Abstract
Safety in consumer goods is maintained by product safety laws and associated regulations. However, the legislation and regulations are specific to product categories and legal jurisdictions, thus impeding one's ability to understand what a recall is and isn’t, and how it differs from related phenomena (e.g., product-harm crisis). The authors aim to provide such an understanding. They reviewed 510 reports from academics, managers, governments, and regulators; conducted interviews with 25 practitioners; and used 10 recall data sets to identify seven fundaments of recall. They synthesize the fundaments to propose a definition and a decision tree of recall, which can help inform academics, journalists, managers, lawyers, and safety advocates regarding what term is appropriate in what context. The authors apply the fundaments to identify similarities and differences between a recall and a harm crisis, the term used frequently in marketing research in association with recall. The fundaments also enable the authors to make five recommendations each for lawmakers and regulators in an effort to guide the academic and practitioner discourse on product recall.
The word recall, due to its negative connotations, appears to be the least preferred term in the trade. Such avoidance could downplay the seriousness of the negative impact on consumers.
—Authors’ interview with representatives of the Canadian Food Inspection Agency
On August 13, 2021, the U.S. National Highway Traffic Safety Administration (NHTSA) opened a formal investigation into 12 crashes related to a defect in the Autopilot feature of Tesla cars (Boudette 2021; NHTSA 2021a). Unlike other car manufacturers, which require owners to take their defective cars to dealers, Tesla updated the Autopilot software in its defective cars without informing the NHTSA and the affected owners (NHTSA 2021c). The NHTSA stated, “As Tesla is aware, the Safety Act imposes an obligation on manufacturers of motor vehicles and motor vehicle equipment to initiate a recall by notifying NHTSA” (NHTSA 2021c, p. 1; italics added). However, Tesla CEO Elon Musk (2023) has long maintained that the term “recall” is “anachronistic and just flat wrong.” In a world where software is an increasingly common component of physical products, Musk famously wrote in January 2014 that “the word ‘recall’ needs to be recalled” (Russolillo 2014, p. 1).
In another incident, following the crashes of multiple Boeing 737 Max airplanes, Boeing issued a “service bulletin” to operators (Boeing 2019), and the U.S. Federal Aviation Administration (FAA) released a “directive” (FAA 2019). However, consumer safety advocate Ralph Nader called for a recall of the aircraft (NPR 2019). Although Nader's usage of the term “recall” is consistent with the common vernacular, Boeing and the FAA used different terms. The U.S. Food and Drug Administration (FDA) uses the terms “recall,” “correction,” “market withdrawal,” “routine servicing,” and “stock recovery” for what seem to be similar phenomena related to unsafe health care products that the FDA regulates. 1 Lastly, in the United Kingdom, manufacturers and regulatory agencies alike use the terms “product safety report,” “product safety alert,” and “product recall” for notifications of defective and unsafe goods. 2
The preceding incidents exemplify at least two problems regarding recalls. First, with products becoming increasingly software operated (e.g., electric vehicles), manufacturers’ obligations with respect to recall might need to be revisited. Second, the legislation and regulation related to product safety and recall are specific to product categories and legal jurisdictions. The specificity is understandable because no legislation and regulation can be complete (Ghosh and John 2005). However, an unintended consequence of this specificity is that the legal and regulatory language has become inconsistent across product categories and jurisdictions and might contradict laypeople's understanding of product recall, as suggested by Tesla's and Boeing's examples. This piecemeal, inconsistent, and occasionally contradictory language has led to a proliferation of alternative terms for recall (Chappell 2013; Dowdell, Govindaraj, and Jain 1992; Linares 2013). These multiple terms adversely impact consumers’ “initial decision to pay attention” (Consumer Product Safety Commission [CPSC] 2003, pp. 8–10), a metric deemed consequential by the U.S. government (Federal Register 2016, 2018).
We reviewed 68 reports from governments and regulatory agencies, 36 reports and white papers from practitioners, and 410 published and unpublished manuscripts from academics. Next, we interviewed 25 representatives of three types of stakeholders: (1) 8 practitioners from eight manufacturing firms, insurance providers, and consulting firms; (2) 15 representatives from seven regulators from Australia, Canada, New Zealand, and United States; and (3) an employee each from two safety advocacy organizations. This exercise helped us identify the terms academics have used with recall. Next, we used ten recall data sets—three of which we received under the Freedom of Information Act—made available by seven regulators across five countries. These data sets cover U.S. recalls of aircraft, animal feed, biologics, consumer goods, drugs, food, meat and poultry products, medical devices, and vehicles; European recalls of consumer goods; Canadian recalls of food, medical devices, and vehicles; U.K. recalls of food and vehicles; and recalls of food in Australia and New Zealand. Our review offers the following contributions.
We use academics’ (Table 1), managers’ (Table 2), and governments’ and regulators’ (Table 3) definitions of recall; practitioner interviews (Web Appendix A); and recall data sets to identify seven fundaments of recall (Table WB1 in Web Appendix B) aimed to clarify and augment stakeholders’ understanding of the term (see, e.g., Figures 1 and 2). Importantly, we condense the seven fundaments to offer a parsimonious yet inclusive definition of recall. We also provide a decision tree (Figure 3) to help academics, journalists, managers, lawyers, and safety advocates discern what a recall is and—just as importantly—what it is not. We demonstrate the value of the definition and decision tree by undertaking two tasks. First, because the marketing term “product-harm crisis” overlaps with recall, we draw similarities and differences between recall and harm crisis. Second, we document how the limitations in the product safety legislation and regulation have led to three types of biases in the empirical findings (Table WB1 in Web Appendix B). This demonstration alerts academics and practitioners to the importance of knowing what legal term to use for what event.
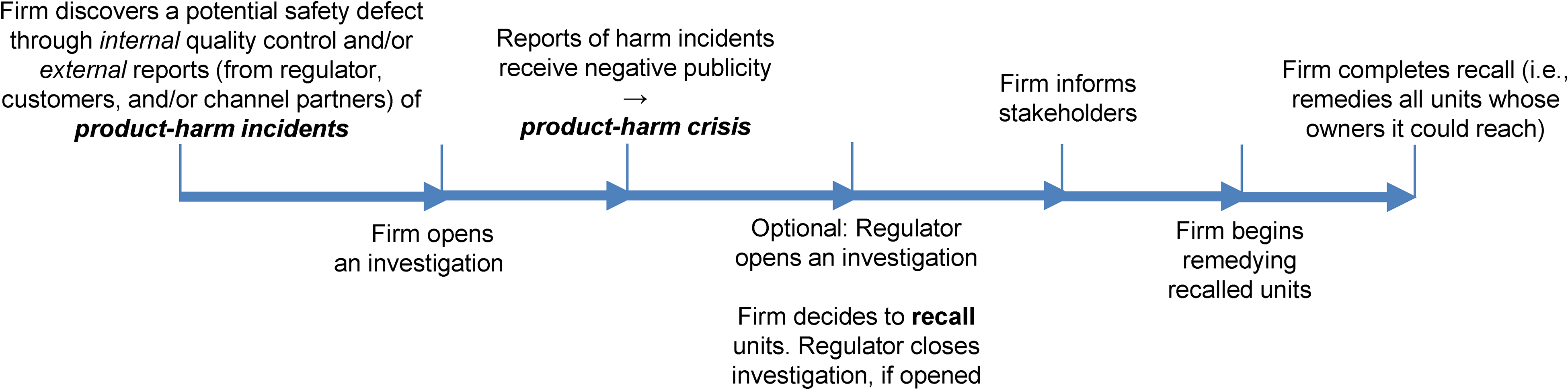
A Process Model of Product Recall.
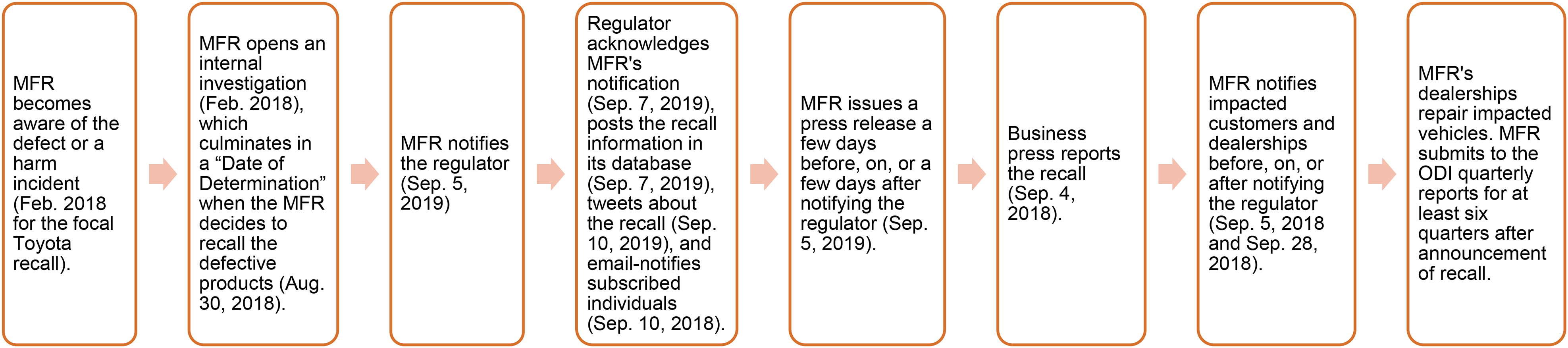
Application of Process Model to an Automobile Recall.
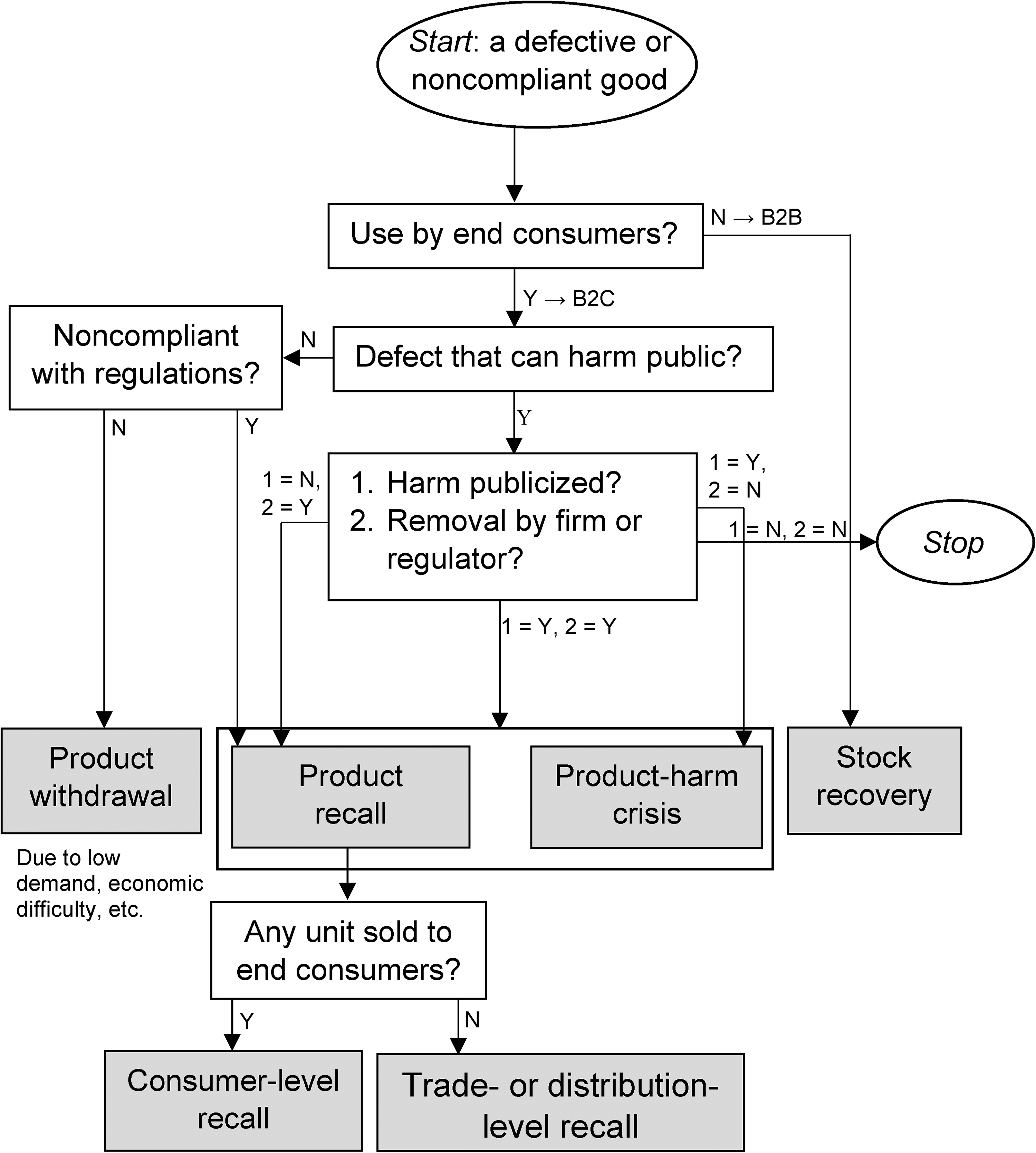
A Decision Tree to Distinguish Product Recall from Related Terms.
Academics’ Definitions of Recall and Related Terms.

Notes: We use bold typeface to highlight key words and phrases in the definitions. For a complete list of references in this table, see the Web Appendix.
Managers’ Definitions of Recall.

Notes: We use bold typeface to highlight key words and phrases in the definitions. For a complete list of references in this table, see the Web Appendix.
Governments’ and Regulators’ Definitions of Recall.
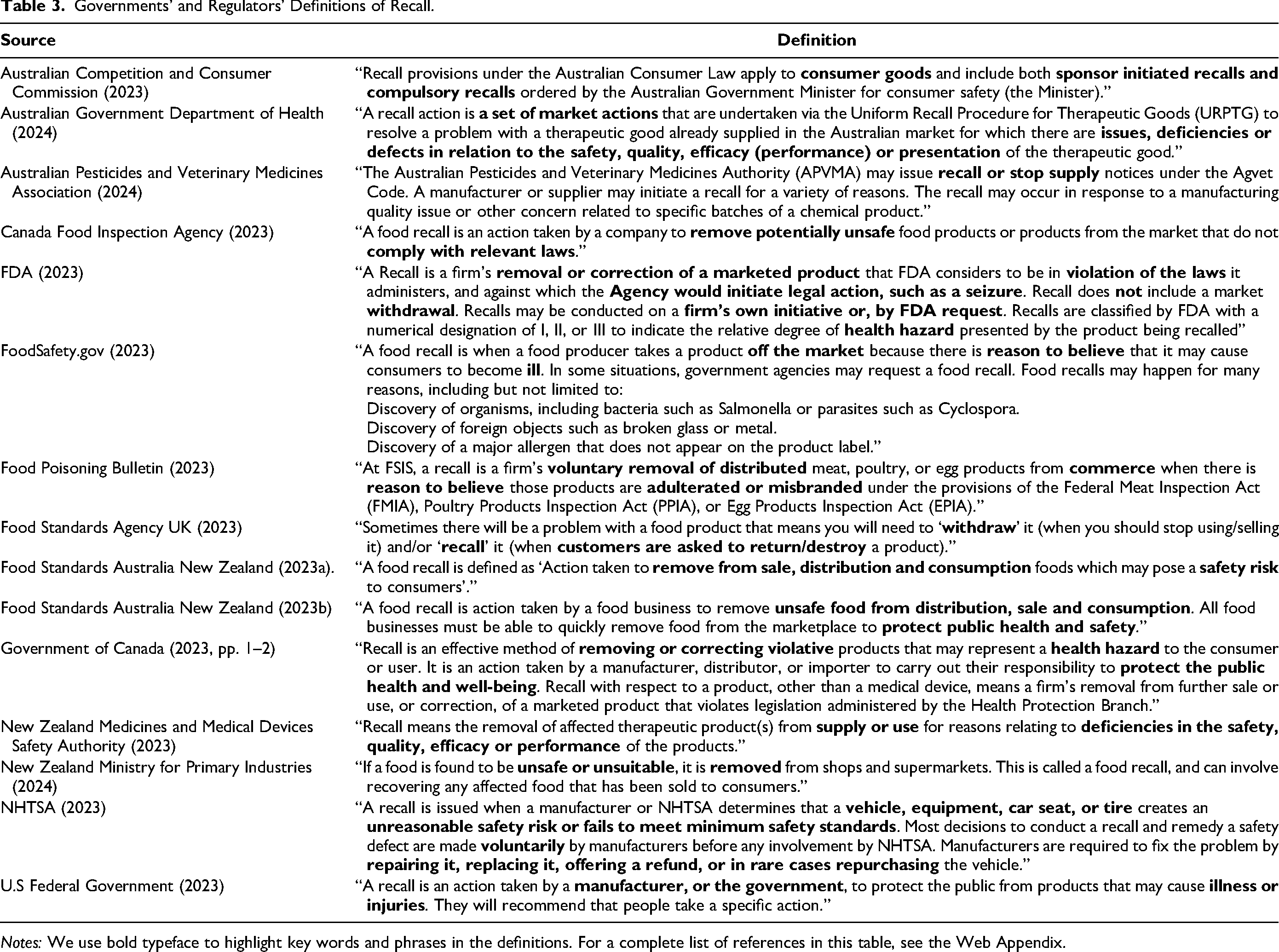
Notes: We use bold typeface to highlight key words and phrases in the definitions. For a complete list of references in this table, see the Web Appendix.
Next, we draw on the seven fundaments to make recommendations for lawmakers and regulators. While we provide suggestions to create a more comprehensive public policy on product recall, we acknowledge factors that limit lawmakers and regulators from achieving this completely. This acknowledgment is timely because more products than ever now include a software component. A software defect calls for an over-the-internet “update” rather than a physical recall of the product, per Elon Musk's argument (Klender 2023; Russolillo 2014). Yet, as the opening anecdote illustrates, the NHTSA is following the law in asking Tesla to inform the NHTSA before rolling out the software updates.
Our recommendations acknowledge that because products keep evolving, it is not possible to draft laws and safety standards that foresee all potential safety defects and violations. However, policies—legislative and regulatory—can still improve a firm's (1) awareness of what to do when it learns that its products are unsafe or noncompliant with safety standards and (2) subsequent remediation of the product.
Method: Database Development and Interview Protocol
We started with a review of the multidisciplinary and multistakeholder body of work on recalls and a descriptive analysis of ten recall data sets. Next, we conducted multiple interviews with managers and regulators. The review and interviews yielded a “thick description” of the recall phenomenon.
Database Development
We started developing the database of manuscripts on recall by first searching ABI/INFORM Global, EBSCO, Google Scholar, and JSTOR for articles with “product recall” in their title, abstract, or author-specified keyword list. To stay as current as possible, we supplemented the published articles with proceedings of the six leading academic management, marketing, and operations conferences. This effort yielded recall articles from 57 journals and 10 conference proceedings. We also posted messages on the mailing lists of marketing academics (ELMAR), operations researchers (the Production and Operations Management Society), and management researchers (the Strategy Division of the Academy of Management), soliciting unpublished research on recall. We also found ten masters’ theses, seven doctoral dissertations, and six book chapters. Lastly, we collected 36 white papers and reports from industry associations and advisory, consulting, and insurance firms, 3 and 68 reports from 13 government organizations from five countries/regions 4 (collectively referred to as practitioner papers).
Articles published in academic journals undergo page setting, allowing one to convert the PDF file of the article into a markup language (e.g., XML) format. We developed a Python program that took as input our database of PDF files of all journal articles. It converted each PDF file into an XML file and extracted the title, abstract, and author-specified keywords for each article. The program next created a term frequency–inverse document frequency (TF-IDF) matrix for the corpus and topic-modeled each abstract.
Researchers often substitute the word “product” in “product recall” with product-category-specific words (e.g., car/automobile recalls, poultry recalls, drug recalls). Similarly, branding research that considers the recalling brand (rather than the recalling firm or the recalled product) as its level of analysis uses the phrase “brand recall.” Research that emphasizes the tangibility of the recalled product uses the term “goods recall,” and studies that take a supply or distribution perspective to goods use “stock recall.” Lastly, researchers use “product-harm recall” and “safety recall”—terms that respectively emphasize consumer harm and the lack of safety as the underlying causes of the product recall—as synonyms of product recall. Therefore, we added all these terms to our search criteria.
In our review, we also learned that researchers use at least four terms interchangeably with “recall.” These terms are “crisis,” “withdrawal,” “seizure,” and “recovery.” We included these alternative suffix words in our search criteria as well (Web Appendix C discusses how recall differs from these constructs). At the end of this exercise, we identified the following nine terms that researchers have used interchangeably with product recall: brand crisis, brand recall, goods recall, goods withdrawal, product crisis, product-harm crisis, product seizure, product withdrawal, and stock recovery. Our database comprised 410 academic manuscripts, distributed as 242 published and 93 unpublished manuscripts, 30 manuscripts in conference proceedings, 38 doctoral dissertations, seven masters’ theses, and five book chapters across 12 academic disciplines. We also obtained 65 practitioner papers and 57 academic manuscripts on related phenomena (e.g., product-harm crisis, product withdrawal). Reading each paper in the database helped us develop a strong understanding of the domain of recall. We added to this understanding by conducting in-depth interviews.
Interview Protocol
The objective of the interviews was to elicit from practitioners facts and opinions about product recall and to reconcile their perspectives with our literature review. We identified the prospective interviewers from our reading of practitioner reports as well as white papers and reports published by the interviewee, either independently or on behalf of their employing organization. We contacted 64 organizations through email, phone, and social media. In our messages, we explained who we were and why we were contacting the organization, and cited the organization's reports on product recall, thus establishing why the organization was a strong fit with our research. We sent a reminder one week after the first message. Of these 64 organizations, 17 agreed to answer our questions. Table WA1 lists the interviewees’ job titles, 5 employer names, and reference recall articles that led to their selection.
We followed the interview approach used by marketing academics (e.g., Challagalla, Murtha, and Jaworski 2014; Grewal, Johnson, and Sarker 2007). This approach has been adopted by academics who have interviewed managers (e.g., Morgan and Zane 2022), regulators (e.g., Grier and Schaller 2020), and nonprofits (Nardini et al. 2022) regarding topics at the intersection of marketing and public policy. Specifically, at the beginning of an interview, the lead author explained the objective of the research, emphasizing that the information provided would be used for academic (i.e., noncommercial) research. Because interviewees expressed reservations about audio recording, we instead took notes as they answered our questions. Next, we sought information from the interviewees about their roles and responsibilities in their employer organizations. We asked whether they were comfortable letting us disclose their names, job titles, and employer's name. After that, the interview became a more directed conversation in which we guided the interviewees through the following questions:
Please cite an example of a product recall. What characteristics of this event qualify it as a recall? Please cite an example of an event that is similar to product recall but given a different label. What label is this event given and why? What characteristics of this event prevent it from being qualified as a recall? Does it matter if we call the event recall or something else? If yes, how does it matter, and for whom? What, in your opinion, are the current pressing managerially relevant questions or problems on the phenomenon of product recall?
We occasionally probed the interviewees to triangulate information from previous interviews we had conducted with other interviewees and from documents that we had read. We used the interviews to obtain verifiable facts from the interviewee's employer organization and the interviewee's opinions. Interviews lasted an average of 30 minutes. Lastly, the first author obtained feedback from the interviewees on the fundaments, definition, and decision tree derived from the interviews and literature. The interviewees were receptive to this material and believed it would be helpful to insurers, consultants, and product managers, thus validating our proposed fundaments, definition, and decision tree.
Product Recall and Related Terms: Definitions by Academics, Managers, Governments, and Regulators
We present academics’ terms and their definitions that relate to recalls, drawing three theoretical insights. Next, we consider managers’ definitions of recall, identifying three points of divergence. Lastly, we discuss governments’ and regulators’ notions of recall, highlighting three areas where their perspectives diverge. We use these points of divergence to propose our seven fundaments of recall.
Academics’ Use of Product Recall and Related Terms
A product recall involves interactions between a business and several of its stakeholders, such as product users, suppliers, distributors/retailers, journalists, lawyers, regulators, governments, and safety advocates. Consequently, recall has received attention from academics in business and nonbusiness disciplines. The upside of this attention is that academics have a truly transdisciplinary base of knowledge that can inform governments and businesses. However, the downside is that these disciplines have used different terms for the same concept and same terms for different concepts. Table 1 states the various terms that overlap with recall, the definitions of such terms, the disciplines that have used these terms, and representative articles in those disciplines.
We draw three insights from these terms and their definitions. First, academics have viewed recall from the theoretical lenses of crisis, controversy, transgression, failure, irresponsibility, performance, scandal, negative event, dubious behavior, wrongdoing, misconduct, publicity, and strategy. Whereas some terms frame the firm as the actor that caused the event (e.g., withdrawal, recovery), others frame the firm as an agency-lacking entity that is negatively impacted by the event (e.g., negative event, crisis). We subsequently elaborate on this difference in a section that teases out the similarities and differences between recall and harm crisis. Second, whereas product recall exists at the most disaggregate product unit of analysis, other terms consider brand, product market, and firm as the unit of analysis. Notably, each of these three units represents a higher order of aggregation relative to the product. Third, some terms take a capability/competence view, which suggests that the event is unintentional, whereas others (e.g., transgression, scandal) cast a moral/ethical perspective, which suggests intentionality.
Managers’ Use of Product Recall and Related Terms
Table 2 reports recall definitions by industry associations, management consulting/advisory firms, law firms, and safety advocacy organizations. The definitions vary on three phenomenon-related characteristics: whether they include (1) defect, noncompliance, or both as the cause of the recall, (2) whom the defect/noncompliance impacts and how, and (3) what types of remedy the recalling organization offers.
