Abstract
Delayed sleep phase disorder (DSPD) is a circadian rhythm sleep disorder. Patients with DSPD have problems initiating sleep if they go to bed at a conventional time, and they often have problems waking at desired times. If they rise early in the morning, they usually experience severe sleepiness during morning hours. In the present study, we investigated the short- and long-term effects on measures of subjective and objective sleepiness and cognitive function of bright light and melatonin treatment alongside gradually advanced rise times in adolescents and young adults. Four treatment conditions were used in the short-term intervention (2 weeks): dim light (placebo) + placebo capsule, bright light + placebo capsule, dim light (placebo) + melatonin capsule, and bright light + melatonin capsule. This was followed by a long-term intervention (3 months) including 2 conditions: no treatment and combined bright light + melatonin treatment. Effects of treatment on sleepiness and fatigue were the primary outcome measures, and effects on cognitive function were secondary outcome measures. On a gradual advancement of the rise time schedule, all treatment conditions (bright light, melatonin, combination, and placebo) were almost equally effective in improving subjective daytime sleepiness, fatigue, and cognitive function in the 2-week study. The 2-week intervention showed no effect on objective sleepiness. Long-term treatment increased some of the positive effects seen after 2 weeks. The combined bright light and melatonin treatment improved subjective daytime sleepiness, fatigue, and cognitive function in the 3-month study. The no-treatment group returned to baseline values on most variables. In conclusion, a gradual advancement of rise times seems to produce positive effects on subjective sleepiness, fatigue, and cognitive performance during short-term treatment of patients with DSPD. However, the benefits from gradually advanced rise times seem to wear off, suggesting that the continuation of bright light and melatonin treatment is beneficial to maintain positive effects over time.
Keywords
Delayed sleep phase disorder (DSPD) is a circadian rhythm sleep disorder in which the sleep period is delayed with respect to conventional norms (American Academy of Sleep Medicine, 2005). Patients with DSPD have been described as unable to advance their sleep phase by behavioral means (American Academy of Sleep Medicine, 2005), and the disorder is associated with impairments related to school or work functioning, alcohol and substance abuse, and psychological and relational disturbances (American Academy of Sleep Medicine, 2005; Dagan and Eisenstein, 1999; Regestein and Monk, 1995; Saxvig et al., 2012). Hence, DSPD is a disorder with several psychosocial challenges (Wilhelmsen-Langeland et al., 2012). The prevalence of DSPD is estimated to be 7% to 16% in adolescence (American Academy of Sleep Medicine, 2005; Gradisar et al., 2011b).
DSPD does not involve a dysfunction of the sleep period itself but simply a delay of the circadian rhythm (Thorpy et al., 1988; Weitzman et al., 1981). Thus, patients with DSPD have problems initiating sleep if they go to bed at a conventional time, and they accordingly experience severe sleepiness during morning hours (American Academy of Sleep Medicine, 2005; Thorpy et al., 1988; Weitzman et al., 1981). Our research group has previously shown that the sleep architecture itself is not different from healthy controls when patients with DSPD are allowed to sleep at self-chosen times (Saxvig et al., 2013b). Because the sleep period is delayed in patients with DSPD, they have more slow-wave sleep between 0600 h and 0800 h than controls (Saxvig et al., 2013b). Sleep curtailment typically leads the patients to catch up on sleep when they can, that is, by napping and by extending sleep on weekends and holidays, which only maintain and potentially increase the sleep phase delay the following nights (Crowley et al., 2007).
The effect of sleep deprivation is known to negatively affect cognitive function, but little is known about how these functions are influenced in patients with DSPD. Shekleton and colleagues (2010) have reviewed studies on neurobehavioral performance in patients with insomnia. The most consistent impairment was shown on tests measuring different aspects of attention and executive function, and their results called for the inclusion of standard psychometric tests in future studies of sleep disorders (Shekleton et al., 2010).
A successful advancement of the sleep period and circadian rhythm is expected to reduce or eliminate negative consequences such as daytime sleepiness in patients with DSPD (Gradisar et al., 2011a). Timed bright light exposure and melatonin administration seem to be the most widely used treatment options for DSPD in clinical practice (Bjorvatn and Pallesen, 2009). The timing of light is crucial (Bjorvatn and Pallesen, 2009), as light before the minimum core body temperature (CTmin) delays the circadian rhythm, whereas light after CTmin advances the circadian rhythm (Khalsa et al., 2003). Bright light after awakening has proven to be effective in advancing the sleep phase in patients with DSPD (Lack et al., 2007; Rosenthal et al., 1990). Rosenthal and colleagues (1990) conducted the only study that we know of on patients with DSPD, assessing the effects of bright light treatment on objective sleepiness (sleep propensity) by the Multiple Sleep Latency Test (MSLT) (Carskadon et al., 1986). Their bright light group (2500 lux) showed increased sleep latency at 0900 h and 1100 h (but not at other times during the day) compared to the dim light group (300 lux). Cole and colleagues (2002) found reduced subjective morning sleepiness in a bright light group (2700-lux masks) compared to a dim light group (<0.01-lux masks), but only in the most delayed subjects. Recently, cognitive behavioral therapy in combination with bright light therapy showed promising effects on advancement of the sleep period, subjective sleepiness, and fatigue in adolescents with DSPD (Gradisar et al., 2011a).
Similar to the effects of light, the effects of exogenous melatonin administration also follow a phase-response curve (Lewy et al., 1998). Previous studies have shown that the sleep period may be advanced by the use of melatonin in the evening, usually taken a few hours before expected sleep onset (Dahlitz et al., 1991; Kayumov et al., 2001; Mundey et al., 2005; Nagtegaal et al., 1998; Rahman et al., 2010). Only 2 of these studies included measures of subjective daytime sleepiness/alertness. Dahlitz and colleagues (1991) found no changes on self-reported alertness following melatonin treatment. Similarly, Kayumov and colleagues (2001) found no overall difference on measures of subjective sleepiness, fatigue, and alertness between a melatonin group and a placebo group. However, when adhering to an imposed sleep schedule (midnight to 0800 h), subjects taking melatonin were less sleepy and fatigued than the placebo group.
There is currently not sufficient empirical evidence to recommend specific treatment guidelines for DSPD (Gradisar et al., 2011a), and more controlled studies are consequently warranted (Bjorvatn and Pallesen, 2009; Gradisar et al., 2011a; Mundey et al., 2005). Despite the fact that DSPD is most common in adolescence and young adulthood, we only know of 1 treatment study on adolescents with DSPD (Gradisar et al., 2011a). In the present study, we investigated the short- and long-term effects on subjective and objective sleepiness and cognitive function of bright light and melatonin treatment alongside gradually advanced rise times in adolescents and young adults. The treatment protocol was designed based on knowledge of the advancing effects of melatonin and bright light on the circadian rhythm when timed correctly. The treatment protocol in the present study has been used for many years in clinical practice by some of the coauthors and is described in detail in a review (Bjorvatn and Pallesen, 2009). The effects of 4 short-term treatment conditions were investigated: dim light (placebo) + placebo capsule, bright light + placebo capsule, dim light (placebo) + melatonin capsule, and bright light + melatonin capsule. This was followed by a long-term intervention (approximately 3-month duration) including 2 conditions: no treatment and combined bright light + melatonin. In the present study, we investigated the effect of treatment on subjective and objective daytime function (on a simulated real-life school/work setting starting at 0800 h), with sleepiness and fatigue as primary outcomes and cognitive function as a secondary outcome. Sleep data (sleep diary, actigraphy, and DLMO) were also collected but are described in another article (Saxvig et al., 2013a).
Materials and Methods
Participants
Recruitment and Inclusion/Exclusion Criteria
The participants were recruited to take part in a clinical trial (http://www.clinicaltrials.gov/; NCT00834886) through advertisements at high schools, a college, a university (i.e., e-mails, flyers, posters, and stands) and in relevant media. A link to the study’s website was distributed, which contained information about the disorder, the study protocol, and contact information.
