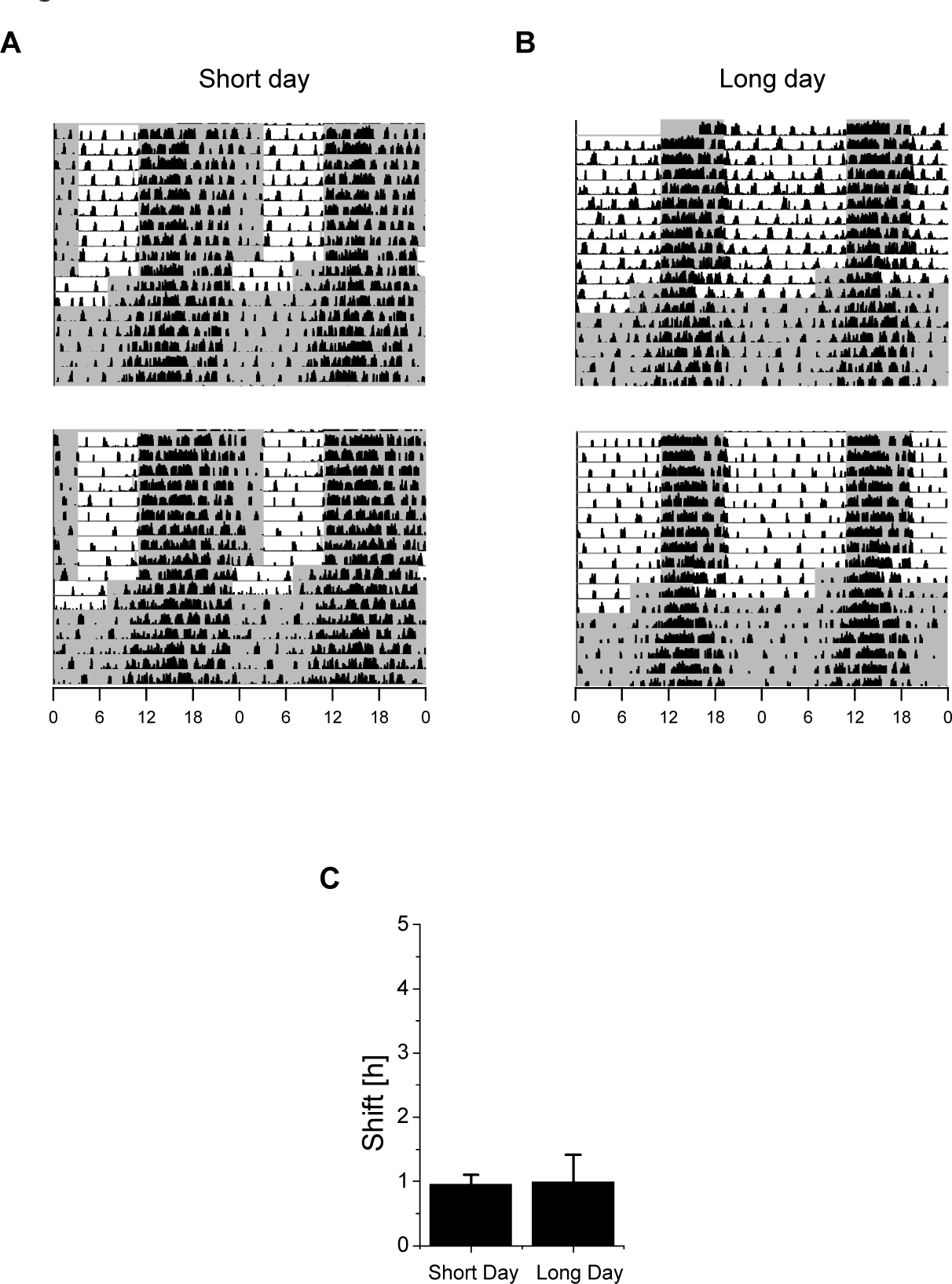
Phase advance in the activity behavior of mice following a 4-h phase advance in the LD cycle. (A, B) Behavioral activity was recorded using a passive infrared detector for mice entrained to either a short (A) or a long (B) photoperiod; see Figure 2 for details. (C) Summary of the advances in the behavioral activity of mice entrained to short days (n = 9) and long days (n = 9). The values did not differ significantly (p = 0.933).
