Abstract
To adapt to seasonal variations in the environment, most mammalian species exhibit seasonal cycles in their physiology and behavior. Seasonal plasticity in the structure and function of the central nervous system contributes to the adaptation of this physiology in seasonal mammals. As part of these plasticity mechanisms, seasonal variations in proliferation rate and neuron production have been extensively studied in songbirds. In this report, we investigated whether this type of brain plasticity also occurs in sheep, a seasonal species, by assessing variations in cell proliferation in the sheep diencephalon. We administered the cell birth marker 5′-bromodeoxyuridine (BrdU) to adult female sheep in July and December, during long and short photoperiod, respectively. The BrdU incorporation was analyzed and quantified in the hypothalamus, a key center for neuroendocrine regulations, as well as in other structures involved in relaying neuroendocrine and sensory information, including the median eminence, the pars tuberalis of the pituitary gland, and the thalamus. In December, 2-fold and 6-fold increases in the number of BrdU+ nuclei were observed in the hypothalamus and thalamus, respectively, when compared with July. This variation is independent of the influence of peripheral gonadal estradiol variations. An inverse seasonal regulation of cell proliferation was observed in the pars tuberalis. In contrast, no seasonal variation in cell proliferation was seen in the subventricular zone of the lateral ventricle. Many of the newborn cells in the adult ovine hypothalamus and thalamus differentiate into neurons and glial cells, as assessed by the expression of neuronal (DCX, NeuN) and glial (GFAP, S100B) fate markers. In summary, we show that the estimated cell proliferation rates in the sheep hypothalamus, thalamus, and pars tuberalis are different between seasons. These variations are independent of the seasonal fluctuations of peripheral estradiol levels, unlike the results described in the brain nuclei involved in song control of avian species.
Seasonal changes in the environment compel most vertebrates to adapt their physiology in order to ensure their reproductive success and, therefore, the survival of their species. Consequently, many species display seasonal rhythms in their physiological functions and behavior. In avian species, marked structural changes in nuclei implicated in the song control system (Parry and Goldsmith, 1993; Brenowitz, 2004) together with variations in the production of new neurons (Goldman and Nottebohm, 1983; Nottebohm, 1985) have been repeatedly found to be associated with seasonal reproduction. Seasonal changes in cell proliferation rates have also been described for another nonmammalian vertebrate, Rana temporaria. The incorporation of [3H]thymidine by hypothalamic cells was higher in May/June during the sexual activity period than in September (Chetverukhin and Polenov, 1993). In seasonal mammals, seasonal variations in extension of neuronal inputs and glial ensheatment have been seen in sheep (Sergeeva and Jansen, 2009; Xiong et al., 1997), and structural changes have also been hypothesized in hamsters following the demonstration of seasonal variation in nestin expression at the base of the third ventricle (Barrett et al., 2006; Ebling and Barrett, 2008). Furthermore, in adult mammals, the issue of seasonal neurogenesis or gliogenesis in relation to seasonal reproductive physiology has been assessed in a few species. Large seasonal fluctuations in cell proliferation were detected in the dentate gyrus of adult meadow voles in relation with seasons (Galea and McEwen, 1999). In golden hamsters, altering the photoperiod can lead to changes in cell proliferation levels in several brain areas, including the hypothalamus, with an increase in cell proliferation following a transition from long to short days (Huang et al., 1998). In this study, we asked whether seasonal-dependent cellular plasticity occurs in sheep, a seasonal species, by comparing the rates of cell proliferation in Ile de France ewes in July and December during long and short days, respectively. We analyzed the sheep mediobasal hypothalamus (MBH) for the presence of newly generated cells following the intravenous administration of the cell proliferation marker bromodeoxyuridine (BrdU). Numerous BrdU+ cells were observed not only scattered in the MBH parenchyma and in the subependymal and ependymal cells lining the third ventricle but also in the thalamic parenchyma. Moreover, a moderate number of BrdU+ cells were found in distinct regions of the median eminence and the pars tuberalis (PT). We next showed a higher rate of cell proliferation in the hypothalamus in December as compared to July. This increase is independent of the variations in the peripheral estradiol levels. In the thalamus, initially used as a control structure, a marked seasonal variation of cell proliferation was unexpectedly found. In contrast, no seasonal variation in cell proliferation was observed in the subventricular zone (SVZ). Using colabeling with cell type–specific markers followed by confocal analysis, we demonstrated that the newly generated cells in the adult sheep hypothalamus and thalamus can differentiate into both neuronal and astrocytic cell types.
Materials and Methods
Animals
All animals were handled and cared for in accordance with the EC Directive of 24 November 1986 (86/609/EEC) and with authorization A37110 from the French Ministry of Agriculture. All procedures were approved by the Regional Ethics Committee (Comité Régional d’Ethique pour l’Expérimentation Animale, region of Centre-Limousin [CREEA]). Adult (2-year-old) primiparous Ile de France (IF) ewes (n = 10, 55.2 ± 4.8 kg body weight [BW] for the July experiment; and n =10, 61.3 ± 6.6 kg BW for the December experiment) were used for proliferation studies and for survival studies (n =10, 58.3 ± 4.2 kg BW). All ewes were kept in outdoor pens with a natural photoperiod and fed daily with hay and pellets and had free access to water. For experiments on cell proliferation (July and December), half of the ewes (n = 5) were ovariectomized and implanted subcutaneously with a 2-cm silastic capsule containing 17β-estradiol at least 3 weeks before the beginning of the experiments to maintain low physiological concentrations of estrogen (approximately 2 pg/mL) (Evans et al., 1994). They were maintained under a natural daylight regimen.
