Abstract
The decarbonisation of the steel and cement industry is of utmost importance in tackling climate change. Hence, steel production in modern integrated steel mills will be shifted towards electric arc furnaces in the future, in turn causing dwindling supplies of blast furnace slag, which is used as a supplementary cementitious material inter alia to reduce the CO2 emissions of cement production. Achieving a sustainable circular steel and building material economy requires the valorisation of currently landfilled steel slags and investigating utilisation options for electric arc furnace slag, which is increasingly being generated. For this purpose, different metallurgical residues and by-products were treated by carbothermal reduction in an inductively heated graphite crucible and then rapidly cooled by wet granulation, yielding a slag fraction similar to granulated blast furnace slag and a metal fraction valuable as a secondary raw material. A spreadsheet-based model was developed to calculate residue combinations to accomplish target compositions of the slag and metal fractions to fulfil previously identified requirements of the targeted cementitious and ferrous products. The results demonstrate the high accuracy of the model in predicting the properties (e.g. main oxide composition) of the generated slag and metal fraction, which fulfil the needed requirements for their use as (i) a supplementary cementitious material and (ii) a secondary raw material in steel production.
Keywords
Introduction
With a worldwide production of 4300 Megatonnes (Mt) for cement and 1950 Mt for crude steel in 2021, both commodities represent enormous importance for the global economy (International Energy Agency [IEA], 2022; Worldsteel Association, 2022). Their very energy-intensive production still relies predominantly on the use of fossil fuels, resulting in 1202 million tonnes of oil equivalent (Mtoe), which accounts for 4.94 Gigatonnes (Gt) of carbon dioxide (CO2) emissions in 2019 (IEA, 2020). However, the ambitious goal of being the first climate-neutral continent formulated within the European Green Deal includes the net-zero emissions plan for steel and cement production by 2050 (Fetting, 2020). Besides integrating carbon-capture and storage/utilisation (CCS/CCU) technologies, these targets can only be achieved by substantially shifting from fossil-fuel-based steel manufacturing processes to deploying green steel technologies (IEA, 2020). Therefore, a substantial drop in European crude steel production via the blast furnace (BF)/basic oxygen furnace (BOF) route will be expected in the next two decades. This pathway is also followed by the Austrian steel manufacturer voestalpine, who plans to start operating two new electric arc furnaces (EAF) in 2027, one each at Linz and Donawitz site, with the use of hydrogen technology playing a significant role in this process in the future (voestalpine AG).
In the cement industry, inevitable process emissions occur during clinker production due to the decarbonisation of limestone. In addition, further CO2 emissions are caused by, for example, thermal energy generation from fossil fuels or transport and mining processes. Besides strategies for compensating CO2 emissions by CCS/CCU technologies (IEA, 2020), a widely established method to reduce the CO2 intensity of cement production is the substitution of clinker with supplementary cementitious materials (SCMs) (Lothenbach et al., 2011). Currently, fly ash from coal combustion and ground granulated blast furnace slag (GGBFS) from pig iron production are used preferentially and with increasing demand from cement producers. Nonetheless, the availability of fly ash and GGBFS will decrease due to the decline of coal-fired power plants and the expected transformation of the steel industry described above (IEA, 2018, 2020). Although new materials, such as calcined clays, have been identified as suitable SCMs in the last decades, there is a need for additional alternative SCMs to compensate the reduced availability of ‘traditional’ SCMs and to reduce the global clinker-to-cement ratio from 0.72 in 2019 to 0.61 in 2070 (Favier and Wolf, 2018; IEA, 2018; Leitner et al., 2014). Additionally, blended cements consisting of Portland cement clinker and latent-hydraulic slags like GGBFS can increase durability, especially under chemical attack and aggressive environments (Amran et al., 2021; Hewlett and Liška, 2019).
Along the different steel production routes, various side streams, such as slags, dust, sludges and gases accumulate, whereas slags make up 90% of the total waste and by-product mass. Besides the uptake of unwanted detrimental steel constituents, slags from iron- and steelmaking fulfil a vital function for steel manufacturing. However, depending on process conditions, even considerable quantities of valuable metals like iron (Fe), manganese (Mn) and chromium (Cr) are inevitably incorporated mostly in their oxidic state into the slag (Worldsteel Association, 2021). Therefore, a highly promising potential for valuable metal recovery, especially in EAF slag, can be exposed, whereby iron represents the highest content ranging between 5 and 42 wt% (Teo et al., 2020). Furthermore, it is known that high amounts of metal oxides, together with high crystalline contents and subsequently low amorphous or glassy contents, can cause low hydraulic reactivity of such steel slags in cementitious systems (Ehrenberg, 2006a, 2006b; Péra et al., 1999) and thereby make them unsuitable as reactive SCMs (Steindl et al., 2022).
Using steel slags in the cement industry via carbothermal reduction for recovering valuable metals and repurposing the residual slag as cementitious material has been the subject of extensive research. Yang et al. 2022 pointed out the challenge posed by the high iron oxide content in steel slags for their application in the cement industry, which can be effectively reduced through carbothermal reduction. However, a significant issue arises from the reduction of phosphorous, which simultaneously dissolves into the iron, hindering its reuse as a secondary raw material. Yu et al., 2022 investigated the optimal process conditions to mitigate this phenomenon. Cao et al. 2023 explored a method of co-modifying BOF slag with copper slag to recover valuable metals with low phosphorus content via carbothermal reduction while stabilizing the remaining slag. Additionally, Liu et al. (2017) studied the valorisation of BOF slag by incorporating SiO2 and Al2O3, emphasising the importance of process control in achieving the desired metal purity and slag structure. Kim et al. (2015) investigated the application of reduced EAF slag as a substitute for cement clinker, finding results comparable to commercial GGBFS in terms of grindability and compressive strength.
Therefore, this work aims to modify the chemical composition and properties of different metallurgical residues from steel production via BF/BOF and EAF routes through an innovative mix design and carbothermal reduction. This valorisation allows the upcycling of the materials into a slag and a metal fraction, which can be used as a SCM as well as a secondary raw material in the iron and steel industry. The process also allows purposeful modification of the resulting slag and metal compositions by adding specific secondary correction materials during the carbothermal treatment.
