Abstract
According to the state of the art, most of the mixed copper and copper alloy scrap and residues are processed in a copper smelter. Despite the environmental and economic advantages relative to primary production, the recycling of copper and its alloying elements (zinc, tin, lead, nickel, etc.) requires significantly more energy and cost than remelting unmixed or pure scrap fractions such as separate collected material or production scrap. To date, however, less attention has been given to the mechanical purification of mixed scrap. Therefore, sorting by alloy-specific components (SBASC) using an industrial X-ray fluorescence (XRF) sorting system was tested on the coarse metallic fraction (10–32 mm) of mixed foundry residues. The findings show that XRF–SBASC can recover higher-grade copper concentrates (reaching 98.3% Cu), leaded brass and complex alloys, such as aluminium bronze and red brass with high purities, for the use in the production of new materials. XRF–SBASC can therefore contribute to a more resource efficient metal recycling, mainly by reducing the energy consumption and loss levels in copper metallurgy.
Keywords
Introduction
Copper and copper alloys are widely used due to their unique properties, such as excellent electrical conductivity and corrosion resistance (Davis, 2001). Since 2012, demand has grown from 24.7 Mt to around 31.1 Mt, an increase of around 26% (IWCC, 2023), driven by a global population growth, economic growth and a transition to a more sustainable society (Kleijn et al., 2011). Nevertheless, the environmental impact (global warming, air pollution, acidification, etc.) of copper products is high (Norgate et al., 2007). In 2016, copper production caused almost 0.4% of total global greenhouse gas (GHG) emissions (Hertwich, 2021) which are mainly related to electricity supply (Norgate & Rankin, 2000). In contrast, recycling of copper and copper alloy scrap is much more environmentally friendly than primary production, reducing energy consumption by up to 85% (Rankin, 2011). However, the savings in energy levels and the associated GHG emissions highly depend on the metallurgical recycling route and therefore on the scrap quality. Basically, there are two recycling options for copper and copper-based materials (Cusano et al., 2017; Schlesinger et al., 2022): direct recycling (remelting) and conventional recycling (smelting and refining). To date, most scrap is processed via the smelting and refining (S&R) route, which requires high energy input (Jolly, 2013) and results in high processing costs (Langner, 2011). This route is similar to that of the primary smelter, and it focuses on cathode production. It includes several stages to sequentially increase the copper (Cu) concentration by removing metallic and non-metallic impurities (Schlesinger et al., 2022): smelting/reduction (~80% Cu), conversion (~96% Cu), fire refinement (~98.5% Cu) and electrolytical refinement (>99.99% Cu). Consequently, depending on the copper content, some stages can be bypassed. Although low-grade materials, such as foundry residues (~10–40% Cu), have to undergo all refining steps, a mixed red brass scrap (~70–85% Cu) can be directly charged to the converter (Samuelsson & Björkmann, 2014; Schlesinger et al., 2022). High-grade copper scrap, such as No. 1 copper scrap with 98% Cu, requires little refining and can therefore be charged to the fire refining furnace (anode furnace), bypassing the smelting and conversion stages. By reprocessing smelter by-products, such as slag and flue dust, the removed alloying elements can be partially recovered (Antrekowitsch et al., 2014; Ayres et al., 2002; Hanusch & Bussmann, 1995). However, the recovery process is complex and often involves metal losses, therefore limiting the potential for energy savings and GHG reductions (Ciacci et al., 2016).
In contrast, scrap, such as pure scrap from manufacturing or fabrication, is kept strictly separate in terms of composition, and it can be directly recycled, significantly reducing the energy footprint (Jolly, 2013; Loibl et al., 2021). Furthermore, direct recycling is a simpler and more cost-effective process from a metallurgical point of view than the complex S&R route (Graedel et al., 2011; Langner, 2011). Direct recycling has the advantages of retaining the values of the added alloying elements that will be lost if the materials are sent to a smelter and of saving the cost of refining (Schlesinger et al., 2022). Therefore, for both economic and environmental reasons, the aim should be to increase the amount of material entering the direct recycling route. In practical terms, complex scrap and residues that are not kept separate must be mechanically processed and sorted prior to metallurgy. Conventional processing techniques mainly include shredding/crushing, screening, magnetic separation and eddy current separation (Heinrichs et al., 2014; Martens & Goldmann, 2016). To obtain recyclates with high market values, recycling companies often use more sophisticated separation technologies, such as sensor-based sorting (SBS) (Kutila et al., 2005; Neubert et al., 2014; Wens, 2015). However, SBS technologies, such as X-ray transmission or colour recognition, reach their limits in regard to sorting by specific alloying components (SBASC). SBASC is only possible using spectrometry techniques, such as X-ray fluorescence (XRF), which allows differentiation based on chemical composition (Macherauch & Zoch, 2014). Therefore, in recent years, industrial XRF sorters have been increasingly used to separate stainless steel, copper, brass and zinc scrap (Neubert et al., 2014; Pfandl et al., 2020).
However, the use of XRF sorters for copper-based scraps and residues, especially those with complex compositions, offers further applications with undeveloped potential. This phenomenon is particularly true for mixed residues from the copper alloy foundry industry. Due to the high production volumes of copper alloys, such as brass alloys, economically significant quantities of by-products, such as slag and dross, are generated each year (Kilicarslan et al., 2014). These by-products contain significant metal values bound in the metallic parts (coarse fraction) and in the non-metallic fraction (fine fraction, also referred to as ash) (Abdel Basir and Rabah, 1999; Maleki et al., 2022; Timur et al., 2000). Although several studies have already investigated the ash treatment (Kahvecioglu et al., 2013; Kilicarslan & Saridede, 2014), less attention has been given to the metallic fraction. Remelting into a new copper alloy, however, requires materials with specified compositions and sorting by alloy-specific components (SBASC) if it consists of mixed alloy metals. In particular, the need is illustrated by the example of the European standard for ingots and castings (European Committee for Standardization, 2017), which distinguishes between a total of 50 copper alloys. To meet the compositional requirements and to adjust the composition, foundries usually blend different scraps before charging the furnace to produce a copper alloy (Cusano et al., 2017; Jolly, 2013). Consequently, there is a high demand for more specified scrap that can be supplied through SBASC using XRF sorters.