Inclusion criteria were 1) living in Bergen, Norway; 2) aged 16 to 25 years; 3) good general health as specified by the exclusion criteria (see below); and 4) fulfilling the diagnostic criteria for DSPD. The participants were diagnosed according to the criteria found in the International Classification of Sleep Disorders, 2nd version (ICSD-2) (American Academy of Sleep Medicine, 2005), operationalized for this study as the following: 1) problems falling asleep in the evening, 2) falling asleep after 0200 h at least 3 days a week, 3) ability to sleep until early afternoon, 4) problems waking up in time for school/work, 5) early wake-up times associated with extreme daytime sleepiness, 6) good subjective sleep quality and duration when given the opportunity to sleep at self-chosen times, and 7) verbally self-reporting the aforementioned sleep problems as chronic (>6 months).The DSPD diagnosis was confirmed as required by the ICSD-2 criteria by sleep diary data covering a 1-week period and showing a delayed sleep pattern.
Exclusion criteria were sleep disorders other than DSPD, moderate to severe psychopathology (see later for procedure), conditions assumed to affect sleep (i.e., migraine, B12 deficiency), all serious somatic disorders (i.e., rheumatoid arthritis, diabetes), medications or treatments assumed to affect sleep (i.e., sedative antihistamines, antidepressants, hypnotics), substance abuse, night work, intelligence quotient <70, breast feeding, and pregnancy.
A total of 264 potential participants responded to the initial invitation. In total, 60 persons fulfilled the basic criteria for inclusion and were scheduled for a meeting. Of those, 10 withdrew prior to the scheduled meeting. The remaining 50 potential participants were screened with the Structured Clinical Interview for DSM-IV (SCID-I) diagnoses (First et al., 1997), the IQ test Raven’s Progressive Matrices (Raven, 2000), and a pregnancy test (only females) and further set up for polysomnographic screening. A total of 40 participants (see Figure 1 for withdrawal/exclusion categories) were included and successively randomized for participation. None of the participants had ever before been diagnosed with DSPD, nor had they previously received treatment for DSPD. Inclusion and data collection were performed from the fall of 2008 until early 2012. Figure 1 illustrates the participant flow throughout the study.
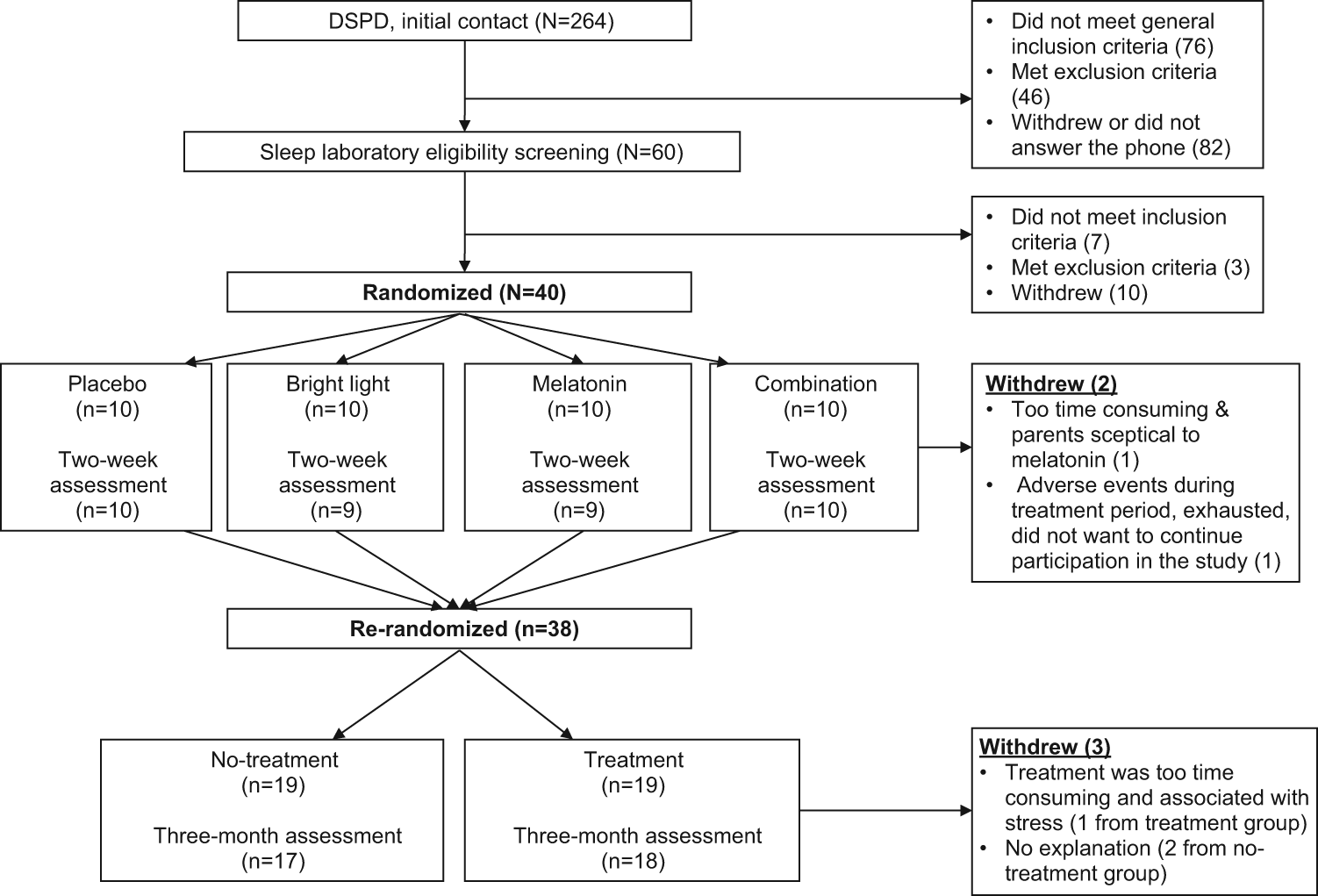
Participation flow throughout the study.
Ethics
Informed consent was obtained after a full explanation of the study protocol. For participants under 18 years of age, parents were required to sign the consent form before inclusion and to give verbal consent. All participants received compensation (approximately US$80) for their time invested in the study. The study was approved by the Regional Committee for Medical and Health Research Ethics in Western Norway, the Norwegian Social Data Service, and the Norwegian Medicines Agency.
Setting and Facilities
All meetings with participants took place at the sleep laboratory at the Faculty of Psychology of the University of Bergen. Inclusion meetings took place between 0800 h and 1700 h, and polysomnography took place between 1800 h and 2330 h. All daytime testing protocol meetings started at 0800 h to 0830 h and ended at about 1400 h to 1500 h. Lighting conditions in the testing rooms were 200 lux. Light was set to 500 lux in the laboratory, where participants stayed during breaks. The laboratory had no windows. Hence, participants were not exposed to daylight while being tested. Although there are seasonal variations in daylight in Norway, these variations are likely to be spread across the sample and should consequently not influence the results. The first author (A.W.-L.) and coauthor I.W.S. administrated all tasks throughout the study (with help from a research assistant, coauthor Ø.V.).
Instruments
Demographic Information
Data were collected regarding age, gender, self-reported average school grades, intelligence (Raven, 2000), symptoms of anxiety and depression (Zigmond and Snaith, 1983), school/employment status (high school or college/university students, employed or unemployed), living conditions (with or without their parents), bedtime, and rise time.
Subjective Self-Evaluations of Sleepiness and Fatigue
The Karolinska Sleepiness Scale (KSS) (Akerstedt and Gillberg, 1990) is a scale in which subjects rate their concurrent sleepiness level. The scale is verbally anchored with steps ranging from 1 (“very alert”) to 9 (“very sleepy, fighting sleep, effort to stay awake”). The Epworth Sleepiness Scale (ESS) (Johns, 1991) is a questionnaire providing a trait measure of daytime sleepiness. The subject rates the probability of falling asleep/dozing off in 8 different everyday situations on a scale ranging from 0 (no probability) to 3 (high probability). We used the validated Norwegian version of the ESS (Pallesen et al., 2007). For the Fatigue Questionnaire (FQ) (Chalder et al., 1993), we used a total fatigue score that was generated from 7 items related to physical fatigue and 4 items related to mental fatigue. We administered the validated Norwegian version of the FQ (Loge et al., 1998).
