Abstract
Since the majority of valuable components in spent lithium-ion batteries, such as lithium, exists in the electrode materials, common studies focused on the treatment of the cathode materials, which ignored the harm of residual electrolyte. The cavitation and thermal effects produced by ultrasonic can not only be used for the separation of electrode materials, but also have a wide range of applications in the field of sewage pollutant degradation. This work used ultrasonic to treat simulated electrolyte (propylene carbonate (PC)) solution of spent lithium-ion batteries, explored the effect of ultrasonic power, the addition amount of H2O2 solution (30 wt%) and reaction temperature on the degradation of electrolyte, and analysed the ultrasonic degradation reaction from the perspective of reaction kinetics. And the synchronous experiment of cathode material separation and electrolyte degradation was conducted under the optimal conditions. The results showed that the highest degradation efficiency of PC in the electrolyte was 83.08% under the condition of ultrasonic power of 900 W, the addition of H2O2 solution (30 wt%) of 10.2 mL, reaction temperature of 120°C and reaction time of 120 minutes, and the separation efficiency was 100%. This work reduced the environmental and health risks in the cathode material separation process and was conducive to the green development of spent lithium-ion battery recycling technology.
Keywords
Introduction
Since Sony Corporation of Japan took the lead in commercializing lithium-ion batteries (LIBs) in 1991, LIBs have been widely used in wireless communication equipment, portable instruments and other household and industrial fields due to their excellent electrochemical properties such as high specific energy, long cycle life and small self-discharge (Wang et al., 2016). As a result, production and demand for LIBs have been increasing. As the average life of LIBs is 1–3 years, the number of spent LIBs is bound to explode with the expansion of production. It is estimated that the output of spent LIBs in China will increase from 700 tons in 2012 to 464,000 tons in 2025, with an average annual growth rate of 59.0% (Hou, 2015). The resource benefits of spent LIBs are outstanding. The electrode materials usually contain 5–20% Co and 5–7% Li (Zhang, 2015), both of which are strategic resources with great recycling value (Gu et al., 2016). Therefore, the separation and resource utilization of spent LIB electrode materials has always been a research focus at home and abroad.
Compared with the research on the resource utilization of electrode materials, less attention has been paid to the treatment and disposal of the electrolyte. The mass fraction of organic electrolyte is about 3.5% in a spent lithium-ion battery. The electrolyte is mainly composed of organic solvent and lithium electrolyte. At present, carbonates are widely used as organic solvents in LIB electrolyte, including propylene carbonate (PC), ethylene carbonate (EC), diethyl carbonate (DEC), ethyl methyl carbonate (EMC), dimethoxyethane, etc. The most commonly used electrolyte lithium salts are LiPF6, LiBF4 and LiAsF6.
After repeated charging and discharging of LIB, most of the organic electrolyte diffuses to the electrode material or the membrane (Liu et al., 2014). If the electrolyte is treated improperly in the process of LIB resource utilization, it will seriously pollute the environment and endanger the health of people (Dai et al., 2013). At present, the treatment methods of spent LIBs electrolyte mainly include lye absorption, heat treatment and ultrasonic treatment. Li Fei used NaOH solution to absorb the electrolyte exposed in the process of dismantling the spent LIB, which removed the main fluoride and reduced the fluorine pollution caused by the volatilization of the electrolyte (Li, 2017). Although the lye absorption method is controllable, safe and efficient, it requires a large investment due to the need for liquid nitrogen freezing and crushing equipment. Under the conditions of 600°C, 30 minutes vacuum evaporation time and 1.0 kPa residual gas pressure, Sun et al. pyrolysed all the organic electrolyte in a vacuum pyrolysis device. The pyrolysis products were mainly fluorocarbon organic compounds (Sun and Qiu, 2011). The heat treatment method is practical and efficient, but the treatment process is complicated. If the conditions are not well controlled, the pyrolysis process is easy to produce toxic and harmful gases. Due to the variety, volatility, low content, complex composition, the downward trend of price, and the rapid development of electrolyte production technology, the recycling value of electrolyte of spent LIBs is low. Therefore, the focus of electrolyte treatment should be placed on degradation of organic matter.
Ultrasonic technology is an advanced oxidation technology with high efficiency and environmental protection. The conversion of ultrasonic energy is accompanied by physical and chemical changes. Related media will cause a series of effects under the action of ultrasound, such as mechanical effects, cavitation effects and thermal effects. These special physical and chemical effects have been widely used in many fields such as cleaning dirt, treating wastewater, sonochemical reactions and medicine (Cheeke, 2012; Shen, 2014). In the previous study by our group, ultrasonic technology has been applied to the separation process of spent LIB cathode materials. Under certain experimental conditions, the separation efficiency of LiCoO2 (a cathode material) reached 99% (Zhao et al., 2020). Besides, the special cavitation effect of ultrasonic can be applied to the degradation and removal of organic pollutants in wastewater, which is a new sewage treatment technology developed in recent years. It has simple operation, mild degradation conditions and fast degradation speed, with excellent development potential and broad application prospects (Yusof et al., 2016; Zhang, 2009).
