Abstract
Objective
Smoking has been associated with psychiatric and somatic comorbidities in bipolar disorder (BD) populations. However, studies in older age BD (OABD) populations are sparse. We hypothesized that among individuals with OABD, current and former smokers would have worse psychiatric and somatic comorbidities parameters compared to never smokers.
Method
Our study used baseline cross-sectional data from 27 international studies reporting data on adults 50 years old and older (N = 984). Smoking status was categorized into current smokers, former smokers, and never smokers. The distribution of demographic and clinical variables was assessed. The associations between smoking status and the clinical variables were examined using multivariable models that adjusted for age, sex, and study. Multivariable models were repeated, restricting to individuals with and without cardiovascular or respiratory (cardiorespiratory) comorbidity.
Results
Our study sample was 52.8% female with a mean age of 62 years and included 347 (35.3%) never smokers, 222 (22.6%) former smokers, and 415 (42.2%) current smokers. After controlling for age, sex, and study, current depression was more prevalent in former versus never smokers and current versus never smokers. Cardiovascular comorbidity was more prevalent among former than never smokers. More current versus never smokers were taking antipsychotic medications and more current versus never smokers having lifetime substance use disorders. When stratifying by the presence of cardiorespiratory comorbidity, the only statistically significant association was higher functioning in never versus current smokers in participants without cardiorespiratory comorbidity, though non-statistically significant relationships were present between lifetime smoking and depression across strata.
Conclusions
The relationship of smoking with depression and substance use disorders is largely independent of age, sex, and, for the depression relationship, cardiorespiratory comorbidity. More smokers taking antipsychotic medications suggests that smoking is associated with a more severe BD course. Cardiovascular comorbidity may serve as a motivating factor for smoking cessation.
Plain Language Summary:
This study examines how smoking impacts mental and physical health in older adults with bipolar disorder (BD). It compares the health of individuals aged 50 and older with BD who currently smoke with those who formerly smoked and those who have never smoked. The study found that smoking is linked with worse health outcomes in this population. It shows that depression, cardiovascular issues, and possibly substance use disorders are more common in those who smoke than those who never smoked. As cardiovascular issues are especially common in patients who formerly smoked, these cardiovascular issues may motivate individuals with BD to quit smoking.
Introduction
Smoking has a prevalence of 30% to 70% in individuals with bipolar disorder (BD), 2–3 times higher than in the general population.1,2 Smoking has been associated with greater BD symptom severity and suicidality,3–5 which may be secondary to smoking-induced oxidative stress or neuroinflammation6,7 and BD treatment non-adherence. 5 Smoking has also been implicated in cardiovascular and metabolic comorbidities in BD populations.8,9 Almost 50% of deaths in patients with BD are associated with tobacco use. 10 Mental health symptoms may also complicate attempts to quit smoking.11,12
The relationship between smoking, BD, and somatic illness is complex. While several studies have shown an association between smoking and metabolic and cardiovascular burden in individuals with BD,8,9 antipsychotic use may partly explain this burden. 8 Another study has demonstrated an association between smoking cessation and metabolic and cardiovascular burden. 13
In older adults with BD (OABD) in particular, the relationship between smoking status and psychiatric and somatic comorbidities is yet to be elucidated. OABD refers to BD patients aged 50 and over, regardless of age of illness onset. 14 The population of OABD is growing and is associated with an elevated risk of somatic comorbidities, impaired psychosocial functioning, and cognitive deficits. 15 This somatic comorbidity burden may be partly secondary to health behaviours, including smoking and alcohol use. 16
Our multisite OABD collaboration has allowed us to examine the prevalence of smoking in OABD and the relationship of smoking status with demographic, somatic, and psychiatric parameters. We hypothesized that (1) among individuals with OABD, former smokers and current smokers would have worse psychiatric and somatic comorbidity burden (e.g., current mood symptoms, substance use disorders [SUDs], medical comorbidities, functioning) compared with never smokers; (2) individuals with OABD who have cardiovascular or respiratory (cardiorespiratory) comorbidities would demonstrate a relationship between smoking status and clinical parameters, as these vulnerable patients may have difficulty properly managing their BD and other somatic comorbidities.