Materials and methods
The following chapter defines the metallurgical residues from two steel plants and the added correction materials. Furthermore, the mixture calculation, the process steps of the carbothermal reduction, the chemical and mineralogical analysis methods and the conducted reactivity tests are presented.
Material characterisation
The materials investigated in this work can be divided into the main input materials, which are, on the one hand, metallurgical residues from voestalpine Stahl Donawitz GmbH (VASD) – hot metal desulphurisation slag (HMDS) and continuous casting slag (CCS) of an integrated steel mill – and on the other hand from Stahl- und Walzwerk Marienhütte GmbH (SWM) – EAF slag (EAFS) and ladle slag (LS). Although these residues comprise the main input materials for the subsequent treatment steps, correction materials were used to modify the chemical composition of the generated output materials, mainly their contents of CaO and SiO2. For this purpose, BOF slag (BOFS) from the same integrated steel mill and fly ash (FA) from paper production were used as calcium sources and Siemens-Martin slag (SMS) from the VASD landfill site, as well as two different charges of fine waste glass (FG1 and FG2) were added as SiO2 sources. Figure 1 depicts the chemical composition of all main input and correction materials.
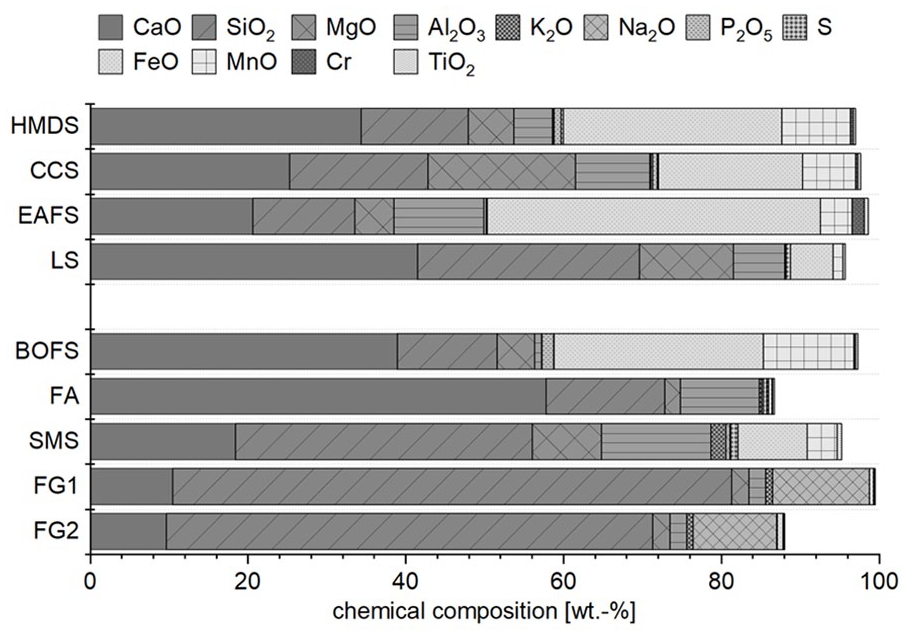
Chemical composition of the main input and correction materials used in wt%. Differences to 100 wt% are corresponding to the loss on ignition (LOI).
All investigated materials (initial and treated, see below) were crushed to <5 mm grain size. Portions were further ground for subsequent analyses and tests using a laboratory disc mill to a target Blaine fineness of 4000 cm2 g−1. X-ray fluorescence (XRF) analysis was used to determine the chemical composition of all materials. A few grams of the ground sample were ignited at 950°C for 1 hour to determine the loss on ignition (LOI). Then, glass pellets were prepared using lithium metaborate/tetraborate and 0.6–1.0 g of ignited sample. The glass pellets were then measured using a Bruker Tiger S8 (Bruker AXS GmbH, Karlsruhe, Germany) or Malvern Panalytical Epsilon 4 (Malvern Panalytical PLC, Malvern, Worcestershire, UK) spectrometer to determine the main element composition and the content of several trace elements.
The amorphous content and the mineralogical composition of all materials were determined by powder X-ray diffraction followed by Rietveld refinement with Highscore Plus 3.0.5 (Malvern Panalytical PLC, Malvern, Worcestershire, UK). Powdered samples were prepared using the back-loading technique and measured using a PANalytical X-Pert PRO diffractometer (X’Pert Pro: Malvern Panalytical PLC, Malvern, Worcestershire, UK) (Co-Kα radiation, 40 kV, 40 mA) in the range of 4 and 85°2θ with 0.008°2θ step size and 45 seconds count time per step. The amorphous content was determined by the external standard method using corundum powder (NIST SRM 676a).
Mixture calculation and limitations
The chemical composition of all available input materials was put into a database for the mixture calculation. Different materials were selected for the four mixtures based on the main input material(s) composition and the suitable correction material(s). Subsequently, limitations had to be defined for the properties and composition of the slag fraction that must be fulfilled for usage in the cement industry. A quality assessment schema was set up based on a literature review of established standards and publications to derive these target compositions of the slag fraction. This assessment matrix lists the most important chemical, mineralogical and material properties of GGBFS, such as its chemical composition and hydraulic activity (see Table 1). The chemical composition was then used to derive boundary conditions for the following mathematical modelling.
Quality assessment criteria for commercial GGBFS selected from standards (15167-1, 2006; B 3309-1, 2010a; B 3309-2, 2010b) and literature referred to in the table (Steindl et al., 2023).