Given the existing lack of research and the environmental and economic advantages of direct recycling of copper alloy scrap separated into composition classes, SBASC was tested in large-scale trials using an XRF sorter on the metallic fraction of a mixed foundry residue sample. The developed processing concept covered two major objectives: (a) separating the alloyed material to obtain recyclates that can be returned to foundries for alloy production and (b) recovering high-grade copper concentrates that require less refining. The main focuses of this study are investigations of the efficiency and performance in terms of XRF–SBASC, but also on potential applications of the sorting products for recyclers such as foundries, semis-fabricators and smelters.
Materials and methods
Test material
The material used for the sorting trials is a sample (~4.3 tonnes) of a pre-treated metallic fraction (10–32 mm) of mixed foundry residues, where the pre-treatment includes crushing, ball milling and screening. The main constituents are metallic copper and copper alloy particles, in the form of droplets, nuggets, cuttings or unmolten scraps. For simplicity, the material covered in this study will be referred to as scrap. Due to the complex composition and the wide variety of different types of scraps, the material mix contains several valuable alloying elements (tin, nickel, manganese, etc.) in varying proportions in addition to copper.
Multi-stage mechanical sorting process
Recovery occured in a multi-stage sorting process. In total, the processing concept comprised eleven sorting stages (Figure 1).
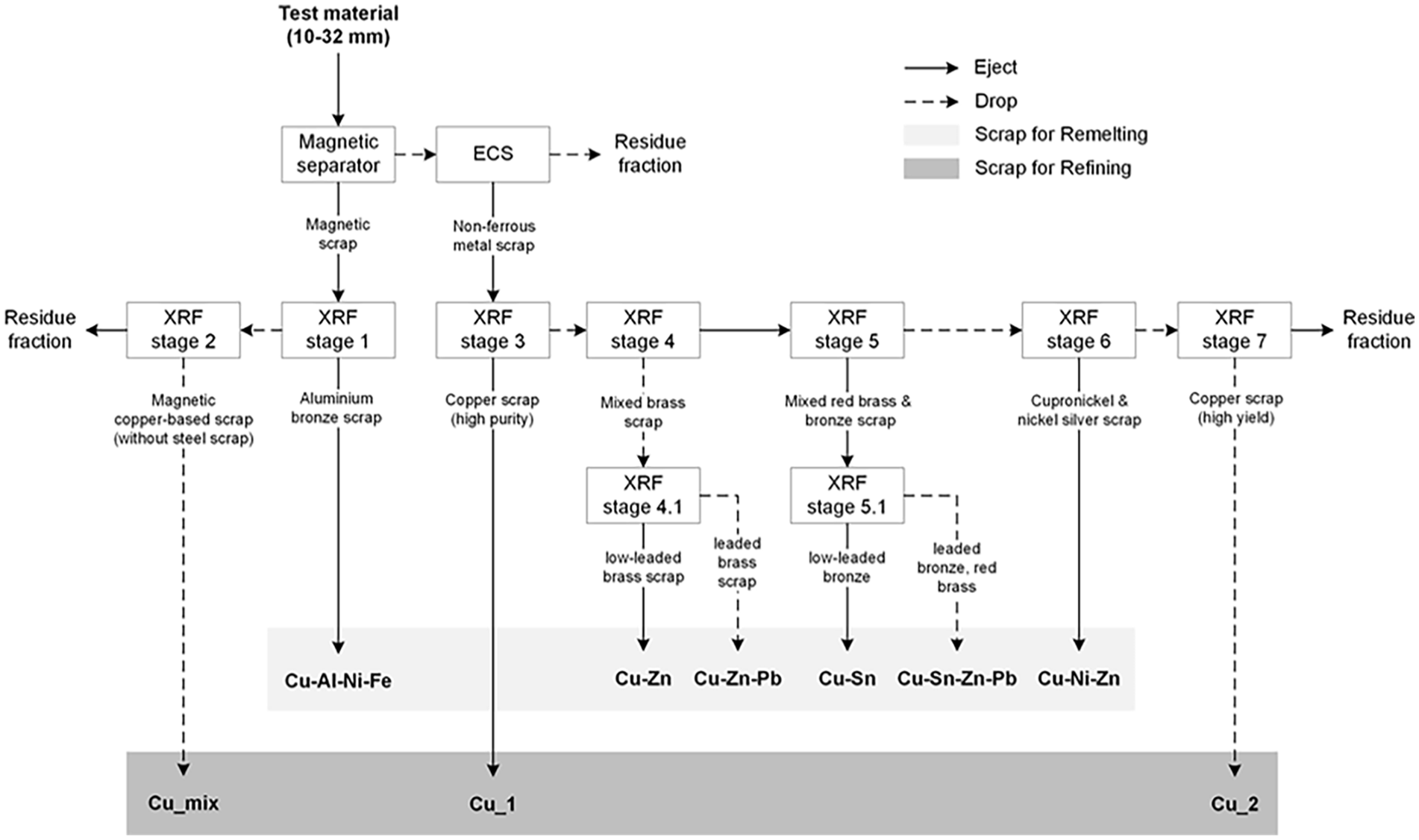
Processing concept for the large-scale trials. ECS: eddy current separation and XRF: X-ray fluorescence.
The pre-sorting included a magnetic separation stage (neodymium magnetic drum) to enrich aluminium bronze scrap alloyed with iron (Fe), nickel (Ni) and ferrous metal scrap. The eddy current separator removed nonmetallic impurities such as slag, plastics, wood and textiles, from the non-magnetic fraction.
The XRF–SBASC was performed in several subsequent stages. Due to the ball-shaped geometry of the objects, an XRF chute sorter (STEINERT CHUTEC® CSS 140 XF L) was used.
