Abstract
Male workers in copper smelting are exposed to copper, lead, and arsenic. This study aimed to assess the effects of combined exposure to these metals on male reproductive hormone levels and assesses malondialdehyde (MDA) as an oxidative stress parameter. The study was conducted on 40 copper smelter workers compared with 40 non-exposed workers. Laboratory investigations included levels of serum copper, blood lead, serum arsenic, follicle-stimulating hormone (FSH), luteinizing hormone (LH), testosterone, and MDA. Levels of copper, arsenic, lead, FSH, and LH were significantly increased compared to controls. However, a statistically significant decrease in the mean value of testosterone was found among exposed workers. Positive correlations between serum copper and both serum FSH and MDA levels were statistically significant as were correlations between serum arsenic and MDA levels. Testosterone levels showed significant negative correlations with both copper and arsenic among exposed workers. A linear regression model of copper, arsenic, and lead levels as independent variables with FSH, LH, and testosterone as dependent variables revealed a significant negative association between serum copper and testosterone levels. The current study concluded that combined exposure to copper, arsenic, and lead in secondary copper smelters had a negative impact on male reproductive hormone levels that may be mediated by oxidative stress.
Introduction
Copper smelter production is currently increasing because of increased demand for copper metal in electrical, electronic, construction, transportation, industrial machinery, and other products that include electrical wires, air conditioning, power, and telecommunication utilities (Li et al., 2013). According to Forsén et al. (2017), various trace elements from impurities present in the copper ores are emitted during the production process. The process is a significant source of atmospheric arsenic and copper.
Rambhatla and Mills (2016) concluded that some heavy metals in the environment are considered endocrine disruptors, affecting the hypothalamic-pituitary-gonadal axis. This can be manifested in exposed males in the form of reproductive and sexual dysfunction. According to OSHA’s Hazard Communication Standard (HCS, 2012), reproductive toxicity definition includes any adverse effects on sexual function and fertility in adults of both sexes. These include changes in the structure and function of the reproductive organs and modifications in any other functions dependent upon the reproductive system integrity. Mantovani and Maranghi (2005) stated that male reproductive toxicity directly affects the reproductive system and endocrine balance. Doumouchtsis et al. (2009) mentioned that lead exposure and hormonal imbalance caused reproductive impairment. Lead accumulation affects the hypothalamic-pituitary axis. Kumar (2018) stated that lead has toxic effects on human male reproduction by decreasing hormone production even in moderate- to low-level exposures.
One of the key characteristics of male reproductive toxicants is the alteration in the production and levels of reproductive hormones and the induction of oxidative stress (Arzuaga et al., 2019). Renu et al. (2018) stated that oxidative stress is one of the mechanisms involved in arsenic-mediated male reproductive toxicity in humans and experimental studies. Other mechanisms include inhibiting spermatogenesis, testosterone pathway, inflammation, genotoxic effects, and activation of heat shock proteins. Combined occupational exposure to copper, arsenic, and lead and their reproductive toxicity has not been widely investigated among copper smelters with their presumed mechanism.
Aim of work
The study aim was to assess the effects of combined exposure to copper, lead, and arsenic on male reproductive hormone levels and to assess MDA as an oxidative stress parameter among workers in a secondary copper smelter.
Materials and methods
A comparative cross-sectional analytical study was conducted in a factory for nonferrous industries in Cairo, Egypt. One of its sectors is a secondary copper smelter. The research Ethical Committee of Kasralainy Hospital, Faculty of Medicine, Cairo University approved this study with approval number (MS 352,020). The study was done during the period from February to March 2020. The study population consisted of two fertile male groups: an exposed (40 workers) and a non-exposed control group (40 workers). All workers in the copper smelter, satisfying inclusion criteria, were included in the study. Inclusion criteria for exposed workers were sharing in the smelting process for at least the preceding couple of years with ages ranging from 20 to 55 years for both groups. Exclusion criteria for both groups included those who had diabetes or were receiving hormonal therapy or antioxidant medication. The non-exposed group consisted of 40 male workers in other sectors of the same factory not occupationally exposed to the copper smelting process.
