Abstract
Circadian variation in cardiac autonomic nervous system activity and behavior during the day shifts of shift workers has not hitherto been clarified. This study examined diurnal 24-h variation in heart rate variability (HRV), sleep-wake cycle, physical activity, and food intake during the day shift in rotating shift workers. The subjects were female nurses and caregivers working at a health care facility (14 day workers and 13 rotating shift workers). Each subject was asked to undergo 24-h electrocardiograph and step count recordings. Coarse graining spectral analysis was used for approximately 10-min segments of HRV (600 beats) to derive the total power (TOT: >0.04 Hz), integrated power in the low-frequency (LF: 0.04-0.15 Hz) and high-frequency (HF: >0.15 Hz) ranges, the ratio of HF power to TOT (HF nu), and the ratio of LF power to HF power (LF/HF). Double cosinor analysis was used to obtain 24-h and 12-h period variations in variables of HRV and physical activity. While no difference was found in the acrophases of either period for step counts or in the 12-h period of HRV variables between the groups, the acrophases of the 24-h period for HRV variables were delayed by 1.3 to 5.5 h in rotating shift workers, and their differences in HF power, HF nu, and LF/HF reached a significant level (p < 0.05). On the days of the experiment, retiring time, waking up time, total time in bed, sleep efficiency, and mealtimes and energy intake for each diet did not differ between the groups. These results suggest that there is a possibility of an abnormal phase angle between circadian variation in cardiac autonomic nervous system activity and the sleep-wake cycle during the day shift in shift workers.
Keywords
Shift workers have a higher risk of health problems such as cardiovascular disease, abnormal metabolism, obesity, and cancer compared with day workers (Akerstedt et al., 1984; Boggild and Knutsson, 1999; Costa, 2003; Fujino et al., 2006; Prasai et al., 2008; Sookoian et al., 2007). Misalignment of the circadian clock in the suprachiasmatic nucleus (SCN) with the sleep-wake cycle (i.e., circadian misalignment) has been indicated as one of the causes for these health problems (Arendt, 2010; Salgado-Delgado et al., 2010b; Walters et al., 2003). The misalignment has been observed during a simulated shift in the sleep-wake cycle. For example, an abrupt 8-h delay in sleep time caused spontaneous misalignment between the phase of the circadian rhythm of plasma melatonin, which is a sensitive endocrine marker of the circadian clock under dim light conditions, and the sleep-wake cycle after the shift (Scheer et al., 2009). Another study showed that the phase of endocrine markers of the circadian clock was delayed by a few hours during the second day after delaying sleep time 8 h (Goichot et al., 1998). However, after 7.5 to 8.0 h of nocturnal sleep was maintained regularly for 8 days, the phase difference between the circadian rhythm of plasma melatonin and the sleep-wake cycle did not differ between shift workers and day workers (Wehrens et al., 2012).
In real-life shift work situations, diurnal variation in heart rate variability (HRV) has been examined in rotating shift workers. There is a close association between the circadian clock and regulation of the cardiovascular system because of projections from the SCN to the autonomic subdivision of the paraventricular nucleus of the hypothalamus (Scheer et al., 2001). HRV has been used extensively as a way to assess cardiac autonomic nervous system activity noninvasively in both laboratory and free living settings (Akselrod et al., 1981; Malliani et al., 1991; Saul, 1990). Previous studies have shown that the diurnal pattern of cardiac autonomic nervous system activity in shift workers estimated by HRV during the night shift was altered from that during the day shift (Freitas et al., 1997; Furlan et al., 2000). In a normal circadian pattern, nocturnal sympathetic nervous activity decreased compared with during the daytime (Boudreau et al., 2011; Vandewalle et al., 2007). In contrast, it increased compared with during the daytime on the day of the night shift, and it was higher on the day of the night shift than on the day shift (Freitas et al., 1997; Furlan et al., 2000; Mitani et al., 2006). However, it remains unclear whether the reported changes in the diurnal pattern of autonomic nervous system activity during the night shift represent normal alignment or misalignment between the circadian clock and the sleep-wake cycle in shift workers, because the diurnal pattern of HRV and the estimated autonomic nervous system activity can be affected by behavioral factors, including the sleep-wake cycle (Freitas et al., 1997), working time (Furlan et al., 2000; Mitani et al., 2006), physical activity, and food intake.
Whether rotating shift workers have misalignment between the circadian clock and the sleep-wake cycle during the day shift has also not been determined. The misalignment during the day shift might be a significant cause of health problems, especially for shift workers who engage in the day shift for most of their working time. Thus, we considered that an appropriate next step would be to compare diurnal variation in HRV and the sleep-wake cycle during the day shift between rotating shift workers and day workers while controlling behavioral factors. The phase relationships between the diurnal rhythm of HRV and the sleep-wake cycle for day workers might be used as normal control data.
Therefore, in this study, our purpose is to examine diurnal variation in HRV, the sleep-wake cycle, physical activity, and food intake during the day shift in rotating shift workers and fixed day workers and to compare diurnal variations in time and frequency domain variables in about 10-min segments of HRV over 24 h and the behavioral factors between the groups in real-life situations.
