Abstract
Various products made from biodegradable polymers have been increasing rapidly in the market since the use of non-biodegradable materials has been banned, particularly for the disabled packaging materials. Burning remains the most popular method that is increasingly used in treating city wastes. The impact of these polymers on environmental during thermal degradation and combustion is an important issue for city waste management. In this work, the thermal degradation and combustion behaviours of the most popular synthetic biodegradable polymers in the market, poly(lactide acid) (PLA), poly(e-caprolactone) (PCL), poly(butylene succinate) (PBS), poly(butylene adipate-co-terephthalate) (PBAT) and polyhydroxyalkenoates (PHA), are investigated. Both isothermal and non-isothermal thermal decomposition in oxygen and nitrogen environment were studied using thermogravimetric analysis combining with differential scanning calorimeter and coupled with Fourier transform infrared spectroscopy and gas chromatograph/mass spectroscopy. The combustion behaviour was investigated by a combustion colorimeter. The study results show that thermal degradation temperatures are PCL > PBS > PLA > PBAT > PHA. The thermal decomposition of all the polyesters started from scission reaction (cis-elimination), and then a stereoselective cis-elimination, which resulted in the formation of trans-crotonic acid and its oligomers. They all decomposed into CO2 and water in excess oxygen environment above 800°C. Various chemical products with smaller molecules were detected under oxygen-free conditions, including oligomers and unsaturated carboxylic acid. The order of the total heat release of the materials from high to low is as follows: PHA > PCL > PBAT > PBS > PLA. The combustion values of these polyesters are lower than those of polyolefins; thus, they will not damage furnace used currently. The results provide some important and useful data for managing these new city waste.
Introduction
Various products made from biodegradable polymers have been increasing significantly since the use of non-biodegradable materials has been banned for disabled packaging products in most countries of the world, especially in the last few years (Filiciotto and Rothenberg, 2021; Moshood et al., 2021; Rujnić-Sokele et al., 2017; Sanjay et al., 2021; Solis and Silveira, 2020; Zhu et al., 2020). Poly(lactide acid) (PLA), poly(e-caprolactone) (PCL), poly(butylene succinate) (PBS) and poly(butylene adipate-co-terephthalate) (PBAT), polyhydroxyalkanoates (PHAs) or poly[(R)-3-hydroxybutyrate] (PHB) are typically well known as the most popular synthetic biodegradable polymers in the market so far. Theoretically, these synthetic biodegradable polymers are all aliphatic polyesters, which are biodegradable during industrial composting. However, combustion is still one of the most popular and increasing methods used to treat city wastes, particularly in the developed countries or cities. In 2019, 85% plastics were recycled in Japan, in which 60% was combusted (PWMI, 2021). Guangzhou, one of the biggest cities in China, has 16 combustion stations and treated its entire wastes (3000 tonnes/day) in 2019 (Li and Wang, 2022). As these biodegradable polymers are new resources of city wastes, to control the burning process and the thermal decomposed products, it is important to investigate their pyrolysis and combustion behaviours (Kopinke et al., 1996a, 1996b; Wachsen et al., 1997; Signori et al., 2009). Thus, the environmental issues of these polymers, especially during thermal decomposition, should be considered even they are biodegradable.
Polymers can be degraded under oxygen-free or oxygen conditions, which are, respectively, called pyrolysis and combustion. Their final products are significantly different. The pyrolysis and combustion properties of the most popular polyolefins (such as polyethylene (PE), polypropylene (PP), polystyrene (PS), polyvinyl chloride, etc.) have been extensively studied (Antoniou and Zorpas, 2019; Grammelis et al., 2009; Ławi’nska et al., 2022; Silvarrey and Phan, 2016; Soni et al., 2021; Sorum et al., 2001; Zhang et al., 2021). Thermal degradation of polyesters has attracted great attention since they are not as thermal stable as polyolefins, and they can be degraded during storage, processing, applications and recycling (Al-Itry et al., 2012; Duan et al., 2019; Gao et al., 2014; Khalid et al., 2017; Meng et al., 2019). Most of the previous work mainly focused on thermal degradation under oxygen-free condition since the processing conditions are normally oxygen-free. Practically, thermal degradation is very important for all the biodegradable polyesters and has been extensively studied (Kopinke et al., 1996a, 1996b; Signori et al., 2009; Wachsen et al., 1997). For example, Aoyagi et al. (2002) have studied and compared the thermal degradation of PHB, PCL and PLA under oxygen-free conditions. These three polyesters showed quite different time-dependent profiles of weight loss and were averagely polymerized under isothermal condition, reflecting their different thermal stability and degradation mechanisms. Thermogravimetric analysis (TGA) and pyrolysis coupled with gas chromatograph/mass spectroscopy (GC/MS) show that PHB is degraded by a random chain scission (cis-elimination), while PCL is degraded by an unzipping depolymerization from the hydroxyl end of the polymer chains. In contrast, the thermal degradation behaviour of PLA was extraordinarily complex because various reactions occurred concurrently. Lu et al. (2012) studied the thermal degradation of PBS under nitrogen condition using TGA-Fourier transform infrared spectroscopy (FTIR). FTIR spectra revealed that the major products were anhydrides, which were formed via two cyclic intramolecular degradation mechanisms by breaking of weak O–CH2 bonds around succinct groups. More recently, Li et al. (2020) studied the effect of Halloysite nanotubes (Hal) on the thermal degradation performance of PBAT. It was found that Hal catalysed the generation of aromatic compounds during decomposition and the consecutive dehydrogenation reaction of carbonization to char.
On the other hand, the thermal degradation of polymers in oxygen environment normally happens during burning. An example is the combustion of city wastes (Vilardi and Verdone, 2022; Voukkali et al., 2021). It is expected that most of the final products of combusted carbon–hydrogen–oxygen materials should be CO2 and HO2. The thermal degradation of various biodegradable polymers in oxygen environment has been reported previously (Al-Itry et al., 2012; Persenaire et al., 2001; Rizzarelli and Carroccio, 2009). It was confirmed that most of the products of combusted these materials are CO2 and HO2. However, there is often oxygen deficit during burning, especially during the burning of unpredictable city wastes. Thus, it is necessary and important to investigate the thermal degradation of these polymers in both oxygen and oxygen-free environments.
It is expected that the products obtained from burning these biodegradable polyesters with city wastes should be a mixture and complex (Grammelis et al., 2009; Silvarrey and Phan, 2016; Soni et al., 2021). Furthermore, combustion value is very important in designing a furnace and controlling the burning process. The combustion value is reasonably high for most of the polyolefin-based polymers since they are all made of petrol-based products (Grammelis et al., 2009; Silvarrey and Phan, 2016). The combustion value of the city wastes is generally higher in developed countries or cities since it contains more packaging materials made from plastics and papers (Sarakikya and Kiplagat, 2015). The combustion value of city wastes could be 4500–10,000 kJ/Kg. With the increasing combustion value of city wastes, the damages caused by furnace have become a popular problem for city waste treatment (Chen et al., 2013; Mastral et al., 1999). It is important to evaluate these new resources in the city waste.
