Abstract
This study introduces an innovative approach for enhancing oil–water emulsion separation using a polyethersulfone (PES) membrane embedded with a nanocomposite of graphene oxide (GO) and silver oxide (AgO). The composite membrane, incorporating PES and polyvinyl chloride (PVC), demonstrates improved hydrophilicity, structural integrity and resistance to fouling. Physicochemical characterization confirms successful integration of GO and AgO, leading to increased tensile strength, porosity and hydrophilicity. Filtration tests reveal substantial improvements in separating various oils from contaminated wastewater, with the composite membrane exhibiting superior efficiency and reusability compared to pristine PES membranes. This research contributes to the development of environmentally friendly oil–water separation methods with broad industrial applications.
Introduction
Water scarcity and excessive water consumption by industries have emerged as critical global concerns in the 21st century (Al-Ghouti et al., 2019; Sustainable development goal 6 Synthesis Report on Water and Sanitation, 2018). This growing recognition of the finite nature of our freshwater resources has spurred heightened attention to the management of industrial wastewater, particularly in sectors such as food production, steel manufacturing, petrochemicals, textiles and more. These industries frequently discharge significant volumes of oily wastewater into natural water bodies. Furthermore, the transportation of crude oil via sea and ocean routes has been associated with oil spills that inflict severe damage on marine and aquatic ecosystems (Saharan et al., 2022).
Over the past decade, there have been numerous oil spill incidents, underscoring the urgency of addressing this pressing issue. Notable incidents include an oil spill of approximately 138,000 tonnes in the East China Sea in 2018, a spill of approximately 1240 tonnes from the Walsh County Keystone Pipeline, and an oil spill of approximately 184.87 tonnes in Russia in 2020 (Saharan et al., 2022, 2023). These incidents not only result in substantial financial losses but also cause long-term and often irreparable harm to marine ecosystems. Thus, there is an escalating demand for continuous, large-scale oil–water separation processes that can safeguard both water and oil resources while meeting stringent environmental standards (Xu et al., 2018).
In response to these standards, industrial wastewater must undergo de-oiling and demineralization treatments before it can be responsibly disposed of or recycled. Currently, industries employ various hybrid technologies to address these challenges, including adsorption, flotation, biological treatment, cyclonic and gravity separators, coagulation-flocculation and evaporation (Ebrahimi et al., 2018; Nasiri and Jafari 2017; Veil et al., 2004). However, these methods are not without their limitations, such as low oil recovery rates, the use of hazardous chemicals and the time-consuming nature of the processes. In light of these constraints, there is an increasing need for advanced materials and strategies that can enhance the efficiency of oil–water emulsion separation.
Recently, highly hydrophilic membranes have gained recognition for their fast, efficient and eco-friendly role in the membrane filtration of oil–water mixtures. These membranes offer a promising solution to address the contamination resulting from oil spills and the discharge of industrial oily effluents.
Despite the promise of membrane filtration, fouling remains a significant challenge inherent in this technology. Membrane fouling can occur through the deposition of an oil layer on the membrane surface or by the blocking of membrane pores by oil droplets. The consequences of fouling are substantial, resulting in a significant decline in flux, or the flow rate of permeate through the membrane. Fouling can be attributed to various factors, including the adsorption of oil components within the membrane, the formation of a cake layer on the membrane’s surface and the blockage of membrane pores. The structure of the membrane’s pores, the characteristics of its surface and the operational and process conditions all play pivotal roles in determining the extent of membrane fouling (Dickhout et al., 2017; Rı́os et al., 1998).Therefore, the development of tailored membrane materials becomes essential to mitigate fouling and its associated adverse effects, such as flux decline and decreased retention. To address these challenges, many research groups have explored the integration of additives or nanocomposites that incorporate various metal oxides into membranes. Among the metal oxides utilized, silver oxide (AgO), silicon oxide, iron oxide, molybdenum oxide and graphene have gained prominence due to their demonstrated efficacy in extending the lifespan and enhancing the performance of membranes (Igwe et al., 2013; Zhu et al., 2017).
Notably, graphene oxide (GO) has emerged as a compelling candidate for improving membrane performance when incorporated into nanocomposites. The unique properties of GO, including its heightened hydrophilicity, high mechanical strength and favourable dispersion in polymer solvents, make it a standout choice (Amiri et al., 2017; 2019; Khorasanizadeh et al., 2023; Mir and Salavati-Niasari, 2013; Panahi et al., 2023; Panahi-Kalamuei et al., 2014; Salavati-Niasari et al., 2009; Teymourinia et al., 2017; Zinatloo-Ajabshir et al., 2021). Additionally, AgO has been chosen for its superior antibacterial activities. The successful integration of nanocomposites into membrane materials has demonstrated substantial improvements in various parameters. For example, Arefi-Oskouia et al. achieved a remarkable 45% improvement in water flux and a 30% increase in the flux recovery ratio (FRR) by incorporating a MoS2/PES nanocomposite membrane compared to a bare PES membrane (Ikhsan et al., 2018). Furthermore, Ikhsan et al. reported high permeate flux and enhanced oil rejection by blending a PES membrane with halloysite nanotube-hydrous ferric dioxide (Abdel-Aty et al., 2020). The recent advancements in membrane technology demonstrate a growing interest in developing efficient ‘water-removing’ materials tailored for addressing the challenges posed by oil-in-water emulsions (Shen et al., 2023; Yang et al., 2022). Notably, Ge and collaborators successfully engineered a composite membrane combining microspheres and nanofibers, showcasing a remarkable capability for microscale oil-in-water emulsion separation through innovative electrospinning and electrospraying techniques (Ge et al., 2017). In a parallel effort, Shao et al. introduced a polyacrylonitrile-based nanofiber membrane featuring a multi-hydrophilic cross-linked network. This design facilitated rapid and effective separation of oil/water emulsions, exemplifying the versatility of nanofiber membranes (Cheng et al., 2020). Equally noteworthy is the work of Xu et al., who achieved the creation of a stable membrane by incorporating carbon nanotubes into a mixed matrix polymeric membrane through vacuum filtration, demonstrating its efficacy in oil/water emulsion separation (An et al., 2018). Furthermore, Yu et al. elevated the capabilities of membrane technology by developing a GO-modified Al2O3 microfiltration membrane via a vacuum method. Their investigation revealed superior performance characterized by high permeate flux and impressive oil rejection in oil/water emulsion separation (Hu et al., 2015). Meanwhile, Khakpour et al. (2019) explored the impact of GO/nanodiamond additives on PVC membranes, illustrating that the integration of nano-additives enhanced hydrophilicity, pure water flux and rejection during ultrafiltration. These diverse studies collectively emphasize the tremendous potential of nanocomposites in revolutionizing membrane technology for efficient oil-in-water emulsion separation. The cumulative findings underscore the significance of ongoing research endeavours in harnessing nanomaterials to address the pressing challenges in wastewater management and environmental remediation.