Materials and Methods
Subjects
Female nurses and caregivers working at a health care facility for the elderly in Choshi city, Chiba prefecture, Japan, participated in this cross-sectional study. The participants were 27 healthy female Japanese (14 day workers and 13 rotating shift workers) who did not take any over-the-counter or prescription medications for more than a month before the experiment. Their mean age, height, weight, and body mass index (BMI) were 40 years (range, 25-53 years), 159 cm (range, 150-170 cm), 57 kg (range, 44-79 kg), and 22.6 kg/m2 (range, 17.9-29.2 kg/m2), respectively. Inclusion criteria for study participants were working 40 to 46 hours a week (i.e., full-time worker) at least 5 months continuously in the current job and shift before the experiment; having no previous admission to hospital for cardiovascular disease; and having an age between 25 and 55 years. Their mean total duration of experience in their current job was 5.4 years (standard deviation [SD], ±5.1 years). The day workers worked a fixed day shift (0900-1800 h), and the rotating shift workers worked day (0900-1800 h) and night (1800-0900 h) shifts in a rotating 2-shift system. The average number of day and night shifts in the rotating shift was 3 days (range, 1-5 days) and 1 night (range, 0-2 nights) per week, respectively. Thus, the number of night shifts in the rotating shift was 4 or 5 nights per month. The measurements were conducted after 3.1 ± 1.6 days of day shift or day off after a single night shift. Each subject gave her written informed consent to participate after the test protocol, aim, and prospective effect on health had been fully described. All the study procedures were reviewed and approved by the Institutional Review Board of the National Institute of Occupational Safety and Health, Japan.
Protocol
Each subject was asked to undergo 24-h electrocardiograph (ECG) recording on the days of the day shift. Bipolar V5-lead ECG was recorded from the beginning of the day shift (0900 h) using a digital ambulatory ECG recorder (RAC3103, Nihon Kohden Corp, Tokyo, Japan). ECG signals were amplified and digitized at a sampling frequency of 250 Hz. Step counts were simultaneously recorded every 2 min using an electronic physical activity monitor (Lifecorder EX4, Suzuken Corp, Tokyo, Japan) (Togo et al., 2005). The subjects wore their activity monitor attached to a waist belt. The subjects were instructed to record in a diary their retiring and waking up times, start and end of times for work, and mealtimes. The total time in bed was calculated from recorded retiring and waking up times during off-duty hours (1800-0900 h). Sleep-wake identification during time in bed was conducted by means of data from Lifecorder using the algorithm proposed by Enomoto et al. (2009).
Foods and beverages and the amounts of each consumed on the days of the experiment were also reported through dietary records. The subjects were asked to provide details of all food and beverages consumed (e.g., ingredients, quantities, cooking methods, and whether homemade or readymade) and any nutritional supplements. At the end of the 24-h ECG recording, well-trained dietitians reviewed the dietary records for completeness and accuracy, and then the total energy intake during the recording was calculated using the Standard Food Composition Table published by the Science and Technology Agency of Japan (2005). Meals taken between 0500 and 1000 h, 1100 and 1500 h, and 1700 and 2200 h were defined as breakfast, lunch, and dinner, respectively (Yoshizaki et al., 2010). Furthermore, the ratios of energy intake for breakfast, lunch, and dinner to total energy intake were calculated.
All the subjects worked the day shift or took a day off on the day before the day of the experiment, and their routine work schedules and contents during the days of the experiment were identical. The subjects were instructed that they should refrain from smoking, ingesting alcohol and caffeine, and engaging in prolonged and/or strenuous exercise and should maintain habitual daily routines on the day before and the days of the experiment. The subjects were all studied during the follicular phase of their menstrual cycles (i.e., between 5 and 12 days after the self-reported onset of menses).
Time and Frequency Domain Analysis of Heart Rate Variability
The R-R interval (RRI) was derived from the ECG by using the first-derivative-based QRS detection method (Hamilton and Tompkins, 1986). All RRIs were scanned for extra or missing beats that could affect the results of the time and frequency domain analysis. The abnormal intervals were corrected by either the insertion (for missing beats) or the omission (for doubled or tripled beats) of beats. The number of beats corrected manually in this way was <0.5% of the total number of beats during the 24-h recording (about 100,000 beats).
Time (mean and SD of RRI) and frequency domain measures were calculated for approximately 10-min segments of HRV (600 beats) every 10 minutes. The HRV data of each segment were aligned sequentially to obtain an equally spaced sample using the mean RRI (DeBoer et al., 1984). After any linear trends were eliminated by linear regression, coarse graining spectral analysis was used for 10-time-shifted subsets of 512 beats to break down the total power into regular periodic and fractal components (Yamamoto and Hughson, 1991, 1994). The total power of the periodic component (TOT: >0.04 Hz), the integrated spectral power of periodic components in the low-frequency (LF: 0.04-0.15 Hz) and high-frequency (HF: >0.15 Hz) ranges, the ratio of HF power to TOT (HF nu), and the ratio of LF power to HF power (LF/HF) were calculated (Task Force, 1996). HF power or HF nu and LF/HF have been used to assess cardiac vagal tone (Akselrod et al., 1981; Malliani et al., 1991; Saul, 1990) and cardiac sympathovagal balance (Togo and Takahashi, 2009; Yamamoto and Hughson, 1991, 1994), respectively.
Double Cosinor Analysis
Cosinor analysis was performed on each subject’s data using the method of least squares regression to assess the timing and amplitude of the diurnal variation in selected HRV variables (mean and SD of RRI, LF power, HF power, HF nu, and LF/HF) or step counts for each subject after the HRV variables obtained or step counts totaled every 10 minutes were averaged for each 1-h bin for each subject (Tabata et al., 2000; Teicher and Barber, 1990), respectively. Since the duration of the daytime active period is longer than that of the nighttime quiescent period, traditional cosinor analysis was not suitable for analyzing step count and HRV variables (Massin et al., 2000), which were influenced not only by the circadian clock but also by behavioral factors (Scheer et al., 2001; Vandewalle et al., 2007). Therefore, we used double cosinor analysis, in which the simultaneous fit of two cosine functions was performed, one with a period of 24 h and the other with a period of 12 h (Krauchi and Wirz-Justice, 1994; Sakamoto et al., 2008). In brief, the cosine function was represented as y = M + A1 cos (ω1t + ϕ1) + A2 cos (ω2t + ϕ2), where M is the mesor, A1 is the amplitude of the 24-h period, A2 is the amplitude of the 12-h period, ω1 = 2π/24, ω2 = 2π/12, t is time, ϕ1 is the acrophase of the 24-h period, and ϕ2 is the acrophase of the 12-h period. According to this cosinor technique, two variables of the 24 h (amplitude and acrophase) and the 12 h (amplitude and acrophase) characterized patterns associated with the circadian and ultradian (12-h) rhythms in each of the HRV variables or step counts, respectively. The significance of the diurnal rhythm was tested by the zero-amplitude test (Nelson et al., 1979; Vandewalle et al., 2007). Compared with single cosinor analysis, double cosinor analysis can more flexibly address the fact that the duration of the waking period is approximately twice as long as that of the sleep period.