BrdU Treatments
For the test experiments performed in November, a single intravenous (i.v.) injection of either 20 or 80 mg/kg of BrdU (Sigma-Aldrich, Saint Quentin Fallavier, France) was tested. For the proliferation studies, a single dose of 20 mg/kg BrdU was administered between 0900 h and 1030 h. To assess whether the observed hypothalamic and thalamic proliferation rates varied between seasons, we compared the number of BrdU+ nuclei in July and in December during the long and short photoperiod, respectively. We hypothesized that, if any, cellular plasticity mechanisms may occur several weeks before the reproductive status transition, in July before the onset of the sexual activity period that starts in August in the IF breed, and in December before the beginning of the sexual rest period that starts mid-January. The first experiment was carried out in the beginning of July 2008 during the sexual rest period. A second experiment following the same experimental scheme was performed with another 10 IF ewes in the beginning of December 2008 during the sexual activity period. Ewes were euthanized 24 hours later with an overdose of sodium pentobarbitone (25 mg/kg; Merial, Lyon, France) preceded by an injection of heparin (25,000 IU, i.v.).
To examine the phenotype of the hypothalamic and thalamic BrdU+ cells, 4 i.v. injections of BrdU (20 mg/kg) were administered at 12-hour intervals to 2 groups of ewes (intact and ovariectomized plus estradiol implant) over a 48-hour period in December. The objective of multiple injections was to maximize the pool of BrdU+ cells to be able to determine their fate 4 weeks later. Ewes went back into their outdoor pens and were euthanized 4 weeks later at the beginning of January.
Tissue Preparation
At the end of each experiment, animals were decapitated. The heads were perfused through both carotid arteries with 2 L sodium nitrite (1%) in NaCl (0.9%) followed by 4 L of cold paraformaldehyde (4%) in 0.1 M phosphate buffer (pH 7.4) (Batailler et al., 2004), and their brains were processed for immunohistochemistry as detailed below.
Two blocks of brain encompassing part of the hypothalamic and thalamic structures (Suppl. Fig. S1A) and the SVZ (Suppl. Fig. S1B) were collected. After a 48-hour period of postfixation, blocks were soaked in 20% sucrose for cryoprotection. The blocks of brains were then processed for immunohistochemistry as detailed below.
Histological Procedures and Immunohistochemistry
Cryostat sectioning
Just before sectioning, the brain blocks were frozen by immersion in nitrogen-cooled isopentane. Coronal sections (10 µm) were processed and mounted directly onto SUPERFROST PLUS (Fisher Scientific, Illkirch, France) slides and kept at −80 °C. Immunohistochemical reactions were carried out using single peroxidase and double immunofluorescence methods on cryostat sections incubated overnight at room temperature with primary antibodies and for 90 minutes at room temperature with secondary antibodies. Single staining was carried out using indirect peroxidase techniques with peroxidase-conjugated secondary antibodies detected with 3,3’diaminobenzidine (DAB) in 50 mM Tris-HCl (pH 7.6) containing 0.025% hydrogen peroxide for 10 minutes followed by washing with 50 mM Tris-HCl (pH 7.6). For double staining, indirect immunofluorescence procedures using fluorochrome-conjugated antibodies were used. Antibodies were diluted in Tris-buffered saline (TBS) (pH 7.4) containing 0.3% saponin and 0.2% bovine serum albumin (BSA).
Immunoperoxidase protocol
Hypothalamic and thalamic coronal sections (10 µm) were collected from the premammillary recess over a 2.6-mm rostral direction (Suppl. Fig. S1A). Every tenth cryostat section was processed for BrdU immunohistochemistry using DAB-peroxidase detection. Coronal sections (10 µm) from the posterior SVZ were collected from the merging of the anterior commissure over a 1-mm rostral direction (Suppl. Fig. S1B). Every tenth cryostat section was processed for BrdU immunohistochemistry using DAB-peroxidase detection. Sections from the SVZ block that were labeled with BrdU were also used as positive controls for BrdU labeling. BrdU immunohistochemistry included an initial step to unmask and denature endogenous DNA using 2 N HCl for 30 minutes at room temperature. Sections were incubated in blocking buffer (TBS, 0.3% saponin, and 2% BSA) for 30 minutes at room temperature. A counter stain using neutral red was performed to visualize the shape and size of the nuclei of cells close to the BrdU-labeled cells.
Quantifications of cell proliferation
The BrdU+ nuclei were quantified using computer-assisted morphometry software (Mercator, Explora Nova, La Rochelle, France). Coronal sections of the diencephalon were divided into 3 areas. The hypothalamus (H) area includes 4 mm laterally on each side of the third ventricular walls and up to the upper limit of the third ventricle dorsally. The lateral limits of the thalamic area (T) correspond to the lateral limits of the hypothalamic area. Dorsally, the thalamic area extends up to 4 mm from the upper limit of the third ventricle. The median eminence (ME) corresponds to the structure on the floor of the third ventricle. A periventricular zone (PVZ) is first isolated from the hypothalamic and thalamic areas. Its perimeter is positioned 1 mm all around the third ventricular walls. Following quantification analysis, this zone was included in the hypothalamic area. Quantifications were performed on 4 coronal sections separated by 600 µm per animal corresponding to the MBH. The counter was unaware of the experimental group. Results are expressed as BrdU+ cell count means ± SEMs for 4 slices from 5 animals per group. Quantification was also performed in the same way for the SVZ of the lateral ventricle. The PT is the tissue surrounding the ME on the anterior part of the diencephalon sections. The BrdU+ nuclei in the PT were quantified on the sections used to quantify hypothalamic cell proliferation, and the results are expressed as mean number of BrdU+ cells (per mm2) ± SEM for 5 animals per group.
Immunofluorescence
Double labeling was performed in the same way with the addition of an initial step to unmask and denature endogenous DNA. For heat-induced epitope retrieval, 10 mM citrate buffer (pH 6.0) was applied. Cross-reactivity of the secondary antibodies was systematically checked by varying the sequence of staining on various experiments. Nonspecific labeling was assessed on alternate slides processed without the primary antibody. To further confirm the status of BrdU+ cells as dividing cells, BrdU and Ki67 colabeling was performed (Li et al., 2008).