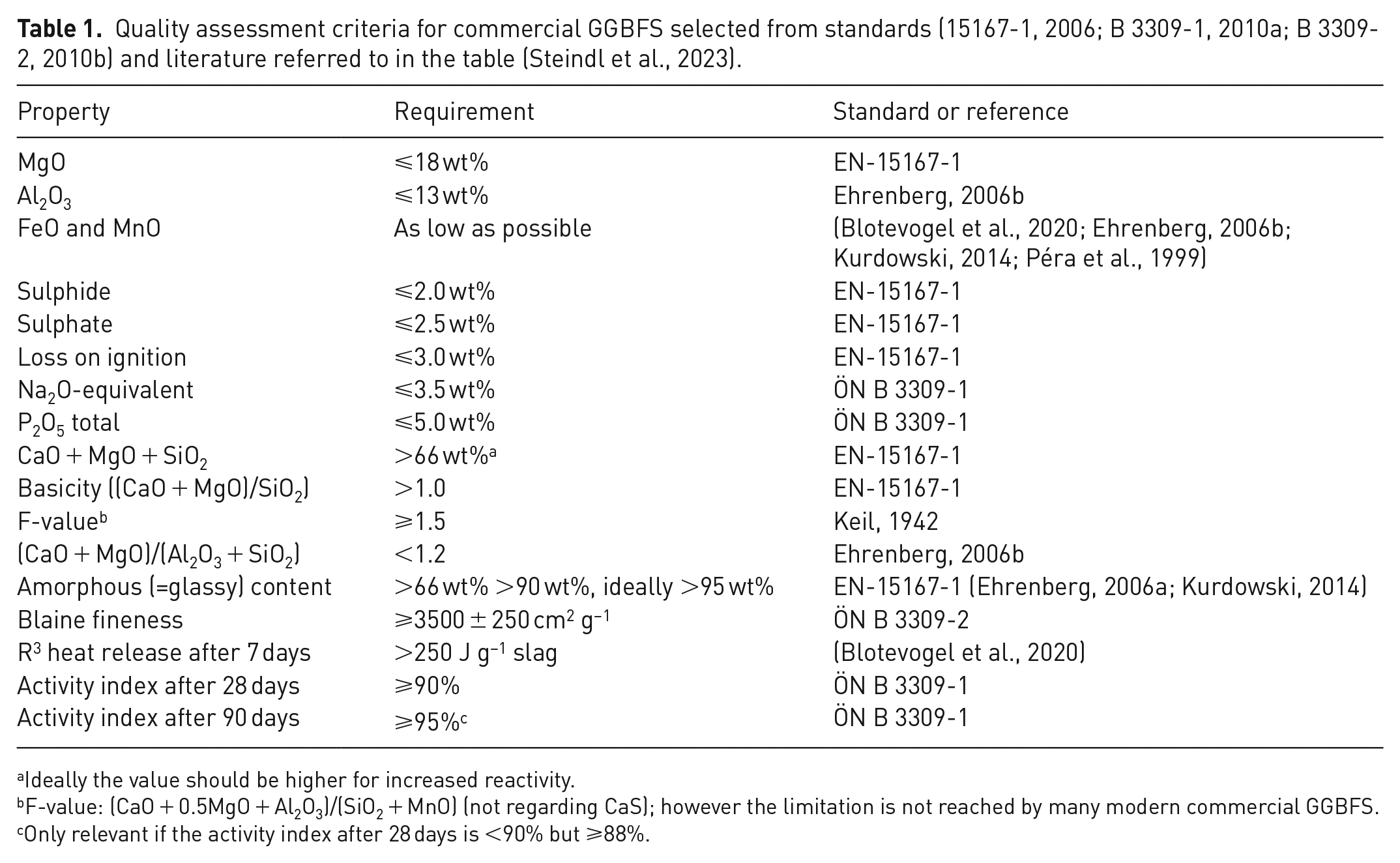
Ideally the value should be higher for increased reactivity.
F-value: (CaO + 0.5MgO + Al2O3)/(SiO2 + MnO) (not regarding CaS); however the limitation is not reached by many modern commercial GGBFS.
Only relevant if the activity index after 28 days is <90% but ⩾88%.
At the start of the calculation process, a summation procedure was initiated for each oxide or element, followed by normalisation to a standard value of 100%. The LOI, which comprises compounds lost during the thermal treatment, was subtracted. This resulting mixture was then mathematically subjected to a carbothermal reduction by applying transfer coefficients for Fe, Mn, Cr and Ti. Hereafter, the parameters of the calculated slag fraction were compared to the predefined limitations of the cement industry. If these did not match, the ratio of the main and correction materials was varied by the Microsoft (MS) Excel Solver until a suitable mixture was found or the calculation was aborted, and new input materials had to be defined (Eisner, 2023).
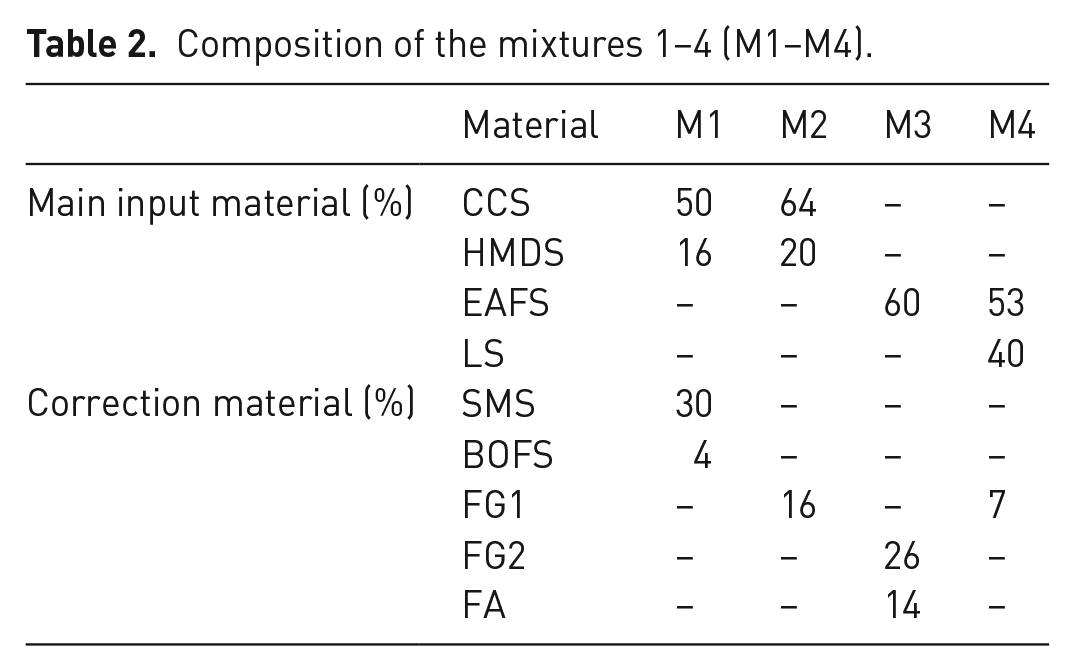
Table 2 shows the composition of the four calculated mixtures of the before-mentioned main input and correction materials. Hereby, the intention was to maximise the amount of main input material within the mixture.
