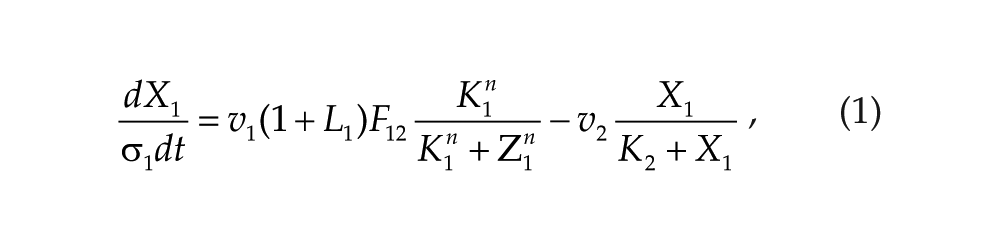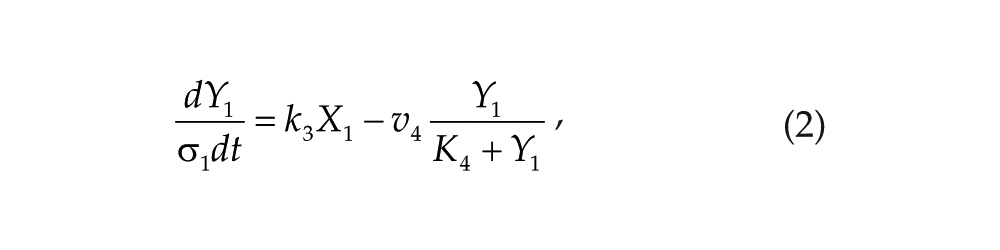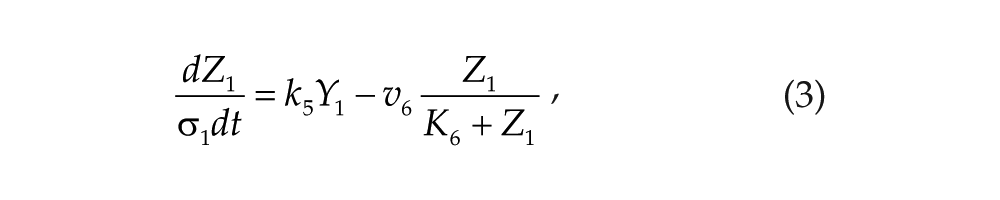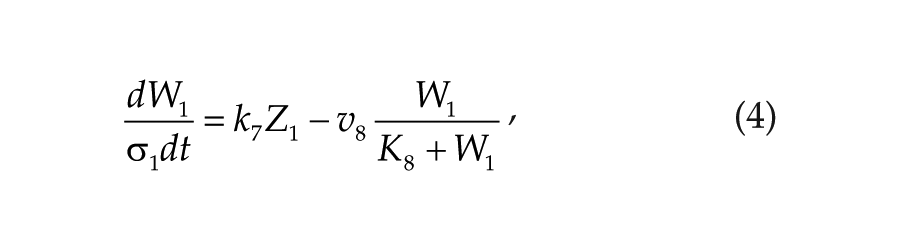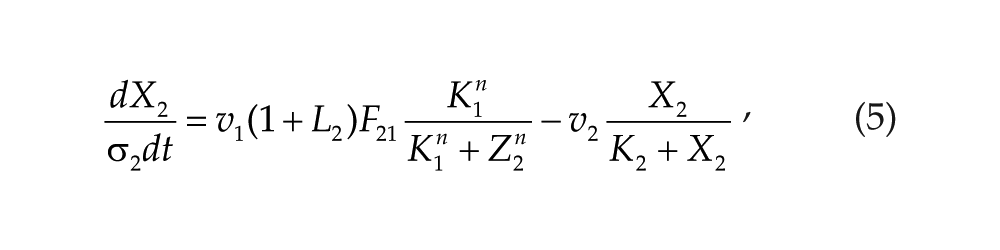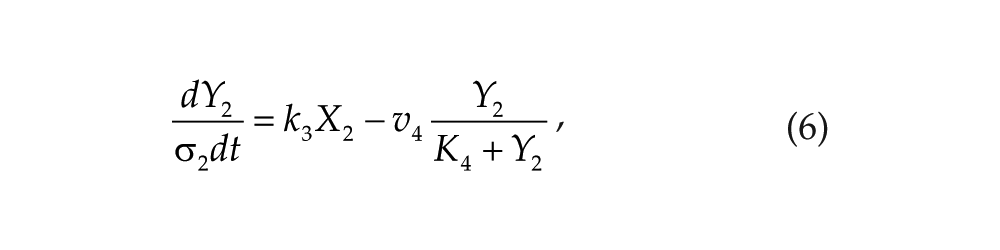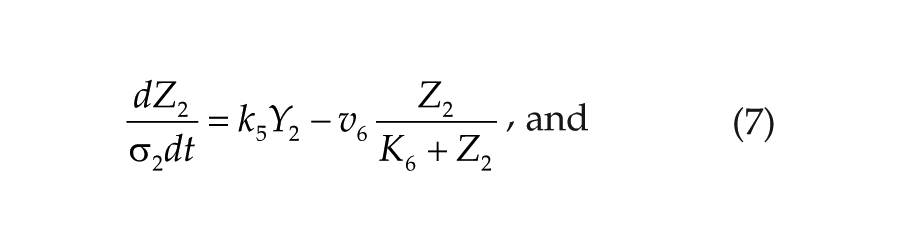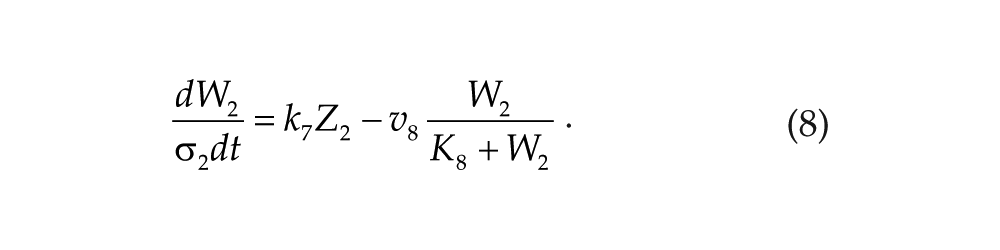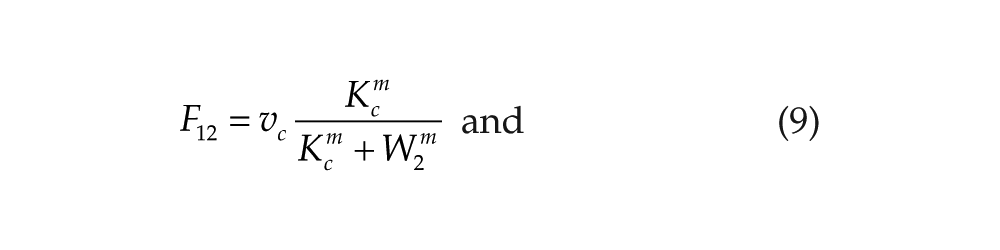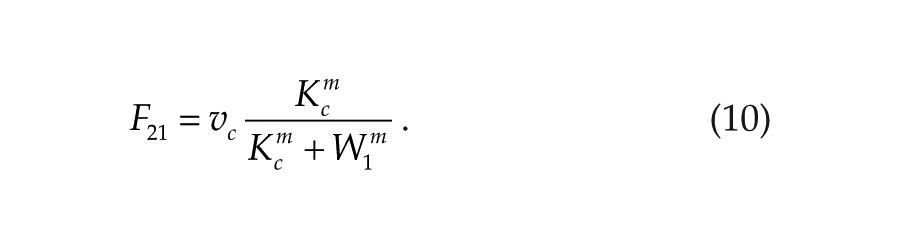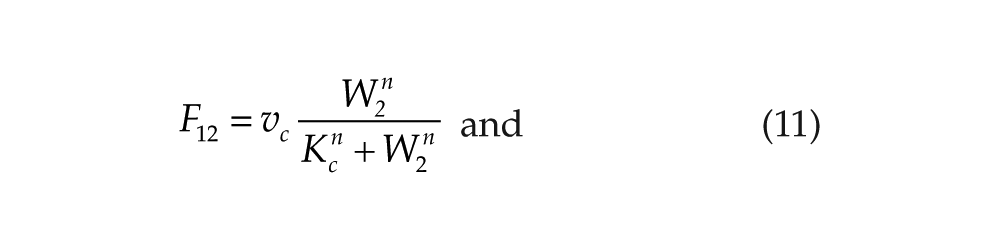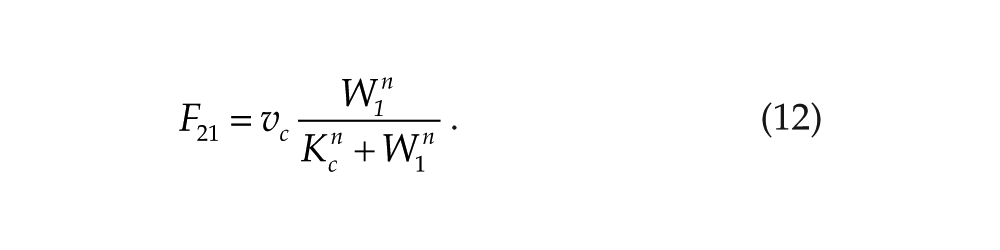Abstract
The circadian timekeeping system appears more complex in birds than in mammals. In mammals, the main pacemaker is centralized in the suprachiasmatic nuclei, whereas in birds, the pacemaker involves the interplay between the pineal and hypothalamic oscillators. In order to investigate the consequence of this complex mechanism, we propose here a mathematical model for the bird circadian clock. The model is based on the internal resonance between the pineal and hypothalamic oscillators, each described by Goodwin-like equations. We show that, consistently with experimental observations, self-sustained oscillations can be generated by mutual inhibitory coupling of the 2 clocks, even if individual oscillators present damped oscillations. We study the effect of constant and periodic administrations of melatonin, which, in intact birds, acts as the coupling variable between the pineal and the hypothalamus, and compare the prediction of the model with the experiments performed in pinealectomized birds. We also assess the entrainment properties when the system is subject to light-dark cycles. Analyses of the entrainment range, resynchronization time after jet lag, and entrainment phase with respect to the photoperiod lead us to formulate hypotheses about the physiological advantage of the particular architecture of the avian circadian clock. Although minimal, our model opens promising perspectives in modeling and understanding the bird circadian clock.