Methods
Overview
The Global Aging & Geriatric Experiments in BD Database (GAGE-BD) sample comprises pooled data from BD studies from several countries. GAGE-BD methods have been described elsewhere. 17 Demographic and clinical variables were harmonized across studies given that studies used different measures to assess similar domains.17,18 The aggregate sample, as of April 2024, used baseline cross-sectional data from 27 international studies reporting data on adults ages 50 years and older with BD (N = 984). These studies involved 18 study sites spanning 10 countries. Contributing studies are listed in Supplemental Tables S1 and S2. Local institutional review boards or ethics committees at originating sites provided approval.
Study Measures
The primary independent variable of smoking status (exposure of interest) was categorized into current smokers, former smokers (participants who had previously smoked and quit, per self-report), and never smokers (reference category). Demographic variables included age, sex, marital status (currently married, currently not married) and employment status (employed, unemployed).
Clinical variables included depression status, severity of manic symptoms, lifetime psychiatric hospitalization, functioning, lifetime SUD, current antipsychotic use, body mass index (BMI), and the presence of somatic comorbidity affecting various organ systems (see below).
Lifetime SUD was dichotomized into current/past SUD or no SUD. Current antipsychotic use was dichotomized into taking or not taking antipsychotic medications. Depression was dichotomized into present or absent. The dichotomous variable was created from a variable that harmonized scores from the Hamilton Depression Rating Scale (HAM-D),
19
the Montgomery-Asberg Depression Rating Scale (MADRS),
20
and the Center for Epidemiologic Studies Depression Scale (CES-D).
21
The cut-offs for depression being present were
Somatic comorbidity was assessed in individual studies using various methods, including standardized measures such as the Cumulative Illness Rating Scale and the Charlson Comorbidity Index, 25 or clinical determination of comorbidity categories based on chart review, self-report, or physical examination. Somatic comorbidity was harmonized into eight binary variables (present/absent) within the following organ systems: cardiovascular, respiratory, gastrointestinal, hepatic/pancreatic, renal, genitourinary, musculoskeletal, and endocrine. Each comorbidity was assessed as a “Yes/No” question that include several example diagnoses (e.g., for the gastrointestinal system, gastric or duodenal ulcer, bloating/constipation, gastrointestinal reflux disease, Celiac disease, Crohn's disease, ulcerative colitis, colon cancer, etc., were included as examples).
Data Analysis
OABD individuals were compared by their smoking status using the non-parametric Kruskal–Wallis test for continuous variables and Fisher's exact test for categorical variables. The associations between smoking status, the independent variable of interest, and the clinical variables were examined further using multivariable models that adjusted for age, sex, and study. These adjustments were made due to sex differences in the relationship between smoking and psychiatric and somatic health parameters,26,27 age-related metabolic factors associated with differential smoking effects,28–30 and possible effects attributable to different study designs. Multivariable models included binomial logistic, multinomial logistic, and mixed effect generalized linear models when appropriate for the binary, nominal, and continuous clinical outcomes with study as a random-effect across all models. Additionally, we repeated these multivariable models restricting to individuals with and without cardiorespiratory comorbidity. Adjusted odds ratios and beta values for categorical and continuous outcomes, respectively, are reported. A two-sided alpha of 5% was considered statistically significant.
Results
Overall Sample
The 984 participants in this study were divided into three groups as determined by their smoking status. This resulted in 347 (35.3%) never smokers, 222 (22.6%) former smokers, and 415 (42.2%) current smokers. Lifetime smoking prevalence was 64.7%. The sample included participants with the following demographics (Table 1): mean age 62.0 years (SD 8.0), 52.8% females, 54.3% single and 45.7% married, and 70% not working. Expanding on the age distribution, all participants were 45 years or older, 27% were between 46 and 55 years, 43% were between 56 and 65 years, 22% were between 66 and 75 years, and 8% were 75 years and older. In terms of psychiatric variables, 47.6% had current depression, 70.2% had current manic symptoms (29.2% had YMRS score of 1 to 4, while 41.0% had YMRS score greater than 5), 54.2% had a lifetime SUD, 27.6% had a psychiatric hospitalization, and 50.5% were on antipsychotic medication. The mean GAF score was 58.7 (SD 18.6), indicating moderate difficulty in functioning. In terms of somatic variables, 42.2% had cardiovascular comorbidity, 33.3% had respiratory comorbidity, 76.5% had gastrointestinal comorbidity, 7.0% had hepatic/pancreatic comorbidity, 7.0% had renal comorbidity, 18.5% had genitourinary comorbidity, 37.7% had musculoskeletal comorbidity, and 30.5% had endocrine comorbidity. Mean BMI was 28.3 (SD 6.1).