As illustrated in Figure 2, the general functional principle is described as follows (Neubert et al, 2014; Steinert, 2013): The feed material is segregated by a vibratory feeder, it slides down the chute and it passes through the detection area where the X-ray tubes emit X-rays onto the material. Within milliseconds, the energy dispersive XRF spectrometer detects and interprets the resulting secondary X-rays emitted to determine the elements. The software in the data processing unit analyses the spectra and classifies each object. Depending on the different sorting objectives of each stage, the classified objects are assigned to target objects, which are mechanically ejected by compressed air jets or to non-target objects that are dropped. A special feature is the integrated 3D laser triangulation (3D-LT) sensor. The 3D-LT sensor information increase the accuracy of classification and separation of the individual objects. To optimise the SBASC for copper-based scrap, the classification software was upgraded prior to the sorting trials. As a first step, over 2,000 scrap objects of similar material were manually grouped according to their specific alloying components using a handheld XRF spectrometer. The sorter then analysed the spectra of these groups, which were embedded in the software to extend the classification.
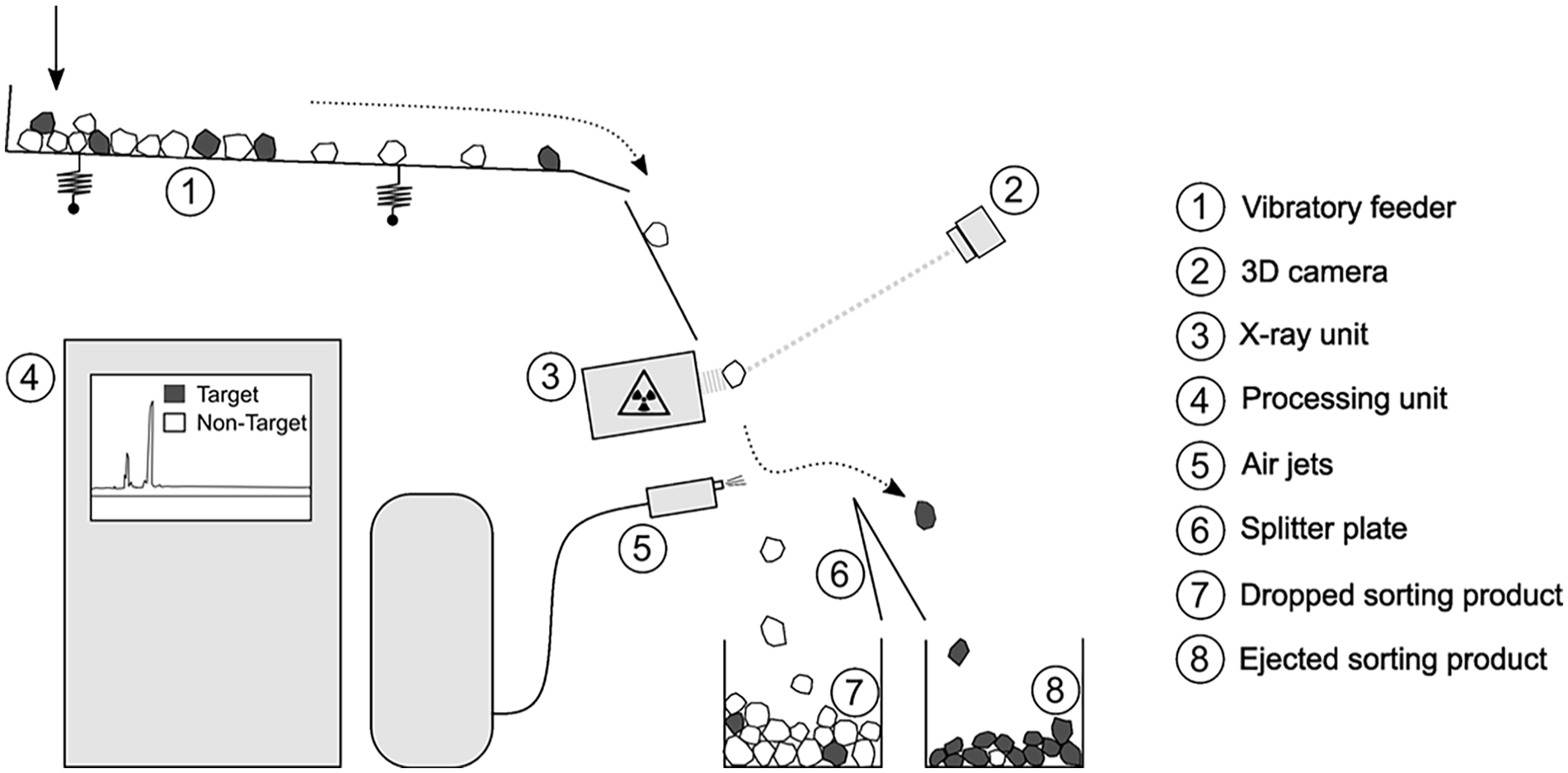
Functional principle of the XRF chute sorter (based on (Steinert, 2013)).
The sorting objective of XRF stage 1 was to recover the aluminium bronze scrap with high purity from the magnetic product. In XRF stage 2 the remaining material, that is, the dropped fraction of stage 1, was returned to the sorter to enrich a mixture of magnetic copper alloys (in particular special brass containing the alloying elements Fe/Ni and thus being magnetic) in Cu_mix by targeted separation of ferrous materials.
The non-ferrous eddy current product was sorted in XRF stage 3 to recover the high-purity copper objects (target: >95% Cu), whereas XRF stage 4 focused on enriching mixed brass scrap from the remaining material. Mixed brass was expected to comprise more than 50% of the remaining material of XRF stage 3; thus, it was dropped in the sorter. This sorting approach reduces the consumption of compressed air, which is the main variable cost factor in SBS processes (Bunge, 2012). In XRF stage 4.1, the mixed brass scrap was further sorted according to the Pb content.