Discussion
Previous studies have reported that the SCN with high-amplitude electrical activity rhythms phase-delay significantly more than low-amplitude SCN rhythms upon stimuli of equal sizes (vanderLeest et al., 2009). The aim of this study was to investigate the effect of a phase-advancing perturbation on high- and low-amplitude SCN rhythms. We first confirmed that the activity in the SCN is more tightly synchronized in mice entrained to a short photoperiod than in mice entrained to a longer photoperiod; this increased synchronization was reflected by a narrower peak width and a higher amplitude in the SCN’s activity rhythm of the short photoperiod–entrained mice. These results are consistent with previous reports of higher amplitude rhythms in synchronized neuronal populations (Rohling et al., 2006; Schaap et al., 2003; vanderLeest et al., 2009). We then induced a phase advance in the LD cycle and observed a large phase advance in the SCN of mice entrained to short photoperiods and a significantly smaller advance in the SCN of mice entrained to long photoperiods. These results confirm former studies on the phase-delaying behavior of high- and low-amplitude SCN rhythms (vanderLeest et al., 2009). Our findings are, however, in contrast with the predictions of our own limit cycle oscillator simulations, in which we tested the validity of our protocol, and with predictions from limit cycle oscillator theory in general. We argue that neurons within a synchronized network population, with presumably consistent phase-shifting responses, will result in a large-amplitude PRC of the population ensemble, whereas neurons within a desynchronized network will show diverse responses to the perturbing stimulus, resulting in a small-amplitude PRC (vanderLeest et al., 2009; Meijer et al., 2012). Therefore, our results are relevant, as they illustrate that limit cycle oscillator behavior at the SCN network level is distinct from limit cycle behavior at the single-cell level. We also observed striking differences between the phase-shifting capacity of the SCN and the behavioral phase-shifting capacity. Specifically, in mice with long photoperiods, the phase advance in the animal’s behavioral activity rhythms was considerably larger than the advance in the SCN itself, suggesting that other brain structures, in addition to the SCN, are in control of resetting behavioral activity rhythms.
In Vitro Results
In vitro recordings of the control (i.e., non–phase-shifted) SCN revealed high levels of electrical activity during the subjective day and low levels of electrical activity during the subjective night; this pattern was observed in mice with short and long days. In both groups, the maximum SCN activity (SCNTmax) was close to midday. The waveform characteristics under short and long photoperiods are consistent with previous electrophysiological (VanderLeest et al., 2007; Brown and Piggins, 2009; vanderLeest et al., 2009) and molecular (Hazlerigg et al., 2005; Johnston et al., 2005; Inagaki et al., 2007; Naito et al., 2008; Myung et al., 2012) studies. Specifically, the waveform was narrow in the SCN of short-day mice (due to the tightly synchronized activity between the individual neurons in the network) and broad in the SCN of long-day mice (due to asynchronous activity between the individual neurons) (vanderLeest et al., 2009). Thus, by entraining the mice to either a short or long photoperiod, we could drive the SCN’s neuronal population into either high or low synchrony.
We found that the SCN of mice entrained to short days phase-advanced significantly more than the SCN of long-day mice. In vitro measurements revealed that the SCN rhythm of the short-day group was significantly phase advanced by 4.2 h and showed, accordingly, complete synchronization to the shifted LD cycle, while the SCN rhythm of the long-day group remained in phase with the LD cycle prior to the shift. To examine the effect of behavioral activity on the phase-shifting capacity of the SCN, we repeated this experiment in the absence of a running wheel. This experiment was particularly relevant, as behavioral activity is more condensed in relative short nights (i.e., longer photoperiods) and therefore may have influenced our results. However, in vitro, we again found a significantly larger phase advance in the SCN of mice entrained to a short as compared to a long photoperiod (3.0 h v. 0.4 h, respectively), confirming that the phase-advancing response is larger in the synchronized network (i.e., in the SCN from short days) than in the nonsynchronized network (i.e., in the SCN from long days). Thus, the shifts obtained in vitro were independent of the presence of behavioral activity.