Descriptive Analysis of Smoking Status Groups.
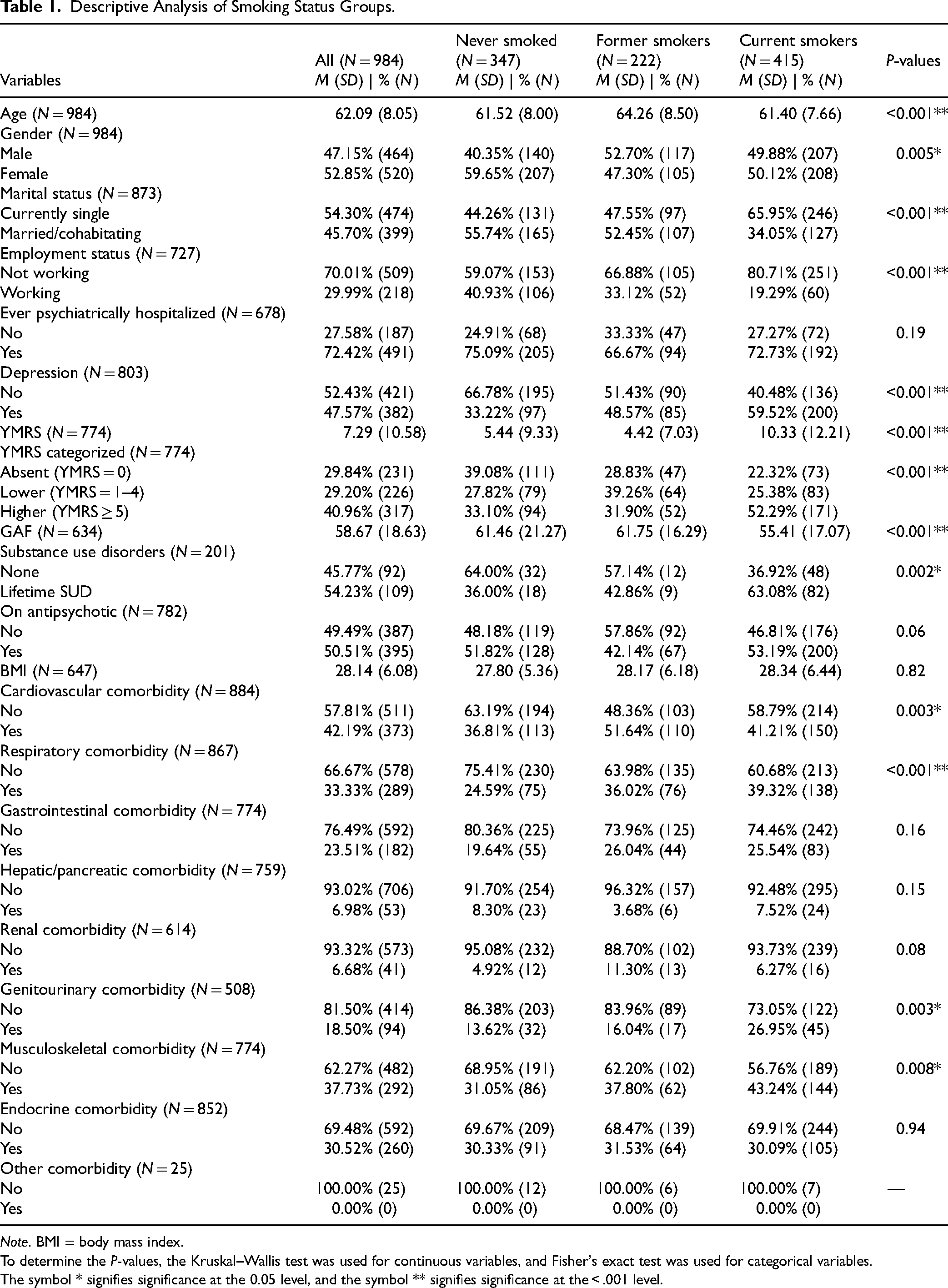
Note. BMI = body mass index.