This study was performed following the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. The workers who participated in the study provided written consent. The studied groups were subjected to laboratory investigations: (a) Serum arsenic and copper and blood lead levels were measured by hydride-generation atomic absorption spectrophotometry with Zeeman background. (b) Reproductive hormones, including serum FSH, LH, and free testosterone, were measured by radioimmunoassay (RIA). Hormonal reference levels were as follows: Serum FSH: 2–15 mIU/ml, Serum LH: 1.5–9.3 mIU/ml and Serum Testosterone: 300–1000 ng/dL. (c) Malondialdehyde (MDA) in plasma. All were analyzed by high-performance liquid chromatography (HPLC).
Statistical analysis was carried out with SPSS- version 26 using non-parametric Mann–Whitney and Kruskal–Wallis tests for comparing quantitative variables, Chi-square for categorical data, and Spearman correlation. Linear regression analysis was done to predict hormone levels using different metals (Chan, 2004).
Results
Copper, arsenic, lead, follicle-stimulating hormone (FSH), luteinizing hormone (LH), testosterone, malondialdehyde (MDA) levels, and erectile dysfunction complaints among the studied groups.
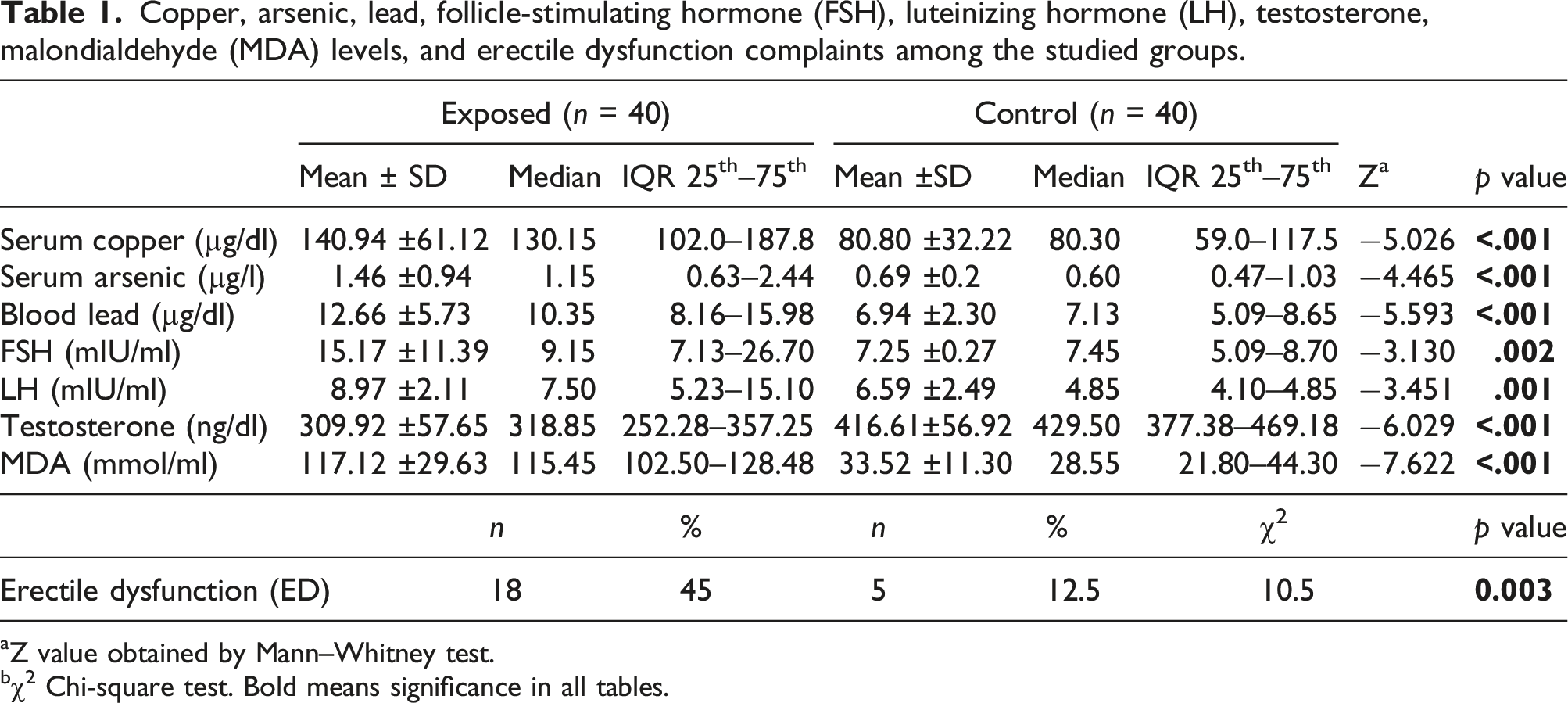
aZ value obtained by Mann–Whitney test.
bχ2 Chi-square test. Bold means significance in all tables.