Therefore, on the basis of ensuring the ultrasonic separation effect of the cathode materials of spent LIBs, the appropriate operating conditions of ultrasonic degradation of simulated electrolyte solution were investigated. This work explored the effect of ultrasonic cavitation technology on the degradation of organic pollutants in the separated liquid phase to achieve simultaneous separation of cathode materials and treatment of pollutants. Furthermore, the potential environmental pollution will be reduced in the process of separation and subsequent recovery, which will help optimize the recycling treatment and disposal process of spent LIBs.
Experiment
Materials
The electrolyte of spent LIBs used in this work mainly contains four organic substances which are DEC, EC, EMC and PC. Among these four organic substances, the structure of PC is the most complicated. Besides, considering from the molecular weight, hazard and degradation difficulty, PC is more harmful and difficult to degrade if it is exposed to the environment. Therefore, PC was selected as the simulated organic matter in this work.
The spent LIBs used in this work were obtained from a regular waste treatment company. All chemical reagents including hydrogen peroxide, ethyl acetate, PC, and dichloromethane were under analytical grade and purchased from Sinopharm Chemical Reagent Co., Ltd, and all solutions used in this work were prepared with deionized water.
Apparatus
The experimental reaction device was an ultrasonic reactor. It consisted of an ultrasonic generator, heating device, stirring device and the temperature control of the instrument.
Ultrasonic degradation of simulated electrolyte solution
Add 0.2 mL PC into 500 mL deionized water with a pipette to prepare simulated electrolyte solution. The solution was mechanically stirred for 1 minute to make it evenly mixed and poured into the ultrasonic reaction device. Then the ultrasonic reaction device was closed with reaction temperature programme and ultrasonic mode (work 5 seconds-stop 2 seconds) set, and the ultrasonic generator was powered on. The ultrasonic degradation experiments of simulated electrolyte solution were conducted to explore the effects of reaction time, ultrasonic power, the addition amount of H2O2 solution (30 wt%) and reaction temperature on the degradation of PC. Based on the ultrasonic degradation of simulated electrolyte solution, the synchronous ultrasonic experiment of cathode material separation and electrolyte degradation was conducted under optimal conditions. In order to ensure the ultrasonic separation effect of anode material of spent LIBs and prevent the aluminium foils from breaking, which may bring difficulties to the subsequent recovery, all experiments were carried out under a certain range of experimental conditions (reaction time ⩽ 100 minutes, ultrasonic power ⩽ 900 W, the addition amount of H2O2 solution (30 wt%) ⩽ 10.2 mL, reaction temperature ⩽ 120 °C).
Analytical methods
The concentration of PC in solution was measured by a gas chromatography mass spectrometer (GC-MS, Trace DSQII-MS).
The degradation efficiency of PC (
where
Results and discussion
Influence of factors on ultrasonic degradation of PC
Effect of reaction time and ultrasonic power
Ultrasonic power is a decisive factor for ultrasonic reactions. Only when the ultrasonic power reaches a certain level can the bubbles vibrating at the sound field frequency be closed. The higher the ultrasonic power, the faster the closing speed, the stronger the pressure wave generated, and the higher the temperature and pressure at the hot spots, and other cavitation effects will be more intense, which triggers and initiates a series of sonochemical reactions (Zhang et al., 2002). The experiments were conducted under the addition of H2O2 solution (30 wt%) of 5.1 mL and the temperature of 100°C. The effects of different reaction time (20, 40, 60, 80, 100 minutes) and different ultrasonic power (180, 540, 900 W) on the degradation efficiency of PC were explored and showed in Figure 1.
