Abstract
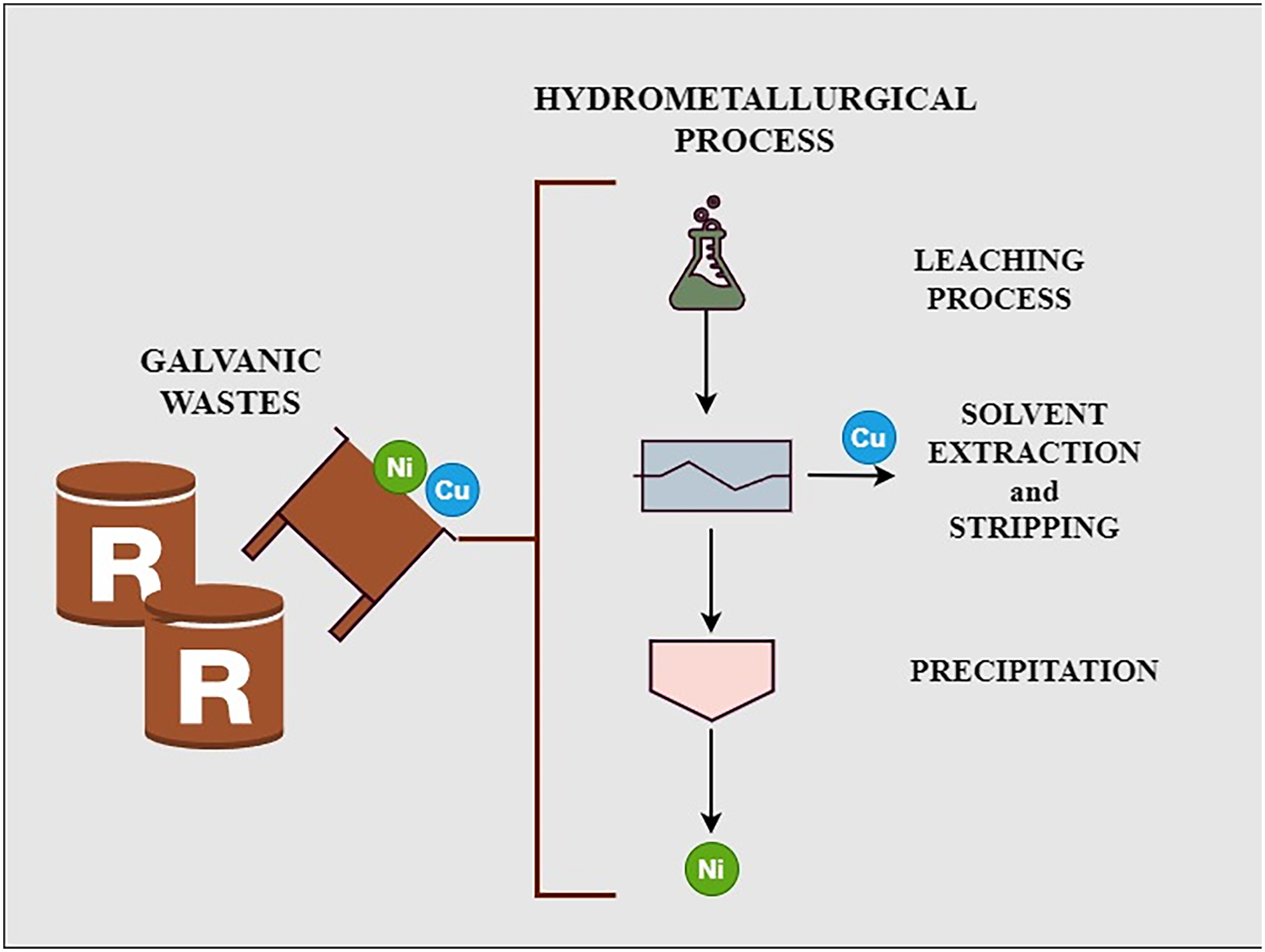
The article focuses on solvent extraction to recover nickel (Ni) and copper (Cu) from leaching solutions obtained by dissolving galvanic sludges with residual effluents of the metal processing industry. D2EHPA diluted in Metryl GI060 was tested as the organic solvent. The first experiments were performed on synthetic solutions to define and optimize the solvent extraction processes. The optimal conditions were subsequently tested to treat real solutions containing Ni and Cu from the leaching of galvanic sludge. The results showed that the real solution behaves differently from the synthetic ones, so new optimization tests were added to maximize Cu extraction from Ni. Under the optimized conditions (pH 4.5, 15% v/v D2EHPA and aqueous/organic (A/O) equal to 2), the extraction yield for Cu was 91%, whereas it was 1% for Ni. The organic solution was subjected to stripping with sulfuric acid (1 M, A/O volume ratio equal to 1). The stripping yield was 99.7%. Two phases were obtained: CuSO4 aqueous solution and the organic phase with D2EHPA/Metryl GI060, which can be recirculated to treat galvanic sludge. NaOH precipitated more than 98% of Ni from the solvent extraction raffinate. The final solid was composed of 4.31 wt% Cu and 39.58 wt% Ni. Finally, a flowsheet with a mass balance for treating 1 tonne of galvanic sludge was proposed.
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