In this work, the pyrolysis and combustion behaviours of the most popular synthetic biodegradable polymers, PLA, PBS, PBAT, PCL, PHA, are systematically investigated and compared. Both isothermal and non-isothermal thermal conditions in oxygen and oxygen-free environment were studied using TGA coupled with FTIR and GC/MA, as well as differential scanning calorimeter (DSC). The combustion values were measured by a combustion calorimeter. The results will provide useful data not only for application conditions (thermal stability), but also for post-treatments (burning with city waste).
Materials and methods
Materials
All the biodegradable polymers used in this work are commercially available. PBAT (C1200) was purchased from BASF (Ludwigshafen, Germany); PLA (3001D) was obtained from Nature Works (Blair, Nebraska, USA); PHAs (1005) was purchased from Mirel (USA); PCL (6800) was purchased from Perstorp (Malmö, Skåne län, Sweden); PBS (TH803S) was purchased from Xinjiang Blue Ridge Tunhe Co. Ltd. (Changji, Xinjiang, China). Before all the materials were used, they were dried in a vacuum oven at 60°C for 12 hours to remove free moisture containing in the samples.
TGA and DSC
A TGA-DSC analysis (STA-8000 calorimeter, PerkinElmer, Waltham, MA, USA) system was used to investigate the thermal properties of the biodegradable polyesters. The samples with about 7 mg were placed in a sample pan and heated from 30 to 800°C by 10°C/min in nitrogen and oxygen atmosphere, respectively. Derivative thermogravimetry (DTG) peak weas used to represent decomposition or pyrolysis temperatures.
TGA with FTIR
The TGA-FTIR analysis was performed using simultaneous TGA (TGA STA8000, PerkinElmer) coupled with FTIR spectrometer (PerkinElmer). Approximately, 10 mg of the sample was heated under nitrogen and O2–N2 mixture gas flow rate of 20 mL/min from 30°C to 800°C at the heating rate of 20°C/min The evolved gases were transported to the FTIR spectrometer through a connecting line (heated at 250°C to prevent the evolved gases from condensing). The FTIR spectra were recorded at a resolution of 4.0 cm−1 from 4000 to 550 cm−1, and 16 scans were conducted for each spectrum. The thermogravimetric (TG) curve and the three-dimensional (3D) FTIR spectra were recorded. The chemicals from decomposed samples were analysed by FTIR.
TGA with GC/MS
Analysis of volatiles was also performed using GC-MS (Clarus SQ8; PerkinElmer) combined techniques. The temperatures of the lines and syringes were maintained at 200°C. About 10 mg of the sample was used in each test at a heating rate of 20°C/min under an atmosphere of high purity nitrogen. The temperature was set to increase from 30°C to 800°C, and the flow rate was controlled at 20 mL/min. Each sample was pyrolyzed at the temperatures corresponding to the maximum weight loss peaks of DTG, respectively. The chromatographic column used was Elite-5 ms (PerkinElmer) (0.25 μm × 0.25 μm × 30 m) and the maximum operating temperature was 350°C. The carrier gas was He with a purity of 99.999%. The flow rate was maintained at 1.0 mL/min, and the split ratio was 20:1. The column oven was heated from 50°C (for 2 minutes) to 300°C (for 5 minutes) at the heating speed of 10°C/min. The mass spectrometer was operated at 70 eV in EI mode. The mass-to-charge ratio was set at 45 to approximately 450, and the scanning rate was 0.002 seconds−1. The chromatographic peaks were analysed and identified by the NIST library and related literature.
Microscale combustion colorimeter
The combustion heat was measured using a microscale combustion colorimeter (MCC; Govmark, Mcc-2, Dayton, Ohio, USA), according to ASTM D7309-19a. Sample of about 5 mg was heated in a crucible under the nitrogen 80/oxygen 20 mixed gases environment from room temperature to 750°C at a heating rate of 1°C/s. Heat release capability (HRC), peak of heat release rate (PHRR), total heat released (THR) and temperature of maximum combustion (Tmax) were measured during the combustion process. Calorimetric measurements were performed in triplicate (using the same pattern sample) and reported as representing the mean value of the three experiments.
Results and discussions
Combustion behaviours
The combustion behaviours are very important for the treatment of burning city waste. The combustion behaviours of different biodegradable polymers were evaluated by a MCC. Figure 1 shows the heating release rate of the different biodegradable polymers measured by the MCC. The order of the combustion temperatures for the polymers, from high to low is as follows: PCL > PBS > PLA > PBAT > PHA, which corresponded with the results of DSC and TGA. It has been noticed that most combustion in oxygen environment finished below 450ºC. More detailed results of the combustion behaviours of the polymers are given in Table 1.
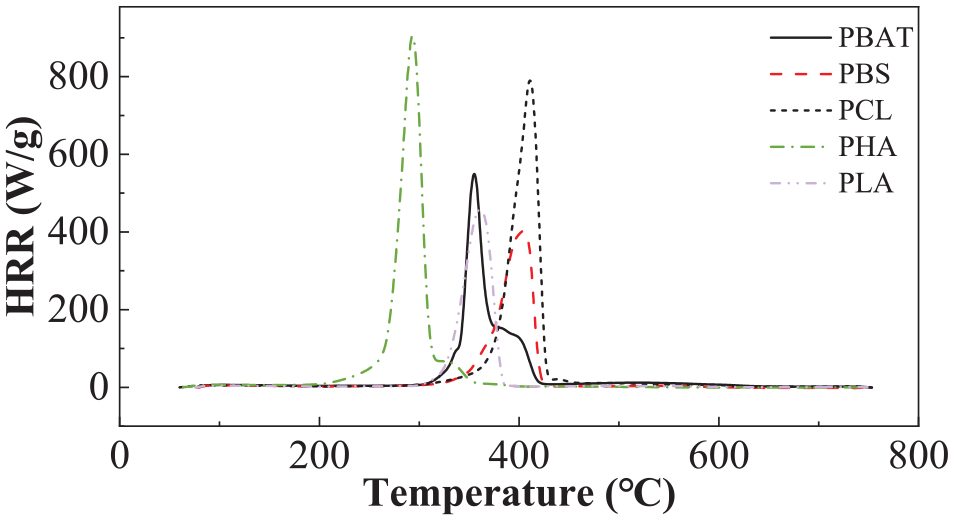
Heating release rate detected by MCC.
Combustion behaviours of the various materials.

HRC: heat release capability; PBAT: poly(butylene adipate-co-terephthalate); PBS: poly(butylene succinate); PCL: poly(e-caprolactone); PHA: polyhydroxyalkenoates; PHRR: peak of heat release rate; PLA: poly(lactide acid); THR: total heat release; Tmax: temperature of maxim combustion.