In light of the notable advantages offered by GO and AgO, this study seeks to advance the field by using these materials to fabricate novel GO–AgO nanocomposites. These nanocomposites are intended to be incorporated into polyethersulfone (PES) membranes. The integration of GO–AgO nanocomposites into PES membranes holds the promise of enhancing their performance in oil–water emulsion separation.
This study charts an innovative path in the quest to engineer advanced filtration membranes with unparalleled efficiency. The research involves the fabrication of PES membranes decorated with GO–AgO nanocomposites and their subsequent characterization using various spectral techniques. These advanced membranes are then deployed for the separation of oils not only from laboratory-prepared oil–water mixtures but also from real-world industrial wastewater contaminated with oil. The study presents a comprehensive investigation into the efficacy of these novel membranes and their potential for addressing pressing environmental and industrial challenges. The introduction of GO–AgO nanocomposites into PES membranes represents a novel and promising approach in the realm of membrane technology. The research aims to address the critical issue of oil–water emulsion separation by offering advanced materials and strategies. By exploring the potential of these nanocomposites, this study is poised to make significant contributions to the field, offering a sustainable and efficient solution to the challenges posed by industrial wastewater and oil spills.
Experimental details
Materials
All of the chemicals viz. silver nitrate (AgNO3), sodium borohydride (NaBH4), PES, polyethylene glycol (PEG, pore former), polyvinyl chloride (PVC), n-hexane, N,N-dimethylformamide (DMF) and sodium dodecyl sulphate (SDS) used were purchased from Sigma-Aldrich. The crude coconut and mustard oils were procured from nearby oil mill at Dosarka, Ambala, Haryana, and the same were used without any further purification. Petrol was acquired from neighbouring fuelling stations.
Synthesis of GO and GO–AgO nanocomposites
GO was synthesized utilizing graphite powder following Tour’s approach as described in our previous research article (Goyat et al., 2022). Using a (REMI) sonicator, 50 mg of synthetic GO powder was disseminated in 50.0 mL of distilled water at room temperature for 60 minutes. The 0.008 M concentration (0.0679 g) of AgNO3 is then dispersed in 50 mL of deionized water. The dispersed AgNO3 solution is then added drop by drop to the GO suspension followed by the addition of 0.5 M (1.892 g) NaBH4 100 mL solution as a reducing agent. The resultant mixture is stirred for 5 hours at 80°C and then allowed to cool at room temperature. The resulting product was filtered through Whatman filter paper no. 1 and rinsed 4–5 times with distilled water followed by pure ethanol. The produced dark grey product was then dried at 40°C for 6–8 hours in a hot air oven and then crushed to get the fine powder, and the obtained nanocomposites were used in the polymeric membranes synthesis as described below in Section ‘Fabrication of GO–AgO-blended PES-based composite membrane’.
Fabrication of GO–AgO-blended PES-based composite membrane
For the fabrication of PES-based composite membranes, the phase inversion technique was opted (Wang et al., 2014; Zinadini et al., 2014). PES pellets (10 g) were initially dehydrated for 30 minutes at 80°C in a hot air oven then constantly stirred them with DMF (85 mL) for 4 h at 60°C. Then 2.0 g of PVC, 2.0 g of PEG and 1.0 g of GO–AgO nanocomposites were added, and the mixture was stirred for another 2 hours. A translucent gel-like uniform casting solution was obtained. With the help Filmography Doctor Blade and a clean glass plate, membrane casting was done. The glass plate was instantly dipped into a water tub at room temperature to initiate phase inversion. The synthesized membrane was kept undisturbed in the water tub overnight to eliminate any remaining solvent (DMF). Finally, the obtained membrane was air-dried for 6–8 hours at 40°C before undergoing various characterization and application part.
Characterization of the prepared GO–AgO Nanocomposite (NC) material and its decorated PES membrane
The above synthesized GO–AgO nanocomposites were characterized using energy dispersive X-ray spectroscopy (EDS) and field emission scanning electron microscopy (FESEM). Furthermore, the morphology of the prepared membranes was also evaluated using EDS mapping or FESEM. The strength of the membrane was calculated using a universal testing machine (UTM). To check the strength of the membrane, tests were conducted using a piece of (15 mm long and 10 mm broad) membrane at a stretching speed of 10 mm second−1.
Hydrophilicity, porosity and water permeability test
Using a goniometer, the hydrophilicity of the fabricated membranes was determined. Firstly, all of the membranes were dried and vacuum-preserved for 3 hours at 25°C. Then the contact angles (CAs) between the designed membranes and the water droplets were measured. For each synthesized membrane, approximately nine to ten CAs took place, and the mean value with standard deviation was subsequently taken into consideration. In order to measure the membrane porosity, the dry membrane samples (pure PES membranes and GO–AgO-decorated membranes) were cut into specific dimensions (2 cm × 2 cm) and then immersed in deionized water for around 48 hours. The membranes were gently wiped, then weighed. Then, they were air-dried and placed in desiccators for 24 hours before being weighed once again, to determine the weight in the dry condition (Ganesh et al., 2013; Kou and Gao, 2011). Finally, the membrane porosity (ε) was calculated by using Equation (1):
where ω2 is the weight of wet membrane, and ω1 is the weight of dry membrane (g), A (cm2) is the area of the membrane,
Furthermore, the water permeability of the synthesized membranes (pure PES membrane and GO–AgO-decorated PES membrane) was calculated according to Equation (2):
where J is the flux of water (Lm−2 hour−1); V is the permeate volume (L); s is the used membrane area (m2); t is the procedure time (hour).
Separation of oil–water mixtures via continuous filtration setup
The fabricated GO–AgO PES membranes were employed in the continuous filtration studies to separate several types of laboratory prepared oil–water mixtures (Vatanpour et al., 2011). To assess the performance and stability of the GO–AgO PES membrane, seven cycles of filtration have been performed. A volume of 1.0 L oil–water mixture was processed in one cycle, and each cycle ran for approximately 15–20 minutes depending on the viscosity of the oil samples. After completion of each cycle, the membranes were rinsed with deionized water to get rid of any adhering material implemented for the next cycle.
