Abstract
To promote the sustainable development of the liquor/ethanol industry and environment protection, alternative ways to dispose of anaerobic digestion residue (ADR) are urgently required. This research aims at studying the effects of different residence times (RTs) (30, 60 and 120 min) and heating rates (HR) (2.5, 5.0 and 10.0°C min-1) under 700°C on characteristics of ADR biochar as well as the optimization of ammonium (NH4+) adsorption. Results showed that, with the increasing RT and HR, the aromaticity as well as the content of fixed carbon and elemental carbon of ADR biochar increased, but the pyrolysis yield, volatile matter content, elemental hydrogen, oxygen and polarity decreased. Biochar prepared at 60 min and 5.0°C min-1 under 700°C presented the best development of orderly and honeycomb shape structures, highest specific surface area and maximal amount of NH4+ adsorption (3.15 mg N g-1). The multilayer heterogeneous adsorption process dominated the NH4+ adsorption behaviour. And the maximal amount of NH4+ adsorption was achieved with 4 g biochar L-1 at pH 11.0 along with the order of the competitive effect of K+ > Na+ > Ca2+ > Mg2+. Furthermore, NH4+ adsorption was exothermic. Thus, the present study demonstrated that ADR biochar has potential to adsorb NH4+ from NH4+ polluted water to meet environmental standards.
Keywords
Introduction
Distillers’ grains, the primary by-product of liquor/ethanol production, were produced about 20 million and 100 million tonnes in China in 2009 (Yang et al., 2012) and 2014 (Zuo et al., 2016), respectively, and this amount is still increasing by 20% annually. Over 80% of distillers’ grains was consumed by the beef, dairy, swine and poultry industries (Wood et al., 2014) for its 25–35% protein, 7.2% fibre and 3–13% fat content (Bhadra et al., 2009). Distillers’ grains could also be used as fertilizer source for plant growth (Nelson et al., 2009). However, the weak acidity and alcoholic toxicity of distillers’ grains may cause potential harm to animal, crops and environment (Yang et al., 2012). Besides, an oversupply of distillers’ grains due to the increasing demands and production of fuel ethanol may surpass the livestock industry’s demand. Thus, to maintain the sustainable development of the liquor/ethanol industry and environment protection, new value-added uses of distillers’ grains should be pursued.
Lately, distillers’ grains have been used as the feedstock of anaerobic digestion for biofuel production due to their high organic matter content (Jáuregui-Jáuregui et al., 2014). And the anaerobic digestion system generated high energy content of hydrogen (143 kJ g-1) and methane (55 kJ g-1) (Mishra et al., 2017). The maximum gaseous energy from the distillers’ grains anaerobic digestion (98.07 kJ g-1) was higher than that from de-oiled cake (69.58 kJ g-1) and algal biomass (67.82 kJ g-1) anaerobic digestion. And the gaseous energy production was highly dependent on the anaerobic digestion conditions including initial pH, temperature and urea addition (Budiyono et al., 2013). However, the development of biofuel production will inevitably increase the amount of anaerobic digestion residue (ADR), the main by-product of anaerobic digestion, which may cause repeated pollution (Buitrón et al., 2014). Currently, soil application is the most frequent method to dispose of ADR for its high content of organic matter and nutrients (Hupfauf et al., 2016) as well as its greater microbial stability and hygiene (Alburquerque et al., 2012). However, drawbacks after soil application of unprocessed ADR have been reported previously. For example, nutrients contained in ADR were easily leached from soil due to their high mobility (Zhu et al., 2014). Besides, the ADR could increase the N2O emission from soil (Galvez et al., 2012). Pyrolysis provides a new option to manage ADR which can avoid the shortages mentioned above.
Biochar, the solid product of organic material pyrolysis under oxygen-limited conditions, was characterized by various physicochemical properties that were significantly influenced by both the biochar feedstock and the processing conditions of pyrolysis temperature, residence time (RT) and heating rate (HR) (Luo et al., 2019; Mubarik et al., 2016; Rehrah et al., 2016; Taskin et al., 2019). And the resulting properties of resultant biochar determined its following agricultural or environmental application (Regkouzas and Diamadopoulos, 2019; Tanure et al., 2019). Biochars prepared at low temperature were favourable to soil application, in contrast, high temperature biochars were beneficial to contaminants’ removal (Agrafioti et al., 2013). Our previous work has demonstrated that the ammonium (NH4+) adsorption amount by ADR biochar increased 201% when pyrolysis temperature increased from 300 to 700°C (Zheng et al., 2018a). Long RT could enhance the pore size of the biochar (Tsai et al., 1997). Long RT and high HR decreased the biochar yield (Kwapinski et al., 2010). When HR increased from 10 to 50°C min-1, the volatile matter, specific surface area and micro-pore volumes of safflower seed press cake biochar decreased, but increased the fixed carbon content as well as O/C, H/C and (O+N)/C ratio (Angın, 2013). Higher ash content was found in biochars from manure, corn stover and grass than that from other feedstocks (Brewer et al., 2009). Compared to wood biochar, straw biochar had a higher specific surface area (Kloss et al., 2012). However, only a few researches were conducted to determine the carbonization performance of ADR (Stefaniuk and Oleszczuk, 2015; Zheng et al., 2018a), and they were mainly focused on the influence of pyrolysis temperature. Little information is available on the influence of various RTs and HRs on the physicochemical properties of ADR biochar.
Ammonium, the most common soluble pollutant in natural waters, was easily used to accelerate water eutrophication that was harmful to both the aquatic ecosystem and economic development (Fan et al., 2019) through its role as an oxygen sink during nitrification (Li and Liu, 2009). Hence, it is of great importance to develop effective ways for the removal of NH4+ from the aquatic environment. In comparison to the NH4+ control technology of chemical precipitation (Huang et al., 2017), ion exchange (Wang et al., 2016) and so on, the adsorption process by biochar has received most attention due to its economic, efficient, simple operation and eco-friendliness (Zhang and Wang, 2016). However, the NH4+ adsorption affinity was significantly related to the biochar properties especially its specific surface area, porous structure and thermostability that are determined by the biochar feedstock and pyrolysis conditions (Fan et al., 2019; Tang et al., 2019; Zheng et al., 2018a). Most studies were concentrated on the adsorption of NH4+ onto biochar prepared at different feedstock and pyrolysis temperatures (Li et al., 2018; Shin et al., 2018; Zhang et al., 2017; Zheng et al., 2018b). To our best knowledge, the link between pyrolysis RT and HR on NH4+ sorption is still unclear.
Prior to the experiment, 700°C was selected as the ADR pyrolysis temperature due to its maximal amount of NH4+ adsorption (Zheng et al., 2018a). This study aims to: (1) determine the characteristics of ADR biochars prepared at different RT and heating rate; (2) provide the optimum RT and HR for ADR pyrolysis based on the maximal amount of NH4+ adsorption; and (3) understand the effect of biochar dosage, solution pH, temperature and competing ions on NH4+ adsorption efficiency. Our results will expand the disposal approach of ADR which will assist both the environment protection and the liquor/ethanol industries’ sustainable development.