It is seen from Table 1 that PHA has the highest HRC, PHRR and THR, while PBS has the lowest during combustion. Their order from high to low is: PHA > PCL > PBAT > PBS > PLA. Higher THR means the material can release more energy during combustion, so PHAs have been considered as potential biofuel (Zhang et al., 2009). All of these biodegradable polyesters have reasonable low residues (<0.02%) after burning, indicating they all can be thermally decomposed completely at above 800°C.
Products from thermal decomposition and combustion
All the samples were heated in both nitrogen (oxygen free) and air (oxygen) environment using a TG facility. The thermal behaviours detected by DSC were also recorded during heating correspondingly. The TG was connected with a FTIR or GC/MS separately so that the gases produced during heating were characterized by FTIR and GC/MS, respectively. The purpose of this work is to focus on pyrolysis and combustion behaviours of the polymers; thus, their thermal properties at lower temperature, such as phase transitions (glass transition temperature Tg, and melting temperature Tm) were ignored since they have been extensively investigated for material processing and applications.
Poly(e-caprolactone)
The chemical structure of PCL is the simplest among the others since it is synthetized from the open ring polymerization of e-caprolactone. Figure 2 shows the thermal pyrolysis and decompositions of PCL in oxygen or nitrogen environment detected by DSC, TGA and FTIR. In Figure 2(A), only one endothermic peak at about 430.3°C was observed in nitrogen environment, indicating that the pyrolysis could be at one stage in oxygen-free conditions and the processing required energy. Two exothermic peaks at about 435.3°C and 567.4°C were observed during decomposition in oxygen environment; it is an oxidation (burning) process; thus energy is released. The two exothermic peaks showed that decomposition could be a two-step mechanism (Iwabuchi et al., 1976; Luderwand, 1977; Persenaire et al., 2001). The first step is a polymer chain cleavage via cis-elimination and the consecutive second step is an unzipping depolymerization from the hydroxyl end of the polymer chain. Both TGA and DTG curves in nitrogen and oxygen environment almost overlapped at about 428.3°C and 430.6°C, respectively (see Figure 2(a)), which corresponded with the endothermic peak measured by DSC in nitrogen environment. The single peak in DSC and DTG curves suggests a single-step degradation of PCL (Aoyagi et al., 2002). Persenaire et al. (2001) pointed out that the cis-elimination reaction and the unzipping depolymerization proceed sequentially at very close temperatures; hence, these two steps may not be resolved by a conventional DTG technique. The mechanisms of both one and two steps should be further clarified.
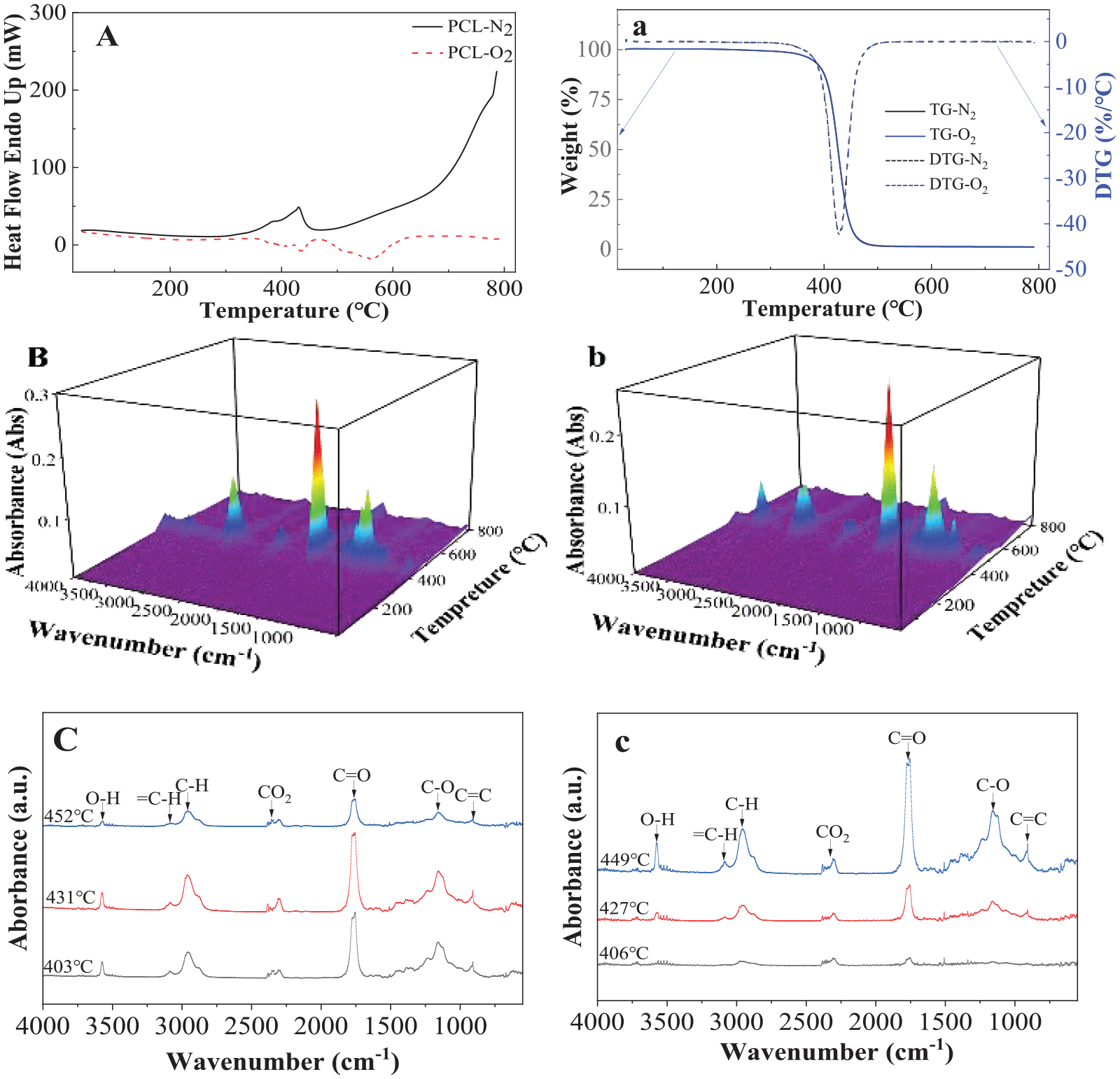
Thermal pyrolysis and decompositions of PCL detected by (A) DSC in nitrogen and oxygen environment; (a) TGA in nitrogen and oxygen environment, (B) 3D images of FTIR in oxygen environment; (b) 3D images of FTIR in nitrogen environment, and (C) FTIR curves measured at 406°C, 427°C and 449°C, respectively, in oxygen environment; (c) FTIR curves measured at 403°C, 431°C and 452°C in nitrogen environment.