Objective Sleepiness (Arousal)
The Alpha Attenuation Test (AAT) is a quantitative method for assessing objective sleepiness. It was developed based on the fact that the EEG power spectrum in the alpha frequency band increases with eyes open and decreases with eyes closed as the participant becomes sleepier (Stampi et al., 1995). The participants were instructed to sit comfortably in an office chair in a soundproof chamber, to relax but to remain awake, and to look at a red “X” on the wall straight ahead of them. They were asked to close their eyes and to open their eyes consecutively 3 times at 2-minute intervals. Thus, the total duration of the AAT was 12 minutes. Electrodes were montaged according to the AASM Manual for the Scoring of Sleep and Associated Events (Iber et al., 2007). Data were collected online with ambulatory Embla Titanum recorders (Natus Neurology, Embla Systems Inc., Broomfield, CO) and Somnologica software package (ResMed, Poway, CA). A single EEG derivation was used for spectral analyses (O1). Power spectrum analysis criteria were set to the following: alpha band power = 8 to 11.99 Hz, 1-second epochs, and 256-point fast Fourier transform (FFT) overlapped by 0%, where the FFTs are averaged with the selected epoch duration. We divided the average alpha spectral power of eyes closed by the average alpha spectral power of eyes open, which gives a ratio of the “alpha attenuation coefficient” (AAC) (Stampi et al., 1995). The higher the AAC, the higher the alertness level.
Cognitive Function
The present study included a set of standardized psychometric tests to measure aspects of attention and executive function. The Conners’ Continuous Performance Test (CPT-II) Version 5.1 for Windows is a test of different aspects of attention (Conners and MHS Staff, 2004). It lasts 14 minutes. The subject is instructed to press the space bar on the computer keyboard whenever a letter appears on the screen, except for the letter “X.” The CPT-II is a test assessing the ability to maintain focused alertness over a relatively long period of time. In the present study, we included an overall reaction time measure (RT for hits), 2 measures of accuracy (number of omissions; responses that should have been given but were not) and commissions (wrong responses given when there should be no response), and 2 measures of consistency (hit RT standard error and variability). Three subtests of the Wechsler Adult Intelligence Scale-III (WAIS-III) (Wechsler, 1997)—the Digit Symbol, Letter-Number Sequencing, and Symbol Search Tests—were included as measures of working memory/processing speed. During the Digit Symbol Test, digits and symbols are presented as pairs, and additional digits and symbols must be paired by the subject. The Letter-Number Sequencing Test requires that the subject order numbers and letters that are presented in an unordered sequence. In the Symbol Search Test, the subject must match symbols that appear in different groups. Finally, 3 subtests of the Delis-Kaplan Executive Function System (D-KEFS) (Delis et al., 2001) were included as measures of executive function. From the Color Word Interference Test (CWIT), we included the third condition in which the subjects are presented color words printed in incongruently colored ink (CWIT-Inhibition) and the fourth condition, which requires the subject to switch between either naming the color of the incongruently colored words or reading the color names when presented with a brace (CWIT-Inhibition/Switching). From the Verbal Fluency Test (VFT), we included the condition in which the subjects are asked to switch between reporting items from 2 different categories (VFT-Category/Switching).
Compliance and Side Effects
A form to be completed each day of the 2-week treatment period was developed in order to measure compliance for the timing/duration of light exposure and if the capsule had been taken and at what time. Points were given for light exposure (1 point), capsule taken (1 point), and rise time (1 point) at the prescribed time (±1 hour). A full score on all measures was rated as 100% compliance. For the 3-month follow-up, points were given for light exposure (yes = 1 point) and melatonin capsule taken (yes = 1 point). A full score was rated as 100% compliance. Side effects during treatment were retrospectively recorded at the 2-week assessment (participants noted any possible side effects on a form developed for the study) and at 3-month follow-up. In cases of adverse events, guidelines for Good Clinical Practice were followed (Switula, 2000).
Procedure/Design
Participants underwent the same daytime testing protocol at 3 assessment points: before treatment (baseline assessment), after 2-week treatment (2-week assessment), and after 3 months of treatment/no treatment (3-month assessment). Prior to the baseline assessment, participants had been instructed to go to bed and rise at self-determined times for 4 consecutive days in order to avoid sleep deprivation. On the assessment days, the participants were instructed to rise at 0700 h and to meet at the sleep laboratory at 0800 h. Test sessions started about every hour from 0900 h to about 1300 h with intermediate breaks, with the purpose of simulating a typical day at school or a day at work (between 0800 h and 0900 h, the participants had breakfast, and electrodes for the AAT were montaged). The AAT, KSS, and CPT-II were all administered 3 times each assessment day, in the mentioned order, at approximately 0900 h, 1100 h, and 1300 h. All other tests were administered once. The protocol included measures of subjective sleepiness and fatigue (KSS, ESS, and FQ), objective sleepiness/arousal (AAT), and performance on tests of cognitive function (CPT-II, WAIS-III subtests, and D-KEFS subtests).
Participants were randomized into 1 of 4 treatment conditions lasting for 2 weeks in a double-blind, placebo-controlled design. The 4 treatment conditions were dim light (placebo) and placebo capsules (placebo group), bright light and placebo capsules (bright light group), dim light (placebo) and melatonin capsules (melatonin group), and bright light and melatonin capsules (combination group). All groups were instructed to use dim/bright light and placebo/melatonin capsules on a gradual advancement schedule regarding rise times and treatment times. In a follow-up study, participants were re-randomized into 2 groups receiving either the treatment combination of bright light and melatonin alongside gradually advanced rise times (treatment group) or no treatment (no-treatment group) in an open-label trial for approximately 3 months.
Treatment Protocol
The treatment protocol was based on the approach described by Bjorvatn and Pallesen (2009). The participants were instructed to sleep until spontaneous awakening on the first day of treatment and then to expose themselves to the study light source for 30 to 45 minutes immediately after awakening, with eyes directed towards the lamp. Melatonin/placebo capsules were to be taken 12 hours after light treatment exposure was initiated. Because of the soporific effects of melatonin, capsules were not to be taken before 2000 h in the evening. This instruction ensured that the treatment was individualized according to how delayed each participant’s circadian rhythm was. Rise time was advanced by 1 hour every day until the preferred rise time was reached (each participant chose their target rise time) and then maintained until the end of the 2-week treatment period. If the participant overslept, the instructions were to expose oneself to the study light lamp immediately upon awakening, to take a capsule 12 hours later, and then to advance by 1 hour every day until the target rise time. No instructions were provided regarding bedtime. Alcohol usage was prohibited during the 2-week treatment period; no instructions regarding alcohol use were given during the 3-month follow-up study.
Bright White Light and Dim Red Light
Light lamps were ML-10,000 manufactured by Miljølys Inc. (Tynset Elektronikk, Tynset, Norway). ML-10,000 is a light box (47 × 17.5 × 29 cm) containing 3 fluorescent bulbs (rendering average index of 80; light temperature of 4000 K) (Ecotone, P1-L, Philips, Oslo, Norway). Lamplights were either bright white (approximately 10,000 lux at 50-cm distance) with a clear cover screen or dim (approximately 400 lux at 50-cm distance) with a red cover screen. Dim light lamps are traditionally assumed to have a minimal effect on the endogenous circadian rhythm (Lewy et al., 1980), and dim light (300 lux) was used as the control condition in the study by Rosenthal and colleagues (1990), who found superior effects in their active condition (2500 lux).
Melatonin Capsules and Placebo Capsules
Hard capsules were packed by Kragerø Tablettproduksjon Inc. (Kragerø, Norway) for the 2-week intervention and contained either 3 mg of fast-release melatonin (5-methoxy-N-acetyltryptamine) or 3 mg of maydis amylum (maize starch). Melatonin was purchased from Nature’s One (Asaman Inc., Avon, MA). We used the original capsules of 3 mg from Nature’s One for the 3-month follow-up study.
Blinding
The 2-week treatment study was double blinded. Participants were informed that they would receive either red or white light but not that the light intensity was different. Lamps were given to the participants, which were packed in boxes concealing the color of the cover screen and differentiated by a letter code (A and B), and the participants were instructed not to reveal the color to the study administrators at any time (blinding of light boxes was performed by 2 university employees not otherwise involved in the study). Participants were further informed that the capsules contained either melatonin or maize starch. The melatonin and placebo capsules were packed in identical containers differentiated by a number code (1 and 2). In case of adverse events, those who blinded the lamp boxes kept the code for the capsules and the lamps throughout the data collection period. The 3-month follow-up study was not blinded.