To determine the frequency of BrdU+ cells coexpressing cell-specific markers (see Suppl. Table S1 for information on the antibodies used), dual fluorescence-labeled sections were observed with a laser-scanning Olympus FluoView confocal system (Tokyo, Japan). For each marker, 10 BrdU+ cells per region on each of 3 equidistant (600 µm) brain sections per animal from n = 5 animals were examined, and results were expressed as a percentage of BrdU + cells double labeled with a specific cell marker ± SEM. Sections were stained with the Hoechst (33258, Molecular Probes, Eugene, OR) marker, which labels the nuclei, for 2 minutes in 0.1% TBS. Each field was scanned over 25 to 35 focal planes using a minimum of 1024 × 1024 pixel resolution through sequential analysis of the 3 laser tracks. Confocal scans were processed with FluoView 500 software (Olympus). Cells were scored positive for a given phenotypic marker when the labeling was unequivocally coupled to a BrdU+ cell in all 3 dimensions on at least 5 successive optical sections.
Statistical Analysis
Statistical analyses were performed with StatView (SAS Institute Inc., Cary, NC, USA). For comparisons, 1-way and 2-way analyses of variance (ANOVAs) were performed using Tukey post hoc tests when appropriate. Differences were considered statistically significant at p <0.05.
Results
Validation of BrdU as a Marker of Hypothalamic Cell Proliferation in Sheep
We first optimized the dose of BrdU to enable a clear visualization of BrdU-immunoreactive nuclei in the diencephalon of the adult ewe. Two doses of BrdU were administered (20 mg/kg or 80 mg/kg, i.v.) to a limited number of ewes (n = 4). Because the higher dose of BrdU (80 mg/kg) did not induce a higher number of BrdU-labeled nuclei, we used 20 mg/kg for subsequent i.v. injections.
BrdU+ nuclei were found scattered throughout the diencephalon parenchyma, and BrdU+ nuclei were often observed as contacting pairs (Fig. 1, all insets), indicative of recent mitogenic activity (Taupin, 2007). BrdU+ cells were seen in the hypothalamus (Fig. 1A), the thalamus (Fig. 1B), the ME (Fig. 1C), the PT (Fig. 1D), the cells lining the third ventricle or ependymal cells (Fig. 1E), and the subependymal zone of the third ventricle (Fig. 1F). Ki67, a cell cycle protein that is expressed during the cell cycle, was used as an endogenous marker of cellular proliferation (Scholzen and Gerdes, 2000). Around 8% (8.2% ± 3.2%) of BrdU+ were double labeled with Ki67 in the diencephalons (data not shown). This value reveals the high mitotic activity of some of the BrdU+ cells that are capable of a re-entry in the cell cycle 24 hours after a first division. To confirm the progenitor nature of the BrdU+ cells, we used Sox2, a transcription factor marker of self-renewing, multipotent stem/progenitor cells (Bennett et al., 2009; Zhao et al., 2008). Over 32% of BrdU+ cells (32.5% ± 4.2%) coexpressed the stem/progenitor marker Sox2. Figure 1G illustrates 2 BrdU+ cells double labeled with Sox2.
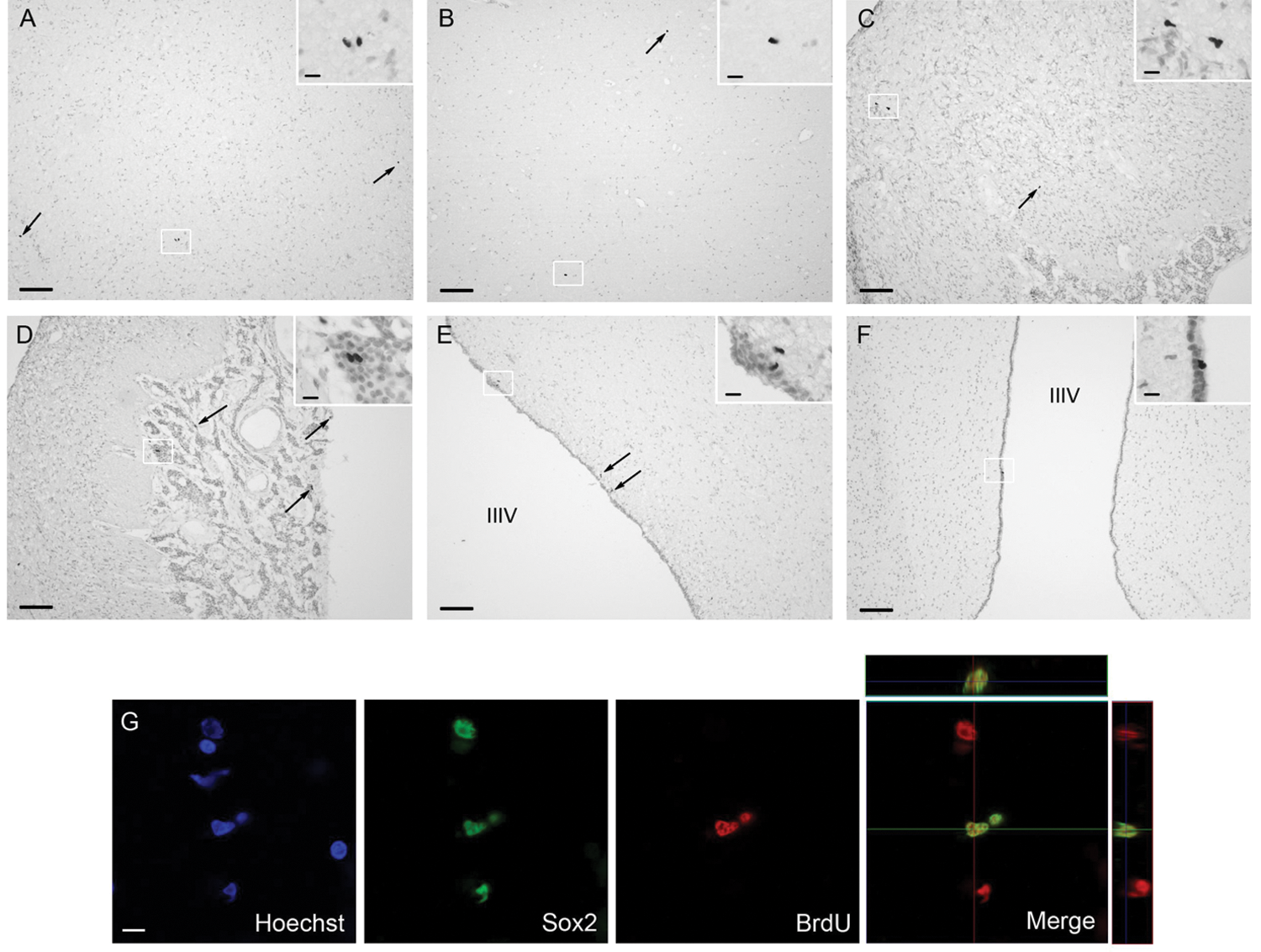
BrdU+ cells in the diencephalon and pituitary structures after BrdU injection. Brdu/Sox2 double labeling in adult sheep hypothalamus and thalamus. Low power photomicrographs showing BrdU-labeled cells 24 hours after BrdU injection, in sections counterstained with neutral red in the hypothalamic (A) and thalamic (B) parenchyma, the median eminence (ME) (C), the pars tuberalis (PT) (D), ependymal cells (E), and subependymal cells (E). BrdU+ cells are shown with arrows and in enlarged photomicrographs in insets. Scale bars: 100 µm (A-F) and 10 µm (all insets). Confocal microscopy image showing Hoechst staining and single and dual labeling for BrdU+ and the neural stem/progenitor marker Sox2. Scale bar: 10 µm.