Figure 2(B) and (b) shows 3D FTIR curves recorded during heating in oxygen and nitrogen environment, respectively. It is seen that no signal was detected below 350°C and above 500°C, indicating pyrolysis and decomposition in oxygen or nitrogen environment mainly happen in this temperature range. Figure 2(C) and (c) gives the specific FTIR curves at selected temperatures based on the highest peaks during decomposition and pyrolysis in oxygen and nitrogen environment. It is seen that the peaks detected at different temperature are almost the same, indicating the decomposition mechanisms are similar at different temperatures and environments. It has been noticed that with increasing temperature in the range between 350°C and 500°C, most of the peak heights decreased (gradually finishing) in oxygen environment but increased (starting) in nitrogen environment. That means pyrolysis and decomposition occur quicker in oxygen environment than in nitrogen environment, as expected.
Only one production peak of caprolactone was detected by GC/MS analysis (see Supplemental Scheme 1), which also supported a single-step degradation mechanism of PCL. Based on the above results, it can be concluded that pyrolysis in nitrogen environment is one-step mechanism, but decomposition in oxygen environment may be multi-step mechanisms.
Polyhydroxy alkenoates
Figure 3 shows the thermal decompositions and pyrolysis of PHA in oxygen or nitrogen environment detected by DSC, TGA and TGA-FRIR. DSC (see Figure 3(A)) shows that there is one sharp endothermic peak at about 300.1°C in nitrogen environment. Besides the two weak exothermic peaks at about 252.2°C and 531.4°C, there is also an endothermic peak at about 303.5°C observed during decomposition in oxygen environment. It is interesting to know that the endothermic peaks observed at around the same temperatures (300.1°C and 303.5°C) were detected in both nitrogen and oxygen environment. The endothermic peak could be responsible for the cyclization of PHA into a quasi-poly(benzoxazole) (PBO) structure and the second stage is the random scission of the PBO backbone (Zhang et al., 2003). Previous studies have shown that PHA has a two-stage thermal decomposition process, where the first stage corresponds to its cyclization into a quasi-PBO structure and the second stage is the random scission of the PBO backbone (Aoyagi et al., 2002; Zhang et al., 2003). PBO has been recognized as a thermal stable and flame resistance material (Wolfe et al., 1981) that needs energy to be decomposed even in oxygen environment.
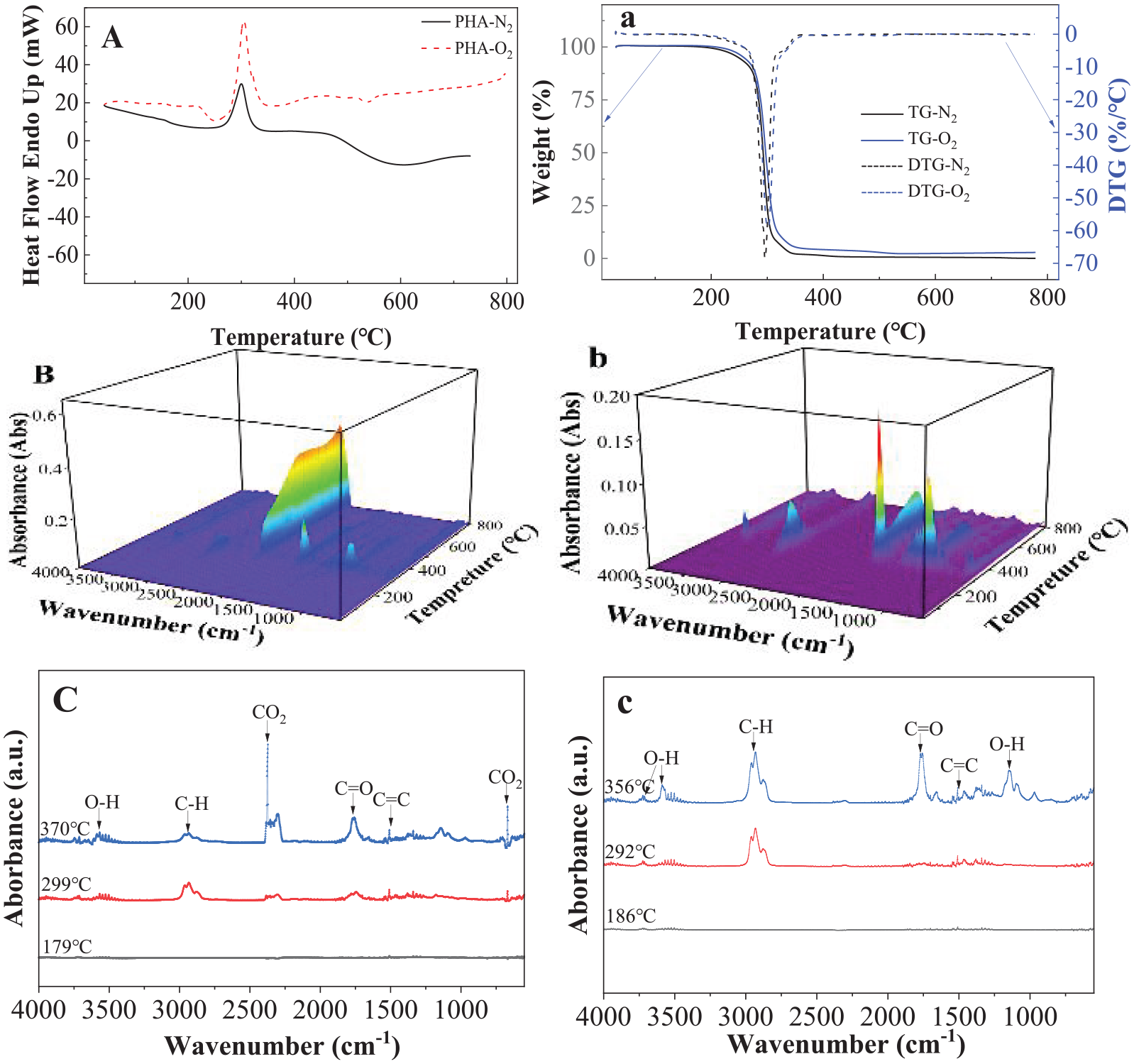
Thermal pyrolysis and decompositions of PHA detected by (A) DSC in nitrogen and oxygen environment; (a) TGA in nitrogen and oxygen environment, (B) 3D images of FTIR in oxygen environment; (b) 3D images of FTIR in nitrogen environment, and (C) FTIR curves measured at 179°C, 299°C and 370°C, respectively, in oxygen environment; (c) FTIR curves measured at 180°C, 292°C and 356°C in nitrogen environment.
Both TGA and DTG curves in oxygen and nitrogen environment almost overlapped and had only one peak at about 295.6°C and 302.6°C, respectively (see Figure 3(a)). The thermal degradation of PHB in nitrogen environment has been suggested to occur almost exclusively by a non-radical random chain scission reaction (cis-elimination); it involves a six-membered ring transition state (Galego and Rozsa, 1999). The dominant reaction pathway is a stereoselective cis-elimination that leads to the formation of trans-crotonic acid and its oligomers. Despite the extensive data reported in the literature, some aspects are not completely understood.