Randomization
The randomization lists were created (4 groups for the 2-week intervention and 2 groups for the 3-month follow-up) using the Internet-based program Research Randomizer (http://www.randomizer.org/form.htm). Participants were chronologically assigned to the respective groups on inclusion.
Statistical Analyses
Data were analyzed using SPSS Statistics version 19.0 (IBM, Armonk, NY). In cases of withdrawal from the study or missing data, baseline values were carried forward in an intention-to-treat analysis to ensure that clinical effectiveness was not overestimated (Hollis and Campbell, 1999).
Demographic variables were compared between groups using 1-way ANOVA and t tests for independent samples (age, self-reported school grades, intelligence, anxiety, depression, bedtime, and rise time) and the Pearson χ2 test (gender, high school/university or college, living situation) to investigate whether the groups differed on these parameters at baseline. One-way ANOVA was also performed on all outcome measures at baseline between the four 2-week intervention groups and between the two 3-month groups.
Effects of the 2-week treatment period were compared between the 4 groups using 2-way ANOVA (4 treatment groups × 2 assessment points) for repeated measures of the KSS, AAT, and CPT-II (values at all 3 sessions: 0900 h, 1100 h, and 1300 h). Only the mean values of the 3 sessions are presented in the tables because no additional information was provided by analyzing each of them separately. Two-way ANOVA for repeated measures between the baseline and 2-week assessment was also performed for the WAIS-III subtests, the D-KEFS subtests, and for the questionnaires (ESS and FQ). Raw scores (as opposed to scaled scores and t scores relating to a normative average) were used for analysis of all cognitive performance tests (CPT-II, WAIS-III, and D-KEFS). For compliance and measures of side effects, 1-way ANOVA (percentage of compliance) and the χ2 test (compliance ≥50% = yes, compliance <50% = no; side effect categories; and reported side effects = yes, did not report side effects = no) were performed to test for differences between the groups.
At the 3-month assessment, the same variables were compared between the groups (no treatment/treatment) with respect to both the baseline assessment and the 2-week assessment using 2-way ANOVA for repeated measures (2 treatment groups × 2 assessment points). Interaction effects were further assessed using t tests for paired samples. Cohen d was calculated between the baseline and 2-week assessment and between the baseline and 3-month assessment using an online calculator (http://easycalculation.com/statistics/effect-size.php) with the following formula: d = M1 − M2/(sqrt(SD12 + SD22)/2).
Results
Baseline Assessment
There were no differences in terms of demographic variables between the 4 groups in the 2-week treatment study at baseline (Table 1). For the 3-month follow-up, participants in the treatment group were somewhat older than the participants in the no-treatment group (p = 0.042) (Table 1). At baseline, there were no differences between the 2-week groups or the 3-month groups on any of the primary or secondary outcome measures.
Demographic information at baseline for the 2-week intervention groups, the 3-month follow-up groups, and all participants (N = 40).
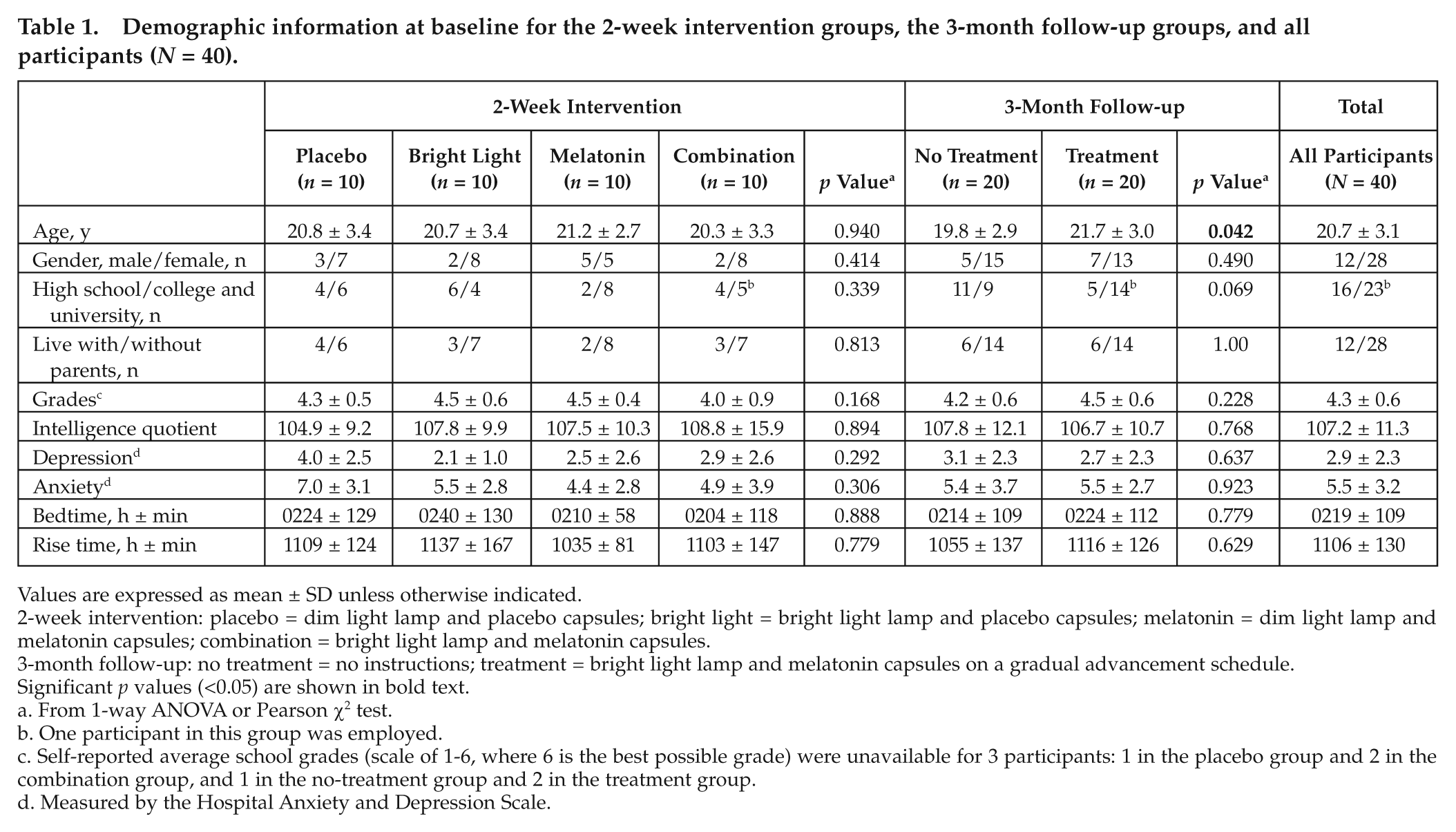
Values are expressed as mean ± SD unless otherwise indicated.
2-week intervention: placebo = dim light lamp and placebo capsules; bright light = bright light lamp and placebo capsules; melatonin = dim light lamp and melatonin capsules; combination = bright light lamp and melatonin capsules.
3-month follow-up: no treatment = no instructions; treatment = bright light lamp and melatonin capsules on a gradual advancement schedule.
Significant p values (<0.05) are shown in bold text.
From 1-way ANOVA or Pearson χ2 test.
One participant in this group was employed.
Self-reported average school grades (scale of 1-6, where 6 is the best possible grade) were unavailable for 3 participants: 1 in the placebo group and 2 in the combination group, and 1 in the no-treatment group and 2 in the treatment group.
Measured by the Hospital Anxiety and Depression Scale.
2-Week Assessment
Subjective sleepiness and fatigue
At the 2-week assessment, all groups showed a reduction in subjective sleepiness measured with the KSS (p < 0.0005). There were no interaction effects and hence no differential improvement on the KSS between the 4 groups (Table 2). Similarly, subjective sleepiness measured by the ESS showed an effect of time (p = 0.028). All groups had improved subjective sleepiness measured by the ESS at the 2-week assessment compared to baseline but with no clear significant interaction. All 4 groups were less fatigued at the 2-week assessment with no interaction effect (Table 2).
