Abstract
This toxicology study was conducted to assess the impact of formaldehyde, a common air pollutant found in Chinese gymnasiums, on the brain function of athletes. In this research, a total of 24 Balb/c male mice of SPF-grade were divided into four groups, each consisting of six mice. The mice were exposed to formaldehyde at different concentrations, including 0 mg/m3, 0.5 mg/m3, 3.0 mg/m3, and 3.0 mg/m3 in combination with an injection of L-NMMA (NG-monomethyl-L-arginine), which is a nitric oxide synthase antagonist. Following a one-week test period (8 h per day, over 7 days), measurements of biomarkers related to the nitric oxide (NO)/cGMP-cAMP signaling pathway were carried out on the experimental animals post-treatment. The study found that: (1) Exposure to formaldehyde can lead to brain cell apoptosis and neurotoxicity; (2) Additionally, formaldehyde exposure was found to alter the biomarkers of the NO/cGMP-cAMP signaling pathway, with some changes being statistically significant (
Introduction
Formaldehyde (FA) is a significant indoor air pollutant in China due to its use in various products such as food, paints, wood processing, furniture, disinfectants, and preservatives (Wang et al., 2022; Ge et al., 2020; Sakr et al., 2003; Wu et al., 2013; Zhu et al., 2022; Lin et al., 2022; Tang et al., 2015). As a result of its emission from these products, it has been identified as a common air pollutant in gymnastics practice rooms. Despite its low level in the body, FA plays a crucial role in metabolic processes (Ye et al., 2013; Tang et al., 2013). Elevated levels of FA in the body can cause damage to critical systems, particularly the central nervous system, leading to difficulties in learning and memory impairment (Yao et al., 2021). Studies conducted on animals have shown that exposure to FA can result in significant deterioration in learning ability and memory, as well as morphological changes in rat hippocampal tissue (Li et al., 2020; Wei et al., 2022; Songur et al., 2003). Additionally, research by Lu et al. (2008) and others have indicated that exposure to high concentrations of FA can result in oxidative damage to brain tissue in mice, as well as changes in learning and memory-related genes. Studies have shown that levels of FA pollution are linked to central nervous system lesions, and construction workers exposed to FA have exhibited symptoms of neurotoxicity (Elinson 1984; Shi et al. 2006). However, the exact mechanisms by which FA induces neurotoxicity are still not fully understood.
Formaldehyde is a naturally occurring compound within the body (Agathokleous and Calabrese, 2021). Despite constant levels of formaldehyde in the blood of rats after inhaling it, neurotoxicity is still present (Kleinnijenhuis et al., 2013). As a result, researchers have been investigating potential mechanisms that could explain the neurotoxicity of formaldehyde. Studies have shown that exposure to FA can result in increased levels of nitric oxide (NO) in the brain of rats (Songur et al., 2003). Additionally, research conducted by Tang et al. (2013) has found that FA can lead to an increase in NO levels, which in turn can suppress hydrogen sulfide (H2S) and ultimately result in neurotoxicity. In the body, NO can form nitrosyl complexes with the heme iron in soluble guanylyl cyclase (sGC). The mentioned process activates the enzyme, leading to an increase in the intracellular cyclic guanosine monophosphate (cGMP) level. This has been observed in studies conducted by Li et al. (2020) and Tang et al. (2013). cGMP is a secondary messenger that can activate the downstream cGMP-dependent protein kinase (PKG) and induce inhibition of phosphodiesterase PDE-2. This adjustment of the cAMP and cAMP/PKA signaling pathway triggers a series of cascading cell responses, which plays a role in the transmission of information (Tegeder et al., 2004) and eventually leads to changes in downstream gene expression. The cellular signaling pathways of NO/cGMP and cAMP are crucial in various physiological processes, as reported by Basini et al. (2010) and Heydarpour et al. (2013). NO is a gaseous diffusible neurotransmitter that differs from classic neurotransmitters and is involved in various physiological and pathological reactions in the central nervous system (CNS) (Ledo et al., 2004). Nitric oxide synthase (NOS) generates NO from L-arginine (L Arg) (Stuehr et al., 1991). Under normal conditions, NO is synthesized by eNOS and iNOS.