Keywords
As mammals, birds display circadian rhythms in their behavior and physiology. Although sharing similar genetic components, the circadian systems in birds and mammals appear different (Bell-Pedersen et al., 2005). In mammals, the central circadian pacemaker is located in the suprachiasmatic nuclei (SCN) of the hypothalamus. The SCN neurons can, upon synchronization, generate a robust, self-sustained circadian rhythm. Through neuronal and humoral output signaling pathways, the SCN then entrains circadian clocks located in peripheral tissues, such as the pineal, lungs, heart, liver, and others (Reppert and Weaver, 2002). In birds, the circadian pacemaking system appears to be more complex (Gwinner and Brandstätter, 2001). It requires the interaction of the pineal, the hypothalamus, and the retina, which all contain circadian clocks but produce a robust circadian rhythm only upon mutual interactions. To understand the dynamic consequences of such an organization and to highlight their possible physiological advantages, we developed and analyzed (by numerical simulation) a mathematical model.
The role of the pineal gland in the avian circadian system was recognized a long time ago (Gaston and Menaker, 1968; Takahashi and Menaker, 1979; Deguchi, 1979). Through the circadian release of melatonin, the pineal mediates various physiological functions, including the daily rest/activity cycle, seasonal changes in reproduction, migration, as well as circadian singing rhythm (Wang et al., 2012). Gaston and Menaker (1968) showed that pineal removal abolishes free-running circadian rhythms of locomotion in the house sparrow. Deguchi (1979) showed that the activity of N-acetyltransferase (AA-NAT), the key enzyme of melatonin synthesis, undergoes a marked circadian rhythm in the pineal gland of chickens. In addition, since the pineal was known to receive direct light input (independently on the retina), this organ was placed at the center of the circadian pacemaker. The role of the pineal in the circadian clock was further demonstrated by transplantation of the pineal (Zimmerman and Menaker, 1979): Receiver birds recover their circadian rhythm in the same phase as the one of the donor.
The above observations demonstrate the crucial role of the pineal in the avian circadian system. However, even in pinealectomized sparrows, some oscillatory components remain. First, the locomotor activity can be entrained by light-dark (LD) cycles in the same phase as in intact birds (Gaston and Menaker, 1968). Second, a transient, damped circadian rhythm of the locomotor activity is still displayed after transfer from LD to constant darkness (DD) (Gaston and Menaker, 1968). Furthermore, it was shown that melatonin rhythm is damped both in cultured, dispersed pineal cells (Robertson and Takahashi, 1988) and at the explanted tissue level (Takahashi et al., 1980), suggesting that other components are required to maintain the circadian rhythm in the pineal. By anatomic analogy with the situation encountered in mammals, Takahashi and Menaker (1982) identified the hypothalamus as another necessary component of the avian circadian pacemaker and proposed the idea that the circadian organization is based on mutual interactions between the retina, the pineal, and the hypothalamus (Menaker, 1982; Cassone et al., 1993; Brandstätter and Abraham, 2003) (Fig. 1).
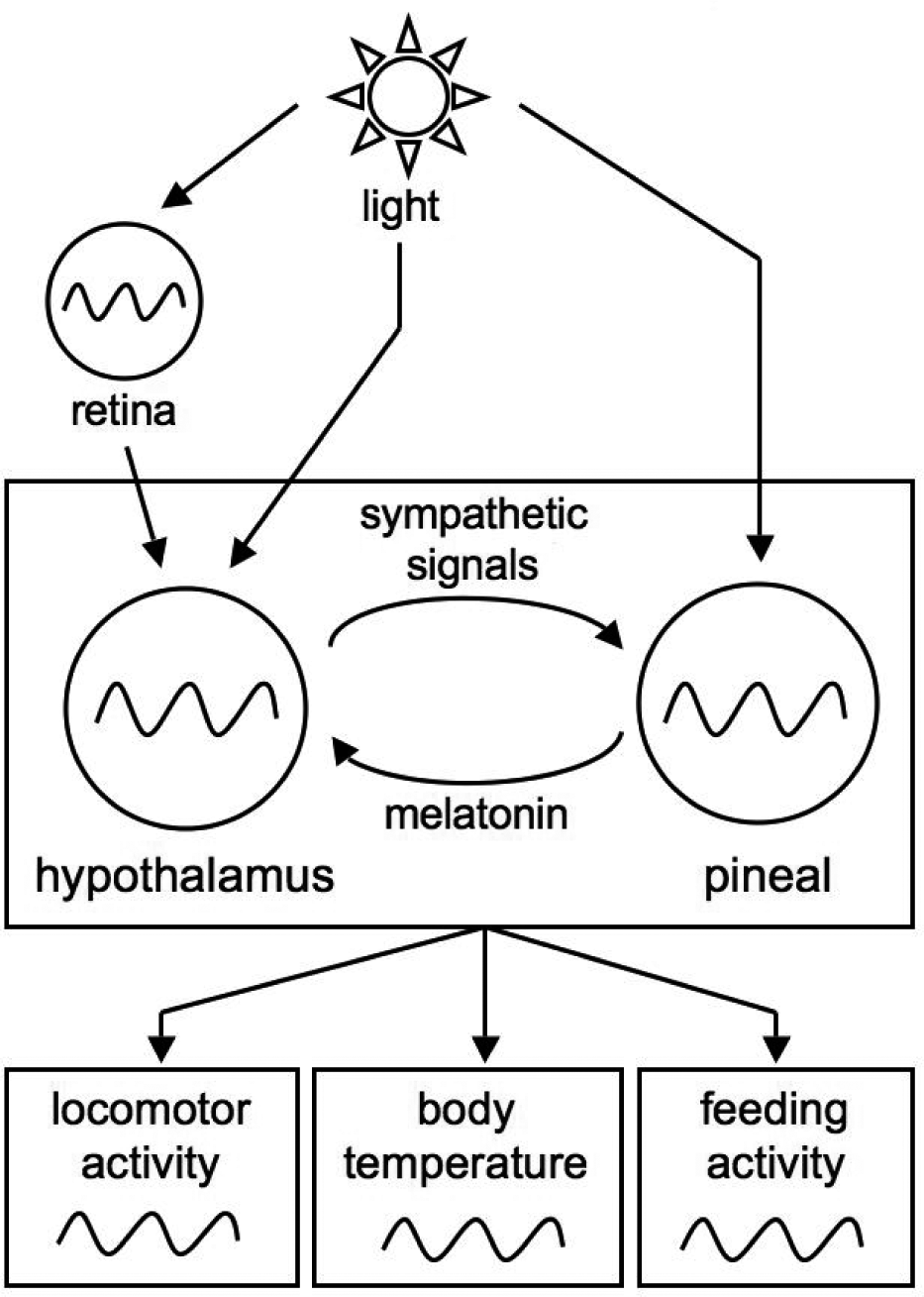
Scheme of the avian circadian system. The circadian system relies on the mutual interactions of the hypothalamic and pineal oscillators. Light acts on both organs via different pathways. Light entrains the hypothalamus through the retina, which also contains a circadian oscillator, but also directly (via encephalic photoreceptors), and acts directly on the pineal (via pinopsin photoreceptors). The avian circadian clock controls many physiological and behavioral outputs including the body temperature and locomotor activity.
This mechanism by which robust oscillations are generated through the interaction of several (possibly damped) oscillators was coined “internal resonance” by Gwinner (1989). It leads to several theoretical predictions (Gwinner and Brandstätter, 2001): Experimental elimination of endogenous melatonin would dampen the hypothalamic oscillator and thereby 1) increase the domain of entrainment by LD cycles and 2) permit faster resychronization after jet lag. Two types of experiments have been carried out to test these predictions in sparrows. On the one hand, the rhythm of the locomotor activity in pinealectomized birds can be entrained by a periodic, exogenous administration of melatonin of different periods (Gwinner and Benzinger, 1978; Heigl and Gwinner, 1995). On the other hand, experiments in which the LD cycle is delayed by 6 h have shown that pinealectomized birds resynchronize faster to the LD cycle (Hau and Gwinner, 1995; Kumar and Gwinner, 2005).