Seasonal Regulation of Cell Proliferation in Sheep Hypothalamus, PT, and SVZ of the Lateral Ventricle
For the PVZ area, although a clear trend towards an increase in cell proliferation was observed in December, the difference between seasons was not significant (65.0 ± 14.3 and 112.2 ± 13.7 BrdU+ cells per 4 sections in July and December, respectively). This area was then included within the hypothalamic zone. In December, we found a significant increase in the number of BrdU+ nuclei in the hypothalamus when compared to BrdU+ cells quantified in July (Fig. 2A and 2B). The increase in BrdU+ cells in the thalamus during December compared to July was more prominent (Fig. 2A and 2B). In ovariectomized ewes carrying an estradiol implant to maintain constant estrogen levels, the increase in BrdU+ cells was not significantly different from that seen in intact ewes (Fig. 2C), suggesting that circulating levels of estradiol do not play a key role in regulating seasonal proliferation. In the ME, no variation between seasons was shown in the proliferation rates. The mitogenic activity in the PT was compared between July and December. We found a significantly higher level of proliferation activity in July (Fig. 3A and 3B). Similar to the central areas, the significant difference was still observed under constant estradiol levels. We then analyzed the proliferation rate in the SVZ of the lateral ventricle, a known neurogenic region. We found that new cell production was not different between seasons, indicating that the seasonal changes in new cell production are region specific (Suppl. Fig. S2).
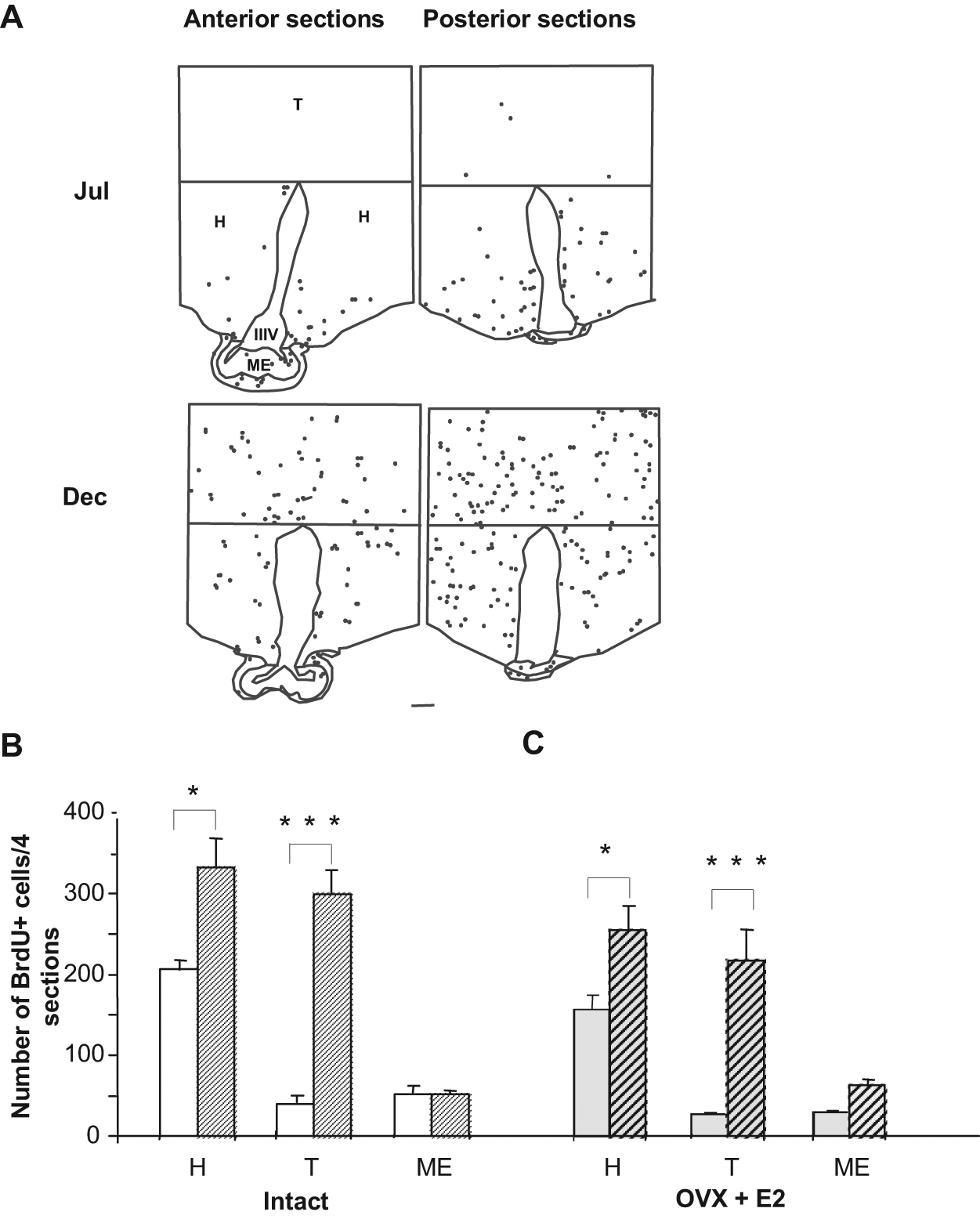