To determine the P-values, the Kruskal–Wallis test was used for continuous variables, and Fisher's exact test was used for categorical variables.
The symbol * signifies significance at the 0.05 level, and the symbol ** signifies significance at the < .001 level.
The distribution of several demographic and clinical variables differed across never smokers, former smokers, and current smokers in unadjusted comparisons (Table 1), including: former smokers were older than current or never smokers (P < 0.001), more never smokers were women (P = 0.005), fewer current smokers were married (P < 0.001), and fewer current smokers were employed (P < 0.001). Psychiatric and somatic differences included: more current smokers were depressed (P < 0.001), more current smokers had significant manic symptoms (P < 0.001), more current and former smokers had lifetime SUDs (P = 0.002), current smokers were lower functioning (P < 0.001), more former smokers had cardiovascular comorbidity (P = 0.003), and more current smokers had respiratory (P < 0.001), genitourinary (P = 0.003), and musculoskeletal comorbidity (P = 0.008). Depression, lifetime SUD, respiratory comorbidity, genitourinary comorbidity, and musculoskeletal comorbidity followed a gradient, with current smokers experiencing higher comorbidity burden than former smokers, and former smokers experiencing higher comorbidity burden than never smokers. Current smokers experienced a higher manic symptom burden and decreased functioning compared to never and former smokers, though never and former smokers demonstrated similar scores for these variables. Cardiovascular comorbidity was highest in former smokers.
After controlling for age, sex, and study (Table 2), current depression was more prevalent in former versus never smokers (odds ratio 1.71 [95% confidence interval 1.08, 2.73]) and current versus never smokers (1.50 [1.04, 2.59]). Cardiovascular comorbidity was more common in former versus never smokers (1.67 [1.12, 2.50]), but not in current versus never smokers (1.01 [0.68, 1.51]). More current versus never smokers were on antipsychotics (1.50 [1.00, 2.25], P = 0.051), and more current versus never smokers had lifetime SUDs (2.91 [0.82, 10.42], P = 0.10), but these results were not statistically significant.
