Abstract
Liquid fertilizers (LFs) produced by microwave-assisted acid hydrolysis of livestock and poultry wastes were applied to potted hot pepper (Capsicum annuum L.) to evaluate their potential to be used as amino acid LFs. A preliminary experiment was conducted to determine the optimum acid-hydrolysis conditions for producing LFs from a mixture of pig hair and faeces (P) and another mixture of chicken feathers and faeces (C). Two LFs were produced under the optimum acid-hydrolysis conditions (acidification by sulphuric acid (7.5 mol L−1) in a microwave (200 W) for 90 minutes), and a commercial amino acid LF (Guo Guang (GG)) was used for comparison. P, C and GG fertilizers were tested in potted hot pepper cultivation at two doses, whereas no fertilizer application served as the control (CK). P and C fertilizers significantly increased the fruit yield compared with GG fertilizer, particularly at the higher dose. Moreover, the treatments improved the fruit vitamin C and soluble sugar contents in the order of C > P > GG compared with CK. These results could be attributed to more types of amino acids in C fertilizer than in P and GG fertilizers. The results also indicated that the prepared fertilizers could significantly increase the shoot and root dry weight, soil available nitrogen and phosphorus contents and nitrogen, phosphorus, and potassium (NPK) uptake by plants compared with CK. In conclusion, microwave-assisted acid hydrolysis could effectively convert unusable wastes into valuable fertilizers comparable or even superior to commercial fertilizers.
Introduction
Livestock production has been continuously growing to meet the increasing demand for meat worldwide (Ilea, 2009), particularly in developing countries such as China (Liu et al., 2019). It has been estimated that the production will double by 2050 (Ilea, 2009). From 2007 to 2017, pork production in China increased by 26.6%, which is equivalent to 55 million tons (Zhao et al., 2022). Moreover, China is one of the largest consumers of chicken meat in 2020, and the consumption amount rose to 15.7 million tonnes in 2021 (Osadchuk et al., 2020). To date, China has become the top pork and chicken egg producer in the world (Yang, 2021; Zhang et al., 2021). This huge industry inevitably generates large quantities of leftovers such as livestock faeces, which can contaminate the soil ecosystem, surface and underground water, spread pathogens and negatively affect all living organisms around if not properly disposed or used (Kumar et al., 2013). In addition, as part of livestock and poultry wastes, pig hair and chicken feathers can also pose serious threats to the environment (Prasanthi et al., 2016; Zhu and Hiltunen, 2016).
As a matter of fact, chicken feathers and pig hair are valuable sources of amino acids due to their high contents of keratin (Branska et al., 2020; Esteban et al., 2010), which can be hydrolysed to reduce their hazards (Chen et al., 2015), and the obtained product can stimulate plant growth and productivity (Casadesús et al., 2020). Moreover, chicken and pig faeces are important sources of nutrients that may partially substitute synthetic chemical fertilizers if properly recycled (Van der Meer et al., 2008). Thereby, the hydrolysis of livestock and poultry wastes is an effective way to reduce their hazards and maximize their economic benefits (Chen et al., 2015).
Decomposition of protein into amino acids via enzymatic hydrolysis is a slow and costly process (Chen et al., 2015; Thangavelu et al., 2019); in contrast, chemical hydrolysis such as acid hydrolysis is a more rapid, simple and effective method (Thangavelu et al., 2019). The main function of acid hydrolysis process is to break the chemical bonds, which bind functional groups together such as glycosidic, amide and lactone bonds (Chen et al., 2019), and then the acidic products such as protein hydrolysates can be neutralized to generate liquids with abundant amino acids and inorganic salts (Sandeaux et al., 1998). In this aspect, microwave technology has been demonstrated as an efficient thermal treatment method of biomass, which can shorten chemical reactions time by up to 80% (Kostas et al., 2017). Moreover, microwave-assisted treatment with certain chemicals such as acids can greatly reduce the energy consumption because microwave radiation can penetrate the organic compounds, resulting in the release of them as soluble substances (Zhang et al., 2018).
As nitrogen (N) is an essential nutrient for crop production, agrochemical N fertilizers are widely used in agriculture production (Sainju et al., 2019). However, excessive application of chemical N fertilizers not only leads to huge economic loss due to N loss through volatilization, nitrification, denitrification and N leaching (Zhang et al., 2022b) but also causes serious environmental threats such as water eutrophication (Soleimani et al., 2022) and nitrous oxide emissions (Aryal et al., 2022), which will aggravate the trend of global warming. In contrast, organic N, particularly amino acid N, can be mineralized by soil microorganisms to release available N for plants, thereby ensuring continuous N supply for plants and promoting N retention in soils (Lu et al., 2018). Amino acids can serve as fertilizers to boost plant productivity and enhance nutrient uptake by plants (Radkowski et al., 2018), which is obviously superior to the excessive use of synthetic chemical fertilizers. Besides, some reports have highlighted the capability of the grown plants to directly absorb intact amino acids (Teixeira et al., 2018).
As previously reported, acid hydrolysis of animal wastes can produce efficient and valuable fertilizers. However, there has been limited research on the conversion of slaughterhouse wastes into valuable products (Kang et al., 2019), particularly by acid hydrolysis. Additionally, to the best of our knowledge, no research has been conducted on the production of fertilizers from the combination of faeces and feathers or hair by hydrolysis. Therefore, the main objectives of this study were: (a) to screen the optimum acid-hydrolysis conditions (acid concentration, microwave power and radiation period) for the production of liquid fertilizers (LFs) from a mixture of pig hair and faeces and another mixture of chicken feathers and faeces by microwave-assisted acid hydrolysis and (b) to evaluate the effect of these produced fertilizers on plant growth and productivity. To this end, hot pepper was selected as a test plant considering its importance as a vegetable crop worldwide (Rajametov et al., 2021), and a commercial amino acid LF Guo Guang (GG) was selected as a reference. We hypothesized that the prepared LFs could improve the growth performance of hot pepper plants, increase the uptake of nutrient cations and to a less extent nutrient anions by plants and be comparable or even superior to commercial fertilizers in enhancing plant growth and productivity. The results may provide important insights into the improvement of plant growth and productivity while sustaining the environment through appropriate treatment of unusable organic wastes.
Materials and methods
Sample collection and preparation
Four different livestock and poultry wastes (pig hair, chicken feathers, pig faeces and chicken faeces) were used as raw materials in this study. Pig hair and chicken feathers were collected from nearby slaughterhouses, and then washed under running tap water and oven-dried at 70°C for 24 hours. Thereafter, these wastes were crushed and packed in sealed bags. In terms of pig faeces and chicken faeces, the samples were collected from nearby farms and then air-dried for 72 hours, crushed and packed in sealed bags.