NG-monomethyl-L-arginine (L-NMMA) is a NOS antagonist commonly utilized in research related to NO-related diseases (Perner et al., 2001). cAMP and cGMP are two biochemical substances that are present in various human and animal tissues and have independent biological activities. As an essential intracellular second messenger, they have the ability to affect cell function by delivering information and regulating cell metabolism (Kong et al., 2022). They play a significant role in various physiological and pathological functions. cAMP and cGMP are interrelated and interact with each other, as shown in Figure 1. In many cases, they have obvious antagonistic functions (Chen and Chai 2002). Pathological changes in the mouse cerebral cortex (H&E staining). (a) Control group, (b) 0.5 mg/m3 FA, (c) 3.0 mg/m3 FA, (d) 3.0 mg/m3 FA+L-NMMA.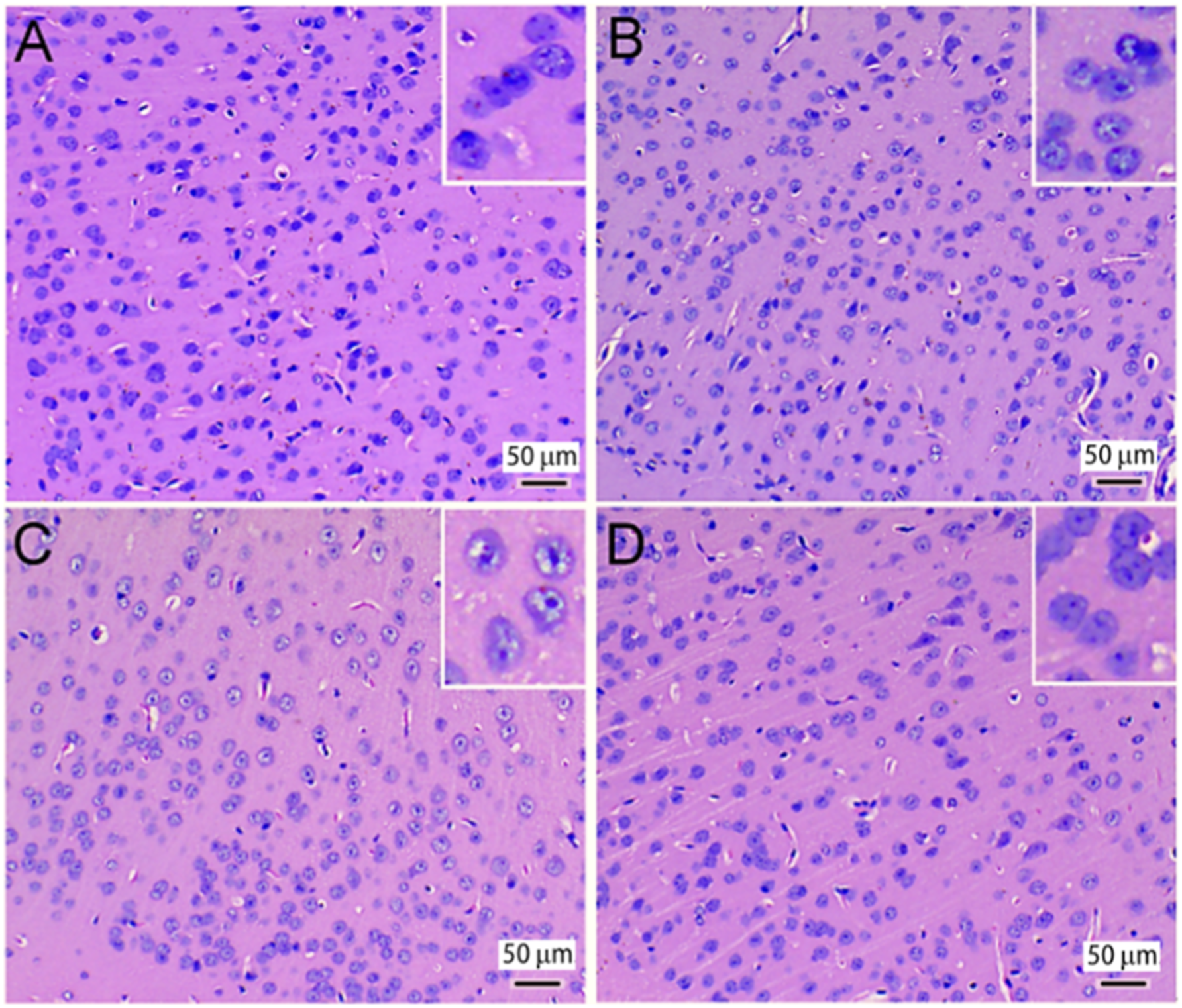
This study aimed to investigate the specific mechanisms of neurotoxicity caused by gaseous formaldehyde in mice, focusing on the NO/cGMP and cAMP signaling pathway. The mice were exposed to different concentrations of formaldehyde (0 mg/m3, 0.5 mg/m3, 3.0 mg/m3), based on the occupational exposure limits set by the People's Republic of China Ministry of Health. In the experimental group treated with the NOS antagonist (L-NMMA), mice were exposed to a concentration of 3.0 mg/m3 of formaldehyde. The levels of cAMP, cGMP, NO, PKA, and PKG were measured in the cerebral cortex, hippocampus, and brainstem to better understand the mechanism of formaldehyde-induced neurotoxicity in mice. Moreover, NOS activity was also examined.
Materials and methods
Equipment, reagents, and kits
The following equipment and reagents were used in this study: a WH-2 small intelligent environment and climate chamber (Wuhan Yuxin Technology Development Co., Ltd., China), Centrifuge-5415R (Eppendorf, Germany), 4160-2 Formaldehyde Analyzer (Interscan, USA), HW-2250 tissue slicer (Huiwo Technology Co., Ltd, China), 4% formaldehyde (Sigma-Aldrich, USA, 100496), and L-NMMA (Sigma-Aldrich, USA, M7033).