Association of Smoking Status and Clinical Variables (Including Comorbidities), Adjusted for Age, Gender, and Study.
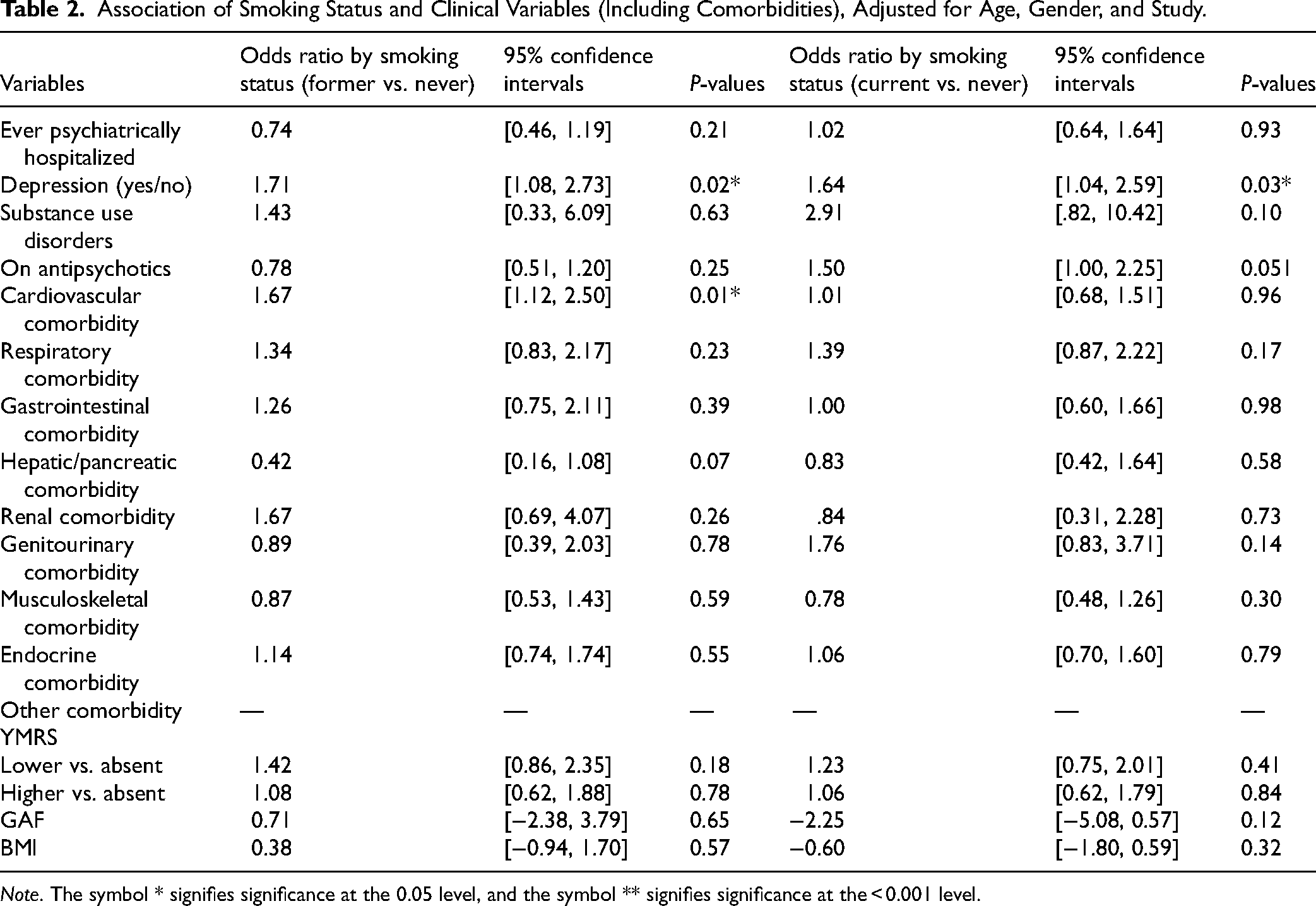
Note. The symbol * signifies significance at the 0.05 level, and the symbol ** signifies significance at the < 0.001 level.
When stratifying by the presence versus absence of cardiorespiratory comorbidity (Table 3), there were few differences in clinical variables by smoking status. The only statistically significant association was higher functioning in never versus current smokers in participants without cardiorespiratory comorbidity (−4.66 [−8.82, −0.50]). More current versus never smokers (12.99, [0.97, 174.27], P = 0.053) and more former versus never smokers (15.83 [0.61, 408.90], P = 0.10) had a lifetime SUD in participants with cardiorespiratory comorbidity, though this result was not statistically significant, and the analysis was limited by small sample sizes. This relationship was not present in participants without cardiorespiratory comorbidity for current versus never smokers (1.16 [0.23, 5.95], P = 0.86) or former versus never smokers (2.83 [0.60, 13.27], P = 0.19). There were non-statistically significant associations between lifetime smoking and depression across the comorbidity strata: current versus never smokers without comorbidity (1.63 [0.87, 3.08], P = 0.13), former versus never smokers without comorbidity (2.00 [0.99, 4.04], P = 0.052), current versus never smokers with comorbidity (1.81 [0.95, 3.45], P = 0.07), former versus never smokers with comorbidity (1.52 [0.81, 2.87], P = 0.20).
Comparison of Clinical Outcomes by Smoking for Groups with versus without Cardiovascular/Respiratory Comorbidities (N = 887).