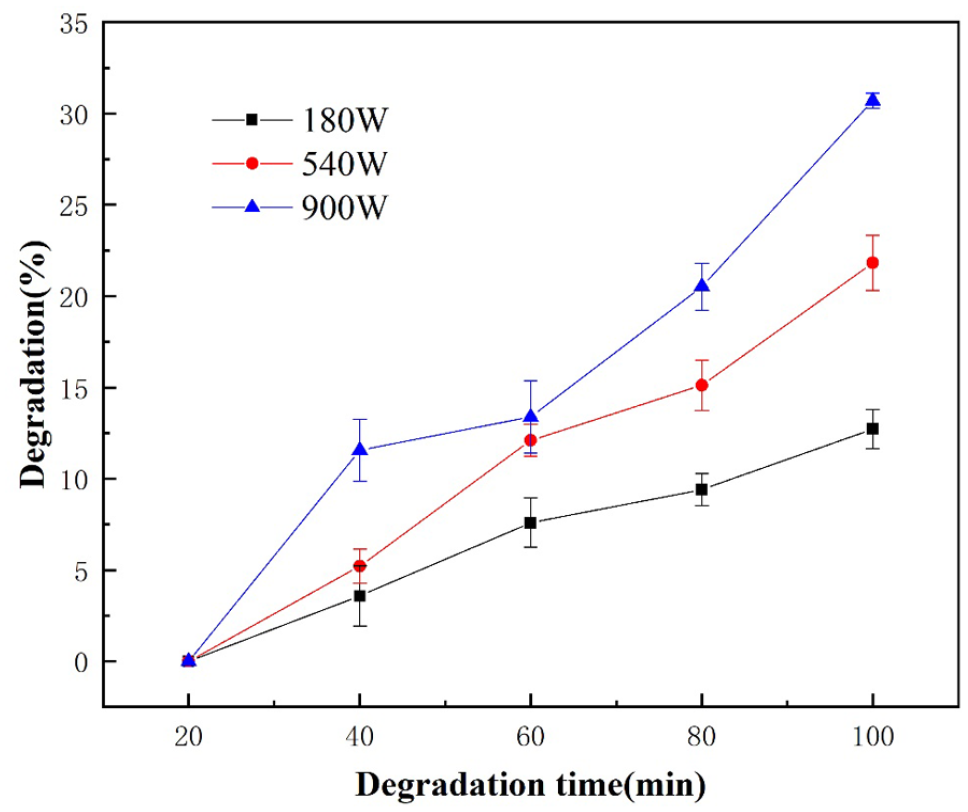
Effect of ultrasonic power on the degradation efficiency of PC.
Under the ultrasonic power of 180, 540 or 900 W, with the increase of reaction time, the degradation efficiency of PC gradually increased. It can be explained that with the increase of reaction time, cavitation bubbles were continuously generated and were in constant contact with PC in the solution, which promoted the degradation of PC and led to a linear increase in the degradation rate with the extension of reaction time.
Under the same reaction time, with the increase of ultrasonic power, the degradation efficiency of PC also increased. The results indicated that higher ultrasonic power had a greater effect on the degradation efficiency of PC.
When the addition of H2O2 solution (30 wt%) was 5.1 mL and the temperature was 100°C, the effect of ultrasonic power on the apparent reaction rate constant of ultrasonic degradation of PC was explored. The results of the fitting and regression of the PC degradation experimental data were shown in Figure 2.
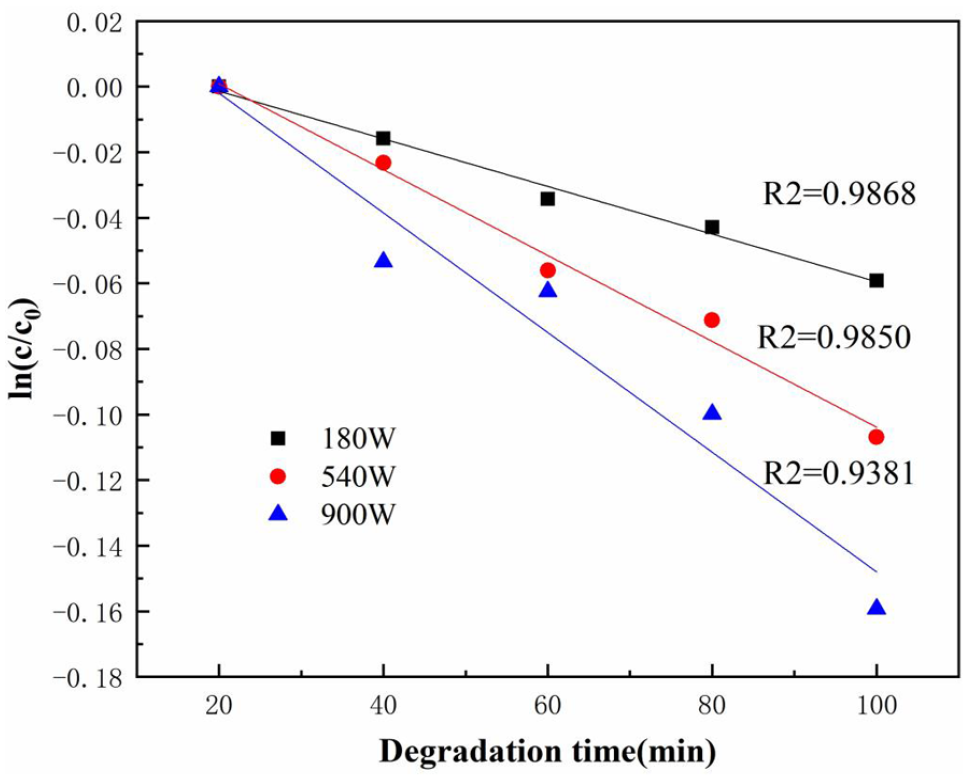
Fitting diagram of ln(c/c0) − t of the degradation of PC under different ultrasonic power.