Scraps containing tin (Sn) as their main alloying element such as tin bronze, lead bronze and red brass were enriched in XRF stage 5, whereas the high-leaded and low-leaded scrap were separated in XRF stage 5.1. The recovery of nickel-rich scraps, such as nickel silver and cupronickel was tested in XRF stage 6. XRF stage 7 was a final downstream step to recover the remaining copper scrap, focusing on high-yield sorting by ejecting the lower-grade copper scrap and other unwanted material in the residue fraction. The average throughput of each XRF sorting stage was between ~3.0 t h−1 and ~3.5 t h−1 (working width: 1.4 m).
After sorting, the recovered products were sampled for the melting experiments and purity assessment. An overview of the methodological approach is given in Figure 3.
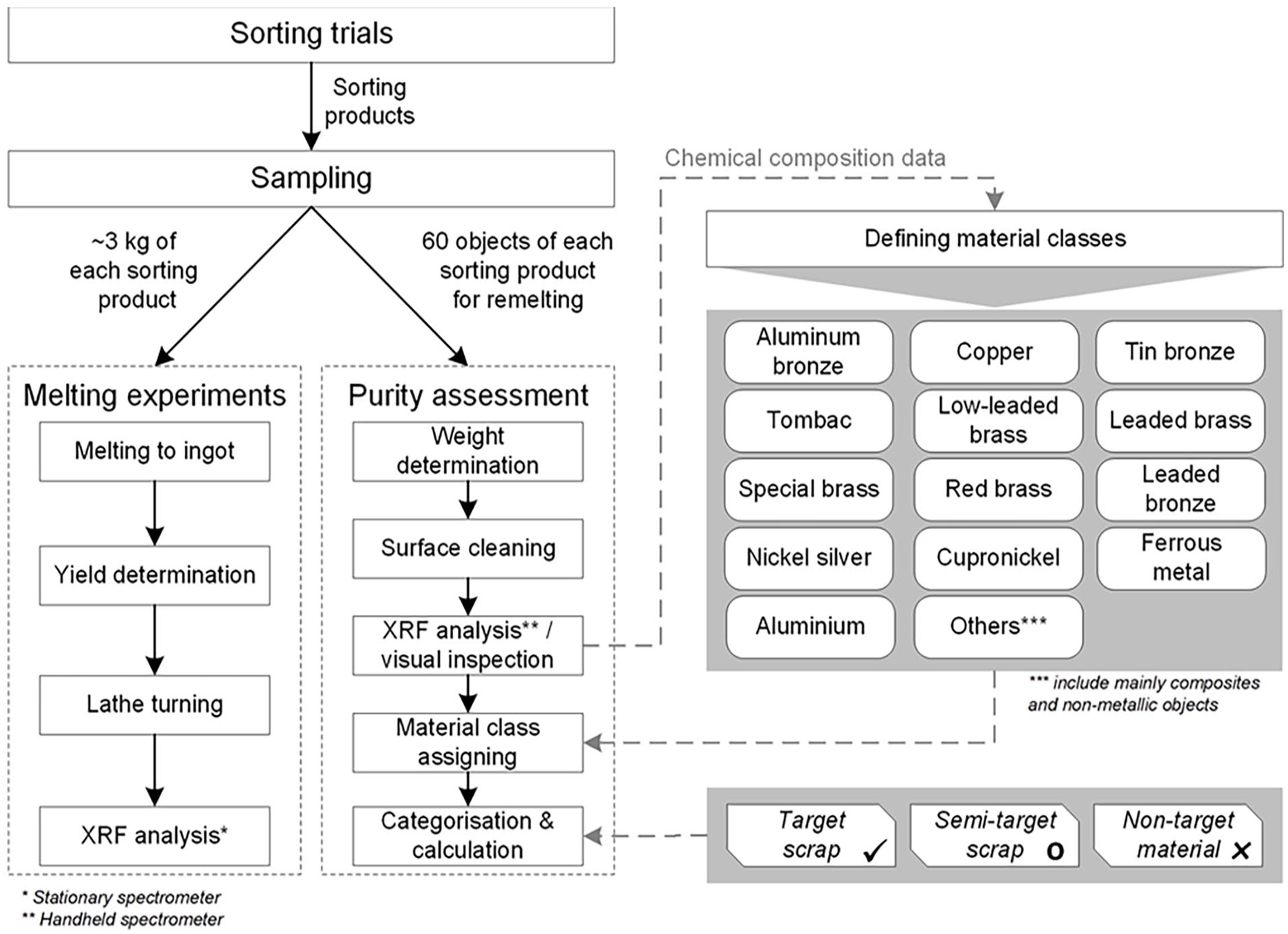
Methodological approach for the purity assessment and melting experiments.
Purity assessment
Purity is a key parameter in terms of sorting processes (Bunge, 2012), and it was used in this study to assess the sorting performance of each stage.
This assessment required a comprehensive characterisation of the sorting products, that is, a determination of the correctly and incorrectly sorted materials. For this purpose, 60 objects were randomly sampled from each product that was intended to be used as remelting material (Cu–Al–Ni–Fe, Cu–Zn, Cu–Zn–Pb, Cu–Sn and Cu–Sn–Zn–Pb). After sampling, the individual objects were weighed and classified using a mobile XRF spectrometer (handheld). Material classes were defined and the objects were assigned to these classes based on the handheld XRF results of visual inspection (e.g. foreign materials). In general, the major advantage of the complementary use of the handheld XRF for classification is that it allows a more detailed differentiation of the alloys than a differentiation based solely on visual and cognitive recognition, using standard tools, such as a magnet, file and acids (Brooks and Gaustad, 2021).
