Abstract
Circadian disruption is a common by-product of modern life. Although jet lag and shift work are well-documented challenges to circadian organization, many more subtle environmental changes cause circadian disruption. For example, frequent fluctuations in the timing of the sleep/wake schedule, as well as exposure to nighttime lighting, likely affect the circadian system. Most studies of these effects have focused on nocturnal rodents, which are very different from diurnal species with respect to their patterns of light exposure and the effects that light can have on their activity. Thus, the authors investigated the effect of nighttime light on behavior and the brain of a diurnal rodent, the Nile grass rat. Following 3 weeks of exposure to standard light/dark (LD; 14:10 light [~150 lux] /dark [0 lux]) or dim light at night (dLAN; 14:10 light [~150 lux] /dim [5 lux]), rats underwent behavioral testing, and hippocampal neurons within CA1, CA3, and the dentate gyrus (DG) were examined. Three behavioral effects of dLAN were observed: (1) decreased preference for a sucrose solution, (2) increased latency to float in a forced swim test, and (3) impaired learning and memory in the Barnes maze. Light at night also reduced dendritic length in DG and basilar CA1 dendrites. Dendritic length in the DG positively correlated with sucrose consumption in the sucrose anhedonia task. Nighttime light exposure did not disrupt the pattern of circadian locomotor activity, and all grass rats maintained a diurnal activity pattern. Together, these data suggest that exposure to dLAN can alter affective responses and impair cognition in a diurnal animal.
Keywords
Biological rhythms are highly adaptive, aligning individuals to daily fluctuations in the external environment, as well as synchronizing internal homeostatic processes. The master mammalian circadian clock is located in the suprachiasmatic nuclei (SCN) and regulates timing of subordinate oscillators throughout the central nervous system and periphery. External lighting is important in synchronizing the circadian system and maintaining daily temporal organization. Prior to the widespread adoption of electrical lighting, individuals’ biological clocks were entrained to a consistent pattern of light and dark; in contrast, modern light exists in several temporal patterns. Moreover, shift work, transmeridian travel, and inconsistent sleep schedules have rapidly increased during the past century. Because the change in nighttime lighting has occurred so rapidly in terms of evolutionary history, it is likely that significant physiological and ecological perturbations have resulted. For example, disruption of the circadian system results in adverse health conditions such as heart disease (Ha and Park, 2005), cancer (Schernhammer et al., 2001; Davis and Mirick, 2006), and metabolic dysfunction (Reiter et al., 2011).
Circadian disruption and light at night are implicated in impaired cognition. Rats housed in constant illumination perform poorly in the Morris water maze (Ma et al., 2007; Ling et al., 2009; Fujioka et al., 2011). Constant light causes tau hyperphosphorylation, increased expression of endoplasmic reticulum (ER) stress-related proteins, thinner synapses, and increased superoxide dismutase and monoamine oxidase (Ling et al., 2009). Similarly, rats housed in constant light display impaired spatial learning in the Morris water maze with accompanying changes in long-term depression in the CA1 area of the hippocampus (Ma et al., 2007). Constant light also impairs learning and memory in mice, which may be related to decreased neurogenesis (Fujioka et al., 2011). Furthermore, mice undergoing experimental jet lag decrease neurogenesis and have prolonged deficits in learning and memory as evaluated in a conditioned place preference task (Gibson et al., 2010).
In addition to influencing learning and memory, circadian disruption changes mood (Monteleone et al., 2011). Seasonal lighting, abnormalities in the circadian clock (Benedetti et al., 2008), and sleep disorders are associated with depression (Bunney and Bunney, 2000). Constant light alters anxiety and depressive-like behaviors in mice (Fonken et al., 2009; Martynhak et al., 2011). Furthermore, Siberian hamsters exposed to dim light during the dark phase increase depressive-like responses and have reduced spine density in the CA1 area of the hippocampus (Bedrosian et al., 2011).
In all of these studies, only nocturnal rodents were used. Both circadian influences on behavior and masking effects of light are very different in nocturnal and diurnal species. Thus, in the present experiment, we examined behavioral and brain responses of diurnal male Nile grass rats to dim light at night (dLAN).