Materials and method
ADR
The ADR, the solid fraction of digestate, was collected from a 500 m3 biogas plant located in Pingdu, Qingdao, China (36°28′N, 119°35′E). The main feedback of the biogas reactor was the Maotai-flavour distillers’ grains. The operation temperature was between 35 and 37°C with a 40 days retention time. The ADR samples were then mixed, air dried, deionized water washed, oven-dried (105°C, 24 h) and unified to the size of 0.5–1.0 cm. Detailed characteristics of the ADR were shown in our previous paper (Zheng et al., 2018b).
Biochar production
Batch tests of 40 g ADR were carried out with porcelain crucibles covered by caps in a muffle furnace (KSL-1200X, Kejing Materials Tec., China) at 700°C. A nitrogen (99.999%) flow of 0.5 dm3 min-1 was supplied constantly. After reaching the setting temperature along with the HR of 5.0°C min-1, the ADR in the muffle furnace was kept for 30, 60 and 120 min, respectively. When the RT was fixed to 60 min, the HRs of 2.5, 5.0 and 10.0°C min-1 from room temperature to 700°C were set. Subsequently, the samples in the muffle furnace were cooled to the room temperature under the N2 atmosphere. The biochar yield was then obtained from the final weight of residue. The resulting biochar was crushed using a ceramic mortar and pestle, then sieved to < 0.25 mm. Finally, the biochar samples were stored in a desiccator (25°C) for further use. The biochar from RTs of 30, 60, 120 min and HRs of 2.5, 5.0 and 10.0°C min-1 were assigned as BC30, BC60, BC120 and BC2.5, BC5, BC10. Notably, the BC60 and BC5 were uniformly designated as BC60/5 for their same pyrolysis conditions.
Characterization of ADR and ADR biochar
The cellulose, hemicellulose and lignin content of ADR were determined by the Van Soest method. Briefly, to isolate the cellulose, hemicellulose and lignin from the ADR sample, the detergent fibres’ extraction was conducted by an FIWE6 extraction unit (VELP, Italy). Then the content of cellulose, hemicellulose and lignin was analysed according to the procedure developed by Van Soest (Goering and van Soest, 1970). Proximate analyses of biochar were determined according to the methods described by Zheng et al. (2018a). The content of ash and volatile matter were determined by heating biochar samples at 760°C for 6 h under air and 900°C for 7 min under N2 in a KSL-1200X muffle furnace (Kejing Materials Tec., China), respectively. The content of fixed carbon, after that, was calculated by difference. Ultimate analyses were measured on a vario EL III elemental analyser (Elementar Co., Germany). The content of O (%
Adsorption isotherm experiment and model fitting
Stock solution of NH4+ (1000 mg N/L) from NH4Cl was prepared in deionized water. The desired concentrations were freshly prepared from the stock solution.
To determine the optimum RT and HR for ADR pyrolysis based on the maximal amount of NH4+ adsorption, the adsorption isotherm experiments were conducted according to Zheng et al. (2018b). Briefly, 0.4 g biochar was mixed with 40 ml NH4Cl solutions of known concentrations (10–150 mg N L-1) in 50 ml centrifuge tubes. The solution pH was then adjusted to 7.0 by 0.1 mol L-1 HCl or NaOH. After that, the centrifuge tubes were orbital shaked at 140 revolutions per minute (rpm) for 120 h at room temperature (25°C). The supernatant was collected by filtration (0.45
The specific amount of NH4+ adsorbed at equilibrium (

where
The adsorption capacity of ADR biochar for NH4+ was defined with the most commonly used isotherm models of Langmuir, Freundlich, Langmuir–Freundlich, Redlich–Peterson and Temkin (Zheng et al., 2018b). The Langmuir model frequently describes the homogeneous adsorption process. Yet the other four models, as the empirical or semiempirical equations, present the heterogeneous adsorption progress (Yao et al., 2013). The corresponding equations are used as in equations (2), (3), (4), (5) and (6):
Langmuir:

where
Freundlich:

where
Langmuir–Freundlich:

where
Redlich–Peterson:

where
Temkin:

where
Batch adsorption of NH4+
After the optimization of RT and HR, the effect of ADR biochar mass, solution pH, competing ions and operating temperature on NH4+ adsorption was investigated. Briefly, for ADR biochar mass, 100 mg N L-1 was mixed with different ADR biochar mass (1–20 g L-1), while the solution pH and temperature were kept at 7.0 and 298.15 K, respectively. For solution pH, 0.4 g ADR biochar was mixed with 40 ml 100 mg N L-1 ammonia solution along with the solution pH which was adjusted to a range of 2.0 to 12.0 using 0.1 mol L-1 HCl or NaOH at the temperature of 298.15 K. For competing ions, K+, Na+, Ca2+, Mg2+ were chosen due to their common existence in various types of wastewater and their competition with NH4+ for adsorption sites. Specially, 40 ml 100 mg L-1 K+ (or Na+/Ca2+/Mg2+) prepared from 100 mg N L-1 ammonia solution was mixed with 0.4 g ADR biochar in the temperature of 298.15 K. Finally, the temperature effect was determined at 298.15, 308.15, 318.55 and 328.15 K, respectively, while other operating conditions were constant with the ADR biochar mass and solution pH test. Other parameters such as equilibrium time, shaking speed and the method of NH4+ measurement, were identical to the adsorption isotherm experiment.
The equilibrium removal percentage of NH4+ (% Removal) was determined as in equation (7) (Kizito et al., 2015):

Thermodynamic parameters such as change in Gibbs free energy (Δ



Δ
Statistical analysis
All the results were given on oven-dry basis. The data of adsorption isotherms were collected in triplicates, and then fitted to isotherm models using Origin Pro 9.1. The determination coefficients (
Results and discussion
Biochar characterization
Proximate analysis and elemental compositions
The proximate and elemental analyses of ADR biochars as a function of RT and HR are presented in Table 1. The biochar yield decreased with increasing RT due to the slow decomposition of hemicellulose (17.81%), cellulose (26.96%) and lignin (28.72%) of ADR. 10.0°C min-1 of HR significantly (
Effect of different residence time (RT) and heating rate (HR) on main properties of anaerobic digestion residue biochars.