Statistical Analysis
A nonpaired t test or Mann-Whitney U test was used for comparison of continuous variables between the day workers and the shift workers, after the normal distribution of variables was confirmed. All the statistical analyses were performed with SPSS software (IBM SPSS 20.0 for Windows, SPSS Japan, Tokyo, Japan). A p value of <0.05 was considered statistically significant using a 2-tailed test.
Results
Figure 1 depicts double plots of ensemble-averaged 10-min step counts and HRV variables over 24 h for each group. Ensemble-averaged fitted curves calculated from double cosinor analysis are also shown for each group. While the average fitted curve of 10-min step counts reached the highest point (zenith) at 1240 h and nadir at 0130 h, respectively, for both day workers and shift workers, times for zenith and/or nadir in selected time and frequency domain variables for HRV were delayed in shift workers compared with day workers (Figure 1). Table 1 shows group means of means for 10-min HRV data over 24 h. There were not any significant differences in means of step counts or HRV variables over 24 h between the groups (Table 1).
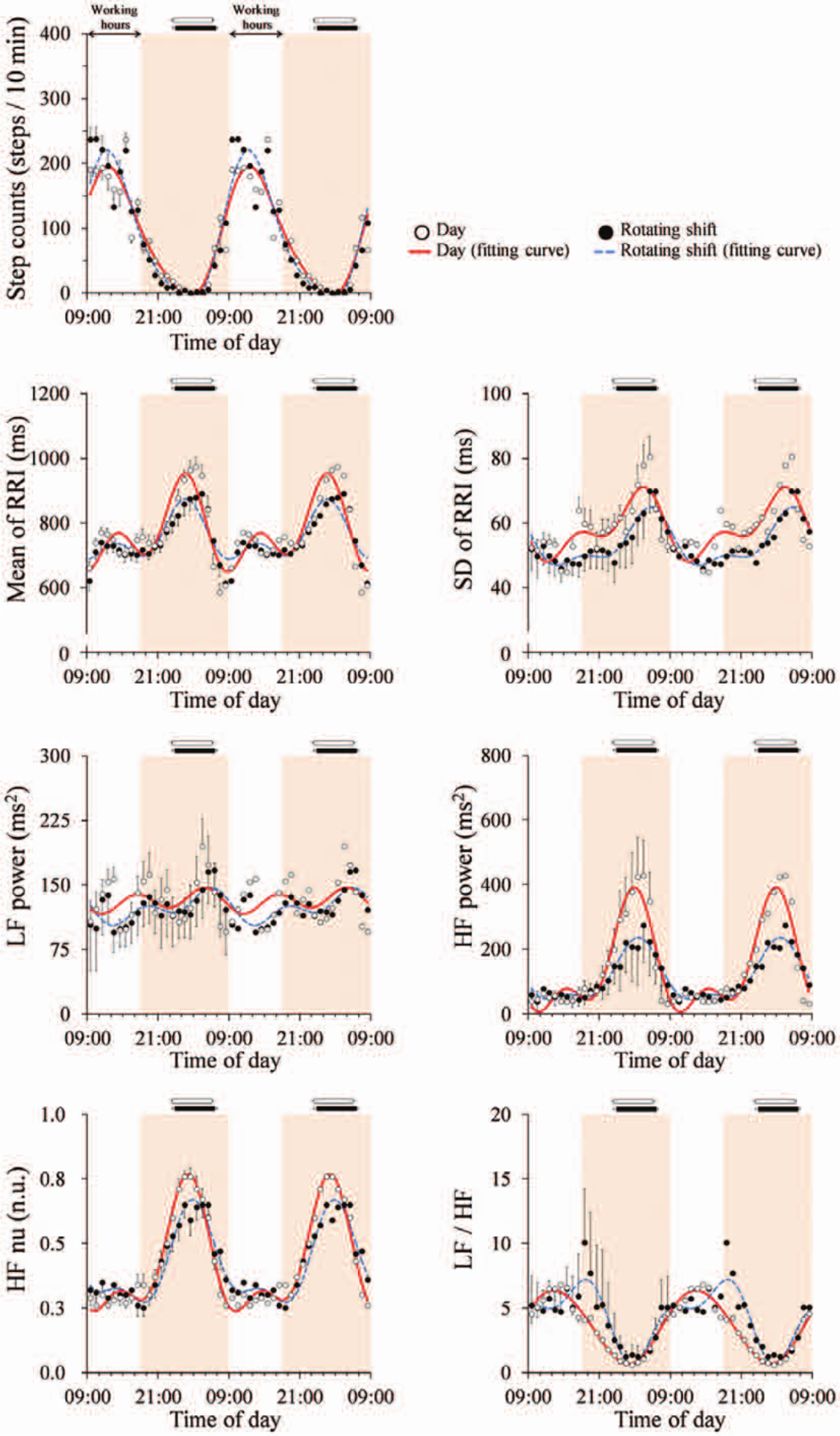
Ensemble-averaged 10-min step counts and heart rate variability variables during the day shift over 24 h for day workers (open circles, n = 14) and rotating shift workers (filled circles, n = 13). Values are shown as mean ± SE. Ensemble-averaged fitted curves by using double cosinor analysis for day workers (solid line) and shift workers (dashed line) are also depicted. Working hours (0900-1800 h) are indicated by the white zone. Horizontal white and black bars indicate sleeping hours in day workers and shift workers, respectively. Data are double-plotted to better visualize rhythmicity. LF = low frequency (0.04-0.15 Hz); HF = high frequency (>0.15 Hz); HF nu = the ratio of HF power to total power (>0.04 Hz); LF/HF = the ratio of LF power to HF power.