Figure 3(B) and (b) shows that the 3D FTIR curves recorded all the singles during heating in oxygen and nitrogen environment, respectively. It is seen that there are no signal detected below 200°C, indicating pyrolysis and decomposition in both nitrogen and oxygen environment mainly happened above this temperature. Figure 3(C) and (c) gives the specific FTIR curves at selected temperatures based on the highest peaks during pyrolysis and decomposition in oxygen and nitrogen environment. It is seen that CO2 peak dominated the products detected in oxygen environment, which indicates that PHA was fully burned. Some C–H and C–O were detected during pyrolysis in nitrogen environment. Most of the production detected by GC/MS (see Supplemental Scheme 2) appeared at one major peak, but multi-chemicals were identified. This shows that the processing is a non-radical random chain scission reaction.
Poly (lactide acid)
The thermal stability and decomposition of PLA have been extensively investigated, especially under oxygen-free condition since one of the weaknesses of PLA is its sensitivity to heat and moisture (Al-Itry et al., 2012; Aoyagi et al., 2002). Figure 4 shows the thermal decompositions and pyrolysis of PLA under both oxygen and nitrogen conditions detected by DSC, TGA, FTIR and GC/MS in this work. An endothermic peak at a temperature of 382.6°C was detected in nitrogen environment by DSC (see Figure 4(A)), which presented the pyrolysis of PLA. An exothermic peak at 385.2°C was detected in oxygen environment, which is the thermal decomposition or oxidation of PLA. It has been noticed that decomposition in both nitrogen and oxygen environment occurs almost at the same temperature, which means the pyrolysis and decomposition occurred at the same temperature and indicates that PLA could be a one-step decomposition mechanism (Badia et al., 2012; Cam and Marucci, 1997; Jamshidi et al., 1988). DTG (Figure 4(a)) shows that PLA lost about 97.2% and 99.2% weight at a temperature above 400°C under nitrogen and air conditions, respectively (see Figure 4(B)). This means there is some percentage (about 2%) of ash under nitrogen conditions. There is only one very close DTG peak at 372.1°C and 377.8°C under oxygen and nitrogen conditions, respectively. The single decomposition peak indicates that PLA could be a one-step decomposition mechanism, whether under oxygen or oxygen-free conditions.
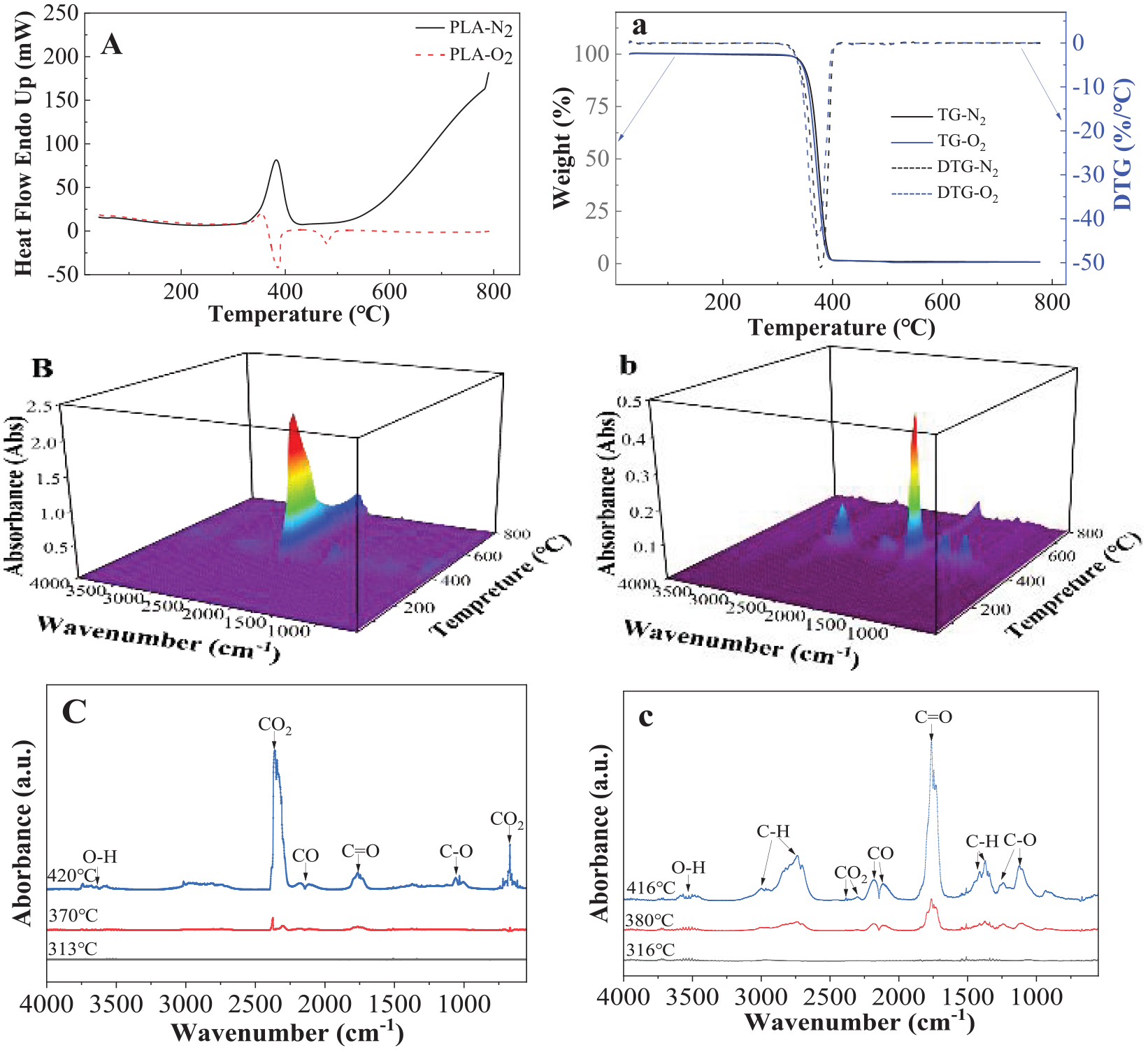
Thermal pyrolysis and decompositions of PLA detected by (A) DSC in nitrogen and oxygen environment; (a) TGA in nitrogen and oxygen environment; (B) 3D images of FTIR in oxygen environment; (b) 3D images of FTIR in nitrogen environment; (C) FTIR curves measured at 313°C, 370°C and 420°C, respectively, in oxygen environment; (c) FTIR curves measured at 316°C, 380°C and 416°C in nitrogen environment.
Figure 4(C) and (c) shows the FTIR spectra of the PLA decomposed and pyrolyzed at different temperatures under oxygen and nitrogen conditions, respectively. It is seen that CO2 peak at 2400 cm−1 dominated the products decomposed in oxygen environment, while C=O is the main product pyrolyzed in nitrogen condition. Specific FTIR curves for the selected temperature show that there are a few decomposed small molecules below 320°C even though the polymer chains started the cis-elimination (Badia et al., 2012).
Multi-GC peaks were detected (see Supplemental Scheme 3) during decomposition in nitrogen environment and various chemicals were proposed by the NIST library. Moisture, hydrolysed monomers and oligomers, molecular weight and residual metals can affect the thermal stability and final products of PLA (Cam and Marucci, 1997; Jamshidi et al., 1988).
Poly(butylene succinate)
Figure 5 shows the thermal decompositions and pyrolysis of PBS in oxygen and nitrogen environment detected by DSC, TGA, TGA-FTIR and TGA-GC/MS. Figure 5(A) shows that there is only one endothermic peak at about 416.1°C in nitrogen environment. There are two exothermic peaks at about 431.0°C and 502.1°C, respectively, in oxygen environment. Rizzarelli and Carroccio (2009) reported oxidized polymer chains originating from the decomposition of hydroperoxide intermediate by radical rearrangement reactions (see Supplemental Scheme 4). The latter products subsequently underwent chain scission processes, which can be accurately traced from the chemical species identified. Georgousopoulou et al. (2016) also observed a radical reaction resulting in branching/recombination degradation reactions. There is only one DTG peak at 413.0°C and 393.5°C under oxygen and nitrogen conditions, respectively.
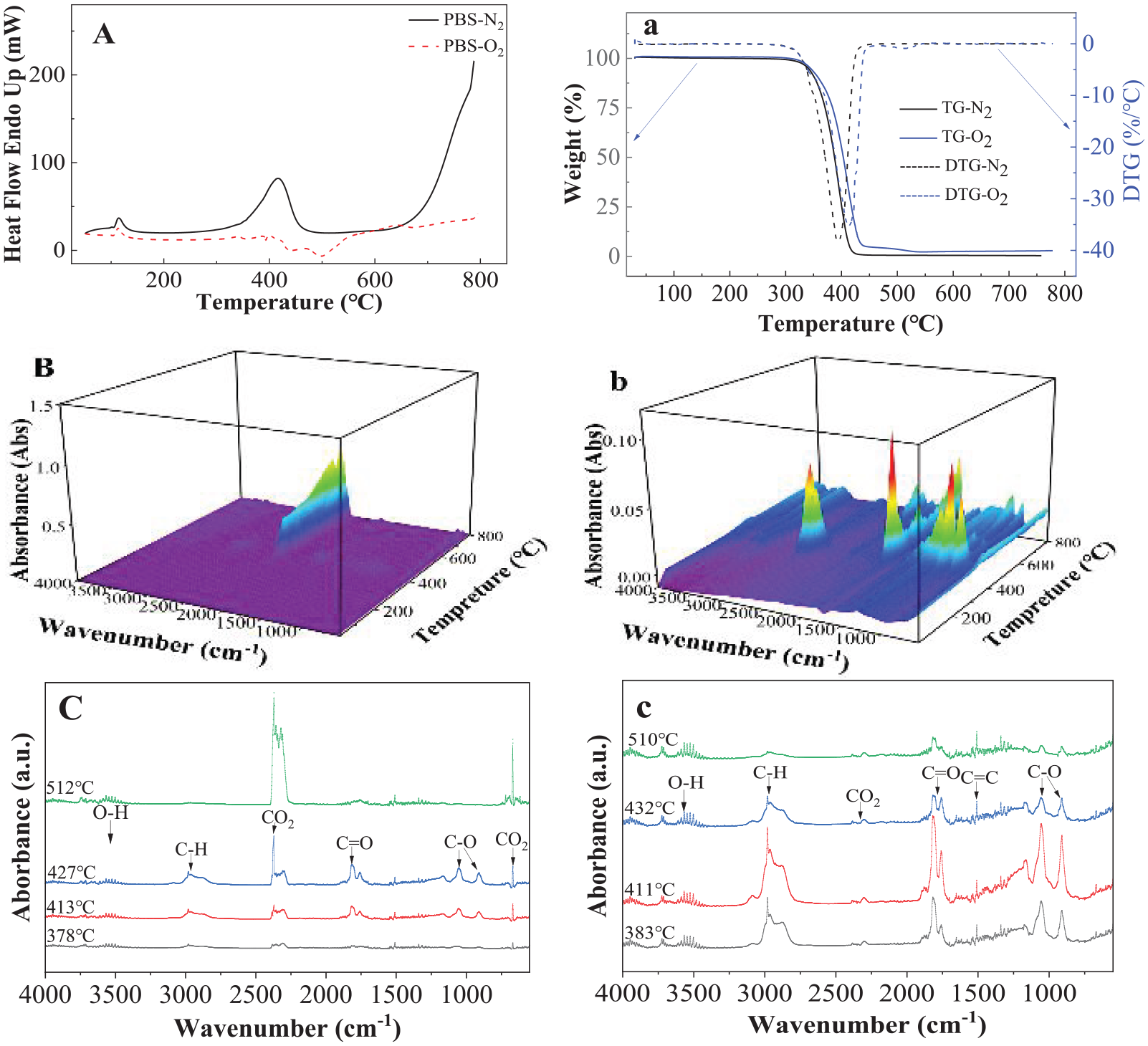
Thermal pyrolysis and decompositions of PBS detected by (A) DSC in nitrogen and oxygen environment; (a) TGA in nitrogen and oxygen environment, (B) 3D images of FTIR in oxygen environment; (b) 3D images of FTIR in nitrogen environment, and (C) FTIR curves measured at 178°C, 413°C, 427°C and 512°C, respectively, in oxygen environment; (c) FTIR curves measured at 383°C, 411°C, 432°C and 510°C in nitrogen environment.
Figure 5(B) and (b) shows the 3D FTIR curves that recorded all the singles during heating in oxygen and nitrogen environment, respectively. No peak was detected below 350°C. It is seen that CO2 peak at 2400 cm−1 dominated the products decomposed in oxygen environment, especially at higher temperature. Multi-peaks were detected in nitrogen environment, indicating PBS could be pyrolyzed complexly in nitrogen environment. Figure 4(C) and (c) gives the specific FTIR curves at selected temperatures based on the highest peaks during pyrolysis and decomposition. The CO2 peak at 2400 cm−1 in oxygen environment increased with increasing temperature. The multi-peaks detected at different temperatures in nitrogen environment showed mainly C–O and –OH.
Figure 5(D) shows the GC/MS curves of PBS decomposed in nitrogen environment with chemicals identified by the NIST library. It is seen that there is one GC peak detected at a very short time.