According to the linear relationship of the diagram and the coefficient of determination R2, the ultrasonic degradation of PC followed a pseudo first-order kinetic reaction within the studied ultrasonic power range. Through linear regression calculation, the pseudo first-order apparent reaction rate constant increased with the increase of ultrasonic power. It can be explained that the increase of ultrasonic power led to an increase in cavitation energy, which lowered the cavitation threshold and the cavitation was more intense. The temperature and pressure in the cavitation bubbles increased correspondingly, resulting in more free radicals, which were more likely to act on PC, thereby increasing the ultrasonic degradation efficiency of PC (Liang, 2011; Liu, 2009). Since the degradation efficiency of PC was the highest under the ultrasonic power of 900 W, the subsequent experiments were conducted at this ultrasonic power.
Effect of the addition amount of H2O2
In order to improve the degradation efficiency of PC, H2O2 was added to the reaction system which had a synergistic effect with ultrasonic treatment. The addition of H2O2 can lead to an increase in the generation of ˙OH and other free radicals under the action of ultrasound, which may promote the degradation of organic matter (Zhao, 2008). Under the condition of ultrasonic power of 900 W and reaction temperature of 100°C, the effect of different addition amount of H2O2 solution (30 wt%) (0, 5.1, 10.2 mL) on the degradation efficiency of PC was explored and the results were shown in Figure 3.
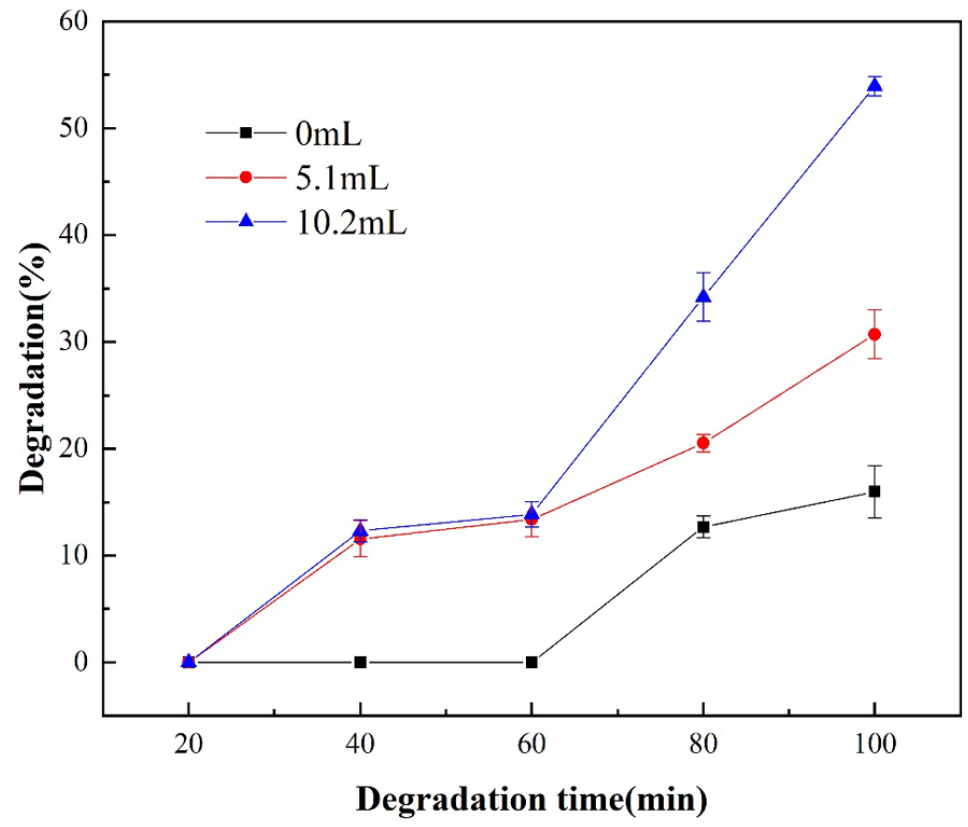
Effect of addition amount of H2O2 solution (30 wt%) on the degradation efficiency of PC.
With the increase of the amount of H2O2 solution added, the degradation efficiency of PC was significantly improved. When the reaction time was 100 minutes and the addition amount of H2O2 solution increased from 0 to 5.1 mL, the degradation efficiency of PC increased from 15.96 to 30.71%. When the addition amount of H2O2 solution continued to increase to 10.2 mL, the degradation efficiency reached 53.93%.
In order to further explore the mechanism of the promotion of the degradation reaction of PC by the addition of H2O2 solution, the effect of the amount of H2O2 solution added on the apparent reaction rate constant of ultrasonic degradation of PC was explored under the conditions of ultrasonic power of 900 W and the temperature of 100°C. The results of the fitting and regression of the PC degradation experimental data were shown in Figure 4.