Comparison of the number and distribution of new cells in the sheep hypothalamus and thalamus between July and December. (A) Representative schematic drawings of anterior (left panels) and posterior (right panels) brain sections divided into hypothalamus (H), thalamus (T), and median eminence (ME). Each dot represents one BrdU+ nuclei. IIIV = third ventricle. BrdU+ cells are detected in the entire rostrocaudal area of the mediobasal hypothalamus (MBH) section in July (Jul, upper panels) and December (Dec, lower panels). Scale bar: 1 mm. (B) Number of BrdU+ cells in July (white) and December (hatched) in intact ewes. (C) Number of BrdU+ cells in July (gray) and December (gray-hatched) in ovariectomized ewes treated with an estradiol implant (OVX + E2). Results are expressed as the mean number of BrdU+ cells from 4 sections in July (plain) and December (hatched) in the 3 regions detailed in A. Error bars represent mean ± SEM (n = 5 animals per group). *p < 0.05, ***p < 0.001.
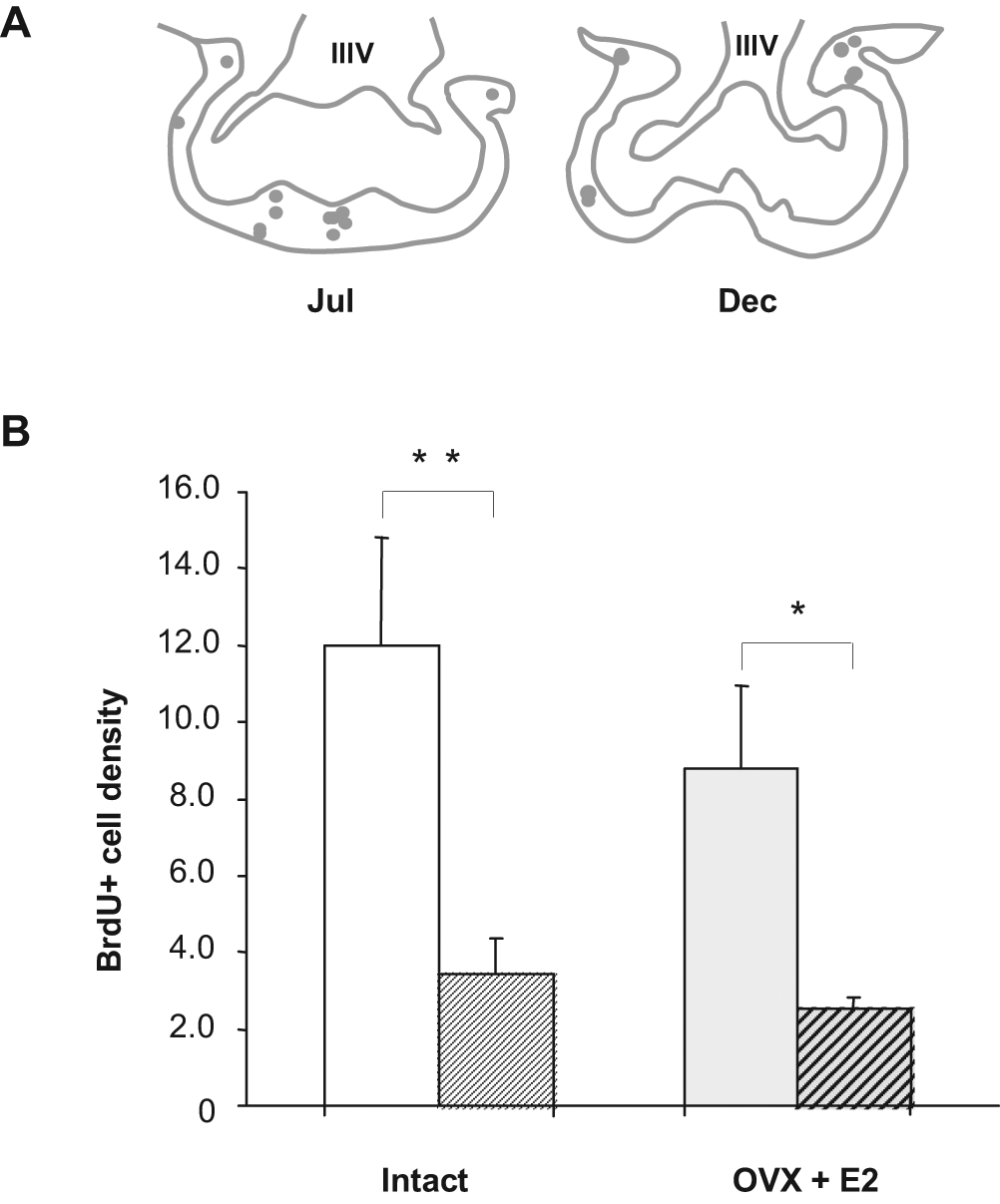
Comparison of the densities of new cells in the pars tuberalis (PT) in July and December. (A) A representative schematic drawing of a 10-µm coronal section of the PT in July (left) and in December (right). Each dot represents one BrdU+ nuclei. (B) Left histogram: BrdU+ cell densities in July (white) and December (hatched) in intact animals. Right histogram: BrdU+ cell densities in July (gray) and December (gray-hatched) in ovariectomized ewes treated with an estradiol implant (OVX + E2). Results are expressed as mean densities of BrdU+ cells on 4 sections in July (plain) and December (hatched). Error bars represent mean ± SEM (n = 5 animals per group). *p < 0.05, **p < 0.005.