As in mammals, circadian oscillations in birds originate at the genetic level. The initial demonstration of rhythmic clock gene expression in the hypothalamus of birds was by Brandstätter et al. (2001). These authors identified and cloned the homolog of the mammalian Per2 gene in sparrows and measured a diurnal rhythm of its expression in the hypothalamus. The subsequent discovery of homologs of Per3, Cry1/2, Bmal1, Clock, and RevErbα (Yoshimura et al., 2000) let us suppose that the genetic mechanism of rhythm generation in birds is similar to the mammalian one: It most likely relies on interlocked feedback loops, the core regulation being a delayed, negative feedback loop (Bell-Pedersen et al., 2005). There are, however, a few differences worth mentioning. 1) No homolog of Per1 was found in birds. 2) The light signaling pathway in mammals requires perception by the retina, whereas in birds, besides the retina-dependent pathway, light can directly reach the pineal (Okano et al., 1994) or the hypothalamus (Foster and Soni, 1998). 3) CRY proteins do not act as photoreceptors in mammals, whereas they seem to have retained this function in birds (Bailey et al., 2002).
The avian SCN can be subdivided into the visual SCN (vSCN) and the medial SCN (mSCN) (Cantwell and Cassone, 2006). Only the vSCN contains melatonin receptors (Rivkees et al., 1989) and expresses circadian rhythms of physiological functions such as brain metabolism (Lu and Cassone, 1993). Abraham et al. (2002, 2003) measured the rhythmic expression of Per2 in the 2 parts of the SCN and found that the phase and amplitude are slightly different in these 2 regions: The amplitude is slightly reduced and the phase delayed in the vSCN (designated LHN for lateral hypothalamic nuclei in their paper) compared to the mSCN. Chong et al. (2003) measured Bmal1 expression in the retina and in the pineal and showed that Bmal1 rhythm is synchronous in these 2 organs and appears in opposition of phase with Per2 expression in the SCN. There are no simultaneous data on a given clock gene in different organs, but, as Per2 and Bmal1 are expressed (more or less) in phase opposition in the mammalian SCN, it seems likely that the circadian oscillations produced by the SCN, the pineal, and the retina clocks are roughly in phase.
By which molecular mechanism are the pineal and hypothalamic genetic clocks coupled? Melatonin, synthesized and released in a circadian fashion in the pineal, is the most obvious candidate (Lu and Cassone, 1993; Cassone et al., 2009). Melatonin receptors are present in the SCN cell membrane. The mechanism by which melatonin regulates the expression of clock genes is not yet elucidated. The Per2 gene, as well as other clock-controlled genes, contains multiple E-boxes, which are recognized by the Clock/Bmal1 dimer, but also the cyclic nucleotide response element, which responds to cAMP signaling. It is possible that melatonin influences Per2 expression by reducing cAMP levels through G-proteins (Cassone et al., 2009). As an output of the pineal oscillator, melatonin may thus act negatively on hypothalamic cells. The mechanism by which the hypothalamic clock regulates the circadian rhythms in the pineal is not fully understood but may be mediated by sympathetic norepinephrine. Indeed, this hormone is released in a circadian manner by the SCN and controls the rhythmic production of melatonin through the inhibition of AA-NAT (Cassone et al., 1990) as well as the expression of some clock genes (Karaganis et al., 2008). These observations are thus consistent with the neuroendocrine loop model proposed by Cassone and Menaker (1984), postulating the mutual inhibition of the 2 oscillators. We should stress, however, that there is no evidence of a direct effect of melatonin (or norepinephrine) on SCN (or pineal) clock gene expression; the coupling mechanisms likely involve intermediary signaling components.
Based on these experimental observations, we propose here, for the first time, a molecular model for the avian circadian system. Numerous models, with various degrees of details, have been developed for the circadian clock in Drosophila, Neurospora, plants, and mammals. They all rely on a central molecular network describing the generation of a self-sustained rhythm (limit-cycle oscillations) in a single cell (Leloup et al., 1999; Leloup and Goldbeter, 2003; Forger and Peskin, 2003). In the model proposed here for birds, the self-sustained rhythm results from mutual interorgan coupling. We describe properties of such a system and their physiological consequences. Some experimentally testable predictions are formulated. At this stage, the model remains simple and qualitative, but directions for future improvements are indicated.
Model
The model that we propose for the circadian clock of birds is based on mutual coupling between the hypothalamic and pineal oscillators. The scheme of the model is depicted in Figure 2. Each organ is treated as a single oscillator (we implicitly assume that intercellular coupling mechanisms ensure the intercellular synchronization in a given organ).
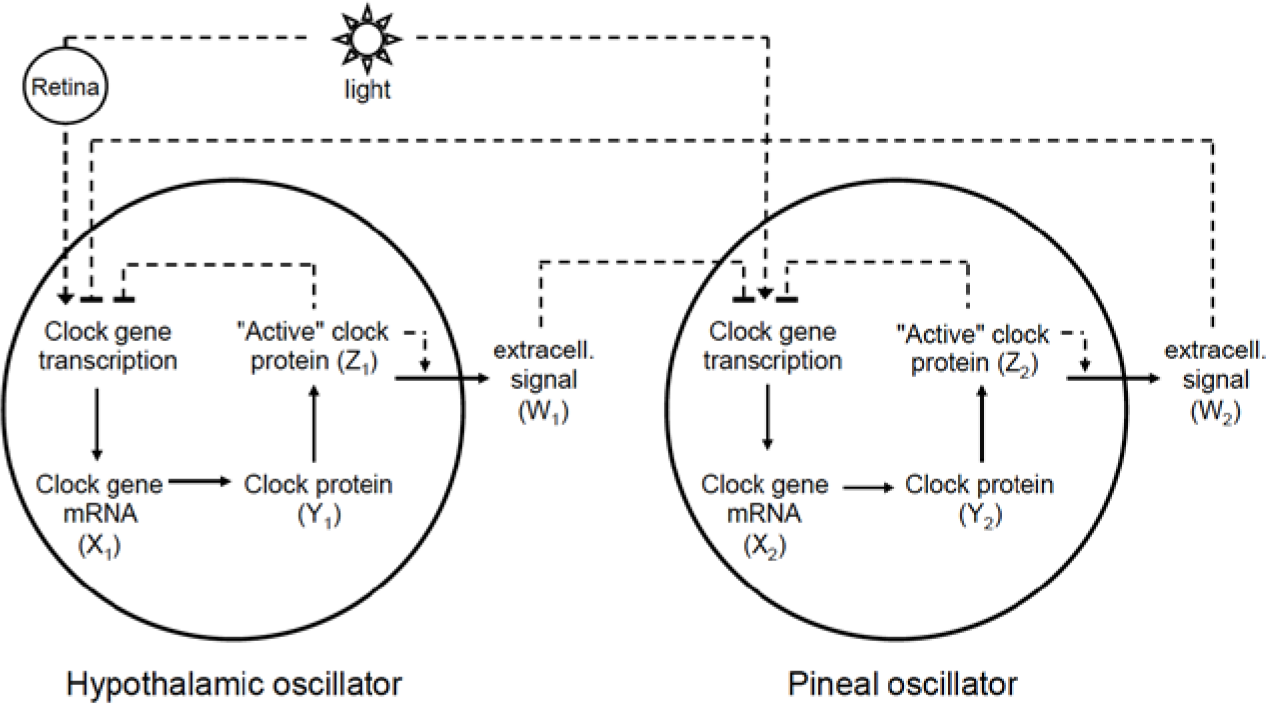
Scheme of the model. We modeled the core avian circadian clock with 2 coupled Goodwin-like oscillators. Each oscillator is based on a negative feedback loop mechanism (X
Hereafter, oscillator “1” will refer to the hypothalamic oscillator, and the corresponding subscript “1” will refer to the variables of that oscillator. Oscillator “2” and subscript “2” will refer to the pineal oscillator.
Each oscillator is modeled by a 3-variable Goodwin-like oscillator (Goodwin, 1965; modified by Gonze et al., 2005). The Goodwin model is a minimal 3-component oscillator, reflecting the core negative feedback of the circadian clock (Ruoff et al., 1999). Variable X can be interpreted as the concentration of the clock gene mRNA (e.g., Per2), Y as the concentration of the corresponding clock protein, and Z as the concentration of the “active” (e.g., phosphorylated) clock protein, acting as a transcriptional inhibitor.
For each oscillator, we also added a clock-controlled variable, W. This variable is used for coupling. For the pineal gland, we will interpret W2 as melatonin. For the hypothalamic oscillator, a possible candidate for W1 is norepinephrine. In each organ, the production of the coupling compound is induced by the “active” clock protein. The time evolution of the variables of our model is governed by the following differential equations:
Parameters σ1 and σ2 are time-scaling factors that allow us to change and adjust the period (and only the period) of each oscillator. Michaelian degradation is chosen to favor the oscillations for a moderate value of the Hill coefficient, n (Gonze et al., 2005).