Production of the LFs by microwave-assisted acid hydrolysis
Pig hair and faeces were mixed at a mass rate of 1:1 to produce a mixture sample (P), whereas chicken feathers and faeces were also combined at a mass rate of 1:1 as another mixture sample (C). Two types of LFs were produced from the two mixtures by microwave-assisted acid hydrolysis after acidification with sulphuric acid (H2SO4) at the mass/volume ratio of 1:10.
Two preliminary experiments were conducted using the orthogonal design to determine the optimum acid-hydrolysis conditions for producing LFs based on the nitrogen (N)-amino acid yield of the hydrolysate. In these experiments, three concentrations of H2SO4 (A) (5, 7.5 and 10 mol L−1 (H+)), three microwave powers (B) (100, 200 and 400 W) and three radiation periods (C) (30, 60 and 90 minutes) were arranged in two standard horizontal orthogonal tables L9 (33) using Taguchi orthogonal array for three factors and three levels (Cimbala, 2014), resulting in nine treatments for each experiment. Each treatment was conducted in triplicate. The N-amino acid yield of the hydrolysate was determined using the formol titration method as described by Gump et al. (2002).
The optimum acid-hydrolysis conditions were screened to produce LFs as follows: (1) to produce the P fertilizer, 12 g of pig hair was mixed with 12 g of pig faeces and acidified with 240 mL H2SO4 (H+ = 7.5 mol L−1), and then exposed to the microwave radiation at 200 W for 90 minutes. Hydrolysates were collected and their total nitrogen (TN), phosphorus (TP) and potassium (TK) were determined before neutralization and dilution as presented in Table 1. Afterwards, hydrolysates were neutralized to pH 5.0 using calcium hydroxide Ca(OH)2, filtered and stored for further analysis. (2) The same procedures and quantities were used to produce the C fertilizer from a mixture of chicken feathers and faeces.
Chemical properties of experimental fertilizers before and after neutralization and dilution.
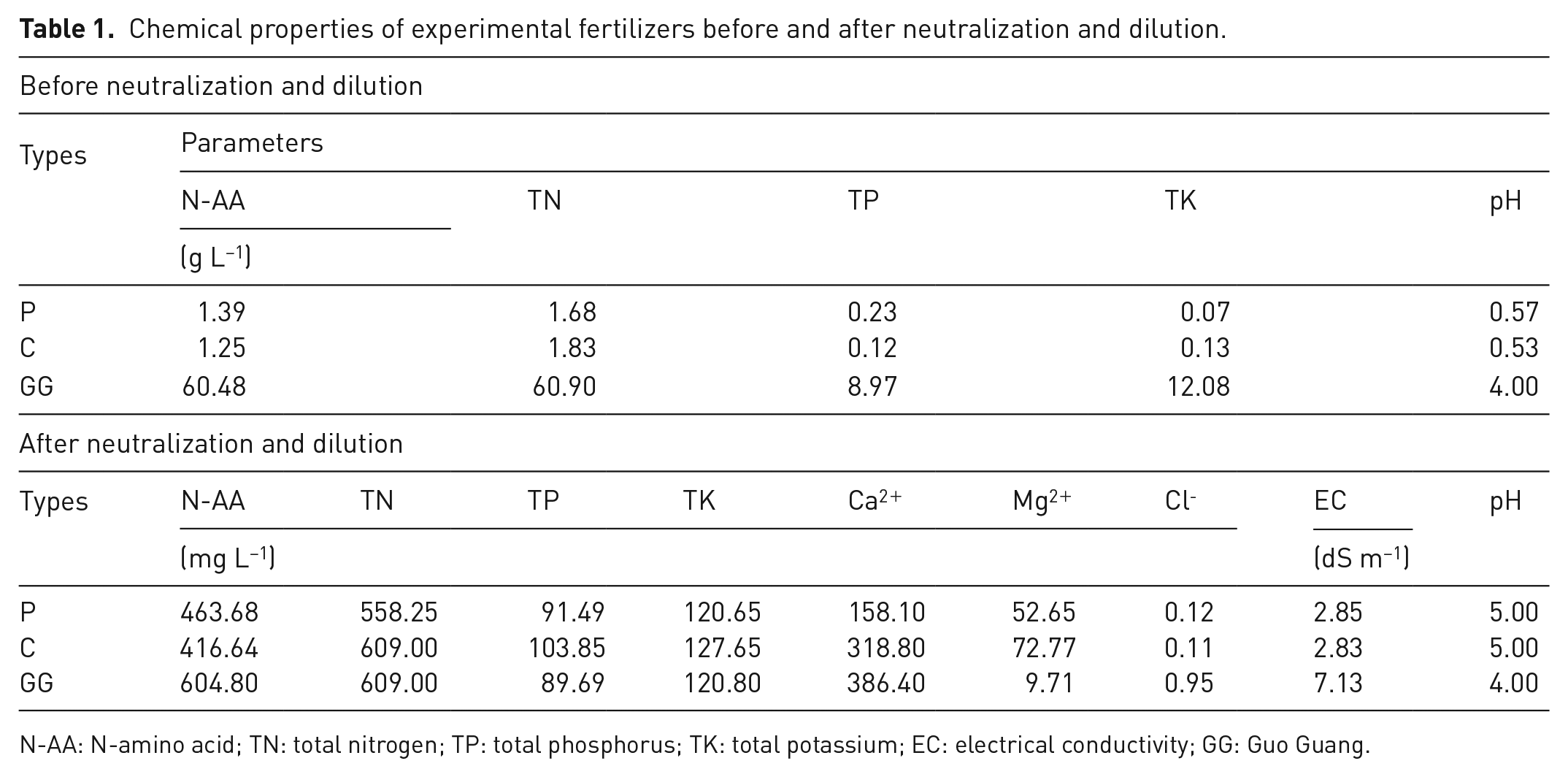
N-AA: N-amino acid; TN: total nitrogen; TP: total phosphorus; TK: total potassium; EC: electrical conductivity; GG: Guo Guang.
To evaluate the efficacy of the prepared LFs on plant growth and productivity, hot pepper (Capsicum annuum L.) was selected as a test plant and a commercial amino acid LF (GG) was used as a reference. Furthermore, monocalcium phosphate Ca(H2PO4)2 and potassium sulphate K2SO4 were used to enrich the prepared fertilizers with phosphate and potassium to make their concentrations comparable to those of the GG LF. The GG fertilizer was diluted by 100 folds with deionized water, whereas the P and C fertilizers were diluted by 3 folds to converge the concentrations of macronutrients (NPK) in all fertilizers. The LFs were analysed before application to the soil (after neutralization and dilution) as presented in Table 1 according to Gupta (2000). The amino acid types and contents in the fertilizers were determined using the automatic amino acid analyser according to the Agricultural Standards of China (NY/T 1975-2010), and the results are presented in Table 2.