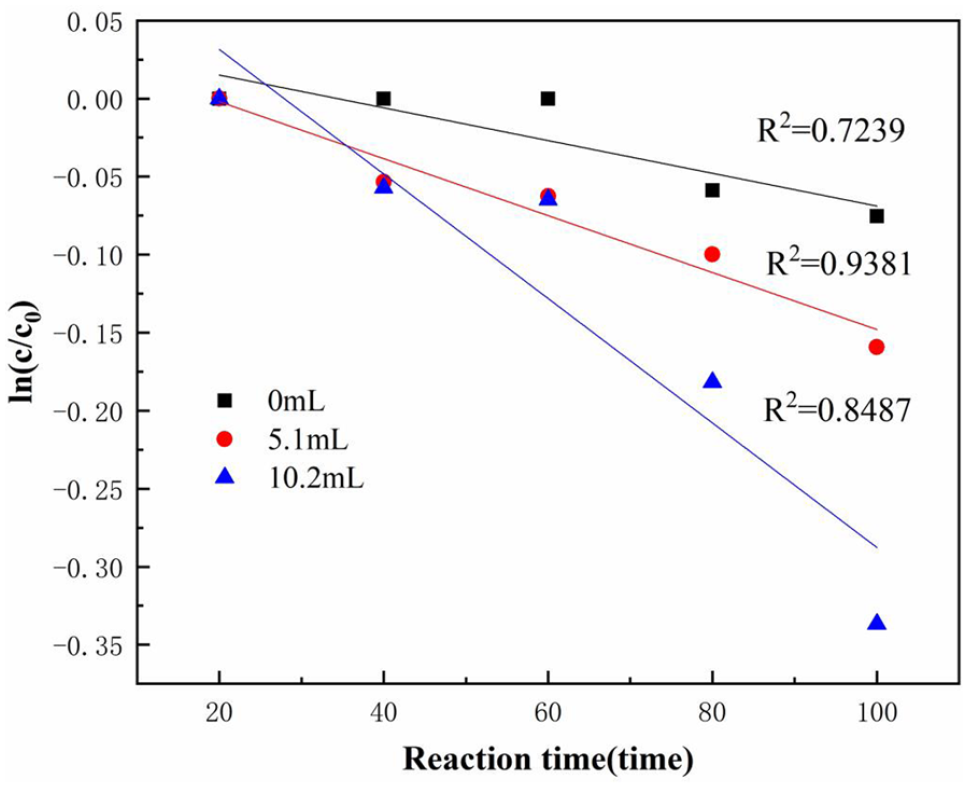
Fitting diagram of ln(c/c0) − t of the degradation of PC under different addition amount of H2O2 solution (30 wt%).
According to the linear relationship of the diagram and the coefficient of determination R2, the ultrasonic degradation of PC followed a pseudo first-order kinetic reaction within the studied addition amount range of H2O2 solution, and the pseudo first-order apparent reaction rate constant tended to increase with the increase of addition amount of H2O2 solution. It indicated that, in the ultrasonic reaction system, the increase in the amount of H2O2 solution added promoted the chemical reaction balance to advance in the direction of generating ˙OH, and strengthened the generation of ˙OH, which increased the concentration of highly oxidizing free radicals. The contact probability per unit volume between free radicals and PC increased, which promoted the PC degradation reaction, thereby increasing the degradation efficiency (Zhao, 2008).
Effect of reaction temperature
In the liquid phase reaction, high temperature can reduce the activation energy of the reaction and increase the activity of the reaction molecules, which can accelerate the rate of the degradation reaction. The experiments were conducted under the ultrasonic power of 900 W and the addition of H2O2 solution (30 wt%) of 10.2 mL. The effect of different reaction temperature (80°C, 100°C, 120°C) on the degradation efficiency of PC was explored and showed in Figure 5.