Materials and Methods
Animals
Male grass rats (Arvicanthis niloticus) used in this study were bred at The Ohio State University from a wild stock obtained from LS. Grass rats were bred under a standard light-dark (LD) cycle (14:10 light [~150 lux] /dark [0 lux]). All animals were provided food (ProLab RMH 2000, LabDiet) and filtered tap water ad libitum. Experimental grass rats were weaned between 21 and 24 days of age and housed with same-sex siblings in polypropylene cages (40 cm × 20 cm × 20 cm) with straw bedding. Colony rooms were maintained at a temperature of 20 ± 4 °C and a relative humidity of 50% ± 10%.
At 10 weeks of age, grass rats were singly housed, randomly assigned a number, and either maintained in LD or placed in dLAN (14:10 light [~150 lux]/ dim [~5 lux]). Blood samples were collected at zeitgeber time (ZT) 6 via retro-orbital bleed after 2 weeks for corticosterone analysis, and 1 week later, grass rats underwent behavioral testing to assess cognitive and affective behaviors. Testing occurred in the following order: Barnes maze, sucrose anhedonia, and forced swim test. The sucrose anhedonia test occurred between ZT 8 and 13; all other tests were conducted between ZT 1 and 6. All experimental procedures followed The Ohio State University Institutional Animal Care and Use Committee and in accordance with the recommendations of the National Institutes of Health and the Guide for the Care and Use of Laboratory Animals.
Corticosterone Radioimmunoassay
Retro-orbital blood samples (~020 mL) were collected from grass rats anesthetized with isoflurane vapors for radioimmunoassay (RIA) of corticosterone concentrations, prior to the onset of behavioral testing. Blood samples were centrifuged at 4 °C f or 30 min at 3.3 g, and plasma aliquots were aspirated and stored in sealable polypropylene microcentrifuge tubes at −80 °C until assayed for corticosterone concentrations using RIA. Total plasma corticosterone concentrations for grass rats were determined in duplicate in an assay using an ICN Diagnostics 125I double antibody kit (Costa Mesa, CA). The high and low limits of detectability of the assay were ~1000 and 5 ng/mL, respectively. All procedures followed those described by the manufacturer guidelines.
Behavioral Testing
Barnes maze
The Barnes maze is a brightly lit arena with 18 evenly spaced holes, one leading to a dark box and the others blocked off with black inserts (Sunyer et al., 2007). On the first day, animals were acclimated to the maze; a bright light and loud fan were turned on as they were guided from the center of the maze to the target hole. After entering, the bright light and fan were turned off, and the grass rats were left undisturbed for 30 sec. Animals then underwent 4 days of training consisting of three 90-sec trials separated by 10-min intervals in the home cage. One day after the last training trial, animals were given a 60-sec probe trial in which the escape box was blocked off. Latency to find the target hole and number of errors were scored during all trials.
Sucrose anhedonia
Consumption of a 2% sucrose solution between ZT 8 and ZT 13 was recorded in all grass rats to measure sucrose anhedonia (Willner et al., 1992). Before presentation of the sucrose solution, grass rats were administered water in modified water bottles for 3 consecutive days, to control for novelty of the bottles. The bottles were weighed before and after the 5-h sample time; the next day, animals were provided a choice between a 2% sucrose solution and water. Sucrose consumption was normalized to water consumption.
Forced swim test
To assess depressive-like responses, grass rats were placed in ~17 cm water (22 ± 1 °C), within an opaque, cylindrical tank (diameter = 24 cm, height = 53 cm). Swimming behavior was videotaped for 5 min and scored by a condition-blind observer with the Observer software (Noldus Corp, Leesburg, VA). Latency to float and time spent floating served as dependent measures; both are used in rodents, including grass rats, to assess depressive-like response (Porsolt et al., 1977; Ashkenazy-Frolinger et al., 2010).
Locomotor activity analyses
Following behavioral testing, grass rats were implanted intraperitoneally (i.p.; under sterile conditions) with telemeters (PDT-4000; Minimitter, Bend, OR) while under isoflurane anesthesia. Surgical wounds were treated topically with Betadine (Sigma Chemical, St. Louis, MO) to discourage infection, and grass rats were injected (i.p.) with buprenorphine (0.1 mg/kg; Sigma Chemical) in sterile saline to alleviate pain during recovery. Following surgery, grass rats were placed in a clean cage, which was placed on a receiver (Minimitter) connected to a computer. Receivers collated emitted body temperature and movement activity frequencies continuously over 30-min intervals and converted them to raw data based on preprogrammed calibration curves for each transmitter.