Subjective and objective sleepiness and fatigue in participants with DSPD before (baseline assessment) and after treatment (2-week assessment) with bright light and melatonin in a randomized, double-blind, placebo-controlled design.
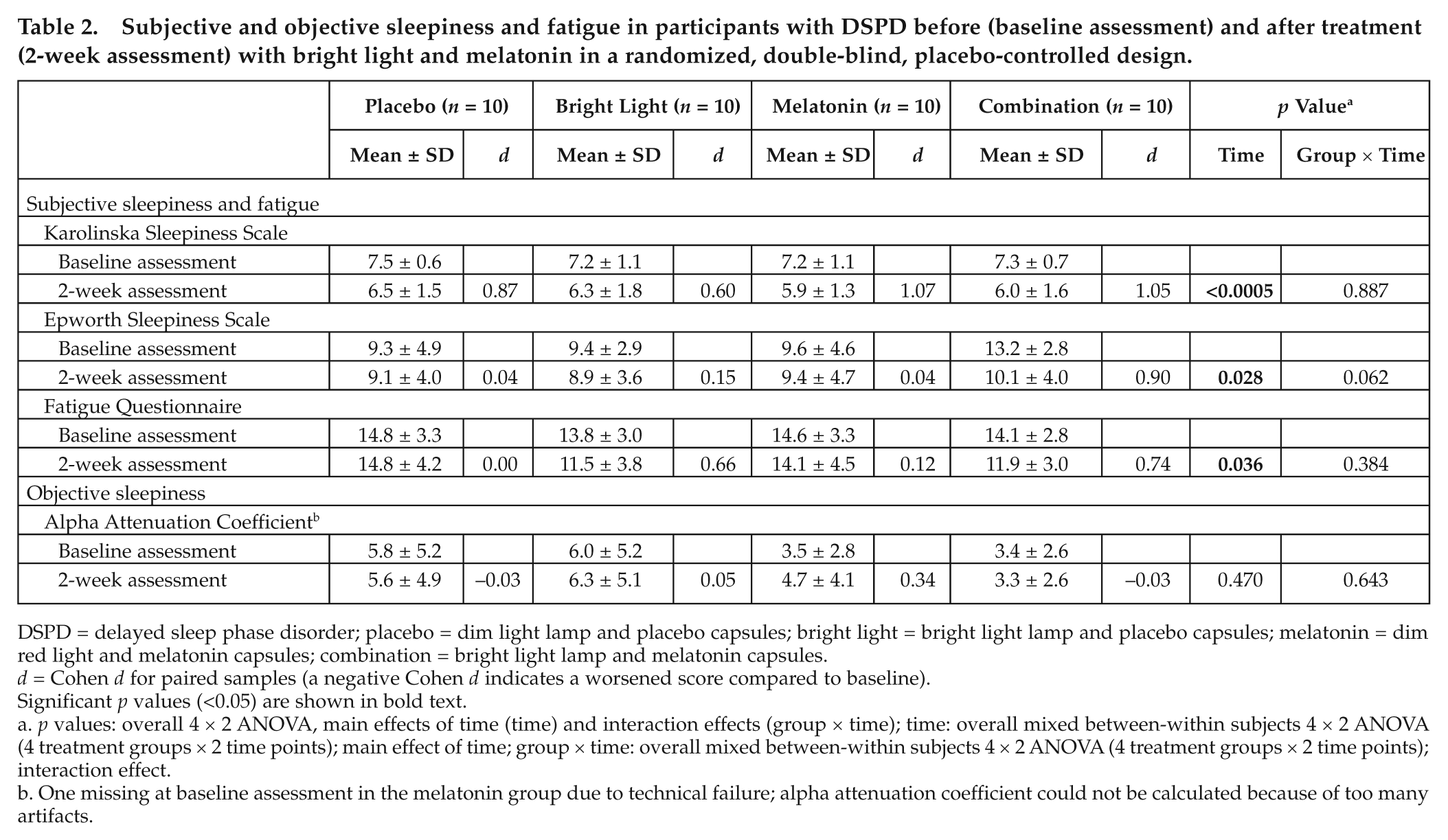
DSPD = delayed sleep phase disorder; placebo = dim light lamp and placebo capsules; bright light = bright light lamp and placebo capsules; melatonin = dim red light and melatonin capsules; combination = bright light lamp and melatonin capsules.
d = Cohen d for paired samples (a negative Cohen d indicates a worsened score compared to baseline).
Significant p values (<0.05) are shown in bold text.
p values: overall 4 × 2 ANOVA, main effects of time (time) and interaction effects (group × time); time: overall mixed between-within subjects 4 × 2 ANOVA (4 treatment groups × 2 time points); main effect of time; group × time: overall mixed between-within subjects 4 × 2 ANOVA (4 treatment groups × 2 time points); interaction effect.
One missing at baseline assessment in the melatonin group due to technical failure; alpha attenuation coefficient could not be calculated because of too many artifacts.
Objective sleepiness/arousal
There were no differences between baseline and the 2-week assessment on the AAC (Table 2).
Cognitive performance
There was no effect of time or interaction on any of the CPT-II measures for RT or accuracy. There was an overall improvement over time on all working memory/processing speed variables (WAIS-III), but no interaction effect between the 4 groups was found (Table 3). There was no effect of time or interaction on CPT-II consistency measures. There was an overall improvement with time on all executive function measures (VFT-Category/Switching, CWIT-Inhibition, and CWIT-Inhibition/Switching) across all groups, with no interaction effect (Table 3).
Cognitive performance measures in participants with DSPD before (baseline assessment) and after treatment (2-week assessment) with bright light and melatonin in a randomized, double-blind, placebo-controlled design.
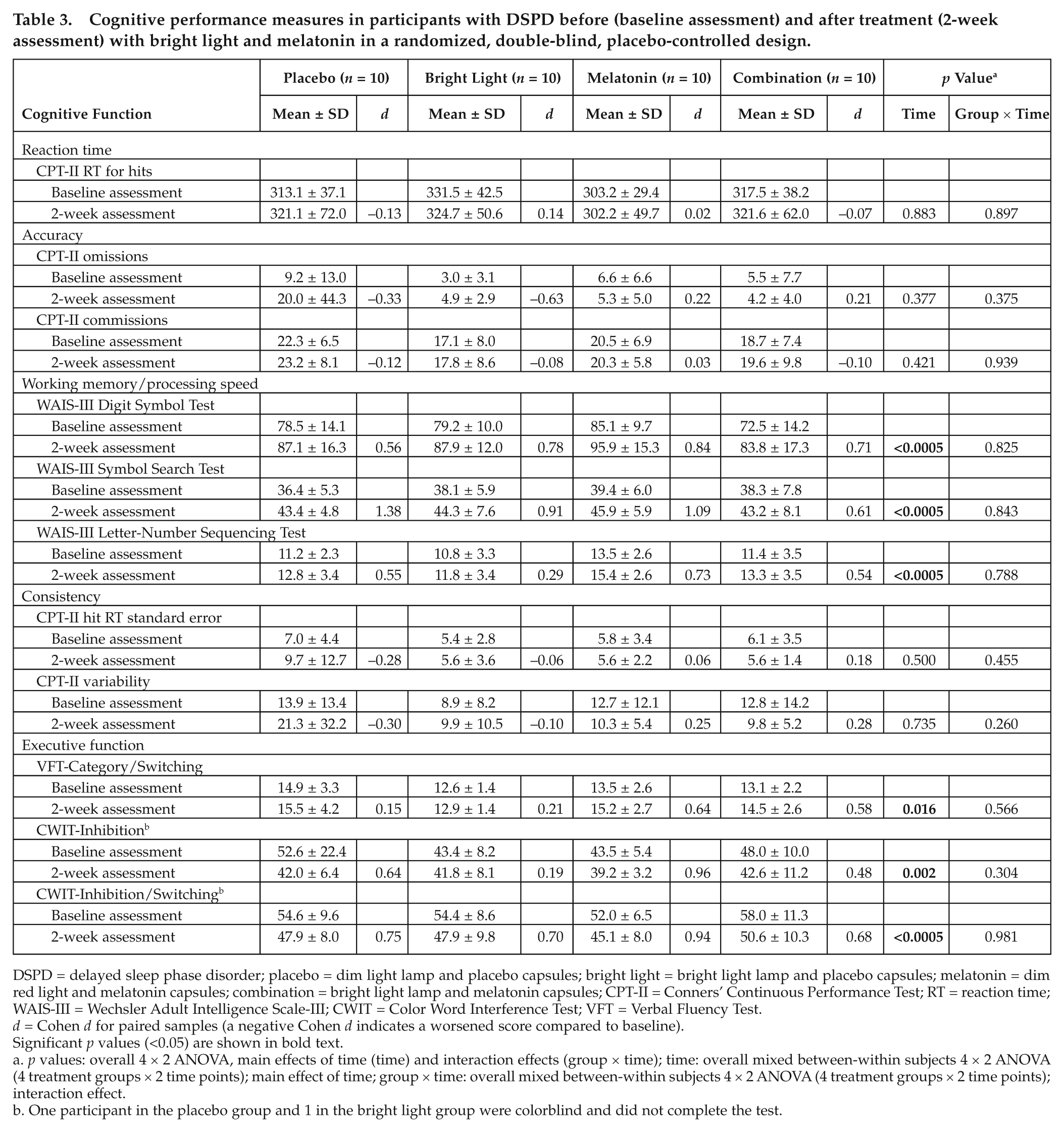
DSPD = delayed sleep phase disorder; placebo = dim light lamp and placebo capsules; bright light = bright light lamp and placebo capsules; melatonin = dim red light and melatonin capsules; combination = bright light lamp and melatonin capsules; CPT-II = Conners’ Continuous Performance Test; RT = reaction time; WAIS-III = Wechsler Adult Intelligence Scale-III; CWIT = Color Word Interference Test; VFT = Verbal Fluency Test.