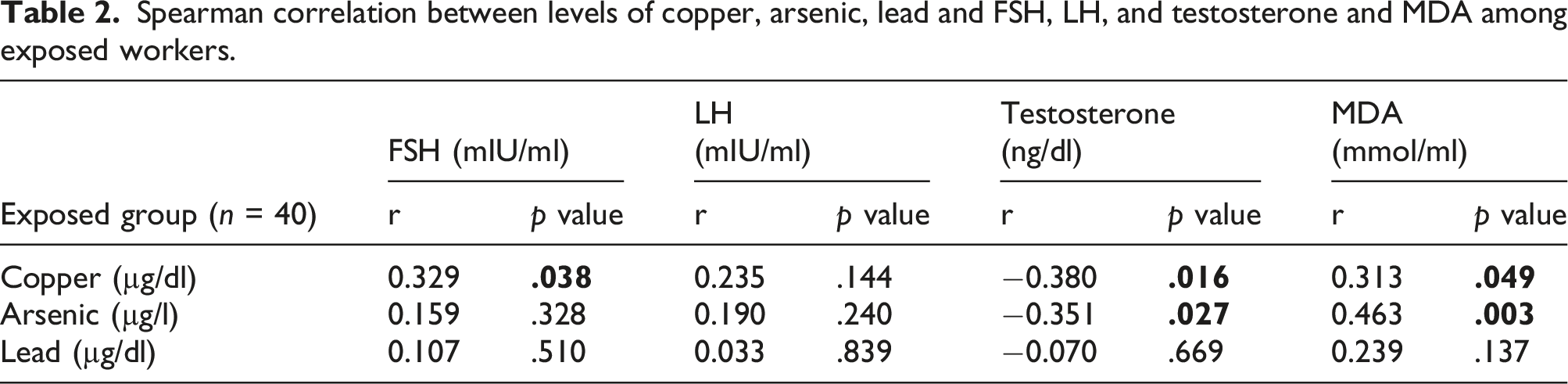
Spearman correlation between levels of copper, arsenic, lead and FSH, LH, and testosterone and MDA among exposed workers.
Spearman correlation between levels of copper, arsenic, lead, male reproductive hormones, and duration of employment among exposed workers.
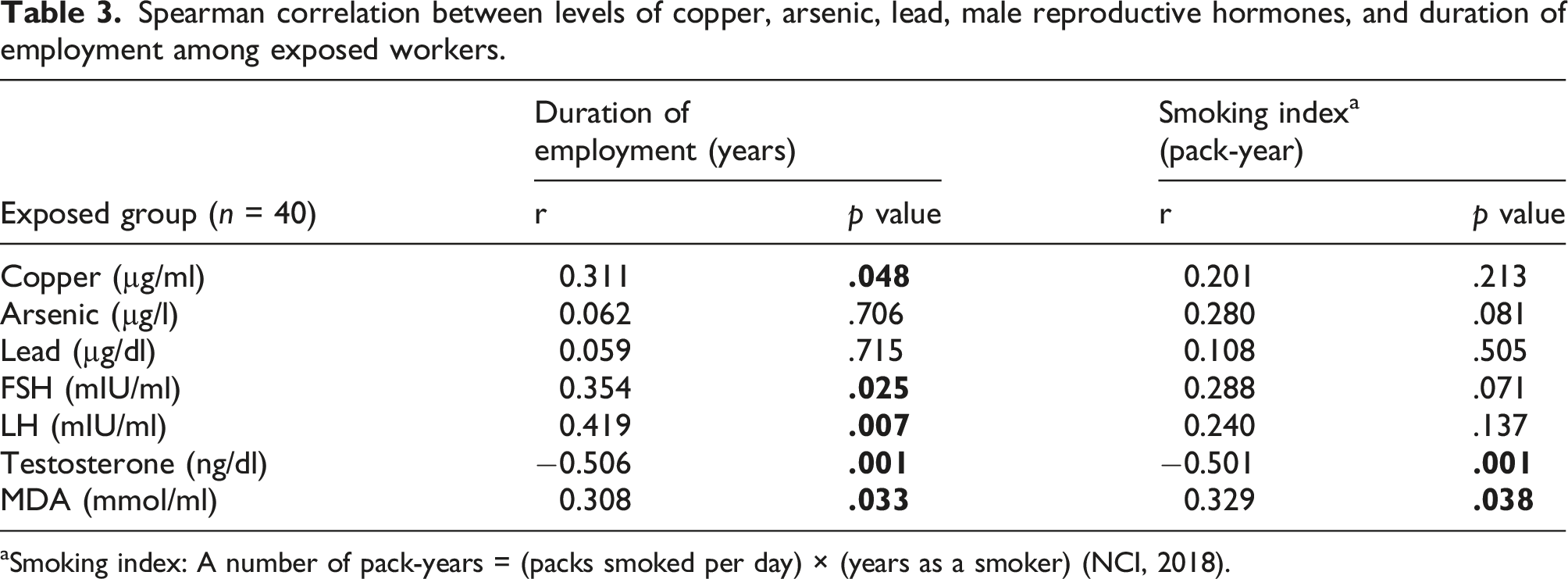
aSmoking index: A number of pack-years = (packs smoked per day) × (years as a smoker) (NCI, 2018).