Hippocampal Morphology
Grass rats were killed between ZT 3 and ZT 5, and brains were removed and processed for Golgi impregnation using the FD Rapid GolgiStain Kit (FD NeuroTechnologies, Inc., Ellicott City, MD) according to the manufacturer’s instructions. Brains were sliced at 100 µm, thaw mounted onto gelatin-coated slides counterstained with cresyl violet (Sigma), dehydrated, and coverslipped. Brains were assessed for hippocampal cell morphology and spine density in the dentate gyrus (DG), CA1, and CA3 using a Nikon E800 brightfield microscope. Tracings were done with Neurolucida software (MicroBrightField, Burlington, VT) at a magnification of 200× for neuronal morphology and 1000× for spine density. Six representative neurons were selected per area, per animal. Whole cell traces were analyzed using NeuroExplorer software, for cell body size and perimeter, as well as dendritic length (MicroBrightField). Sholl analysis defines dendritic complexity by the number of dendritic branch points at fixed intervals from the cell bodies (Sholl, 1956) and was conducted on apical and basilar dendrites. From each neuron, >20 µm was selected in the apical and basilar areas, respectively (except in the DG, where granule cells lack bidirectional projections). All spine segments selected were at least 50 µm distal to the cell body. Spine density (spines per 1 µm) was calculated for each trace and averaged per cell, per area, and per animal.
Statistical Analyses
Comparisons for behavior analyses and hormone concentrations were conducted using a one-way analysis of variance (ANOVA). Neuronal characteristics and spine densities were averaged per animal and then analyzed using a one-way ANOVA. Sholl analyses were also averaged per animal and then analyzed using a repeated-measures ANOVA with lighting condition as the between-subject factor and distance from the cell body as the within-subject factor. For each Barnes maze session (1-4), latencies and error rates, respectively, were averaged per session for each grass rat; data were subject to repeated-measures ANOVA (lighting condition as the between-subject factor and session as the within-subject factor). Fourier analysis was used to determine whether locomotor activity was rhythmic and followed 24-h periodicity using Clocklab software from Actimetrics (Wilmette, IL). Grass rats were considered rhythmic when the highest peak occurred at ~1 cycle per day with an absolute power of at least 0.005 mV/Hz as previously described (Kriegsfeld et al., 2008). FFT power values for 0.083 cycles per day were compared between lighting conditions by one-way ANOVA. Percentage of daytime activity and total daily activity were also analyzed by one-way ANOVA. The above statistical analyses were conducted with StatView software (v. 5.0.1; SAS Institute, Cary, NC). Nonlinear regression analysis was used in GraphPad Prism software (v. 4; GraphPad Software, La Jolla, CA). In all cases, differences between group means were considered statistically significant if p ≤ 0.05.
Results
Somatic Measures
There were no differences in body or reproductive tissue mass (p > 0.05; data not shown).
Activity Analysis
Regardless of lighting conditions, grass rats displayed equivalent levels of total daily locomotor activity and locomotor activity rhythms. Furthermore, both grass rats maintained in LD and dLAN showed a diurnal pattern of activity that is typical of this species. There were no differences in onset, offset, peak, or amplitude (p > 0.05; Fig. 1A, B).

Representative actograms from a grass rat housed under (A) a standard light/dark cycle or (B) dim light at night.
Learning and Memory
Grass rats exposed to dLAN decreased learning abilities in the Barnes maze. dLAN grass rats had an increased latency to reach the target hole and a higher error rate as compared with grass rats housed under dark nights. There was a main effect of lighting condition, such that dLAN increased latency to reach the target hole over the course of training trials (F 1, 42 = 6.064, p < 0.05; Fig. 2A). Post hoc analysis revealed dLAN increased latency to reach the target hole on days 2 and 4 of the training trials (p < 0.05). Furthermore, there was a main effect of lighting condition with respect to error rate; grass rats housed under dLAN increased errors over the course of training trials (F 1, 42 = 8.184, p < 0.05; Fig. 2B). On day 2 of the training trials, dLAN grass rats showed an increased error rate compared with grass rats housed under standard LD conditions (p < 0.05).