Consistently, with the neuroendocrine loop mechanism (Cassone and Menaker, 1984), we hypothesize that the coupling variables W1 and W2 act as inhibitors for the other oscillator. The coupling functions are then defined by
We will denote this mutual negative coupling by (–,–). The case of mutual activation, designated as (+,+), can be described by the following coupling functions:
Light acts on the core clock via at least 3 different pathways. It is perceived by the retina, which, in turn, projects into the hypothalamus. There is also a retina-independent light pathway affecting the hypothalamic oscillator, involving encephalic photoreceptors (Foster and Soni, 1998). Finally, a direct input of the light into the pineal, via pinopsin photoreceptors, was also identified (Okano et al., 1994). Since the molecular nature and the possible difference in these input pathways are not yet elucidated, we will assume, for simplicity, that light acts in the same way on both oscillators. Thus, in the following simulations, we will take L = L1 = L2. By analogy, with the situation in mammals, we will assume that light induces the transcription of the core clock gene. Light-dark cycles are modeled by the square wave variation of L.
Numerical simulations have been performed with Matlab (MathWorks, Natick, MA, USA) and checked with XPP-AUTO (Pittsburgh, PA, USA; Ermentrout, 2002).
Results
Coupling-Induced Limit-Cycle Oscillations
The hypothalamic and pineal oscillators are each modeled by the same Goodwin-like model. This 3-variable oscillator is based on a delayed negative feedback loop. Since the pioneer work by Goodwin (1965), numerous studies have already shown that such a system can produce self-sustained oscillations. Here, we have selected parameter values so that 1) the coupled system displays limit-cycle oscillations with a circadian period (Fig. 3A and 3C) and 2) in the absence of coupling, the oscillations are damped (Fig. 3B and 3D). The degree of damping is in agreement with the observation of Robertson and Takahashi (1988), who measured melatonin released by dispersed chick pineal cells in DD.
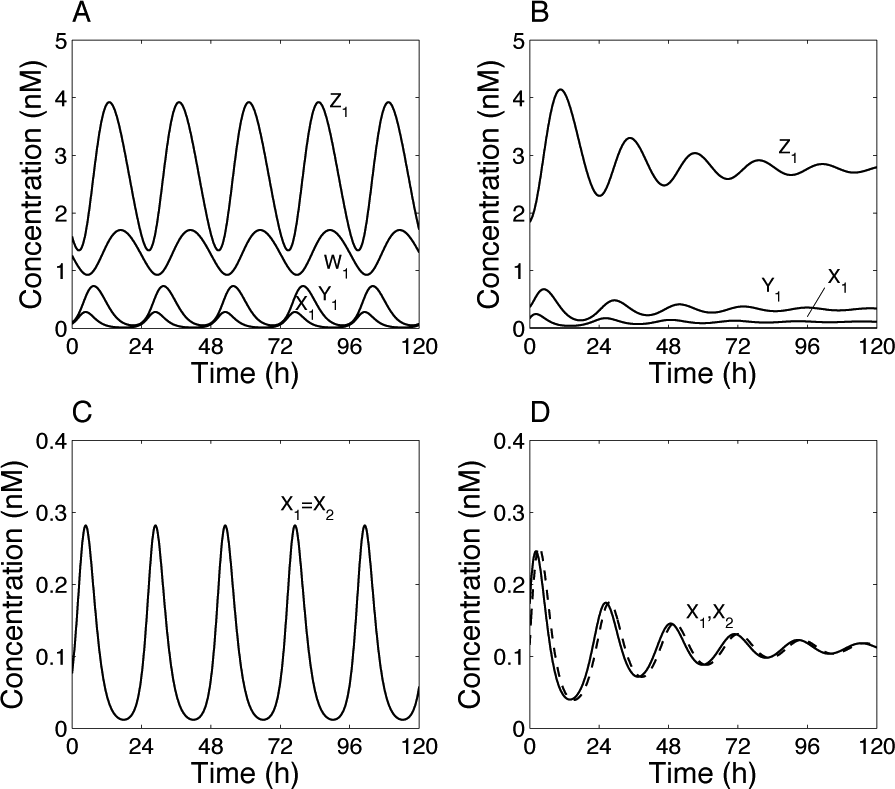
Self-sustained oscillations induced by coupling between the pineal and hypothalamic oscillators. The results have been obtained by numerical integration of equations 1 to 8 with the coupling functions of equations 9 and 10. (A, C) Coupled system. (B, D) Uncoupled system (F12 = F21 = 1). Parameter values: σ1 = σ2 = 1, Kc = 1 nM, m = 3, v1 = 1 nM/h, K1 = 1 nM, n = 3, v2 = 0.45 nM/h, K2 = 1 nM, k3 = 1 h–1, v4 = 0.45 nM/h, K4 = 1 nM, k5 = 1 h–1, v6 = 0.45 nM/h, K6 = 1 nM, k7 = 0.1 h–1, and v8 = 0.2 nM/h. Limit-cycle oscillations (A, C) are independent of the initial conditions. Initial conditions (B, D) correspond to a point on the limit cycle: X1(0) = 0.17, Y1(0) = 0.37, and Z1(0) = 1.84 (a slightly different initial point is taken for the second oscillator to distinguish the 2 curves in panel D). Concentrations are tentatively expressed in nM. The period for the coupled system is 24.14 h, while the “period” for the uncoupled (damped) oscillator is 22 h.
Thus, mutual coupling of 2 damped oscillators can induce self-sustained (limit-cycle) oscillations. Oscillations are synchronized in phase (Fig. 3C). Parameter values have been adjusted so that the period of the oscillations is about 24 h. For the same parameter values, but in the absence of coupling, the period of the damped oscillations is slightly shorter, about 22 h. A manual screening of parameter values showed that obtaining limit-cycle oscillations from coupling of the damped oscillators is not too difficult, but this is often accompanied by a shift in the period. In other words, the period of the coupled system is often not close to the average of the period of individual oscillators and may even be very different. This shift in the period was already reported when synchronizing a population of Goodwin-like oscillators (Gonze et al., 2005).
These coupling-induced limit-cycle oscillations illustrate the “internal resonance” mechanism suggested by Gwinner and Brandstätter (2001), based on the neuroendocrine loop mechanism (Cassone and Menaker, 1984). Our model provides the first implementation of such a mechanism using a simple molecular model for circadian rhythms. Hereafter, we describe the dynamic properties of this model and relate them to available experimental data.
Robustness to Asymmetry between the 2 Oscillators
In the above simulations, identical parameter values were chosen for the 2 oscillators. The system is thus completely symmetrical. In reality, the 2 systems are not expected to be perfectly identical. To assess the robustness of their behavior and to ensure that in-phase synchronization is not an artifact due to the symmetry of the model, we examined the effect of asymmetry in the 2 oscillators. We introduce a difference in the period of the 2 oscillators by changing the time-scaling parameter σ2 of “oscillator 2.”
Figure 4A shows how the period of the coupled system changes as a function of σ2. We can first verify that for σ1 = σ2 = 1, we have a 24-h period. A small difference in the period of the 2 oscillators may increase or decrease the period of the coupled system, but as long as the difference is not too large, the period remains circadian. We can also observe that if σ2 is too large or too small, oscillations in the coupled system are lost. Limit-cycle oscillations occur for 0.8 < σ2 < 1.25. Out of this range, oscillations are damped. To maintain a self-sustained rhythm, the period of 2 oscillators should not differ too much. Similar results are obtained for small asymmetry in the kinetic parameters (not shown).