Multivariate linear regression to detect FSH, LH, and testosterone as dependent variables using copper, arsenic, and lead as independent predictors.
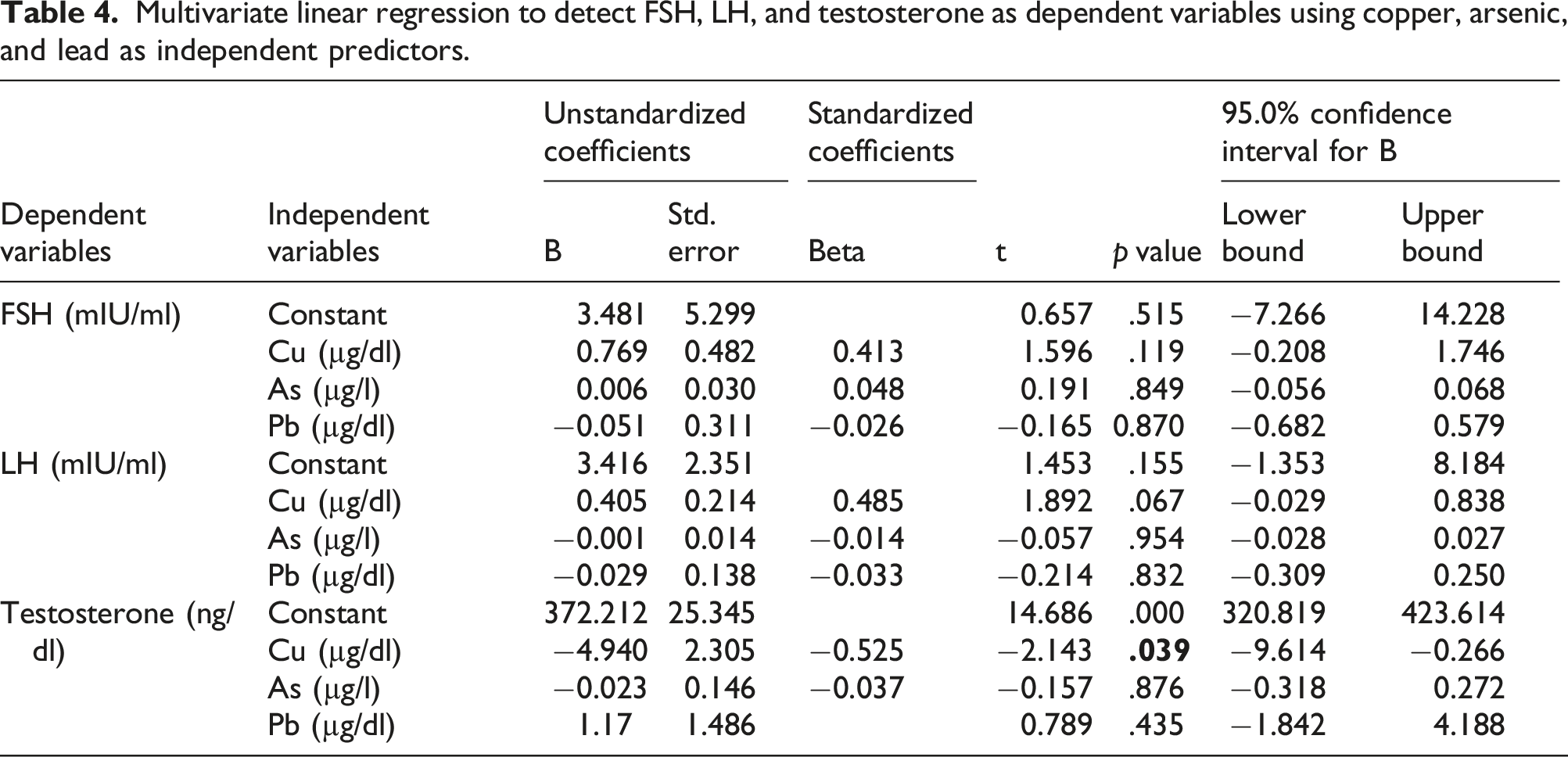
As for FSH, the overall model explained 19.9% of the variation in serum FSH (R2 = 0.199) and it was useful in explaining serum FSH levels, F (3, 36) = 2.988, ρ < 0.05.