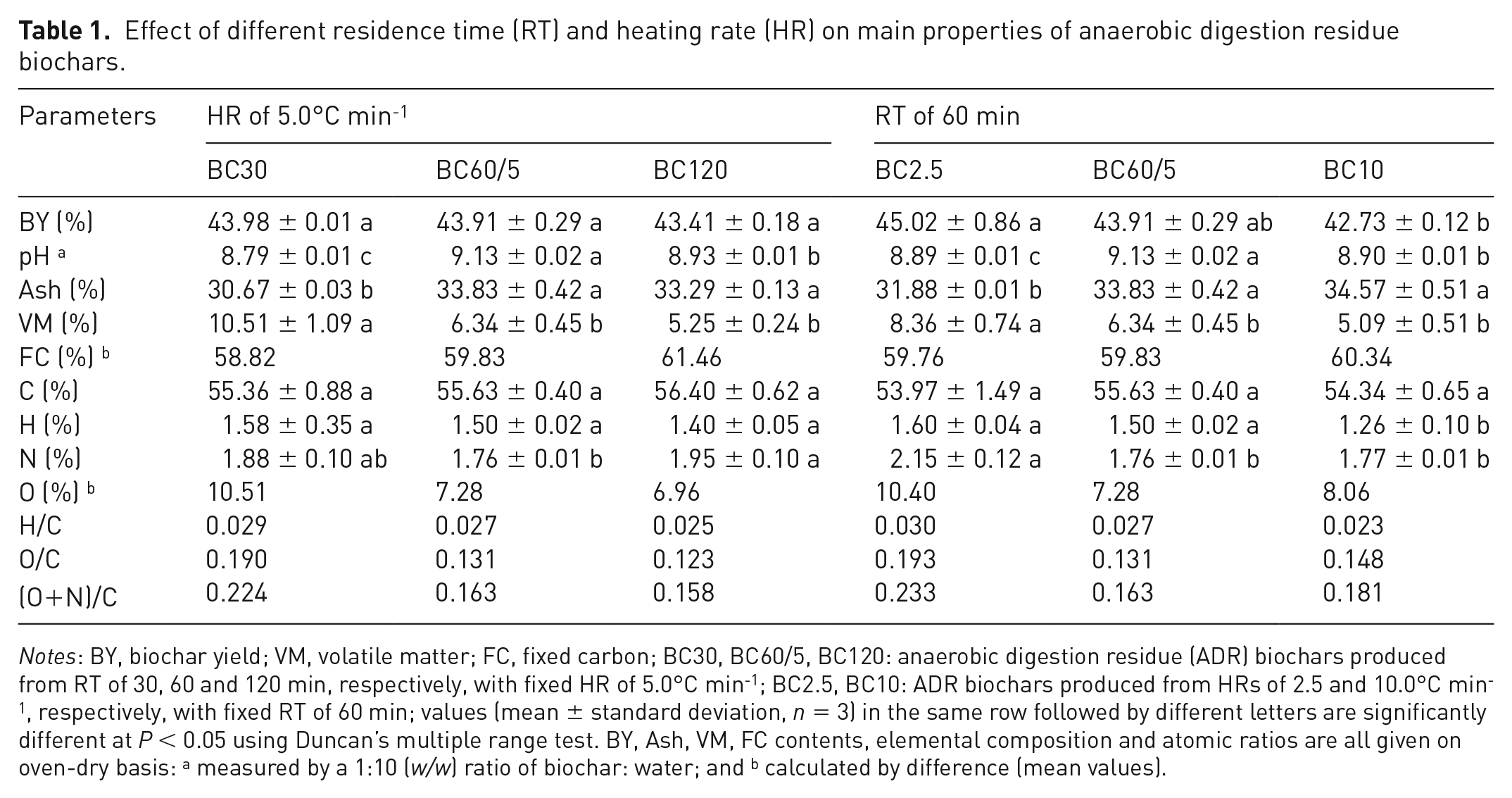
The ash content significantly (
The carbon content was limited in the range of 53.97% to 56.40% with no significant differences (
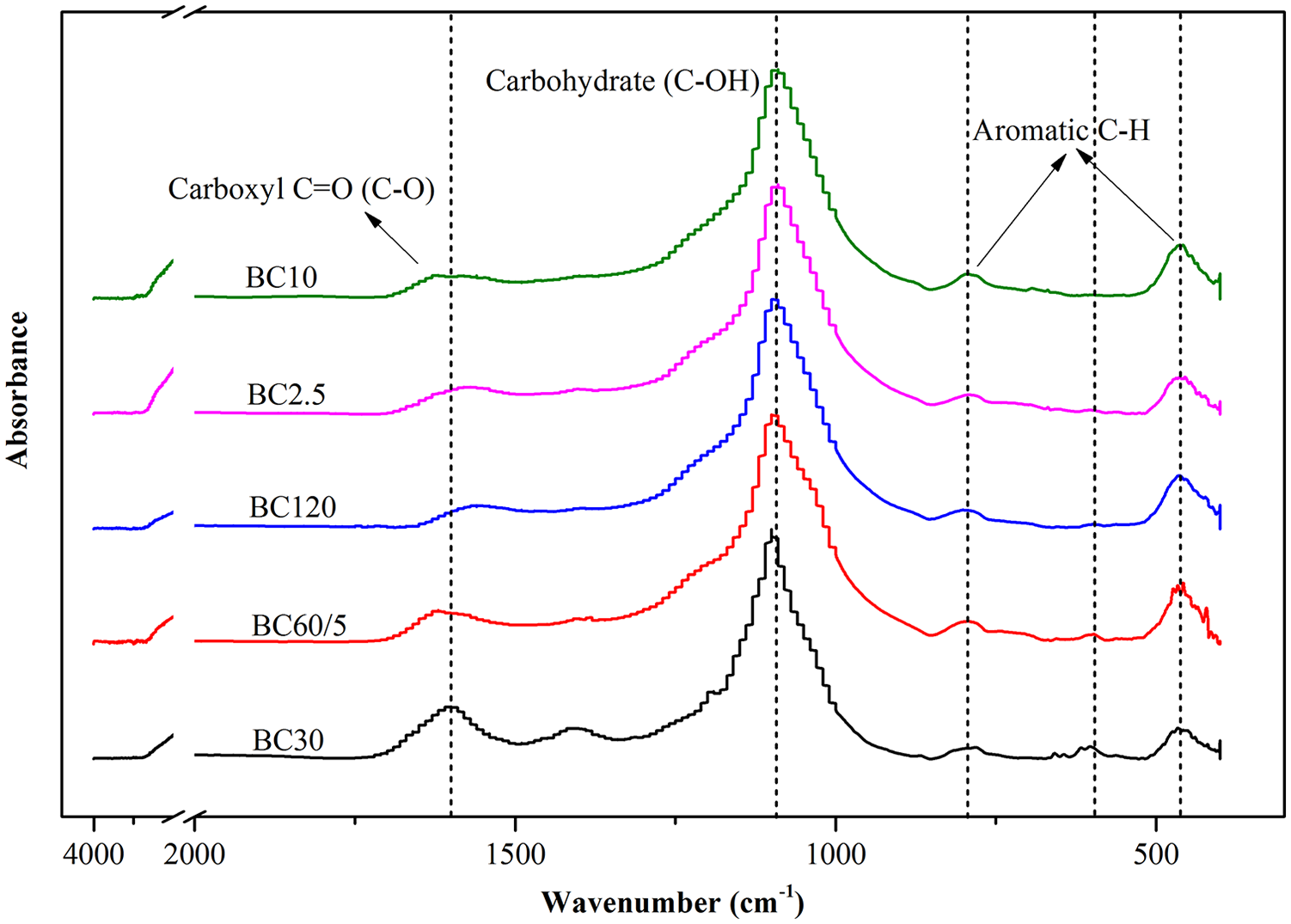
Fourier transform infrared spectroscopy spectra of anaerobic digestion residue biochars.