General characteristics of subjects and measured variables during experimental days on the day shift.
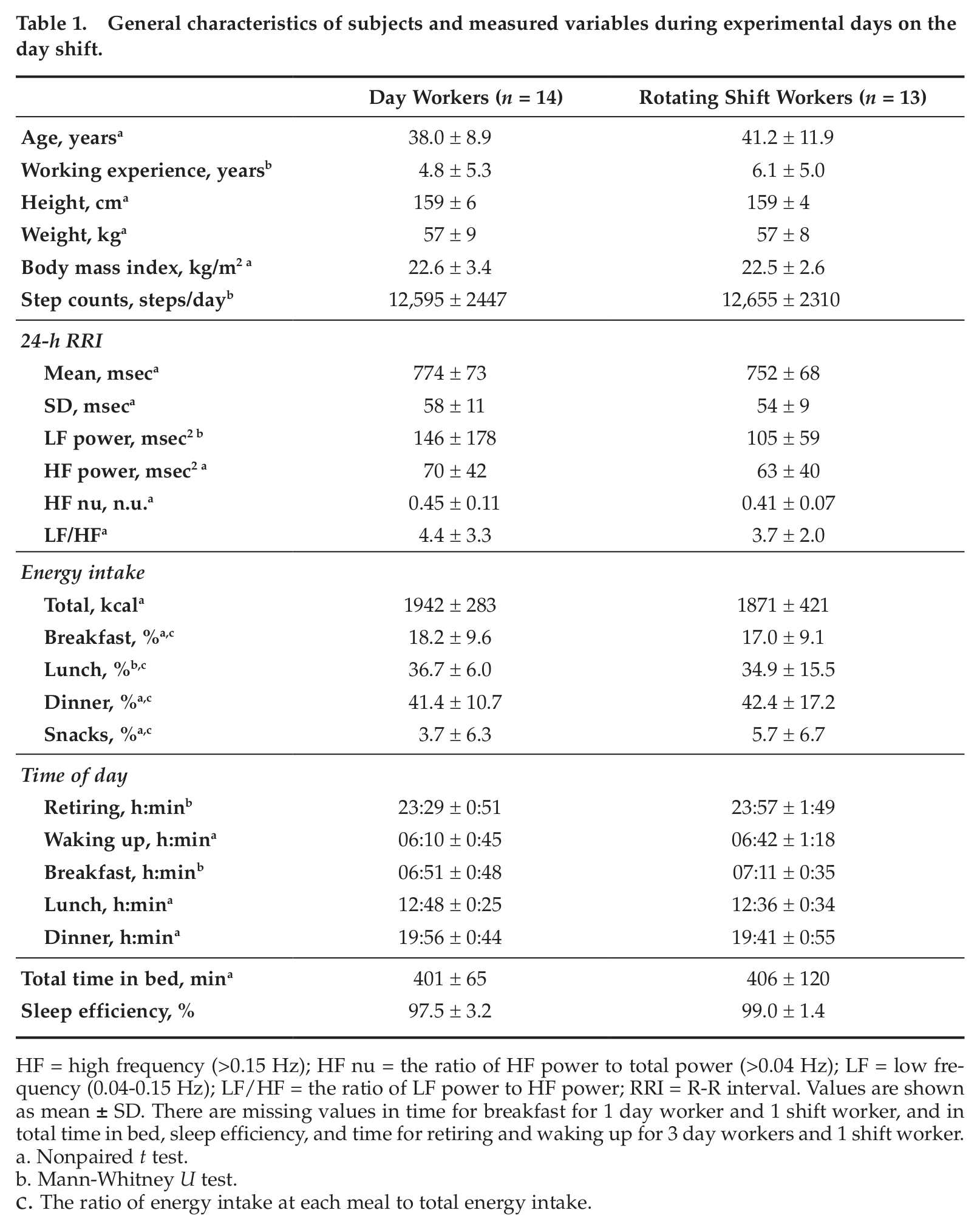
HF = high frequency (>0.15 Hz); HF nu = the ratio of HF power to total power (>0.04 Hz); LF = low frequency (0.04-0.15 Hz); LF/HF = the ratio of LF power to HF power; RRI = R-R interval. Values are shown as mean ± SD. There are missing values in time for breakfast for 1 day worker and 1 shift worker, and in total time in bed, sleep efficiency, and time for retiring and waking up for 3 day workers and 1 shift worker.
Nonpaired t test.
Mann-Whitney U test.
The ratio of energy intake at each meal to total energy intake.
For the fitted curves of 10-min step counts for each subject, zenith or nadir, time at zenith or nadir, and amplitude (50% of the zenith-nadir difference) did not differ between the groups, although the mesor was significantly (p < 0.05) higher in rotating shift workers than in day workers (Table 2, Figure 2). Meanwhile, there were significant differences in the fitted curves of 10-min HRV variables for each subject (Figure 2). The time at zenith for SD of RRI, LF power, HF power, and LF/HF was significantly delayed in rotating shift workers compared with day workers (Figure 2). The time at nadir for HF power and HF nu was significantly delayed in rotating shift workers compared with day workers (Figure 2).