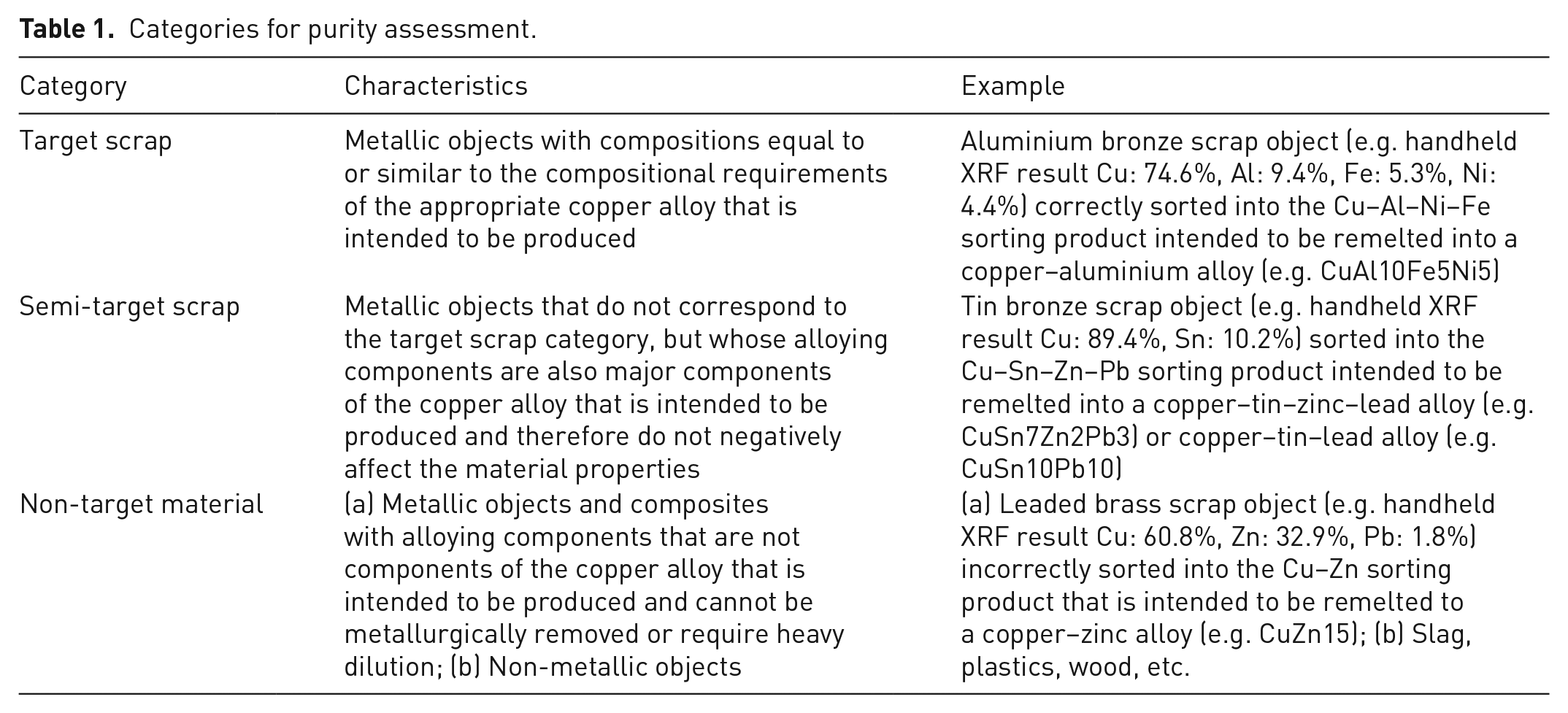
Prior to purity calculation, for each sorting product, the material classes were categorised as (a) target scrap (TS), (b) semi-target scrap (STS) and (c) non-target material (NTM). A detailed description of the categories is given in Table 1.
Categories for purity assessment.
The purity

The procedure was therefore similar to manual sorting, but performed in reverse, based on the data collected from each object by the characterisation.
Melting experiments and chemical analysis
Purity alone as a single parameter is useful to assess the sorting performance. However, it does not provide any information on the chemical composition, which is essential for foundries and semi-fabricators wanting to use the recyclates to produce new copper alloys. Another important aspect is the metal yield obtained when the material is remelted, as melting losses (through slag or skimmings) represent a direct monetary loss and should therefore be kept to a minimum.
For this purpose, melting experiments were performed, followed by chemical analysis using a stationary wavelength dispersive (WD) XRF spectrometer. Sampling and sample preparation were carried out according to LAGA PN 98 (Ländergemeinschaft Abfall, 2001) to obtain a 3 kg sample of each sorting product. After removal of free iron, each sample was remelted with flux (borax) in an induction furnace to produce homogeneous sample for chemical analysis. The molten metal was poured into a mould for cooling.
The metal yield

Finally, the cooled metal sample was analysed to determine the chemical composition. Prior to the analysis, the sample was cut to an appropriate size to fit into the WD XRF spectrometer, turned on a lathe and polished to obtain a clean sample surface.
Results and discussion
Purity of sorting products for remelting
Purity was assessed by the share of correctly and incorrectly sorted scrap objects in the respective samples for the six copper alloy sorting products for remelting. Figure 4 shows the sample composition broken down into TS, STS and NTM.