Amino acid types and contents in experimental LFs.
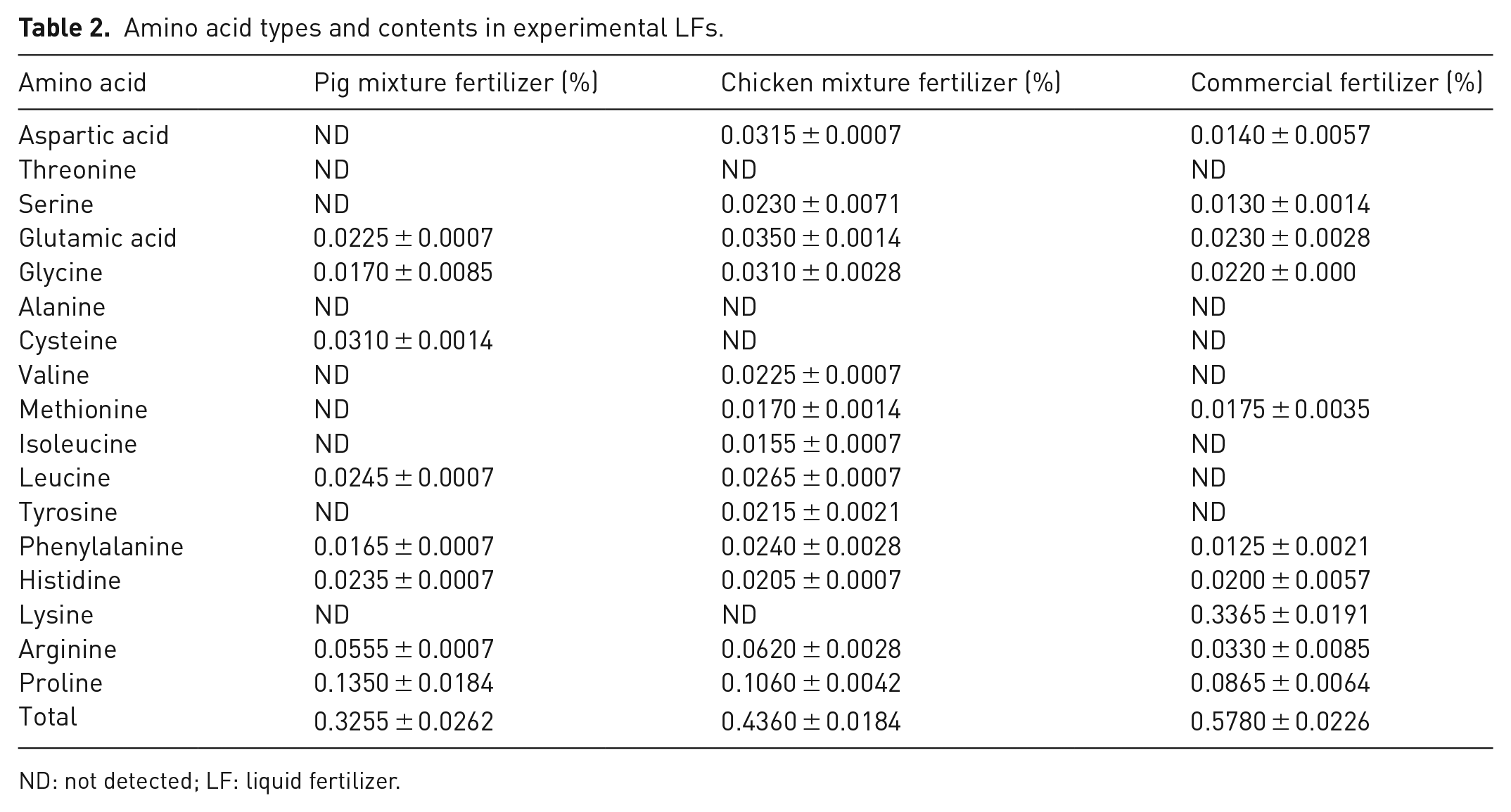
ND: not detected; LF: liquid fertilizer.
Soil sampling and analysis
One hundred kilograms of soil was sampled at 0–30 cm depth from a field (30°27′15.7″N, 112°40′28.6″E) in Qianjiang city of Hubei Province, China. The samples were mixed, air-dried, crushed and sieved through 2-mm sieve for determination of physical and chemical properties as follows. The particle size distribution was analysed following Piper (1950); the soil electrical conductivity (EC) was measured in saturated soil paste extract using EC meter and the soil pH was determined in soil–water suspension (1: 2.5) using a pH meter (Jackson et al., 1973). Soil organic matter content was analysed using the total organic carbon analyser (vario TOC select) (ISO, 1995), and the cation exchange capacity was determined by the ammonium acetate method as described by NIAST (2000). The chemical and physical properties of the investigated soil are presented in Table 3.
Chemical and physical properties of experimental soil.