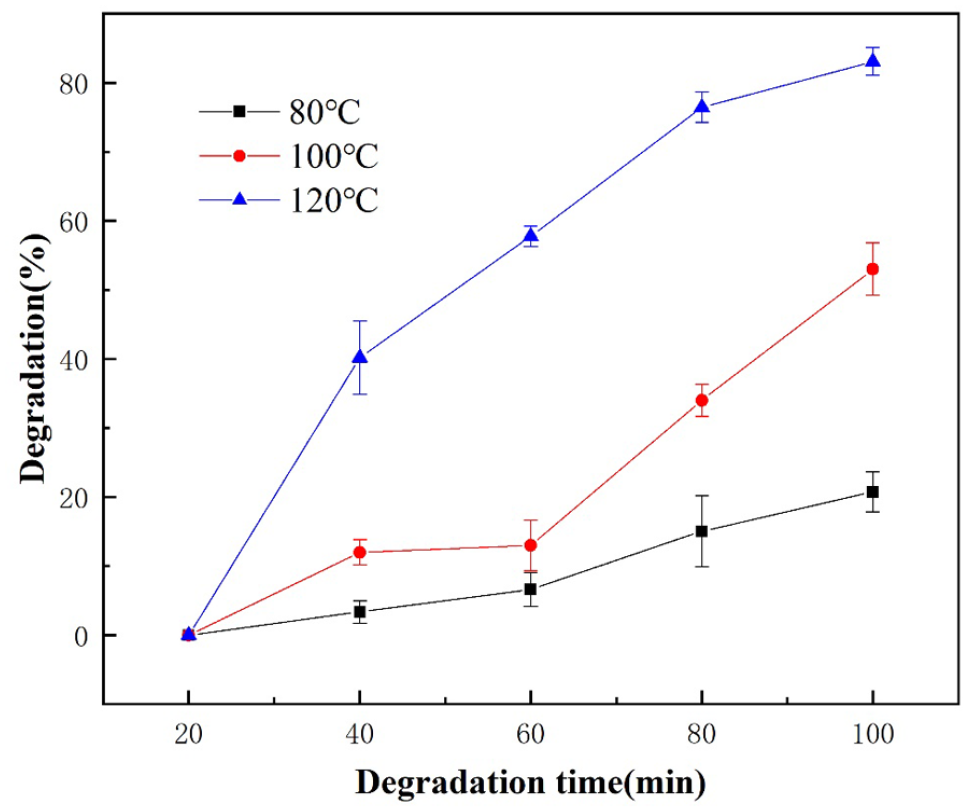
Effect of reaction temperature on the degradation efficiency of PC.
When the reaction time was 100 minutes and the reaction temperature increased from 80°C to 100°C, the degradation efficiency of PC increased from 20.73 to 53.93%. As the reaction temperature continued to increase to 120°C, the degradation efficiency reached 83.08%. The results showed that reaction temperature played an important role in the degradation of PC.
Under the conditions of ultrasonic power of 900 W and the addition of H2O2 solution (30 wt%) of 10.2 mL, the effect of the reaction temperature on the apparent reaction rate constant of ultrasonic degradation of PC was explored. The results of the fitting and regression of the PC degradation experimental data were shown in Figure 6.
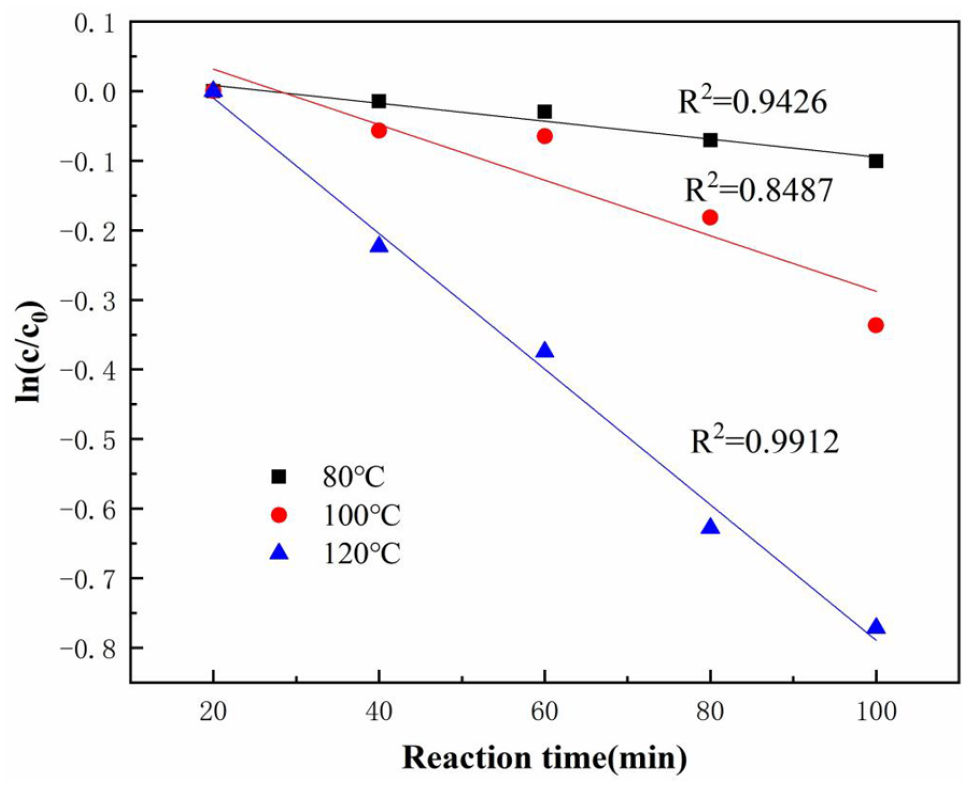
Fitting diagram of ln(c/c0) − t of the degradation of PC under different reaction temperatures.