Variables of fitted curves for 10-min step counts and heart rate variability over 24 h.
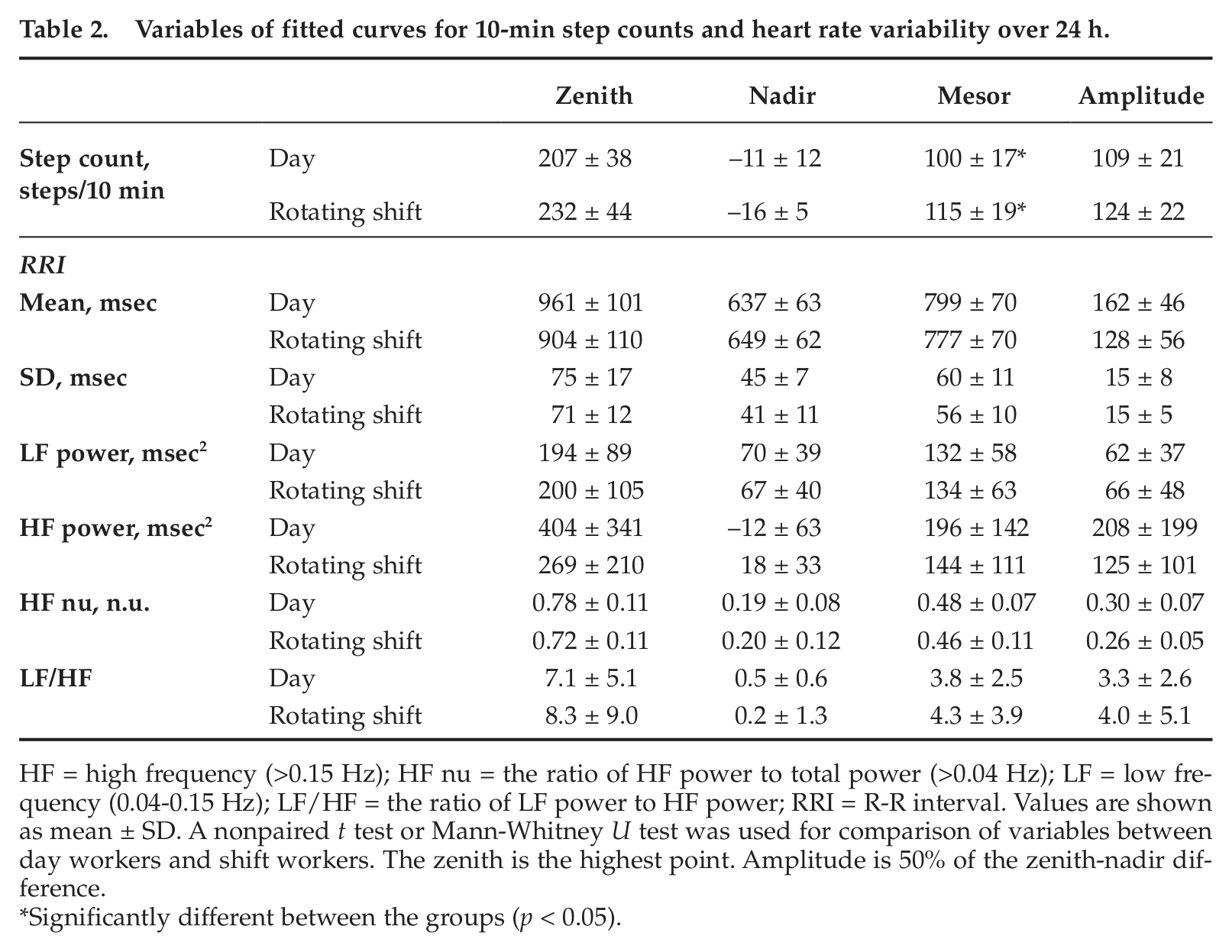
HF = high frequency (>0.15 Hz); HF nu = the ratio of HF power to total power (>0.04 Hz); LF = low frequency (0.04-0.15 Hz); LF/HF = the ratio of LF power to HF power; RRI = R-R interval. Values are shown as mean ± SD. A nonpaired t test or Mann-Whitney U test was used for comparison of variables between day workers and shift workers. The zenith is the highest point. Amplitude is 50% of the zenith-nadir difference.
Significantly different between the groups (p < 0.05).