The test kits used included Nitric Oxide assay kit (abcam, USA, ab65328), Nitric oxide synthase kit (Abcam, USA, ab211083), Lowry protein assay kit (Thermo Fisher, USA, 23240), Mouse cAMP ELISA test kits (Abcam, USA, ab65355), Mouse cGMP ELISA test kits (Abcam, USA, ab133026), Mouse PKA ELISA test kits (Abcam, USA, ab139435), and mammal PKG ELISA test kits (MyBioSource, USA, MBS3805365).
Experimental animals and their selection
The study used 24 healthy male Balb/c mice that were specific pathogen-free. These mice were between 5 and 6 weeks old and weighed 23 ± 1.5g. The mice were purchased from the Experimental Animal Center of Hubei Academy of Preventive Medicine in Wuhan, China and were kept in standard environmental conditions with a 12 h light-dark cycle, 50%–60% humidity, and a temperature of 20°C–25°C. They were provided food and water ad libitum. Before the study began, the mice were quarantined for 7 days for health status assessments. They were then randomly divided into four experimental groups, with six mice in each group.
Experimental protocol
Grouping and treatments

Biological sample preparation
Following cervical dislocation, the mouse brains were removed and placed in PBS buffer on ice. The brains were rinsed with 4°C PBS buffer to remove any blood, and then the cerebral cortex, hippocampus, and brain stem were isolated. The tissues from each region were weighed and homogenized with pre-cooled PBS to create a 10% solution. The resulting supernatant was centrifuged at low temperatures and all procedures were conducted at 0∼4°C. The samples were sub-packed and stored at -70°C for future use.
Histological examination
The mouse cerebral cortex was fixed in a 4% buffered paraformaldehyde solution for 24 h at 4°C. Afterward, the samples were dehydrated and embedded in paraffin. At least three sections of 5 μm per tissue were prepared and stained with hematoxylin and eosin (H&E). The prepared sections were then observed under a light microscope (Leica DM 4000B, Leica, Wetzlar, Germany) to identify any histopathological changes in the mouse cerebral cortex.