Collection and preparation of oil-in-water mixtures
The total five different types of oil–water mixture were filtered through the prepared membranes as given in Table 1. The first two samples (petrol and hexane) were mixed with water approximately in 1:5 ratios, respectively. Furthermore, by utilizing the SDS surfactant and intense mechanical shearing, stable oil–water mixtures of these two samples were prepared. The next two crude oil samples (coconut and mustard oils) were obtained from the nearby oil mills and were filtered as obtained. Furthermore, a real sample of oil–contaminated water having an unknown concentration of oil was collected from the nearby oil restaurant at Ambala Cantt, Haryana, India.
Different type of oil–water samples used to investigate the study.

Furthermore, the filtration was carried out using continuous filtration setup, and the oil-removal efficiency was calculated using Equation (3).
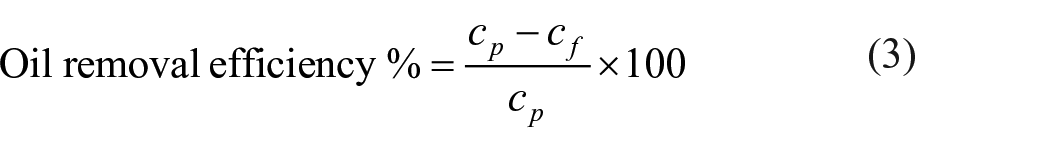
where cp is the concentration of oil present in solution before filtration, and cf is the concentration of oil obtained after filtration.
Antifouling test
The antifouling test is one of the most important tests to check the fouling of the membrane after filtration cycle. Usually, the membranes porosity decreases as the number of filtration cycles increases. It may be due to the contaminants (organic, inorganic and microbes) present in the water, which forms layer on the surface of the membrane results in its clogging. On the other hand, the microbes also assemble on the surface of the membranes, and they start consuming the membrane as their feed, leading to membrane leakage.
In this test firstly, the freshly prepared membrane was flushed with deionized water for 30 minutes and the flux (F0) that is, the volume of water filtered was measured and then through the same setup, oil-in-water emulsions were filtered for further 30 minutes. After this, the membrane was removed from the filtration setup and was washed with deionized water. The washed membrane was again used in the filtration setup and was further flushed with deionized water for the same time, and the volume of the filtrate (Fi) was measured. The water FRR was then calculated using Equation (4),

where (F0) is the pure water flux of the membrane before filtration, and (Fi) is the flux of membrane after filtration of oil–water emulsions.
Results and discussions
Characterizations of GO–AgO nanocomposite
The GO–AgO nanocomposites were successfully synthesized and were characterized by EDX and FESEM techniques. FESEM images of the GO–AgO composites obtained are shown in Figure 1. The FESEM findings revealed that no significant aggregation of AgO particles across the surface of GO nanosheet was found. The synthesized GO–AgO particles had a spherical, petal-like shape and were evenly dispersed on the surface of GO sheets (Cao et al., 2019; Jin et al., 2022; Yu et al., 2018). Additional EDX spectra confirmed the presence of carbon (C), silver (Ag) and oxygen (O) elements in the designed GO–AgO nanocomposites.
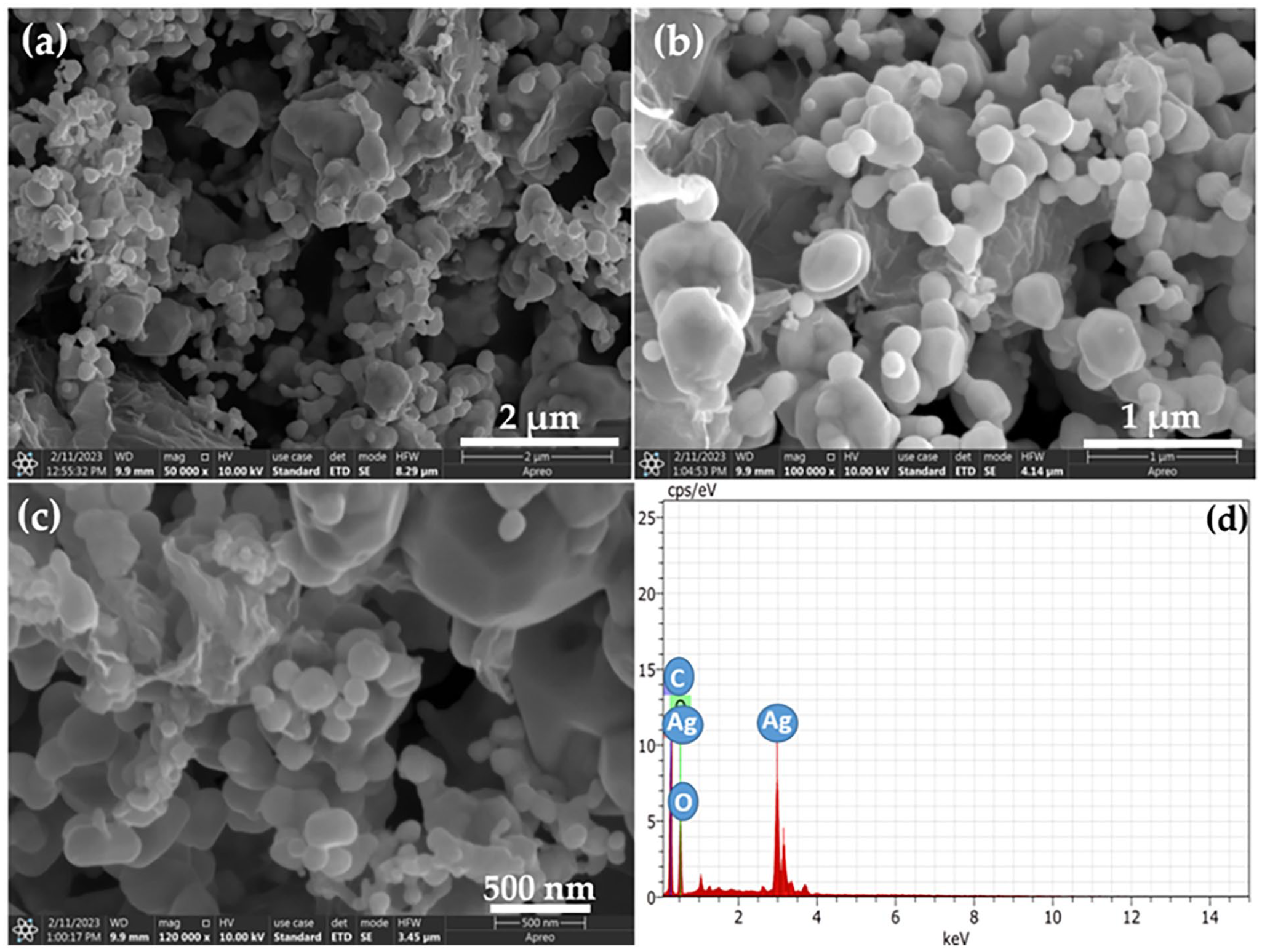
(a–c) Typical FESEM images, and (d) EDX spectrum of GO–AgO nanocomposites.