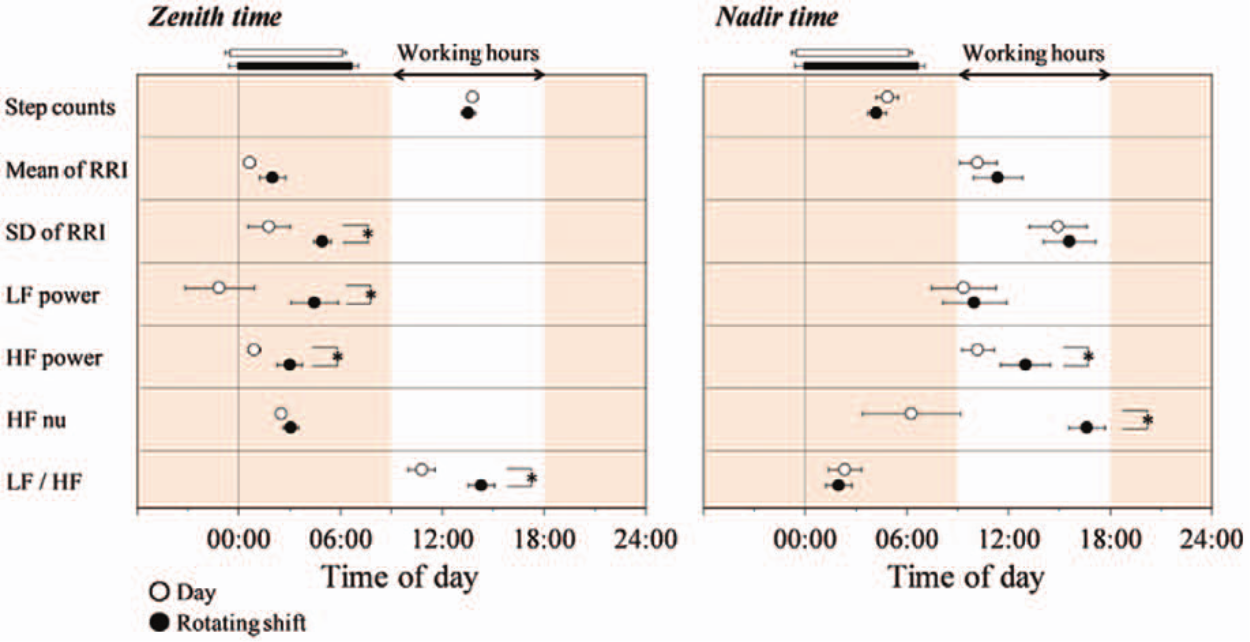
Time at zenith (the highest point) and nadir of fitted curves for step counts and heart rate variability over 24 h for day workers (open circles, n = 14) and rotating shift workers (filled circles, n = 13). Values are shown as mean ± SE. Working hours (0900-1800 h) are indicated by the white zone. Horizontal white and black bars indicate sleeping hours in day workers and shift workers, respectively. *Significantly different between the groups (p < 0.05).
We evaluated 24-h and 12-h period variations for step counts and HRV variables calculated by double cosinor analysis for each group (Table 3, Figure 3). Table 3 shows group means of fitting parameters for each variable. While the acrophases of the two periods for step counts did not differ significantly between the groups, the acrophases of the 24-h period for HRV variables were delayed by 1.3 to 5.5 h in rotating shift workers compared with those in day workers, and their differences in HF power (2.5 h), HF nu (1.3 h), and LF/HF (2.2 h) reached a significant level (p < 0.05) (Figure 3). The amplitude for the 24-h period (Table 3) and any variables for the 12-h period in step counts and HRV variables (Table 3, Figure 3), except for the means of RRI and HF power for the 12-h period, did not differ significantly between the groups (Table 3, Figure 3).
Amplitude of 24-h and 12-h period cosine waves for 10-min step counts and heart rate variability over 24 h.
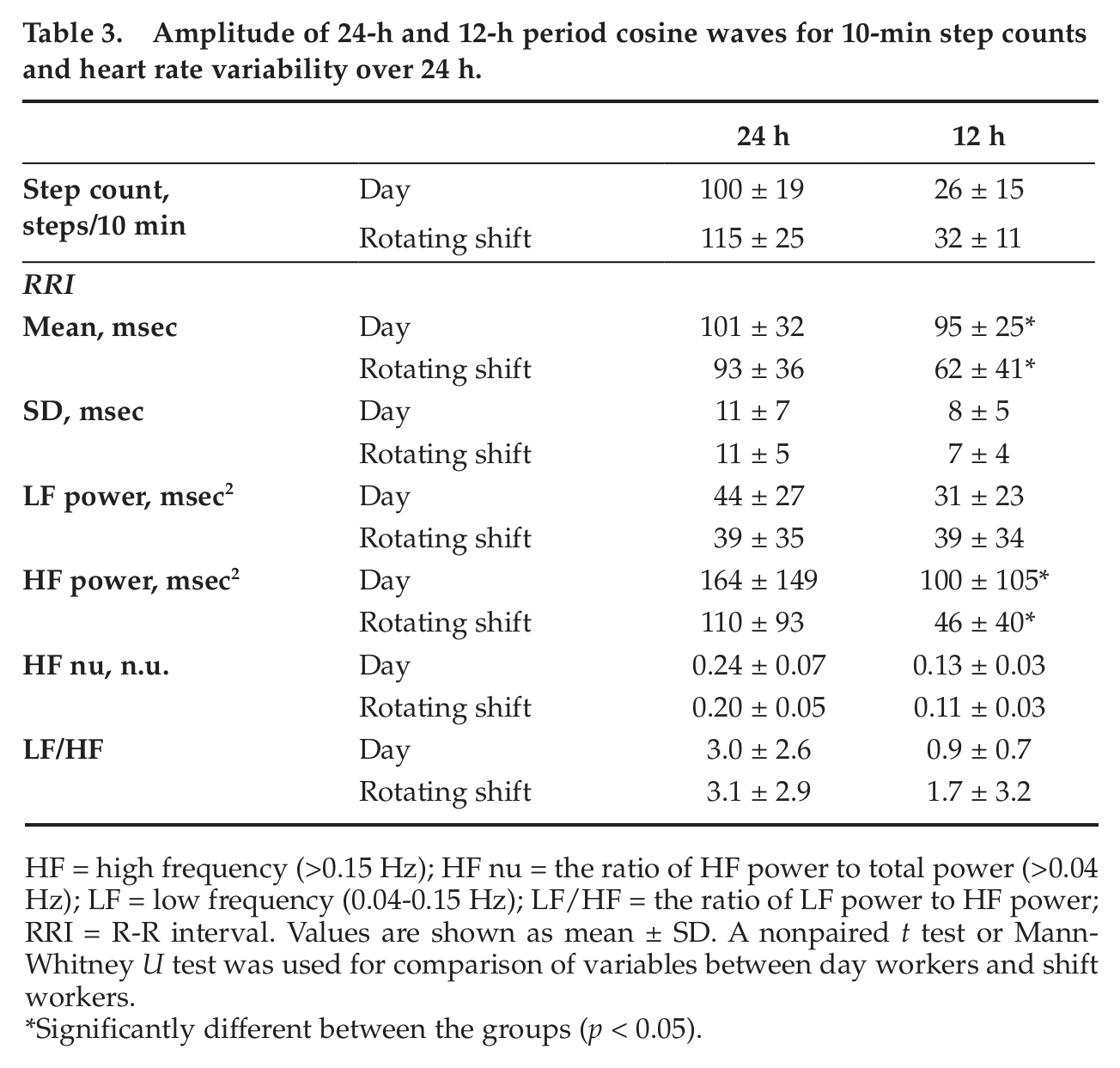
HF = high frequency (>0.15 Hz); HF nu = the ratio of HF power to total power (>0.04 Hz); LF = low frequency (0.04-0.15 Hz); LF/HF = the ratio of LF power to HF power; RRI = R-R interval. Values are shown as mean ± SD. A nonpaired t test or Mann-Whitney U test was used for comparison of variables between day workers and shift workers.
