Abstract
Spina bifida meningomyelocele (SBM) is a neural tube defect that involves dysraphism of the spinal cord and extensive reorganization of the brain. The authors assessed the relationship between chronotype, diurnal preferences, and sleep problems in individuals with SBM and healthy controls. Although individuals with SBM showed the characteristic decelerating quadratic relationship between age and chronotype, the curve was displaced, peaking at a younger age in controls compared with SBM (23.4 vs. 29.2 years). Groups did not differ in morningness-eveningness preferences. Individuals with SBM endorsed more sleep problems than controls. Further examination of the relationship between entrainment and sleep in SBM is warranted.
The timing of sleep relative to the 24-h day changes across the life span, with progressively later sleep-wake times during childhood and adolescence (Carskadon et al., 1993) and earlier times in aging adults (Carrier et al., 1997). Sleep problems are common in neurodevelopmental disorders like SBM (Glickman, 2010; Harvey and Kennedy, 2002). SBM is associated with sleep apnea (Waters et al., 1998), abnormality of hypothalamic structures (Raybaud and Miller, 2008), and compromise of the optic nerve (Del Bigio, 2010), due to the Chiari II malformation of the cerebellum and brainstem, and due to hydrocephalus. To explore the idea that sleep may be disordered because of anomalies in brain regions associated with circadian rhythmicity, we measured chronotype, diurnal preferences, and sleep problems in 202 individuals with SBM and 62 typically developing, healthy age peers (Suppl. Tables S1 and S2).
Chronotype was measured using questions from the Munich Chronotype Questionnaire (MCTQ) to calculate the midpoint of sleep on free days, corrected for sleep duration (MSFsc; Suppl. Table S3; Roenneberg et al., 2003). MSFsc is correlated with objective measures of circadian timing (i.e., phase angle of entrainment) and is sensitive to developmental changes and genetic mutations associated with sleep pathologies (Allebrandt and Roenneberg, 2008). The relationship between age and chronotype was quadratic within each group and overall (SBM: F(2,188) = 7.64, p < 0.0006, R2 = 0.075, significant linear and quadratic terms for age t(188) = 3.69, p < 0.0003; t(188) = −3.57, p < 0.0004, respectively; Controls: F(2,55) = 7.04, p < 0.0019, R2 = 0.204, significant linear and quadratic terms for age t(55) = 2.04, p < 0.05, t(55) = −3.72, p < 0.0005; Overall: F(5,243) = 5.31, p < 0.0001, R2 = 0.10, significant linear and quadratic terms for age t(243) = 3.97, p < 0.0001, t(243) = −3.73, p < 0.0002). The positive linear estimate and negative quadratic estimate means that chronotype increases with age but the instantaneous slope of the curve decelerates constantly with age, revealing an overall parabolic (inverted U) shape (Fig. 1). There were no significant effects of group or group by age interactions in the overall model (all p > 0.1) and no effect of sex or lesion level group, a proxy for ambulatory status in SBM (Fletcher et al., 2005), when these variables were incorporated into the models (all p > 0.05).