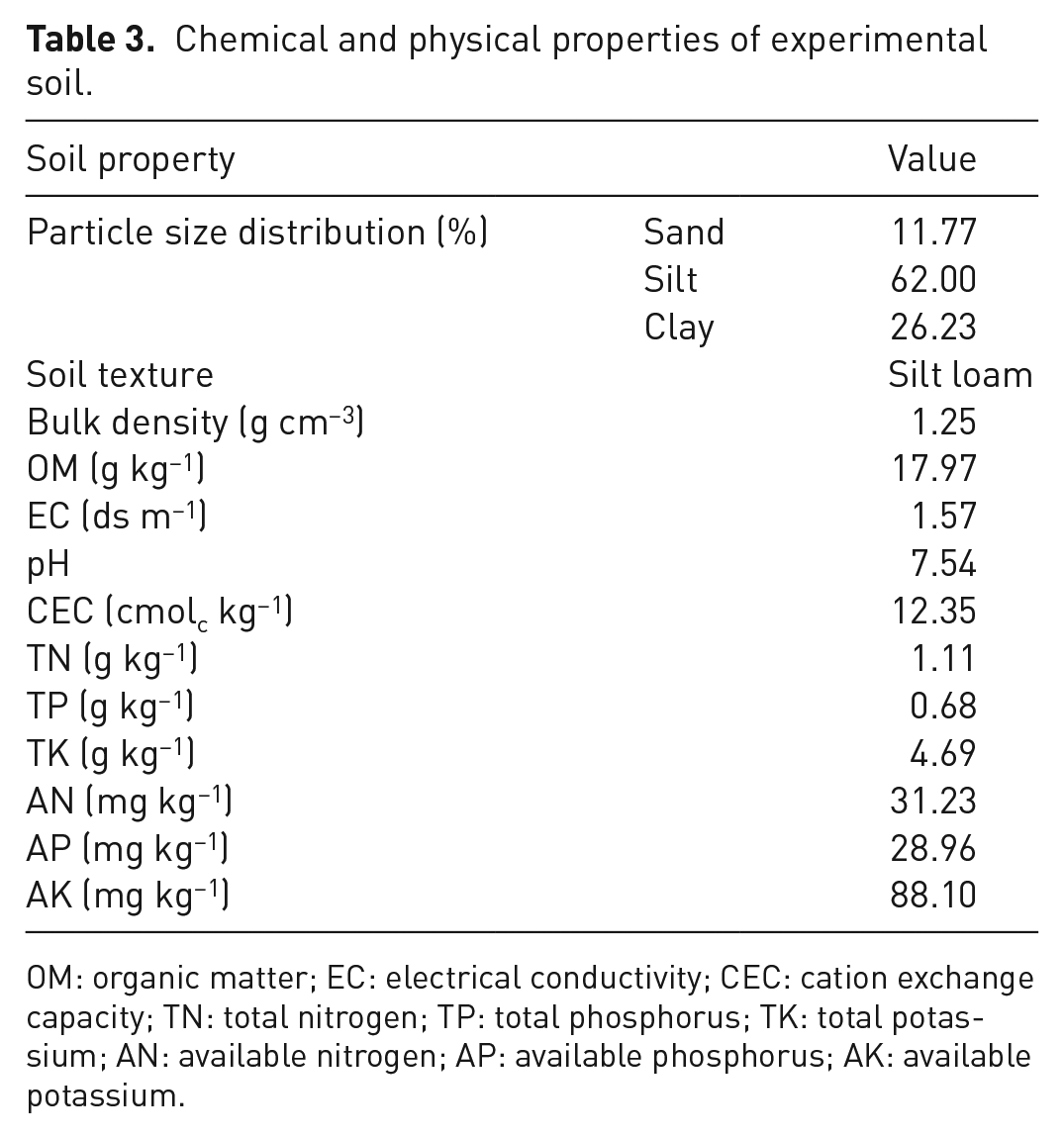
OM: organic matter; EC: electrical conductivity; CEC: cation exchange capacity; TN: total nitrogen; TP: total phosphorus; TK: total potassium; AN: available nitrogen; AP: available phosphorus; AK: available potassium.
Pot experiment for hot pepper (C. annuum L.) cultivation
A pot experiment following a factorial randomized complete block design was conducted under greenhouse conditions at Huazhong Agricultural University for 10 weeks to evaluate the efficacy of the prepared LFs on hot pepper (C. annuum L.) growth and yield compared with a commercial amino acid LF (GG). A total of 21 plastic pots (22.5 cm diameter top, 16.5 cm diameter base and 18 cm depth) were used, and each was filled with 4 kg soil and planted with two hot pepper seedlings.
P, C and GG fertilizers were applied weekly per pot at two doses of 50 and 100 mL. The untreated pots served as the control (CK). Therefore, the experiment comprised seven treatments (CK, P-100, P-50, C-100, C-50, GG-100 and GG-50), and each treatment was replicated three times. The pots were kept under greenhouse conditions (temperature of 25 ± 5°C, natural sunlight and relative humidity of 46 ± 8%) for 10 weeks under continual irrigation with deionized water to keep the soil moisture content at 70% of the water holding capacity.
Plants, fruit and soil analysis
At the end of the experiment, the fruit, shoots and roots of pepper plants were collected and weighed to obtain their fresh weights. The roots and shoots were oven-dried at 70°C for 2 days to determine the dry weight, and then crushed and digested with H2SO4 + H2O2 according to Hseu (2004). TN, TP and TK contents in different plant parts (shoots and roots) were analysed following the methods previously described (NIAST, 2000), using Kjeldahl, ultraviolet spectrophotometric and flame photometric methods, respectively. The vitamin C (Vit C) content in fruit was determined by the photometric method (Arya et al., 1998), and the fruit soluble sugar (SS) content was detected using the anthrone–sulphuric acid method (Leng et al., 2016).
After pepper harvesting, soil samples were also collected from the rhizosphere of each pot, air-dried and sieved through a 2-mm sieve to analyse their available macronutrient (NPK) contents (Gupta, 2000).
Statistical analysis
Data analysis was conducted using SPSS statistical software version 22. The mean values were calculated as an average of three replicates before analysis. Statistical differences among groups were determined by one-way analysis of variance and Duncan test at the probability of 5%. OriginPro software version 8.5.1 was used to draw the figures.
Results and discussion
Optimum acid-hydrolysis conditions of the experimental wastes
The N-amino acid yields obtained from different acid-hydrolysis conditions of pig and chicken wastes are presented in Table 4. As a result, the maximum N-amino acid yield was obtained under the conditions of A2B2C3 (7.5 mol L−1, 200 W and 90 minutes) for P and C fertilizers (12.54 and 10.68%, respectively). The mean value of the sum of each level Li was calculated and presented in Table 4 to determine the impact of the three factors on N-amino acid yield. The calculated mean value range (R = Limax – Limin) of each level represented the impact of each factor on the N-amino acid yield. In terms of the chicken wastes, the R values followed the order of RB > RC > RA, suggesting that microwave power (B) is the most important factor in the hydrolysis process. This finding is consistent with that of Chen et al. (2015), who revealed that microwave heating power is the main factor that accelerates the hydrolysis of poultry feathers without the use of any catalyst. In contrast, the R value for the hydrolysis conditions of pig wastes followed the order of RC (5.18) > RA (4.43) > RB (2.08), indicating that time is the most important factor for the hydrolysis process of pig wastes. This result is in agreement with that of Esteban et al. (2010), who reported that reaction time of pig hair hydrolysis process was an essential factor for amino acid yield, and the highest rate of hydrolysis process was obtained during the first 90 minutes. In addition, another study reported that the yield of amino acid from hair hydrolysis increased with increasing reaction time due to the extraction of more amino acids (Nagarjuna et al., 2020).
Effects of different acid-hydrolysis conditions on N-amino acid yield and the mean values of hydrolysis factors for pig and chicken wastes.