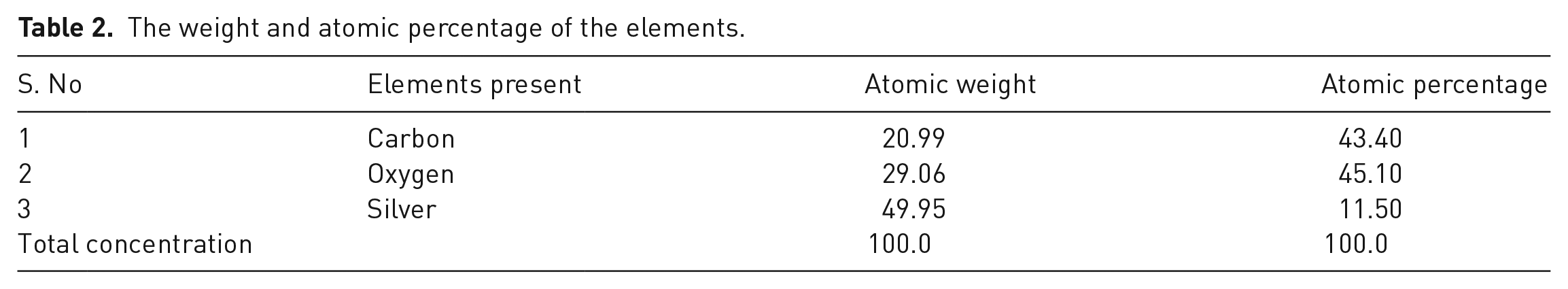
The EDX spectrum shown in Figure 1(d) offers valuable insights into the elemental composition of the synthesized GO–AgO nanocomposites. Consistent with expectations, distinct peaks corresponding to the presence of Ag, O and C elements are prominent, corroborating the successful integration of these elements within the nanocomposite structure. This comprehensive characterization affirms the successful synthesis of GO–AgO nanocomposites with uniform dispersion and establishes a foundation for their subsequent incorporation into the filtration membrane fabrication process. Furthermore, the weight and atomic percentage of each elements is shown in Table 2.
The weight and atomic percentage of the elements.
Morphological study and membrane tensile strength (MTS)
Upon confirming the distinctive characteristics of the GO–AgO nanocomposite, these nanoparticles were seamlessly integrated into the membrane fabrication process. The morphologies of the resulting membranes were meticulously examined utilizing FESEM. The micrographs in Figure 2((a)–(c)) vividly depict the porous nature of all membranes, with noteworthy variations in pore density and size compared to the pure PES membrane (Ashraf et al., 2022; Dmitrieva et al., 2022). The EDS spectra in Figure 2(d) further validate the successful incorporation of the nanocomposite, revealing the presence of C, O, chlorine (Cl) and sulphur (S) elements – primary constituents of both PES and PVC.

Typical (a–c) FESEM images, (d) EDS spectrum and (e) EDS-mapping for the PES membrane.
Furthermore, EDS mapping (Figure 2(e)) was conducted to visualize the distribution of these elements across the surface of the PES membrane. The results demonstrated a uniform distribution of each element, with no observable agglomerations. The quantitative composition of these elements in the membrane is presented in Table 3, as elucidated by the EDS mappings. This thorough analysis not only corroborates the successful integration of the GO–AgO nanocomposite into the membrane structure but also provides a detailed insight into the uniform distribution of constituent elements, essential for understanding the material’s morphological characteristics. The morphological study serves as a foundation for assessing the membrane’s structural integrity and its potential implications on performance, paving the way for subsequent discussions on tensile strength and other mechanical properties. These insights contribute to the overall understanding of the nanocomposite-embedded membrane and its suitability for application in diverse separation processes.
The compound percentage of the elements as according to the EDS mapping.

Upon the integration of GO–AgO nanocomposites onto the membrane surface, a discernible increase in the number of pores with comparable radii pore diameters was observed, as illustrated in Figure 3((a)–(c)), in contrast to the pure PES membrane. However, this phenomenon is not uniform, as the introduction of these additives initially enhances pore size and hydrophilicity, but only up to a certain threshold. Beyond this limit, the microscopic particles tend to aggregate, resulting in an increase in solution viscosity. This aggregation, in turn, imposes a kinetic barrier to phase separation, leading to a decrease in membrane pores and hydrophilicity (Fekete et al., 2023; Ismail et al., 2023).

Typical (a–c) FESEM images, (d) EDS spectrum and (e) EDS-mapping for the GO–AgO PES membrane.
To elucidate the chemical composition of the fabricated composite membrane, the EDS spectra were acquired, as depicted in Figure 3(d). The distinctive peaks corresponding to Ag, O, S, Cl and C elements affirm the successful integration of these elements within the nanocomposite structure. To further assess the uniform distribution of the added nanocomposites, EDS mapping was conducted, revealing an even dispersion of each element across the membrane surface, with no significant agglomeration observed (Figure 3(e)). Visual observations and literature reports indicate an increase in the thickness of the composite membrane. This thickness augmentation can be attributed to the incorporation of GO–AgO nanocomposites, which widen the interlayer gap between membrane layers. The elemental composition of the prepared composite membrane is detailed in Table 4, offering a quantitative representation of the incorporated elements and reinforcing the successful synthesis of the nanocomposite-embedded membrane. This comprehensive morphological and compositional analysis lays the groundwork for a nuanced understanding of the membrane’s structural features, essential for evaluating its performance and potential applications in separation processes.
The compound percentage of the elements as according to the EDS mapping.