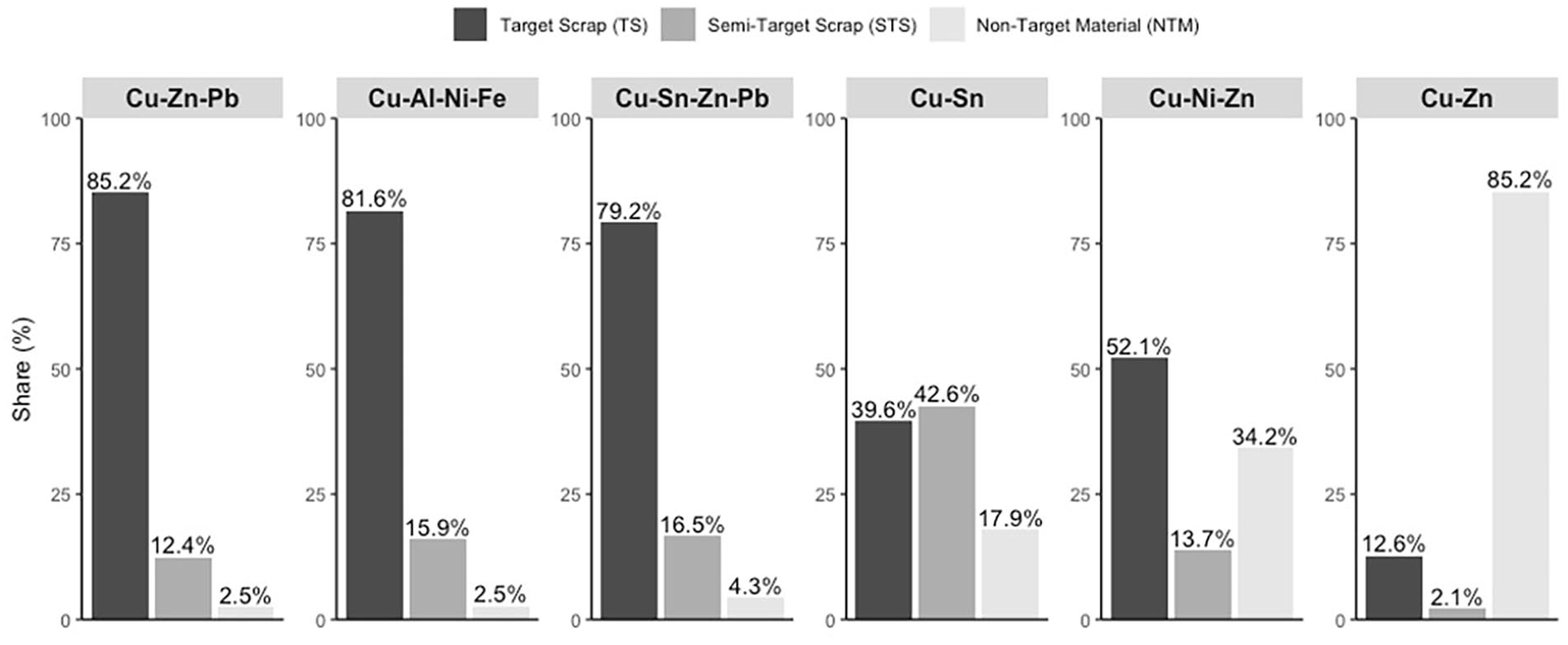
Composition of the sorting products for remelting in terms of target-scrap, STS and NTM based on the characterisation of 60 objects per sample.
The purities (sum of TS and STS) range from ~15% to 98%, with four of the six sorting products examined achieving a purity greater than 82.2%. The best sorting performance and the highest share of TS can be seen for Cu–Zn–Pb with ~85%, Cu–Al–Ni–Fe with ~82% and Cu–Sn–Zn–Pb with ~79%. In addition, these sorting products contain between ~12% and 17% STS. For example, the STS of Cu–Al–Ni–Fe includes copper scrap alloyed with small amounts of Ni and Fe. In terms of the specifications for copper–aluminium alloys, this scrap is not a contaminant here, and it is expected to result in a change in the chemical composition, for example, a higher copper content in the melting charge. Similarly, the Cu–Sn–Zn–Pb sorting product contains certain amounts of copper (~7%), leaded brass (~4%) and bronze scrap (~5%), the constituents of which are also commonly present in copper–tin–zinc–lead alloys. The STSs in Cu–Zn–Pb include copper (~2%) and brass scrap (~10%), resulting in an overall purity of about 98%. In contrast, the purity of the ejected product (Cu–Zn) is relatively low (~14%), demonstrating that the separation of unleaded or low lead brass scrap (<0.5% Pb) from the mixed brass scrap is challenging. As a result, ~85% is NTM, which mainly includes leaded brass scrap (~81%). The brass and tombac scrap, which represent the TS, have a small share in the sorting product of ~13%. A possible explanation for the poor TS recovery rate might be that the difference in the chemical composition is relatively small. In industrial sorting processes the high throughput is accompanied by short measurement times of a few milliseconds, which reduces the accuracy of element detection and makes differentiation more difficult. Longer measurement times and associated low throughputs can potentially overcome this limitation, that example, by using a belt sorter with slow belt speed and mechanical ejectors.
In contrast, Cu–Ni–Zn and Cu–Sn show different distributions: While the share of TS is lower with ~52% for Cu–Ni–Zn and ~40% for Cu–Sn, the share of STS is more than twice as high with ~32% and ~43% respectively. For Cu–Sn, the STS consists only of copper particles. This observation may be explained by the fact that tin bronze scrap often has a higher copper content than red brass and leaded bronze scrap, and it is closer to pure or low-alloyed copper. The lower proportion of copper particles remaining as STS in the dropped Cu–Sn–Zn–Pb sorting product (~7%) supports this hypothesis. The NTM, accounting for about 18%, consists mainly of red brass scrap (~13%) which was incorrectly ejected and slightly contaminates the sorting product with unwanted alloying elements (Zn, Pb). However, the overall purity of ~82% indicates a good sorting result. The Cu–Ni–Zn sorting product has purity of ~66%. Although the TS includes cupronickel scrap (~27%) and nickel silver scrap (~26%), the STS mainly consists of brass scrap (~8%). The NTM are mainly non-magnetic special brass scrap alloyed with aluminium (~19%) and aluminium bronze scrap (~6%). Aluminium is an impurity in both copper–nickel alloys and copper–nickel–zinc alloys and is therefore unwanted in this sorting product. Energy dispersive XRF spectrometers are limited in the detection of lighter elements such as aluminium and silicon (Neubert et al., 2014), which can be a possible reason for these incorrectly sorted materials.