Poly(butylene adipate-co-terephthalate)
Figure 6 shows the thermal decompositions and pyrolysis of PBAT in oxygen and nitrogen environment detected by DSC, TGA, TGA-FTIR and TGA-GC/MS. It is seen from Figure 6(A) that there are two endothermic peaks at about 352.9°C and 421.3°C detected in nitrogen environment. There are some smaller exothermic peaks between 360°C and 450°C and big exothermic peak at about 521.3°C observed during decomposition in oxygen environment. Like PBS, the oxidized polymer chains originated from the decomposition of the hydroperoxide intermediate by radical rearrangement reactions. The latter products subsequently underwent chain scission processes and the radical reaction resulted in branching/recombination degradation reactions (Ibrahim et al., 2011; Iwabuchi et al., 1976). The multi-peaks at lower temperature are similar to those of PBS (see Figure 5(A)); the peak at higher temperature, 520°C is logically for the decomposition of aromatic co-polyester (terephthalic acid). Two DTG peaks at 366.5°C/370.7°C and 421.6°C/428.1°C were detected in oxygen and nitrogen environment, respectively. Obviously, this is due to the decomposition of aliphatic co-polyester (adipic acid and 1,4-butanediol) at lower temperature and the decomposition of aromatic co-polyester (terephthalic acid) at higher temperature.
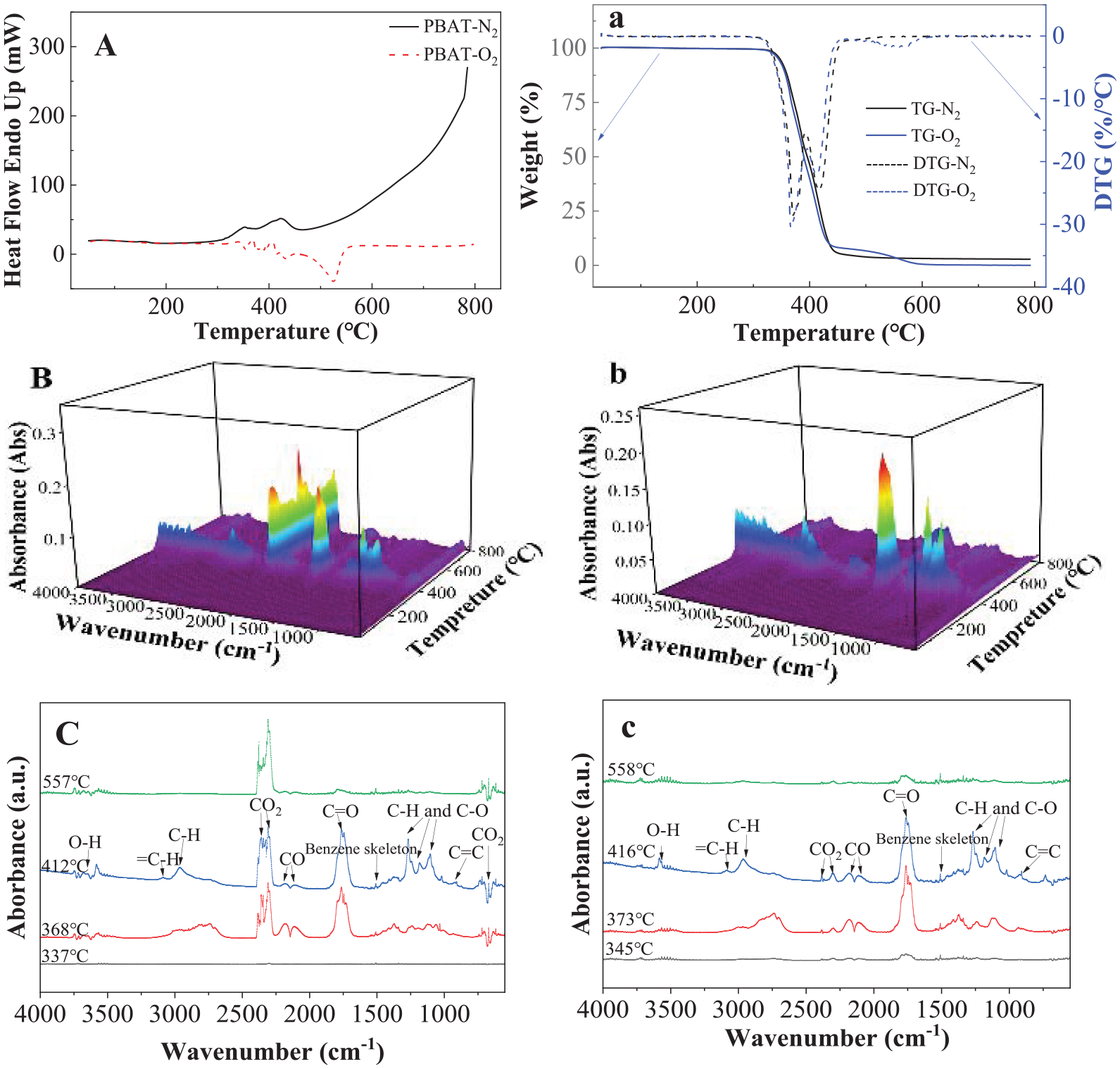
Thermal pyrolysis and decompositions of PBAT detected by (A) DSC in nitrogen and oxygen environment; (a) TGA in nitrogen and oxygen environment, (B) 3D images of FTIR in oxygen environment; (b) 3D images of FTIR in nitrogen, and (C) FTIR curves measured at 337°C, 368°C, 412°C and 557°C respectively in oxygen environment; (c) FTIR curves measured at 340°C, 373°C, 416°C and 558°C in nitrogen environment.
Figure 6(B) and (b) shows 3D FTIR curves recorded all the singles during heating in oxygen and nitrogen environment, respectively. Unlike PBS, multi-peaks were detected for PBAT in oxygen environment. Apart from the main CO2 peak observed at 2400 cm−1, multi-peaks for CO, C=O, CH, OH, etc. were also detected. Multi-peaks were observed in both nitrogen and oxygen environment (see Figure 5(b)). Figure 6(C) and (c) presents the specific FTIR curves at selected temperatures based on the highest peaks during decomposition and pyrolysis in oxygen and nitrogen environment, respectively. Multi-peaks were detected at different temperatures and each peak position was almost the same in both oxygen and nitrogen environment. However, CO2 at 2400 cm−1 is the highest peak in oxygen environment while C=O at 1750 cm−1 is the highest peak in nitrogen environment. Multi-peaks were detected by GC (see Supplemental Scheme 5), indicating the chemicals obtained from decomposition in nitrogen are much more complex. It is expected since the chemicals are obtained from the decomposition of both aliphatic and aromatic co-polyesters.
Discussion
The thermal degradation of neat polymer generally involves the initial decomposition, intermediate products recombination reaction and char formation process. The thermal decomposition mechanism of the polymers could follow one or more chemical mechanisms of random-chain scission, unzipping, chain-stripping and cross-linking. The thermal degradation of polyesters has been the subject of several studies and has been summarized as a special group. The basic reaction pathways are intramolecular ester exchange (transesterification) and cis-elimination (Garozzo et al., 1986; Kopinke et al., 1996; Signori et al., 2009). Some reviews are available on ester pyrolysis in the gas phase, which gives a comprehensive picture of structure–reactivity relationships for cis-elimination. The rates of ester pyrolysis by cis-elimination appear to be the same whether studied in pure liquid or gaseous state.