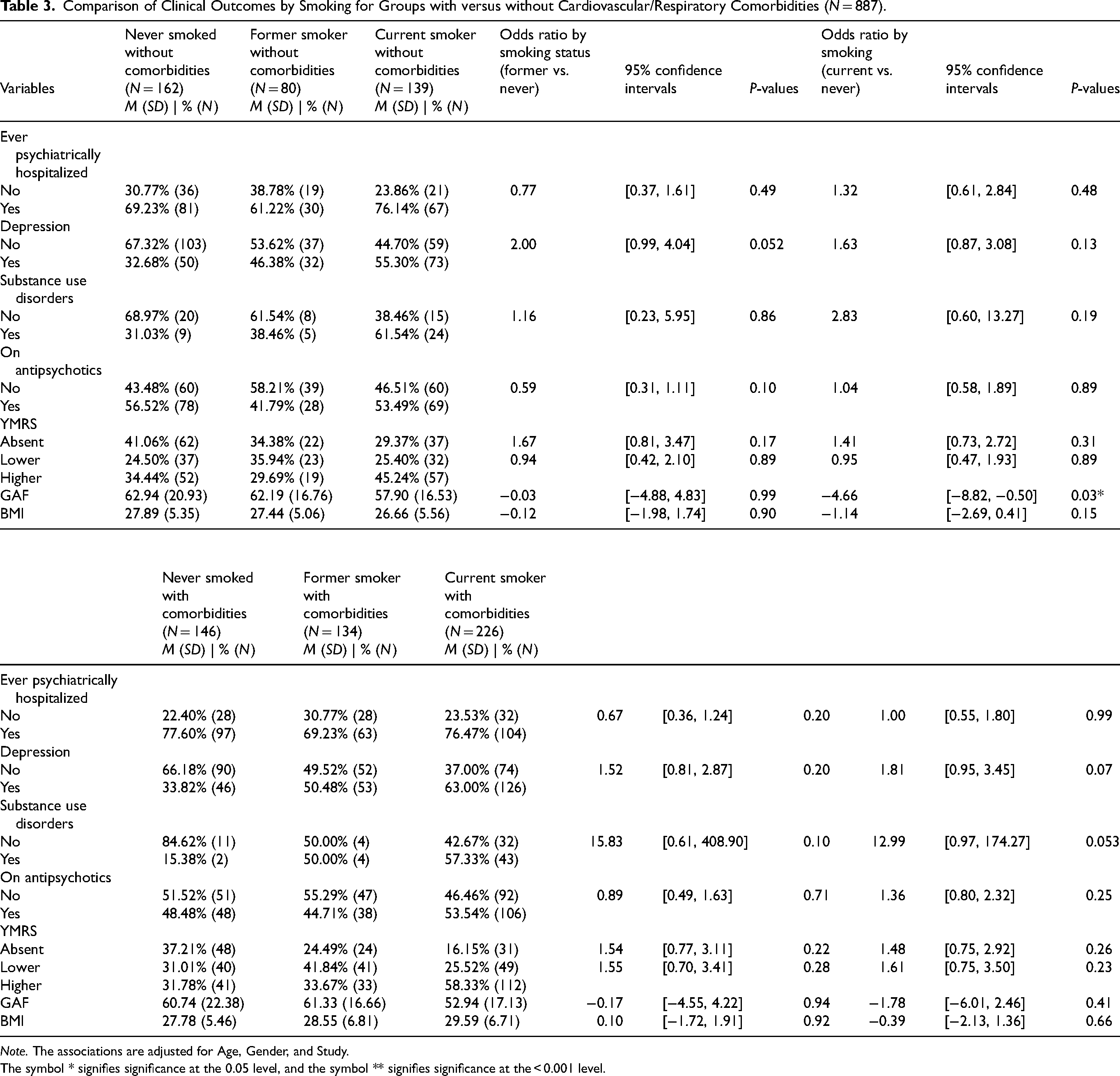
Note. The associations are adjusted for Age, Gender, and Study.
The symbol * signifies significance at the 0.05 level, and the symbol ** signifies significance at the < 0.001 level.
Discussion
In our large sample of OABD, cigarette smoking was correlated with poorer socioeconomic, psychiatric, and somatic parameters, though some unexpected relationships emerged. This study adds to the sparse literature on the complex relationships between BD course, somatic comorbidity, smoking, and smoking cessation in OABD.
The prevalence of current smoking in our sample (35.3%) was slightly lower than that in a recent Dutch study in OABD that showed a current smoking prevalence of 44.6%. 15 Rates of smoking are generally high in the Netherlands, 31 possibly explaining the decreased smoking prevalence rates in our study. To our knowledge, ours is the first study is to report on lifetime smoking prevalence in OABD populations. It is difficult to compare OABD smoking prevalence to smoking prevalence in younger populations as studies vary widely, 1 though OABD population prevalence rates align with general BD prevalence ranges. In seriously mentally ill populations, chronic somatic and psychiatric disease burden along with stressful life experiences may drive difficulties with smoking cessation 11 and explain the persistence of smoking behaviour into older age.