Composition of the mixtures 1–4 (M1–M4).
The transfer coefficients for iron, manganese and chromium into the slag fraction for the mixture calculation were estimated through pre-experiments. It was, therefore, estimated that 2% of iron, 50% of manganese, 20% of chromium and 50% of titanium would end up in the slag fraction after the carbothermal reduction.
The amount of carbon powder as a reducing agent was determined considering the theoretical reduction reactions (1)–(4). To the calculated stoichiometric carbon demand, a surplus of 10% was added to guarantee that the mentioned metal oxides can be carbothermally reduced completely.
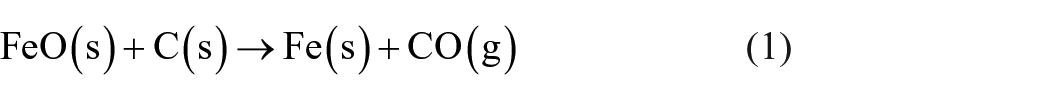
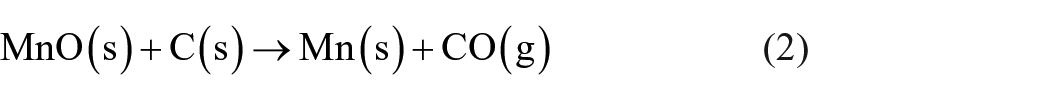
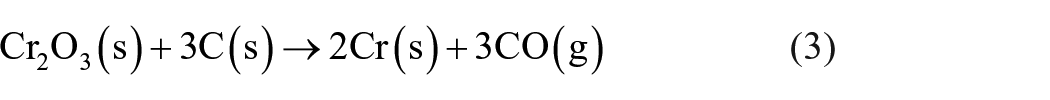
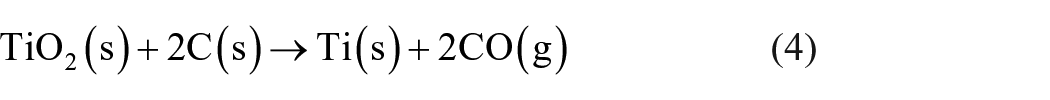
Experimental
Generally, the valuable substances in the residual materials will be isolated into two fractions: a metal fraction as a secondary raw material and a reactive slag fraction that meets the required quality for use as a supplementary cementitious material. The desired carbothermal reduction of the residual mixture is the prerequisite for recovering or separating the valuable metals. During the rapid cooling of the melt, a glassy solidification of the slag fraction must be achieved to ensure its hydraulic activity in the Portland cement system and, subsequently, the strength development in the hardening material.
The calculated input materials were mixed according to the results of the MS Excel Solver, and the required carbon powder was added. Each mixture was tested twice with an input mass of around 1.4 kg. The mixtures were melted in an inductively heated reactor above 1400°C in approximately 110 minutes. Then, a holding time of about 30 minutes at the maximum temperature was implemented. The reactor design contained an inner graphite crucible with a height of 181 mm and a diameter of 120 mm placed in an outer ceramic crucible and two layers of ceramic fibre blanket as thermal insulation. The top was closed with a graphite lid and ceramic fibre blankets. This structure should enable a reduction under the CO/CO2 atmosphere, whereby the entry of atmospheric oxygen cannot be prevented entirely. This reactor was positioned within the coil directly above the water jet of the granulation basin. A hole previously drilled in both crucibles was plugged with a graphite stopper, which was pulled from above as the holding time had passed to initiate the tapping process. The systematic cooling via wet quenching helped form a slag and metal granulate mixture collected in a net within the basin. The obtained granulate was then dried at 105°C for 24 hours. In the following, the material was crushed by a laboratory jaw crusher (Retsch BB50) to a particle size of 0.1 mm and sieved in a sieve tower (Retsch AS2002). Since the metal fraction is less brittle, it accumulates in the screen with larger mesh sizes, facilitating a rough first step of separating metal and slag fraction followed by a subsequent manual magnetic separation. Figure 2 summarises the process steps of the practical implementation.