d = Cohen d for paired samples (a negative Cohen d indicates a worsened score compared to baseline).
Significant p values (<0.05) are shown in bold text.
p values: overall 4 × 2 ANOVA, main effects of time (time) and interaction effects (group × time); time: overall mixed between-within subjects 4 × 2 ANOVA (4 treatment groups × 2 time points); main effect of time; group × time: overall mixed between-within subjects 4 × 2 ANOVA (4 treatment groups × 2 time points); interaction effect.
One participant in the placebo group and 1 in the bright light group were colorblind and did not complete the test.
Dropouts, compliance, and side effects
Two participants withdrew from the 2-week study (see Figure 1 for explanation). Compliance was 84.5% in the combination group, 77% in the melatonin group, 86.3% in the bright light group, and 87.3% in the placebo group, with no difference between the groups (p = 0.567). One participant in the combination group and 2 in the melatonin group were rated as noncompliant (<50%) (p = 0.306). Side effects were reported by 15 participants. Of these 15 participants, 6 were in the combination group, 2 in the melatonin group, 4 in the bright light group, and 3 in the placebo group (p = 0.409). Side effects reported were headache (n = 5), nausea (n = 3), discomfort in the eyes (n = 5), and skin irritation (n = 2). None of the side effects were serious or long lasting. At the 2-week assessment, there was advancement in the rise time of almost 2.5 hours and bedtime of about 1 hour across all groups, with no interaction effect (Saxvig et al., 2013a).
3-Month Assessment
Because there were no interactions differentiating the 4 treatment conditions at the 2-week assessment, the 2-week data were therefore collapsed (2-week groups were combined) when they were used in the analysis for the 3-month follow-up.
Subjective sleepiness and fatigue
At the 3-month follow-up, there was an effect of time (p < 0.0005) as well as an interaction effect (p = 0.022) on the KSS in favor of the treatment group (Table 4). The treatment group reported reduced subjective sleepiness compared to baseline at all 3 time points (0900 h, p = 0.002; 1100 h, p < 0.0005; and 1300 h, p = 0.013). The no-treatment group did not score differently at 3-month follow-up compared to baseline on any test session. Subjective sleepiness measured by the ESS showed a positive effect of time compared to baseline but no interaction. However, there was both an effect of time and an interaction effect when comparing the 2-week to the 3-month assessment, revealing that the treatment group reduced their score over time (p < 0.0005). The no-treatment group did not score differently on the ESS at the 3-month assessment compared to the 2-week assessment (p = 0.180) (Table 4). There was an effect of time and an interaction effect on fatigue at the 3-month assessment compared to baseline. The treatment group was less fatigued (p = 0.004), while the no-treatment group scored no differently from baseline on the fatigue scale (p = 0.894) (Table 4).
Measures of subjective and objective sleepiness and fatigue in participants with DSPD at 3-month follow-up compared to the baseline assessment (baseline to 3 months) and 2-week assessment (2 weeks to 3 months).
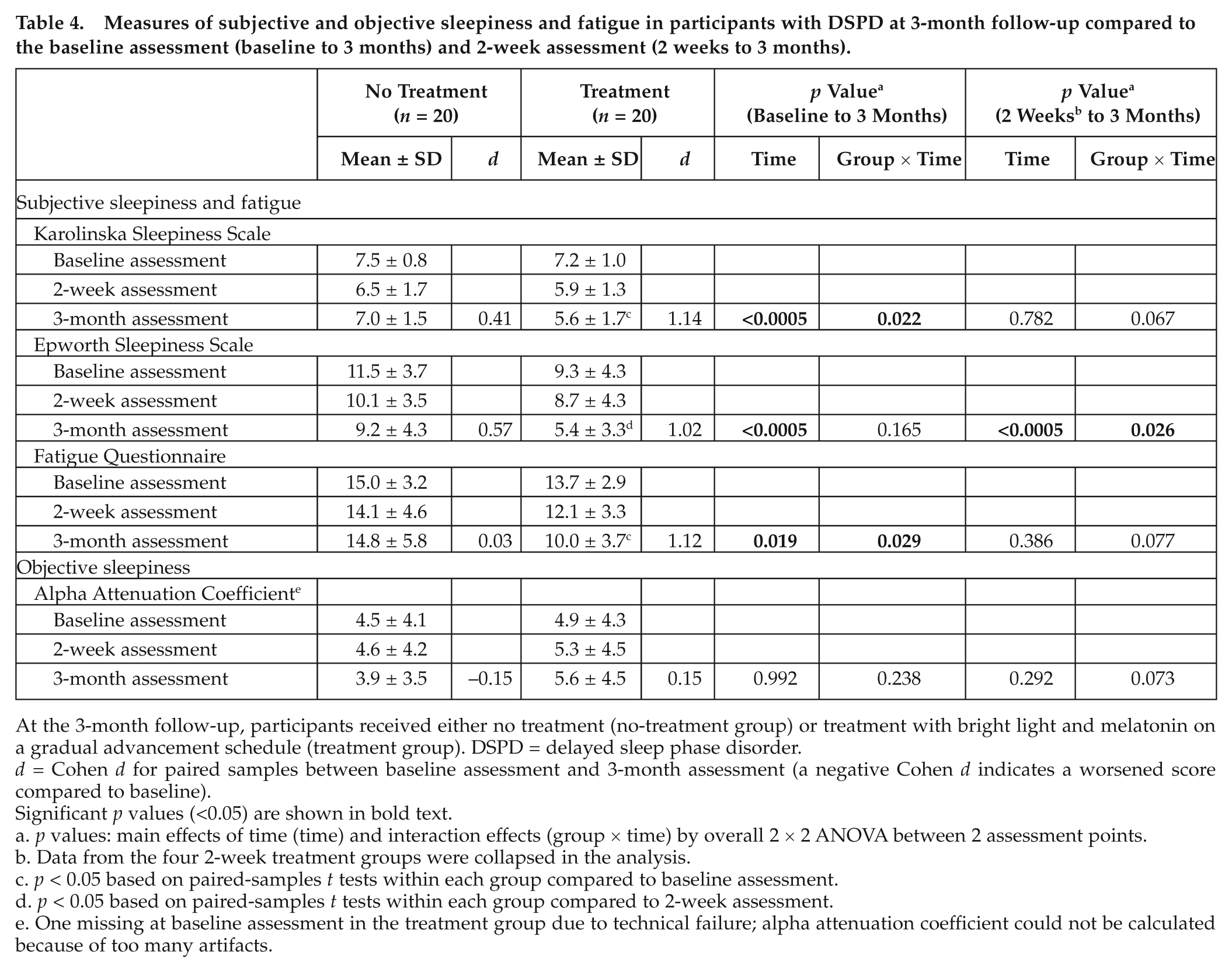
At the 3-month follow-up, participants received either no treatment (no-treatment group) or treatment with bright light and melatonin on a gradual advancement schedule (treatment group). DSPD = delayed sleep phase disorder.
d = Cohen d for paired samples between baseline assessment and 3-month assessment (a negative Cohen d indicates a worsened score compared to baseline).