Our study highlights that smoking cessation may have particularly complex consequences for physical health. Some studies in BD populations have identified that smoking cessation is associated with adverse metabolic consequences.8,13 However, these metabolic consequences may be driven by antipsychotic use and age 8 and may not be associated with an increased cardiovascular risk, according to a prospective study of adults with serious mental illness. 13
We found that former, but not current, smokers have higher rates of cardiovascular comorbidity than never smokers in OABD, even after controlling for age and sex. This novel finding may be attributable to our older population being more vulnerable to the metabolic consequences of smoking cessation. 28 However, in some studies, smoking cessation has been associated with improved metabolic parameters.29,30 An alternative explanation is that the population of former smokers quit smoking as a result of their cardiorespiratory diagnosis, a motivating factor. This explanation would suggest that psychiatrists should regularly provide psychoeducation about somatic consequences of smoking to increase patients’ motivation. This psychoeducation strategy aligns with the effectiveness of advertisements that elicit disgust and fear about drug consequences to promote abstinence.32,33 Identifying motivators for smoking cessation is crucial in BD populations, who quit smoking less frequently than the general population. 11
Our hypothesis that smoking status would be associated with increased somatic comorbidity was partially borne out. However, the association between smoking and various somatic comorbidities (musculoskeletal, genitourinary, and endocrine) was absent after adjusting for age, sex, and study, illustrating that the initial associations were likely driven by differences in age between subgroups, with more somatic comorbidity in older participants.
We found that lifetime smoking history (either former or current smoking) was associated with the presence of depression even after adjusting for age, sex, and study. Non-statistically significant associations remained after stratifying for cardiorespiratory comorbidity status. Though associations between cardiovascular disease and depressive symptoms have been reported, 34 our study illustrated that the relationship between smoking and depression is largely independent of cardiorespiratory comorbidity. To our knowledge, this is the first time this complex relationship has been elucidated. We also found that more smokers were taking antipsychotics, which suggests that smoking is associated with a more severe BD course. These findings support our hypothesis and extend several studies’ finding of the association between smoking and psychiatric comorbidity in general BD populations to OABD populations.3–5,8 Treating BD symptoms such as impulsivity may help with smoking cessation. Further, clinicians should discuss the association of smoking with a more severe BD course to increase patients’ motivation for smoking cessation.
However, after controlling for age, sex, and study, the association between smoking and manic symptoms was no longer statistically significant. Though smoking has been correlated with lifetime manic and depressive episodes 4 and earlier age of onset of manic symptoms, 3 it is unclear whether current smoking is associated with current manic symptoms. In fact, there have been cases of smoking cessation being associated with mania onset. 35 Our results indicate that metabolic factors related to age or sex may mediate whether current smoking behaviour is associated with manic symptoms. Further research is needed.
After controlling for age, sex, and study, a non-statistically significant relationship remained between smoking and lifetime SUD. This relationship is consistent with prior studies’ finding of smoking being correlated with other SUDs.35,36 After stratification by cardiorespiratory comorbidity, a non-statistically significant association remained between smoking and other SUDs in the cardiorespiratory comorbidity subgroup only. This novel finding aligns with the Diagnostic and Statistical Manual of Mental Disorders (5th edition) definition of a SUD, which includes continuing substance use despite negative health consequences of use. 37 Clinicians should proactively screen patients who continue smoking despite cardiorespiratory comorbidity for comorbid SUDs.
We also found that the lower functional status of current or former smokers compared to never smokers remained significant even after accounting for age, sex, study, and cardiorespiratory comorbidity. This may suggest other factors contribute to OABD smokers’ functioning, such as our findings of increased depression, decreased marriage rates, and decreased employment in smokers. Other unmeasured factors may also contribute, such as decreased cognitive abilities in smokers in certain BD and older age populations.38,39 Alternatively, smoking behaviour may represent an attempt to cope with a decreased ability to function in some individuals. Clinicians should help cultivate healthier coping strategies in OABD populations to decrease smoking-related risks.