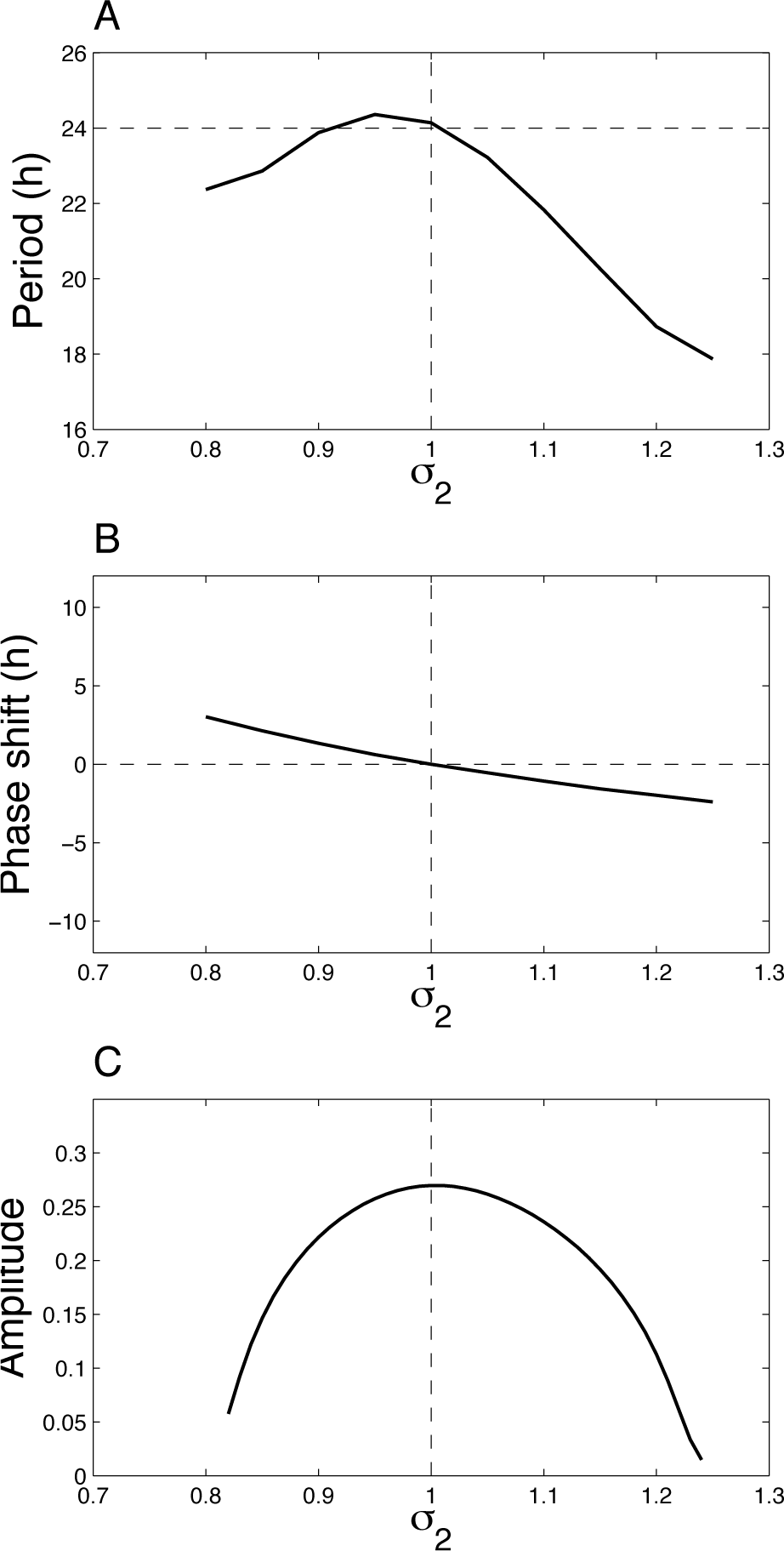
Effect of asymmetry in the 2 oscillators on the dynamics of the system. (A) Period, (B) phase relationship, and (C) amplitude of the oscillations (defined as the difference between the maximum and the minimum of X1) as a function of σ2. Note that limit-cycle oscillations occur for 0.8 < σ2 < 1.25. Out of this range, oscillations are damped, and the system converges to a steady state. Parameter values are as in Figure 3.
Figure 4B presents the phase shift between the 2 oscillators as a function of σ2. The asymmetry in the 2 oscillators does not yield large phase shifts; the 2 oscillators remain synchronized almost in phase. Figure 4C shows that the amplitude of the oscillations is maximal for σ1 = σ2 = 1 and decreases when the oscillators are asymmetric. This bell curve is reminiscent of other coupled oscillatory systems, where internal resonance is also associated with amplification of the oscillations (Nayfeh and Mook, 1979). This behavior is observed for any small asymmetry in the kinetic parameter values of the 2 oscillators.
The system is thus robust towards small variations and asymmetry in kinetic parameters. The model, however, predicts that mutations that strongly affect the period of 1 of the 2 oscillators would rather lead to a loss of oscillations than to complex dynamics.
Importance of the Type of Coupling for the Mode of Synchronization
In the previous simulations, we considered negative mutual coupling in which the output variable (W1) of each oscillator acts negatively on the other oscillator. In biological terms, it means that melatonin produced by the pineal will act negatively (most likely via a signal transduction pathway) on clock gene expression in the hypothalamus. Conversely, the sympathetic signal coming from the hypothalamus will inhibit clock gene expression in the pineal gland. This (–,–) coupling is described by the coupling functions in equations 9 and 10 and leads to in-phase synchronization.
The case of mutual positive coupling, denoted as (+,+), can be simulated by replacing the inhibitory functions in equations 9 and 10 by the activatory functions in equations 11 and 12. In this case, we observe antiphase synchronization (Suppl. Fig. S1A). As done for (–,–) coupling, this behavior was found to be robust towards small asymmetry in the 2 systems. If we now consider asymmetric coupling, of the (–,+) kind, using F12 defined by equation 9 and F21 defined by equation 12, we obtain out-of-phase synchronization, which is also characterized by asymmetry in the amplitude of the 2 oscillators (Suppl. Fig. S1B).
Thus, the nature of coupling determines the mode of synchronization (a similar situation was described in a model of coupled metabolic oscillators [see Gonze et al., 2008]). As explained in the introduction, there is currently no clear evidence to favor one type of synchronization in birds, but some data suggest that oscillations in clock gene expression are rather in phase. In the following sections, we only discuss the case of mutual inhibition (and the associated in-phase synchronization).
Effect of Exogenous Administration of Melatonin
Many experiments have been devoted to the study of the effect of constant and periodic administration of melatonin in pinealectomized birds. We model pinealectomy by setting X2 = Y2 = Z2 = 0, and the level of melatonin is then controlled by the variable W2, which is described by an arbitrary function reflecting the exogenous level of melatonin. We examine here the effect of constant and periodic melatonin treatments on the hypothalamic oscillator.
We have already seen that in the absence of melatonin, the oscillations are damped (Fig. 3). In Supplementary Figure S2, we see that a nonnull constant level of melatonin accelerates damping of the oscillations. At very high melatonin concentrations (W2 = 3), there are no more oscillations; all variables rapidly stabilize at constant values. We can also notice that, due to the inhibitory effect of melatonin (W2) on gene expression, the steady state of X1 becomes lower as the level of melatonin increases. We may thus anticipate that the locomotor activity (presumably activated by the clock protein) will also be attenuated. This is consistent with the experimental data (Gwinner and Benzinger, 1978).
The effect of a periodic administration of exogenous melatonin is illustrated in Figure 5. We model such a treatment by a square wave variation of W2. In Figure 5A, the periodic administration of melatonin is close to the variation observed in endogenous conditions, that is, when the 2 oscillators are coupled. In that case, the hypothalamic oscillations can readily be entrained at 24 h and are very similar to the ones obtained in the coupled system (Fig. 3C). When the amplitude of melatonin’s periodic forcing is increased (Fig. 5B), oscillations become irregular in their amplitude (they undergo period-doubling bifurcation) but remain phase locked.
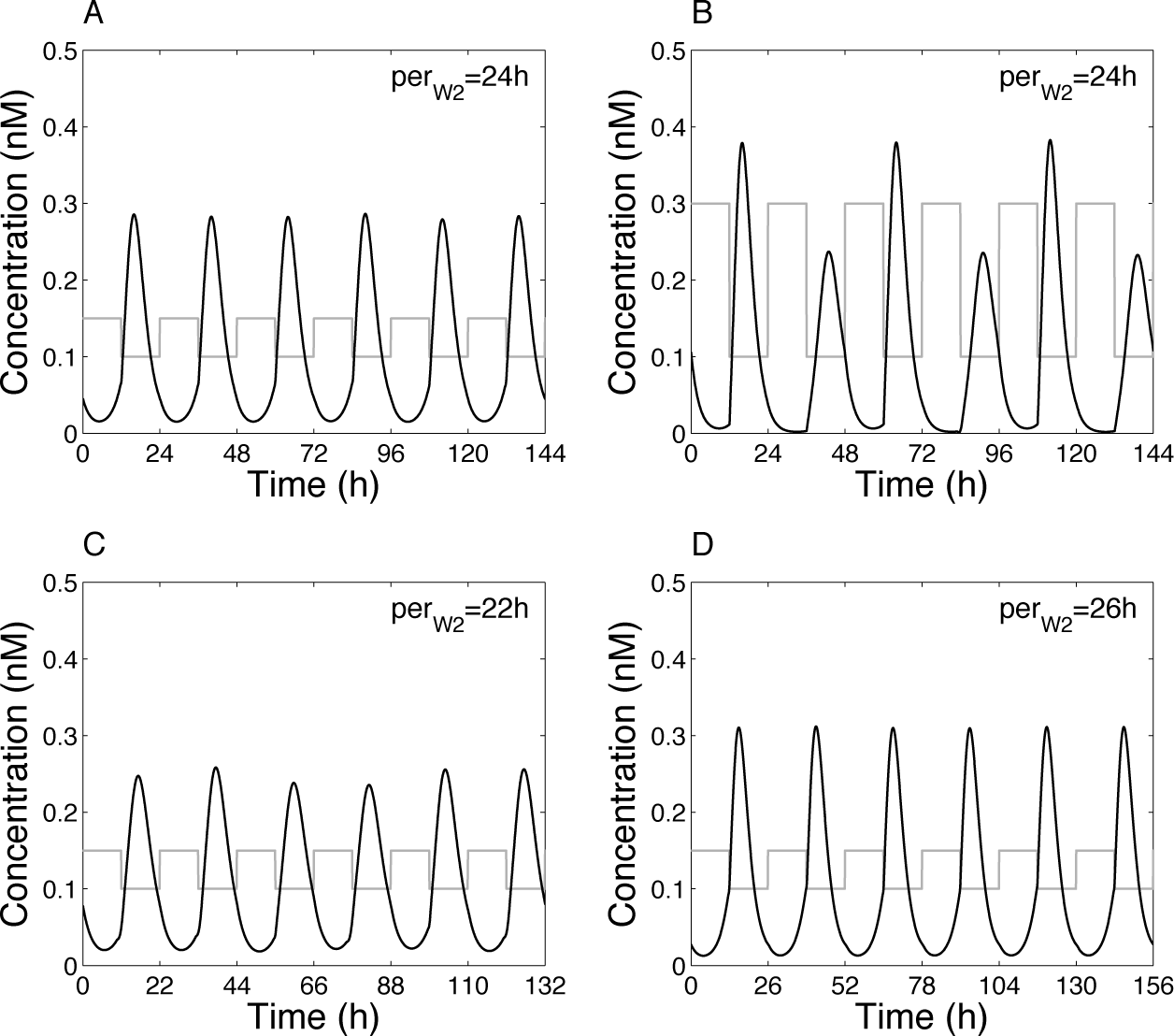
Effect of various periodic melatonin treatments on the hypothalamic oscillator. (A) W2 varies between 1 and 1.5 nM (i.e., close to the values of W2 when the 2 oscillators are coupled), with a period of 24 h. (B) W2 varies between 1 and 3, with a period of 24 h. (C) W2 varies between 1 and 1.5, with a period of 22 h. (D) W2 varies between 1 and 1.5, with a period of 26 h. In each panel, W2 is indicated by the gray curve. Note that, for readability, the variable W2 has been divided by a factor of 10. Parameter values are as in Figure 3.