Surface properties
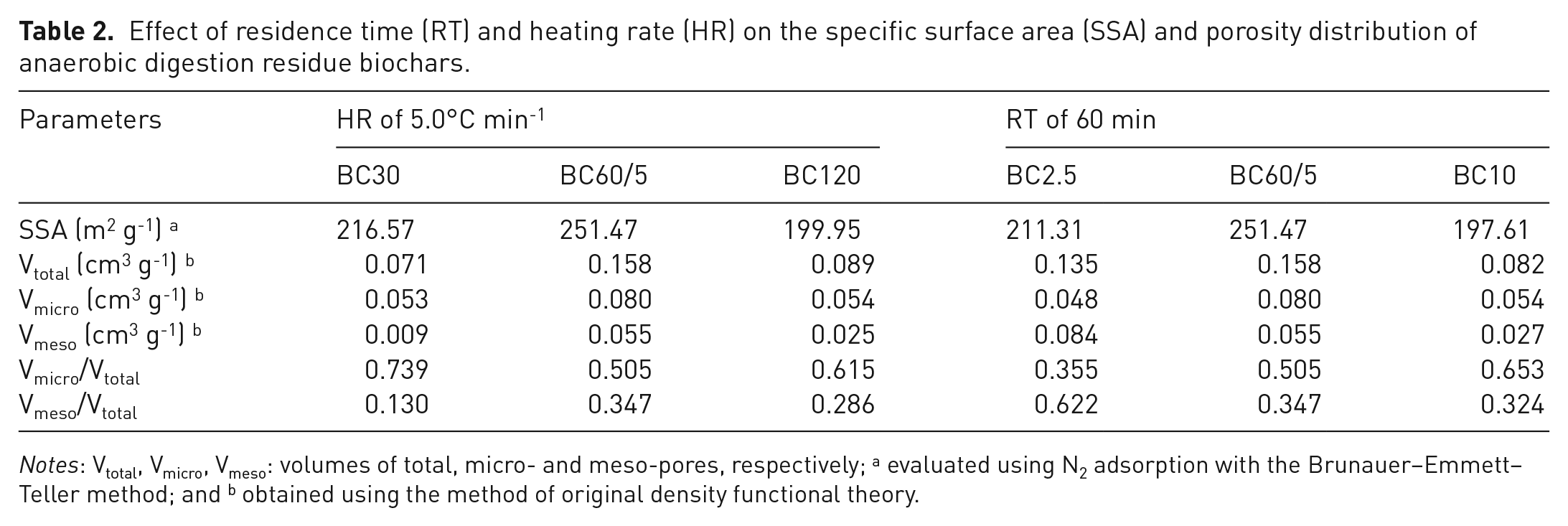
As shown in Table 2, the SSA of ADR biochar gradually increased from 216.57 and 211.31 m2 g-1, respectively, to 251.47 m2 g-1 with increasing RT from 30 to 60 min and HR from 2.5 to 5.0°C min-1, then decreased to 199.95 and 197.61 m2 g-1, respectively, with further increase of RT and HR. The same trend for volumes of micro-pores (Vmicro) and total pores (Vtotal) was also observed. This could be attributed to the release of volatiles (Mui et al., 2010). Longer RT and lower HR were both favourable to the diffusion of volatile products from biochar particles. But shorter RT and higher HR could lead to the accumulation of volatiles between and within biochar particles. The decrease of SSA, Vmicro and Vtotal that exceed a certain value of RT and HR could be explained by the collapse of micro-pores to meso-pores (Zheng et al., 2018a) as shown in Figure 2. Besides, the SSA was significantly and positively correlated with Vmicro (
Effect of residence time (RT) and heating rate (HR) on the specific surface area (SSA) and porosity distribution of anaerobic digestion residue biochars.

Scanning electron microscopy images of anaerobic digestion residue biochars.
The N2 adsorption–desorption isotherms and pore sizes’ distribution of ADR biochars are illustrated in online Supplementary Figure S2 and Figure 3, respectively. For RT, the N2 isotherms exhibited type I (micro-pore adsorption) behaviour according to the International Union of Pure and Applied Chemistry classification (Brunauer et al., 1940), indicating the formation of micro-porous structure which was subsequently confirmed by the high ratio of Vmicro/Vtotal which ranged from 0.505 to 0.739 (Table 2). For HR, the N2 isotherm of BC2.5 was of type IV, which was indicative of the wide distribution of meso-pores demonstrated by the high Vmeso/Vtotal ratio of 0.622 (Table 2). The N2 isotherms of BC60/5 and BC10 were then evolved to type I, reflecting the evolution of micro-porous, and it was confirmed by the increased ratio of Vmicro/Vtotal (Table 2). As shown in Figure 3, the micro-pores of 0.50–0.75 nm and 1.25 nm in diameter were the main pore sizes in the ADR biochars, and they both increased considerably when RT increased from 30 to 60 min and HR from 2.5 to 5.0°C min-1, subsequently decreasing with further increase of RT and HR.
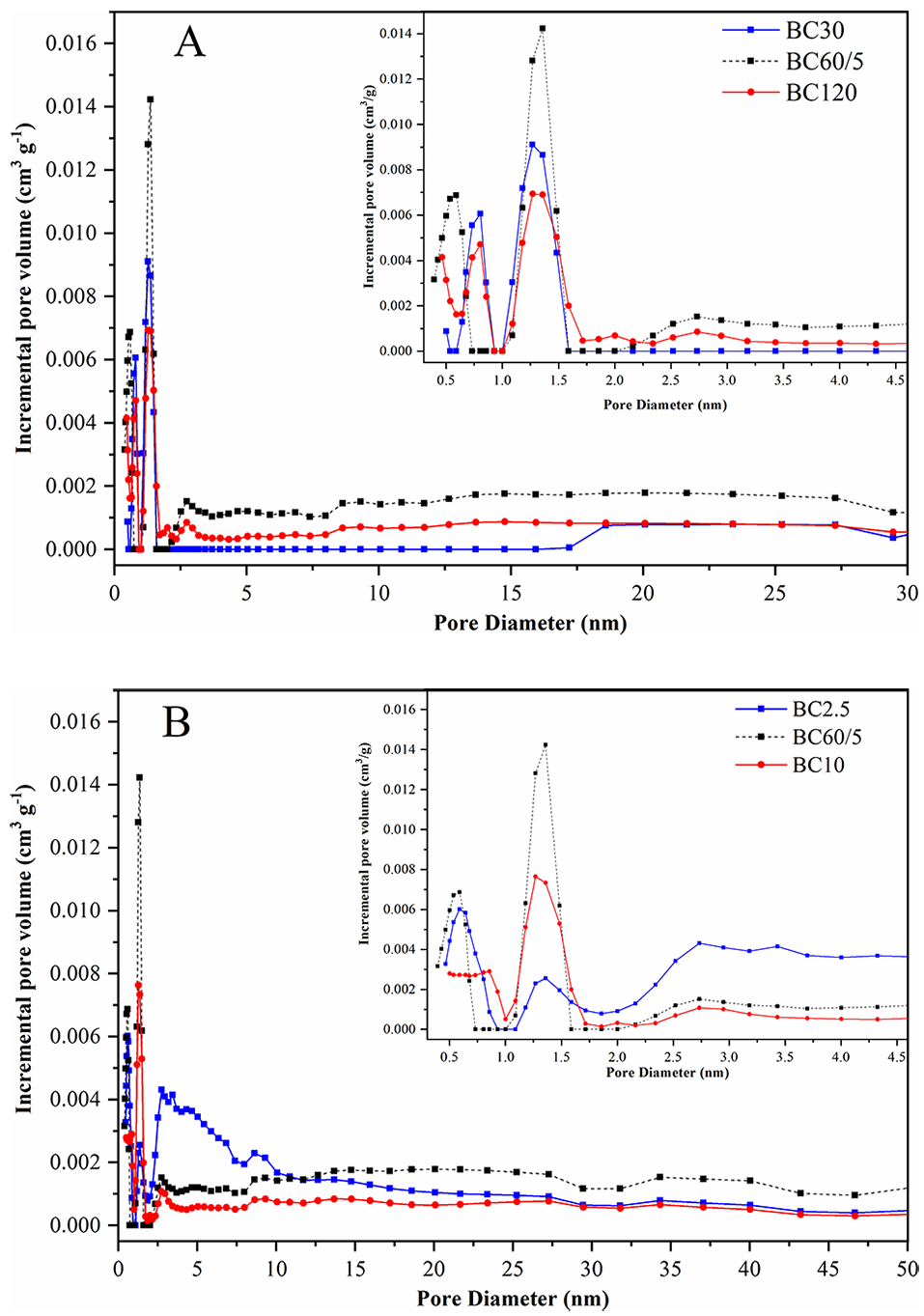
Effect of residence time (RT) (A) and heating rate (HR) (B) on the pore sizes distribution of anaerobic digestion residue (ADR) biochars. BC30, BC60/5, and BC120: ADR biochars produced from RT of 30, 60 and 120 min, respectively, with fixed HR of 5.0°C min-1. BC2.5, BC10: ADR biochars produced from HRs of 2.5 and 10.0°C min-1, respectively, with fixed RT of 60 min.