Advances v. Delays
Testing the response to advances of the LD cycle was prompted by studies showing that the mechanisms underlying advances and delays differ. For example, light-induced glutamate release activates NMDA receptors, which triggers Ca2+ influx, activates nitric oxide synthase (NOS), and increases nitric oxide (NO) production. Blocking NMDA receptors or NOS activation inhibits glutamate-induced phase advances and phase delays (Ding et al., 1998). Several studies, investigating these intracellular signal transduction cascades, revealed that the divergence in the delaying and advancing signaling pathways occurs downstream of NO (Ding et al., 1998; Gillette and Mitchell, 2002; Antle et al., 2009; Schwartz et al., 2011). Important for light-induced resetting of the SCN are the clock genes mPer1 and mPer2 (Albrecht et al., 1997; Obrietan et al., 1998; Zylka et al., 1998; Field et al., 2000; Yan and Silver, 2002). Intracerebroventricular delivery of mPer1 antisense oligonucleotides reduced the light-induced phase delay in locomotor activity (Akiyama et al., 1999). Moreover, applying mPer1 antisense oligonucleotides suppressed glutamate-induced delays in neuronal firing rhythms in vitro (Akiyama et al., 1999). Finally, behavioral studies in hamsters revealed elevated Per1 mRNA levels after entrainment to short T-cycles (advances), whereas entrainment to long T-cycles (delays) altered Per2 expression (Schwartz et al., 2011). Our results show that the SCN’s response to an advancing shift is large for high-amplitude SCN rhythms and small for low-amplitude SCN rhythms, regardless of whether the mice were entrained in the presence or absence of a running wheel. Thus, despite the divergence between the advancing and delaying intracellular signaling pathways, the advances and delays appear to follow the same rule.
Behavioral Results with a Running Wheel
In response to a 4-h advance of the light cycle, followed by DD, we found a small advance in running-wheel activity in mice with short days (1.3 h). The magnitude of the phase shift differs from the shift of the SCN in vitro (4.2 h). It has been previously shown that a shift in behavior is smaller than in the SCN (Vansteensel et al., 2003; van Oosterhout et al., 2012). The difference was attributed to interactions between non-SCN structures and the SCN, which attenuate the behavioral shift and even the shift of the SCN when it is recorded in vivo. In contrast, when the SCN is measured in isolation, the endogenous phase is observable. Thus, in mice with short photoperiods, it is possible to explain the presence of both a small shift in behavior and a large shift in the SCN.
In contrast to this, mice that entrained to long days showed a large and immediate shift in wheel-running behavior after a 4-h phase advance of the LD cycle (3.7 h). The advance in wheel-running behavior of mice exposed to long days was found to be much larger than the advance measured in the SCN isolated from mice exposed to long days in the presence of a running wheel (1.4 h). This result is rather surprising and raises the question of how to explain the large shift in the behavioral activity of mice in long photoperiods, despite the relatively small shift in the SCN itself.
To investigate this apparent difference in phase-advancing capacity between behavior and SCN activity, we examined the behavioral responses more closely. The phase advance in running-wheel activity in mice with long days was large in magnitude and did not show transients, unlike light pulse–induced advances. As a result of the shift in the light regime, the mice spent the latter part of the day in darkness, resulting in intensified behavioral activity. Dark pulses are considered to be nonphotic stimuli and cause phase advances between midday and late day, where they induce behavioral activity (Janik and Mrosovsky, 1993; Biello and Mrosovsky, 1996). Although our protocol was designed to expose the mice to shifts in the light cycle, nonphotic stimuli of unequal strength existed between the short and long day groups (Reebs and Mrosovsky, 1989a; Reebs and Mrosovsky, 1989b; Yamanaka et al., 2008). Additional research would be required to identify brain structures that may function as secondary oscillators and shift to a larger extent than the SCN in response to behavioral activity.