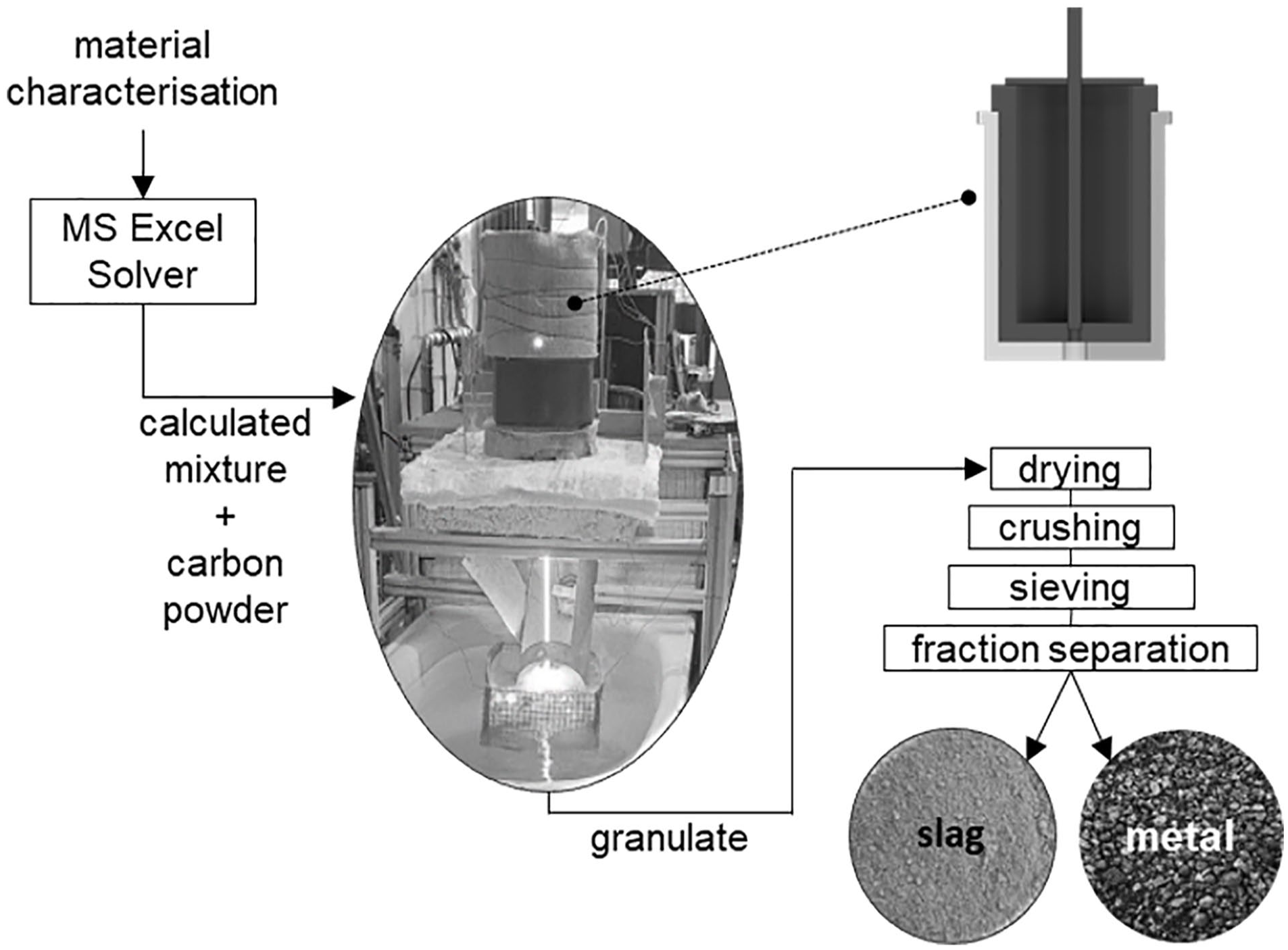
Schematic depiction of the carbothermal reduction process steps, the reactor design and the obtained fractions.
Binder suitability testing
Following chemical and mineralogical characterisation (see above), the binder suitability of the upcycled slag fractions was determined in terms of the hydraulic (re)activity of the resulting powder.
The ability of all initial and carbothermally treated materials to react hydraulically was determined with the R3 testing procedure (Li et al., 2018). Lab-grade portlandite and calcite powders were dry-mixed with the material to be tested (all kept at 40°C); then a KOH-K2SO4-solution was mixed in for 90 seconds. An isothermal calorimeter (Calmetrix I-Cal 4000H, Calmetrix Inc., Arlington, Massachusetts, USA) was used to measure the heat release from roughly 85 g total sample mass over 7 days at 40°C. The specific heat release in respect to the slag mass was calculated from the baseline-corrected heat release curve.
The activity index (AI) of the materials was determined according to (15167-1, 2006): standard mortar prisms were fabricated following (196-1, 2016) using a binder containing CEM I 42.5 R (Austrian standard cement) and slag. Although EN 15167-1 would require a ratio of CEM I 42.5 R to slag of 1:1, instead a ratio of 3:1 was used following the Austrian standard (B 3309-1, 2010). The reason for this, was that a slag content of 25 wt% in the binder (corresponding to commonly used CEM II/B-S composite cements) was judged to be more practical. The compressive strength of the demoulded and water-stored prisms (40 × 40 × 160 mm) was determined after 28 and 90 days and compared to reference values obtained from prisms fabricated in the same way with 100 wt% CEM I 42.5 R in the binder. The strength ratio (compressive strength of prisms with 25 wt% slag divided by compressive strength of reference prisms with 100 wt% CEM I 42.5 R) was used to calculate the activity index as a measure of the hydraulic activity of the slag; as an example, an AI of 100% would mean that the compressive strength of the cement–slag mixture is equal to that of pure cement. Since six compressive strength values were obtained for every sample and reference, the activity index’s standard deviation could be calculated by error propagation.
Transfer coefficients of Fe, Mn and Cr
For the analysis of the transfer coefficients of the valuable metals, first of all, the mass input of the elements Fe, Mn and Cr was determined through the chemical composition of the individual mixture before the carbothermal reduction. After the thermal treatment and processing steps, the data from the slag fraction analysis could be used to determine the mass of the elements in the obtained slag fraction under consideration. Finally, the transfer coefficients were calculated as the quotient of the mass of the element in the obtained slag fraction to the total input mass of the element. The portion not found in the slag is assigned to the metal fraction or is referred to as ‘removed’. This flow includes reduced Fe, Mn and Cr, which remain in the crucible after the tapping as well as the processing losses.
Results and discussion
The following chapter presents and discusses the results regarding the accuracy of the MS Excel Solver, the suitability of the generated slag fraction to function as a SCM, and the chemical composition of the obtained slag and metal fractions. In addition, the transfer coefficients of Fe, Mn and Cr are reviewed.
Accuracy of the mixture calculation
Figure 3 compares the chemical composition of the calculated slag fraction using the MS Excel Solver after thermal treatment and the actual analysed material. As for each mixture, two experiments were carried out, the mean value is depicted in combination with the deviation indicator.
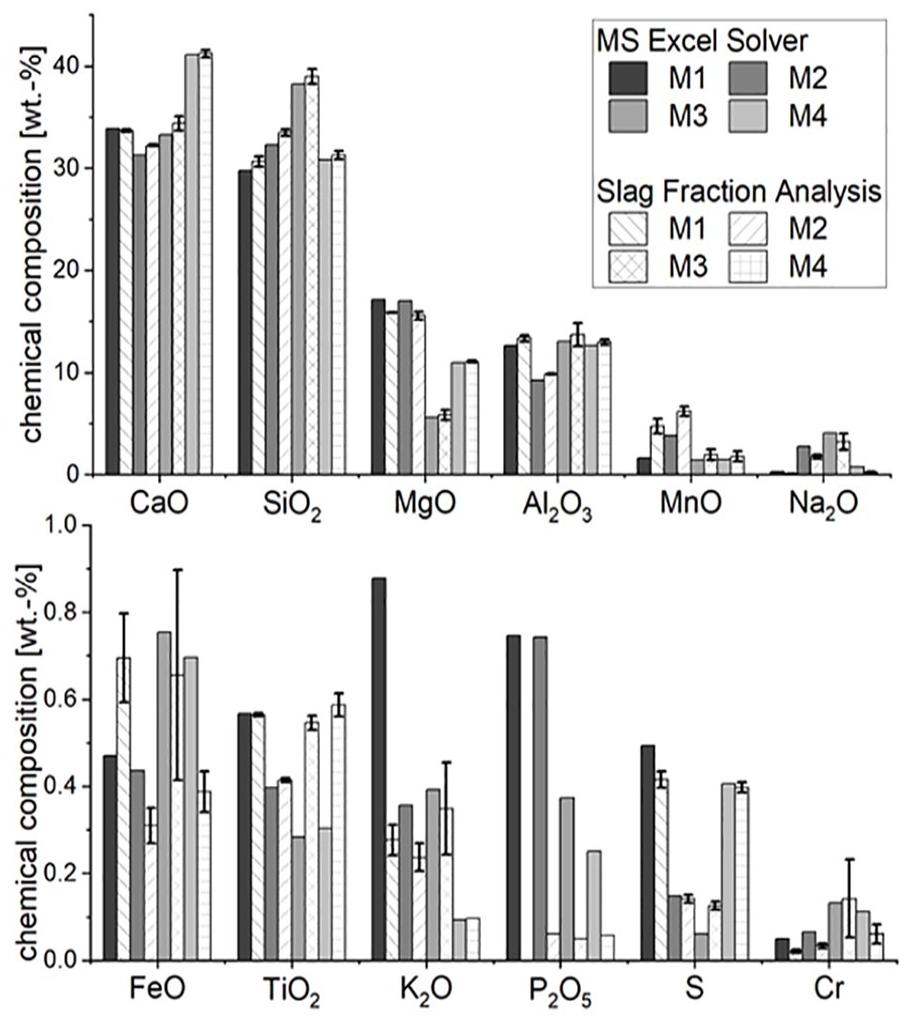
Deviation of the chemical composition of the calculated and the analysed slag fraction in wt%.
Firstly, the results are compelling since the match between the targeted product calculated by the MS Excel Solver and the analysis of the generated product is very high as far as the main input is concerned. It can be seen in Figure 3 that the minor deviations concerning the corresponding mass fraction of the oxide calculated by the MS Excel Solver are the main oxides CaO (−1.5%), SiO2 (−2.5%), MgO (2.9%) and Al2O3 (−4.9%). However, more significant differences can be detected for Na2O (53.7%) and K2O (27.4%). The functionality of the spreadsheet-based model can explain this, as no gaseous phase was taken into account. Therefore, every oxide or element that is not mathematically transferred into the metal fraction automatically ends up in the calculated slag fraction. Furthermore, the determined P2O5 does not match the value of the MS Excel Solver. The average deviation of 88,8% stems from the phenomenon that is not considered: phosphorus preferentially forms compounds with iron and is therefore found in the metal fraction. Furthermore, no significant differences could be found between the mixtures with the metallurgical residues from VASD (M1 and M2) and from SWM (M3 and M4), as there are outliers in all directions.
As mentioned above, at this stage, the MS Excel Solver cannot accurately predict the content of MnO, Na2O, K2O and P2O5 due to not considering the gaseous phase and the distinctive characteristics of phosphorus. Therefore, the spreadsheet-based model works best in respect to deviations if the content of these oxides is low.
Slag and metal fraction analysis
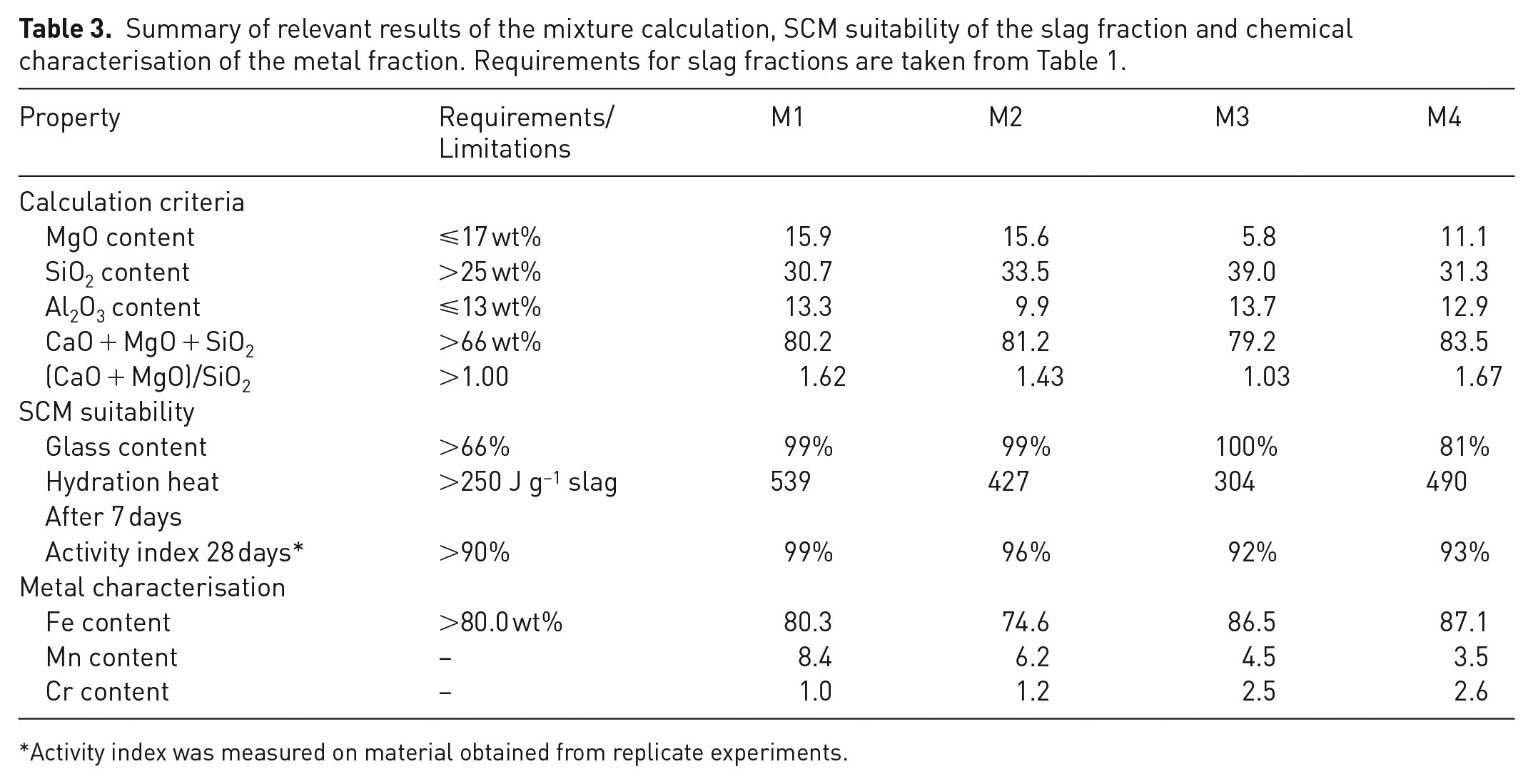
The boundary conditions for the mixture calculation consist of different oxide proportions as well as parameters such as the sum of CaO, MgO and SiO2 and the basicity. These limits were all met very well by the analysed slag fractions within an assumed accuracy of the XRF analysis of ±1 wt%.
The cumulative hydration heat after 7 days and the activity index after 28 days were identified as measures of the latent hydraulic properties, which are arguably the most important properties determining the usability of the slag fractions as SCMs. Both parameters are above the specified requirements for all mixtures (see Table 2); however M3 exhibits significantly lower hydration heat release than the other materials. The heat release as measured by the R3 protocol and the activity index could also be correlated to the chemical composition, as materials with a high basicity ratio [(CaO + MgO)/SiO2], such as M1 and M4, exhibited higher heat release than materials with an intermediate (M2) or low basicity (M3) (R2 of the linear correlation = 0.93). In direct comparison, M4 exhibits lower heat release and a smaller activity index than M1 although the basicity of M4 is higher. This circumstance is likely due to the presence of a significant portion of crystalline phases in M4, lowering the amorphous content – 81 wt% in contrast to 99 wt% – and therefore the amount of reactive, glass-like slag.
These findings confirm the strong relation between the hydraulic reactivity and the chemical and mineralogical composition of slags, which has been proposed for similar materials before (e.g. Blotevogel et al., 2020; Steindl et al., 2023). These relations could be used beneficially to estimate the suitability of carbothermally treated slags and other SCMs based on their chemical composition and amorphous content in the future. It will also provide more precise boundary conditions for mathematical models such as the one presented herein.
Judging from the chemical composition, high amorphous content and high reactivity, the slags are very similar to common GGBFS (Li et al., 2018; Steindl et al., 2023). However, to provide more information about the influence of certain components (e.g. Al2O3, K2O and Na2O), further carbothermal treatment experiments should obtain more experimental data. Additionally, further research is needed to provide information regarding the durability (e.g. shrinkage, resistance to chemical attacks such as sulphate attack) and long-term performance of building materials (mortars, concretes) made with binders containing carbothermally treated slags.
For the generated metal fraction, a high iron content of >80 wt% was desired. This requirement was met or exceeded by all mixtures except for M2, see Table 3.
Summary of relevant results of the mixture calculation, SCM suitability of the slag fraction and chemical characterisation of the metal fraction. Requirements for slag fractions are taken from Table 1.
Activity index was measured on material obtained from replicate experiments.
According to the results presented in Table 3, mixture M1 shows the best performance characteristics and can therefore be determined as optimal for the present work.
Transfer coefficients of Fe, Mn and Cr
As mentioned in the methods, transfer coefficients (TC) into the slag for Fe, Mn and Cr were estimated to calculate the mixtures. As depicted in the Sankey diagrams in Figure 4, the reduction of iron oxide was almost complete and consistent for all mixtures and tests (TCFe,slag = 1.5 ± 0.7%). It can also be seen that the transfer coefficient of Mn shows a more significant deviation (TCMn,slag = 49.9 ± 15.3%), whereas the difference within the two trials for M3, as well as M4, was most significant. The actual transfer coefficients into the slag for Cr were lower than expected (TCCr,slag = 7.2 ± 3.0%), meaning the transfer into the metal fraction worked well.