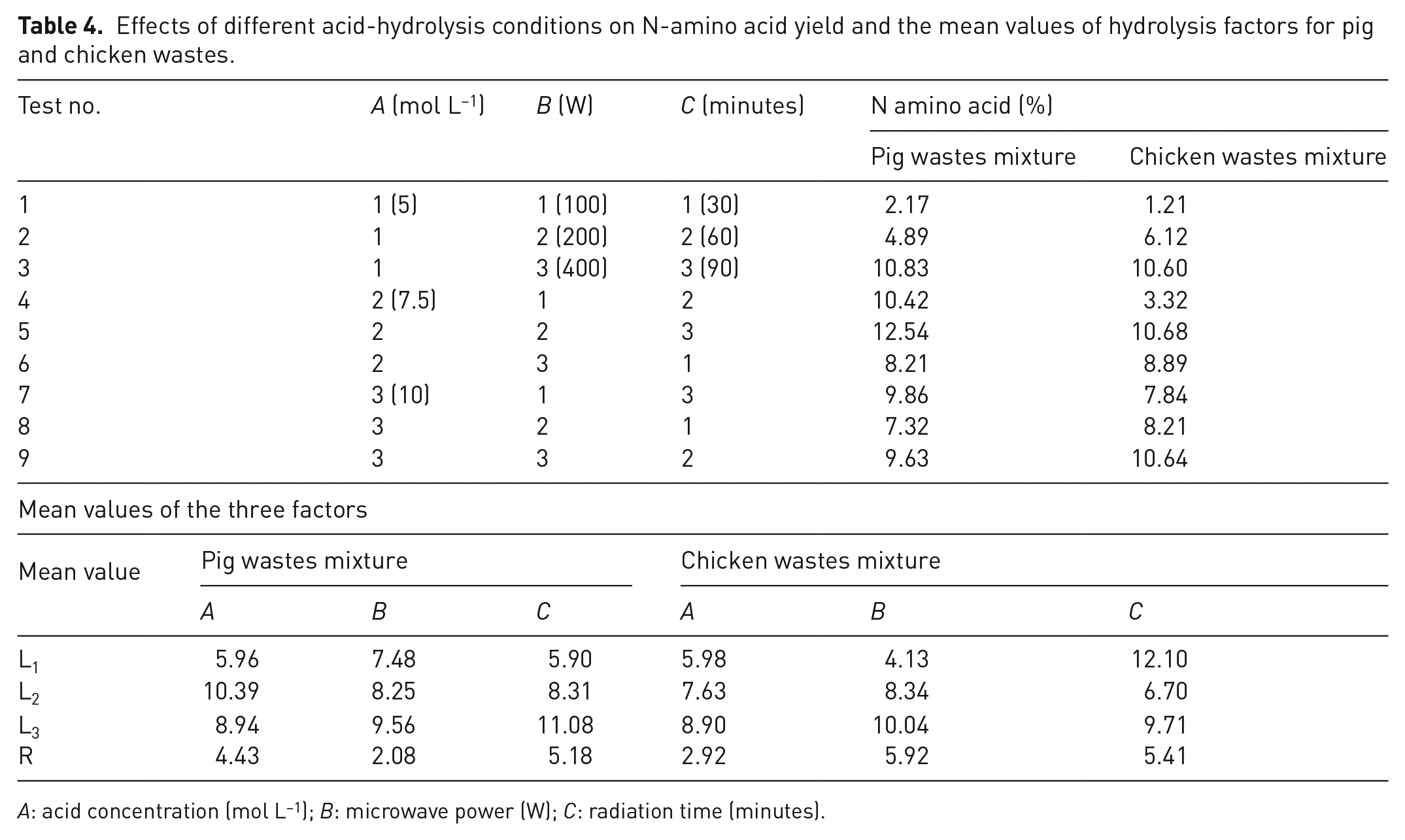
A: acid concentration (mol L−1); B: microwave power (W); C: radiation time (minutes).
Characteristics of the prepared LFs
The chemical characteristics of commercial and the prepared LFs (before neutralization and dilution) are listed in Table 1. The results showed that the pH values of P and C fertilizers were 0.57 and 0.53, respectively, which were extremely low as they were obtained from acid hydrolysis. Therefore, the pH of both P and C LFs was adjusted to pH 5.0 using Ca(OH)2 (Seong et al., 2016) to avoid the adverse effect of excess acidity on plant growth. The results also showed that P and C fertilizers had extremely high TN-to-TK ratios (23.59 and 14.73, respectively) relative to the commercial fertilizer GG (5.04), which might be a plant growth-limiting factor (Kang et al., 2019). Thus, potassium sulphate K2SO4 was used to increase the potassium content in P and C fertilizers to bring their TN-to-TK ratios close to that of GG. Similarly, monocalcium phosphate Ca(H2PO4)2 was added to P and C fertilizers to maintain the TN-to-TP ratio at the same level in all LFs.
Afterwards, all LFs were diluted using deionized water, and their chemical properties before application to the soil (after neutralization and dilution) were determined and shown in Table 1. The TN contents in P, C and GG fertilizers were 558.3, 609 and 609 mg L−1, whereas the corresponding N-amino acid yields were 463.7, 416.7 and 604.8 mg L−1, respectively, suggesting that the most nitrogen in the commercial fertilizer GG was in the form of amino acids. In terms of other nutrients, GG (386.4 mg L−1) and C fertilizer (318.8 mg L−1) showed higher Ca2+ contents than the P fertilizer (158.1 mg L−1), whereas C and P fertilizers (72.77 and 52.65 mg L−1, respectively) had higher contents of Mg2+ than GG (9.71 mg L−1), indicating a high Mg2+ content in the animal wastes.
The EC of the P, C and GG fertilizers was 2.85, 2.83 and 7.13 dS m−1, respectively (Table 1). The EC of GG was significantly higher than that of P and C fertilizers, which could be related to its high chloride (Cl−) content as presented in Table 1, where Cl− contents of the P, C and GG fertilizers were 0.12, 0.11 and 0.95 mg L−1, respectively. Therefore, LFs hydrolysed from pig and chicken mixtures are safer than the commercial fertilizer GG.
The types and contents of amino acids in the LFs were also determined and the results are shown in Table 2. GG exhibited the highest amino acid content (0.54%), whereas the C and P fertilizers only contained 0.38 and 0.28% of total amino acid, respectively. In addition, the C fertilizer had 13 types of amino acids, followed by GG fertilizer (10 types of amino acids) and P fertilizer (8 types of amino acids).