Determination of cAMP, cGMP, PKA, and PKG contents
The levels of cAMP, cGMP, PKA, and PKG in the mouse cerebral cortex, hippocampus, and brainstem were determined according to the instructions provided with the test kits. The standard curves were plotted according to the instructions and were utilized to determine the levels of cAMP, cGMP, PKA, and PKG in the samples.
Determination of NO content and NOS activity
The NO assay kit and NOS kit instructions were followed to determine NO levels and NOS activity in the mouse cerebral cortex, hippocampus, and brainstem. Protein content was quantified using the Folin-phenol measured protein quantification kit. The NO content and NOS activity in different brain regions were then calculated as per the kit requirements.
Statistical analysis
The data are presented in the form of mean ± SEM. Statistical graphs were generated using GraphPad Prism 5.0. Statistical analyses were performed using IBM SPSS Statistics 19.0. As multiple sets of measurement data were obtained in this experiment, differences among groups were tested with ANOVA and followed by an LSD test. A
Results
Pathological changes in the mouse cerebral cortex
Figure 1(a) shows the control group with normal morphology, while Figure 1(c) shows the cerebral cortex tissue of the 3.0 mg/m3 FA exposure group with neuronal degeneration, apoptosis, uneven coloring, cell swelling, vacuolar degeneration, and nuclear pyknosis. Even in the low FA concentration group (Figure 1(b)), apoptosis of neuronal cells was observed. However, the antagonist group (Figure 1(d)) showed less neuronal degeneration than the high concentration FA group, indicating that +L-NMMA alleviated the toxic effects of FA on cerebral cortex cells and tissue. (see Figure 1).
Influence of FA exposure on the NO/cGMP-cAMP signaling pathway in the mouse cortex
Upon exposure to gaseous formaldehyde, NOS activity was observed to increase (Figure 2(A)) in comparison with the control group. While NOS activity did not change significantly in the 0.5 mg/m3 group, it was significantly higher in the 3.0 mg/m3 group ( The levels of biomarkers measured in the mouse cerebral cortex. (Mean±SEM, 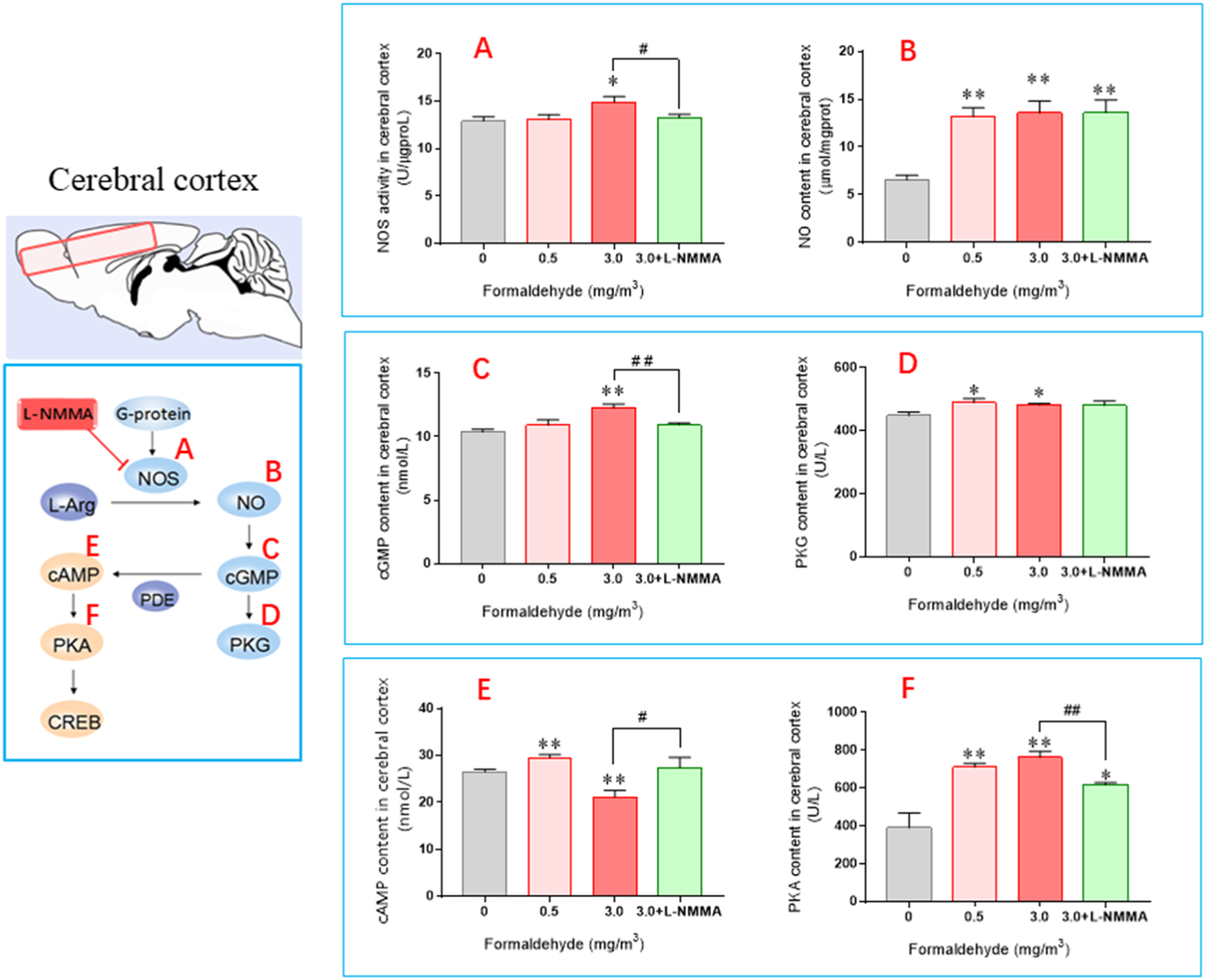
The study showed that the experimental group had a significant increase in cGMP content compared to the control group, as seen in Figure 2(C). The 3.0 mg/m3 group had a highly significant increase (
The study found that cAMP levels were significantly elevated in the 0.5 mg/m3 group (
Influence of FA exposure on the NO/cGMP-cAMP signaling pathway in the mouse hippocampus
In Figure 3(a), NOS activity increased compared with the control group, with the 0.5 mg/m3 group showing a significant increase ( The levels of biomarkers measured in the mouse hippocampus. (Mean±SEM, 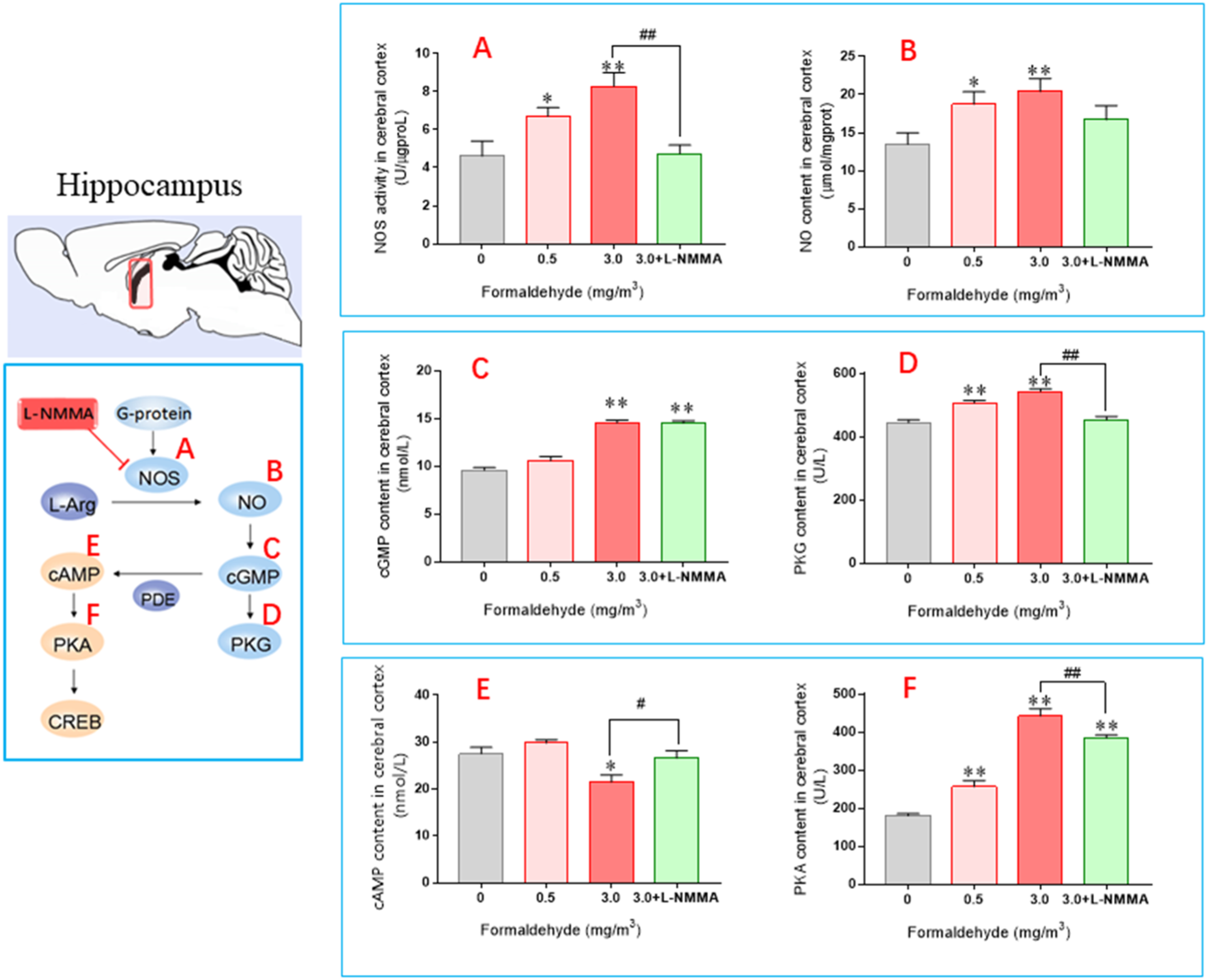
