Abstract
The recovery of lithium from spent lithium iron phosphate (LiFePO4) batteries is of great significance to prevent resource depletion and environmental pollution. In this study, through active ingredient separation, selective leaching and stepwise chemical precipitation develop a new method for the selective recovery of lithium from spent LiFePO4 batteries by using sodium persulphate (Na2S2O8) to oxidize LiFePO4 to FePO4. The impact of various variables on the efficiency of lithium leaching was investigated. Moreover, a combination of thermodynamic analysis and characterization techniques such as X-ray diffraction and X-ray photoelectron spectroscopy was employed to elucidate the leaching mechanism. It was found that 98.65% of lithium could be selectively leached in just 35 minutes at 60°C with only 0.2 times excess of Na2S2O8. This high leaching efficiency can be attributed to the stability and lack of structural damage during the oxidation leaching process. The proposed process is economically viable and environmentally friendly, thus showing great potential for the large-scale recycling of spent LiFePO4 batteries.
This is a visual representation of the abstract.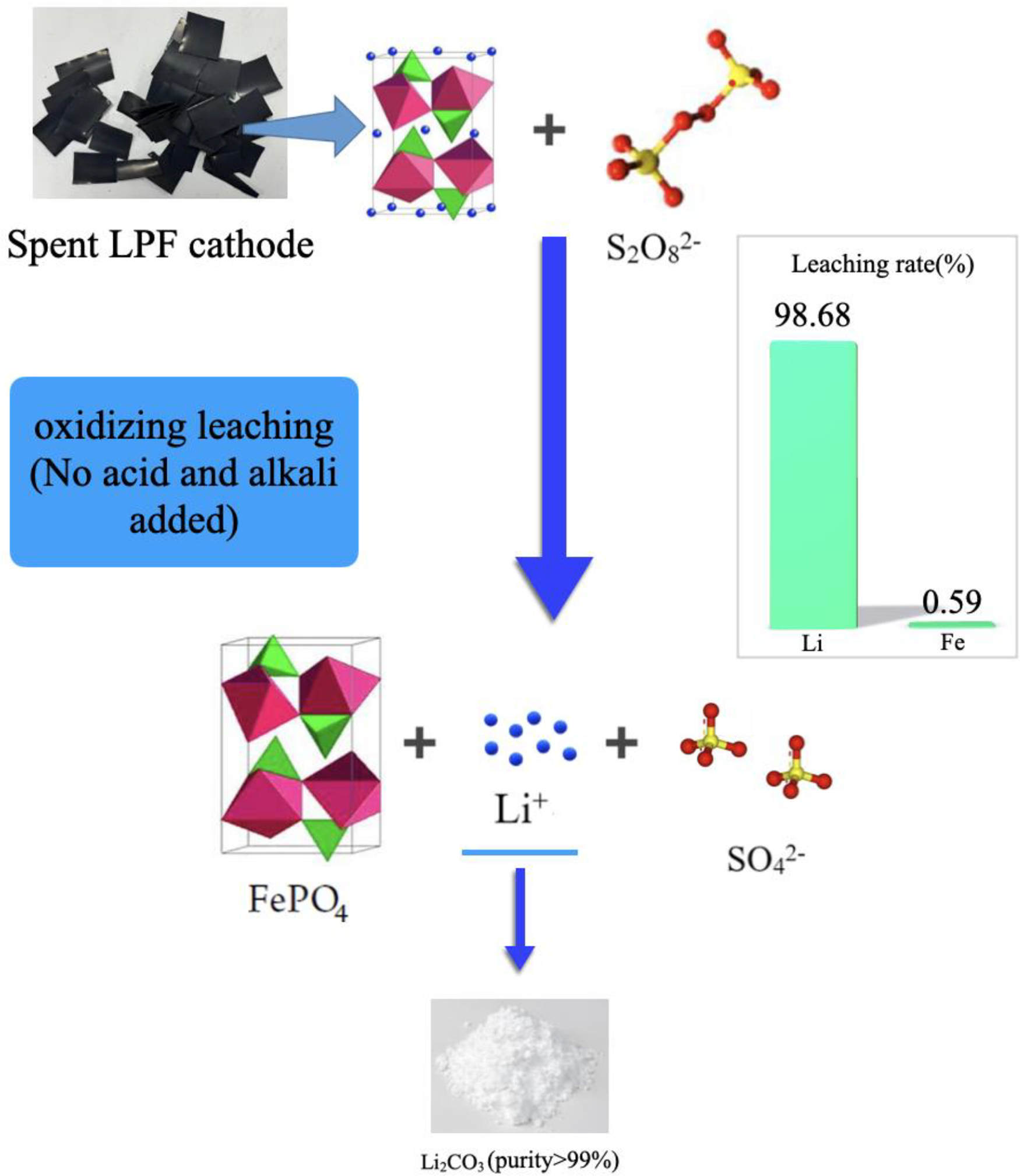
Introduction
Lithium-ion batteries (LIBs) are regarded as the most promising devices for both energy storage systems and electric vehicles (Liu et al., 2019a). Lithium iron phosphate (LiFePO4), being a typical representative cathode material, has been extensively applied in electric vehicles and energy storage stations due to its excellent structural stability, high safety and good economic viability (Liu et al., 2014). With the increasing demand for high performance and generation updates of LIBs, even though LiFePO4 can be used in a step-by-step manner, the green and efficient treatment of scrapped LIBs becomes an inevitable issue for resource and environmental sustainability (Fan et al., 2020). Additionally, the valuable element Li in LiFePO4 batteries presents significant commercial and economic potential for battery recycling, whereas the hazardous phosphorus element necessitates the environmental requirement for reuse and can easily transform into urban solid waste (Zhao et al., 2020). Formulating appropriate utilization and recycling strategies for LiFePO4 poses a major challenge for the thriving development of LIBs.
Traditional metallurgical extraction techniques, such as acid leaching (Yadav et al., 2020; Zhang et al., 2014), co-precipitation (Yang et al., 2017) and high-temperature treatment (Song et al., 2017), have been utilized as efficient value-added technologies for LiFePO4 battery recycling. However, the inherent properties of olivine-structured LiFePO4, characterized by strong Fe–O and P–O ionic bonds, severely impede structural degradation and element separation (Huang et al., 2001). Therefore, although the laboratory has conducted many studies on the process of acid leaching and resynthesis of LiFePO4 materials (Bian et al., 2016; Shin et al., 2015; Wang et al., 2018), this recycling process is still not feasible in practical applications. Unlike traditional acid leaching processes, some scholars used low concentration H2SO4 as the leaching agent and H2O2 as the oxidant, achieving good leaching rates for Li 96.9%, P 1.95% and Fe 0.027% (Li et al., 2017).
Both high and low concentrations of acids seem to be inevitably used to disrupt the structure of LiFePO4. We have noticed that some researchers have replaced organic electrolytes in LiFePO4 batteries with water-based electrolytes such as Li2SO4, LiNO3 or LiCl to isolate problems caused by the reaction between organic electrolytes and electrodes (Li et al., 1994; Tron et al., 2017). During this charging process, LiFePO4 in the cathode is oxidized to FePO4 by applying a positive potential. Then, lithium ions are de intercalated from the cathode into the liquid electrolyte, which is stable during electrochemical cycling (Hou et al., 2013). This achievement provides us with ideas that the used LiFePO4 cathode may be oxidized into FePO4 by chemical oxidants, and then the most valuable element lithium can be released into aqueous solution, which can be easily separated from insoluble residues.
In this article, we use sodium persulphate (Na2S2O8) as an oxidant to study the detailed leaching strategy of LiFePO4 materials. The effects of reaction time, temperature, solid–liquid ratio and the mass ratio of Na2S2O8 to LiFePO4 materials on the leaching efficiency of lithium-ions are discussed. Combined with thermodynamic equilibrium, the thermodynamic possibility and efficiency of the whole process are discussed. Then, the differences between the conventional acid leaching method and this new method are compared and discussed, and the advantages are demonstrated through the aspects of environmental and economic benefits.
Materials and methods
Materials and chemicals
The chemical reagents used in this work include Na2S2O8, sodium hydroxide and sodium carbonate (purchased from Sinopharm Chemical Reagent Co., Ltd, Shanghai, People’s Republic of China) and were of analytical grade. The spent LiFePO4 batteries were provided by ZCycle Co., Ltd, Shanghai, China. Moreover, its main components are (wt.%) 4.22% Li, 22.38% Fe and 19.39% P.
Leached experiment
Firstly, the spent batteries were discharged completely, disassembled and the cylindrical electrode material removed. The next two significant steps are:
(1) Separation of LiFePO4 from spent batteries
The LiFePO4 electrode material was cut into 3 × 3 cm sheets, placed in 70 hot water, and gently stirred for 15 minutes. The LiFePO4 material falling off the aluminium foil were separated, dried and powdered after washing with deionized water.
(2) Selective leaching of cathode materials
The control variable method was used to study the influence of various critical factors on the experimental results. The leaching experiment was carried out in a 250 mL beaker, which was placed in a magnetic stirring constant temperature water bath at 60°C. Mixed the cathode material powder with Na2S2O8 powder according to the mass ratio of 1.2:1, The solution was stirred at a speed of 300 r minute−1. After 35 minutes, the suspension was separated by suction filtration.
According to the reaction equation between Na2S2O8 and LiFePO4, the reaction produced sodium sulphate and iron phosphate precipitation. The reaction equation is as follows:
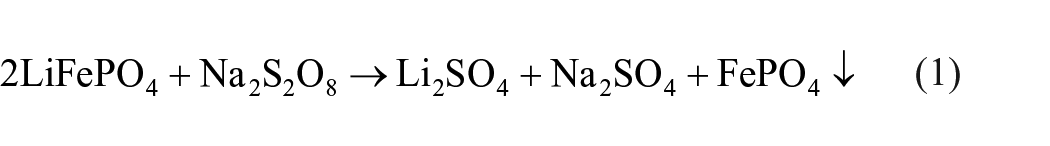
Amongst them, the function of Na2S2O8 is to oxidize divalent iron into trivalent iron to convert LiFePO4 into iron phosphate precipitation (Mahandra and Ghahreman, 2021). In the process, lithium is discharged into the solution and leached, which is similar to the charging and discharging process of the battery and will not destroy its olivine crystal structure. The whole leaching reaction realizes the separation of iron and lithium (Shentu et al., 2021), and the selective leaching of lithium was realized.
An inductively coupled plasma atomic emission spectrometer (ICP-OES SPECTRO ARCOS II) was used to identify and determine the concentration of metal elements in the filtrate. At the same time, to avoid the experiment’s randomness and ensure the experiment’s reproducibility, repeated experiments under the same conditions were carried out twice for all variable influence experiments.
After washing the filter residue with deionized water several times, placed it in a drying oven and dry it at 80°C for 12 hours and calcined it at 650°C for 5 hours in a tubular furnace to remove carbon. Li+ in the filtrate was recovered in the form of lithium carbonate precipitation by adding sodium carbonate. Iron and other metal impurities were removed by adjusting the pH of the lithium-containing leaching solution, then concentrating the solution by heating it. According to the inverse ratio between the solubility of lithium carbonate in an aqueous solution and temperature (Liang et al., 2004), sodium carbonate powder (Zhao et al., 2019) is directly added at 90°C to precipitate lithium carbonate. After filtration and hot water cleaning, Li2CO3 products were recovered.
Analytical method
The concentrations of various metallic elements in the leachate are determined by an inductively coupled plasma atomic emission spectrometer for identification and determination. The leaching rate of various elements
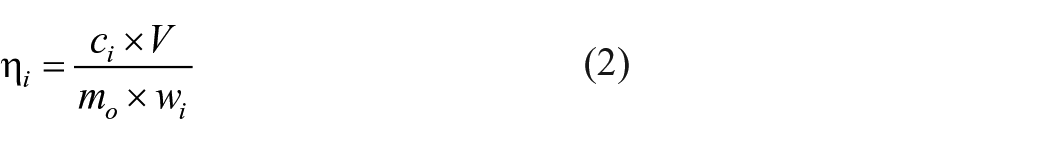
where
Results and discussion
Effect of Na2S2O8 dosage on lithium leaching rate
Only 81.42% of Li can be leached when the mass ratio is 0.8 (Figure 1(a)). Then the leaching rate of Li increased gradually and reached 97.89% with the increasing dosage of Na2S2O8. When the mass ratio reached 1.2, with a further increase in Na2S2O8 dosage, no change of Li leaching is observed, but the leaching of Fe exhibits a slight increase. The rationale behind this phenomenon lies in the fact that a minor excess of Na2S2O8 will oxidize H2O and result in solution acidification, as illustrated by the following chemical equation:
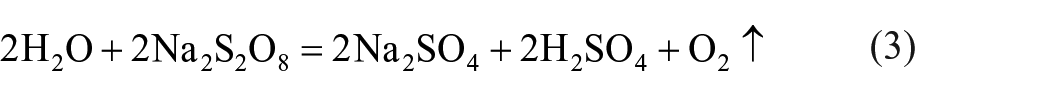
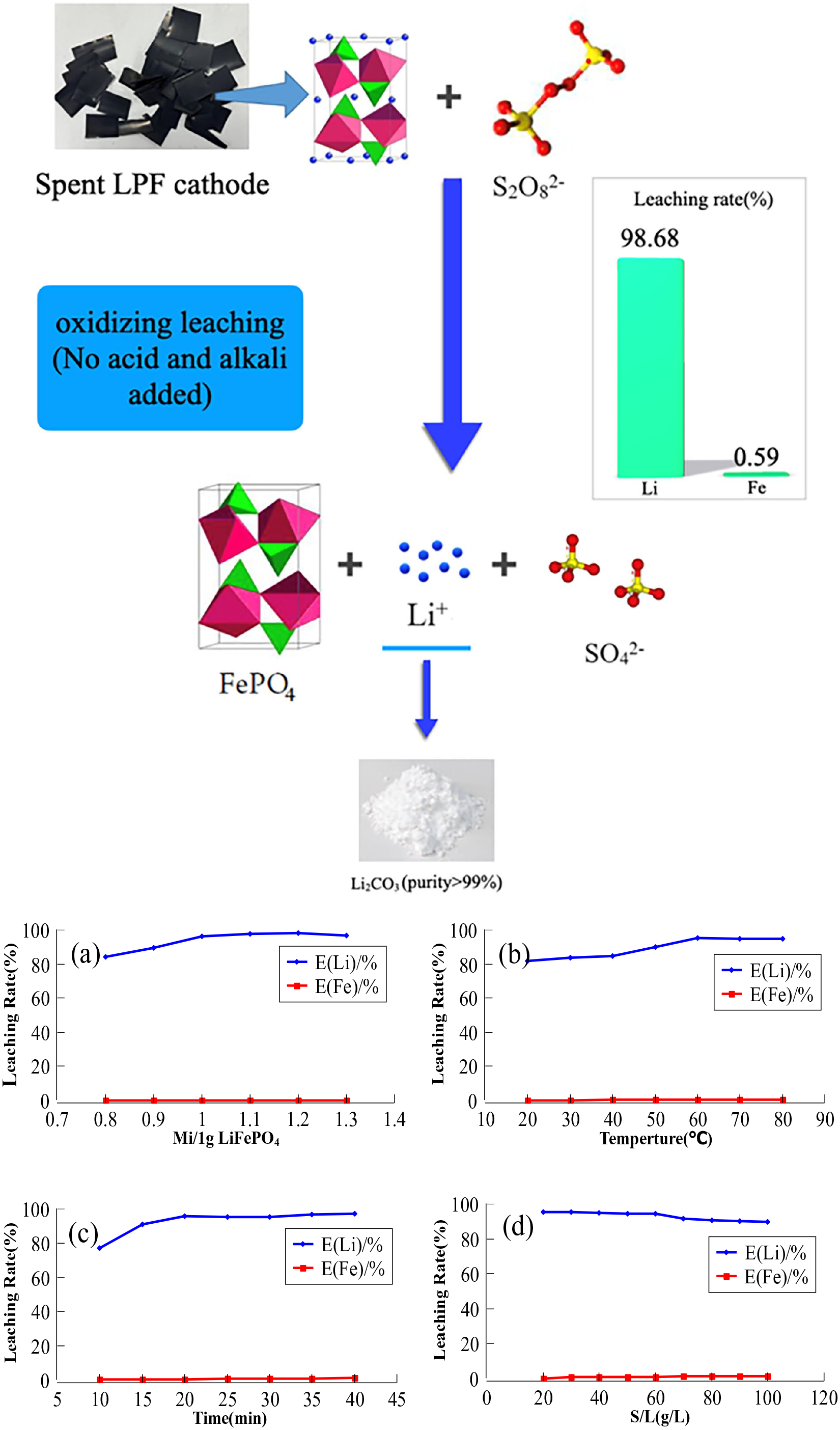
(a) Effect of sodium persulphate content on lithium leaching rate. (b) Effect of different temperatures on lithium leaching rate. (c) Effect of different time on lithium leaching rate. (d) Effects of different solid–liquid ratios on lithium leaching rate.
So, little soluble Fe ions will be generated in this weak acid solution. Combined with the analysis of experimental phenomena and results, the mass ratio should be controlled at 1.2.
Effect of different temperatures on lithium leaching rate
The leaching rate of lithium-ion is only 81.58% after reaction at 20°C for 35 minutes (Figure 1(b)). The lithium-ion leaching rate is increasing with the continuous temperature increase. When the temperature raised to 60°C, the leaching rate reached the maximum value of 95.26%, the leaching rate maintained at this level in the subsequent heating up. The leaching rate of iron ion is controlled by 1% at all temperatures. The metal leaching process is endothermic, so increasing the reaction temperature creates more favourable thermodynamic conditions for the leaching process (Behera and Parhi, 2016). Therefore, with the increase in temperature, the balance of chemical reaction is promoted, the speed of chemical reaction is accelerated and the leaching rate of lithium-ion is improved. It can be seen from the curve analysis that the lithium-ion leaching rate has not significantly improved after 60°C. Therefore, considering the energy consumption caused by high temperature, the reaction temperature should be controlled at 60°C.
Effect of different time on lithium leaching rate
It can be seen that after reaction at 60°C for 10 minutes, the leaching rate of lithium-ion is only 76.87% (Figure 1(c)). With the increase in reaction time, it reached 95.23% at 30 minutes and 96.47% at 35 minutes. After that, there was no significant increase or fluctuation. The leaching rate at 10 minutes was much lower than that at 35 minutes, which may be explained by insufficient mixing of cathode material and oxidant for a short period of time. After 35 minutes, there seems to be no significant improvement in leaching rate. According to this experimental phenomenon, we can conclude that a high lithium-ion leaching rate (96.47%) can be guaranteed without prolonging the reaction time after 35 minutes. Considering the time cost and ensuring a high lithium-ion leaching rate, the reaction time should be set to 35 minutes.
Effects of different solid–liquid ratios on lithium leaching rate
When the solid–liquid ratio is 20 g L−1, the maximum lithium-ion leaching rate is 95.47% (Figure 1(d)). The rate of lithium-ion leaching decreased gradually as the solid–liquid ratio increased. Before the solid–liquid ratio was 60 g L−1, it maintained a relatively high lithium-ion leaching efficiency (more than 92%), but with the further increase in the solid–liquid ratio, the lithium-ion leaching efficiency decreased significantly. A smaller solid–liquid ratio allows for more complete contact between the positive electrode material powder and Na2S2O8 solution(Jha et al., 2013), it was more conducive to the mass transfer process between solid and liquid, so the leaching rate was higher (Li et al., 2015). Combined with the analysis of experimental phenomena and results, the solid–liquid ratio should be controlled within 60 g L−1.
Kinetics analysis
Leaching lithium from spent LiFePO4 cathode is a solid-liquid heterogeneous reaction, which occurs on the outer surface of unreacted LiFePO4 particles. The coexistence of LiFePO4 and FePO4 phases in chemical lithium removal has been reported (Chen et al., 2006; Ramana et al., 2009). In the oxidation leaching process, the volume involved in the phase transition decreases (6.81%) when the lithium-ion gets rid of the crystal structure of LiFePO4 (Allen et al., 2007). According to the principle of hydrometallurgy when the FePO4 layer is formed in the reaction process, the product formation and phase volume reduction make the leaching process will conform to the shrinking core model (Li et al., 2018; Meng et al., 2018). The shrinking core model was used to analyse the leaching kinetics of lithium at different temperatures in order to study the apparent mechanism of lithium leaching by Na2S2O8. Assuming that the particles of the cathode scrap have a spherical geometry and the process is controlled by internal diffusion through the product layer, the integrated equation of the model can be expressed as follow:

The integrated Equation (5) of the shrinking core model can be expressed as follow when the process is controlled by chemical reactions at the interface:

where
To investigate the kinetics of the present work, the data were fitted to both equations. The plots of
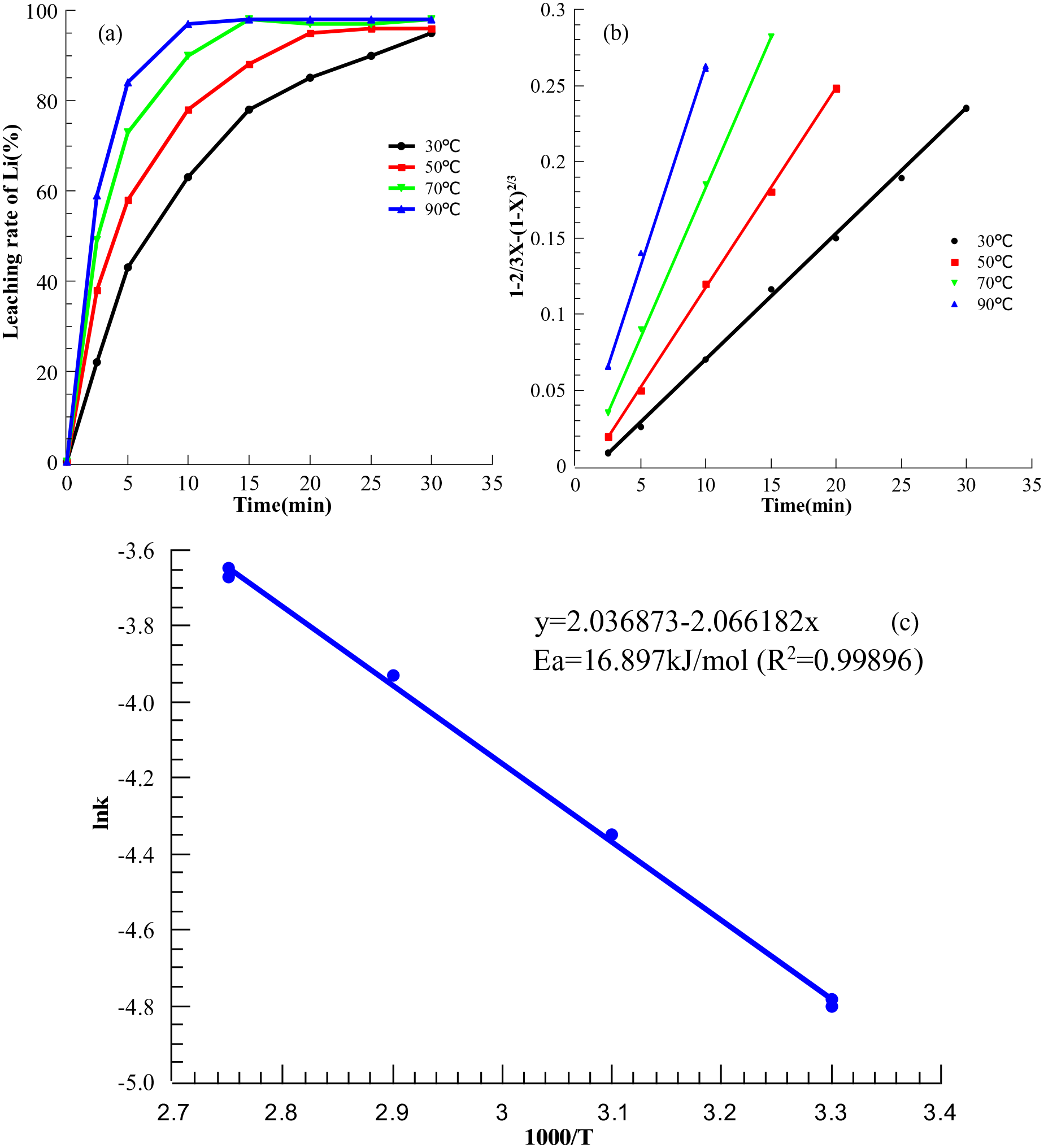
(a) The leaching rate of Li at different temperature and time. (b) Data fitting diagram of internal diffusion model at different temperature. (c) lnk-1000/T diagram of internal diffusion.
Arrhenius equation is used to describe the relationship between reaction rate constant and reaction temperature (Wang et al., 2019):


where
The activation energy of leaching reactions is usually calculated by the linear form of the Arrhenius equation, according to the plots of ln
Thermodynamic analysis
Figure 3(a) is the E-pH diagram of Fe–P–Li–H2O thermodynamic equilibrium at 333.15 K, and the specific calculation method is given (Shentu et al., 2021). The pH potential diagram at 298.15 K was discussed (Zhang et al., 2019). However, the leaching temperature of this work is 333.15 K. As can be seen from Figure 3(a), both iron phosphate and LiFePO4 have their stable regions in an aqueous solution, and there are two paths to realize the conversion from LiFePO4 to iron phosphate (Fan et al., 2018). Initially, the first path with a blue arrow in Figure 2(a). It is divided into two main processes: LiFePO4 is totally dissolved into Li+, Fe2+ and PO43+ and then Fe2+ is oxidized into Fe3+ in the solution through the oxidation of oxidant so that Fe3+ precipitates in the form of FePO4. This path is the conventional acid leaching method discussed earlier, which will consume much acid solution, produce a lot of acid spent water and increase the cost of subsequent treatment. The second path is marked with a yellow arrow. It shows direct conversion method from LiFePO4 to iron phosphate. It is evident that this method will save a lot of costs (Verma et al., 2019). When the pH is in the range of 2–6, the oxidation potential of Na2S2O8 can meet the requirements of the range of LiFePO4. Therefore, the conversion from LiFePO4 to iron phosphate can be realized without introducing impurities and acid-based solution to realize the selective leaching of lithium-ions.
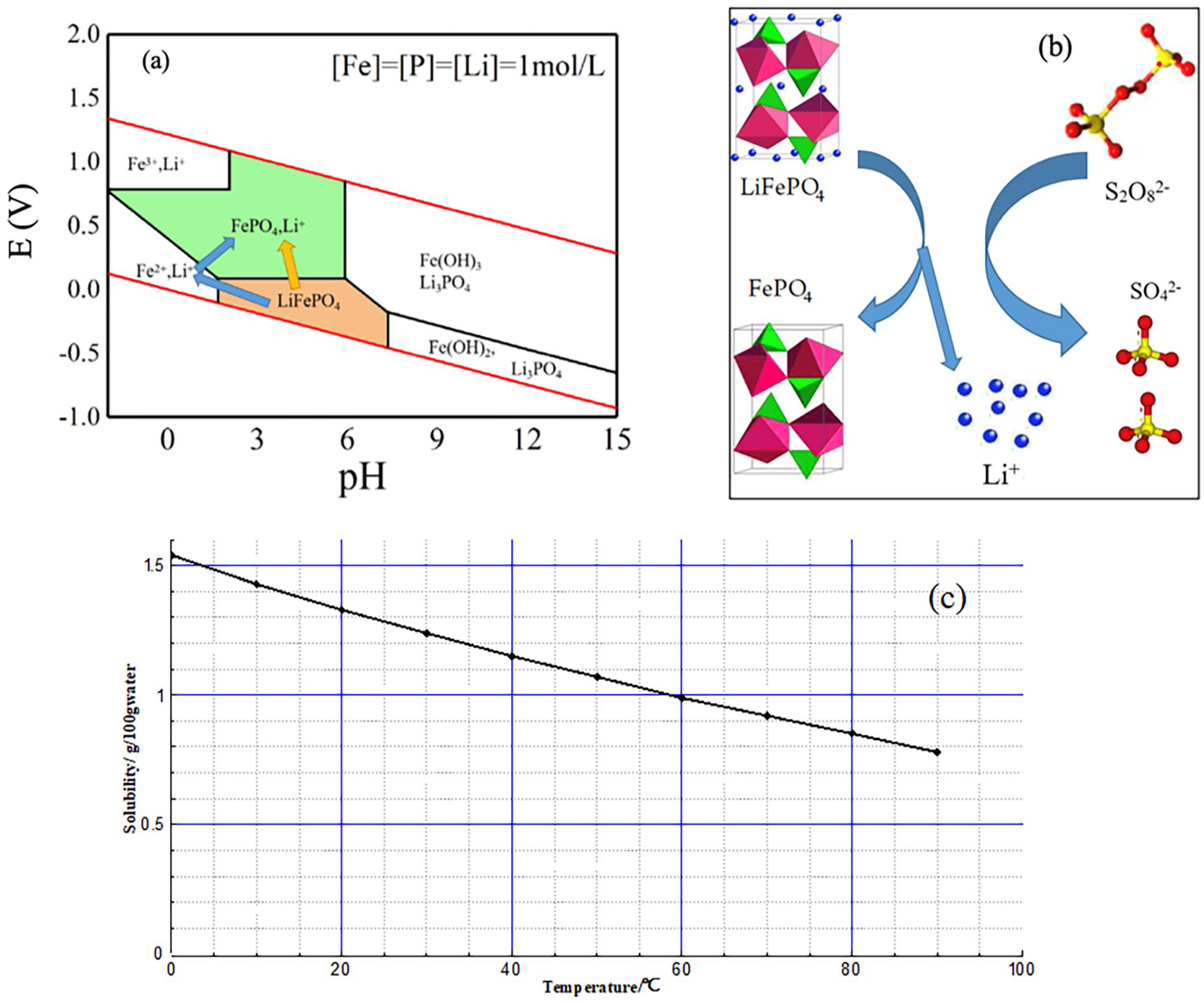
(a) E-pH diagram of thermodynamic equilibrium of Fe–P–Li–H2O at 333.15 K. (b) Schematic diagram illustrating the leaching mechanism. (c) Solubility curve of lithium carbonate.
Figure 3(b) is a schematic diagram of the leaching mechanism of the reaction between persulphate and LiFePO4. There is a redox reaction between LiFePO4 and S2O82−, and then FePO4 with the same olivine crystal structure is generated. At the same time, lithium-ions are released from the crystal into the aqueous solution. We can see that the key to this process is that the crystal structure has not been damaged and changed before and after lithium-ion leaching, which is similar to the phase transition in the charging process of LiFePO4 battery (Zhang et al., 2019).
Economic analysis
Taking 1 kg of spent LiFePO4 batteries as an example to compare the economic consumption of each process between conventional acid leaching method and our Na2S2O8 oxidation leaching method on a laboratory scale. The specific details are as follows:
The pretreatment process of 1 kg spent LiFePO4 battery mainly includes discharge and disassembly, and 367 g cathode electrode materials can be obtained (battery components are provided by China ZCycle Co., Ltd, Shanghai, China). In this process, the input cost of raw materials (1 kg pent LiFePO4) is 1$ (Liu et al., 2019b). In addition, there is no difference between the conventional acid leaching method and this method in the process.
In the separation step of the positive active material and aluminium foil, the conventional acid leaching method mainly uses NaOH to dissolve the collector aluminium. Theoretically, 84 g NaOH is required, so the cost is 0.234$ (the price of NaOH is 2.789$ kg−1, which is from the website https://www.reagent.com.cn/). In addition, 6 L water is required to dissolve NaOH, and 2 L water is required for cleaning. The water cost in Shanghai, China is 0.766$ m−3 (including 0.481$ m−3 for water supply and 0.285$ m−3 for sewage treatment), so the investment in water consumption is 0.0061$. So, the total cost of the separation step is 0.24$. In this method, the active material can be separated from the aluminium foil by cutting the material into 3 × 3 cm thin slices, placing it in 1 L hot water at 70°C, and gently stirring for 15 minutes. Therefore, the water charge is 0.00076$, and the electricity charge is 0.02$ (the industrial electricity charge in Shanghai, China, is 0.136$ kW−1 hour−1, and the power of thermostatic water bath is 0.6 kW). Therefore, the total cost of this method in the separation step is 0.02076$.
Some researchers (Peng et al., 2021) took acetic acid as an example to analyse and calculate the various costs of the conventional acid leaching method in the leaching stage, which is 0.2$. In this method, 440 g NaS2O8 is required in the leaching stage, and the input is 0.186$ (the price of NaS2O8 is 0.425$ kg−1, which is provided by the website https://mall.chemsrc.com/search?s1=true&s=7775-27-1).
In the process of precipitation of Li2CO3, the conventional acid leaching method has a low pH value due to the addition of high concentration of acid. The metal impurities contained in the material and the FePO4 generated usually dissolve into the lithium rich leaching solution, which requires an additional pH adjustment process to remove impurities. Therefore, it is necessary to neutralize the excessive acid and alkali in the mother liquor, which leads to the consumption of additional alkali liquor. This step is unnecessary in this method because Na2S2O8 is added according to the stoichiometric ratio.
In conclusion, compared with the conventional acid leaching method, this method has lower economic consumption in each process, so it is economically feasible.
Recovery of lithium from leaching solution
After discussing the leaching rates of lithium and iron by the above four variables, we obtained the lithium-containing leaching solution through the best experimental conditions. The metal impurities such as iron were removed by adjusting the pH of the solution, and then the solution was concentrated. Sodium carbonate powder was added at 90°C to precipitate lithium carbonate (Zhao et al., 2019). The solubility of lithium carbonate in an aqueous solution is inversely proportional to temperature (Liang et al., 2004), as shown in Figure 3(c). After filtration and washing with hot water, Li2CO3 products can be effectively recovered.
Process flow introduction
Based on the above research, in order to recover valuable metals from spent batteries more efficiently and orderly, a selective leaching chemical precipitation route was proposed (Figure 4). The optimized recovery route is summarized as: when the mass ratio of Na2S2O8 to LiFePO4 powder is 1.2, the water bath heating temperature is controlled at 60°C, the stirring speed is 300 r minute−1, the system solid–liquid ratio is within 60 g L−1, the reaction time is 35 minutes, and the lithium ion leaching rate can reach 98.68%. Due to the use of Na2S2O8, the impurities in lithium containing solution are lower than those in methods.
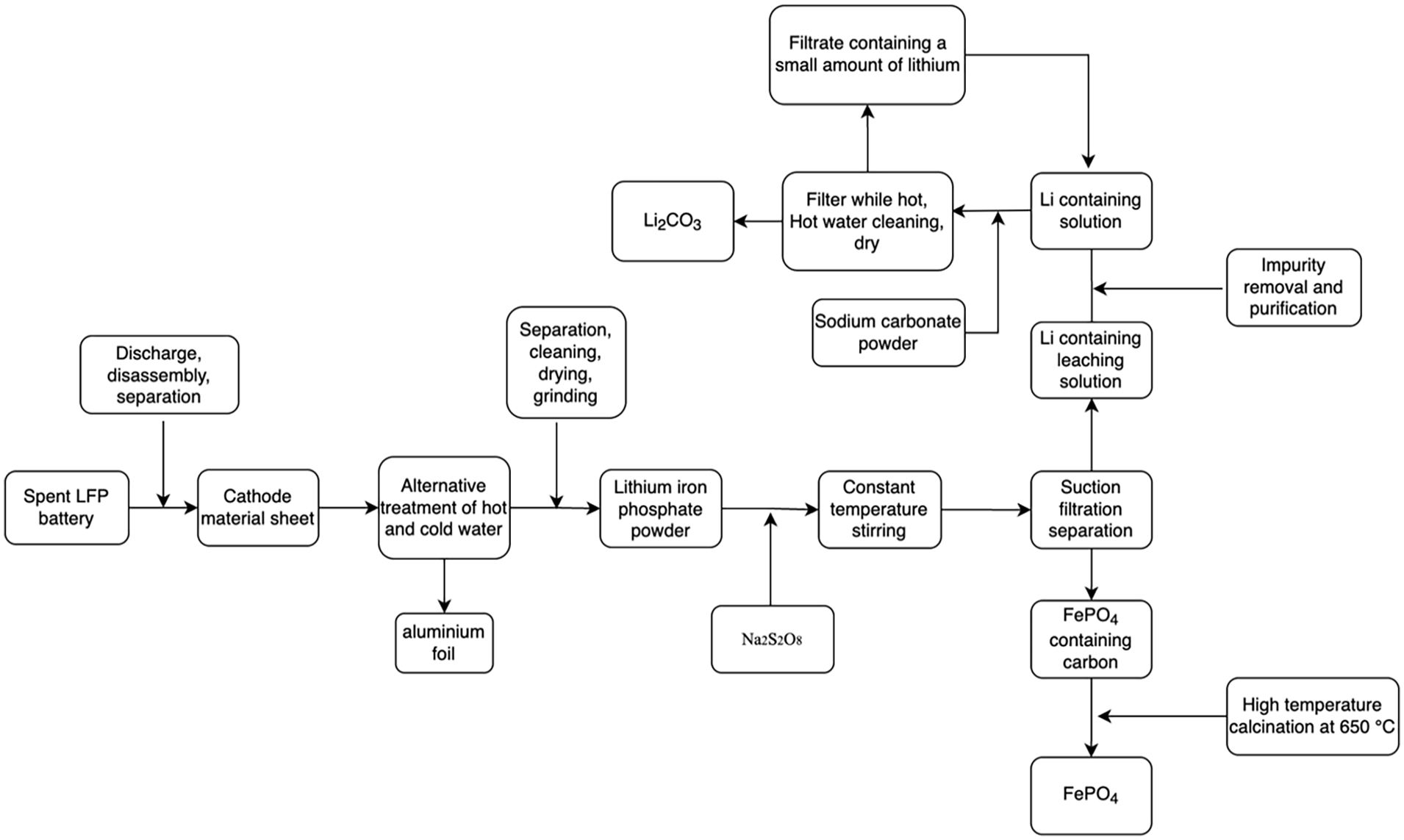
Flowchart for leaching and recovery process.