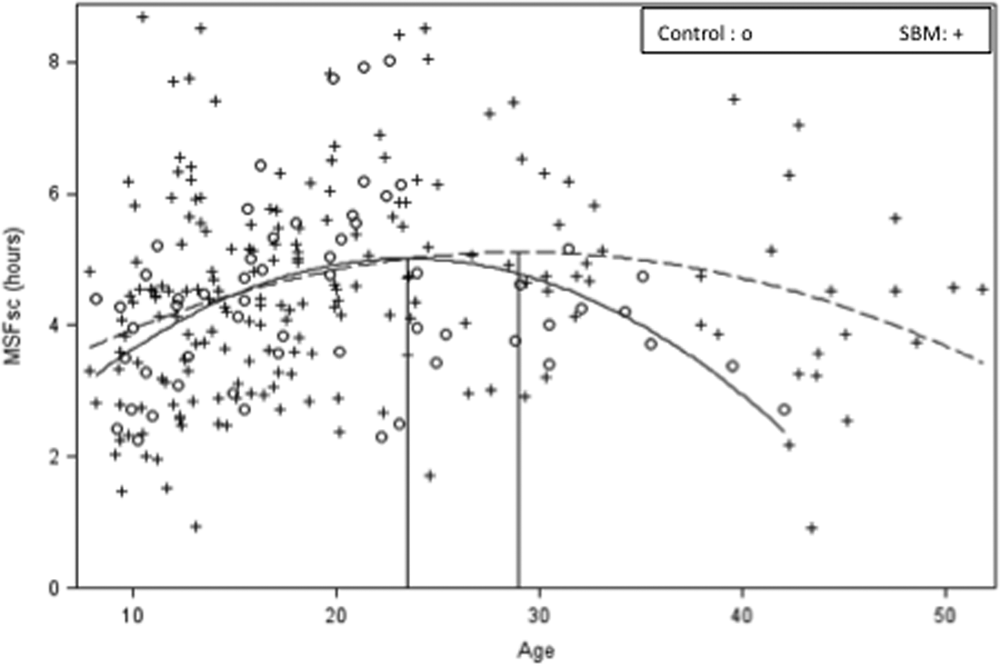
Timing of the midpoint of sleep on free days, corrected for sleep duration (MSFsc) as a function of age in SBM and controls. The curves represent the quadratic relationship of MSFsc, in hours, to age in controls (solid line) and SBM (dashed line). The vertical lines indicate the age at which the curve of the line changes direction (slope = 0) for each group. Age was centered for statistical analyses but is displayed in the graph in years. Note: One participant whose MSFsc score was negative (i.e., sleeps more on work nights than on free nights) is not represented in the figure but his data are included in all analyses.
MSFsc peaked at age 23.4 in controls and age 29.2 in SBM, identified by the derivative of the quadratic equation, that is, the point where the slope of the curve changes direction (where it equals zero). Inspection of Fig. 1 suggests differences in the rightward portion of the 2 curves (i.e., after MSFsc peaks in SBM), so we centered age at the SBM peak and found that the slope of the tangent line for controls was less than zero at this age (linear age by group interaction, t(243) = −2.15, p < 0.04); we found no interaction when age was centered at the control peak. This result is tempered by the nonsignificant group by (quadratic) age interaction, but it does suggest that at the age when MSFsc peaks in SBM, controls are already returning to earlier sleep-wake times.
Diurnal preference was measured using questionnaires that examine individual perceptions about “best” times of day for sleep, work, or other activities (Carskadon et al., 1993; Horne and Ostberg, 1976). Morningness-Eveningness Questionnaire (MEQ) scores have been correlated with measures of circadian rhythms in temperature, melatonin, and cortisol, although this relationship is not straightforward. Because diurnal preferences are measured with different instruments in children and adults, separate analyses were conducted for each age group. In children, there were no relationships between preference for morning versus evening activity and age (p > 0.05), nor were there differences between SBM and controls, or among controls, participants with lower spinal lesions, and those with upper spinal lesions (all p > 0.05). For adults, there was a linear relationship between age and diurnal preferences, t(122) = 3.46, p < 0.0007, that did not differ between diagnostic or lesion level groups, p > 0.05. Diurnal preferences and age were correlated across and within diagnostic groups (Overall: r = 0.29, p < 0.001; controls: n = 33, r = 0.36, p < 0.04; SBM: n = 92, r = 0.28, p < 0.007)
Sleep problems were explored using different questions for children and adults (Suppl. Table S4). A greater number of sleep problems were reported for both groups with SBM, compared with controls.