Surface morphology
The surface structure of ADR biochars under different RT and HR is visualized in Figure 2. As observed, all the ADR biochars featured regular pore structures’ morphology which contributed to the high SSA (> 190 m2 g-1; Table 2) of ADR biochars. Many pores in a honeycomb shape were clearly found in the biochar surface, and this should be caused by the cellulose, hemicellulose and lignin degradation of ADR during the pyrolysis (Sevilla and Fuertes, 2009). With the increase of RT from 30 to 60 min and HR from 2.5 to 5.0°C min-1, more micro-pores of 0.50–0.75 nm and 1.25 nm were developed which is confirmed in Figure 3. And this maybe the main reason for the increase of SSA, Vmicro and Vtotal (Table 2). However, with the further increase of RT and HR, higher abundance of bigger size pores (Figure 2) along with the decrease of Vmicro and Vmeso (Table 2) were found due to the breaking and merging of the honeycomb structure. Thus, the development of pore sizes of ADR biochars was significantly affected by the RT and HR.
FTIR analysis
The FTIR spectra of ADR biochars from different RT and HR are displayed in Figure 1. Figure 3 shows that the carbohydrate, aromatic and carboxyl groups were predominant, especially the carbohydrate group. Similar intensity of the C-OH stretching vibration (1054 cm-1) corresponding to the carbohydrate groups such as primary, secondary and tertiary alcohols, phenols, ethers and esters (Angın, 2013) was observed. The intensity of antisymmetric C = O (C-O) stretching vibration (1610 cm-1) of carboxyl groups (-COOH) became lower with increasing RT; however, no effect was found in the HR. Besides, the intensity of aromatic C-H stretching vibration (462 cm-1) also increased with increasing RT and HR (Kizito et al., 2015), but the intensity of aromatic C-H out of plane stretching (801 cm-1) (Chen et al., 2014) was not affected. Generally, the effect of RT and HR on the FTIR spectra of ADR biochars was not pronounced except the stretching vibration of aromatic C-H, which was indicative of the enhancement of the aromaticity of ADR biochars produced under longer RT and higher HR.
Adsorption isotherms
The adsorption capacity of ADR biochars for NH4+ was defined with most commonly used isotherm models, namely the Langmuir, Freundlich, Langmuir–Freundlich, Redlich–Peterson and Temkin models. The fitting parameters are presented in Table 3, and the pattern of NH4+ adsorption is illustrated in Figure 4 (RT) and Figure 5 (HR). The fitting results showed that the Freundlich adsorption isotherm model represented the experimental data best with the highest coefficient (
Adsorption isotherms constants and correlation coefficients of NH4+ adsorption by anaerobic digestion residue biochars.
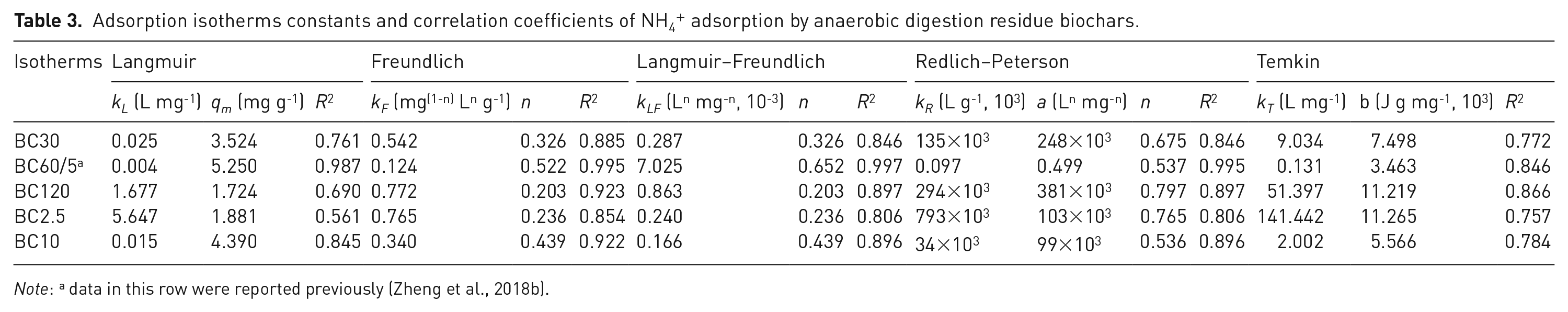
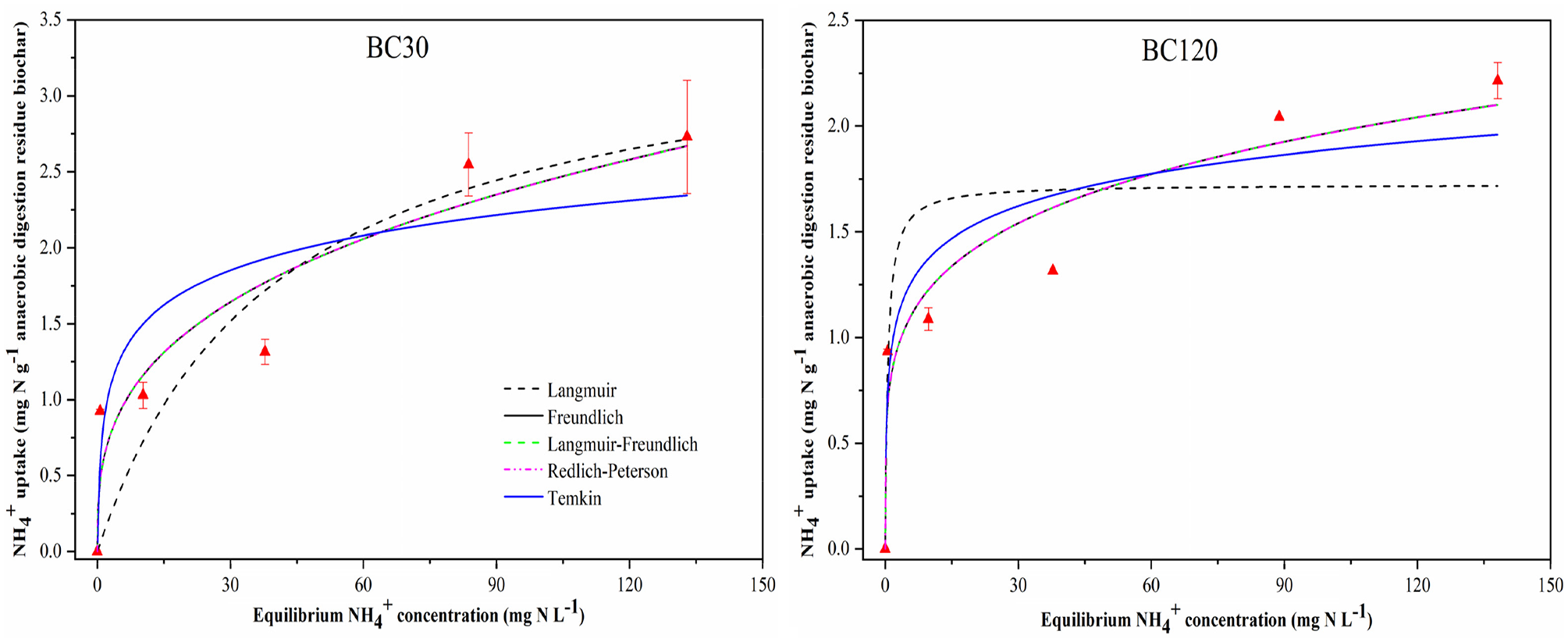
Adsorption isotherm curves of NH4+-N on anaerobic digestion residue biochars produced from different residence times. The fitting results of BC60/5 were previously reported (Zheng et al., 2018b).