Here again, due to the inhibitory effect of melatonin on clock gene expression, we observe that X1 (and Y1, not shown) increases and reaches its maximum level when melatonin is low. There is thus a delay between melatonin’s availability and the high level of X1 and Y1, consistent with the recording of the locomotor activity in pinealectomized birds subject to the periodic administration of exogenous melatonin (Gwinner and Benzinger, 1978).
Heigl and Gwinner (1995) have reported that for very short or very long periods of administration of melatonin, behavior becomes arrhythmic. In our model, when the forcing period is much smaller (22 h) (Fig. 5C) or larger (26 h) (Fig. 5D) than 24 h, oscillations remain entrained at the forcing period and, despite small fluctuations in the amplitude, still remain phase locked. This does not provide a satisfactory explanation for the complete loss of rhythmicity observed in the experiments. Factors unrelated to the core oscillatory mechanism must therefore be responsible for this arrhythmicity.
Effect of Light: Entrainment by LD Cycles and Resynchronization after Jet Lag
Light-dark cycles are modeled by square wave forcing of the parameter L = L1 = L2. This periodic function is charaterized by its period (by default per = 24), amplitude (by default amp = 0.5), and photoperiod, defined as the duration of the light phase divided by the day length (by default php = 0.5). With these default parameter values, both coupled and decoupled oscillators are entrained by an LD cycle of 24 h (Fig. 6A and 6B). The amplitude and phase are, however, different in the 2 cases. These results suggest that the pineal amplifies the oscillations and controls the entrainment phase of the hypothalamic oscillator (and vice versa).
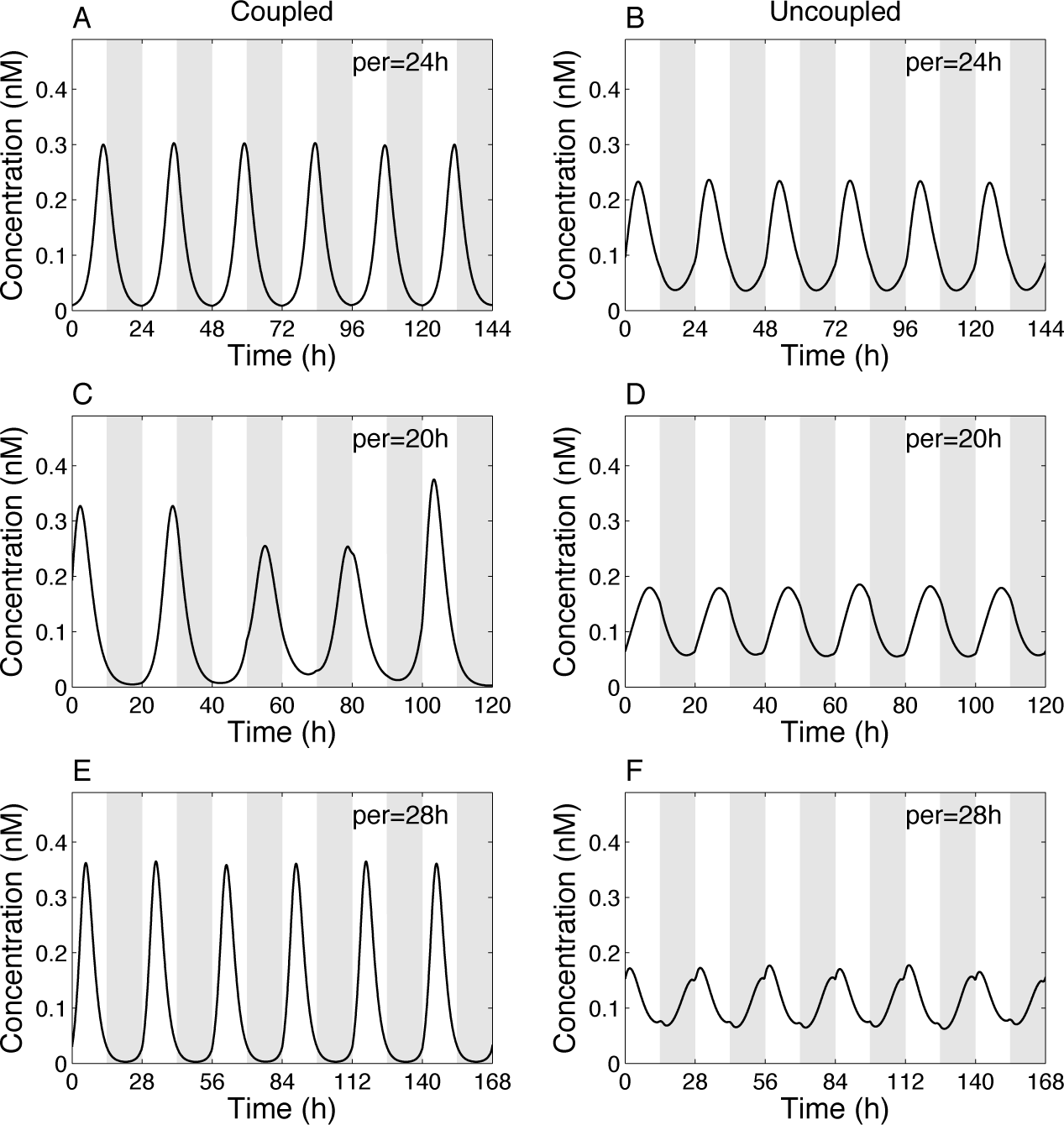
Entrainment by an LD cycle. (A, C, E) Coupled system. (B, D, F) Uncoupled system (F12 = F21 = 1). (A, B) Period of the LD cycle (per) = 24 h. (C, D) per = 20 h. (E, F) per = 28 h. Other parameters for forcing are amp = 0.5 and php = 0.5. Shown is the variable X1. Gray areas denote the dark phases. Parameter values for the oscillators are as in Figure 3.
When the period is reduced to per = 20 h, oscillations in the coupled system are impaired (and not phase locked), while they remain properly entrained in the uncoupled system (Fig. 6C and 6D). When the period is increased to per = 28 h, both coupled and uncoupled oscillators are entrained to 28 h (Fig. 6E and 6F). We can also notice that the amplitude of the forced damped oscillator is reduced compared to the coupled system.
Figure 7A shows the entrainment range of the coupled (solid lines) and uncoupled (dashed lines) systems as a function of the period of the LD cycle for various amplitudes of light (amp). The uncoupled (damped) oscillator is entrained over the whole range of periods tested, whereas the coupled system is entrained only over a limited range of periods, roughly centered around 24 h. Out of the entrainment range, oscillations are not phase locked and are often characterized by a progressive shift of the phase. Figure 7B gives the amplitude of X1 oscillations as a function of the period of the LD cycle. The amplitude of the coupled system is significantly larger than that for the uncoupled oscillator. In summary, the coupled system is entrained over a bounded range of the LD period, but the resulting oscillations have larger amplitudes.