Behavioral Results without a Running Wheel
To test for possible effects of running-wheel activity on the behavioral phase advance, we repeated the behavioral experiments in the absence of a running wheel. Mice that were entrained to a short day without a running wheel had a small (1-h) phase advance after a 4-h advance in the LD regime. This relatively small advance is similar to the 1.3-h advance in the mice that were entrained to a short day in the presence of a running wheel. These results suggest that running-wheel activity has little or no influence on the behavioral phase-advancing capacity of mice that are entrained to a short day. On the other hand, the behavioral phase advance in the long day–entrained mice was reduced significantly from 3.7 h to 1.0 h in mice that were entrained without a running wheel. From these results, we conclude that the phase-advancing response of the long day–entrained mice was induced predominately by running-wheel activity. Because the behavioral shift was much larger than the shift in the SCN, it is highly unlikely that the behavioral shift was driven by a shift in the SCN. Previous studies have reported accelerated re-entrainment of behavior to a shifted LD cycle when the animals had access to a running wheel (Deboer and Tobler, 2000; Yamanaka et al., 2008). Nevertheless, the most intriguing aspect of this finding was the absence of a large shift in the SCN in the presence of a large behavioral shift. Previous studies revealed that peripheral tissues and/or non-SCN brain structures can have a higher phase-advancing capacity than the SCN (Yamanaka et al., 2008; Sellix et al., 2012; Cao et al., 2013; Mohawk et al., 2013). For example, the habenula plays a role in regulating several circadian behaviors, including locomotor activity, and this structure can regulate activity rhythms either on its own or as part of a larger network (Paul et al., 2011). Our results are consistent with these findings and suggest that additional (non-SCN) brain structures also control behavioral activity rhythms.
Conclusions
For many decades, the phase-shifting capacity of the biological clock was believed to reflect the behavior of a limit cycle oscillator. Based on limit cycle oscillator theory, one would expect that high-amplitude oscillations would have a smaller shift than low-amplitude oscillators in response to a given stimulus. This prediction has indeed been confirmed in unicellular organisms, including Chlamydomonas, Euglena, Gonyaulax, Paramecium, and Neurospora (Malinowski et al., 1985; Johnson et al., 1989; Hosokawa et al., 2013). The amplitude of the SCN network is determined by 1) the single-cell amplitude and 2) synchrony among the single cells. In several previous studies, the amplitude of the SCN rhythm was reduced as a result of a reduction in single-cell amplitude rather than as a reduction in neuronal synchrony (Guilding et al., 2013; Vitaterna et al., 2006). According to expectations from limit cycle theory, the increase in the phase-shifting capacity in these mice is explainable by the reduction in single-cell amplitude. In our study, we have modified the electrical activity amplitude of the SCN rhythm, not by varying the amplitude of the individual neurons but by changing the synchrony among them (VanderLeest et al., 2007). We found that selective enhancement of neuronal synchrony leads to a large phase-shifting capacity. This enhanced phase-shifting capacity in the SCN with short days is in agreement with previous studies that reported a larger phase-delaying capacity of high-amplitude SCN oscillations compared to low-amplitude SCN oscillations (vanderLeest et al., 2009). Taken together, these data reveal a positive relationship between neuronal synchronization and phase-shifting capacity at the level of the SCN neuronal network.
Footnotes
Acknowledgements
The authors thank Henk Tjebbe van der Leest and Kit-Yi Yam for excellent technical and intellectual support. They also thank the anonymous reviewers for their excellent suggestions. This research was supported by the Netherlands Organization for Scientific Research (NWO), complexity grant number 645.000.010.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.