As regards LH, the overall model explained 21.6% of the variation in serum LH (R2 = 0.216) and it was not useful (non-significant) in explaining serum LH levels, F (3, 36) = 3.308, ρ = 0.054.
Regarding testosterone, the overall model explained 28.5% of the variation in serum testosterone, and it was significantly valuable in explaining serum testosterone levels (R2 = 0.285), F (3, 36) = 4.784, ρ < 0.05.
A significant negative association between serum copper and testosterone level (t = −2.143, ρ < 0.05) was found. For each one µg/dl increase in serum copper, there was a 4.94 ng/dl decrease in serum testosterone.
Binary logistic regressions to assess the effects of copper, arsenic, and lead exposure (independent predictors) on the likelihood of elevation of both FSH and LH above reference intervals and reduction of testosterone below reference levels (dependent variables).
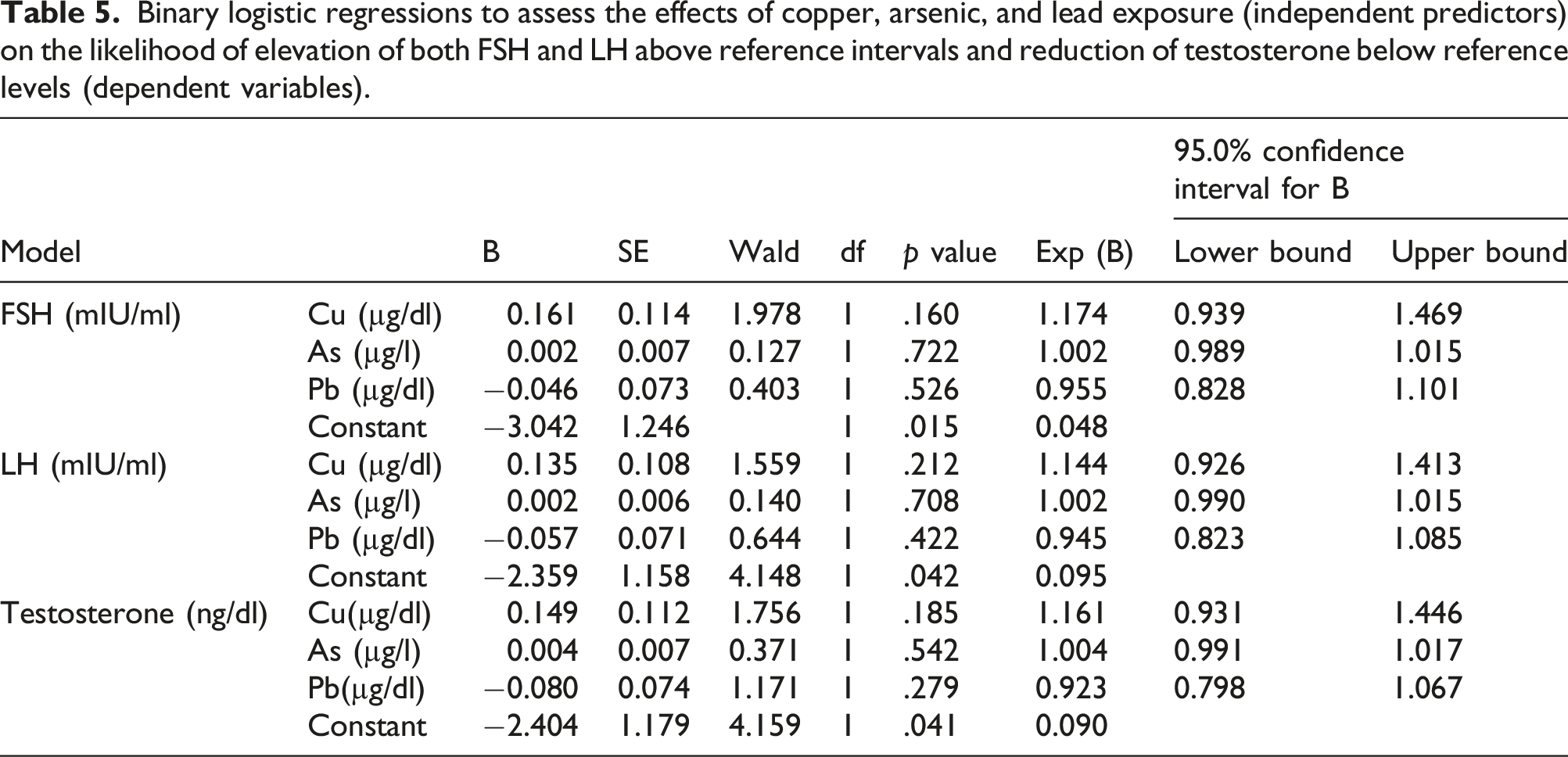
As regards FSH, (χ2 (3) = 9.469,
As regards LH, (χ2 (3) = 7.477,
As regards testosterone, (χ2 (3) = 10.417,
However, none of the independent predictors statistically contributed to the dependent variables.
Discussion
Copper smelter workers are exposed to mixtures of metals including copper, arsenic, and lead that cause reproductive toxicity affecting spermatogenesis (Eidi et al., 2010) and the pituitary-testicular axis (Jana et al., 2006; Doumouchtsis et al., 2009).
The present study found that erectile dysfunction (ED) was significantly more prevalent among the exposed group than the control group. This was supported by Rambhatla and Mills (2016) in their review on the impact of environmental exposure, including heavy metals on male sexual health, similar to Anis et al. (2007), who stated that erectile dysfunction might be related to chronic lead exposure. The blood lead level was significantly higher in the ED group than in the control group. The studied patients with high blood lead levels showed deposition of grayish lead granules in histological sections of the cavernous tissue. Fang et al. (2008) found an increased prevalence of erectile dysfunction among those chronically exposed to arsenic.