In comparison to the control group, the cGMP levels of the 0.5 mg/m3 group remained unchanged (as shown in Figure 3(c)), whereas the levels of the 3.0 mg/m3 and +L-NMMA groups increased significantly (
In Figure 3(e), it was observed that the cAMP content in the 0.5 mg/m3 group did not change significantly compared to the control group, whereas it decreased significantly (
Influence of FA exposure on the NO/cGMP-cAMP signaling pathway in mouse brainstem
In Figure 4(a), it was observed that NOS activity increased in both the 0.5 mg/m3 and 3.0 mg/m3 groups compared with the control group with high significance ( The levels of biomarkers measured in the mouse brainstems. (Mean±SEM, 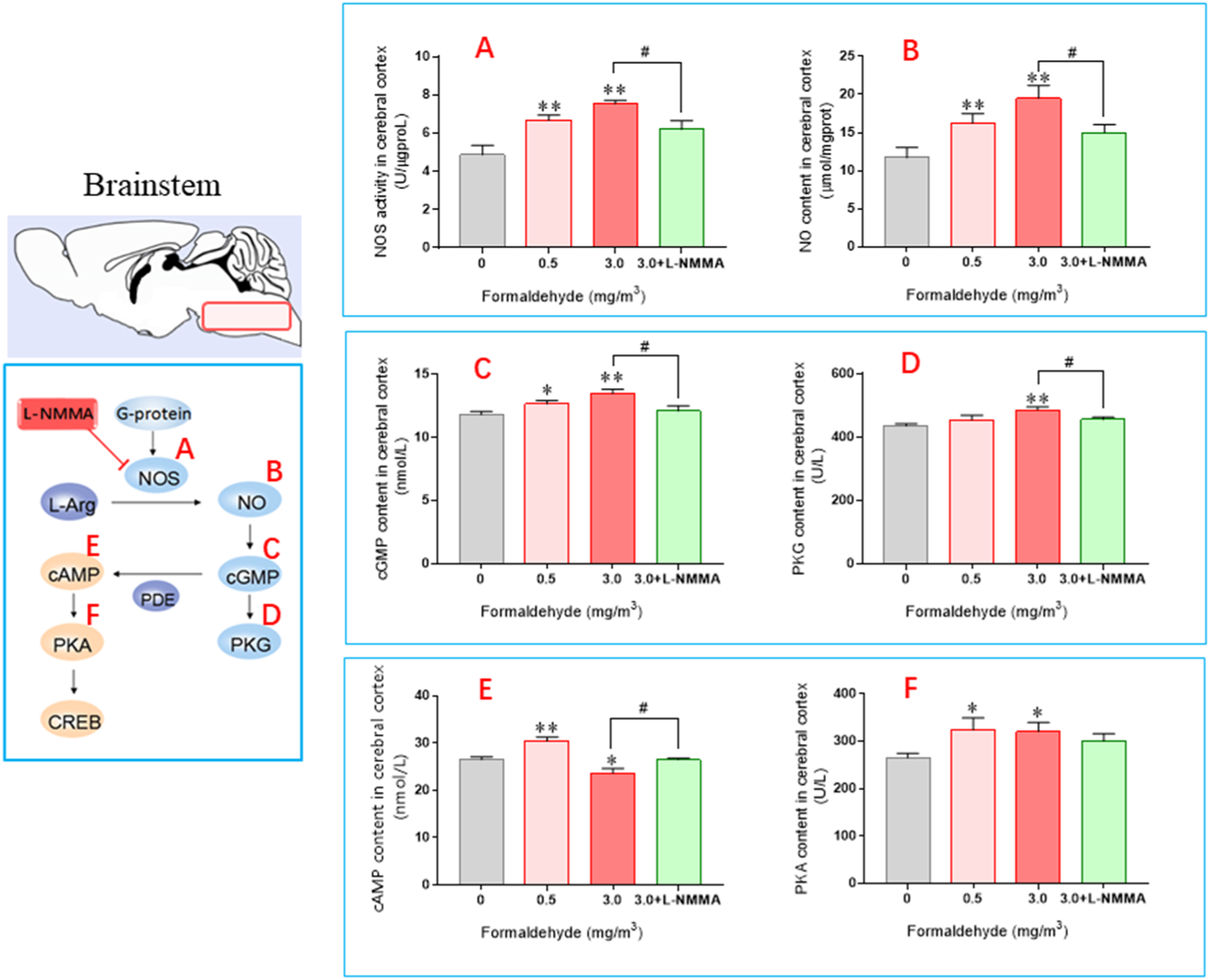
The study found that exposure to formaldehyde at concentrations of 0.5 mg/m3 and 3.0 mg/m3 resulted in significant increases in cGMP content, with the latter group showing a very significant increase (
The study found that cAMP level changes in the brainstem mirrored those in the cerebral cortex, increasing initially and then decreasing. The group exposed to 0.5 mg/m3 had significantly higher levels compared with the control group, while the group exposed to 3.0 mg/m3 had significantly lower levels. The +L-NMMA group had similar