The leachate obtained from the leaching reaction contains high concentration of Li (11.9 g L−1), and the impurity concentration is very low. Therefore, lithium solution can be further concentrated and purified. The high-purity Li2CO3 product is prepared by directly adding Li2CO3 and evaporated. The mother liquor obtained after evaporation can be used for freezing crystallization to prepare Na2SO4 ·10H2O product. Then, the remaining crystallization solution can be used to leach another batch of cathode materials after adding Na2S2O8. For the leaching residues, FePO4 powder exists in the form of precipitation after leaching and can be obtained after filtering and washing. After necessary treatment, the FePO4 powder obtained can be used together with Li2CO3 obtained by this method to prepare regenerated LiFePO4 cathode materials. This process means that the entire route can be recycled without introducing impurities, and the cathode material can be regenerated.
Different from conventional acid leaching methods, there are problems such as long acid leaching time, higher reaction conditions and large amount of acid wastewater. Based on the principle of lithium-ion principle and embedding during battery charging and discharging, this method oxidizes Fe (II) to Fe (III) by oxidation reaction and precipitates in the form of FePO4. Li+ is discharged from the lattice into the liquid phase due to charge repulsion effect, which effectively solves the above problem and guarantees a relatively high leaching rate.
In addition, this method is also environmentally friendly. The core reaction of the whole process is:
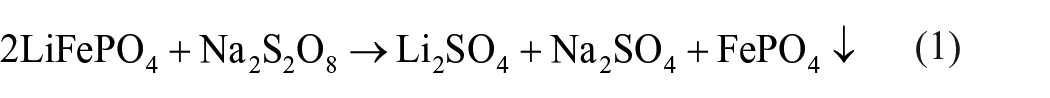
All products have the attribution of utilization. Even a little excessive NaS2O8 will hydrolyse in water. The reaction equation is as follows:
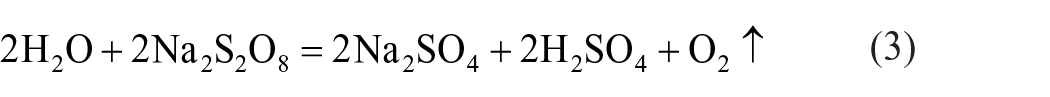
The leachate can be recycled after Li2CO3 is precipitated. Therefore, this method is environmentally friendly and harmless.
Conclusion
A new method for selective recovery of lithium from cathode spent of spent LiFePO4 batteries was proposed. LiFePO4 is oxidized to FePO4 with Na2S2O8, forcing lithium to be removed from the cathode. Under the optimal conditions of 1.2 times the theoretical dosage of Na2S2O8, the solid–liquid ratio is 60 g L−1, the reaction temperature is 60°C and the reaction time is 35 minutes, more than 98% lithium is leached without acid and alkali. The feasibility of this method is also verified by thermodynamic and kinetic analysis, as well as analysis of the E–pH diagram of thermodynamic equilibrium of Fe–P–Li–H2O at 333.15 K, Only lithium ions are separated from the raw material structure and enter the liquid phase during the oxidation leaching process, whereas the original olivine crystal structure of the raw material is not damaged, it is precipitated in the form of FePO4 and used to recover Fe, thus realizing the one-step separation of Li and Fe, as well as the selective recovery of Li. This is difficult to do with conventional acid leaching method. Based on the above, a selective leaching process was proposed to recover the whole spent LiFePO4 battery. Lithium can be effectively leached with shallow impurities. Li2CO3 products can be directly prepared by adding Na2CO3 to the leaching solution. This study provides a simple and environmentally friendly technology for the selective recovery of valuable metals from spent lithium phosphate batteries.
Footnotes
Acknowledgements
The authors thank TES-AMM Corporation (China) Limited for providing us with raw materials for spent LiFePO4 batteries as well as analysis and testing equipment.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