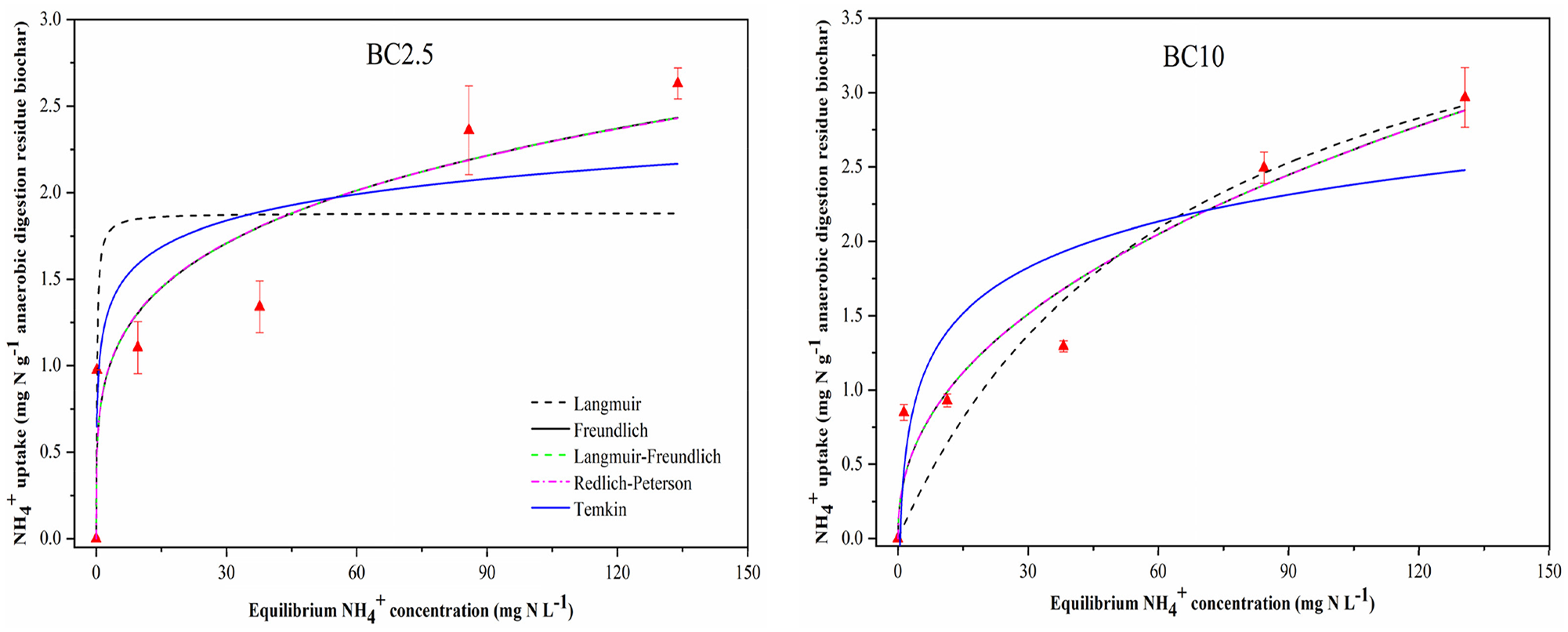
Adsorption isotherm curves of NH4+-N on anaerobic digestion residue biochars produced from different heating rates. The fitting results of BC60/5 were previously reported (Zheng et al., 2018b).
Amount of NH4+ adsorbed by BC60/5 and effect of BC60/5 mass on amount of NH4+ adsorbed
Figure 6 indicates that the percentage of NH4+ removal increased from 20.1% to 27.4% as the mass increased from 1 to 4 g L-1. This can be attributed to the increased SSA of BC60/5 and the enhanced adsorption sites’ availability resulting from the increase of BC60/5 mass (Zhang et al., 2011). However, with a further increase to 20 g L-1, the NH4+ removal percentage was only raised to 30.6% due to the overlap or aggregation of BC60/5 particles that shielded the available adsorption sites (Kizito et al., 2015). Similar NH4+ adsorption behaviour was also observed on biochars from pine sawdust/wheat straw (Yang et al., 2018) and rice husk/wood (Kizito et al., 2015). Obviously, negligible effect on NH4+ removal was observed when the BC60/5 mass was over 4 g L-1. Therefore, 4 g L-1 was regarded as the optimum BC60/5 usage. In contrast, NH4+ adsorption on per gram of BC60/5 significantly decreased from 20.56 to 1.57 mg N g-1 as the BC60/5 mass increased from 1 to 20 g L-1. As the initial concentration of NH4+ kept constant, it is very clear that the ratio of NH4+ amounts to BC60/5 adsorption sites decreased due to the increased actual number of adsorption sites resulting from the enhanced BC60/5 mass (Zhang et al., 2011).