levels to the control group, but significantly higher levels compared with the 3.0 mg/m3 group. Additionally, PKA content increased significantly in the 0.5 mg/m3 and 3.0 mg/m3 groups compared with the control group, but did not change significantly in the +L-NMMA group compared to the control and 3.0 mg/m3 groups.
Discussion
According to a study conducted by Lu et al. in 2008, inhalation of gaseous formaldehyde at higher concentrations (3.0 mg/m3) can have detrimental effects on the learning and memory of mice. The study also found that such inhalation can alter the expression of NMDA receptors in the forebrain of mice. The study found that exposure to a high concentration of formaldehyde (3.0 mg/m3) caused up-regulated expression of NR1 and NR2B subunits and down-regulated expression of the NR2A subunit. However, when a lower concentration of FA (1.0 mg/m3) was examined, this effect was not observed. Therefore, the implication is that exposure to 1.0 mg/m3 of formaldehyde did not have obvious neurotoxicity, while exposure to a higher concentration can be toxic to nerves. The present study aimed to explore the neurotoxicity of formaldehyde mediated by the NO/cGMP- and cAMP-mediated signal transduction pathways. Three groups were exposed to different concentrations of formaldehyde (0 mg/m3, 0.5 mg/m3, and 3.0 mg/m3) for further analysis.