According to the linear relationship of the diagram and the coefficient of determination R2, the ultrasonic degradation of PC followed a pseudo first-order kinetic reaction within the studied temperature range. Through linear regression calculation, the pseudo first-order apparent reaction rate constant increased with the increase of temperature. It can be explained that, in the ultrasonic/H2O2 oxidation reaction system, the increase of reaction temperature increased the number of activated molecules in the reaction system, which was beneficial to accelerate the rate of the PC degradation reaction.
The simultaneous experiment results of the cathode material separation and electrolyte degradation
Based on the results of the ultrasonic degradation of simulated electrolyte solution, the synchronous experiment was conducted under the optimal condition (ultrasonic power of 900 W, the addition of H2O2 solution (30 wt%) of 10.2 mL, reaction temperature of 120°C and reaction time of 120 minutes). Under the optimal condition of simulated electrolyte degradation, the active materials adhering to the cathode materials were completely peeled off, which was conducive to subsequent recovery. It indicated that separation effect was excellent. While the cathode materials were basically separated, the degradation efficiency of PC in the separated liquid phase reached 83.08%, which showed a significant degradation effect. According to the results, the effect of using ultrasound to synchronize the separation of cathode materials and the degradation of electrolyte was remarkable.
Although the recovery of electrode materials and valuable metals is still the focus of research, attention to electrolyte treatment has also been growing. Table 1 showed the treatment of spent lithium-ion battery electrolyte studied by other researchers in recent years.
List of the treatment of spent lithium-ion battery electrolyte.

LIBs: lithium-ion batteries; PC: propylene carbonate.
At present, the recovery of electrolyte still faces the problems of complex operation, high energy consumption, high cost and secondary pollution. Moreover, due to the low electrolyte content, low value and poor quality of recovered products, the recovery of electrolyte has not been suitable for the treatment trend in the future. The novelty of this work is that it focused on the removal of organic pollutants in electrolyte, especially the removal of organic solvent that are less studied at present. The advantages of the ultrasonic cavitation treatment adopted in this work include mild conditions, simple operation and environmental friendliness. The organic pollutants in the electrolyte can be degraded while ensuring the complete separation of the electrode materials. The long-standing pollution control in the recycling process of spent LIBs is solved without additional operations. This work is conducive to the technology development of spent lithium-ion battery recovery in the future.
Analysis of degradation mechanism
Ultrasonic chemical effect is caused by the direct interaction of ultrasonic waves with substance molecules. In a general solution system, the chemical reaction under the action of no sound field is determined by the chemical properties of the reactants, while the chemical reaction under the action of ultrasonic waves mainly comes from cavitation. In a general solution system, chemical reaction without a sound field is determined by the chemical properties of the reactants, while chemical reaction under the action of ultrasonic waves is mainly caused by cavitation. The cavitation effect means that the process of cavitation bubbles shrinking to collapse occurs rapidly in a tiny space, which can form local hot spots with abnormally high temperatures over 5000 K and instantaneous pressures higher than several hundred bar, and produce sonoluminescence, active free radicals, shock waves, high-speed microfluidics and other accompanying effects. The cavitation effect is the main reason why ultrasound plays a role in accelerating the rate of chemical reactions, increasing chemical yields, opening up new pathways of chemical reactions, and expanding the breadth of physicochemical reactions.
The ultrasonic chemical action in the aqueous reaction system of the degradation occurred in three regions, as shown in Figure 7. The first region was the core region of cavitation bubble. When the bubble collapsed inward, a high temperature of 5000 K and a strong pressure of 500 bar were generated in the gas phase in the bubble. Under this condition, the water molecules in the water vapour were split into ˙H and ˙OH. And ˙O were generated in the presence of oxygen and reacted with ˙H to form HO2˙. The second region was the gas–liquid interface region, where the temperature was relatively lower than inside of the bubble. However, the high energy accumulated at the interface caused pyrolysis of the non-volatile organics. The third region was the liquid phase region. The free radicals generated inside the bubble and at the gas–liquid interface diffused into this region and reacted with the medium in the solution.