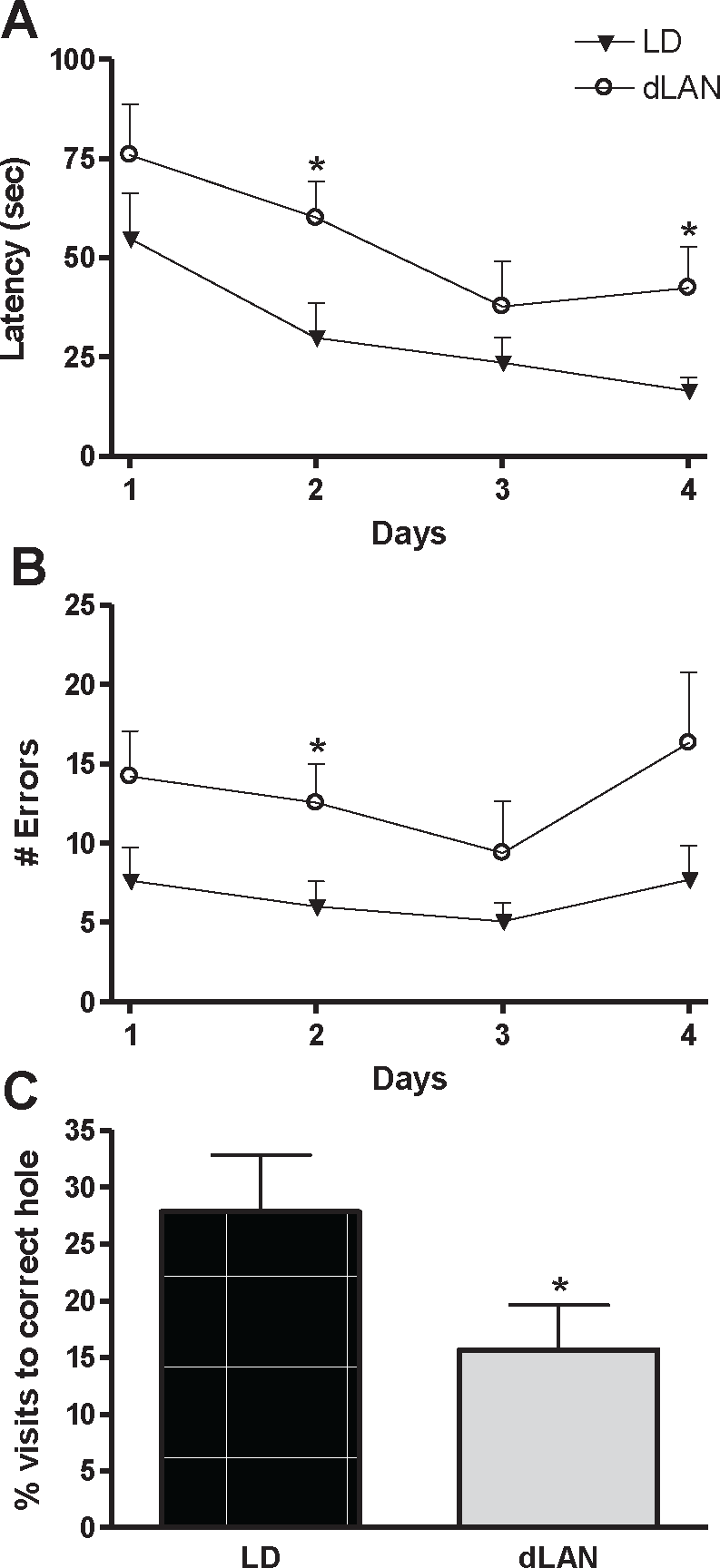
Dim light at night impaired spatial learning and memory in the Barnes maze. (A) Latency to reach the target hole in the Barnes maze by day (average of 3 trials). (B) Number of visits to false holes by session. (C) Relative visits to the target hole versus false holes during the probe trial. LD = light/dark; dLAN = dim light at night.
Twenty-four hours after the final training session, memory retention was assessed with a probe trial. During the probe trial, the target box was removed and behavior on the maze was recorded for 60 sec. dLAN grass rats spent a lower percentage of time investigating the target hole compared with LD grass rats; these data indicate decreased memory retention (F 1, 14 = 7.084, p < 0.05; Fig. 2C). Our lab has previously found no differences in spontaneous locomotor activity between grass rats housed under dLAN and LD in a 5-min brightly lit open field task (unpublished observations). This indicates Barnes maze differences are due to changes in learning and memory and not differences in motivation when exposed to a brightly lit open space.