Determination of the Fate of the Newborn Cells
Four weeks after BrdU administration, we found BrdU+ cells widely scattered in the hypothalamus and thalamus without any apparent clusters. Four weeks after BrdU injection, the total number of hypothalamic BrdU+ cells decreased compared to the total number of BrdU+ cells observed 1 day after injections but remained at a high level both in intact and ovariectomized ewes (Fig. 4A). In the thalamus, the total number of BrdU+ cells is substantially reduced 4 weeks after BrdU administration (Fig. 4B). Immunofluorescent double staining combined with confocal microscopy was used to determine the differentiation state of the new cells. Hypothalamic and thalamic BrdU+ cells were first tested for coexpression with doublecortin (DCX) (Fig. 5A), a transient marker for early migrating postmitotic neurons. Few BrdU+ cells expressing DCX (1%-2%) were detected in both structures. Double labeling with an antibody against NeuN, a marker for mature neurons, indicated that 16.7% ± 7.4% and 22.7% ± 13.2% of BrdU+ cells in the hypothalamus and thalamus, respectively, acquire a neuronal fate (Fig. 5B). BrdU+ cells that colocalize with the mature neuronal marker NeuN were detected in all brain regions studied and within all animals. However, the number of newly generated neurons was variable among individuals, suggesting variations in the time course for neuronal differentiation. Colabeling with an antibody against the astrocytic marker S100B revealed that 73.3% ± 8.8% and 52% ± 20.8% of the new cells from the hypothalamus and thalamus, respectively, display a glial phenotype of the astrocytic lineage (Fig. 5C). Colabeling with an antibody against glial fibrillary acidic protein (GFAP), another astrocytic marker, confirmed these results (70% ± 3.6% and 50.6% ± 9.8% for new hypothalamic and thalamic cells, respectively) (Fig. 5D). In contrast, few, if any, BrdU+ cells expressed 2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase), a marker for oligodendrocytes (data not shown), suggesting that at 4 weeks after BrdU injection, no new oligodendrocytes are generated. Taken together, these results indicate that newly generated cells in the adult sheep hypothalamus and thalamus can differentiate into both astrocytic and neuronal cell types under physiological conditions in vivo.
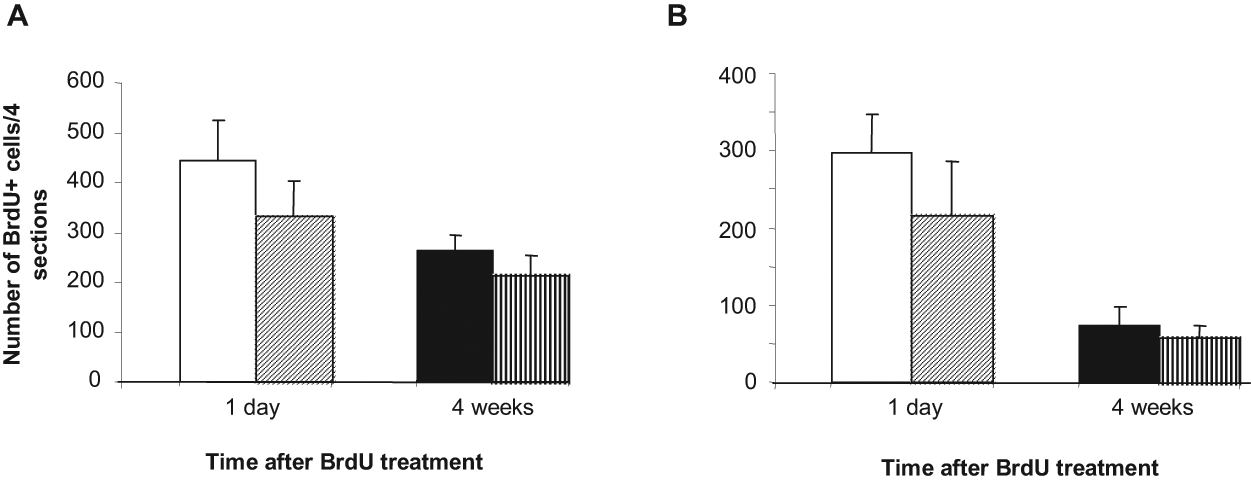
Comparison of the persistence of BrdU+ nuclei at 1 day and 4 weeks following BrdU injections performed in December. Number of new cells in the hypothalamus (A) 1 day (white bars) and 4 weeks (black bars) after injection in intact (plain) and OVX + E2–treated ewes (hatched). Number of new cells in the thalamus (B) 1 day (white bars) and 4 weeks (black bars) after injection in intact (plain) and OVX + E2–treated ewes (hatched). Error bars represent mean number of BrdU+ cells from 4 sections in July (plain) and December (hatched) ± SEM (n = 5 animals per group).
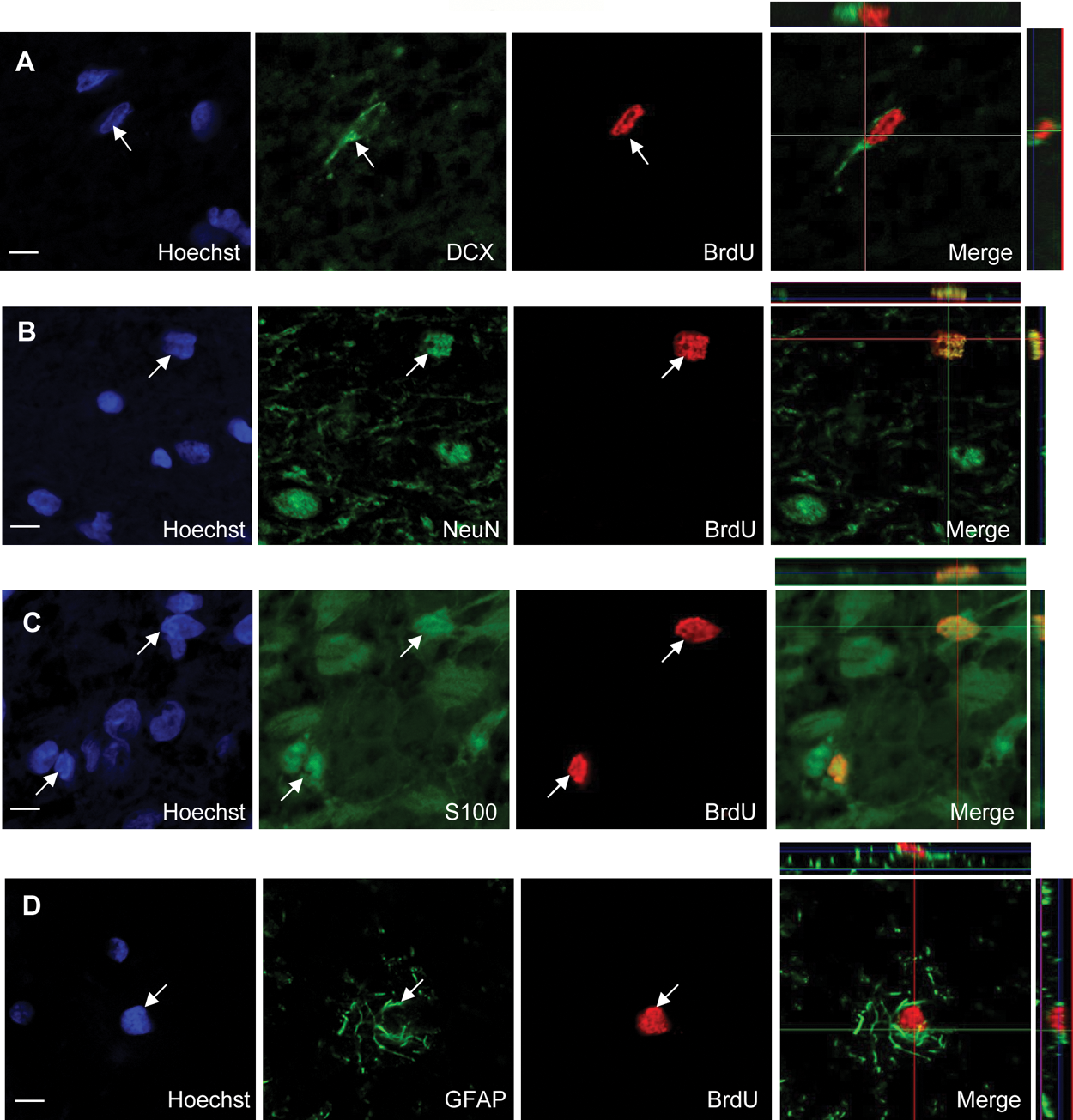
New hypothalamic cells exhibit neuronal and glial phenotypes. Confocal analysis of representative adult hypothalamic sections 4 weeks after BrdU administration. Single labeling for Hoecsht, BrdU, and cell type marker merges images with 3-dimensional confocal reconstruction for BrdU and DCX (A), BrdU and NeuN (B), BrdU and S100B (C), and BrdU and GFAP (D). Scale bar: 5 µm.
Discussion
By combining endogenous and exogenously administered cell proliferation markers, we detected a significant level of cell proliferation in the adult hypothalamus, thalamus, and PT, structures involved in the control of seasonal physiological functions. We further demonstrated that cell proliferation rates change between seasons. The number of cells having incorporated BrdU varied in opposite ways between the central structures (hypothalamus and thalamus) and PT.
Cell Proliferation in the Hypothalamus
We found BrdU+ cells in the vicinity of the third ventricle walls as well as deep within the hypothalamic parenchyma, suggesting that in adult ewes, similar to mice hypothalamus (Kokoeva et al., 2005, 2007), cell proliferation is not confined to definite germinal niches but occurs all over the hypothalamic parenchyma. A possible explanation for this scattered mitogenic activity is the widespread presence of dividing progenitor cells in situ. Nearly a third of BrdU+ cells also express Sox2, strongly suggesting that these dividing cells represent multipotent progenitor cells, as previously reported in the hypothalamus of adult mice and rats (Pencea et al., 2001; Reynolds and Weiss, 1992; Weiss et al., 1996). A growing number of studies report that hypothalamic progenitor cells can proliferate in vitro (for a review, see Migaud et al., 2010). It is not clear yet whether hypothalamic progenitor cells are true stem cells or whether they derive either from recently identified stem cell niches in the ependymal lining and nearby circumventricular organs (Bennett et al., 2009; Perez-Martin et al., 2010; Xu et al., 2005) or originate from the SVZ of the lateral ventricle, 1 of the 2 most active mitogenic zones of the central nervous system. A minor proportion of the dividing cell progenitors may migrate caudally into the hypothalamus, as reported for new neurons in the adult primate brain (Gould et al., 1999).
An absence of cell proliferation has been previously reported in the hypothalamus of adult Merino ewes (Hawken et al., 2009). Several methodological differences between the experimental protocols used could explain this discrepancy. Hawken and colleagues (2009) performed their experiments in Merino, a different sheep breed in which reproductive activity is much less driven by seasons than in the IF breed. In addition, they used a different protocol to immunodetect BrdU+ cells (different primary antibody and high concentration of HCl). In addition, BrdU injections were carried out in November, which corresponds to a long photoperiod and sexual rest in the southern hemisphere. These parameters may have contributed to the absence of BrdU+ cell detection in the hypothalamus.