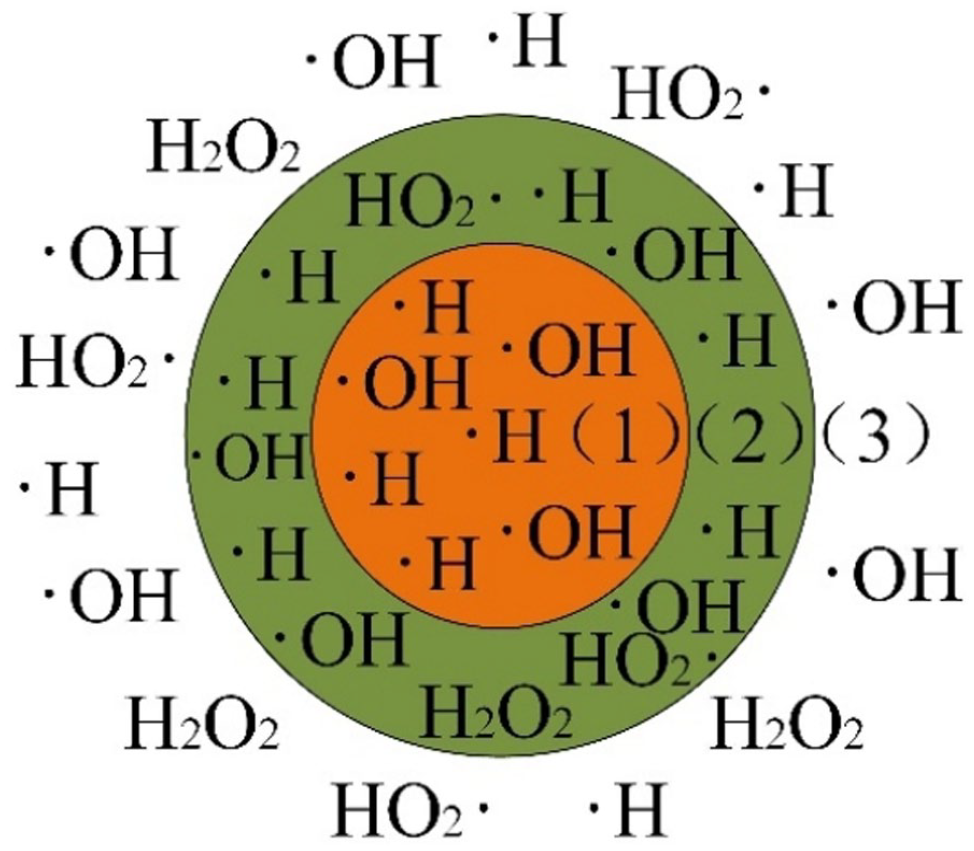
Schematic diagram of the ultrasonic reaction area: (1) core region of cavitation bubble, (2) gas–liquid interface region and (3) liquid phase region.
In the process of ultrasonic degradation of PC, the degradation and removal of PC was mainly the result of the combined action of free radical oxidation, high temperature pyrolysis and supercritical water oxidation. (1) Free radical oxidation: Water vapour or other gases in cavitation bubbles were decomposed into free radicals such as ˙H and ˙OH under extreme conditions of high temperature and high pressure. In addition, O2 in the bubbles was decomposed to generate ˙O. The free radicals combined and reacted with each other to generate various strong oxidants, which greatly enhanced the hydrothermal oxidation of PC. (2) High temperature pyrolysis: The high temperature and high pressure region generated by ultrasonic cavitation broke the chemical bonds of PC. And organics underwent a pyrolysis reaction similar to water phase combustion inside the cavitation bubbles and at the gas–liquid interface. (3) Supercritical water oxidation: Due to the high temperature and high pressure generated by ultrasonic cavitation, the temperature and pressure of the water molecules on the surface of the cavitation bubbles reached the critical temperature of water of 647 K and the critical pressure of 21 bar (Cai, 2015). At this time, the gas–liquid interface disappeared and became a homogeneous phase system. The reaction solution had high material transport and diffusion capacity, and the chemical reaction rate was extremely fast. The oxidant such as free radicals reacted quickly with PC to realize the removal of PC.
Conclusions
The combined action of cavitation effect, mechanical effect and thermal effect induced by ultrasound can separate electrode materials. At the same time, the cavitation effect of ultrasound caused instantaneous high temperature and high pressure at hot spots in the liquid medium, which can further generate strong oxidizing free radicals in the water medium and degrade the residual electrolyte in the electrode materials to control the secondary pollution caused by the electrolyte in the resource utilization process of spent LIBs. Under the optimal condition of ultrasonic power of 900 W, the addition of H2O2 solution (30 wt%) of 10.2 mL, reaction temperature of 120°C and reaction time of 120 minutes, the degradation efficiency of PC in the electrolyte was 83.08%, and the active materials adhering to the cathode materials were completely peeled off, which meant that cathode material separation and electrolyte degradation could be carried out simultaneously and the effect was significant. The whole process realized separation of valuable components and degradation of toxic components, which was beneficial to optimize the recycling process of spent LIBs, reduce environmental risks and human health risks, and it has certain industrial and environmental benefits.
Footnotes
Acknowledgements
The authors greatly appreciate the valuable comments from editor and anonymous reviewers.
Author’s Note
Shuguang Zhu is also affiliated to Engineering Research Center of Building Energy Efficiency Control and Evaluation, Ministry of Education.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the research project of urban water environment security system in north plain of Anhui province (21H0169-DG).