Chronotype and diurnal preferences were negatively correlated in children and adults (children: controls, r = −0.48, p < 0.008; SBM, r = −0.43, p < 0.0001; adults: controls, r = −0.71, p < 0.0001; SBM, r = −0.67, p < 0.0001). Neither of these was correlated with sleep problems in children (all p > 0.05). For adult controls (n = 33), chronotype was correlated with “Awakening in the early morning” (r = 0.46, p < 0.02), but diurnal preference was not related to ratings of sleep problems (all p > 0.05). For adults with SBM, chronotype was correlated with “I get tired” (r = 0.25, p < 0.03) and “Trouble falling asleep” (r = 0.22, p < 0.05); diurnal preference was related to endorsement of all sleep problems (range r = −0.22 to −0.33, median r = −0.26, all p < 0.05) except “Sleep that is restless or disturbed.”
Taken together, our data suggest that the phase relationship between sleep-wake timing and the 24-h day in SBM is not identical to that of typically developing individuals. Although the shape of the curve relating chronotype to age was similar in both groups, the curve for SBM appears much flatter. The lack of a linear relationship when analyses were partitioned by age in SBM supports this finding and contrasts with the robust linear relationships in controls. In addition, the curve in controls trends downward at the time it peaks in SBM.
Although evidence of premature development in SBM includes precocious puberty (Dahl et al., 1996) and early onset of cognitive difficulties characteristic of aging (Dennis et al., 2010), sleep timing patterns were delayed in adults with SBM relative to controls, with the shift to earlier sleep-wake times occurring almost a decade later than that documented previously (Roenneberg et al., 2004). Our results add to the extensive database of age-related changes in chronotype in the general population (Roenneberg et al., 2004) but suggest that there may be clinical groups with different phase relationships.
All adults increasingly preferred morning activities as they aged, but we found no relationship between younger age and increasing preference for evening activities, and no group differences in diurnal preferences, despite group differences in chronotype and a correlation between diurnal preferences and chronotype. Although chronotype and diurnal preferences have been used interchangeably, our data suggest that they are dissociable.
Children and adults with SBM endorsed sleep problems more often than controls. Interestingly, the problems endorsed by adults appear to involve sleep timing and quality. Chronotype was correlated with early morning awakening in controls, reflecting the typical developmental shift in sleep timing. In adults with SBM, chronotype was related to different sleep problems, raising the possibility that disturbances in circadian timing contribute to sleep difficulties in this population. Although chronotype is a more direct measure of sleep than is diurnal preference (Allebrandt and Roenneberg, 2008), diurnal preference was also related to sleep problems in adults with SBM.
Limited mobility might have affected chronotype or diurnal preferences. For example, the availability of assistance in toileting and wheelchair routines might have attenuated the adolescent shift to later activities, especially in individuals with SBM with upper spinal lesions who have more severe mobility challenges. In our sample, 93% of participants with upper spinal lesions were confined to wheelchairs with limited independent functioning in this domain. However, we found no relationship between lesion level group and diurnal preference or chronotype, suggesting that mobility or the structured delivery of services for mobility challenges does not influence morningness-eveningness preferences and that level of physical activity is unrelated to the phase relationship between sleep and the 24-h day in SBM.
Children and adults with SBM have problems in a range of cognitive and motor domains (Dennis and Barnes, 2010); nevertheless, they appear to judge their “best” times of day for sleep, work, or other activities in a manner similar to that of their peers. Because diurnal preferences have been associated with variations in cognitive performance throughout the day in healthy children and adults (Hasher et al., 2002; May et al., 2005), whether chronotype, diurnal preferences, or both are related to cognitive performance or the specific cognitive deficits (Dennis and Barnes, 2010) in children and adults with SBM remains to be investigated.
Footnotes
Acknowledgements
Grant sponsor: Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Grant number P01-HD35946 to J.M.F. and M.D. This research was supported in part by the Ontario Ministry of Health and Long Term Care (to K.E.). The views expressed do not necessarily reflect those of the OMOHLTC.
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