Seasonal Variation of Cell Proliferation Rates in the Hypothalamus
We found an increase in cell proliferation rates in the hypothalamus in December, during the short photoperiod, indicating that new cell generation is enhanced during short days. These results, together with data from other groups showing an increase in neuronal birth and survival in the hamster brain following a transition from long to short days (Huang et al., 1998), suggest that seasons may regulate new cell generation. The functional relevance of this change in hypothalamic cell proliferation rates between short and long days is not yet understood, and it is necessary to test the hypothesis that this process may contribute to the seasonal variations in neuroendocrine secretion and homeostasis including by blocking this cellular proliferation using an antimitotic drug. Nevertheless, the fact that no such variations were observed in the SVZ supports the hypothesis that seasonal changes in proliferation rates are region specific and linked to structures involved in seasonal neuroendocrine regulations.
Photoperiodic transitions were shown to alter cell proliferation in the golden hamster SVZ (Huang et al., 1998); however, this effect was most extreme at the rostral tip of the SVZ. It would be interesting to examine the more rostral section of the sheep SVZ.
Sexual hormones are potent regulators of neuronal turnover in many vertebrate species, and estradiol is a regulator of hippocampal neural progenitor cell proliferation in adult rodents (Cameron and Gould, 1994; Ormerod and Galea, 2001; Tanapat et al., 1999; Ormerod et al., 2004). Therefore, the influence of seasonally changing estradiol levels on seasonal proliferation in adult ewes needed to be taken into account. Our results suggest that the increase in cell proliferation observed during short days is not related to seasonal changes in peripheral estradiol levels. Since seasonal variation in cell proliferation is observed in both intact and in OVX + E2 ewes, one may hypothesize that it is likely to be of functional importance for endogenously driven rhythms (i.e., circannual rhythms), such as reproduction in sheep. This hypothesis could be tested by analyzing cell proliferation variation in free-running animals (i.e., in constant photoperiodic conditions).
The identification of the local or global stimuli that seasonally activate cell proliferation in vivo is underway, and melatonin is a leading candidate. Melatonin, through a circadian rhythm of secretion, is the transducer of photoperiodic information. The duration of melatonin secretion is inversely proportional to day length, allowing this hormone to act as a “time messenger” that synchronizes circannual rhythms to time of year (Malpaux, 2006; Malpaux et al., 1997). In addition, melatonin was recently reported to modulate proliferative activity in the dentate gyrus of early postnatal rats (Kim et al., 2004) and in embryonic neural stem cells (Moriya et al., 2007) and to promote the survival of new neurons in the hippocampus of adult mice (Ramirez-Rodriguez et al., 2009). Thyroid hormones are other key factors that might be involved in the seasonal regulation of cell proliferation. TSH β expression, which is driven by the photoperiod, has recently been shown to regulate the hypothalamo-hypophysial gonadal axis at the PT-brain interface in mammals (Hanon et al., 2008; Yasuo et al., 2010). In addition, when present in some of the hypothalamic nuclei, T3 allows passage into seasonal anestrus (Anderson et al., 2003).
Cell Proliferation Rates and Season in the Thalamus
Cell proliferation has previously been reported in the rodent thalamus (Pencea et al., 2001). Our results showing seasonal changes in cell proliferation rates support the view that adult thalamic nuclei have a greater ability to generate new cells than previously thought. The functional significance of the regulation of this thalamic mitogenic activity is difficult to envision. It may be related to the emerging role of this structure in the processing of sensory stimuli (Saalmann and Kastner, 2009). Our data indicate that 4 weeks after BrdU injections, the number of BrdU+ cells detected is reduced, suggesting that some of these new cells either die or migrate to other brain areas.
Cell Proliferation Rates and Season in the PT
In our study, we show changes in cell proliferation activity in the PT with a higher number of new cells produced in July compared to December. This may be related to the higher level of prolactin secreted during the long days of summer (Thimonier et al., 1978). The seasonal changes in cell proliferation in this structure are of interest considering the recently reported critical role that the PT plays on the photoperiodic regulation of the hypothalamo-hypophysial gonadal axis in mammals (Hanon et al., 2008; Yasuo et al., 2010). A first step in the elucidation of the functional significance of this seasonal variation will be to characterize the phenotype of the new pituitary cells.
Cell Fate
Four weeks after BrdU administration, 16.7% and 22.7% of the hypothalamic and thalamic BrdU+ cells, respectively, expressed the mature neuronal marker NeuN, indicating that neurogenesis is occurring in these brain structures in adult sheep. On the other hand, we found that 70% and 52% of the new cells differentiated into S100B+ astrocytes in the sheep hypothalamus and thalamus, respectively. The same proportion of BrdU+ cells was found to be double labeled with GFAP, confirming that these progenitors also generate astrocytic cells. The newly generated neurons and astrocytes were scattered throughout the parenchyma, confirming the absence of neurogenic niches. No new CNPase+ oligodendrocytes were detected in the sheep hypothalamus and thalamus, in agreement with previous studies in rats (Steiner et al., 2004). However, using another oligodendrocytic marker (APC), Kokoeva and colleagues (2005) reported oligodendrogenesis in the mouse hypothalamus. Whether this reflects a species difference in the time course of glial differentiation remains to be determined. These results indicate that new cells are at least bipotent progenitors, the remaining pool of BrdU+ cells likely corresponding to not yet differentiated cells.
In conclusion, we found that substantial seasonal changes in cell proliferation occur in December, during short days, in the sheep diencephalon including the hypothalamus. These newly formed cells have the capacity to express either a neuronal or an astrocytic phenotype because after 4 weeks, they expressed either the mature neuronal NeuN or the astrocytic S100 or GFAP markers. The next step will be to examine the fine tuning of the seasonal regulation of neurogenesis and gliogenesis in these structures and explore the role of such cellular plasticity on seasonal animal physiology.
Footnotes
Acknowledgements
The authors thank Dr. Vincent Prévot (INSERM U816 Lille) for help with the BrdU immunostaining protocol and the technical staff of the experimental unit UEPAO of the INRA Centre of Tours for taking care of the animals. This work was financially supported by the INRA PHASE department and a grant from the Agence Nationale de la Recherche (ANR) (ANR-09-JCJC-0049-01).
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