Lysine was the predominant amino acid in GG, accounting for 61.94% of its total amino acid content, but it was not found in the prepared fertilizers. Lysine is an important component of root exudates (Ye et al., 2018) with an isoelectric point of 9.74 (Pušnik et al., 2016). Accordingly, this amino acid exists in soil mainly in the form of positively charged ions (Ye et al., 2018), and may also be strongly sorbed on soil surface, which may reduce its bioavailability (Vieublé Gonod et al., 2006). Moreover, lysine can be transferred preferably across the plasma membrane with high affinity compared with negatively charged amino acids (Kanwa et al., 2020).
In the present study, the C fertilizer was rich in the amino acids of proline, leucine and valine. Proline plays a crucial role in plant cellular metabolism, particularly under adverse conditions such as drought and stress (Tegeder and Rentsch, 2010). Unlike that of lysine, the isoelectric point of proline is about 6 (Liu and Wang, 2020). Therefore, proline is mainly present in the form of negatively charged particles, which are not easily adsorbed on soil surface when the soil pH is higher than 7.0, and hence can remain longer in the soil solution. Leucine is another essential amino acid required for signalling cell wall stress (salt stress), which effectively regulates plant growth (Zhao et al., 2018). Besides, leucine is essential for pollen germination and growth (Wang et al., 2018). It has an isoelectric point of 6.0, and exists mainly as negatively charged particles in alkaline soils (Zhou et al., 2019). Valine is also an important amino acid that improves the photosynthetic rate and fruit quality (Li et al., 2020), with an isoelectric point of 6.0, and therefore exhibits negative charge in alkaline media (Sheikhian, 2021). It is noteworthy that the pH of the prepared fertilizers is acidic (pH = 5.0). Therefore, amino acids such as proline, leucine and valine may be added mainly as positive charged particles and could be absorbed by plant roots with high affinity. Even if these amino acids remain longer in the soil solution (pH = 7.54), their negative charges may increase the chelation of nutrient cations (Ghasemi et al., 2014) and promote their uptake by plants.
Effects of LFs on hot pepper growth and yield
Application of the three studied fertilizers significantly (p < 0.05) increased the dry weights of shoots and roots in pepper plants compared with the CK (Figure 1(a)). The dose of 100 mL per pot resulted in more dramatic increases than 50 mL per pot. C and GG fertilizers resulted in higher shoot and root dry weights than P fertilizer. This increase might be related to the higher total amino acid contents in C and GG fertilizers than those in the P fertilizer. Our findings are supported by a previous report that the application of amino acid fertilizer increased the shoot and root biomass of an emergent accumulator plant (Nasturtium officinale R. Br.) (Zhang et al., 2022a).
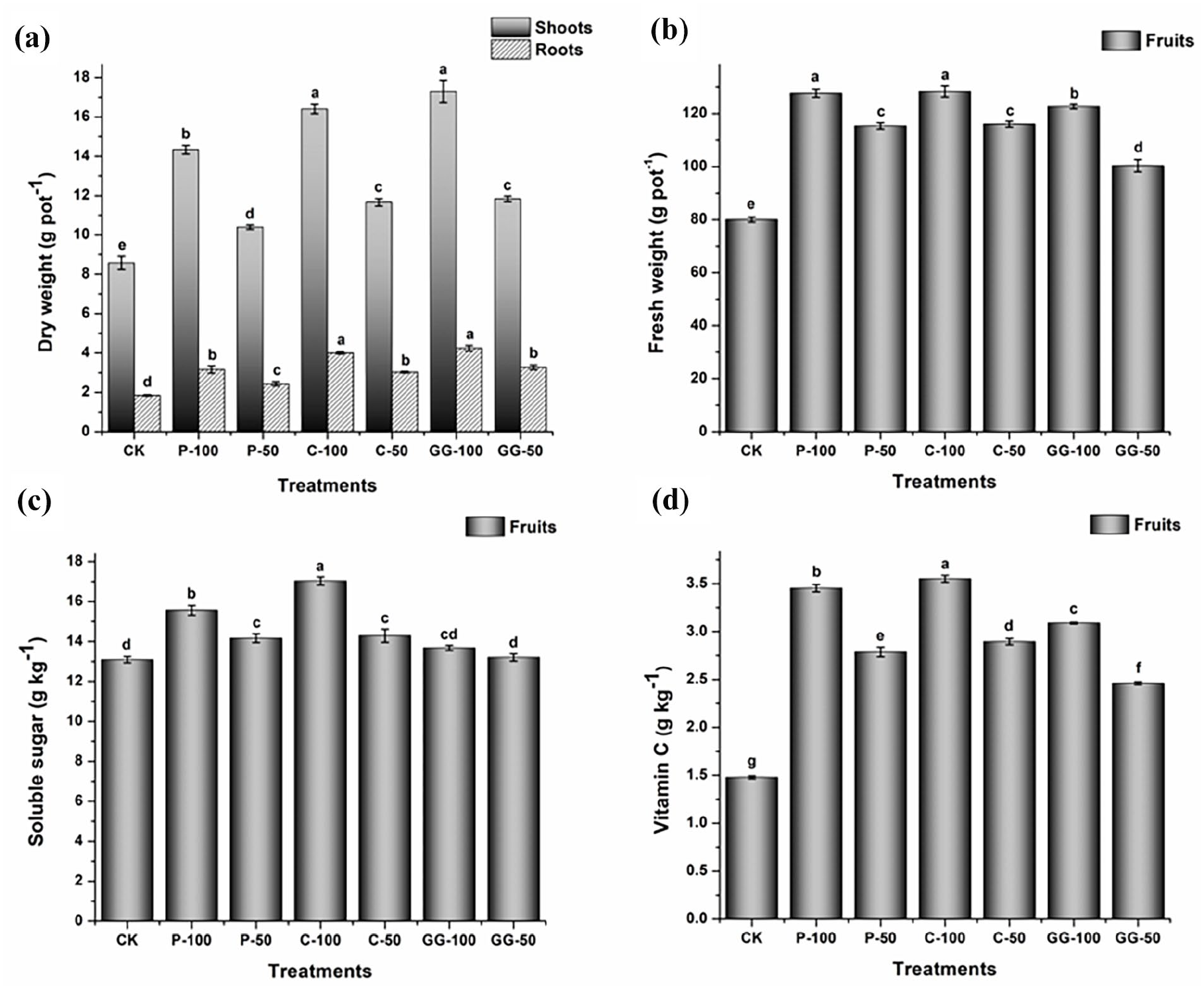
Effects of amino acid LFs on (a) shoot and root dry weights of pepper plants, (b) fruit yield, (c) SS and (d) Vit C contents in the fruit. Results are expressed as mean ± standard deviation (n = 3). Significant difference at p < 0.05 is indicated by different letters on bars as determined by one-way analysis of variance followed by Duncan’s test.