Affective Responses
Depressive-like responses were evaluated using a sucrose anhedonia and forced swim task. Grass rats exposed to dLAN increased depressive-like responses in the sucrose anhedonia test. One grass rat was excluded from analyses because of a leaky water bottle. dLAN grass rats reduced consumption of a sucrose solution demonstrating an anhedonic-like response (F 1, 15 = 4.711, p < 0.05; Fig. 3A).
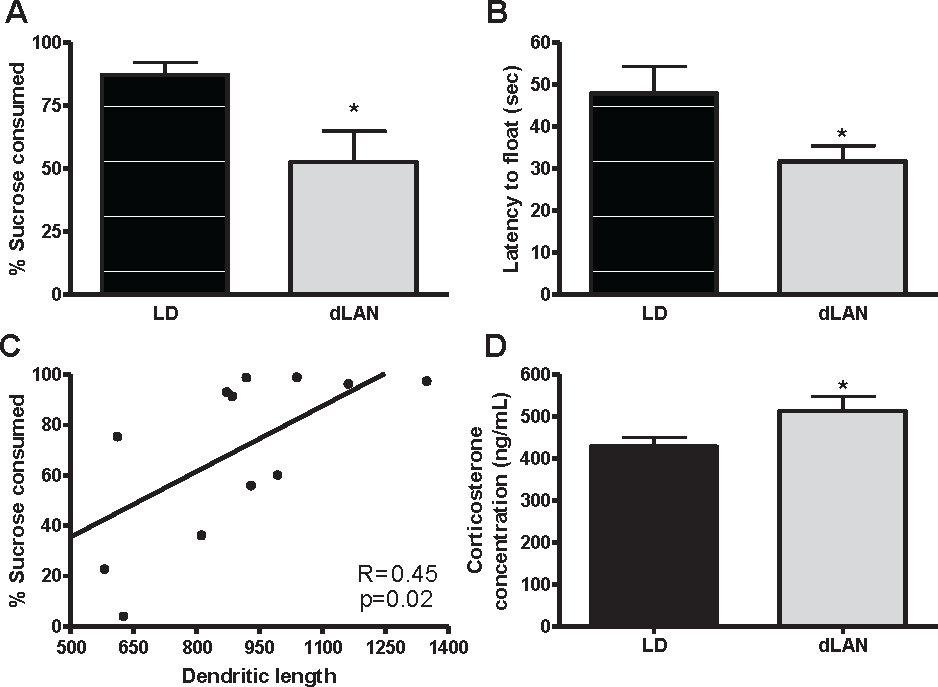
Dim light at night increased depressive-like responses and elevated corticosterone concentrations in male Nile grass rats. (A) Amount of sucrose solution versus total liquid consumed in a sucrose anhedonia task. (B) Latency to first float the forced swim test. (C) Correlation between percentage of sucrose consumed and dendritic length in the dentate gyrus of the hippocampus. (D) Corticosterone concentrations after 2 weeks in lighting conditions. Data are expressed as mean ± standard error of the mean (SEM). *p < 0.05 between groups. LD = light/dark; dLAN = dim light at night.
Grass rats exposed to dLAN also increased behavioral despair in the forced swim task. dLAN grass rats reduced latency to first float, indicating they more rapidly reach a state of behavioral despair (F 1, 16 = 4.774; p < 0.05; Fig. 3B). No differences, however, were observed between groups with respect to float duration, which is the primary depressive-like response evaluated in the forced swim test (p > 0.10).
Hippocampal Neuronal Morphology
Exposure to dLAN is associated with changes in neuronal morphology in the CA1 and DG regions of the hippocampus. Rats housed under dLAN reduced dendritic length in the DG and CA1 basilar dendrites (F 1, 11 = 4.875, 7.357, respectively; p < 0.05; Fig. 4). Furthermore, there was a positive association between dendritic length in the dentate gyrus and sucrose consumption in the sucrose anhedonia test (r = 0.455, p < 0.05; Fig, 3C). Groups did not differ with respect to spine density, cell body area, or cell body perimeter in any area (Table 1).