In Figure 4, we present the calculated MTS for both the pristine PES membrane and the PES membrane adorned with GO–AgO nanocomposites. The PES membrane exhibited a maximum force (Fmax) of 32.4 N, whereas the GO–AgO decorated PES membrane displayed a significantly enhanced Fmax of 59.2 N. This notable increase in tensile strength can be attributed to the combined mechanical properties of the PVC polymer and the GO–AgO nanocomposites embedded in the PES polymer matrix. Furthermore, it was observed that the incorporation of GO–AgO nanocomposites led to a substantial improvement in tensile strength up to a certain threshold. Beyond this critical concentration, agglomeration of the nanocomposites occurred, potentially diminishing the mechanical strength of the membrane (Fekete et al., 2023; Ismail et al., 2023). This finding underscores the delicate balance required in optimizing the concentration of nanocomposites to achieve enhanced mechanical properties without compromising membrane integrity. The augmentation in tensile strength aligns with previous research findings. Zhao et al. demonstrated a significant enhancement in the mechanical characteristics of polymers through the incorporation of GO and its nano-additions (BărdacăUrducea et al., 2020). This reinforces the notion that the integration of nanocomposites can indeed impart superior mechanical properties to polymer matrices. The elucidation of MTS provides crucial insights into the mechanical robustness of the GO–AgO PES membrane, supporting its potential applicability in diverse separation processes. The observed enhancement in tensile strength, while promising, necessitates a nuanced understanding of the optimal nanocomposite concentration to ensure both mechanical resilience and effective separation performance.
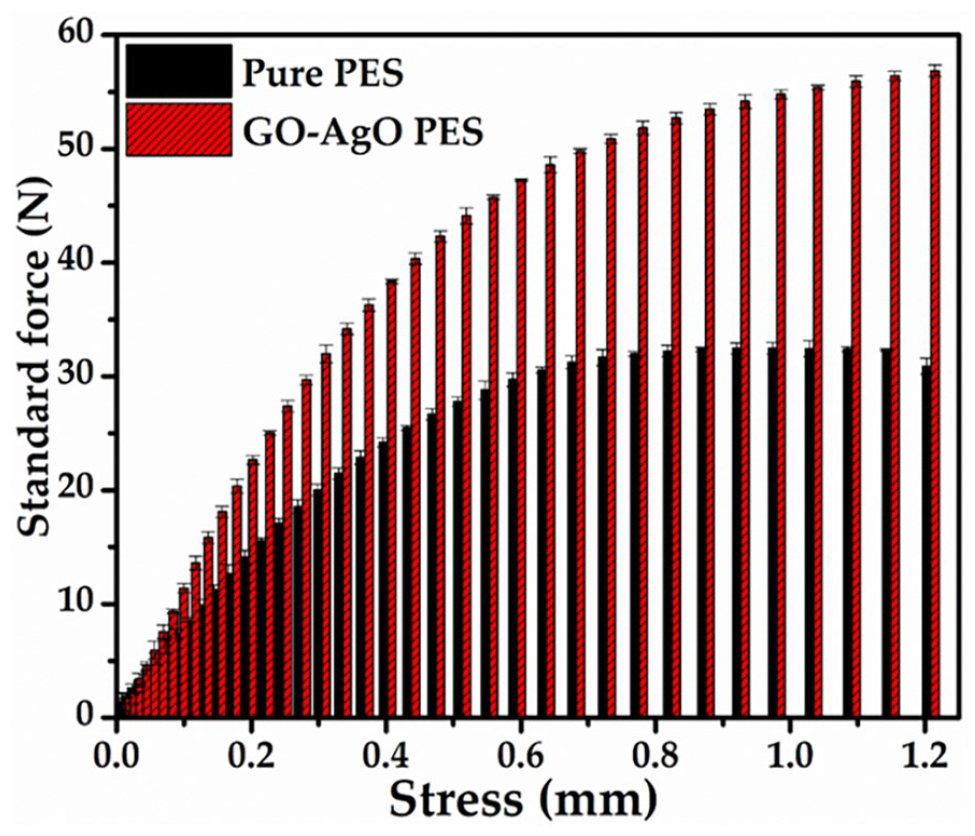
Typical graph for the mechanical strength of the membranes.
Furthermore, an exhaustive series of tests, encompassing abrasion, folding, membrane soaking in water, and drying, were conducted to assess the robustness and resilience of the fabricated membrane. The results yielded exciting outcomes, affirming the membrane’s durability and structural integrity. As depicted in Figure 5, the membrane exhibited remarkable stability, retaining its original shape with no discernible wrinkles or cracks on its surface. To evaluate the membrane’s abrasion resistance, it was affixed to mesh sandpaper and subjected to a 100 g weight while undergoing 200 circular revolutions. The membrane demonstrated exceptional anti-abrasion capabilities, maintaining its structural and functional integrity throughout the rigorous test (Rameetse et al., 2020; Zhao et al., 2010). This impressive performance in the face of abrasion highlights the practical durability of the membrane, suggesting its potential suitability for real-world applications where mechanical wear and tear are significant considerations. These findings collectively underscore the robustness of the membrane, not only in maintaining its structural integrity but also in exhibiting resilience against abrasive forces. Such attributes are crucial for membranes intended for practical applications, particularly in industries where wear resistance is a critical factor for sustained performance. The results of these tests further bolster the potential of the fabricated membrane for deployment in challenging environments, solidifying its candidacy for various separation processes.

Images showing the mechanical stability tests of the prepared composite membrane.
Hydrophilicity, porosity and water permeability of the membranes
In evaluating the hydrophilic and water permeability characteristics of the membranes, key parameters such as contact angle and porosity play pivotal roles. The contact angle of the pristine PES membrane measured at 105° indicated its inherently hydrophobic nature. However, through the strategic integration of PEG and GO–AgO nanocomposites, a notable reduction in the contact angle to 68.6° was achieved, rendering the membrane significantly more hydrophilic, as depicted in Figure 6. This transformation correlates with an evident increase in water flux, ultimately fulfilling the essential requirement for efficient water filtration (Rameetse et al., 2020; Zhao et al., 2010). The incorporation of GO–AgO nanocomposites further augmented the water flux or permeability rate, escalating it from 0.782 Lm−2 hour−1 for the pure PES membrane to an impressive 5.86 Lm−2 hour−1, as calculated using Equation (2). This substantial enhancement underscores the positive impact of the nanocomposites on facilitating rapid water permeation through the membrane. Simultaneously, the membrane’s porosity experienced a significant boost, ascending from 62% for the pure PES membrane to an elevated 88% for the GO–AgO PES membrane. This increase in porosity further reinforces the membrane’s enhanced capacity for water permeability and filtration (Zhong et al., 2021). The developed GO–AgO PES membrane exhibits a substantial shift towards hydrophilicity, characterized by a decreased contact angle, heightened porosity and a remarkable increase in water permeability. These modifications collectively position the membrane as an efficient and effective solution for water filtration applications, promising enhanced performance and broader utility compared to the hydrophobic nature of the pure PES membrane.