Compared with that of the CK, fruit yield was also considerably increased by the application of all fertilizers (p < 0.05, Figure 1(b)), particularly at the dose of 100 mL per pot. Many studies have accredited this positive impact for their contents of amino acids on the growth and yield of hot pepper (Bakpa et al., 2021; Kang et al., 2019), wheat (Popko et al., 2018), lettuce (Khan et al., 2019), soybean (Kocira et al., 2018) and cowpea (Wang et al., 2019). Here, there was no significant variation in fruit yield among the three studied fertilizers when applied at 100 mL per pot. Our results also indicated that P and C fertilizers could improve the SS content in pepper fruit compared with the control and GG fertilizer (p < 0.05, Figure 1(c)). In addition, application of the three LFs enhanced the ascorbic acid (Vit C) content compared with the CK (p < 0.05, Figure 1(d)), particularly at the dose of 100 mL per pot. In this regard, both P and C fertilizers resulted in better quality traits than GG, particularly the C fertilizer. Consistent with our results, previous studies have revealed that the application of amino acid water-soluble fertilizer (AWF) could increase the SS and Vit C in chilli pepper (Bakpa et al., 2021). Notably, in the study of Bakpa et al., AWF was found to contain different types of amino acids such as valine, isoleucine, leucine and tyrosine, which were not found in the GG fertilizer but in the C fertilizer in this study. In addition, AWF contained proline, phenylalanine and arginine, which were found in the GG fertilizer but in lower amounts than in our prepared fertilizers. These results suggest that the improvement of quality traits by C and P fertilizers compared with GG may be related to their more types of amino acids. Similarly, agro-industrial waste extract (rich in peptides and free amino acids) could increase Vit C content in kiwi fruit (Donno et al., 2013). Furthermore, plants can synthesize small amounts of amino acids, and this process is energy consuming (Hildebrandt et al., 2015). Accordingly, amending soils with fertilizers based on amino acids can save the energy of plants and increase their productivity (Popko et al., 2018).
Effects of LFs on NPK availability in soil
Soil contents of available nitrogen (AN), phosphorus (AP) and potassium (AK) are shown in Figure 2. The application of all studied fertilizers significantly increased soil AN and AP contents compared with the CK (p < 0.05). The increase in AN content was more noticeable with increasing fertilizer application dose. In contrast, there was no variation in AP content among different fertilizer treatments. These results suggest the importance of amino acids as a source of N for plants (Rothstein, 2010), besides chelating the cations bound with phosphorus (Souri and Hatamian, 2019) to release it and increase its solubility in soil (Carvalhais et al., 2011). Moreover, fertilizer application resulted in a significant reduction (p < 0.05) of AK content in the soil, with no significant variation among the investigated treatments. This might occur when amino acids stimulate soil biota (Bahadur et al., 2016) to utilize soil available K and temporarily reduce its available content in soil.
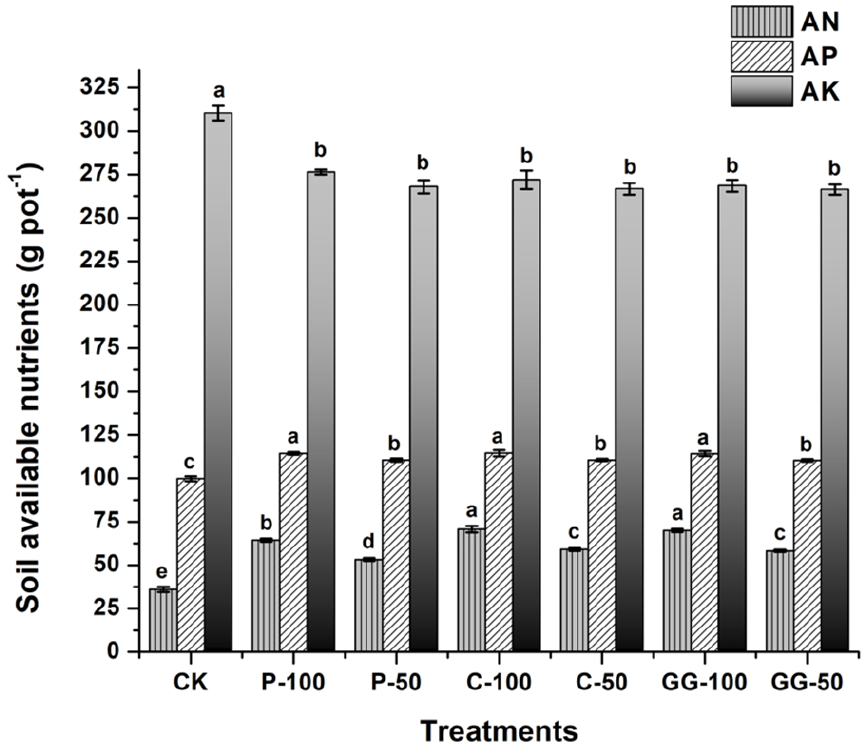
Effects of amino acid LFs on AN, AP and AK contents in the soil. Results are expressed as mean ± standard deviation (n = 3). Significant difference at p < 0.05 is indicated by different letters on bars as determined by one-way analysis of variance followed by Duncan’s test. Similar letters indicate no significant variation among treatments.
Effects of LFs on NPK uptake by plants
N and K uptake per pot by pepper plants increased significantly upon the application of all studied fertilizers (Figure 3), particularly at the dose of 100 mL per pot. These results suggest that amino acid fertilizers can supply instantly accessible N, which can be absorbed faster by the plants than inorganic N (Bakpa et al., 2021). Particularly, C-100 and GG-100 resulted in the most dramatic increase in K uptake, whereas no variation in N uptake was detected (p < 0.05) among the studied fertilizer treatments. In general, N and K contents in shoots were much higher than those in roots, probably because amino acids increase the N use efficiency via improving its phloem transport (Criado et al., 2017). Moreover, amino acids induce beneficial physiological processes (Teixeira et al., 2017) and act as osmolytes to promote ion transport in plants and also protein synthesis (Souri and Hatamian, 2019). Although P fertilizer increased the K availability in the soil, the uptake of the increased available K by plants was lower than that by plants treated with the other two fertilizers (C and GG). The possible reason is that soil K was chelated by cysteine amino acid, which was found in P fertilizer but not in C and GG fertilizers. Besides, cysteine was reported to have high binding energy with ions (Adams et al., 2017), which exceeds the capability of the plants to uptake available K from the soil.