The exposed workers had a significant elevation of heavy metal (copper, arsenic, and lead) levels compared to the control. This is similar to Milnerowicz et al. (2010), who conducted their study on copper-foundry workers occupationally exposed to heavy metals (arsenic, cadmium, and lead) and found that blood lead increased by 9-fold and urine arsenic increased by 10-fold in all groups of smelter workers compared with the appropriate control groups. Smoking habits significantly influence certain reproductive parameters (Telisman et al., 2000). Association between blood lead and testosterone was affected by smoking status (Kresovich et al., 2015). This was consistent with the present finding of a significant negative relationship between testosterone and the smoking index.
Reproductive endocrine parameters included FSH, LH, and testosterone. Pituitary hormones (FSH and LH) were significantly elevated in the exposed group compared to the control. Some hormonal levels were beyond the reference intervals (elevated FSH and LH and depressed testosterone), but these disorders were not significantly affected by any of the studied heavy metals. A positive association was found between FSH and copper. Another study revealed that LH and FSH levels were significantly higher in infertile men (having reproductive toxicity) with occupational exposure to lead than in the group of fertile men without occupational exposure to lead (El Zohairy et al., 1996). A regression model indicated that serum copper was positively associated with LH (Jurasovic et al. 2004). A review discussing copper-induced neuroendocrine reproductive effects both in vivo and in vitro (Roychoudhury et al., 2016) found that copper is essential during gonadotropin-releasing hormone (GnRH) secretion. The present findings were supported by research on the effects of chronic oral exposure to sodium arsenite on hypothalamic-pituitary-testicular activities in mature male albino rats. Their results showed decreased plasma LH, FSH, and testosterone. The researchers explained that arsenic probably affects pituitary gonadotrophins leading to the inhibition of androgen production, so inorganic arsenic has a suppressive effect on androgenesis (Jana et al., 2006).