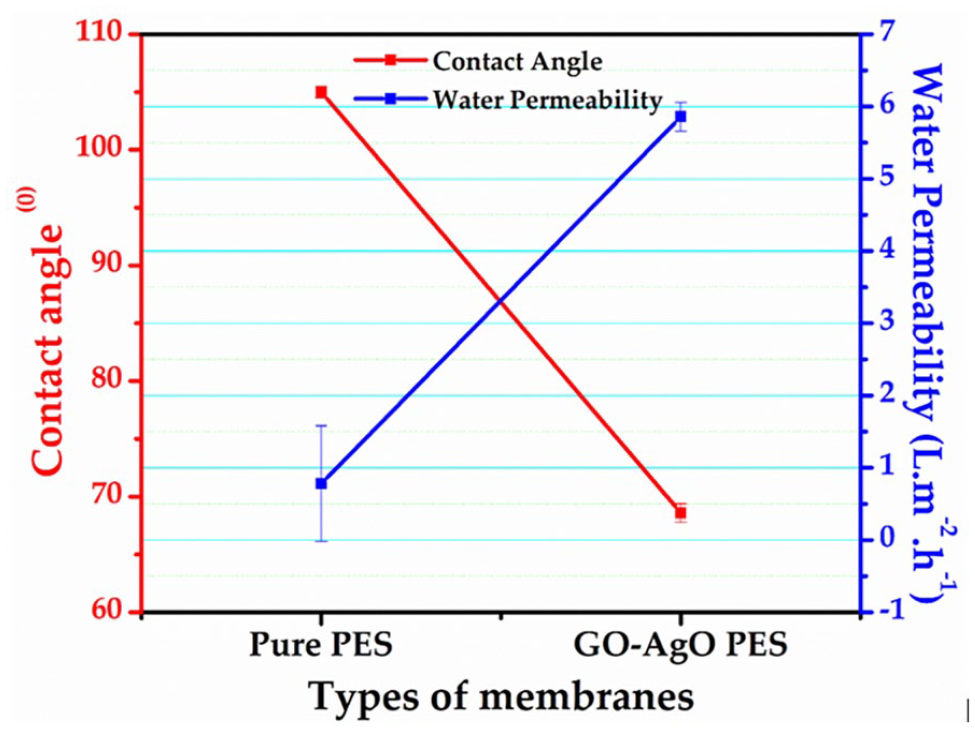
Graph showing the contact angle and water permeability of the membranes.
Antifouling test
The assessment of membrane fouling and its consequential impact on water flux rate is a critical aspect of membrane performance, as fouling contributes to increased hydraulic resistance, influencing membrane lifespan, efficiency and maintenance costs. The water FRR, calculated using Equation (4), serves as a quantitative measure of the membrane’s ability to recover its initial water permeability after encountering fouling challenges. Throughout consecutive filtration cycles, fouling-induced permeability decline was observed, particularly notable for the pure PES membrane, where the flux decrement reached approximately 83%. In stark contrast, the GO–AgO PES membrane exhibited a substantially lower flux decrement, ranging between 28% and 32% across filtration cycles. This significant mitigation of fouling-induced permeability decline underscores the remarkable antifouling properties inherent to the GO–AgO composite membrane. The superior resistance to fouling in the GO–AgO PES membrane can be attributed to the synergistic effects of GO and the membrane’s inherently hydrophilic nature. The heightened hydrophilicity of the composite membrane promotes the absorption of more water molecules, creating a hydration layer on its surface. This layer, in turn, serves as a deterrent against the adhesion of hydrophobic foulants to the membrane surface. The intricate interplay between the hydrophilic nature of the membrane and the unique characteristics of GO establishes a robust defence mechanism, resulting in prolonged efficiency and reduced maintenance costs (Shen et al., 2019; Tan and Rodrigue, 2019). The antifouling test substantiates the exceptional performance of the GO–AgO PES membrane, positioning it as a promising solution for sustained and efficient water filtration applications in the face of fouling challenges.
Mechanism behind oil–water emulsion separation
The notable efficiency exhibited by the composite membranes in oil–water emulsion separation is underpinned by a sophisticated mechanism driven by the strategic integration of GO–AgO nanocomposites into the membrane matrix. This infusion introduces polar functional groups, including –COOH, –C=O, =O and –OH, onto the membrane surface (Rameetse et al., 2020; Vatanpour et al., 2011) . These polar groups collectively render the membrane surface hydrophilic, imparting it with an enhanced ability to attract water molecules from oil–water mixtures. The hydrophilic nature of the membrane surface plays a pivotal role in the establishment of a hydration layer, a critical component in resisting fouling from oil particles. This hydration layer is formed due to the interaction of polar groups with water molecules, creating a barrier that impedes the adhesion of oil particles to the membrane surface. This mechanism is particularly effective in preventing the fouling that would otherwise compromise the membrane’s separation efficiency. Furthermore, electrostatic interactions and hydrogen bonding facilitated by the nanocomposite materials contribute to the formation of the hydration shell. These interactions enhance the stability of the hydration layer, fortifying its resistance against fouling from oil components. The collaborative effects of the hydrophilic membrane surface and the hydration shell create a selective barrier that allows water molecules to permeate through the membrane easily while impeding the passage of oil particles. The mechanism of oil–water emulsion separation involves the synergistic effects of the hydrophilic membrane surface, polar functional groups and the hydration layer, all orchestrated by the incorporation of GO–AgO nanocomposites. This intricate interplay results in the production of clear water filtrate, devoid of any oil content, underscoring the efficacy and selectivity of the composite membrane in addressing the challenges of oil–water emulsion separation (Rameetse et al., 2020).
Oil–water separation test
The oil–water separation test conducted using the specially crafted GO–AgO PES membrane showcased remarkable efficacy in separating five distinct oil–water mixtures, as illustrated in Figure 7. The experimental setup facilitated the successful filtration of 1.0 L oil–water mixtures through the membrane in seven consecutive cycles, with membrane washing with distilled water conducted after each cycle. The oil–water separation capacity, determined through Equation (3), revealed compelling results, affirming the membrane’s effectiveness in addressing diverse oil types within contaminated wastewater samples. In the initial filtration cycle, the modified membrane demonstrated exceptional separation capacities, achieving approximately 97% separation for Petrol, 95.6% for Hexane, 92% for mustard oil, 90% for coconut oil and 89% for other oils present in the wastewater sample (Figure 8). This high efficiency underscores the membrane’s capability to effectively mitigate the presence of varied oil contaminants, positioning it as a versatile solution for real-world oil–water separation challenges. However, as the filtration cycles progressed, the separation capacity experienced a marginal decline of approximately 3–4% after each cycle. This reduction can be attributed to the gradual blockage of pores or active sites on the membrane’s surface, impeding its optimal performance (Dmitrieva et al., 2022; Rameetse et al., 2020; Tan and Rodrigue, 2019; Zhao et al., 2010). This systematic evaluation of oil–water separation capacities reaffirms the practical utility and reliability of the GO–AgO PES membrane in addressing the complexities of oil–contaminated wastewater. The slight decrement in separation efficiency over successive cycles underscores the importance of periodic maintenance and cleaning to ensure prolonged and effective membrane performance in oil–water separation applications.

Image showing the setup used for separation of oil–water mixture.
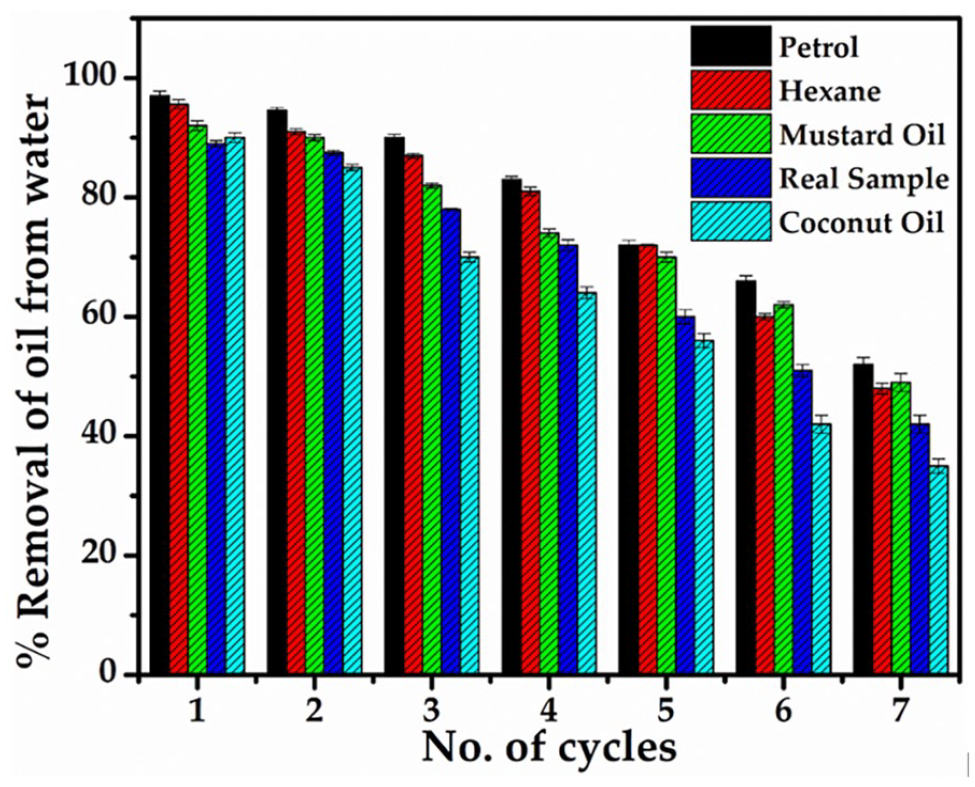
Graph showing the percentage removal of oil from oil–water mixtures.
Table 5 presents a comparative analysis of recently published works on oil–water separation, providing a comprehensive overview of various membrane types and their performance metrics. Notably, our fabricated GO–AgO PES PVP membrane stands out as an innovative and superior solution, exhibiting exceptional separation efficiency across multiple oil types and cycles. Several membranes, such as PVS + SiO2, PSF + bentonite and PVDF + vermiculite NP, have been previously investigated for oil–water separation. Although these membranes demonstrate satisfactory removal percentages, our GO–AgO PES PVP membrane surpasses them in terms of both efficiency and versatility. The fabricated membrane exhibits outstanding separation performance, achieving 97% removal for petrol, 95.6% for hexane, 92% for mustard oil and 90% for coconut oil, surpassing the efficiency of the previously reported membranes. Moreover, the GO–AgO PES PVP membrane showcases remarkable stability and durability, sustaining high performance over seven cycles, a notable improvement over membranes in the literature. This extended cycle durability is a critical factor in real-world applications, emphasizing the practical efficacy of our membrane.
A comparison table of some recently published work for oil water separation.
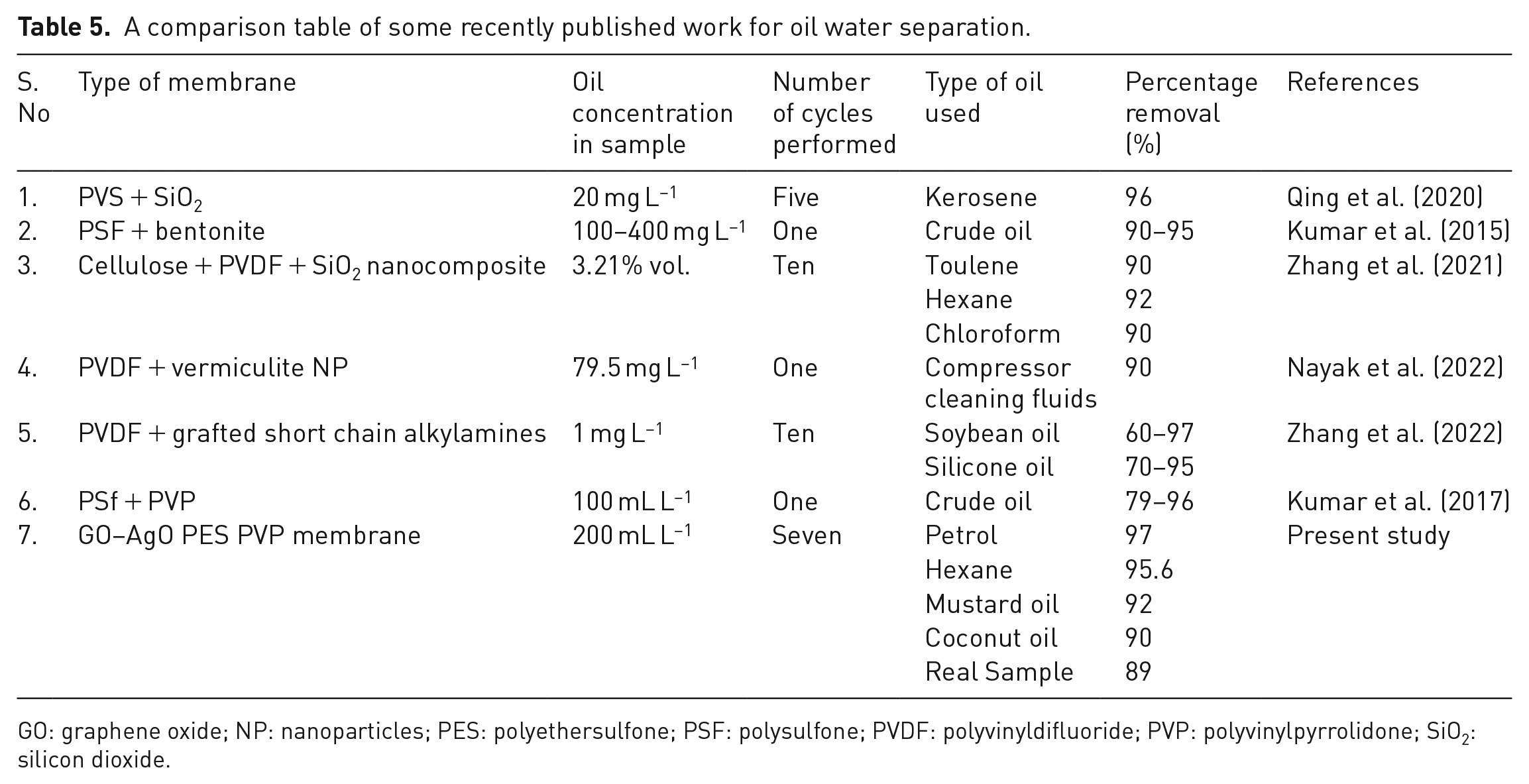
GO: graphene oxide; NP: nanoparticles; PES: polyethersulfone; PSF: polysulfone; PVDF: polyvinyldifluoride; PVP: polyvinylpyrrolidone; SiO2: silicon dioxide.
The comparison highlights the advantages of our fabricated membrane, including high water flux, robust strength and stability. These attributes, combined with excellent oil removal capacity across multiple cycles, position our membrane as an advanced and promising solution for oil–water separation. The meticulous design involving GO and AgO nanocomposites within a PES-polyvinylpyrrolidone matrix proves to be highly effective, offering a promising avenue for addressing challenges in wastewater management and environmental remediation.
The filtration experiments, as depicted in Figure 8, were meticulously conducted over seven cycles to assess the longevity and sustained performance of the fabricated membrane in oil–water separation (Tummons et al., 2020). It was observed that beyond the seventh cycle, the oil–water separation capacity declined below 50%, signalling a reduction in efficiency. Consequently, no further filtering cycles were performed, indicating a practical limit to the membrane’s continuous operational lifespan.
The specific challenges posed by crude coconut oil merit attention in this context. The observed lower efficiency in separating crude coconut oil is attributed to its inherent characteristics, including low viscosity. Crude coconut oil tends to form a dense layer or cake on the membrane’s surface, leading to the clogging of membrane pores. This phenomenon hinders the separation process and contributes to the early reduction in separation efficiency compared to other oil–water samples (Banchik, 2017; Tanudjaja et al., 2019).
The discerning analysis of filtration experiments underscores the importance of understanding the nature of the oil component in oil–water mixtures. Different oils exhibit distinct properties that can significantly impact the membrane’s performance, highlighting the need for tailored solutions based on the specific composition of the wastewater. The limitations observed with crude coconut oil offer valuable insights for further optimization of the membrane design to address a broad spectrum of oil–water mixtures with varying viscosities and compositions.
Conclusion
In summary, this study employed phase inversion technology to fabricate both pure PES membranes and PES membranes incorporated with GO–AgO nanocomposites. Comprehensive characterization was conducted utilizing various spectral techniques, revealing the uniform dispersion of AgO particles and the absence of aggregation on the surface of GO. Moreover, these nanocomposites were successfully integrated into the membrane matrix, leading to the characterization of composite membranes. This investigation clarified the substantial and promising influence of GO–AgO nanocomposites on membrane morphology, contact angle, hydrophilicity and mechanical strength. The incorporation of GO–AgO nanocomposites into the polymer matrix resulted in a multitude of improvements in membrane performance. Notably, the contact angle was significantly reduced from 105° to 68.6°, whereas water permeability, hydrophilicity and tensile strength experienced remarkable enhancements. The prepared membrane was then evaluated as a filtration medium for the separation of oil–water mixtures. The results demonstrated the remarkable efficiency of the modified membrane, which successfully separated approximately 97% of petrol, 95.6% of hexane, 92% of mustard oil, 90% of coconut oil and 89% of various oils from oil-contaminated wastewater samples in the first filtration cycle. Furthermore, the GO–AgO PES membrane exhibited minimal flux decrement when compared to the pure PES membrane. In conclusion, the GO–AgO-decorated PES composite membrane has demonstrated its potential as a highly effective and versatile separation medium for a wide range of oil–water mixtures. Its exceptional performance attributes, including enhanced hydrophilicity and reduced fouling tendencies, position it as a promising candidate for industrial-scale oil–water separation applications. This research contributes to the advancement of membrane technology and offers a sustainable solution for the pressing challenges associated with industrial wastewater treatment and oil spill remediation.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are thankful to the Deanship of Scientific Research at Najran University, Najran, Kingdom of Saudi Arabia for funding under the Research Group funding programme Grant no. NU/RG/SERC/12/36.