However, there are a few possible effects that need to be considered when interpreting the purity results presented in this study. Firstly, as already mentioned above, the XRF sorter is designed for high throughput sorting resulting in short measurement times. These conditions limit the detection accuracy relative to a stationary/handheld spectrometer that allows longer measurement times (often combined with surface cleaning prior to the measurement). In addition stationary/handheld spectrometers allow measurements to be made directly on the surface, which is not achievable with industrial XRF sorters. These conditions cause deviations in the sorting decisions and affect the accuracy of distinguishing between scrap materials with minor differences in chemical composition, as demonstrated by the separation of leaded and low-leaded brass. Secondly, the interpretability of the results obtained is limited by the small sample size and sampling errors may have occurred. Another source of uncertainty is the possibility of false readings (e.g. caused by contaminated material surfaces with e.g. metallic dust or mud), which can lead to the object being assigned to the wrong material class. Consequently, the results are not fully representative of the XRF sorting performance for foundry residues in general. To validate the results and to gain a robust understanding of the variance, further experiments by changing the following parameters are needed: (a) input material compositions, (b) machine settings, such as eject sensitivity and air jet adjustments, (c) sequences of the sorting stages and (d) extent of mechanical pre-treatment (crushing, screening, etc.).
Chemical composition and metal yield of all sorting products
Table 2 shows the results of the melting experiments for the samples of each sorting product, separately for the intended recycling route. The metal yields range between 96.5% and 98.3%, indicating no significant variances between the molten samples. The melting losses (3.5–1.7%) are caused by the formation of dross, volatilisation (mainly of Zn), non-metallic impurities, oxides and unmolten metallic impurities with a higher melting point than copper-based materials (e.g. free iron).
Results of the chemical analysis of the remelted ingots of the focused sorting products.
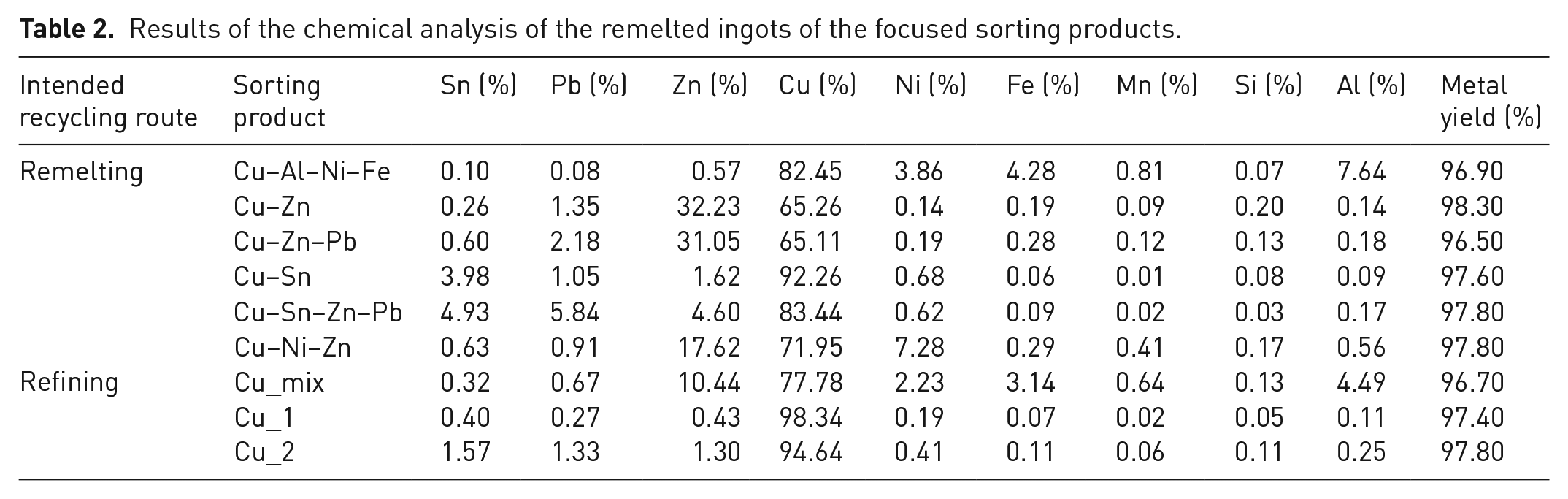
As expected, the results of the chemical analysis of the ingots show clear differences between the recovered sorting products regarding the alloying elements (Sn, Pb, Zn, Cu, Ni, Fe, Mn, Si and Al). Therefore, the products intended for remelting (except Cu_mix) have a significantly higher content of alloying elements than the copper concentrates Cu_1 and Cu_2 intended for refining.
The Cu–Al–Ni–Fe ingot contains high contents of Al (~7.6%), Fe (~4.3%) and Ni (~3.9%), which are characteristic of complex copper–aluminium alloys. The ingot has the highest content of Mn (~0.8%) relative to the other ingots. The contents of Zn (~0.6%), Sn (~0.6%) and Pb (~0.1%) are relatively low and are still above the limit values, considering the specifications for copper and copper alloys. The excessively high contents may be explained by metallic inclusions (e.g. one object covering another) or adhesions caused by the mechanical pre-treatment, that is, shredding and ball-milling. The XRF sorter cannot detect these inclusions, resulting in impurities being inadvertently carried into the sorting product. However, for some alloys, such as CuAl10Ni5Fe4, the chemical composition of the ingot is already close to the requirements. One method of fully meeting the requirements is the addition of high purity scrap to the furnace charge, particularly for Pb reduction. Pb affects the mechanical properties of copper–aluminium alloys (Jablonski, 2014); thus, the content is often limited to 0.05% (0.03% for ingots and castings).