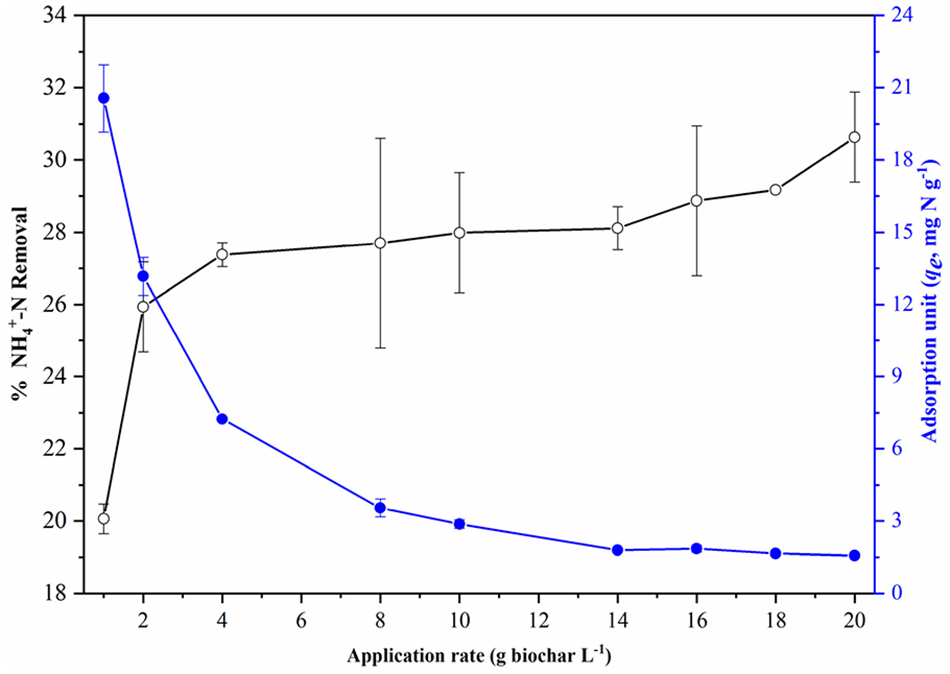
Effect of the BC60/5 mass on the removal and adsorption unit of NH4+-N (C0, 100 mg N L-1; pH, 7.0; temperature, 298.15 K; reaction time, 120 hours).
Effect of solution pH on amount of NH4+ adsorbed
Figure 7 shows the NH4+ removal efficiency and corresponding NH4+ adsorption unit by BC60/5 at various pHs from 2.0 to 12.0. Both the percentage removal and amount adsorbed (mg N g-1) of NH4+ increased rapidly when the pH ranged from 2.0 to 3.0 due to the intense competition for adsorption sites between H+ and NH4+ (Zhang and Wang, 2016) as well as the high protonation of functional groups (C = O, COO-) on the BC60/5 surface that weakened the polar attraction of NH4+ ions in aqueous solution (Novak et al., 2010). However, the NH4+ removal efficiency and corresponding NH4+ adsorption unit were not enhanced when pH increased from 3.0 to 8.0. The alkaline nature of BC60/5 (pH = 9.13; Table 1) led to the similar solution pH due to its OH- release (Zhu et al., 2012), although the decreased H+ amount weakened the competition between H+ and NH4+ (Yang et al., 2018). For 8.0 < pH ⩽11.0, the NH4+ removal percentage and corresponding NH4+ adsorption unit were significantly increased from 19.59 to 58.05% and 1.93 to 4.93 mg N g-1, respectively. This trend could be attributed to the dominant mechanism of electrostatic attraction (Mubarik et al., 2016; Vu et al., 2017). At pH > 11.0, the adsorption took a decreasing trend for the conversion of NH4+ to NH3 (Yang et al., 2018).
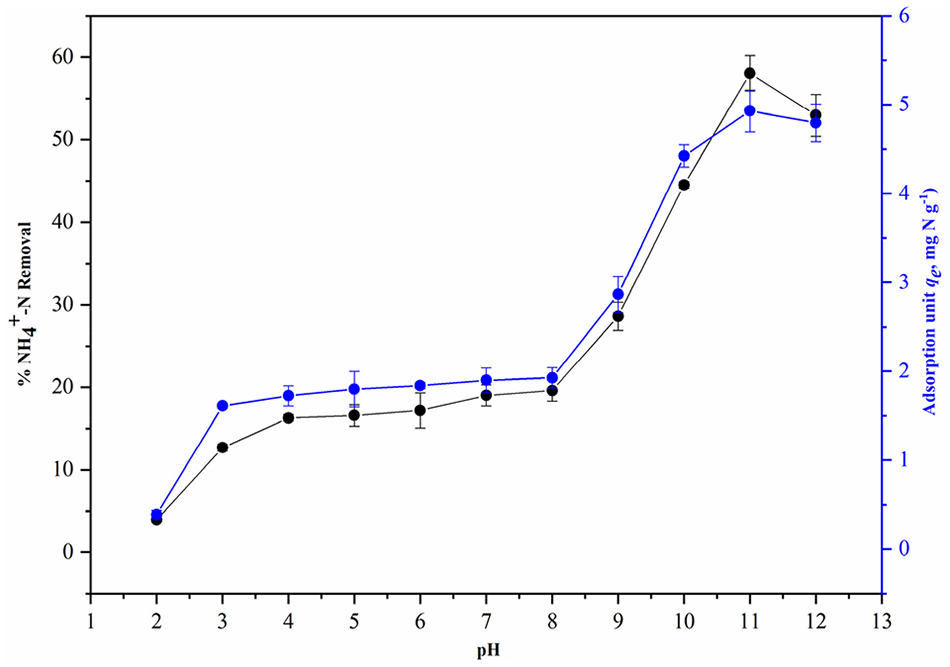
Effect of solution pH on the removal and adsorption unit of NH4+-N by BC60/5 (C0, 100 mg N L-1; mass, 10g L-1; temperature, 298.15 K; reaction time, 120 hours).
Effect of the individual presence of other cations on amount of NH4+ adsorbed
Figure 8 shows the effect of the individual presence of other cations on the NH4+ adsorption. The individual presence of Mg2+, Ca2+, Na+ and K+ led to the significant decrease of NH4+ removal percentage from 27.99% (only NH4+ existed) to 17.39%, 17.18%, 16.52% and 13.76%, respectively. This phenomenon could be explained by the competition between NH4+ and cations for the adsorption sites based on the electrostatic attraction (Yang et al., 2018). And this decrease trend could be linked to the order of ionic radii of K+ > Na+ > Ca2+ > Mg2+ – then the available pore size of formed Mg2+-BC60/5 was bigger than those of Ca2+-, Na+- and K+-BC60/5 which reduced the diffusion rate of NH4+ in the pores (Huang et al., 2017). Thus, negative effect of coexisting cations on NH4+ adsorption of ADR biochar was confirmed. Similar NH4+ adsorption studies using wood/rice biochar (Kizito et al., 2015) and zeolites (Huang et al., 2017) have reported the same effect in the presence of cations.
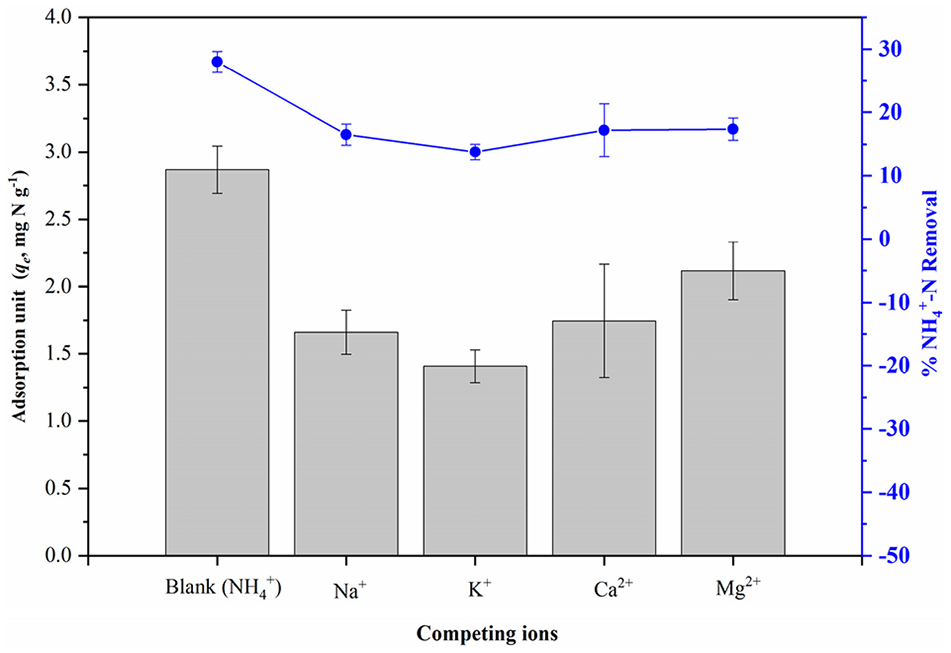
Effect of competing ions (Na+, K+, Ca2+, Mg2+) on the removal and adsorption unit of NH4+-N by BC60/5 (C0, 100 mg N L-1; mass, 10 g L-1; pH, 7.0; temperature, 298.15 K; reaction time, 120 hours).