The above results have shown the thermal degradation and combustion behaviours of the most popular synthetic biodegradable polymers in the market, PCL, PHA, PLA, PBS and PBAT in oxygen and nitrogen environment. They were studied by TGA coupled with DSC and FTIR as well as GC/MA. Table 2 presents the summary of the pyrolysis and decomposition temperatures, and chemicals produced from the decomposition under nitrogen condition. The order of temperatures used for the polymers in both pyrolysis in nitrogen and decomposition in oxygen environment from high to low is as follows: PCL > PBS > PLA > PBAT > PGA. In most cases, the decomposition temperature in oxygen environment is lower than that in nitrogen environment. As expected, pyrolysis in nitrogen environment required energy while decomposition or oxygenation in oxygen environment released energy. It is interesting to know that an endothermic peak was detected by DSC in oxygen environment for PHA, which could lead to its cyclization into a quasi-PBO structure during decomposition.
Pyrolysis and decomposition temperatures, and chemicals obtained from decomposition.
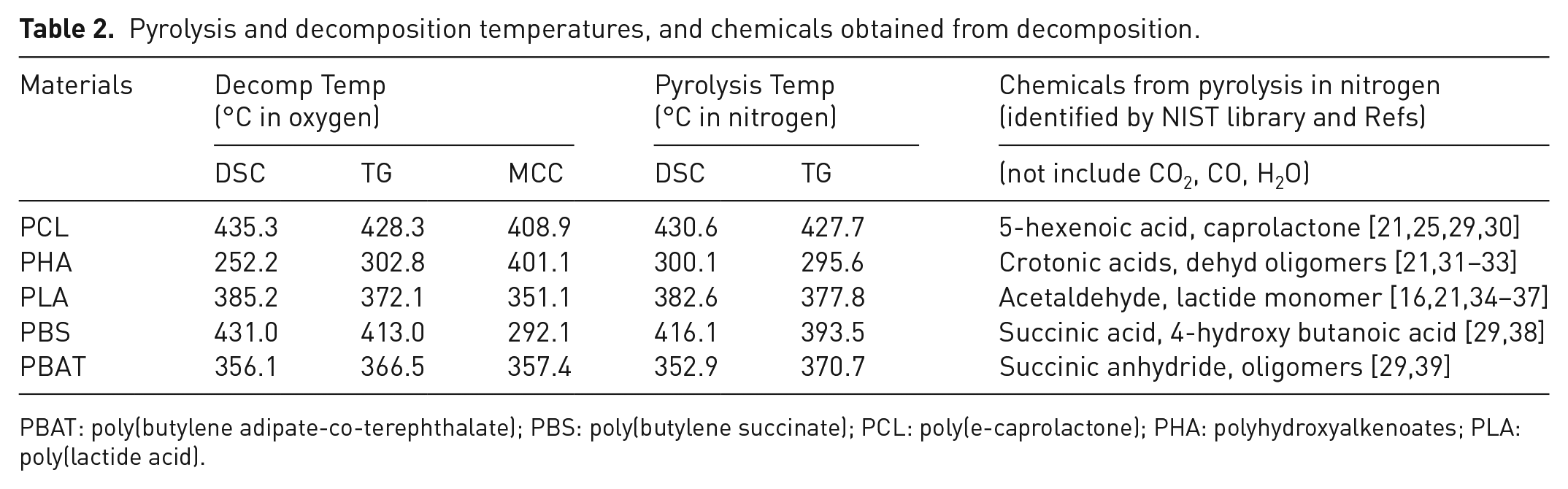
PBAT: poly(butylene adipate-co-terephthalate); PBS: poly(butylene succinate); PCL: poly(e-caprolactone); PHA: polyhydroxyalkenoates; PLA: poly(lactide acid).
The final products decomposed at higher temperature depended on the environment: oxygen or oxygen-free. Most of the chemicals produced in the oxygen environment are CO2 and water. Various small molecules were detected in nitrogen environment. These small molecules identified by the NIST library include oligomers, unsaturated carboxylic acid and CO2, CO, H2O, etc. There are no particular toxic chemicals detected or reported for the most of these biodegradable polyesters. The results indicated there is no particular toxic effect from burning these biodegradable polymers when enough oxygen is provided. All the THR of these biodegradable polyester is between 18 and 28 (see Table 1), which is similar to that of other polyesters PC (20.0), PET (15.5), PMMA (24.9) but lower than that of polyolefins PE (41.3), PP (40.9), PS (38.6) (Snegirev et al., 2012, 2014, 2017, 2019). Thus, they will not damage the conventional furnace used for burning city wastes. The order of HRC, PHRR and THR for the biodegradable polyesters from high to low is as follows: PHA > PCL > PBAT > PBS > PLA.
The results provide some important and useful data for managing these increased new city waste. This work also increased some knowledge in the area of waste management and research.
Conclusion
All these popular biodegradable polyesters are thermally degradable at high temperatures in both oxygen and nitrogen environment. The basic reaction pathways of the polyester thermal degradation are as follows: (a) intramolecular ester exchange (transesterification), which produces cyclic oligomers; (b) cis-elimination, which produces unsaturated carboxylic acid. The order of the thermal degradation temperatures used for these biodegradable polyesters, from high to low is as follows: PCL > PBS > PLA > PBAT > PHA. The final products decomposed at higher temperature depended on the environment: oxygen or oxygen-free. Most of the chemicals produced in the oxygen environment are CO2 and water. Various small molecules were detected in nitrogen environmental. These small molecules identified by the NIST library include oligomers, unsaturated carboxylic acid and CO2, CO, H2O, which confirmed previous reports. There are no particular toxic chemicals detected or reported for most of these biodegradable polyesters. The combustion value of these biodegradable polyesters is lower than that of most polyolefin-based conventional polymers. The order of HRC, PHRR and THR for the polymers from high to low is as follows: PHA > PCL > PBAT > PBS > PLA.
The results indicate that the new resources of city waste from biodegradable polymers will not increase toxic chemicals during burning treatment. Their combustion values are lower than conventional plastics, which means they will not damage the funeral system currently used. The future work should consider the effect of chemical reactions of these polyesters with conventional petrol-based plastics during burning treatment.
Supplemental Material
sj-docx-1-wmr-10.1177_0734242X221129054 – Supplemental material for Thermal degradation and combustion properties of most popular synthetic biodegradable polymers
Supplemental material, sj-docx-1-wmr-10.1177_0734242X221129054 for Thermal degradation and combustion properties of most popular synthetic biodegradable polymers by Hongmei Chen, Fengyi Chen, Hui Chen, Hongsheng Liu, Ling Chen and Long Yu in Waste Management & Research
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge research funds obtained from Natural Science Foundation of China (22178124), the 111 Project (B17018), Science and Technology Plan Project of Guangzhou (202102080150) and National Undergraduate Training Program for Innovation and Entrepreneurship of South China University of China.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