The main alloying element of the Cu–Zn ingot (eject of XRF stage 3.1) is Zn with a content of ~32.2%, followed by Pb with a content of ~1.4%. However, the specifications for copper–zinc alloys limit Pb to, for example, 0.1% for CuZn37 and 0.2% for CuZn40. Although the excessive Pb content probably requires large amounts of new material to be added to the charge for remelting, the content is still ~40% lower than that in Cu–Zn–Pb (~2.2%). XRF–SBASC therefore enables at least a downstream Pb reduction by implementing a second sorting stage in the processing concept. Surprisingly, the composition of the Cu–Zn–Pb ingot (drop of XRF stage 3.1) is relatively similar to that of the Cu–Zn ingot (with the exception of Pb). However, due to the higher Pb content, this sorting product is more suitable for remelting copper–zinc–lead alloys such as CuZn33Pb2. For both Cu–Zn and Cu–Zn–Pb, Al, Si and Mn are minor impurity constituents, which can probably be removed by melt refining, e.g. oxygen blowing. By comparing the ingots of Cu–Sn (eject of XRF stage 4.1) and Cu–Sn–Zn–Pb (drop of XRF stage 4.1), the differences in chemical compositions are greater. Thus, for Cu–Sn, Pb is ~80% lower, Zn is ~65% lower, and Cu is ~10% higher than for Cu–Sn–Zn–Pb. The more surprising result, however, is the ~20% lower Sn content in the Cu–Sn ingot. There are two possible explanations for this unexpected result, which influence each other: On the one hand, as shown in the purity assessment, the Cu–Sn sorting product still contains a certain amount of copper scrap, which may have increased Cu and reduced Sn during the homogenisation of the melt. On the other hand, the leaded bronze scrap recovered in Cu–Sn–Zn–Pb has also a high Sn content, which can account for the higher content. The intended remelting of a copper–tin alloy from the Cu–Sn sorting product is possible, considering the specifications for CuSn10. However, as with the other sorting products, blending with other scraps and/or some refining is required here to adjust the composition. Under similar conditions, Cu–Sn–Zn–Pb can be used to remelt a copper–tin–zinc–lead alloy, such as CuSn5Zn5Pb5.
Another promising result is given by the chemical composition of the Cu–Ni–Zn ingot (eject of XRF stage 5): The relatively high Ni content of ~7.3% proves an effective recovery of Ni-rich materials such as cupronickel scrap. Additionally, the high Zn content of 17.6% indicates the presence of nickel silver scrap, which corresponds to the findings of the purity assessment. For remelting a copper–nickel alloy, such as CuNi10Fe1Mn1, a further sorting step is useful, focusing on the separation of cupronickel and nickel silver scrap. However, this was beyond the scope of the trials due to the small sorting product amount recovered in this stage.
In contrast to the alloy sorting products for remelting, the copper scrap concentrates recovered in XRF stages 1, 2 and 6 are intended for refining, that is, to produce cathodes. By focusing on high purity and thus positive sorting only high-grade copper scrap particles in XRF stage 2, a comparatively high Cu content of ~98.3% is achieved for Cu_1. Due to the low level of impurities, it is expected that this concentrate can be directly charged into the anode furnace for fire refining, bypassing prior refining stages such as smelting and converting. Moreover, the plant operator can benefit from a lower refining charge, while at the same time receiving a higher profit on the sale of the sorting product. The same applies to Cu_2, for which the ingot also has a relatively high copper content of ~94.6%. This result demonstrates the importance for this downstream sorting stage to recover the remaining copper scrap particles by focusing on high-yield sorting. An alternative recommendation would be to adjust the machine settings in stage 3 in terms of ejection sensitivity to replace XRF stage 7. However, the effectiveness of this adjustment has to be tested in further sorting trials. In contrast, the copper scrap recovered in stage 2 still contains high proportions of alloyed scrap, which is particularly evident in the complex composition of the Cu_mix ingot. Two main conclusions can be drawn from this analysis result: although the Zn content of ~10.4% indicates the presence of special brass scrap in this sorting product, the Al content of ~4.5% suggests that there is still a certain amount of aluminium bronze scrap that has not been recovered in stage 1. The implementation of a further downstream XRF stage may therefore be a useful addition. Due to the strict limitations on Pb content for copper–aluminium alloys, it is preferable to focus on the high-yield sorting of special brass scrap.
In summary, the results highlight the importance of melting experiments and chemical analyses to determine both the value and the most resource efficient recycling route for the sorting products obtained.
Conclusions
SBASC was tested on the metallic fraction of mixed foundry residues from the copper alloy industry using an industrial XRF chute sorter. For this purpose, the material was processed in several stages in large-scale sorting trials. The main goal of the study was to investigate the purity and chemical composition of the sorting products obtained. The results proved that under certain conditions the alloy scrap sorting products Cu–Al–Ni–Fe (aluminiumbronze scrap), Cu–Zn–Pb (leaded brass scrap), Cu–Sn (tin bronze scrap) and Cu–Sn–Zn–Pb (red brass and leaded bronze scrap) are suitable for direct recycling, that is, for the production of new copper alloys. Due to the impurities identified in the sorting products, these products require blending with other scrap and/or undergo some refining in order to meet the compositional requirements, for example, those specified in the European standards for ingots and castings (European Committee for Standardization, 2017). Despite the additional material required for blending, this recycling route has the advantage that the alloying elements remain in the associated alloy system rather than being slagged, increasing the resource efficiency, reducing CO2 emissions and decreasing melting costs. Consequently, the added value of accurate alloy sorting can also increase the recycler’s profit. Furthermore, two higher-grade copper concentrates were recovered that are also recyclable with significantly less energy and cost due to the high copper content achieved (Cu_1: ~98.3%, Cu_2: ~94.6%).
Although the study successfully demonstrated SBASC on a complex copper-based scrap, some results indicated possible limitations in terms of the depth of sorting. For example, the separation of leaded and low-leaded brass scrap (Cu–Zn, Cu–Zn–Pb) has illustrated the challenge of sorting scrap with only small differences in the chemical composition. This phenomenon can be attributed to the short measurement times of XRF chute sorters. However, it should be considered that each additional sorting step incurs extra cost and therefore only makes sense if the purity is required for metallurgy or sales purposes. From this point of view, the ambitious goal of separating all alloy families that are present in the foundry residues addressed in this study does not seem effective, nor is it logistically feasible. The decisive factor is that the entire recycling chain, that is, the combination of SBASC and metallurgical recovery, is less energy and cost-intensive than sending the unprocessed material to a smelter. However, a detailed quantification of the environmental and economic benefits of the tested multi-stage processing concept would require a comprehensive assessment and is beyond the scope of this article. As this study was the first to investigate SBASC of copper alloys, further research on the sorting depth is needed to optimise the interface between mechanical processing and metallurgical recycling.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