The brain is a complex system that efficiently integrates information from different parts of the body (Sporns et al., 2004). It is comprised of various regions, each with a primary function. The cerebral cortex regulates the body's movement, the brainstem maintains life by controlling important physiological functions such as heartbeat and breathing, and the hippocampus enables memory and learning (Maguire, 1997). The present study examined the cerebral cortex, hippocampus, and brainstem of mice.
The study's findings indicated that exposure to 3.0 mg/m3 gaseous formaldehyde can significantly alter the NO/cGMP and cAMP signaling pathways. While the 0.5 mg/m3 group did not exhibit obvious neurotoxicity in the exposed mice, there was still some degree of impact on the NO/cGMP and cAMP signaling pathways. This may be due to the activation of the body's defense system in response to low concentration formaldehyde exposure. The study found that there were significant increases in the content of different brain regions in the 3.0mg/m3 group compared with the control group (
PKG is a crucial protein kinase in the NO/cGMP signaling pathway, present in various tissues and cells of diverse organisms. It is considered as one of the central cGMP receptors. The activation of sGC by NO leads to the synthesis of cGMP, which in turn activates PKG to regulate different signaling pathways in the body (Denninger and Marletta 1999). The study's experimental results indicated that formaldehyde affects the NO content in the body, causing a shift in the flow of PKG through the signal transduction pathway of the cascade, and leading to abnormal PKG expression.
Exposure to gaseous formaldehyde resulted in significant changes in cAMP content in the cerebral cortex and brainstem, initially increasing and then decreasing compared to the control group. The high concentration group showed a significant reduction (
According to Erdogan et al. (2008), higher levels of stimulation strength lead to an increase in cAMP levels in the body. Properly elevated cAMP levels can prevent inflammation and protect tissues. In the event of a brain injury, the body releases neurotransmitters and free radicals, which activate the intracellular signal transduction mechanism and result in the opening of the blood–brain barrier. Studies have shown that increasing cAMP levels in endothelial cells can prevent the opening of the blood–brain barrier when exposed to low concentrations of formaldehyde, thus protecting tissues and preventing inflammation (Huang et al., 2021). However, exposure to high concentrations of formaldehyde can lead to excessive release of NO in the body, activating sGC and resulting in a significant increase in intracellular cGMP content. This can cause a decrease in cAMP content through the activation of PDE-2 or inhibition of PDE-3, leading to severe neurotoxicity in mice.
Study limitations: In this study, a significant limitation was limited pathological examinations. Further research should focus on obtaining more and better pathological examination results and their analysis.
Conclusion
According to the present study, formaldehyde exposure can cause brain cell apoptosis and neurotoxicity. The study also found that formaldehyde exposure can alter biomarkers of the NO/cGMP-cAMP signaling pathway. The study suggests that the negative impact of formaldehyde on the brains of mice is related to the regulation of the NO/cGMP-cAMP signaling pathway, as evidenced by the intervention of L-NMMA.
Footnotes
Acknowledgments
The authors thank the other tutors and students in the same laboratory for their help and cooperation.
Author contributions
Conceptualization, methodology, and formal analysis: SD, XY, and PM. Investigation: XH, FC, and WZ. Writing: XH, SD, XY. Funding acquisition: XY, SD. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (21577045), the Health Commission of Hubei Province scientific research project (WJ2021Z006) and Scientific Research Innovative Team of Hubei University of Science and Technology (2023T08).
Ethical statement
Data Availability Statement
Data available on request from the corresponding author, Professor Xu Yang (Hubei Key Laboratory of Genetic Regulation and Integrative Biology, School of Life Science, Central China Normal University, China), email:
