Abstract
Treatment of Palabora vermiculite with ammonium chloride or ammonium nitrate solutions caused the collapse of its structure, resulting in a reduction of the exfoliation onset temperature by more than 200°C. It is speculated that ammonium ions enter the hydrobiotite galleries as guest ions partially replacing the hydration water associated with the magnesium ions. The effectiveness of various modified vermiculite flakes as flame-retardant fillers was evaluated in laminates of high-density polyethylene and plasticized polyvinyl chloride/vermiculite-modified composites. The laminates comprised a high-density polyethylene sheet coated with polyvinyl chloride plasticized with 100 phr of a phosphate ester and filled with 40 phr vermiculite. Cone calorimetry results, measured at a radiant flux of 35 kW m−2, revealed that copper-ion modified vermiculite was the most effective flame-retardant additive. Laminates coated with polyvinyl chloride containing copper-ion-modified vermiculite exhibited a significantly reduced peak heat release rate of 64kW/m2, compared to 677kW/m2 for uncoated high-density polyethylene, demonstrating the potential of polyvinyl chloride–based flame-retardant coatings incorporating copper-ion-modified vermiculite for polyethylene.
Keywords
Introduction
Palabora ‘vermiculite’ is a mixed-layer vermiculite-biotite mineral1–3 containing less than 50% true vermiculite. 4 It exhibits considerable variability in composition, which influences its cation exchange capacity (CEC). 2 The individual flakes of interstratified vermiculite consists of elementary layers comprising vermiculite and biotite.5,6 In these layers, the hydrated magnesium interlayer ions in vermiculite layers are easily exchanged, unlike the potassium cations in the biotite layers. 6
Interstratified mica materials thermally expand at relatively low temperatures,7,8 in contrast to true vermiculite. 9 Rapid heating of vermiculite flakes to high temperatures typically leads to an eight-fold expansion in volume.10,11 This exfoliation process is driven by the explosive release of interlayer water. 8 The onset exfoliation temperature of vermiculite is typically more than 600°C. However, this temperature can be adjusted through targeted ion exchange4,11 or by intercalation with urea. 12
Several studies have investigated the potential of both neat and modified vermiculite, to enhance fire retardancy in bulk polymers,13–16 foams17,18 and coatings.19–22 For vermiculite to function as an effective flame retardant for polymers, its exfoliation should occur at temperatures below the polymer volatilization range, which typically lies between 200–350°C. 16 This is important because volatilization marks the release of flammable gases that sustain combustion; forming a protective barrier before this stage can significantly reduce fire risk. However, the exfoliation of neat vermiculite generally occurs at temperatures well above the melting or decomposition points of common polymers. 11 It is proposed that appropriate ion exchange treatments can lower the onset temperature of exfoliation in vermiculite, enabling it to meet the required temperature range for it to be an effective flame retardant for polymers.11,12 In addition, these treatments are expected to influence the expansion behaviour of vermiculite, which will, in turn, enhance the structure and effectiveness of the resulting heat-insulating layer.
The use of expandable vermiculite as a stand-alone flame retardant additive has been explored in polyurethane and plasticized poly(vinyl chloride) (PVC) at loadings up to 20 wt.%. 16 Cone calorimeter tests showed that the vermiculite expanded to form a heat-insulating layer on the surface layer, aiding thermal stabilization of the condensed phase but with limited impact on vapour phase behaviour. In both systems, the peak heat release rate (pHRR) was reduced. In the PVC matrix, there was also a significant reduction in total heat release (tHR). This was attributed to halogen species, released from PVC, acting as ‘flame poisons’. Vermiculite has also been utilized as a thermal stabilizer for PVC resins. 23
This study focused on developing fire-resistant coatings for polyethylene pipes used in underground mining applications. Based on previous results, 16 a PVC-based coating containing 17 wt.% of various vermiculite modifications, achieved through ion exchange reactions, was explored.
Materials and methods
Materials
Merck Pty Ltd. (Modderfontein, South Africa) supplied all the salts that were used in the ion-exchange reactions. Palabora vermiculite was provided by Mandoval Vermiculite (Pty) Ltd. (Alberton, South Africa). All experiments used a medium particle size fraction, the portion that passed the sieve with 4 mm apertures but which was retained on the sieve with 2 mm apertures. Poly(vinyl chloride) emulsion grade PG680 was sourced from TPC Paste Resin Co. Ltd. (Rayong, Thailand). This free-flowing powder had a K-value of 69. A proprietary blend of synthetic isopropylated triaryl phosphate ester plasticizers was supplied by Chemtura. High-density polyethylene sheets (2 mm thick), were purchased from Maizey Plastics, Pretoria-East, South Africa. Chemically pure (CP) copper nitrate, nickel(II) nitrate, ammonium chloride, ammonium nitrate and potassium nitrate were supplied by SigmaAldrich (now Merck).
Sample preparation
Ion exchange modification
Molar solutions (1 M) of the respective salts were made using distilled water. A total of 2 L of each salt solution was added to 5 L glass containers containing 1.5 kg of unmodified vermiculite. The containers were shaken vigorously three times per day. After 3 to 7 days, the liquid in the containers was drained and replaced with fresh 1 M salt solutions. This process was repeated five times. Following this, the solution in the containers was drained and replaced with distilled water. The glass containers were shaken and left to stand for 1 day before the water was replaced with a fresh quantity. This washing process was repeated daily for 1 week to ensure the removal of all non-intercalated minerals from the surface of the vermiculite flakes. Finally, the modified vermiculite samples were removed from the containers and left to air-dry in aluminium pans.
Preparation of the vermiculite-PVC-polyethylene laminates
Moulds measuring 100 mm × 100 mm and about 6 mm deep were shaped from thick aluminium foil, using 3.08 ± 0.08 mm thick polyethylene sheets as template. The 17 wt.% PVC-vermiculite compounds were prepared using the plastisol method. PVC powder (100 g), plasticizer (100 g) and vermiculite (40 g) were mixed together in a mortar and pestle until homogeneous. Note that no additional PVC heat stabilizer was incorporated into the formulation in view of a previous finding that vermiculite flakes impart thermal stability to PVC. 23 Portions of the paste mixture (30 g) were immediately poured onto the polyethylene sheets (average mass 26.9 ± 0.216 g) placed at the bottom of the prepared moulds and heated for 20 minutes in a convection oven set at 130°C. This resulted in a solid coating but it is not certain that full gelation of the PVC-plasticiser combination was achieved. The average mass of the final laminates was 66.5 ± 1.6 g. They had an average thickness of 5.7 ± 0.7 mm. Separately PVC-only samples sheets were cast. They contained 60 phr of the plasticizer and their mean weight was 49.0 ± 2 g. The total heat release, measured for the neat polyethylene and PVC samples, were scaled to reflect values expected for a sample mass of 66.5 g, which was the average for the PVC-HDPE laminates.
Characterisation
X-ray diffraction (XRD)
X-ray diffraction analysis was performed using a PANalytical X’Pert Pro powder diffractometer. The results were analysed with X’Pert Highscore plus software. The samples were analysed in the θ - θ configuration, using an X’Celerator detector with variable divergence slits and fixed receiving slits, utilizing Fe filtered Co-K radiation (λ = 1.789 Å). Mineralogical identification was based on matching the measured diffraction pattern with the best fitting pattern from the Inorganic Crystal Structure Database (ICSD) database.
X-ray fluorescence (XRF)
The vermiculite samples were roasted in alumina refractory crucibles at 1000°C to determine the loss of ignition (LOI). Small samples (1.0 g) were mixed with 6.0 g lithium tetraborate flux and fused at 1050°C to make a stable fused glass bead. XRF analysis was conducted using a Thermo Fisher ARL Perform’X Sequential XRF analytical instrument, with data processed using Uniquant software. The software analysed all elements in the periodic table from Na and U, reporting only those elements present above the detection limits. Standard sample materials were prepared and analysed in the same manner.
Fourier transform spectroscopy-attenuated total reflection (FTIR-ATR)
FTIR spectra of the vermiculite flakes were recorded on a Perkin-Elmer Spectrum 100 instrument in ATR mode. The spectra represent averages of 32 scans, scanned at a resolution of 2 cm−1. FTIR-ATR spectra were also obtained for a selection of CP salts.
Thermomechanical analysis
The thermomechanical analyses were conducted using a TA Instruments Q400 thermomechanical analysis (TMA) instrument, with data analysed using the TA Instruments Universal Analysis 2000 software. The thickness of the flakes tested ranged from 91 to 231 μm. In each experiment, a single flake was sandwiched between the flat surface probe and the bottom surface of an alumina pan. Flake expansion behaviour was measured using dynamic scans in air using an applied force of 0.02 N. The temperature was scanned, from 50°C to 600°C, at a rate of 10°C min−1 with a nitrogen purge flowing at 50 mL min−1. The expansion behaviour, of at least four different flakes from each vermiculite modification, was determined.
Cone calorimeter testing
Cone calorimeter tests were performed following the ISO 5660 standards24–26 using a Dual Cone Calorimeter (Fire Testing Technology (UK) Ltd.). Three specimens of each composition were tested. The sheet dimensions were 100 mm × 100 mm × 6 mm. They were placed on aluminium foil, covered with a grid and the PVC-vermiculite side of the samples was exposed horizontally to an external heat flux of 35 kW m−2. This heat flux was chosen on the basis of the ISO 5660 standards.24–26
Results and Discussion
X-ray diffraction
The XRD pattern shown in Figure 1 for the neat vermiculite reveals the typical mineralogical composition of Palabora vermiculite. This includes vermiculite (red line) at approximately 14.33Å (7.157°2θ), a mixed layer (interstratified) vermiculite/mica (blue and green lines), identified as hydrobiotite, which is expressed as a duplet at 12.53Å (8.187° 2θ) and 11.92Å (8.591° 2θ), mica (biotite/phlogopite, purple) at 10.10Å (10.155° 2θ). While other phases may be present as minor impurities, apatite (Ca10(PO4)6(OH)2) is likely the most significant.
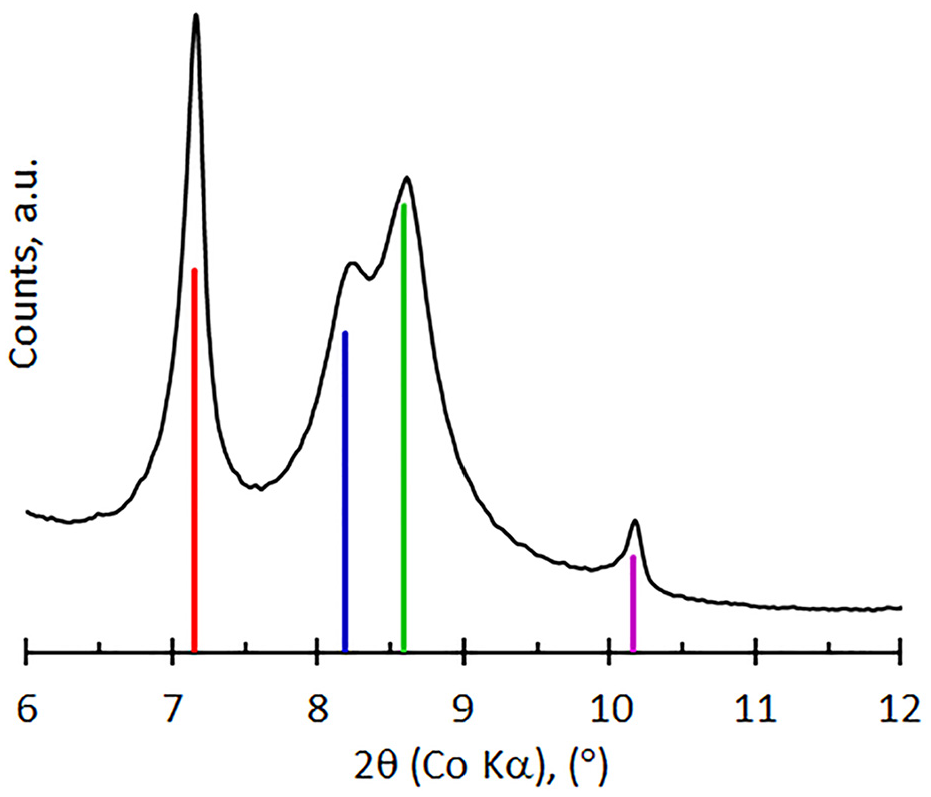
XRD diffractogram for the neat vermiculite showing vermiculite (red line), the interstratified vermiculite/mica (blue and green lines), and mica (purple line) reflections.
The diffractograms for the samples exposed to copper- and nickel nitrate solutions appear very similar to that observed for the neat vermiculite. However, as shown in Figure 2, the patterns found for the samples exposed to NH4Cl and NH4NO3 indicate significant changes. The vermiculite structure has completely collapsed, even the second and third order reflections at higher angles have disappeared. Instead, a strong broad reflection is visible at 10.98 Å (9.34° 2θ). However, the mica reflection remains visible at the higher angle shoulder of this broad peak, with a small, sharp reflection attributed to biotite also discernible. It appears that the interstratified hydrobiotite structure has shifted as a result of ion-exchange, the main reflection shifted to higher angles, from approximately 8° to 9.35° 2θ. However, the remaining features of the original unit cell are preserved, with reflections at higher angles still visible at approximately, 21, 30 and 52° 2θ.
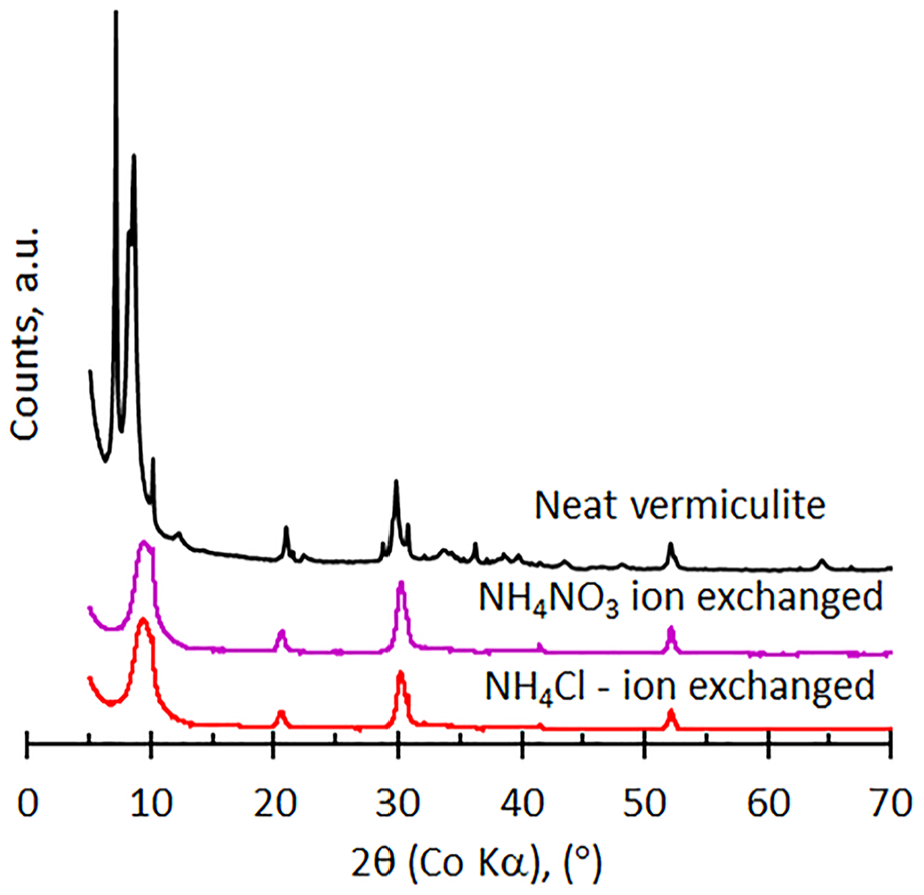
XRD diffractograms for the modified vermiculites compared to the neat vermiculite.
X-ray fluorescence spectroscopy
Table 1 compares the single value XRF results for the neat vermiculite and the samples modified with salt solutions. The LOI values listed for all the samples are relatively consistent at 8.65 ± 0.22 %. Similarly, the MgO/(SiO2 + Al2O3) ratio remained stable at 0.497 ± 0.013 across samples. This stability was unexpected, as it was anticipated that the ion exchange modification would lead to the partial replacement of magnesium ions in the hydrobiotite interlayers. However, the XRF results confirmed that samples treated with copper and nickel salts contained cations from these salts in appreciable amounts.
XRF results obtained for the neat vermiculite and various samples exposed to salt solutions
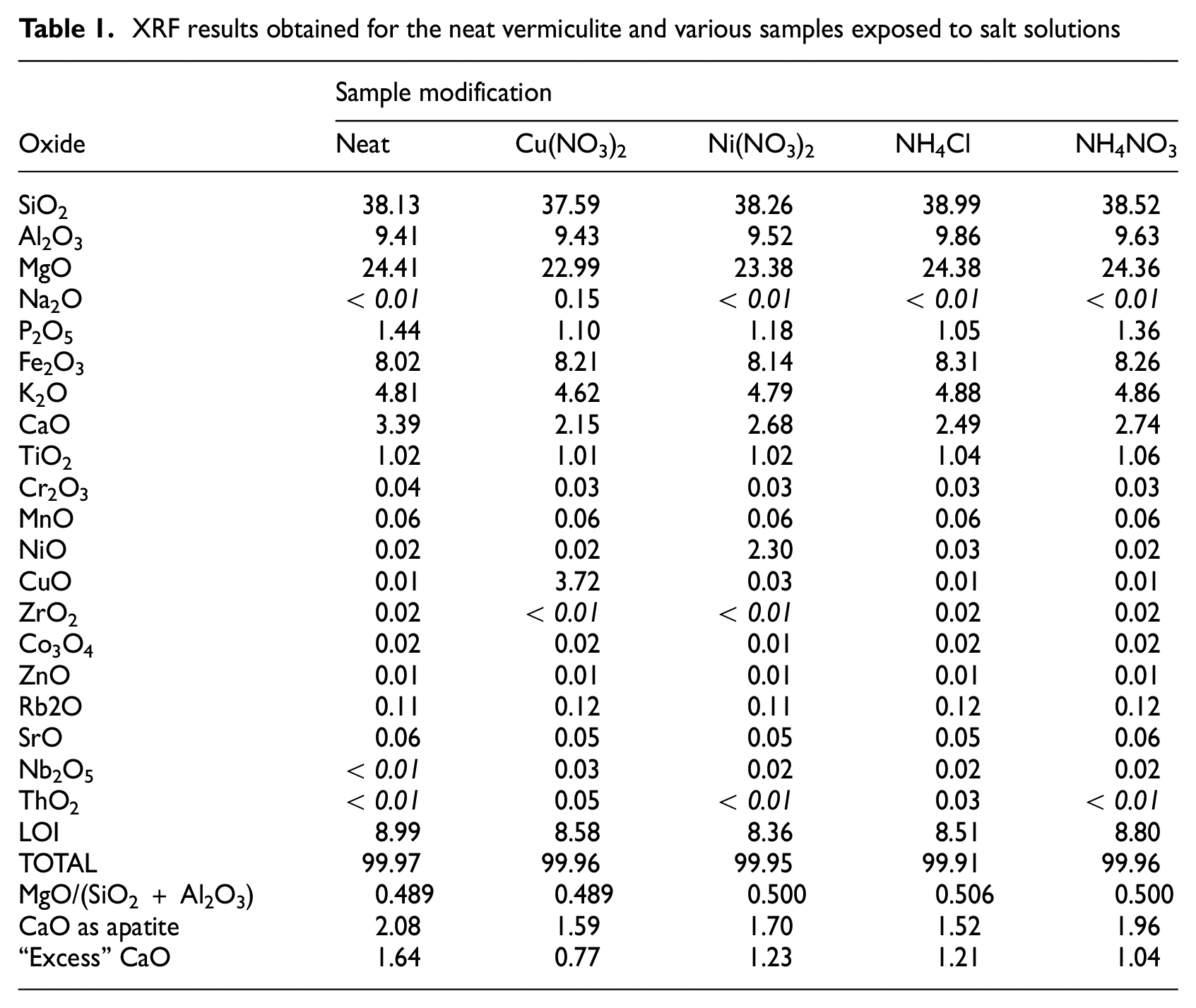
The P2O5 and CaO contents in the neat vermiculite were measured at 1.44 % and 3.39 % respectively. Following treatment with the salt solutions, these values decreased to averages of 1.17 ± 0.12 % for P2O5 and 2.52 ± 0.23 % for CaO. Some of the calcium can be accounted for if it is assumed that the phosphorus present exists in the form of apatite. However, the measured calcium levels exceeded this amount in all the samples. Notably, there was a definite reduction of this ‘excess’ calcium level detected in the modified vermiculite samples (see Table 1). The implication is that at least part of the ion exchange must have resulted in replacement of some of the calcium originally present in the material. Unfortunately, it is not clear what the origin of these calcium ions were although one might be tempted to assume that these calcium ions might have been present in the hydrobiotite galleries.
Fourier transform spectroscopy-attenuated total reflection (FTIR-ATR)
Figure 3 compares the FTIR-ATR spectra for the neat vermiculite and an ammonium nitrate modified sample to the spectra for ammonium nitrate, ammonium chloride and potassium nitrate. The spectra for the copper- and nickel nitrate modified vermiculites did not differ from that recorded for the neat vermiculite. However, the ammonium modified vermiculite features two additional absorption bands, labelled ‘a’ and ‘b’ in Figure 3, and positioned at 1422 cm−1 and 1349 cm−1. Similar positions for bands attributable to ammonium ion and nitrate ion absorption bands are indicated by the spectra for the salts shown in Figure 3. This provided confirmation that ammonium ions are present in the hydrobiotite galleries; perhaps even some nitrate ions too but this is a speculative assertion.
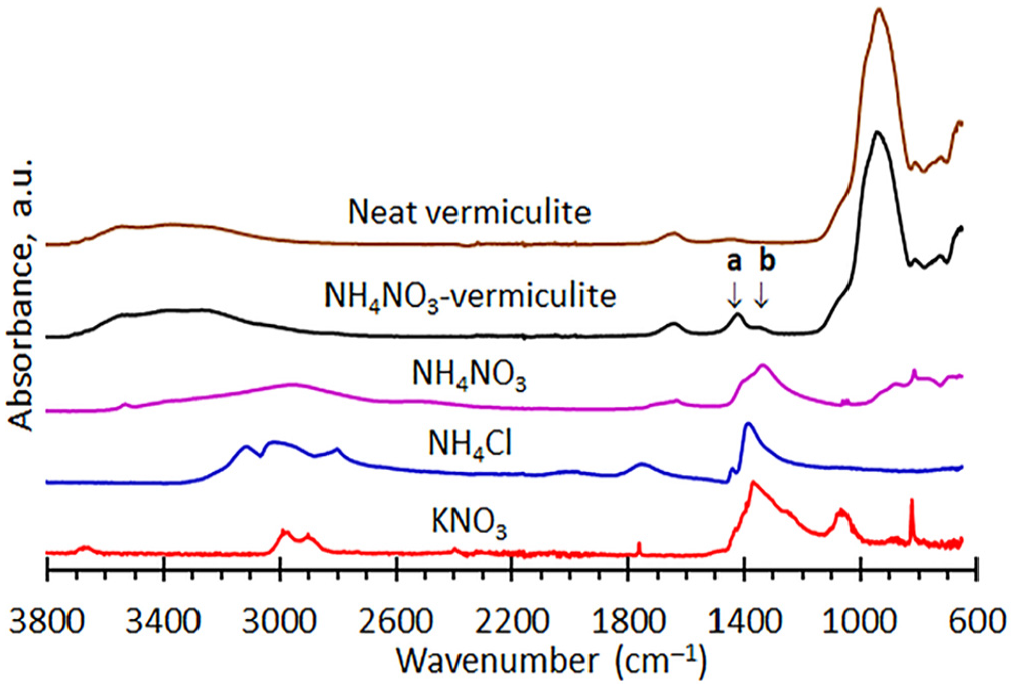
FTIR-ATR spectra for neat vermiculite and selected modifications.
TMA
Figure 4 shows the TMA traces recorded for individual vermiculite flakes ion exchanged using ammonium nitrate solutions. Clearly, there is considerable variation in the observed exfoliation onset temperatures and also in the ultimate expansion ratios. This is a consequence of inhomogeneous nature of the starting material which is, after all, a natural product formed by irregular weathering. The TMA traces obtained with the other vermiculite samples showed similar property variation. Figure 5 summarizes the effect of ion exchange on the exfoliation onset temperatures and the flake expansion ratios. The variability in expansion ratios is particularly large. The expansion ratios showed no correlation with flake thickness, as indicted by a correlation coefficient of −0.066 for the full data set. This suggests that the variability in the mineralogy of the individual flakes, rather than variations in the flake thicknesses, determined the expansion ratio.
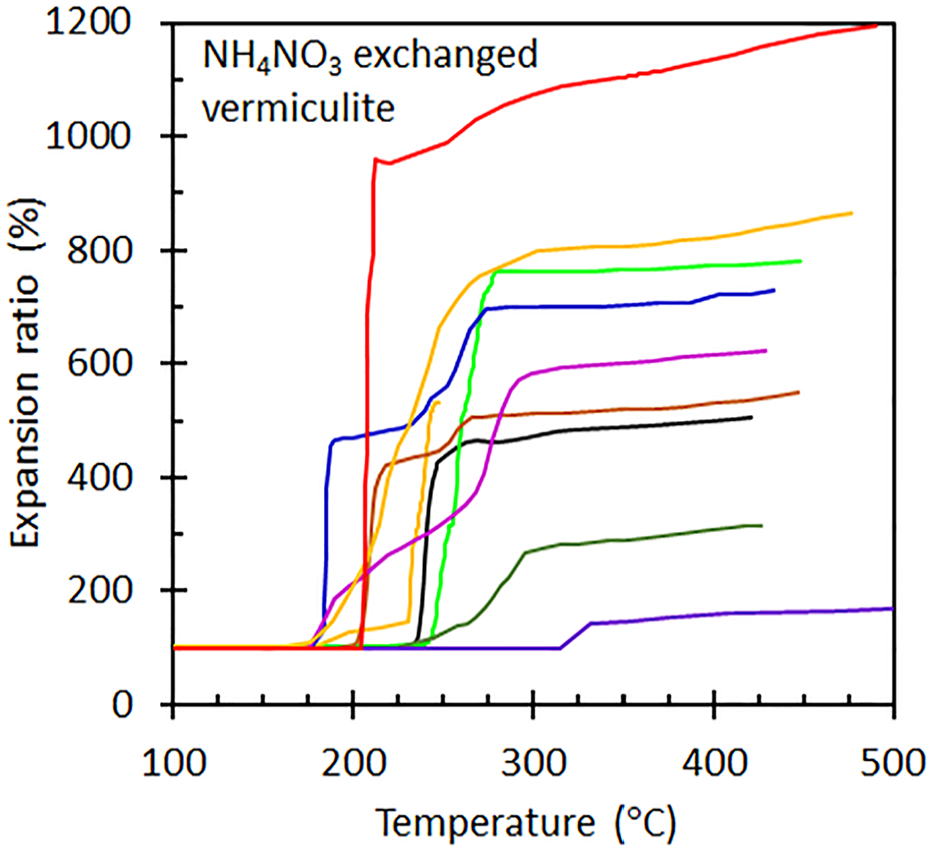
TMA traces obtained for ammonium nitrate treated vermiculite flakes.
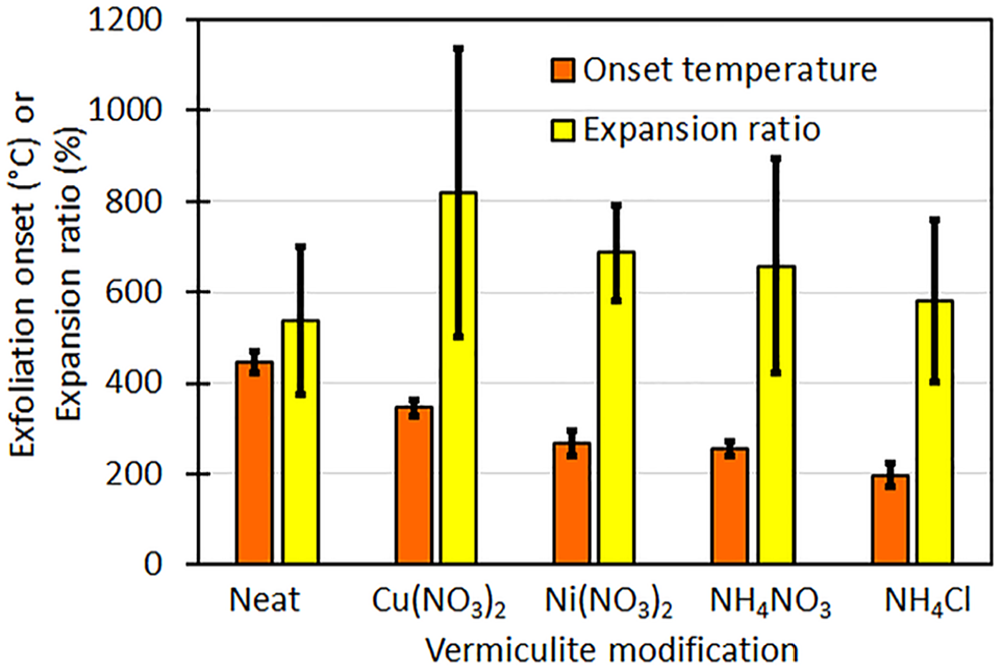
Effect of ion exchange on TMA onset temperatures and expansion ratios.
The various treatments significantly reduced the exfoliation onset temperature of the vermiculite. The neat vermiculite had an average exfoliation onset temperature of 447°C with a standard deviation of 23°C (447 ± 23°C). For vermiculite treated with copper nitrate, nickel nitrate, ammonium nitrate and ammonium nitrate, the exfoliation onset temperatures were reduced to 345 ± 17°C, 267 ± 27°C, 255 ± 18°C and 197 ± 26°C, respectively. These values are presented graphically in in Figure 5, together with the expansion ratios. Notably, the exfoliation onset temperature for the ammonium chloride-treated sample was 59°C lower than the mean value observed for the ammonium nitrate-treated samples. This difference was calculated for 10 independent TMA measurements performed on each of these two vermiculite variants. A two-tail t-test confirmed that the difference in the means in the measured exfoliation onset temperatures is statistically significant (p = 0.00004).
For vermiculite to function effectively as a flame retardant in polymers, exfoliation should occur below the polymer volatilization temperature. 16 For such applications, exfoliation within the range of 200°C to 350°C is considered desirable. 11 Muiambo et al. 11 observed that co-intercalation with urea was necessary to reduce the exfoliation onset temperature to below 210°C for vermiculite exchanged with magnesium, aluminium, or iron(III) ions. In this study, the exfoliation onset temperatures for Palabora vermiculite treated with copper nitrate, nickel nitrate, ammonium nitrate, and aluminium nitrate were found to be 345±17°C, 267±27°C, 255±18°C, and 197±26°C, respectively. These values are significantly lower than those reported for other treatments in previous studies and fall within the target temperature range for flame retardancy.4,11,12 These findings indicate that the vermiculite treatments employed in the current study were effective in achieving the desired thermal behaviour for flame-retardant applications in polymers.
Flame retardancy
Although several studies have focused on developing flame-retardant coatings containing vermiculite, those demonstrating effectiveness typically involved modifications or combinations with other flame retardants, rather than relying on neat vermiculite alone. For instance, Nasirzadeh et al. 27 evaluated the incorporation of vermiculite into a water-based intumescent flame-retardant coating for polypropylene, enabling the material to achieve a V0 rating in UL 94 vertical burn tests. Sethurajaperumal et al. 22 developed a vermiculite-based nanocomposite paint using exfoliated vermiculite nanosheets in an epoxy matrix to coat wooden rods, which effectively reduced flame spread, combustion velocity, and smoke release. Barczewski et al. 28 utilized expanded vermiculite as a synergist with the intumescent flame-retardant ammonium polyphosphate (APP), demonstrating its effectiveness in enhancing flame retardancy.
In this study, the flame-retardant effectiveness of PVC-based coatings containing modified vermiculite were evaluated using cone calorimetry. The heat release rate (HRR) is widely regarded as the most critical parameter for evaluating the flammability and associated fire hazards of materials.29,30 In cone calorimetry, HRR is measured using oxygen consumption calorimetry, enabling the characterization of material combustion behaviour through HRR–time curves. 30
Figure 6 shows representative HRR curves obtained from the cone calorimeter tests conducted on the laminates. The HRR curves for the polyethylene and the PVC laminated based on the Cu(NO3)2-modified vermiculite (see Figure 6(a)) exhibited the shape characteristic of thermally thin samples. 30 Thermally thin samples feature a sharp peak in their HRR curves as the whole sample is pyrolyzed at once. HRR curves characteristic of thermally thick, char-producing samples show a sudden rise to a plateau value. 30 The HRR curves for the other PVC containing samples approached this shape. They showed a rapid rise after ignition followed by a slower downwards taper in the heat release rate.
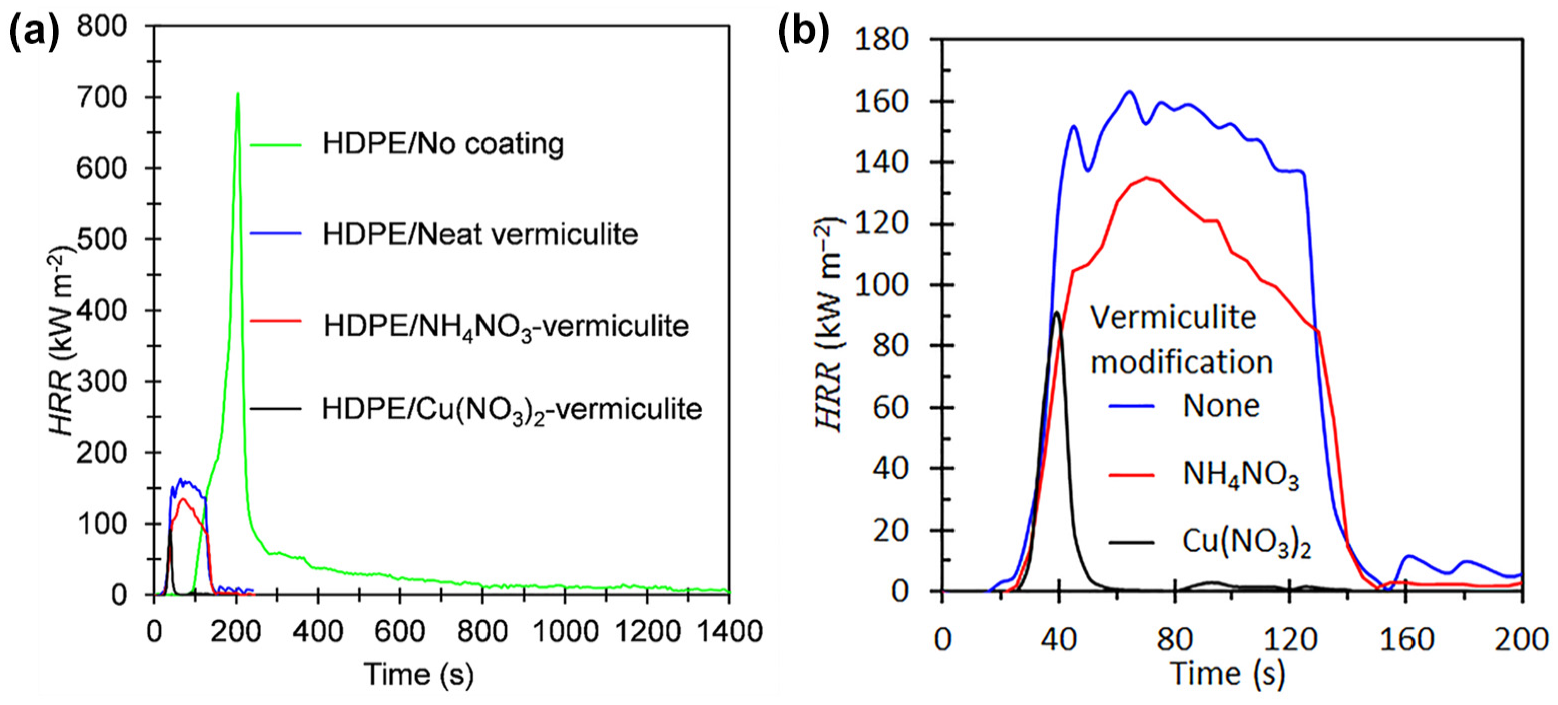
Cone calorimeter heat release rate (HRR) curves for laminates based on the various vermiculite modifications. (a) includes the HRR curve for the neat HDPE and (b) only shows the HRR curves with the modified vermiculite. The sample sheets were backed by aluminium foil and their dimensions were 100 mm × 100 mm × 3.2 ± 0.1 mm. They were exposed horizontally to an external heat flux of 35 kW m−2.
Fire performance indices derived from cone calorimeter tests are summarized in Table 2 and illustrated in Figure 7. The time to ignition (tig), for the neat polyethylene was 96 ± 4 s, whereas for the laminates, the tig was significantly reduced, as observed in Figure 6, and reported in Table 2. For the PVC sheet, tig was 35 ± 3 s, with similar observed for the polyethylene-PVC laminates. This reduction in ignition time for the PVC containing samples is attributed to the presence of the volatile plasticizer. This rationalization is supported by the earlier onset of mass loss observed for the laminates when compared to the neat HDPE.
Cone calorimeter data summary for time to ignition (tign), time to flame out (tflame_out), peak heat release rate (pHRR), time to peak heat release rate (t pHRR ), maximum average rate of heat emission (MAHRE), fire growth rate(FIGRA), total smoke released during whole test (TSR), Petrella fire growth index (pHRR/tign) and residue remaining after test completion

Mass remaining at time t = 200 s.
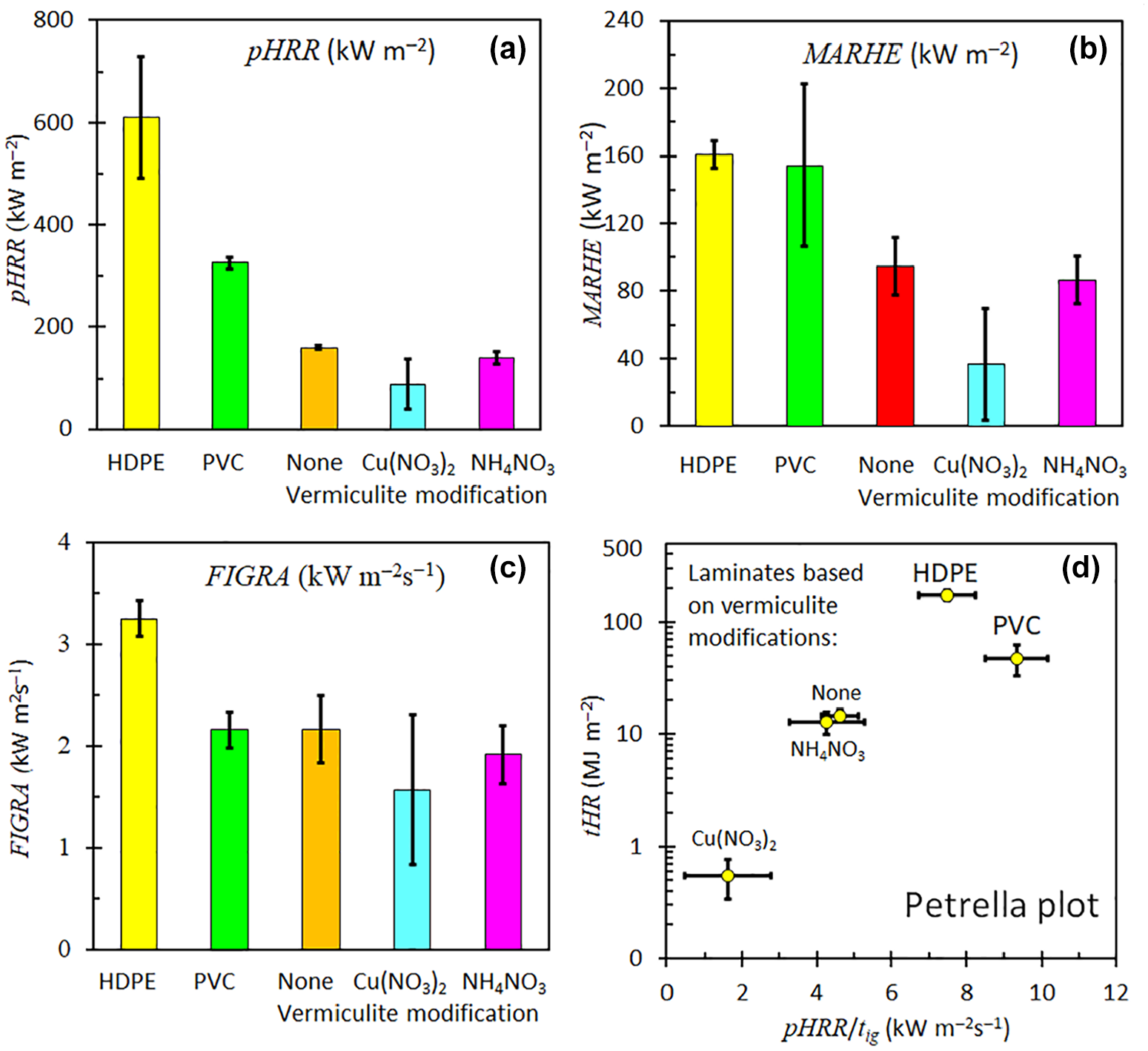
Pictorial summary of fire indices obtained or laminates exposed horizontally to an external heat flux of 35 kW m−2. (a) pHRR (kW m−2), (b) MARHE (kW m−2), (c) FIGRA (kW m−2s−1) and (d) Petrella plot. The sample sheets were backed by aluminium foil and their dimensions were 100 mm × 100 mm × 3.2 ± 0.1 mm.
The time to flame out showed considerable variability. For the neat polyethylene, it was 1350 ± 95 s, while PVC-containing samples had shorter flameout times, with the Cu(NO3)2-modified vermiculite laminate recording the shortest time at 49 ± 5 s.
In cone calorimetry, the peak heat release rate (pHRR) represents the maximum rate at which heat is released per unit area during combustion, serving as a critical indicator of a material’s flammability and potential fire hazard. 31 Table 2 presents peak heat release rates (pHRR) obtained from the cone calorimeter tests. The pHRR for the neat polyethylene was 677 ± 35 kW m−2 whereas for the PVC compound exhibited a significantly lower pHRR of 326 ± 11 kW m−2. All the coated samples demonstrated even lower pHRR values, as shown in Table 2. The lowest recorded pHRR (64 ± 36 kW m−2) was observed for the sample with a PVC coating containing Cu(NO3)2-modified vermiculite. This corresponds to a 91% reduction compared to the pHRR of neat, uncoated polyethylene, indicating a substantial improvement in flame retardancy. In contrast, Miedzińska et al. 32 reported a maximum reduction of only 25% in the pHRR of polyurethane foams incorporating vermiculite modified with casein, chitosan, or potato protein. Similarly, Muiambo et al. 16 achieved a 28% reduction in the pHRR of polyurethane using vermiculite co-intercalated with magnesium ions at a 15 wt.% loading of urea-modified vermiculite. This highlights the superior effectiveness of the copper-modified vermiculite coating in enhancing flame retardancy compared to other treatments reported in the literature.
The cone calorimeter-derived parameters that are pertinent to fire hazard evaluations are the fire load and flame spread. 30 The fire load is the total amount of heat that can be generated by a flammable material if it is ignited. In the cone calorimeter this index is quantified by the total heat released (tHR). The total heat release values projected for the polyethylene and PVC for equivalent surface specific weights are 172 ± 18 MJ m−2 and 47 ± 15 MJ m−2. As indicated in Table 2, much lower values were measured for the laminates. In particular, the tHR for the laminate based on the Cu(NO3)2-modified vermiculite was just 0.55 ± 0.21 MJ m−2. In contrast, previous studies using modified vermiculite reported only marginal or no reduction in the tHR, further highlighting the effectiveness of the treatment employed in this study.16,32
Flame spread is not directly measured in cone calorimeter tests. The fire growth rate (FIGRA) and the fire growth index (pHRR/tign) proposed by Petrella, 33 can be used as proxy estimators for the flame spread. 30 The FIGRA can usually be determined from the expression:

where t pHRR is the time to the peak heat release rate.
Another very important index used to interpret cone calorimeter data is the maximum average rate of heat emission (MAHRE).30,34 The MAHRE parameter is defined as the peak value of the cumulative heat emission divided by time. 34 It provides a measure of the propensity for fire development under full scale conditions. 34 Table 2 lists the times to peak heat release rate together with the FIGRA, MAHRE and fire growth indices. For a material to be effectively flame retarded, these indices should all assume low values. Table 2 shows that all the laminates featured significantly lower values compared to the polyethylene and PVC sheets. The reductions imparted, by the incorporation of the modified vermiculite into the PVC coating, to the pHRR and the MAHRE index are illustrated in Figure 7(a) and Figure 7(b) respectively. The performance of the Cu(NO3)2-modified vermiculite laminate is particularly impressive with respect to lowering the magnitude of both of these two indices. The FIGRA results are plotted in Figure 7(c) and the values obtained for all the PVC materials are similar and significantly lower than that of the polyethylene.
The improved fire performance, with respect to the various fire indices, is tentatively attributed to both the reflection of the incoming radiation and heat-insulating protective barrier properties of the expanded vermiculite flakes. The thermal expansion of the vermiculite flakes results in the formation of a low-density insulating layer at the surface. It reduces heat transport to lower layers of the substrate and this slows down the rate of thermal degradation of the polymer. This, in turn, reduces the rate at which volatile fuel is released. This means that less material was consumed during fire testing of the flame retarded samples. This is confirmed by the higher residue values listed in Table 2.
The fire growth index (pHRR/t ign ) proposed by Petrella 33 is probably a better estimator of flame spread than the FIGRA. 30 The Petrella plot helps to visualize the effect of a flame retardant on the magnitude of both fire hazard parameters.30,33 It is a plot of the total heat released tHR (as fire load) against pHRR/tig (as a fire growth index). Figure 7(d) shows a Petrella plot for the samples evaluated presently. For a material to be effectively flame retarded both the fire load and the fire growth index should assume low values. Figure 7(d) shows a dramatic decrease when the Cu(NO3)2-modified vermiculite was applied as the filler for the PVC coating on the polyethylene substrate.
The smoke generated by fires reduces visibility and this can significantly affect life safety in underground mining applications. 35 Table 2 shows that the total smoke released (TSR) in the cone calorimeter tests was lowest for polyethylene and highest for the PVC compound. On average, the TSR values measured for the laminates were about twice this value but significantly lower than that found for the PVC compound.
In fire deaths the influence of heat is considered to be of minor importance. Mortality is invariably caused by toxic gases 29 in which carbon monoxide is the main culprit, with carbon dioxide of secondary importance.36,37 Table 2 shows that the combustion of the polyethylene generated the least carbon monoxide and the PVC compound the most. Unsurprisingly, the combustion of the polyethylene resulted in the highest carbon dioxide release. Complete combustion of the polyethylene should yield 3.14 kg CO2 per kg substrate but the measured value was only 1.63 ± 0.47. Only part of this difference can be attributed to the fact that a solid char-like residue remained which contained residual carbon. The implication is that the combustion of the released volatiles must have been incomplete. Superficial consideration of the CO values released by the laminates suggests that the combustion of the coated samples poses a greater health risk than neat polyethylene. However, the risk posed by the release of these two gases can be assessed on the basis of the short-term exposure limits (STEL). These values indicate the acceptable average exposure over a 15 min period. The Occupational Safety and Health Administration (OSHA) set regulatory limits for CO and for CO2 of 200 ppm value of 30 000 ppm respectively. 38 The ratio of these concentrations (CO2: CO = 150) indicates the relative hazard posed by the two gases. If the actual ratio exceeds this value, it means that the CO2 controls the smoke toxicity. As seen in Table 2, the measured values of this ratio are in all cases more than an order of magnitude higher than 150. This means that the laminates pose a lower CO2 smoke toxicity hazard than the polyethylene as far as the impact of the gases CO and CO2 are concerned. However, in reality, the actual smoke toxicity posed by the PVC containing materials might still be higher as they also release HCl but unfortunately this was not measured presently. HCl is classified as an irritant effluent during fires, while CO is an asphyxiant, and COã is a simple asphyxiant.
Conclusion
Attempts were made to modify Palabora vermiculite by ion exchange by exposure to aqueous solutions containing ammonium chloride, ammonium nitrate, copper nitrate and nickel nitrate. Elemental analysis using X-ray fluorescence spectroscopy (XRF) showed the presence of copper and nickel after the corresponding solution exposure. However, the magnesium ions in the interstratified layers were not exchanged but the calcium impurity was reduced. The X-ray diffraction (XRD) patterns for the samples exposed to the nickel- and copper nitrate solutions were essentially identical to that of the starting material. However, XRD showed that vermiculite structure of the samples treated with the ammonium ion containing solutions had collapsed. It is speculated that the ammonium guest ions partially replaced the water of hydration present in the original material. TMA showed that exposure of the vermiculite to the salt solutions caused a reduction in the exfoliation onset temperature. The lowest onset temperature (<200°C) was obtained for the sample treated with ammonium nitrate solution.
The effect of the various vermiculite samples on the fire performance polyethylene sheets, coated with vermiculite-filled plasticised PVC, was evaluated using cone calorimeter testing at 35 kW m−2. For this purpose, emulsion grade PVC was plasticized with 100 phr to which was added 40 phr vermiculite. The polyethylene sample sheets measured 100 × 100 × 3.17 mm and weighed 26.9 g. The PVC coating weighed 39.6 g. The cone calorimeter testing result showed that the vermiculite-containing PVC coatings, and in particular the copper ion modified vermiculite, provided excellent fire barrier properties. Laminates with a PVC coating containing copper-ion-modified vermiculite showed a significant reduction in the peak heat release rate (pHRR), with a value of 64kW/m2, compared to 677kW/m2 for uncoated high-density polyethylene (HDPE). This study demonstrates the development of an effective PVC-based flame-retardant coating for polyethylene using copper-ion-modified vermiculite.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
ORCID iDs
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