First, most sources state safety defects as the cause of recall. Few sources (e.g., Food Marketing Institute) acknowledge noncompliance with product standards as an alternative reason, and very few (e.g., Factiva) specify defect and noncompliance as potential determinants of a recall. Second, some definitions take the product's perspective, emphasizing that the defect/noncompliance could impede the product performance and thus impact the product user. Others take the product user's perspective, emphasizing that the product could harm users. Lastly, some definitions take the legal perspective, stating that the product manufacturer is recalling the unsafe product to avoid legal action. Third, some definitions state the types of remedy that the recalling firm offers. These include the recalling firm asking the product owners to return the recalled product and receive either a replacement product or a refund. Alternatively—and often, depending on the product category—the firm offers to repair the product free of charge.
Governments’ and Regulators’ Use of Product Recall and Related Terms
Table 3 documents recall definitions by governments and regulators in Australia, Canada, New Zealand, the United Kingdom, and the United States. We note three variations in these definitions. First, some governments/regulators include non-safety-related concerns as causes for recall. For example, Australia and New Zealand consider products’ (lack of) effectiveness and presentation as reasons for a recall, whereas Canada and the United States view recall as triggered only by a lack of safety. Second, some definitions state the stages of supply chain from where the product is recalled—that is, distribution, retail, and consumption. In contrast, other governments/regulators (e.g., Food Standards Agency UK) view recall as an action that retrieves defective products from consumption stage. Third, some governments (e.g., Australia) and regulators (e.g., FDA) state in their definition that the government can play a role in the initiation of recalls.
Table 3 highlights the inherent incompleteness of regulators’ definitions of product recall, owing to complexity of the phenomenon, changing landscape, and natural inability/unwillingness to consider all possible contingencies, i.e., “ink costs” (e.g., Wuyts and Geyskens 2005). It thus suggests the need to create a definition that transcends the idiosyncrasies of product categories and laws and can be used by managers, academics, and business stakeholders.
The Seven Fundaments of Product Recall
We synthesize recall definitions by academics, managers, governments, and regulators, with insights gained from the 25 practitioner interviews we conducted and our examination of 10 recall data sets to identify seven fundaments of recall (Table WB1 in Web Appendix B). The fundaments in turn allow us to create a process model of recall (Figures 1 and 2) and a decision tree that helps distinguish recall from related terms (Figure 3). Further, the fundaments enable us to draw similarities and differences between recall and harm crisis and identify three biases caused by not acknowledging the differences. Next, we quote academics, practitioners, and regulators to illustrate how we arrived at each fundament.
Fundament #1
The notion of recall exists in the vernacular as well as in the laws that govern the safety of the focal product category. It thus has a legal basis and can be associated with civil and criminal penalties.
6
Importantly, these laws (and thus the term recall) apply to physical goods and not to services. In addition, they apply to goods that individuals use (i.e., consumer goods or goods used by professionals) and not to goods that organizations use as intermediate components or parts. A recall is an action taken by a manufacturer, or the government, to protect the public from products that may cause illness or injuries. (USA.gov 2020) A recall is a firm's removal or correction of a marketed product that the FDA considers to be in violation of the laws it administers and against which it would initiate legal action (e.g., seizure). (FDA 2020) A recall is a firm's voluntary removal of distributed meat, poultry, or egg products from commerce when there is reason to believe those products are adulterated or misbranded under the provisions of the Federal Meat Inspection Act (FMIA), Poultry Products Inspection Act (PPIA), or Egg Products Inspection Act (EPIA). (Food Safety and Inspection Service [FSIS] 2020, p. 3) There is a reason why CPSC has the word consumer. You pick any safety regulator in the country that has supervised recalls … FSIS, NHTSA, CPSC, FDA … all these are goods that end-consumers buy. Federal Aviation Administration and Federal Railroad Administration regulate aircrafts and trains but the term “recall” does not apply to them because aircrafts and trains are bought for companies and not individuals. The FAA and the FRA still supervise their safety but the word ‘recall’ does not apply. The word ‘product’ in CPSC actually refers to tangible goods. By its very nature, a service cannot be recalled. (Author interview with a staff member from the Consumer Product Safety Administration)
A firm that manufactures, distributes, or sells a consumer good is liable for physical harm caused to the consumer or their property by a defect in the good (Patel and Reinsch 2003). 7 The end user can be laypeople or professionals. For example, an appliance manufacturer recalls defective units that laypeople use. Similarly, a medical device manufacturer recalls defective devices that health care professionals use. Indeed, the European Commission classifies each recall by whether the “product user” is a professional or a consumer; the latter refers to a nonprofessional user of the product (see https://ec.europa.eu/safety-gate-alerts/screen/search, accessed April 1, 2024). Relatedly, the term “recall” applies to goods and not to services (see Figure 3; Kashmiri and Brower 2016; Schwarcz 2013).
Marketing implications
Product-related laws—particularly those that center on consumer safety—have a natural connection with marketing. Surprisingly, we have found no marketing manuscript that leverages the legal basis of recall and the implications thereof. We believe that this is a missed opportunity. Further, phrases such as “removal or correction” and “a firm's voluntary removal” suggest that a recall involves firm voluntary disclosure (Astvansh, Ball, and Josefy 2022). Relatedly, a recall involves interactions among businesses, lawyers, politicians, journalists, and regulators. Therefore, recall can serve as a substantive context to develop theory at the intersection of marketing and public policy, marketing and law, and marketing and politics—broadly, marketing and nonproduct market strategy (Astvansh, Wang, and Shi 2022).
Fundament #2
A consumer good must be recalled if it (1) has a defect
8
that creates a substantial risk of injury to the user (i.e., safety defect) and is noncompliant with regulations, (2) does not have a safety defect but is noncompliant with a regulation that is not safety-related, or (3) is compliant with regulations, but has a safety defect. A recall is issued when a manufacturer or [regulator] determines that a [product] creates an unreasonable safety risk or fails to meet minimum safety standards. (NHTSA 2024) A recall may be issued if a [product] (a) is a health hazard, (b) is mislabeled or packaged poorly, (c) is not what it says, (d) is poorly manufactured. (Marks 2020) The [product] … maybe hazardous to health … or may not meet the requirements set in the Act or the Regulations. (Government of Canada 2024). A nut processing company recalling packages of mixed nuts because they may contain undeclared peanuts. Anyone with an allergy or severe sensitivity to peanuts who eats this product may have a serious or life-threatening reaction. People can be allergic to peanuts and still be able to eat tree nuts, which include pecans, walnuts, Brazil nuts, almonds, pistachios, macadamia nuts, and cashews, among others. Since the product was produced and labeled to not include peanuts but it did contain them this presents a potential for bodily injury and the product would need to be recalled immediately. This is characterized as an undeclared allergen. Another example of a product recall would be an accidental contamination. Look no further than the romaine lettuce contamination from Nov 2018. There was a multistate recall for romaine lettuce that was produced in Northern California. This qualified as a recall because the product tested positive for E. coli and there were many sicknesses reported. (Authors’ interview)
When a consumer good has a safety defect and is noncompliant with regulations, it becomes the most eligible candidate for a recall. For example, on March 12, 2008, General Motors recalled vehicles that had a defective tire pressure monitoring system, which “could result in a loss of control of the vehicle” and was thus a safety hazard (NHTSA 2008). Another (less acknowledged) situation is when the good does not have a safety defect but is noncompliant with a regulation that is not safety-related. For instance, on February 22, 2016, Medtronic recalled its medical device “because it was commercially distributed without a cleared premarket [approval] from” the FDA (FDA 2016). Lastly, a situation arises when a good has a safety defect but is still compliant with the existing regulations. For example, in 2014, Tesla found that the charging equipment of its cars could overheat, posing a risk of fire (NHTSA 2014b). Although its cars complied with the regulations, they still posed a safety risk. Elon Musk argued that because Tesla does not require owners to return the cars physically, the company's action should not be termed a recall (Rayman 2014). Regulators, journalists, and safety advocates—stakeholders that report on recalls—may consider including in their reports whether a recall is caused by a defect or noncompliance. Such disclosure would offer greater transparency to business stakeholders such as consumers, politicians, and investors.
Marketing implications
This fundament of recall can help develop and test the theory of corporate responsibility. A defect indicates that a firm's quality-control process missed detecting a flaw and thus represents an act of omission. In contrast, noncompliance—as the term suggests—indicates an act of commission and violation (Copeland, Jackson, and Morgan 2004) and thus places greater responsibility on the firm (Stäbler and Fischer 2020). This nuance is theoretically rich, as it captures a firm's active (vs. passive) behavior. The recall literature has focused on defects as the underlying reason for recall, with no attention (to our knowledge) to noncompliance. This is surprising, because the CPSC and the FDA provide data 9 on noncompliance and the concomitant civil and criminal penalties. Our reading of the data suggests that some noncompliance—for example, the Medtronic case involving sales before the regulator approved the product—is of particular relevance to the marketing discipline. Lastly, this fundament helps clarify that a software update can be a recall if it fixes a safety defect or addresses noncompliance. Because more products now have a software component, manufacturers can address the products’ safety defects and noncompliance over the internet. Thus, consistent with Musk's argument, it would be worthwhile to focus on the means of physically delivering the product back to the manufacturer as opposed to interpreting the term “recall” literally.