Our study was limited by its cross-sectional design, which restricts inferences about causation. 40 Sample sizes were small for certain scales in the stratified samples, leading to wide confidence intervals and a decreased degree of certainty about certain conclusions. 40 For instance, our study may have been underpowered to detect a relationship between smoking history and depression in the stratified samples given the non-statistically significant associations across comorbidity status. The small sample sizes in the stratified samples may also explain the absence of a relationship between smoking status and other clinical variables in the subgroup with cardiorespiratory comorbidity, as per our original hypothesis. Further, the GAGE-BD sample currently lacks harmonized data for a few important variables about BD severity such as suicidal ideation and time spent in mood episodes, though psychiatric hospitalizations and antipsychotic use may serve as proxies for illness severity. Additionally, research samples from multiple studies with different inclusion criteria may not represent all of OABD, which limits generalizability. Finally, our aggregated dataset is derived from studies with different methodologies, which added heterogeneity to some variables such as somatic comorbidity categories and prevented precise mapping onto Strengthening the Reporting of Observational Studies in Epidemiology guidelines.
Future studies should prospectively examine the relationship between smoking and the incidence of psychiatric and somatic complications to support causative inferences about the effects of smoking in OABD. The GAGE-BD team is compiling longitudinal data so that these questions may be addressed in the future. 41 More studies are also needed on the medical factors driving increased rates of cardiovascular disease in former smokers and elaborating on the risks and benefits of smoking cessation.
Conclusions
Our study elucidated the complex relationship between smoking and psychiatric and somatic comorbidities in OABD. One novel finding is that former smokers had higher rates of cardiovascular disease than never smokers, suggesting that cardiovascular disease may serve as a motivating factor for smoking cessation in OABD. Future studies should explore the causative role of smoking in driving comorbidity in OABD.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437251387658 - Supplemental material for Relationship Between Smoking and Psychiatric and Somatic Comorbidities in Older Age Bipolar Disorder: Lien entre le tabagisme et les affections psychiatriques et somatiques concomitantes chez les personnes âgées atteintes de trouble bipolaire
Supplemental material, sj-docx-1-cpa-10.1177_07067437251387658 for Relationship Between Smoking and Psychiatric and Somatic Comorbidities in Older Age Bipolar Disorder: Lien entre le tabagisme et les affections psychiatriques et somatiques concomitantes chez les personnes âgées atteintes de trouble bipolaire by Molly Howland, MD, Arushi Mahajan, MD, Peijun Chen, MD, PhD, MPH, Osvaldo Almeida, MD, PhD, Kürşat Altınbaş, MD, PhD, Hilary Blumberg, MD, Annemiek Dols, MD, PhD, Nicole Fiorelli, MA, Orestes Forlenza, MD, PhD, Beny Lafer, MD, PhD, Andrew Olagunju, MBBS, MSc, Melis Orhan, PhD, Soham Rej, MD, MSc, Martha Sajatovic, MD, Matthew Schurr, PhD, Christian Simhandl, MD, PhD, Jair Soares, MD, PhD, Ashley Sutherland, MA, Shang-ying Tsai, MD, Sara Weisenbach, PhD, ABPP, Joy Yala, MPH and Farren Briggs, PhD in The Canadian Journal of Psychiatry
Footnotes
Conflicts of Interest Disclosure
Orestes Forlenza is a consultant for Milken Institute and Lilly and has received an honorarium from Aetna.
Soham Rej has received a clinician-scientist salary award from the Fonds de Recherche de Québec Santé; has received operating grants from the Canadian Institutes of Health Research, Social Sciences and Humanities Research Council, Mind and Life Institute, Doggone Foundation, Jewish General Hospital Foundation, and Mitacs; and is a shareholder of Aifred Health.
Martha Sajatovic has received grants from Neurelis, Intra-Cellular, Merck, Otsuka, Alkermes, International Society for Bipolar Disorders; royalties from Springer Press, Johns Hopkins University Press, Oxford Press, and UpToDate; and consulting fees from Alkermes, Otsuka, Lundbeck, Janssen and Teva. She has also received compensation for preparation of continuing medical education activities from American Physician's Institute (CMEtoGo), Psychopharmacology Institute, American Epilepsy Society, and Clinical Care Options.
Jair Soares has received grants from Compass Pathways, Mind Med, and Sunovion. He is on the advisory boards for Alkermes and J&J.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the International Society for Bipolar Disorders (ISBD) Bowden Massey Strategic Research Initiative in Bipolar Disorder Award, and made possible by logistical support from the ISBD. Additional support was also provided by the Tim and Joan Jenkins Fund for Psychiatry Research and by Alkermes, Otsuka and Intra-Cellular Therapies, Inc.
ORCID iDs
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