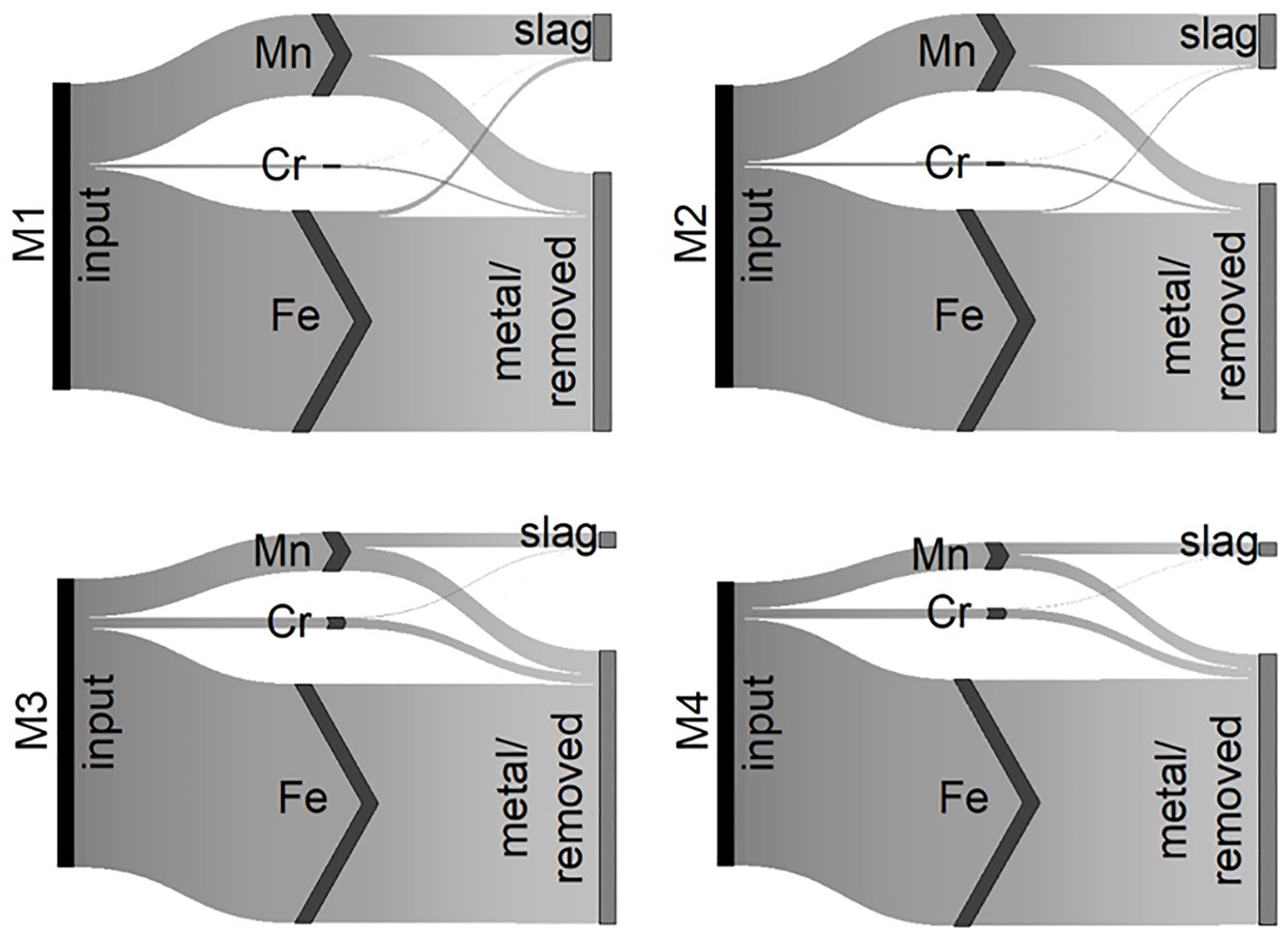
Schematic depiction of the transfer coefficients for Fe, Mn and Cr into the slag and metal/removed fraction.
Conclusion and outlook
A circular economy with high resource efficiency must be promoted to achieve ambitious climate targets, particularly in energy- and CO2-emission-intensive industries such as cement, iron and steel. Therefore, a carbothermal reduction of different metallurgical residues in combination with other industrial residues is proposed and its practical application is demonstrated in the outlined article. The presented treatment approach enables the upcycling of industrial wastes and by-products into secondary raw materials for the production of steel and building materials, while simultaneously helping to reduce CO2 emissions and landfilled volumes of metallurgical residues.
This work proves the general functionality of the developed solver-spreadsheet model to calculate the mix proportions of input materials for the carbothermal reduction treatment based on target compositions of the desired output materials (slag and metal fractions). The slag fractions, derived from various input material combinations such as steel slags, waste glass and fly ashes, were shown to be suitable as supplementary cementitious materials and similar in reactivity and composition to ‘traditional’ granulated BF slags. The iron-rich metal fraction can potentially be used as a secondary raw material for steel production in the BOF or electric arc furnace routes, supplementing or substituting pig iron and scrap steel normally used in the involved processes.
In the future, this spreadsheet model can also be a helpful tool to evaluate and characterise the composition of the released gaseous phases and the physical properties of the liquid and granulated slag. Specifically, targeting and characterising parameters such as the melting temperature and viscosity of the slag and liquid metal have particular relevance for the granulation process. Furthermore, the chemical and mineralogical composition of the slag fractions already could be characterised at the planning stage, enabling the tailoring of slags for specific use cases (e.g. high hydraulic reactivity).
However, influencing factors in the multi-component treatment system and various use cases of the produced slag and metal fractions have not been thoroughly researched. The follow-up project, ‘Recover-Met-Binder’, focuses on characteristic of the metal recovery, further evaluating the yielded slag fractions, and improving and characterising the involved treatment.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research project ‘UpcycSlag-Binder - US-B’ was funded by the Styrian Future Fund and the Climate Protection Fund of the City of Graz and carried out as part of the ‘Green Tech 100 - 1 Earth, 0 Carbon, 0 Waste’ programme. The ‘Recover-Met-Binder – RMB’ research project is funded by the Styrian Future Fund and carried out as part of the ‘Green Tech X’ programme. Additionally, we are grateful for the material supply and metal analysis by VASD and the material supply from SWM.