Fundament #3
A recall need not be the sole purview of the manufacturer. Other value chain participants may also initiate recalls. [A] manufacturer or distributor initiates the recall to take [products] off the market. (FSIS 2020) Remove product from commerce (e.g., by manufacturers, distributors or importers). (Grocery Manufacturers Association 2010, p. 7) The recall strategy will specify the level in the distribution chain to which the recall is to extend. In general, Class I recalls may extend to the consumer or user level, Class II recalls may extend to the retail level, and Class III recalls may extend to the wholesale level. (FDA 2020)
Although usually it is the manufacturer that recalls a good, other supply-chain members (distributor, importer, or retailer; Hora, Bapuji, and Roth 2011) or even the regulator can initiate the recall. Although the legal liability resides with the manufacturer (including importer and distributor), any member of the value chain can remove the unsafe product from distribution and potential consumption. Indeed, the CPSC data include a categorical variable identifying whether the recalling firm is a manufacturer, importer, distributor, or retailer (see https://www.cpsc.gov/Recalls). Similarly, the European Commission's Rapid Exchange of Information System (also known as Safety Gate) reports whether the recall was initiated by manufacturer, retailer, or importer. 10
Marketing implications
Game theorists and structural modelers can view a recall as a strategic game among various members of the supply chain and/or among regulators, with each party maintaining some private information (Çolak and Bray 2016; Singh and Grewal 2023). Further, a firm can work simultaneously with regulators from neighboring countries to coordinate a recall at the broader regional level. 11 International business academics can use this fundament to examine when and how firms, their downstream intermediaries, and regulators might implement a cross-border recall.
Fundament #4
A recall comprises multiple managerial decisions, of which recall initiation is the most visible. A recall action is a set of market actions. (Australian Government Department of Health 2024) Manufacturers are required to fix the problem by repairing it, replacing it, offering a refund, or in rare cases repurchasing the vehicle. (NHTSA 2024)
A product's recall involves many decisions that its manufacturer, retailers, consumers/users, and the regulator undertake (Figure 1 provides a process model of recall). The most visible and legally required decision is the firm's notification to the related safety regulator—that is, recall initiation. Per product liability law, the “firm” here is the entity that is legally liable for selling a product that can potentially harm consumers. While notifying the regulator is the first step, the legislation is unclear on when the firm must inform the regulator. Recent evidence suggests that firms “might intentionally report recalls late to help stock prices” (Seto 2021). Another key decision that affects the manufacturer's, retailers’, and customers’ cost–benefit calculus is the remedy the manufacturer offers—for example, repair, replacement, disposal, or refund (Liu, Liu, and Luo 2016; Mafael, Raithel, and Hock 2022). Ideally, a manufacturer must notify retailers and customers when it has sufficient evidence to believe that the product may be unsafe for users.
Other decisions include managing reverse logistics; notifying stakeholders such as affected users, retailers, and investors; and, in extreme cases, testifying before the government. However, the process begins when the firm discovers a potential defect (Wowak et al. 2021) and ends when the firm has remedied all defective product units (Pagiavlas et al. 2021). Indeed, the NHTSA and the FDA provide data on when and how the firm first discovered a defect, the steps it took to investigate the defect, which suppliers and internal departments the firm involved in the investigation, and so on (Ni and Huang 2018). All these are decisions that can affect recall outcomes (e.g., speed of remedying recalled units, learning) and involve multiple stakeholders, including suppliers, regulators, consumers, retailers, risk insurers, and society.
Marketing implications
This fundament helps us propose a process model of recall (Figure 1) and demonstrate its application in the context of automobile recalls (Figure 2). Although prior research in operations (e.g., Ni and Huang 2018) and marketing (e.g., Liu and Shankar 2015) has offered anecdotes hinting at this process, we extend these anecdotes to offer a process model, which suggests how a recall fits in the life cycle of a product. Future research can use this novel fundament of recall in several ways. For example, machine learning academics can mine a recalling firm's notices to the regulator, the affected consumers, and the dealers and explore the synergy (or lack thereof) with respect to how a firm communicates a recall to different stakeholders. 12 Academics and practitioners can use the data on consumer complaints to determine how long a manufacturer waited to notify retailers and customers.
Fundament #5
A firm's motivations to initiate a recall include (1) demonstrating its responsibility toward consumers, (2) mitigating the risk of product liability, or (3) both. [A recall is] a company's removal or correction of a marketed product … against which a government agency could initiate legal action. (Squire Patton Boggs 2015) [A recall] should be considered as a risk mitigation activity. (Australian Pesticides and Veterinary Medicines Association 2024) A recall is an action … to protect the public from products … that may cause health or safety problems. (USA.gov 2020)
Although recall has a legal basis, it includes a firm's voluntary behavior that demonstrates its responsibility toward its consumers and the public at large. Specifically, a firm may initiate a recall motivated by this responsibility. Conversely, a recall can be an economic decision intended to mitigate the manufacturer's risk of product liability (Krulwich 1984). Interestingly, because negligence 13 laws vary across U.S. states, a firm's product liability differs by state. Of the 50 U.S. states, 4 states and the District of Columbia have adopted the contributory negligence law, which denies a plaintiff (i.e., harmed consumer) any damage compensation if they have been even slightly negligent. All other states have adopted the comparative negligence law, which in turn can be of two types. Thirteen states have the pure comparative negligence law, which awards a plaintiff compensation based on their relative fault. The remaining 33 states use a modified comparative negligence law, which awards a plaintiff compensation if their fault exceeds a specified threshold (50% for 12 states and 51% for 21 states).
Marketing implications
In states that have adopted contributory negligence or modified comparative negligence law, courts set a relatively low standard of proof that consumers neglected returning the recalled products. Consequently, a firm headquartered in these states is more likely to recall defective goods than in states that have implemented a pure comparative negligence law (Zhang et al. 2022). Future research can exploit this legal difference and discontinuity in the threshold across states to test the theory on firm supererogation or volunteerism—that is, whether the firm's recall is driven by the logic of responsibility or the logic of liability (Cavazos, Rutherford, and Berman 2018).
Fundament #6
Not all safety regulators have the legal authority to mandate recalls. Among those that can, the preference is to influence rather than mandate. As a result, a firm's recall could be voluntary/volitional or involuntary—that is, mandated by regulators. The FDA lacks statutory authority to force a recall. Section 1412 of [Title 15] authorizes … the NHTSA to force the manufacturer to notify interested parties and to remedy safety-related defects. [The CPSC] can order that the product be brought into conformity with the standard, be replaced, or that refunds be offered. (Krulwich 1984, pp. 759–70)
Safety regulators enforce product safety laws that vary by product category. However, not every law grants its enforcing regulator the authority to mandate recalls (Copeland, Jackson, and Morgan 2004; Schwartz and Adler 1983). For example, the FDA has the authority to compel recalls of food and medical devices but not that of drugs. When the regulator cannot legally mandate the recalls, it uses its authority to nudge or recommend the firm to initiate the recall voluntarily. We used our data sets to check the term that each regulator uses for what academics call mandatory/involuntary recalls and how such recalls have changed over time. We conclude that among the regulators that have this power, few really exercise it. For example, between 1966 and December 5, 2021, the NHTSA supervised 25,450 recalls, out of which it classified 21% as “influenced.” Importantly, this percentage has slowly increased over time. However, between October 1, 2002, and October 31, 2020, the FDA classified merely .34% of recalls as “mandated” or “influenced”—a percentage that has waned over time. Similarly, regulators in Australia, Canada, Europe, New Zealand, and the United Kingdom rarely mandate a recall. Perhaps as a consequence, safety advocates have accused the safety regulators of kowtowing to firms (e.g., Consumer Reports 2010; Plungis 2018).
Marketing implications
This sixth fundament of recall clarifies regulators’ limitations in surveilling firms and monitoring product safety as well as the legal bases for these limitations (The Center for Public Integrity 2008; Ivory 2015). Game theorists can use the context to study the strategic interactions between a regulator and a firm (Çolak and Bray 2016), what incremental information each party shares with its counterpart, and whether this information ends the game or takes it to the next level. For example, the law requires vehicle manufacturers to submit early warning data regularly to the NHTSA, 14 which the NHTSA uses to decide whether and when to open and close a regulatory investigation into a defect. 15 These are novel data that have not yet been studied.
Fundament #7
Although safety defects and noncompliance are often the outcome of suppliers’ and manufacturers’ negligence, they could also be the result of opportunism and intentional misconduct, which are now more easily detectable, thanks to more regulators providing open application programming interfaces (APIs). [Product recalls are] incidents where organizations negligently or deliberately release flawed or unsafe products into the marketplace. (Hersel et al. 2019, p. 552) A recall is a firm's action to remove product from commerce (e.g., by manufacturers, distributors or importers) to protect the public from consuming adulterated or misbranded products. (Grocery Manufacturers Association 2010, p. 7)
Often, safety defects are the outcomes of a supplier's or a manufacturer's negligence. However, higher traceability of a product's components and activism by nonprofit organizations have made it easier for regulators (manufacturers) to catch manufacturers’ (suppliers’) opportunism and deliberate misconduct. For example, in 2013, West Virginia University's Center for Alternative Fuels Engines and Emissions reported to the U.S. Environmental Protection Agency (EPA) that Volkswagen's vehicles were emitting more nitrogen dioxide than allowed. In response, the EPA found that Volkswagen deceived consumers by cheating on emissions tests (EPA 2019; more broadly, visit https://www.epa.gov/enviro/tri-ez-search). In another incident, several automobile manufacturers found that Takata “routinely manipulated results of airbag inflator tests” (Trudell and Fisk 2016, p. 1). Academics have posited that organizational networks offer suppliers and manufacturers the opportunity and motivation to commit such opportunism and fraud (Ball, Shah, and Wowak 2018). Indeed, car manufacturers have been accused of underreporting their vehicles’ emissions (Carrington 2015; Contag et al. 2017). In another incident, Aston Martin recalled 75% of its sports cars manufactured between 2007 and 2012 because a tier-three supplier used counterfeit raw plastic instead of the specified DuPont plastic (NHTSA 2014a). The potential for counterfeiting means that product manufacturers must watch out for not only their genuine yet defective goods but also their counterfeit counterparts. Detecting misconduct has become easier because more regulators have started offering open APIs to make easier public access to their databases. For example, the Consumer Financial Protection Bureau, CPSC, FDA, FSIS, and NHTSA now provide APIs to allow business stakeholders to improve access to their database. 16 Further, these regulators upload data more frequently than they used to in the past. For example, the European Commission's Rapid Exchange of Information System website reports multiple cases of recalls of counterfeit products (see, e.g., European Commission 2023d).
Academic and managerial implications
Theoretically, a recall in the aftermath of misconduct is particularly diagnostic because the firm undertakes it not out of concern for public safety but to limit government and public sanction. This fundament can thus inform how managers’ interpretation of stakeholder saliency (legitimacy, urgency, and power) and stakeholder claims (alignment or conflict) impact how the firm responds, ranging from accommodative to defensive (Hersel et al. 2019).
Definition of Recall
We distill the seven fundaments to define product recall as an organization's actions—legally mandated or voluntary—intended to prevent from use consumer goods that have a safety defect and/or are noncompliant with regulations. We intend our definition to be parsimonious yet inclusive of academics’, managers’, and regulators’ notions of recall discussed in the preceding section. This synthesis of multiple stakeholders’ perspectives and languages adds value, offering a comprehensive viewpoint on recall. Lastly, our proposed definition casts recall as an organizational phenomenon that has a legal basis with political and social implications. We use the definition to create a decision tree (Figure 3), which we hope can direct stakeholders to the proper term for their phenomenon of interest.
Similarities and Differences Between Recall and Harm Crisis
Our seven fundaments and definition of recall help distinguish recall from harm crisis. 17 Dawar and Pillutla (2000, p. 215) provide the most cited definition of harm crises: “discrete, well-publicized occurrences wherein products are found to be defective or dangerous.” As our decision tree (Figure 3) illustrates, a harm crisis requires two conditions. First, the focal product must have a defect that can cause bodily harm to its users and observers. The harm may be potential or may have already occurred (the literature refers to the latter case as a “harm incident”). Second, the defect information must have become public, usually through news/social media. 18 The literature on harm crisis does not define the word “crisis.” So, some researchers prefer to use the term “product safety issues” instead (Hersel et al. 2019). Based on the vignettes and observational measures used in the harm crisis literature, we infer that crisis refers to high volume and negative valence of news following public-harm incidents. We identify one similarity and three differences between recall and harm crisis (Figure 3).
Similarity
Some researchers (e.g., Liu, Shankar, and Yun 2017; Zhao, Zhao, and Helsen 2011) consider both a harm crisis and a recall as a marketing crisis (Dinner, Kushwaha, and Steenkamp 2019) or an organizational crisis (Clark 1988; Pearson and Clair 1998). However, following previous research (Grewal, Johnson, and Sarker 2007), an organizational crisis refers to an event that (1) has a low probability of occurrence, and (2) threatens the existence of the organization/brand (Grewal, Johnson, and Sarker 2007). Neither harm crisis nor recall meets either criterion.
Implications of the similarity
This nuance can inform the emerging literature on marketing crisis (Dinner, Kushwaha, and Steenkamp 2019) and position it within the broader theory on organizational crisis and its management. Dinner, Kushwaha, and Steenkamp (2019) have redefined the term “marketing crisis” as a publicized negative event “stemming from marketing-mix-related activities” (p. 339). This definition is closer to Dawar and Pillutla's (2000) notion of harm crisis and different from Pearson and Clair's (1998) consideration of organizational crisis as an event with low probability and existential threat. Future research can develop and test these emerging ideas, using recall as a context.
Difference #1
A harm crisis is an unfavorable circumstance in which a firm finds itself, whereas a recall is the firm's corrective response to the circumstance (Hersel et al. 2019). That is, a recall is a solution to a problem (product safety defect and/or noncompliance) and not a problem in itself. Three vehicle recalls initiated in the United States illustrate this difference (see next subsection). The 1996 NHTSA–Chrysler incident is a harm crisis, but not a recall. The 2010–2013 NHTSA–Chrysler episode is an example of a harm crisis and a recall. Lastly, the 2013 Tesla example involves a recall, but no harm crisis (Figure 3).
Implications of the difference
This difference emphasizes that recall is an endogenous decision, and firms strategically decide whether to initiate a recall, and, if yes, when to announce it (see Mukherjee et al.’s [2021] research on herding in recall announcements). Acknowledging recall as a choice variable would enable event studies (e.g., Liu and Shankar 2015) and marketing-mix studies (Cleeren, Dekimpe, and Helsen 2008; Cleeren, Van Heerde, and Dekimpe 2013; Gao et al. 2015; Van Heerde, Helsen, and Dekimpe 2007; Zhao, Zhao, and Helsen 2011) to control for the endogeneity of the timing of the announcement and the marketing-mix adjustments that a recalling firm makes. Such control could offer different empirical insights.
Difference #2
Whereas a harm crisis can occur only if the focal product has a safety defect that has harmed consumers, a recall can follow an instance of noncompliance as well (Figure 3). For example, on May 21, 2017, Volkswagen recalled model-year 2017 Jetta cars because the cars’ Vehicle Identification Number (VIN) markings on the body did not match the VIN plate near the windshield (NHTSA 2017). The VIN mismatch could not have compromised public safety and hence could not have caused a harm crisis.
Implications of the difference
This difference highlights the notion that the causes of a recall (safety defect, noncompliance, or both) are broader than the causes of a harm crisis (safety defect). Further, unlike a defect, noncompliance involves a more active role played by the law and its makers and enforcers, making recall a broader phenomenon than a harm crisis.
Difference #3
Both harm crises and recalls occur in the aftermath of harm incidents. However, a recall is a firm action, whereas a harm crisis refers to publicity surrounding the harm incidents (Figure 3). For example, Gokhale, Brooks, and Tremblay (2014) measured Toyota's stock returns to four events related to Toyota's sudden unintended acceleration defect. The first event is Toyota's minor recall. The researchers found that the stock return to this recall is insignificant. The second event is an accident that received massive publicity. Perhaps surprisingly, the return to this event is also insignificant. The third is Toyota's major recall, to which return is significantly negative. Lastly, NHTSA's report stating that it did not find any defect resulted in positive stock returns for Toyota. This article exemplifies that a harm crisis can involve multiple events, including public-harm incidents, regulatory events, and potentially multiple recall announcements.
Implications of the difference
This feature emphasizes the difference between recall and recall announcement. A recall is often not announced, and thus the broader public (including consumers and investors) usually do not become aware of a recall. We searched Factiva for recall articles published in the Wall Street Journal and received an average of four unique recall articles per year. However, news organizations can follow regulators' databases and social media pages to become aware of recalls and subsequently disseminate the information for the public benefit. Such news coverage of recall is unrelated to a harm crisis.
Three Types of Biases Caused by Not Differentiating Between a Recall and a Harm Crisis
A harm crisis occurs when reports of product-harm incidents receive negative publicity (i.e., harm publicity). In contrast, a recall is a firm's decision to remove a defective or noncompliant consumer good from the distribution chain and consumers. Accordingly, a recall announcement embeds three potential effects: (1) confirmation of a defect or noncompliance (negative effect), (2) acknowledgment of recall costs 19 (negative effect), and (3) prevention of future harm (positive effect). Which of these effects dominates depends on how much negative publicity has preceded the recall announcement and the estimated costs of recall, and whether academics’ empirical specifications include recall publicity and costs.
Empirical marketing research on recall has measured the effects of a recall on the recalling brand's and its rivals’ sales and/or stock returns. Not differentiating between a recall and a harm crisis can lead to one or more of three biases in such research. We briefly discuss these three biases in the context of sales and stock returns as outcome variables.
Partial effect bias (downward)
This bias is caused when the academics seek to estimate the total effect (on sales or stock return) of a harm crisis (see Figure 1), but they sample a shorter time frame, often the one closer to a recall announcement. For example, to measure the total effect of the harm caused by Toyota's unintended acceleration defect, one must consider the period beginning from the first harm incident and ending after the last recall in the sequence of recalls. Consequently, academics measure a partial effect of the crisis. This downward bias in the total effect is unrelated to the outcome variable or the level of temporal aggregation (i.e., daily, weekly) of the explanatory variable and the outcome variable. In that sense, this bias is analogous to academics who misestimate the returns to the launch of a new product as the total returns to innovation, as shown by Sood and Tellis (2009) and Warren and Sorescu (2017).
Data interval bias (downward)
This bias is caused when academics seek to estimate the effects of announcements of recalls (explanatory variable) on sales or stock returns (outcome variables) without controlling for the publicity of harm incidents that may have led to the recall. The omitted harm publicity leads to endogeneity bias in the academics’ estimates. This bias exists only if the explanatory variables and outcome variables are aggregated at a level higher than daily and is thus common in sales studies. Daily values, for example, in event studies, allow for a difference-in-differences estimate, which does not suffer from endogeneity bias caused by the omission of harm publicity.
Endogeneity bias (downward/upward)
This bias is caused when recall-level regressors (e.g., the number of units recalled, recall initiation strategy, time to recall) are assumed to be exogenous to the empirical specification. As our definition suggests and empirical recall research shows (Chen, Ganesan, and Liu 2009; Eilert et al. 2017), these regressors are choices that firms make. Not accounting for their endogeneity leads to biased estimates. Whether the bias is downward or upward depends on the particular regressor and its correlation with the explanatory variables and the outcome variable. This bias exists in event studies that use the raw (as opposed to unexpected 20 ) values of the recall-level regressors. Similarly, the bias occurs in sales regressions if the endogeneity of recall-level regressors is not corrected for. Lastly, stock return response models should consider the unexpected value of recall magnitude (the number of recalls in a year).
Recommendations for Public Policy
We draw on our fundaments to make five recommendations each for lawmakers and regulators. Because products are incorporating more technology and evolving at an unprecedented rate, laws and standards for product safety may not keep up with what is needed. That is, laws and standards cannot realistically be complete. Our recommendations acknowledge and explicitly account for this innate incompleteness in laws for product safety. 21
Recommendations for Lawmakers
Define recall and clarify what it is for the public. Our fundament #1 leads us to recommend that federal governments provide a definition of recall that encompasses product categories, laws, and regulators and thus offers for the public a general understanding of what a recall is. Interestingly, only the Government of Canada and the U.S. federal government define recall (Table 3). Recently, the U.S. federal government seems to have taken a step back on defining recall. The nonprofit organization Internet Archive (https://web.archive.org) reports that the U.S. federal government introduced the website https://www.usa.gov/recalls on September 7, 2015 (https://web.archive.org/web/20150907005341/https://www.usa.gov/recalls), which provided the U.S. government's definition of product recall. However, the website no longer exists. The website's last internet archive is dated March 31, 2023 (https://web.archive.org/web/20230331220621/https://www.usa.gov/recalls). Further, the website removed the definition of product recall between November 29, 2020, and December 19, 2020. 22 We believe a government's definition of product recall is critical to stakeholders’ understanding of the term. The U.S. government not only delisted its previous recalls page (https://www.usa.gov/recalls) but also does not publish the definition on its alternate website (https://recalls.gov/).
As Table 3 highlights, governments vary on whom they are defining recall for. For example, the U.S. government defined recall as “an action … to protect the public from products that may cause illness or injuries.” We appreciate the focus on why a product is recalled, suggesting that the definition is directed at the public. However, the U.S. government defined recall as “an action taken by a manufacturer or the government.” As our fundaments highlight, this definition is incomplete, because a recall can be initiated by a retailer, a distributor/wholesaler, or an importer, without any directive from the manufacturer or regulator. More importantly, which member of the supply chain recalls the product is less relevant for the public. The Canadian government's definition of recall is a case in point. It provides only the information that is relevant to the public and avoids irrelevant details, such as the initiator and related laws. We thus recommend that the federal government replace in its definition the word “manufacturer” with the word “business,” lest the public assume that a retailer's recall, for example, falls outside the government's definition.
Clarify that recall is an action triggered by safety and/or noncompliance concerns and that withdrawal and recovery are distinct from recall. The agencies of Australia and New Zealand include performance and presentation as reasons for recall, whereas Canada, the European Commission, the United Kingdom, and the United States reserve the term “recall” for an action to address safety concerns and noncompliance. Consistent with the broader use, and drawing on fundaments #2 and #3, we suggest limiting the term “recall” to action triggered by safety concerns or noncompliance with product standards. To the extent that product performance and presentation is coded in product standards, “recall” can apply to these non-safety-related issues. An alternative is to use “safety recall” to emphasize to retailers and customers that the product is being recalled because it is unsafe for use. The “safety” prefix would help draw attention and mitigate harm. Although the labels for the action are semantic, emphasizing the cause of the action would help answer safety advocates’ questions about the differences among these terms (e.g., Dingley 2013) and assist managers (Linares 2013) and journalists (NPR 2013) in choosing the appropriate term. This emphasis can also help draw customers’ attention to the recall and lower incidence of product harm (Copeland, Jackson, and Morgan 2004; Jackson and Morgan 1988).
Specify timing and content of a recall notice to affected product users as well as how the notice must be delivered. Fundament #4 leads us to recommend that laws specify the recalling organization's timing and the content of a recall notice. Specifically, laws should guide a firm on how it could determine that the evidence is sufficient to warrant a recall. On the one hand, a premature recall—based on insufficient evidence—could create panic among retailers and product owners. On the other hand, a delayed recall could cause harm. Drawing on fundaments #5, #6, and #7, we recommend that the recall notice to the product owner include a summary of the evidence that led the recalling firm to determine that the product is unsafe. The recalling firm may have received a harm-incident report from product users, retailers, and/or regulators. We recommend that recalling firms disclose the number of incidents and the levels of harm (e.g., injuries) caused in these incidents. Alternatively, the firm's or its supplier's internal quality checks may have led to this determination. Disclosing this information will alert readers to the likelihood and magnitude of harm that the product may cause. Users of the product may then decide whether and when to use the remedy. In addition, the disclosure will allow users to determine how proactive (vs. reactive) the firm has been in owning its responsibility to provide safe products. Indeed, consistent with our suggestion, the NHTSA has initiated a “recall notification grant program” funding research to make recall notices more effective (U.S. Office of Management and Budget 2019). Other regulators might consider following the NHTSA's initiative. Lastly, the laws must be more specific and flexible regarding the various communication media the firm or regulator must use to contact the buyers of the affected products. Depending on how much contact information of a buyer is available, these media may include personalized letters, emails, and text/voice messages.
Persuade product owners to avail of the recalled product's free remedy. Fundament #5 reminds lawmakers that negligence law—which is used when a consumer brings legal liability against a firm—varies by U.S. state. Consequently, firms headquartered in the 37 states that have implemented the contributory negligence or modified comparative negligence law are likely to recall their unsafe products more proactively than firms headquartered in the other 13 states. Interestingly, negligence law reminds us that the firm's responsibility does not end at recall initiation. If a product owner does not avail of the free remedy offered by the manufacturer/retailer, they are at risk for harm. Acknowledging this risk, U.K. law authorizes officers of its Vehicle and Operator Services Agency and police to inspect recalled vehicles and question drivers who have not taken the free remedy (U.K. Legislation 2002).