Significant p values (<0.05) are shown in bold text.
p values: main effects of time (time) and interaction effects (group × time) by overall 2 × 2 ANOVA between 2 assessment points.
Data from the four 2-week treatment groups were collapsed in the analysis.
p < 0.05 based on paired-samples t tests within each group compared to baseline assessment.
p < 0.05 based on paired-samples t tests within each group compared to 2-week assessment.
One missing at baseline assessment in the treatment group due to technical failure; alpha attenuation coefficient could not be calculated because of too many artifacts.
Objective sleepiness/arousal
There was no effect of time or interaction between the baseline and 3-month assessment of the AAC (Table 4).
Cognitive function
There was an interaction effect on the RT measure when comparing the 3-month follow-up to baseline (p = 0.010) and the 2-week assessment (p = 0.011). The effect was due to slower RT for hits in the no-treatment group at 3-month follow-up compared to baseline and the 2-week assessment, while the treatment group did not score differently than baseline at the 2-week or 3-month assessment (Table 5). Three-month follow-up data showed that there was an improved effect of time from baseline on all working memory/processing speed variables. However, there was no interaction effect. There was also an improvement over time at 3-month follow-up compared to the 2-week assessment on the WAIS-III Digit Symbol Test, with no interaction effect (Table 5). There was an interaction effect on the CPT-II consistency measures of hit RT standard error and variability (Table 5). The treatment group scored more consistently than the no-treatment group at the 3-month assessment compared to baseline. There was a positive effect of time on all executive function measures and an interaction effect on the VFT-Category/Switching. Paired-samples t tests showed that the treatment group had improved their performance on the VFT-Category/Switching, while the no-treatment group scored no differently from baseline (Table 5).
Measures of cognitive performance in participants with DSPD at 3-month follow-up compared to the baseline assessment (baseline to 3 months) and 2-week assessment (2 weeks to 3 months).
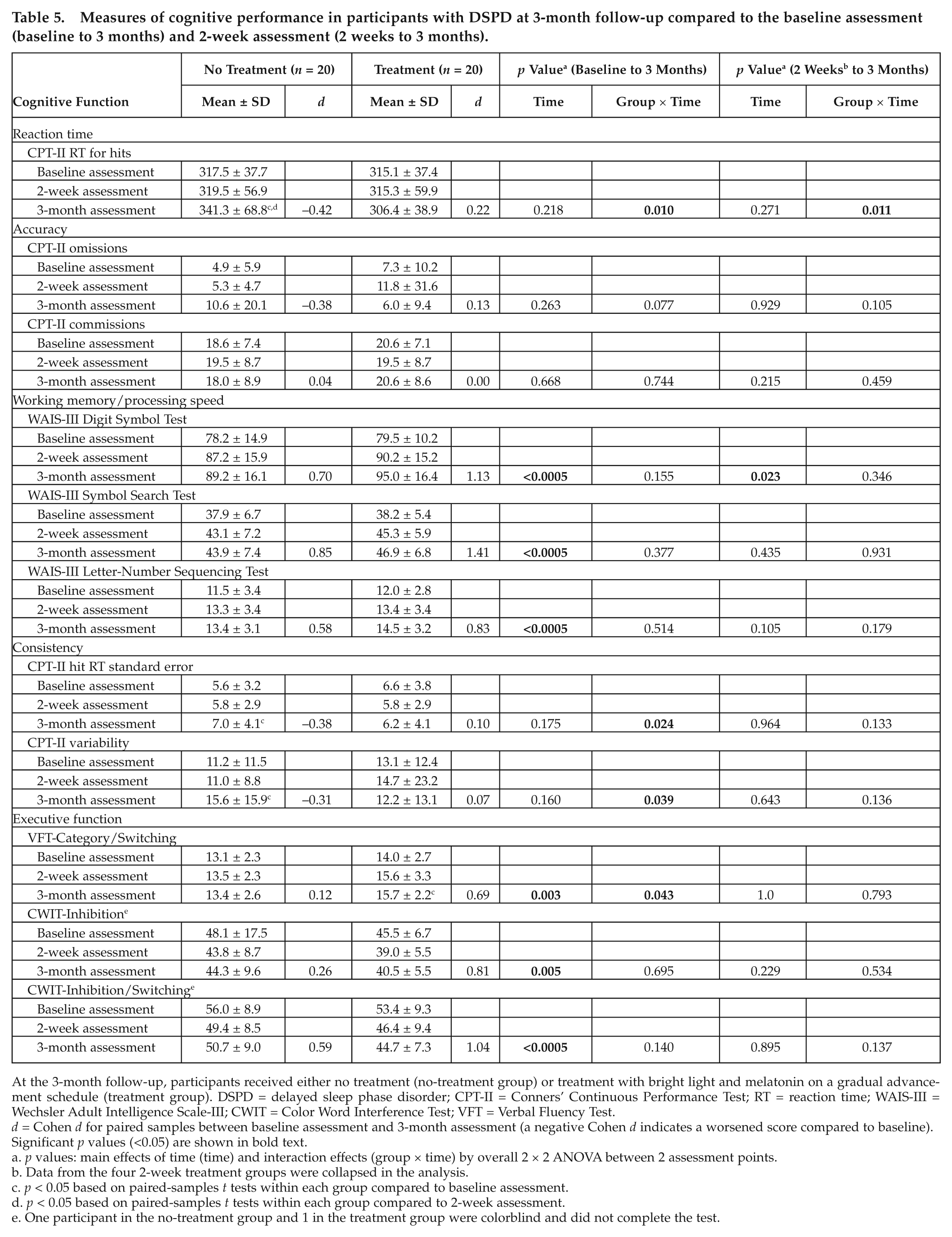
At the 3-month follow-up, participants received either no treatment (no-treatment group) or treatment with bright light and melatonin on a gradual advancement schedule (treatment group). DSPD = delayed sleep phase disorder; CPT-II = Conners’ Continuous Performance Test; RT = reaction time; WAIS-III = Wechsler Adult Intelligence Scale-III; CWIT = Color Word Interference Test; VFT = Verbal Fluency Test.
d = Cohen d for paired samples between baseline assessment and 3-month assessment (a negative Cohen d indicates a worsened score compared to baseline).
Significant p values (<0.05) are shown in bold text.
p values: main effects of time (time) and interaction effects (group × time) by overall 2 × 2 ANOVA between 2 assessment points.
Data from the four 2-week treatment groups were collapsed in the analysis.
p < 0.05 based on paired-samples t tests within each group compared to baseline assessment.
p < 0.05 based on paired-samples t tests within each group compared to 2-week assessment.
One participant in the no-treatment group and 1 in the treatment group were colorblind and did not complete the test.
Dropouts, compliance, and side effects
Three participants withdrew from the 3-month follow-up (see Figure 1 for explanation). Average compliance was 58% in the treatment group when including 19 participants (1 missing value because compliance was not recorded by 1 participant). Thirteen participants (65%) were rated as compliant to treatment (≥50%) and 7 (35%) as noncompliant (<50%). Side effects were reported by 3 participants in the form of headache (n = 1) and discomfort in the eyes (n = 2). Side effects were not serious or long lasting.
The 3-month assessment showed an advancement in the rise time of about 2 hours compared to baseline and bedtime of more than 1 hour in the treatment group, whereas the no-treatment group had returned to the baseline timing of rise time and bedtime (Saxvig et al., 2013a).
Discussion
The effects of bright light and melatonin administered alongside a gradual advancement of rise times were investigated using a randomized, double-blind, placebo-controlled design. After short-term treatment (2 weeks), subjective sleepiness measured by the KSS was reduced across groups by more than 1 point with large effect sizes. However, there were no differences between the 4 treatment conditions. The 2-week treatment also yielded a reduction in subjective sleepiness in all groups assessed by the ESS, with a large effect size for the combination group only, and the treatment also resulted in a reduction of fatigue across the 4 groups. Participants in all 4 conditions improved their cognitive performance on tests of working memory/processing speed and executive function but not on the RT, accuracy, and consistency measures from the CPT-II. In sum, bright light, melatonin, a combination, and placebo alongside a gradual advancement schedule were almost equally effective in improving subjective daytime sleepiness, fatigue, and cognitive function in the 2-week study. This contradicts the findings from previous studies, where subjective sleepiness, objective sleepiness, fatigue, and alertness improved significantly only after active treatment (Gradisar et al., 2011a; Kayumov et al., 2001; Rosenthal et al., 1990).
It has been assumed that patients with DSPD cannot achieve advancement in the sleep phase (leading to a reduction in daytime sleepiness) by behavioral means (American Academy of Sleep Medicine, 2005). However, Sharkey and colleagues (2011) recently found that strict rise schedules could phase advance sleep in participants with a delayed sleep phase. They did not assess sleepiness, but our findings suggest that participants diagnosed with DSPD can also achieve positive effects on subjective daytime sleepiness by gradual advanced rise times. Cole and colleagues (2002) did find a reduction in subjective morning sleepiness in the most delayed subjects, also in the dim light group, but the effect was larger after bright than dim light. The 2-week treatment yielded no positive effects on objective sleepiness (AAT) in our study, in contrast to the findings by Rosenthal and colleagues (1990) of increased sleep latency on the MSLT during morning hours after bright light compared to dim light treatment. However, the AAT may be regarded mainly as a measure of arousal, while the MSLT is a measure of sleep propensity; hence, these 2 objective measures of physiological sleepiness may reflect different sleepiness modalities. In line with this, Alloway and colleagues (1997) found correlations between AAT and MSLT scores for a group of patients with narcolepsy but not for normal sleepers.
Long-term effects of bright light and melatonin were investigated using a randomized, controlled design for 3 months. The treatment group was less sleepy as measured by the KSS compared to baseline and maintained a positive effect after short-term treatment at the 3-month follow-up. On the other hand, subjective sleepiness measures were no different from baseline measures in the no-treatment group; hence, the positive effect of the 2-week treatment was eliminated when active treatment was terminated. Simultaneously, the treatment group had further reduced their subjective sleepiness as measured by the ESS from the 2-week to the 3-month assessment and reported less fatigue after the 3-month treatment compared to baseline. These findings indicate that a treatment of DSPD maintained longer than 2 weeks can lead to better effects on subjective sleepiness and fatigue. This is in line with Rosenthal and collegues (1990), who suggest that maintained treatment is necessary, because otherwise the circadian rhythm tends to drift back to a delayed phase in patients with DSPD. Similar to the effects on subjective sleepiness, the treatment group had maintained an advancement of rise time and bedtime after the 3-month treatment, while the no-treatment group had drifted back to delayed rise times and bedtimes.
Strengths and Limitations of the Study
To our knowledge, this is the first study that systematically investigated the effect on daytime function of bright light and melatonin in the treatment of DSPD. Previous studies on the effects of bright light have included behavioral instructions of advanced rise times, but no studies on the effect of melatonin have included a gradual advancement of rise times in their treatment protocol. This study is the first to assess both short- and long-term effects of treatment using a controlled design combining bright light, melatonin, and a gradual advanced rise time. It is also the first controlled study investigating the effect of bright light and melatonin on sleepiness and performance in adolescents and young adults diagnosed with DSPD. By holding the timing of the test sessions constant and in the morning, circadian confounding effects were reduced. Circadian variations in cognitive performance in relation to local and internal times have been shown, for example, in shift workers (Vetter et al., 2012).
The participants in the present study were thoroughly screened for other pathologies. Hence, anxiety, depression, or attention disorders can thus be ruled out as probable confounders to our results. However, the results may, on the other hand, not be directly transferable to the large number of patients with DSPD who suffer from comorbid disorders (Reid et al., 2012). Furthermore, our participants scored somewhat above the average range on self-reported school grades and intelligence, which may represent a bias towards relatively well-functioning patients with DSPD compared to patients whom we commonly meet in sleep clinics, for example, those who have dropped out of school or who are unemployed because of their sleep disorder.
The conceptualization and operationalization of sleepiness are under constant debate in the research literature (Horne, 2010). Because of this, we included a wide range of tests measuring different modalities of sleepiness and cognitive functions, which are believed to be transferable to performance during a day at school or a day at work. Although improvements seen across groups on tests of cognitive function after 2 weeks are expected due to a practice effect (Horne, 2010; Van Dongen et al., 2003), it could not explain group differences. In addition, we cannot rule out that the improvements seen across the 2-week groups were due to a placebo effect, as all groups were active in following the instructions. Similarly, we cannot rule out that the differences seen across the groups in the 3-month study were due to a placebo effect, as only the treatment group received instructions and was an active group.
The sample size in the present study was quite large, but the small group sizes in the 4-armed 2-week trial may still represent a limitation. There was a small age difference in the 3-month follow-up groups, which possibly could account for some of the differences between the effects in the 2 groups. No other differences between background variables or baseline assessment were found; therefore, it seems unlikely that the difference in age can explain the results.
As regards to the 2-week treatment groups, the dim light lamps were used as placebo because they, according to previous studies, have no chronobiotic effect (Lewy et al., 1980; Rosenthal et al., 1990). However, more recent research has indicated that dim light at optimal times may have phase-setting effects (Duffy and Wright, 2005). Light of 90- to 150-lux intensities has in recent studies been shown to have an effect on the melatonin phase in healthy volunteers (Dijk et al., 2012), and it has been shown that long-wavelength light has smaller effects on the circadian phase than short-wavelength light (Wright et al., 2004). It is possible that the positive effects after the 2-week assessment were due to the effect of light in all groups, and the behavioral instructions to gradually advance rise times in itself lead to earlier exposure to ambient light. Hence, the exact reason why all groups improved on many variables after the 2-week study is not clear. The fact that all groups in the 2-week study (including the placebo group) were active and given the same behavioral instructions may differentiate them from pure wait-list controls, which has been used in other studies (Gradisar et al., 2011a). The no-treatment group in the 3-month follow-up study had become familiar with the positive effects of a gradual advancement of rise times, but they were not encouraged or discouraged to continue this behavior. This fact, however, may differentiate them from a wait-list comparison group. We did not use statistical corrections for multiple comparisons in the analysis of cognitive function, but the conclusions in the present study are supported by the consistency of results within the different functional areas. Another limitation of the study was that we individualized target rise times in the treatment protocol but did not record these and can therefore not report how many participants reached and/or adhered to their target rise time.
In the 2-week study, compliance was high across all groups, and the lack of differentiating results between the active treatment groups and placebo cannot be attributed to differences in compliance. For the 3-month treatment group, compliance was lower but still satisfactory from a clinical viewpoint. If compliance was higher and all participants in the 3-month treatment group complied, the results might have been different. Our aim was to assess the effectiveness of a treatment modality that is readily available in, for example, a primary care setting with limited time per patient. Some patients (4 of our participants were not compliant with instructions during the 3-month treatment) with DSPD will likely need closer follow-up in order to adhere to treatment.
Conclusion
A gradual advancement of rise times seemed to be effective in producing positive effects on subjective sleepiness, fatigue, and cognitive performance in the short-term treatment of DSPD. However, the benefits from a gradual advancement of rise times seem to wear off, suggesting that the continuation of bright light and melatonin treatment is beneficial to maintain positive effects over time.
Footnotes
Acknowledgements
First and foremost, the authors thank the participants for their generous contributions to this study. They also thank Kragerø Tablettproduksjon Inc. (Anne Hopstock) for help with the study medicine and placebo capsules, Rune Mentzoni and Geir Scott Brunborg for help with the blinding procedures, and Elisabeth Flo for help with the procedure of the spectral power analyses for the AAT. Finally, the authors thank the Meltzer Foundation for grant funding used for the execution of this study.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