Significantly different between the groups (p < 0.05).
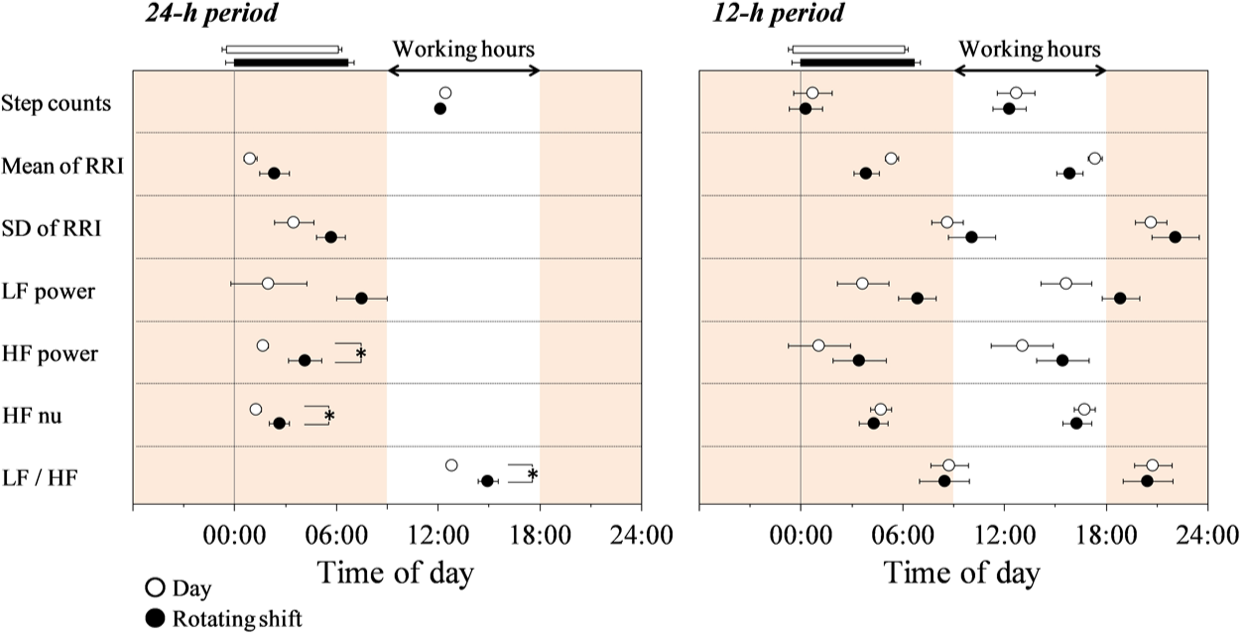
Acrophases of 24-h and 12-h period cosine waves for step counts and heart rate variability for day workers (open circles, n = 14) and rotating shift workers (filled circles, n = 13). Values are shown as mean ± SE. Acrophase (rad) is converted to phase (h) from midnight. Working hours (0900-1800 h) are indicated by the white zone. Horizontal white and black bars indicate sleeping hours in day workers and shift workers, respectively. *Significantly different between the groups (p < 0.05).
On the days of the experiment, retiring time, waking up time, total time in bed, sleep efficiency (i.e., the percentage of the total time asleep after retiring time relative to the total time in bed), amount of energy intake, and meal times did not differ between the groups (Table 1). Times of sunrise (range, 0511-0600 h) and sunset (range, 1718-1808 h) on the days of the experiment also did not differ between the groups. Subjective level of stress was obtained from 8 day workers and 8 shift workers using a visual analogue scale (0-20) on an electronic diary. The levels at 0900 h, 1200 h, 1500 h, and 1800 h on the experimental day did not differ between day workers and shift workers (8.5 ±7.1 and 7.7 ± 3.1, 11.0 ± 1.7 and 9.2 ± 4.1, 12.8 ± 3.1 and 9.2 ± 5.2, and 10.9 ± 6.6 and 9.4 ± 2.7, respectively).