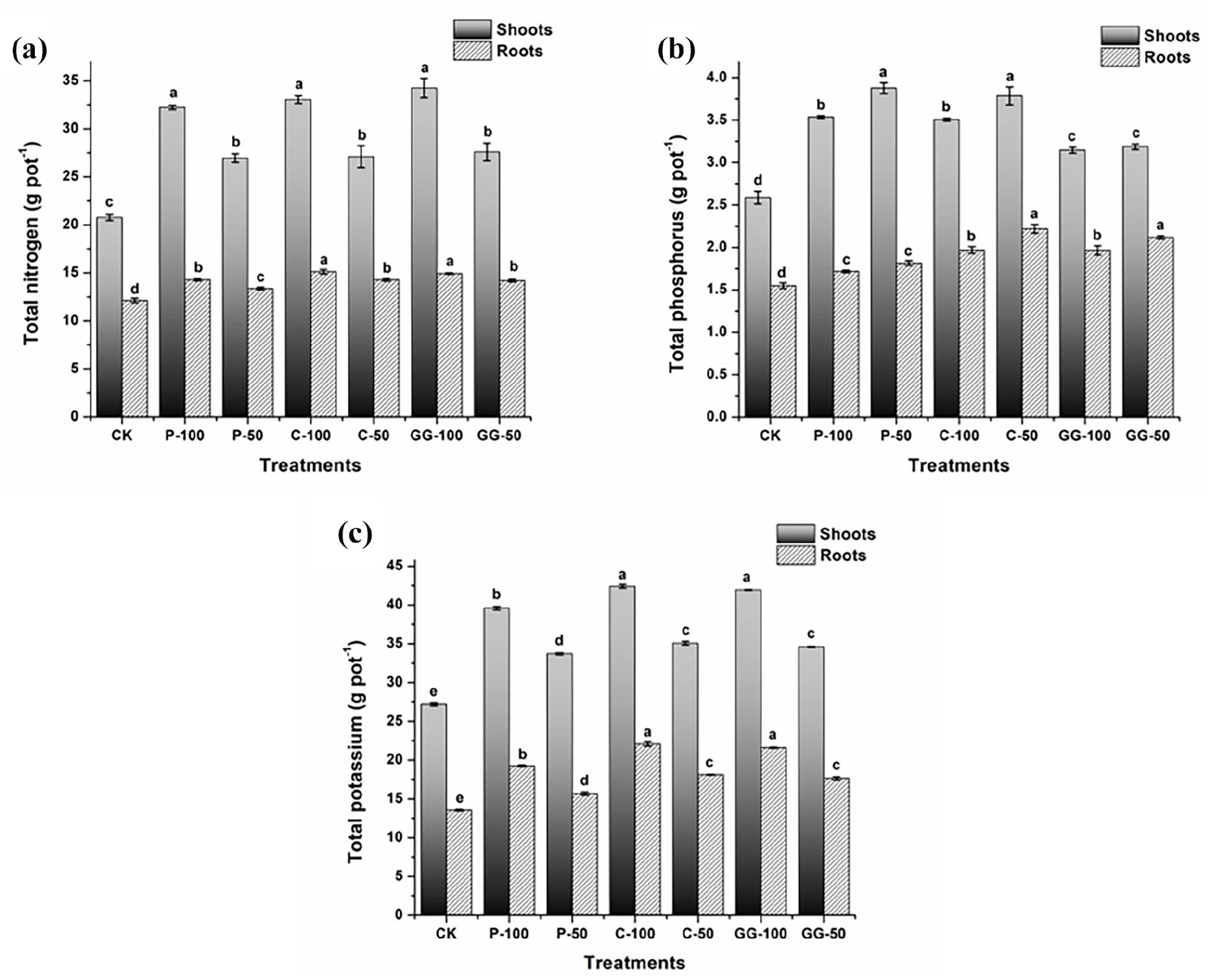
Effects of amino acid LFs on (a) nitrogen, (b) phosphorus and (c) potassium uptake (g per pot) by the plants (shoots and roots). Results are expressed as mean ± standard deviation (n = 3). Significant difference at p < 0.05 is indicated by different letters on bars as determined by one-way analysis of variance followed by Duncan’s test. Similar letters indicate no significant variation among treatments.
Phosphorus uptake by pepper shoots and roots also increased significantly (p < 0.05) under the application of all tested fertilizers compared with the control. Unexpectedly, the increase in phosphorus uptake was more prominent at the dose of 50 mL per pot than at 100 mL per pot. This finding is in agreement with that of Shehata et al. (2011), who reported that application of amino acids at lower doses resulted in higher phosphorus uptake by celeriac plant than application at higher doses. Notably, ‘P-50’ and ‘C-50’ treatments resulted in significantly more dramatic increases (p < 0.05) in phosphorus uptake by plants than commercial GG whether at 50 or 100 mL per pot. These results indicate that the investigated fertilizers could effectively promote the uptake of nutrient cations, and that of nutrient anions to a lesser extent, which is consistent with the results previously reported (Kang et al., 2019).
Although the prepared P and C fertilizers recorded similar results to the commercial fertilizer GG in terms of shoot and root dry weight, N and K uptake by plants, and AN and AP in the soil, they showed certain advantages over the GG fertilizer in fruit yield, quality traits and phosphorus uptake by plants, implying that they are promising and competitive fertilizers to promote plant growth and productivity.
Conclusion
This study demonstrated the feasibility of producing LFs using environmentally harmful livestock and poultry wastes as raw materials. The experimental wastes were hydrolysed using microwave-assisted acid hydrolysis, then the hydrolysates were neutralized to pH 5.0 using Ca(OH)2. Afterwards, K2SO4 and Ca(H2PO4)2 were added to the hydrolysates to increase their phosphorus and potassium contents. The prepared P and C fertilizers could enhance shoot and root dry weight, fruit yield, NPK uptake by the plants and available N and P contents in the soil compared with the control. They could also considerably improve fruit quality traits such as SS and Vit C compared with the commercial fertilizer (GG). In conclusion, our hydrolysed products could be used as valuable fertilizers competitive with the commercial ones. Moreover, microwave-assisted acid hydrolysis is a promising technique for converting environmentally hazardous wastes, such as livestock and poultry wastes, into valuable products that can be used as LFs to increase crop productivity, which is an important aspect of sustainability. The beneficial effects of our prepared fertilizers could be associated with their abundant types of amino acids. However, the mechanism underlying the effect of these amino acids needs to be further elucidated. Moreover, further research is required for the application of these fertilizers at large scale.
Footnotes
Acknowledgements
The authors are grateful to Dr Daniel Zogona for his valuable comments on this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundamental Research Funds for the Central Universities (No. 2662023DKPY003), China and the National Natural Science Foundation of China (No. 52070085).