Although we understand the U.K. government's perspective, we believe such stern measures may invoke reactance from the very constituents these laws are meant to protect. We recommend that governments devise alternative strategies to persuade product owners so they become a part of the solution, as opposed to being mere receivers of problem notifications (i.e., recall notices). For example, a retailer asks shoppers to sign up for loyalty programs so they can accumulate loyalty points and claim rewards. An alternative positioning is that loyalty program allows retailers to trace a stockkeeping unit (SKU) to the shopper. If the SKU were later found to be a part of the recall, the retailer can issue an autogenerated message or phone call to the shopper and inform them of the recall. Such repositioning of the loyalty program could make a retailer more attractive to product suppliers/manufacturers and demonstrate responsibility to shoppers, consistent with our fundament #5. Similarly, a manufacturer often asks a product buyer to register the product with the manufacturer. This ask could emphasize that the registration will allow the manufacturer to contact the buyer if the purchase product is involved in a recall.
Improve cross-country coordination of recalls. Per fundament #6, although the laws do not grant all regulators the power to mandate recalls, lawmakers can encourage/reward regulators to coordinate recalls with their peer regulators in other countries. Interestingly, some regulators are already undertaking such coordination. However, we recommend that this coordination become the norm and not an exception. For example, U.S. federal law requires an automotive manufacturer to report to the NHTSA a recall it issued in a foreign country if the recall relates to a similar automobile or part sold in the United States (U.S. Federal Government 2023). Interestingly, no such law applies to product categories supervised by the FDA, CPSC, 23 or FSIS.
In similar spirit, the European Commission's Rapid Exchange of Information System coordinates European Union regulators’ recalls of consumer goods. The system reports the notifying country and the country of origin for each recall, thus alerting member countries of the source of the problem and the source of information about the problem. Lastly, the Organisation for Economic Co-operation and Development's (OECD's) GlobalRecalls portal “contains consumer product recalls from 47 jurisdictions globally and integrates recalls from the European Union and ASEAN regional recalls portals, [and thus] enhances information sharing across jurisdictions and supports regulators in taking corrective actions.” 24
We understand the difficulty of coordinating actions across jurisdictions. However, we hope federal governments—or an international organization such as the United Nations—put in place a cross-border agency that coordinates not only reporting of recalls but also tracking the recalled products’ removal from use. Such coordination will extend the independent efforts of the European Commission, the OECD, the CPSC, and the NHTSA, potentially lessening redundant efforts by regulators of each impacted country and boosting public welfare.
Recommendations for Regulators
Define recall as applicable to the focal product category/categories: Fundament #1 suggests that, when available, the government definition should provide the foundation on which a regulator—which is governed by safety law(s) specific to the regulated product categories—should build a definition that applies specifically to the product category/categories it monitors. Consistent with this recommendation, the Canadian Food Inspection Agency's definition of recall seems to be a category-specific application of the Canadian government's definition. However, the same cannot be said of other regulators. The FDA's definition of recall, for example, states that a business initiates a recall to avert the FDA's legal action for violating product safety standards. We note that unlike the U.S. government's definition, which focuses on public safety, the FDA's definition casts a legal view and is thus directed at businesses. Further, the FDA defines recall in terms of the outcomes, and the definition is thus tautological (Suddaby 2010; Yadav 2010). We recommend that regulators apply their federal government's general definition to the specific laws and product categories they supervise. They should also create a definition that is specific to businesses they supervise.
Monitor firms’ use of euphemistic terms that might impede customers’ attention to recall: Regulators aim to lower consumer apathy toward recall notices. Our research suggests that a pervasive cause for this apathy is that firms and regulators use euphemisms to refer to a recall. For example, consumers are less likely to read an “advisory” than a “recall notice” and may thus continue to use the harmful product (CPSC 2003). Fundament #1 and the decision tree suggest that the solution lies in using the correct term (i.e., recall) and not any of its euphemisms (Doering 2012). This monitoring can be performed not only by regulators but also by industry associations. For example, the insurance industry uses a diverse set of terms for recall insurance products. Underwriters inform us that this lack of consistency makes their job difficult and causes unnecessary legal disputes if a buyer files a claim.
Make accessible information about safety incidents related to a product: Regulators receive and collect much data, but these data are mostly inaccessible to users. For example, the NHTSA provides data files of vehicles ratings, recalls, investigations, complaints (https://www.nhtsa.gov/nhtsa-datasets-and-apis), and early warning incidents (https://www.nhtsa.gov/vehicle-manufacturers/early-warning-reporting). However, these files are not integrated to present a “complete” picture of the safety of the vehicles of a brand or a manufacturer; the result of this is a lower level of consumers’ safety-related awareness. Such lack of knowledge means that the onus of when to notify product owners lies with the firm (refer to fundaments #5 and #6). We recommend that the NHTSA integrate these disparate data and present a summary of the incidents and harm attributes of the vehicles of a brand or a manufacturer. For example, the NHTSA could create an app in which a vehicle owner can provide their VIN and opt in to be notified whenever a recall involves the VIN or the specific year-make-model is involved in an injury or property damage. Such an app enables the owner to take charge of their safety and makes them involved in the process, as opposed to merely receiving mailed recall notices that may be overlooked as spam mail.
Allow a recalling firm to expedite remediation: Consistent with our fundament #5, some regulators, such as the CPSC, allow the recalling firm to expedite the remediation. This process is understandably costly for the regulator and the recalling firm. However, the upside is that it can inform affected customers in a timely manner and potentially save lives. We recommend that other regulators—particularly those that supervise safety of food and health care products, such as the FDA—consider “piloting” an expedited remediation and compare the benefits and costs before deciding whether to introduce it as a choice variable for the recalling firm. Allowing the recalling firm to expedite remediation—often using technology to trace sold products and social media to reach product owners—may create a collaborative as opposed to adversarial relation between the regulators and those regulated.
Measure and publicly report recall-specific rate of remediation: The U.S. FDA and NHTSA are the only two regulators across all countries that collect data on the rate that a recalling firm remedies the recalled products—a proxy for the firm's demonstration of responsibility (fundament #5). Specifically, the FDA assigns a recall the status of complete when it has reason to believe that the recalling firm has made all efforts to remedy the recalled product units. This translates into a recall completion date. One can therefore compute the number of days between recall initiation and completion. This number measures the rate of remediation. Similarly, the NHTSA requires a vehicle manufacturer to report the number of recalled vehicles repaired in each calendar quarter since the initiation of a recall.
This number enables a stakeholder to measure the quarterly rate of repair. Our examination of other regulator-provided data suggests that no other regulator collects—and, by extension, measures—the rate of remediation. This inattention to measurement limits the regulator's management of recalls and stakeholders’ ability to hold the recalling firm and the regulator accountable for preserving public safety (Beresford 2021; Consumer Reports 2010). We hope other regulators follow the NHTSA and the FDA in measuring this accountability, thus boosting enforcement of the recall-related laws. Following the Fixing America's Surface Transportation (FAST) Act (U.S. Government Publishing Office 2015), we also hope the regulators not only collect these data but also regularly publish reports on how the manufacturers and NHTSA have performed on remediation rate (NHTSA 2017, 2018, 2021b).
Conclusion
Extant legislation and regulations related to product safety and recall are specific to product categories and legal jurisdictions. This specificity leads to inconsistent legislation and regulation across product categories and legal jurisdictions, limiting the development of a shared language. This limitation has led to pervasive misunderstanding among practitioners about what a recall is—and, more importantly, what it is not. We address this misunderstanding by identifying seven fundaments of recall. For each fundament, we draw implications for marketing and public policy. Table WB1 in Web Appendix B summarizes the implications in tabular form. We distill the essence of the seven fundaments into a definition of recall and a decision tree (Figure 3) that stakeholders can use to determine what term to use for what phenomenon. These fundaments help us make five concrete recommendations each for lawmakers and regulators.
Our review has limitations that suggest at least two directions for further research. First, the present effort has focused on untangling conceptual and terminology-related limitations in legislation and regulation relating to recall. This focus allows us to contribute to research on recalls and closely related but distinct phenomena, such as harm crises. Future research can use our definition to meta-analyze the effects of recall and related terms on outcomes such as stock return, sales, consumer attitude, and organizational learning. Second, we have not considered how other related negative events might overlap conceptually and substantively with product recall. Future research might extend our review to consider these other negative events—such as data breach, service failure, ethical misconduct, and brand scandals—and firms’ corrective actions in response to these failures (e.g., service recovery). Such a review can help inform marketing and public policy on the broader domain of negative events.
Supplemental Material
sj-pdf-1-ppo-10.1177_07439156241242419 - Supplemental material for What Is (and Isn’t) a Product Recall?
Supplemental material, sj-pdf-1-ppo-10.1177_07439156241242419 for What Is (and Isn’t) a Product Recall? by Vivek Astvansh, Kersi D. Antia and Gerard J. Tellis in Journal of Public Policy & Marketing
Footnotes
Authors Contributions
This manuscript is based on the first author’s doctoral dissertation.
Joint Editors in Chief
Jeremy Kees and Beth Vallen
Associate Editor
Frank Germann
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