Effect of temperature on amount of NH4+ adsorbed and sorption thermodynamics
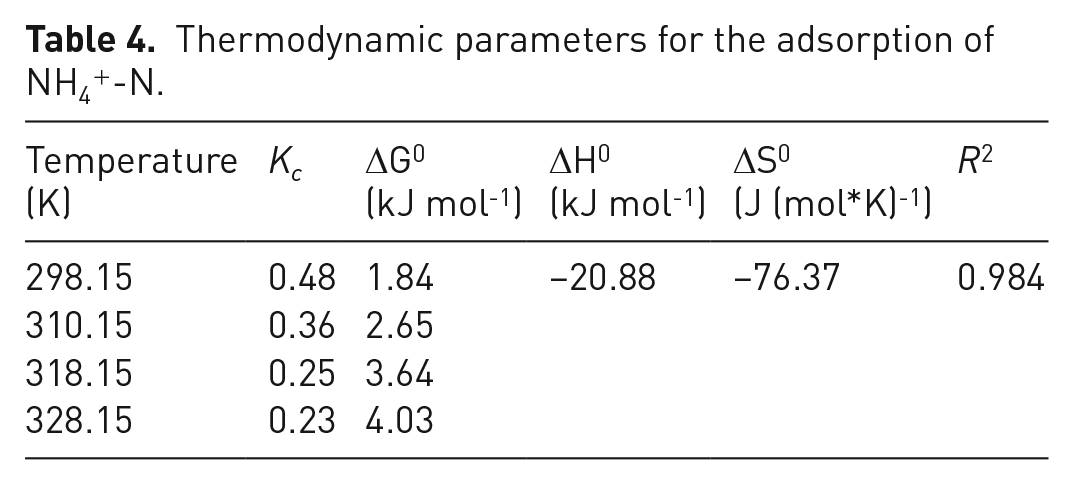
The effect of temperature on the amount of NH4+ adsorbed by BC60/5 was investigated at different temperatures, that is, 298.15, 308.15, 318.15 and 328.15 K, as shown in Figure 9. With the temperature increasing, the removal efficiency and adsorption unit of NH4+ were both significantly decreased. As presented in Table 4, the positive value of Δ
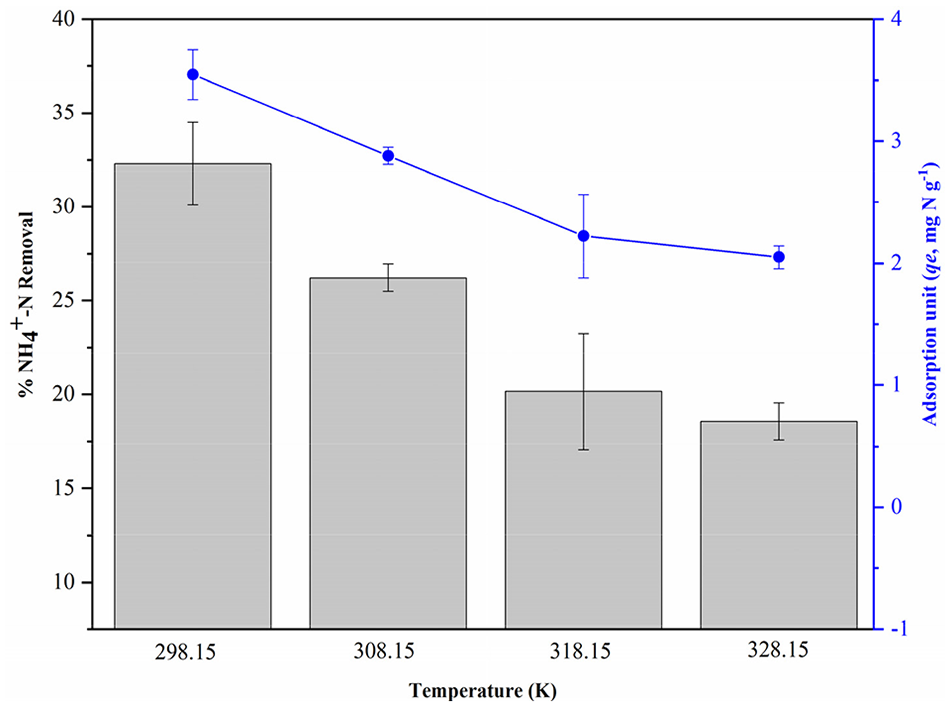
Effect of temperature on the removal and adsorption unit of NH4+-N by BC60/5 (C0, 100 mg N L-1; mass, 10 g L-1; pH, 7.0; reaction time, 120 hours).
Thermodynamic parameters for the adsorption of NH4+-N.
Conclusions
The results from this study indicate that both the RT and HR significantly influenced the physicochemical properties of biochar produced from ADR. Longer RT and higher HR were both favoured to obtain higher aromaticity as well as fixed carbon and elemental carbon content. However, for pyrolysis yield, the content of volatile matter and elemental hydrogen, oxygen, as well as the polarity, an opposite trend was found. RT of 60 min and HR of 5.0°C min-1 of ADR biochar (BC60/5) showed the best development of orderly and cylinder-like structure, highest specific surface area and maximal amount of NH4+ removal. Equilibrium data of NH4+ adsorption were best simulated by Freundlich isotherm. The maximum adsorption capacity of NH4+ was observed under the mass of BC60/5 of 4 g L-1 at pH 11.0 along with the order of the competitive effect of K+ >Na+ > Ca2+ > Mg2+. Besides, the process of NH4+ adsorption was exothermic. It is believed that the present ADR biochar prepared at 60 min, 5°C min-1 and 700°C can be effectively applied in removing NH4+ from NH4+ polluted water to meet water quality standards for the environment.
Supplemental Material
Supplemental_File – Supplemental material for Biochar of distillers’ grains anaerobic digestion residue: Influence of pyrolysis conditions on its characteristics and ammonium adsorptive optimization
Supplemental material, Supplemental_File for Biochar of distillers’ grains anaerobic digestion residue: Influence of pyrolysis conditions on its characteristics and ammonium adsorptive optimization by Xuebo Zheng, Ting Shi, Wenjing Song, Lei Xu and Jianxin Dong in Waste Management & Research
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the project ZR2019BD062 supported by the Shandong Provincial Natural Science Foundation, 2019B04 supported by the Science Foundation for Young Scholars of Tobacco Research Institute of Chinese Academy of Agricultural Sciences, and 2017M612371 supported by the China Postdoctoral Science Foundation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