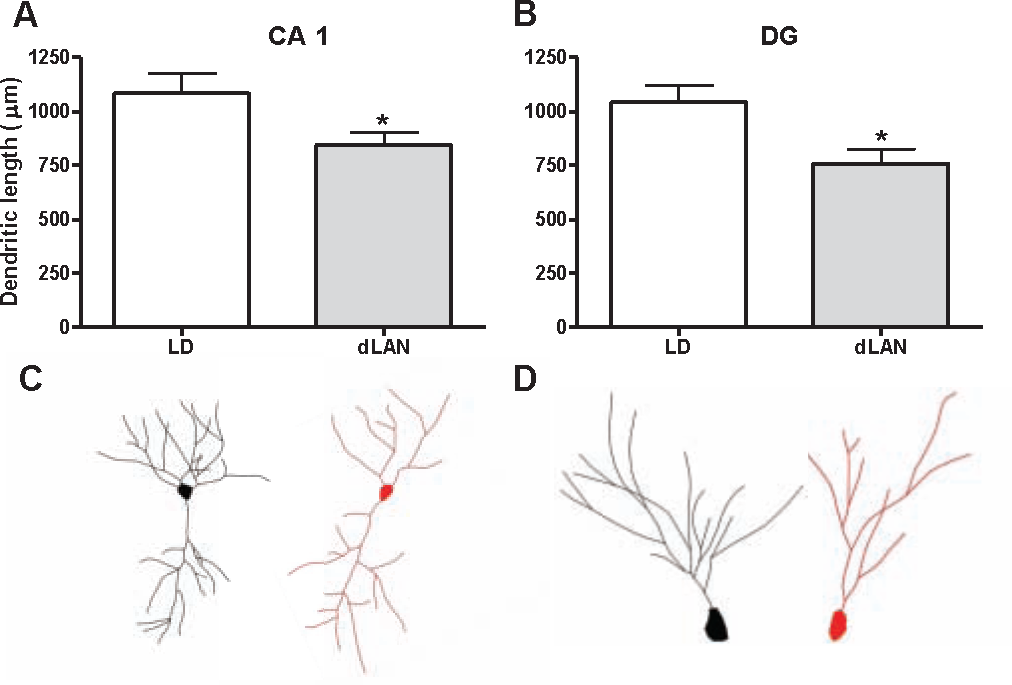
Morphological characteristics of hippocampal neurons. Total dendritic length was reduced in (A) CA1 basilar dendrites and (B) the dentate gyrus (DG). (C) Representative tracing from the CA1 of an LD grass rat (left) compared with a dLAN grass rat (right). (D) Representative tracing from the DG of an LD grass rat (left) and dLAN grass rat (right). Data are expressed as mean ± SEM. *p < 0.05 between groups. LD = light/dark; dLAN = dim light at night.
Morphological characteristics of neurons in the CA1, CA3, and DG of the hippocampus.
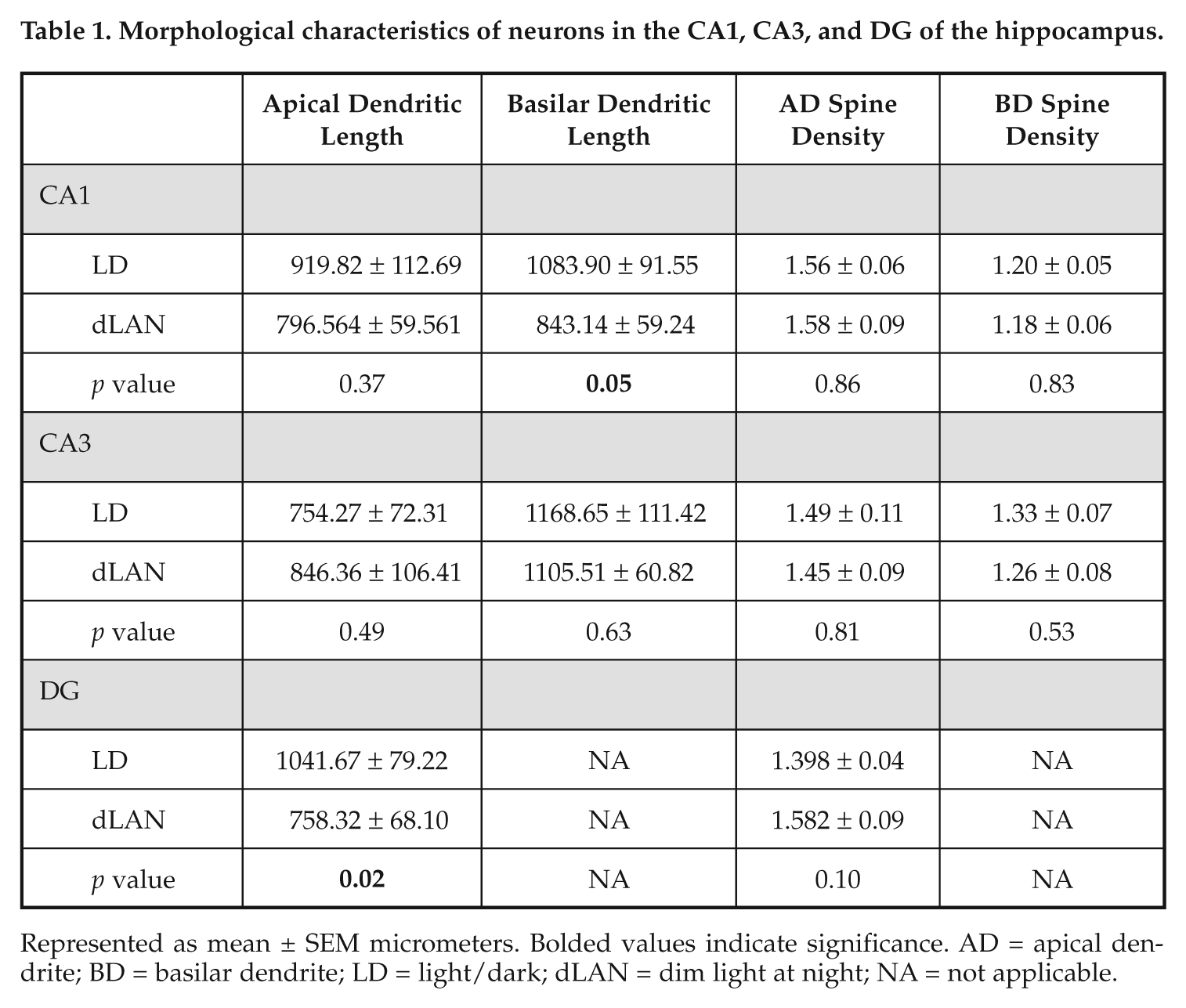
Represented as mean ± SEM micrometers. Bolded values indicate significance. AD = apical dendrite; BD = basilar dendrite; LD = light/dark; dLAN = dim light at night; NA = not applicable.
Corticosterone Concentrations
Grass rats displayed elevated corticosterone concentrations 2 weeks after placement in dLAN compared with LD (F 1, 16 = 4.521, p < 0.05; Fig. 3D).
Discussion
This study investigated the effect of dim nighttime light exposure on depressive-like responses and learning and memory in Nile grass rats, a diurnal rodent. Here we show that exposing grass rats to light at night impaired their spatial learning and memory as evaluated by the Barnes maze (Sunyer et al., 2007). Animals were trained to find a target hole on the maze over the course of 4 days and then evaluated in a probe trial. During the training trials, dLAN impaired performance as compared with LD. dLAN increased the latency for grass rats to reach the target hole and increased the number of visits to incorrect holes. Spatial memory was similarly impaired by dLAN. Twenty-four hours after the final training session, memory retention was assessed with a probe trial. dLAN decreased the percentage of time spent investigating the target hole, indicating decreased memory retention.
These results confirm and extend previous findings (Ma et al., 2007; Ling et al., 2009; Fujioka et al., 2011), indicating that housing nocturnal rodents under light at night impairs spatial learning and memory. Our results demonstrate that nighttime light exposure also impairs spatial learning and memory in diurnal rodents. Furthermore, previous studies have used 24 h lighting of the same intensity, while in this study, grass rats were exposed to a distinctly darker phase. In this study, we replicated previous reports that grass rats housed under LD and dLAN display equivalent levels of total daily locomotor activity and have similar locomotor activity rhythms (Fonken et al., 2011). All grass rats maintained a diurnal activity pattern, which is consistent with previous reports (McElhinny et al., 1997). Constant lighting conditions used in previous studies can result in an arrhythmic activity rhythm (Cambras et al., 2011). These results demonstrate that cognitive impairments occur in animals with light at night in the absence of disruption in locomotor activity rhythm.
The effects of light at night on other cognitive functions remain unspecified (Castro et al., 2005). It is possible that nighttime light exposure specifically targets hippocampal-dependent learning and memory through disruption of circadian processes (Ruby et al., 2008). Alternatively, dLAN may represent a mild chronic stressor producing deficits in learning and memory via reduced neurogenesis, changes in hippocampal architecture, or both processes (McEwen and Sapolsky, 1995; Gould and Gross, 2002; Fujioka et al., 2011). Studies in nocturnal rodents have reported glucocorticoid concentrations to be elevated or unaffected by nighttime light exposure (Abilio et al., 1999; Van der Meer et al., 2004; Fonken et al., 2009; Fonken et al., 2010). Grass rats housed under dLAN elevated plasma corticosterone concentrations. dLAN may be a stronger stressor to diurnal as compared with nocturnal rodents. Circulating glucocorticoid concentrations are predictive of hippocampal atrophy and memory deficits in both humans and rodents (Bodnoff et al., 1995; Lupien et al., 1998). Because corticosterone concentrations were measured only at a single time point, however, conclusions drawn from the results must be constrained.
Grass rats exposed to dLAN increased depressive-like responses in a sucrose preference test. dLAN reduced consumption of a sucrose solution in the sucrose anhedonia test. LD rats consumed a higher percentage of sucrose than water, whereas dLAN grass rats showed no preference for the sucrose solution. This implies that the sucrose solution had diminished hedonic valence for grass rats exposed to dLAN, which models a key feature of human depression (Willner et al., 1992). Results in the forced swim test were equivocal. Increased floating time in the forced swim test is considered “behavioral despair” because rodents putatively stop searching for an escape mechanism (Porsolt et al., 1977). There were no differences in total floating time in the forced swim test between LD and dLAN grass rats, but grass rat housed under dLAN reduced latency to first float. The forced swim test has not been extensively used in grass rats, although one study reported increases in floating duration in grass rats housed in short photoperiods (Ashkenazy-Frolinger et al., 2010). Grass rats are poor swimmers, which suggests the forced swim test may not be a reliable behavioral measure (Duplantier and Ba, 2001).
The depressive-like phenotype of the dLAN grass rats is consistent with our predictions based on depressive disorders related to both stress (Willner, 1997) and circadian dysfunction (Turek, 2007). Furthermore, nighttime light exposure increases depressive-like responses in nocturnal rodents (Bedrosian et al., 2011). Light at night may increase depressive-like responses through changes in hippocampal circuitry. dLAN decreased dendritic length in DG and CA1 basilar dendrites. Furthermore, there was a positive association between dendritic length in the dentate gyrus and sucrose consumption in the sucrose anhedonia test. The hippocampus is a critical structure in the pathophysiology of depressive disorders. Depression is associated with changes in glucocorticoids and hippocampal atrophy (Sheline et al., 1996; Sapolsky, 2000). Moreover, changes in hippocampal morphology are associated with chronic stress and depressive-like responses in rodents (Magarinos et al., 1996; Hajszan et al., 2009; Magarinos et al., 2011).
In summary, our results suggest that exposure to dim nighttime lighting can alter hippocampal neuronal morphology, impair learning and memory, and increase depressive-like responses in a diurnal rodent. Opportunities for exposure to light at night have rapidly increased during the past century. The present results suggest that this exposure may have accompanying maladaptive effects. Finding an appropriate model to test whether changes in environmental lighting are related to mood disorders is critical. Many animal models are potentially confounded by the use of nocturnal rodents that may not experience the same form of disruption as diurnal animals when exposed to light at night. The responses to light at night seen in the present study are comparable to those described in previous reports using nocturnal rodents (e.g., Bedrosian et al., 2011), which increases confidence that nocturnal rodents are appropriate subjects of study of this issue. In addition, our results raise questions about the use of rodent vivaria that have windows in the doors of animal rooms and continuous lighting in the halls.
Footnotes
Acknowledgements
The authors thank James Walton, Zachary Weil, and Tracy Bedrosian for technical assistance and Sallion Wolfe for excellent animal care. This research was supported by NSF grant IOS 04-16897 and by United States–Israel Binational Science Foundation grant 2005-337. L.K.F. was supported by an American Heart Association Pre-Doctoral Fellowship.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