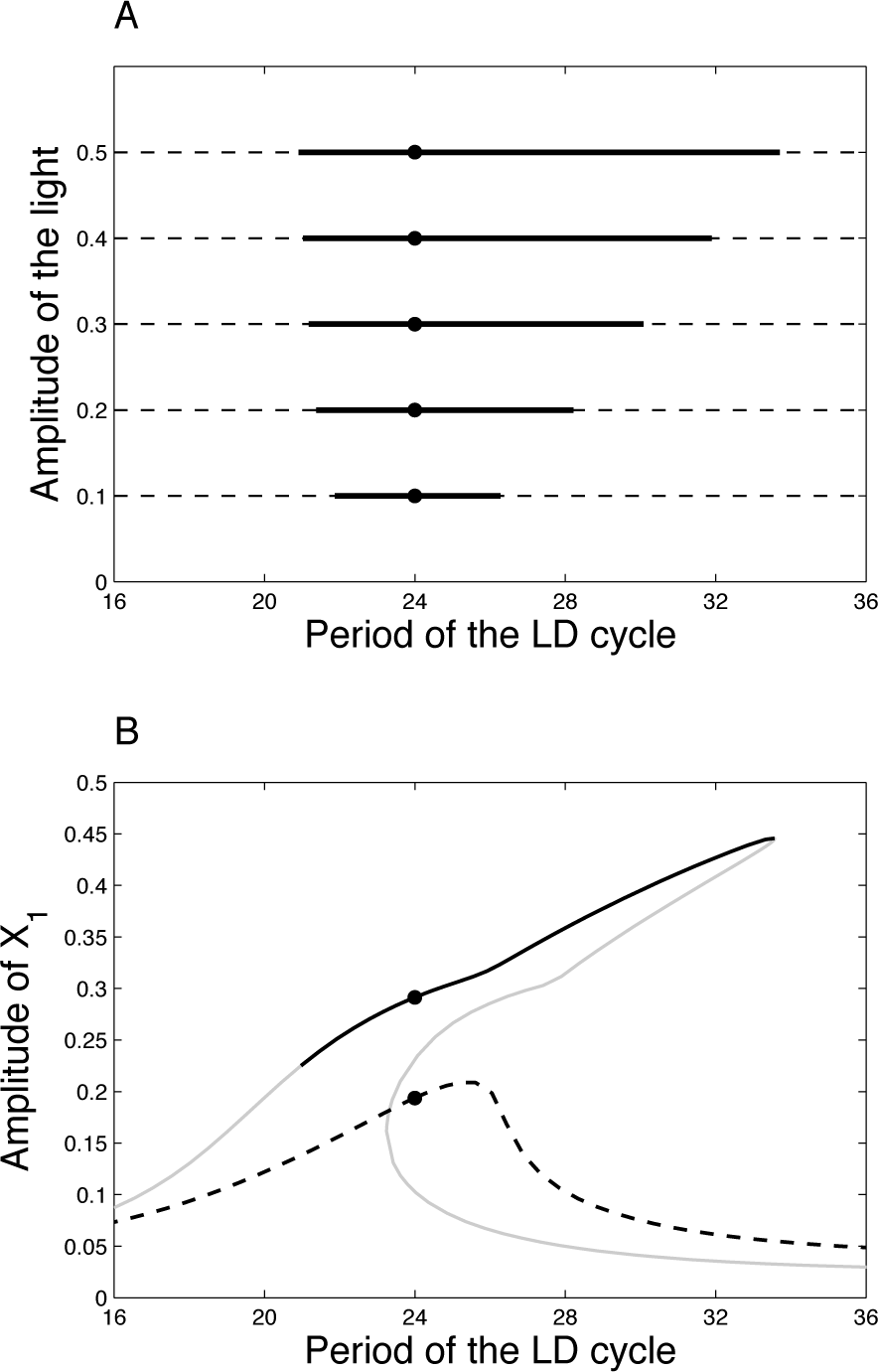
(A) Range of entrainment as a function of the period of the LD cycle for various amplitudes of light. (B) Amplitude of X1 as a function of the period of the LD cycle for amp = 0.5. The gray parts of the curve denote an unstable limit cycle. As a matter of fact, the entrainment region is delimited on the right by a limit point and on the left by torus bifurcation (Nayfeh and Balachandran, 2004). These technical considerations are mathematically investigated in another paper (Woller et al., in preparation) and rely on bifurcation theory. Parameter values for the oscillators are as in Figure 3. In both panels, the solid lines refer to the coupled system, whereas the dashed lines refer to the uncoupled system.
The primary function attributed to circadian rhythms is to facilitate adaptation of the living organisms to the periodic environment. The circadian clock achieves this goal presumably by setting the phase of expression of clock genes and clock-controlled genes with respect to the LD cycle. A key feature is thus the phase of the circadian oscillations, which must be properly controlled and adjusted with regard to the photoperiod. In Figure 8, we report the phase of the entrained system at different photoperiods and compare it to the uncoupled system. Remarkably, the 2 systems display very different patterns. While the coupled system is locked to the LD transition, the uncoupled system seems rather locked to the middle of the day. This difference in the entrainment phase appears also clearly as a function of the forcing period (Suppl. Fig. S3). This supports the idea that coupling between the 2 oscillators allows a proper control of the entrainment phase.
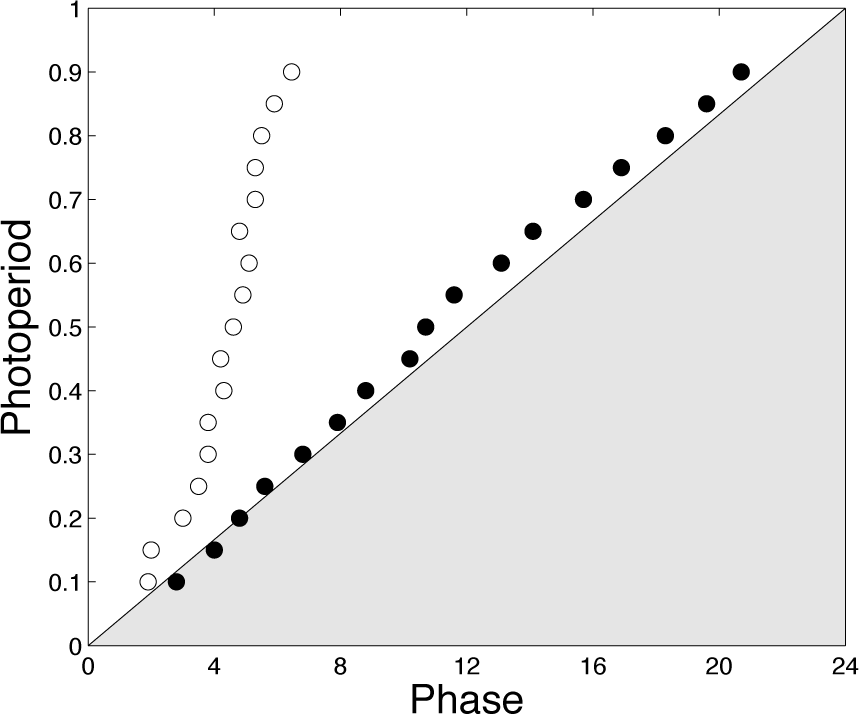
Effect of the photoperiod. The dots correspond to the phase of the maximum of X1. Black dots: Coupled system. White dots: Uncoupled system. The period and amplitude of the LD cycle are per = 24 h and amp = 0.5, respectively. Parameter values for the oscillators are as in Figure 3.
Another related function of the circadian clock is to readjust and coordinate the physiological rhythms after jet lag. The effect of jet lag in our model is illustrated in Supplementary Figure S4: At 72 h, the LD cycle is either delayed by 8 h (Suppl. Fig. S4A and S4B) or advanced by 8 h (Suppl. Fig. S4C and S4D). In both cases, the coupled system takes more time to resynchronize than the uncoupled oscillator. This readjustment of the phase is shown in Figure 9. The uncoupled system recovers the phase faster than the coupled system, usually in a monotonous fashion. Another interesting difference worth underlying is the case illustrated in Figure 9D. In some conditions, the uncoupled oscillator resynchronizes in the same direction as the LD phase shift (orthodromic re-entrainment), whereas the coupled oscillators resynchronize in the opposite direction (antidromic re-entrainment). This faster resynchronization in pinealectomized birds was reported in experimental studies by Hau and Gwinner (1994) and, more recently, by Kumar and Gwinner (2005).
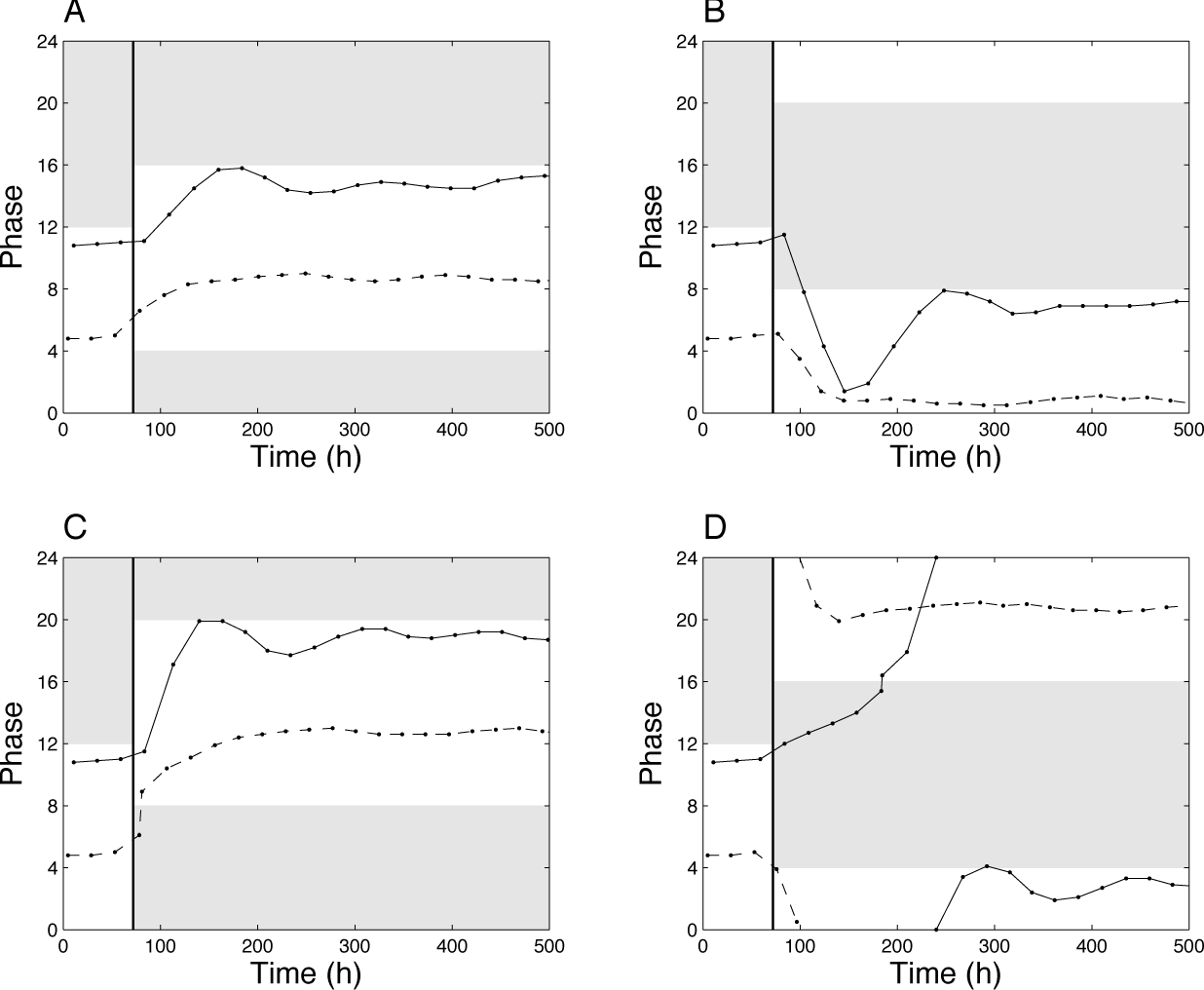
Resynchronization after jet lag. Phase of the maximum of X1 with respect to the LD cycle after jet lag. At 72 h, the LD cycle is delayed by 4 h (A), advanced by 4 h (B), delayed by 8 h (C), or advanced by 8 h (D). Solid lines: Coupled system. Dashed lines: Uncoupled system. Parameter values are as in Figure 3.
Discussion
The molecular mechanism of circadian clocks has been modeled in many organisms from cyanobacteria to fungi, insects, plants, and mammals. Despite an increasing amount of data on the circadian rhythms in birds, no biophysical model has so far been proposed for this broad class of animals. Although sharing many genetic components with the mammalian circadian clock, the bird circadian clock organization appears more complex. The mammalian pacemaker is centralized to the SCN, whereas in birds, the circadian rhythm results from mutual coupling between the pineal gland, hypothalamic, and possibly retinal oscillators (Gwinner and Brandstätter, 2001). We have examined here a molecular model for the avian circadian clock based on this mechanism. Our model is undoubtedly oversimplified with respect to the real bird clock, but it recapitulates the fundamental experimental observations and offers a basis for future extensions.
The pineal and hypothalamic organs are each viewed as a single oscillator and described by a Goodwin-like model. In the presence of mutual coupling, they generate self-sustained oscillations of a limit-cycle type. Depending on the nature of coupling, oscillations are synchronized in phase or in antiphase. Historically, 2 different models have been proposed to explain the circadian system of birds. The first one is the neuroendocrine loop model (Cassone and Menaker, 1984). In this model, robust rhythms arise from mutual inhibition between the pineal and hypothalamic oscillators, while in the second one, called the internal resonance model (Gwinner, 1989), coupling amplifies the oscillators’ amplitude (resonance). Our model, based on mutual amplification via inhibitory interactions, thus incorporates features of both models. When coupling is removed, as occurs when the organ is isolated, oscillations are damped and progressively lost.
Coupling-induced limit-cycle oscillations in a large population of cells have already been investigated as a possible mechanism for the generation of robust and synchronized rhythms in the mammalian SCN (Gonze et al., 2005; Bernard et al., 2007; Webb et al., 2012). The idea that circadian oscillators may be damped was already suggested many years ago (Lewis and Saunders, 1987), and phenomenological models based on mutual coupling between 2 oscillators were already proposed (Strogatz, 1987; Kronauer et al., 1982). Motivated by the avian circadian system, Wu et al. (2000) reported on an abstract mathematical model based on coupled damped oscillators. They found a shift in the period as a function of coupling strength and predicted asymmetry between the 2 oscillators, with the pineal oscillator being weaker than the SCN as well as a 180° phase relationship between the 2 oscillators. While period shift due to coupling is common (Pittendrigh and Daan, 1976; Strogatz, 1987; Gonze et al., 2005), the later predictions of Wu et al. (2000) are not confirmed by our model.
The origin of damping observed at the level of the organs in birds is not clear. The lack of single cell measurements does not allow us to discriminate between the case where each cell displays damped oscillations and the situation where damping results from desynchronization of rhythmic cells. In mammals, there is experimental evidence showing that damping observed in peripheral tissues (Yamazaki et al., 2000) is rather due to desynchronization of the cells, and the function of the SCN is to ensure intercellular coupling and thereby synchronization in peripheral organs (Yoo et al., 2004).
Numerical simulations of our bird circadian model allowed us to investigate the effect of constant and periodic administration of melatonin in pinealectomized birds. Consistently with experimental observations (Heigl and Gwinner, 1995), our results demonstrate that the circadian rhythm in the hypothalamus can be entrained over a certain range of period of exogenous melatonin supply. Out of the entrainment range, when the period of the melatonin rhythms is much higher or much lower than 24 h, oscillations may become irregular in their amplitude, but the oscillations remain phase locked. This does not explain the arrhythmicity observed in the experiments, suggesting that other factors may be responsible for the loss of rhythmicity in these conditions.
Since the primary function of the circadian clock is to synchronize the physiological and behavioral rhythms to the day-night cycles, we study the effect of LD cycles on the bird circadian oscillator. Comparison of the single (damped) oscillator and coupled oscillator models showed that the advantage of the coupled model is enhancing the amplitude of the rhythm at the cost of reducing the range of periods in which entrainment is observed. These results are in agreement with the findings of Abraham et al. (2010), who demonstrated that coupling among circadian clocks results in increased amplitude and rigidity. The coupled and uncoupled models also display different phase locking patterns as a function of the photoperiod. Since the phase of the circadian clock is crucial for adaptation, it is possible that the 2-oscillator structure of the avian circadian clock was selected to ensure proper control of this entrainment phase. It should be noted, however, that other mechanisms, including complex intracellular molecular regulations (Troein et al., 2011) or gating of the light input by the clock (Geier et al., 2005), may also be used to set the entrainment phase.
Finally, the coupled oscillator system was shown to resynchronize slower than the single damped oscillator, with the possibility of an antidromic pattern. Slower resynchronization may be beneficial for birds, as it will allow time for metabolism or the immune system to adapt to the shifted day-light cycle. It should be stressed here that the resynchronization time of oscillators may also be affected by other factors such as the radial relaxation speed of the oscillator (Granada and Herzel, 2009) or the autonomous period or biochemical kinetic parameters of the oscillator (Leloup and Goldbeter, 2013).
The model presented here is a generic model for the avian circadian clock. Interactions between the circadian oscillators and their relative contributions are species specific. For example, pinealectomy has a greater effect on the locomotor rhythm in passerine birds such as the house sparrow than in galliform species such as the chicken and quail (Underwood et al., 2001; Bell-Pedersen et al., 2005). In the latter 2 species, the eyes have been demonstrated to play a greater role, as ocular enucleation, but not pinealectomy, abolishes activity rhythms in these animals (Underwood, 1994). In migratory birds (warblers) as well as in arctic birds (penguins), the level of melatonin was shown to be low (Gwinner and Brandstätter, 2001). The complex and species-dependent structure of the avian circadian clock would provide the birds with increased adaptive properties.
At this stage, our avian circadian model remains simple and qualitative, but it provides a basis for future modeling efforts. Directions for improvements include 1) the extension of the core oscillator mechanism to account for the multiple genes and feedback loops expected to operate at the cellular level; 2) the refinement of light input pathways, including the incorporation of an oscillator model for the retina; 3) the development of a multicellular model accounting for cell-cell coupling and synchronization in each organ; and 4) the study of bird-specific properties. These theoretical models will be developed in parallel with further experimental studies and will hopefully provide a complementary view on the bird circadian system.
Footnotes
Acknowledgements
The authors thank T. Erneux and G. Dupont for fruitful discussions and the anonymous reviewers for their valuable recommendations.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.