Testosterone was significantly decreased in the exposed group compared to the control group. It was significantly negatively associated with copper and arsenic. Rambhatla and Mills (2016) stated that lead and arsenic are androgen suppressors. Morakinyo et al. (2010) found a decreased plasma testosterone level in adult male rats upon exposure to sodium arsenite. A study on Taiwanese men, who were chronically exposed to arsenic in drinking water, found decreased testosterone levels compared with controls (Fang et al., 2008). A study of the copper effect on male reproductive functions among those with low lead exposure found that serum copper was negatively associated with thin sperm (Telisman et al., 2007). Animal studies concluded that copper was associated with abnormal testosterone, LH, FSH concentrations, and increased MDA concentrations (Liu et al., 2016). These studies supported the current results regarding copper and arsenic.
No significant association was found between lead and reproductive hormones. There is evidence of altered male reproductive hormones with chronic occupational lead exposure; however, much of these results were not statistically significant (Balachandar et al., 2020). Male reproductive endocrine function impairment may not be evident, although human semen quality was significantly reduced with moderate exposure to lead (blood Pb < 400 µg/L, exposure duration ≥2 years), which was reported by Telisman et al. (2000). They conducted their study on healthy male industrial workers aged 20–43 years with slight to moderate occupational exposure to lead and found no significant difference between lead-exposed workers and controls regarding reproductive endocrine function parameters (FSH, LH and testosterone), while semen quality parameters were affected. Another study came to the same conclusion as it suggested that lead caused hypothalamic or pituitary disturbance with long-term exposure after initial subclinical testicular damage, as the study found that lead-exposed individuals showed high LH and FSH levels associated with average testosterone concentrations in short-term lead-exposed individuals. In contrast, low testosterone levels did not induce high LH and FSH concentrations in long-term exposed individuals (Doumouchtsis et al., 2009). However, Yu et al. (2010) found that testosterone levels were significantly lower in lead-exposed workers than in the control group, suggesting that exposure to lead may alter male reproductive hormones and impair endocrine function. In contrast, a study conducted by Kresovich et al. (2015) found that blood lead was positively associated with testosterone among those environmentally exposed to heavy metals.
Several mechanisms, including oxidative stress, may mediate male reproductive toxicity in humans and experimental studies. MDA was a parameter of oxidative stress in this study and was significantly elevated in the exposed compared to the control and positively associated with copper and arsenic levels and employment duration. This is supported by Gholamhoseinian et al. (2007), who examined the influence of exposure to copper on MDA production in copper industry workers aged 35–55 years. They found higher serum copper levels among exposed workers in comparison with the controls. The serum MDA concentration in workers was higher than that in the control group. In addition, Escobar et al. (2010) found that urinary arsenic and serum MDA levels were significantly higher among smelter workers than among controls. A review by Zubair et al. (2017) on arsenic-induced toxicity in the animal male reproductive system concluded that arsenic exposure might cause reactive oxygen species generation and oxidative stress, and alterations in gonadotropins, LH and FSH levels, as well as testosterone, were found with severe germ cell degeneration. Anis et al. (2007) found that serum reactive oxygen species were significantly greater among those with high blood lead levels, while serum antioxidant levels were significantly lower among the same individuals when compared with those having low blood lead. Kasperczyk et al. (2008) conducted a study on healthy fertile men, employees of zinc, and lead metalworkers. They concluded that high lead exposure was associated with lipid peroxidation in the form of increased MDA. A study by Wang et al. (2006) indicated that the action of lipid peroxidation was increased by high arsenic levels and the duration of exposure and rendered arsenic-induced MDA production likely due to natural protective system impairment, such as glutathione (GSH) depletion.
Conclusion
The current study concluded that exposure to copper, arsenic, and lead in secondary copper smelters had a negative impact on male reproductive hormone levels that may be mediated by oxidative stress.
Footnotes
Acknowledgments
To our colleague, Dr Marwa Mohammed Fouad, Associate Professor of Occupational and Environmental Medicine, Faculty of Medicine, Cairo University, for her great effort in this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