Discussion
In this study, we have compared diurnal variations in cardiac autonomic nervous system activity during the day shift between day workers and rotating shift workers. To do this, we used double cosinor analysis to obtain 24-h and 12-h period variations in time and frequency domain variables of HRV and physical activity. As a result, the acrophases of the 24-h period for HF power, HF nu, and LF/HF were delayed in shift workers compared with those in day workers, although the acrophase and the amplitude of the 24-h period for step counts did not differ between the groups (Figure 3). Meanwhile, the acrophases of the 12-h period for these variables did not differ between the groups (Figure 3). There were not any significant correlations between the mesor of step counts and the acrophases of the 24-h period for parameters of HRV (r = −0.045 to 0.397, p > 0.05) or the mean of LF power and LF/HF over 24 h (r = −0.151 and 0.079, respectively, p > 0.05). In addition, there were not any significant differences in the sleep-wake pattern, the total step counts, the total energy intake, the ratios of energy intake for breakfast, lunch, and dinner to total energy intake, and each mealtime between the groups (Table 1), indicating that the effects of relatively strong behavioral factors that can modify the circadian variations in HRV (Furlan et al., 2000; Scheer et al., 2001) or sympathetic nervous activity did not differ between the groups. We measured the 24-h ECG after 3.1 ± 1.6 days of day shift or day off after a night shift. Therefore, our results indicate that there is a possibility of an abnormal phase angle between the diurnal 24-h variation in cardiac autonomic nervous system activity and the sleep-wake cycle during the day shift in shift workers for at least 3 days after changing the shift.
For day workers, the acrophases of the 24-h period for mean RRI, LF, and HF nu were around 0000 h, and the acrophase of LF/HF was around 1200 h (Figure 3). A previous study reported that acrophases of the rhythm of RRI, LF and HF, and LF/HF over 24 h during an ultradian sleep-wake cycle procedure for healthy young subjects who were not night shift workers were also around 0000 and 1200 h, respectively (Boudreau et al., 2011). Considering also that heart rate in SCN-intact, but not SCN-lesioned, rats had a clear circadian rhythm and that the diurnal pattern of the HRV could be mediated through projections from the SCN to the autonomic subdivision of the paraventricular nucleus of the hypothalamus (Scheer et al., 2001), our results also suggest that the phase of the circadian variation in cardiac autonomic regulation and/or the circadian clock differs between the groups. Recent laboratory studies have indicated that an abnormal phase angle between the circadian clock and the sleep-wake cycle (approximately 4.0-6.0 h) can have adverse effects on metabolism (e.g., glucose, insulin, triglyceride, and leptin) (Nguyen and Wright, 2009; Ribeiro et al., 1998; Scheer et al., 2009) and might cause health problems such as cardiovascular disease and metabolic disorders (Arendt, 2010; Scheer et al., 2009).
Most previous studies on shift work tolerance have focused on abnormalities during the night shift in order to examine physiological maladaptation to night work (Kantermann et al., 2010). Supplementary to this, our results indicate the possibility that shift workers who engage in night shifts 1 or 2 times per week have a risk of cardiometabolic disorders due to incomplete adaptation to the day shift. However, it has been shown that it is possible for the phase angle between the circadian clock and the sleep-wake cycle for shift workers to return to normal after keeping a 7.5- to 8.0-h nocturnal sleep regularly for 8 days (Wehrens et al., 2012). The level of 24-h averaged autonomic nervous system activity for shift workers was not found to be abnormal (Table 1), which is consistent with the results of a previous study (Wehrens et al., 2012). Thus, a quick improvement in the abnormal phase angle during the day shift may be important in preventing health problems in such rotating shift workers.
Recently, it has been reported that light exposure (Scheer et al., 1999; Ueyama et al., 1999), physical activity (Van Reeth et al., 1994), and dietary behavior (Fuller et al., 2008; Mieda et al., 2006; Salgado-Delgado et al., 2010a) are zeitgebers for the circadian clock. Thus, night work causes a phase shift in the circadian clock (Goichot et al., 1998; Sack et al., 1992). In addition, the circadian misalignment might be partly caused by social schedule (e.g., the working time of the day shift). A discrepancy between biological and social time is described as “social jetlag,” which has been indicated as a risk factor for developing chronic sleep debt (Wittmann et al., 2006), obesity (Roenneberg et al., 2012), and depression (Levandovski et al., 2011) even in adults who do not engage in night shift work. Future study is needed to examine whether controlling such zeitgebers and/or working times improves the misalignment rapidly and prevents health problems in shift workers.
There are some limitations to our study. First, behavioral factors that can modify the circadian pattern of cardiac autonomic nervous system activity could not be completely eliminated in our free living conditions, although relatively strong behavioral factors did not differ between the groups. Our findings need confirmation with a larger sample size, and future studies should use more sensitive measures of circadian phase, such as the acrophase of 24-h melatonin concentration or the dim light melatonin onset test. Second, the effect of the rotating shift system remains unclear. Morikawa et al. (2005) reported that people working on a rotating 2-shift system had a higher risk of increased BMI and type 2 diabetes compared with people working on a rotating 3-shift system, indicating that the type of rotating shift system might affect the degree of phase shift in the circadian clock and diurnal variation in the cardiac autonomic nervous system activity. Third, self-reported data on the onset of menses and meals could be inherently biased.
In conclusion, shift workers had an abnormal phase angle between diurnal 24-h variation in the cardiac autonomic nervous system activity and the sleep-wake cycle during the day shift. Considering that relatively strong behavioral factors such as the sleep-wake cycle, physical activity, and food intake, which can modify diurnal variation in HRV, did not differ significantly between day and shift workers, the phase of the circadian variation in the cardiac autonomic nervous system activity or the circadian clock during days on the day shift for shift workers may misalign with the sleep-wake cycle. Future studies should examine how to adjust the circadian clock to the day shift quickly after the night shift. This may be of importance in preventing health problems in rotating shift workers.
Footnotes
Acknowledgements
The authors thank Kanetaka Watanabe, the director of the Medical Corporation of Doaikai, and the subjects for their cooperation in the present study. This study was supported by a grant from the Japan Society for the Promotion of Science (Grant-in-Aid for start-up 20800085, Scientific Research (C) 22500690, Young Scientists (B) 23700924).
Conflict of Interest Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
